Chemical Diversity, Biological Activity, and Genetic Aspects of Three Ocotea Species from the Amazon
Abstract
:1. Introduction
2. Results and Discussion
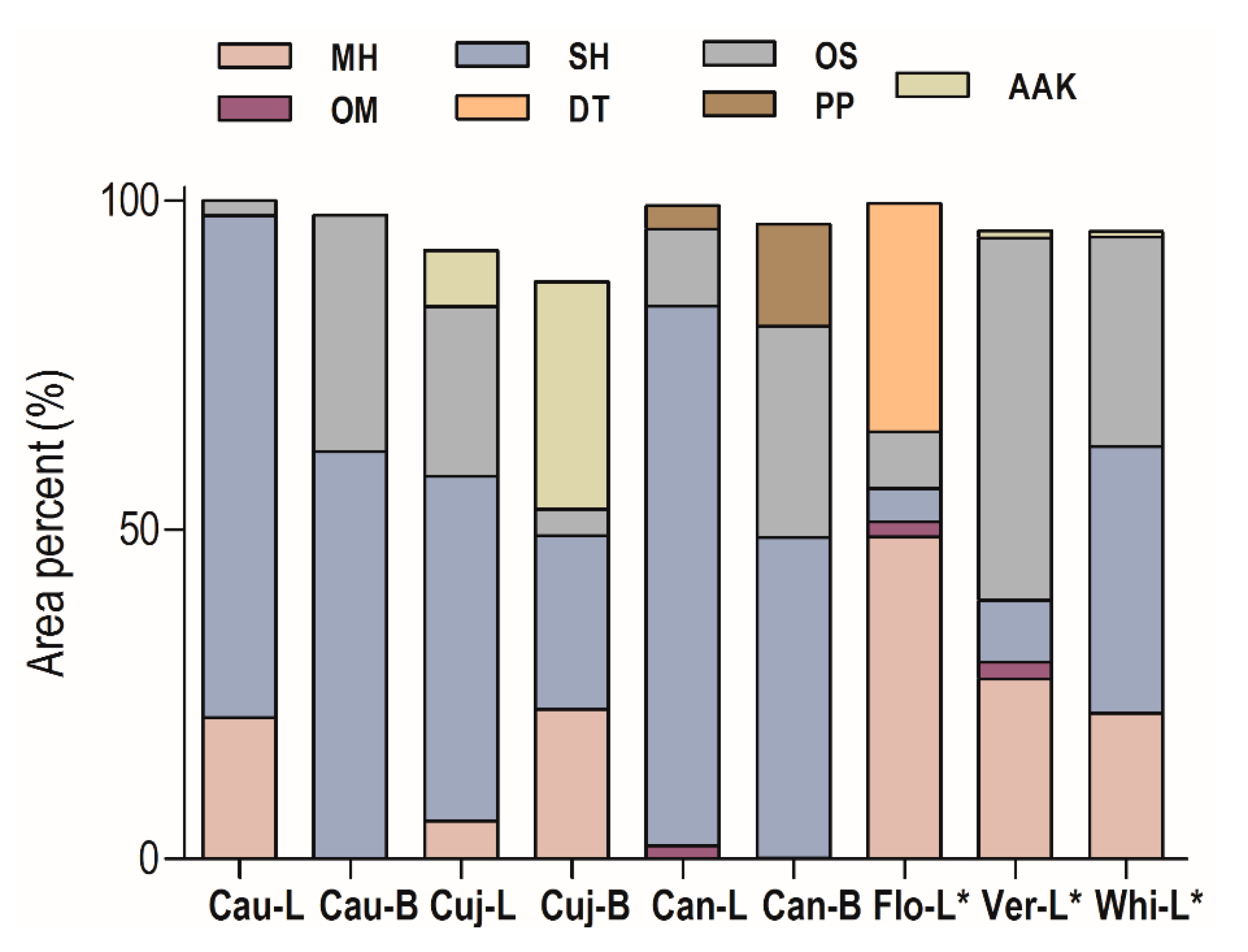
2.1. Essential Oil Chemical Composition
2.2. Antimicrobial and Cytotoxic Activities
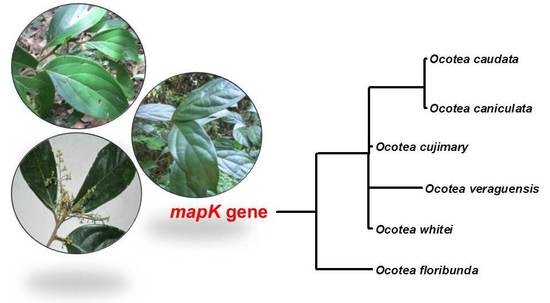
2.3. Phylogenetic Analysis
3. Materials and Methods
3.1. Plant Material
3.2. Essential Oil Extraction
3.3. Gas Chromatographic–Mass Spectral Analysis
3.4. Antibacterial Assay
3.5. Cytotoxicity Assay
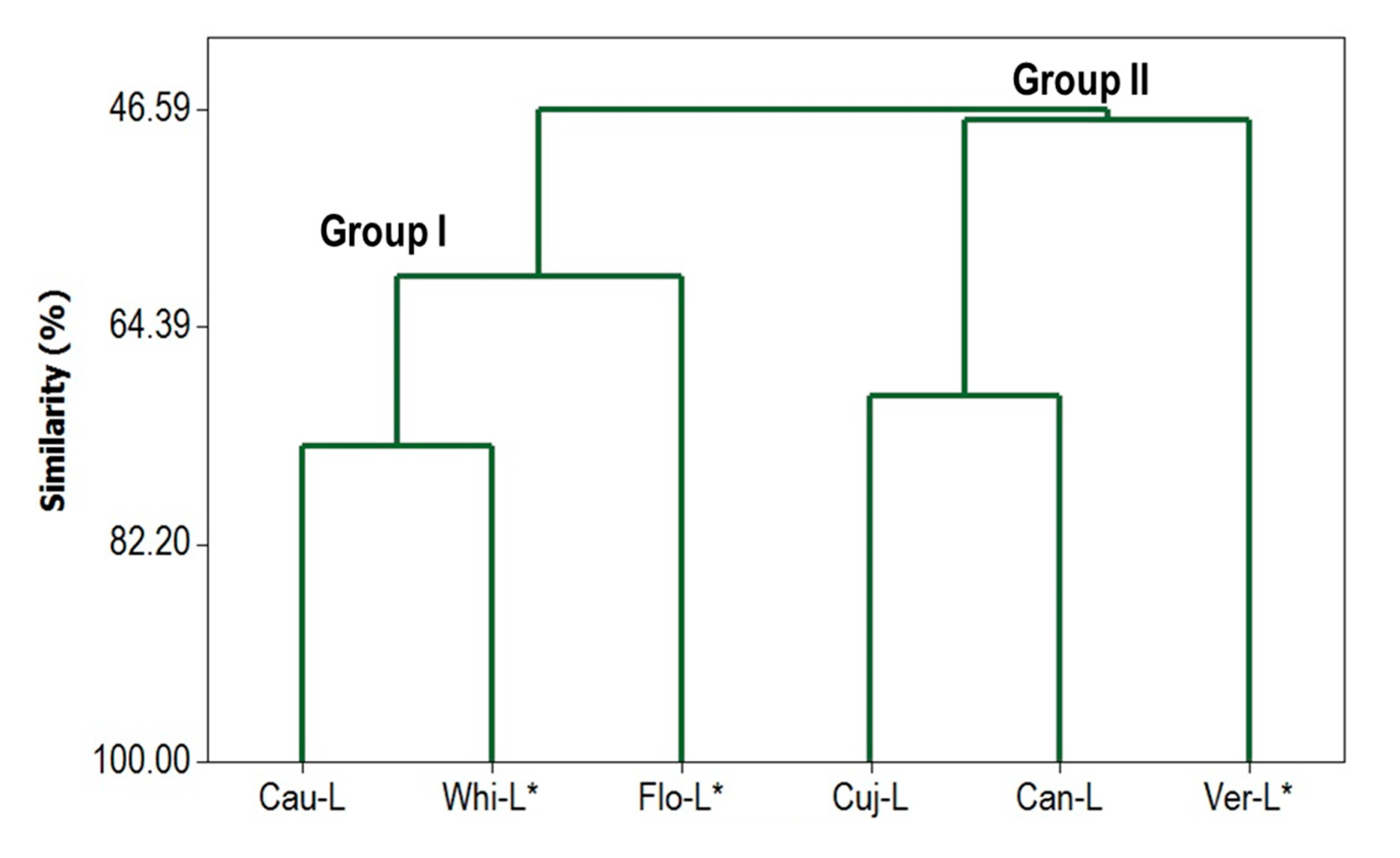
3.6. Multivariate Statistical Analysis of Chemical Composition
3.7. DNA Isolation, PCR Amplification, and Sequencing
3.8. Phylogenetic Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| matK | Megakaryocyte-Associated Tyrosine Kinase |
| MIC | Minimum Inhibitory Concentration |
| RICalc. | Calculated retention index |
| RILit | Literature retention index |
| IC50 | Median Inhibitory Concentration |
| GC | Gas chromatography |
| DMSO | Dimethylsulfoxide |
| RPMI | Roswell Park Memorial Institute medium |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| HEPES | 4-(2-Hydroxyethyl)-1-piperazineethanesulfonic acid buffer |
| PCR | Polymerase Chain Reaction |
References and Notes
- Rohwer, J.G. Lauraceae. In The Families and Genera of Vascular Plants; Kubitzki, K., Rohwer, J.G., Bittrich, V., Eds.; Springer: Berlin, Germany, 1993; Volume 2, pp. 366–391. [Google Scholar]
- Quinet, A.; Baitello, J.B.; Moraes, P.L.R.; Alves, F.M.; Assis, L. Lauraceae. Lista de Espécies da flora do Brasil. Jardim Botânico do Rio de Janeiro. Available online: http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB143 (accessed on 7 March 2017).
- Institute of Research Rio de Janeiro Botanical Garden. Lauraceae. Flora do Brasil 2020 em Construção. Jardim Botânico do Rio de Janeiro. Available online: http://floradobrasil.jbrj.gov.br/reflora/floradobrasil/FB143 (accessed on 7 March 2017).
- Van der Werff, H. A synopsis of Ocotea (Lauraceae) in Central America and southern Mexico. Ann. Mo. Bot. Gard. 2002, 89, 429–451. [Google Scholar] [CrossRef]
- Marques, C.A. Importância econômica da família Lauraceae Lindl. Florest. Ambient. 2001, 8, 195–206. [Google Scholar]
- David, J.M.; Yoshida, M.; Gottlieb, O.T. Phenylpropanoid-catechins from bark of Ocotea porosa. Phytochemistry 1994, 35, 545–546. [Google Scholar] [CrossRef]
- David, J.M.; Yoshida, M.; Gottlieb, O.R. Neolignans from bark and leaves of Ocotea porosa. Phytochemistry 1994, 36, 491–499. [Google Scholar] [CrossRef]
- Barrera, E.D.C.; Suárez, L.E.C. Aporphine Alkaloids from Leaves of Ocotea macrophylla (Kunth) (Lauraceae) from Colombia. Biochem. Syst. Ecol. 2009, 37, 522–524. [Google Scholar] [CrossRef]
- Garcez, W.S.; Yoshida, M.; Gottlieb, O.R. Benzylisoquinoline alkaloids and flavonols from Ocotea vellosiana. Phytochemistry 1995, 39, 815–816. [Google Scholar] [CrossRef]
- Chavez, J.P.; Gottlieb, O.R.; Yoshida, M. 10-Desmethyl-1-methyl-eudesmanes from Ocotea corymbosa. Phytochemistry 1995, 39, 849–852. [Google Scholar] [CrossRef]
- Ballabenia, V.; Tognolinia, M.; Bertonia, S.; Brunib, R.; Guerrinib, A.; Ruedac, G.M.; Barocellia, E. Antiplatelet and antithrombotic activities of essential oil from wild Ocotea quixos (Lam.) Kosterm. (Lauraceae) calices from Amazonian Ecuador. Pharmacol. Res. 2007, 55, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Chaverri, C.; Díaz, C.; Cicció, J.F. Chemical Analysis of Essential Oils from Ocotea gomezii W.C. Burger and Ocotea morae Gómez-Laur. (Lauraceae) Collected at “Reserva Biológica Alberto M. Brenes” in Costa Rica and their Cytotoxic Activity on Tumor Cell Lines. J. Braz. Chem. Soc. 2011, 22, 741–745. [Google Scholar] [CrossRef]
- Morais, L.C.S.L.; Barbosa-Filho, J.M.; Almeida, R.N. Central depressant effects of reticuline extracted from Ocotea duckei in rats and mice. J. Ethnopharmacol. 1998, 62, 57–61. [Google Scholar] [CrossRef]
- Fournet, A.; Ferreira, M.E.; Arias, A.R.; Guy, I.; Guinaudeau, H.; Heinzen, H. Phytochemical and antiprotozoal activity of Ocotea lancifolia. Fitoterapia 2007, 78, 382–384. [Google Scholar] [CrossRef] [PubMed]
- Montrucchio, D.P.; Miguel, O.G.; Zanin, S.M.; da Silva, G.A.; Cardoso, A.M.; Santos, A.R. Antinociceptive effects of a chloroform extract and the alkaloid dicentrine isolated from fruits of Ocotea puberula. Planta Med. 2012, 78, 1543–1548. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cheng, E.; Rakotondraib, L.H.; Brodie, P.J.; Applequist, W.; Randrianaivo, R.; Rakotondrafara, A.; Ratsimbason, M.; Rasamison, V.E.; Kingston, D.G.I. Antiproliferative compounds from Ocotea macrocarpa from the Madagascar dry forest. Tetrahedron Lett. 2015, 56, 3630–3632. [Google Scholar] [CrossRef] [PubMed]
- Costa, I.F.; Calixto, S.D.; Heggdorne de Araujo, M.; Konno, T.U.; Tinoco, L.W.; Guimarães, D.O.; Lasunskaia, E.B.; Leal, I.R.; Muzitano, M.F. Antimycobacterial and Nitric Oxide Production Inhibitory Activities of Ocotea notata from Brazilian Restinga. Sci. World J. 2015, 2015, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ballabeni, V.; Tognolini, M.; Giorgio, C.; Bertoni, S.; Bruni, R.; Barocelli, E. Ocotea quixos Lam. essential oil: In vitro and in vivo investigation on its anti-inflammatory properties. Fitoterapia 2010, 81, 289–295. [Google Scholar] [CrossRef] [PubMed]
- MMA (Ministério do Meio Ambiente). Instrução Normativa n°.6 de 23/09/2008. Reconhece as espécies da flora brasileira ameaçadas de extinção.
- Martins, E.M.; Martinelli, G.; Arbetman, M.P.; Lamont, R.W.; Simões-Araújo, J.L.; Powell, D.; Ciampi-Guillardi, M.; Baldauf, C.; Quinet, A.; Galisa, P.; et al. Development and characterization of microsatellite loci for Ocotea species (Lauraceae) threatened with extinction. Genet. Mol. Res. 2014, 13, 5138–5142. [Google Scholar] [CrossRef] [PubMed]
- Rahali, F.Z.; Lamine, M.; Gargouri, M.; Rebey, I.B.; Hammami, M.; Sellami, I.H. Metabolite profiles of essential oils and molecular markers analysis to explore the biodiversity of Ferula communis: Towards conservation of the endemic giant fennel. Phytochemistry 2016, 124, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Takaku, S.; Haber, W.A.; Setzer, W.N. Leaf essential oil composition of 10 species of Ocotea (Lauraceae) from Monteverde, Costa Rica. Biochem. Syst. Ecol. 2007, 35, 525–532. [Google Scholar] [CrossRef]
- Da Silva, J.K.R.; da Trindade, R.C.; Maia, J.G.; Setzer, W.N. Chemical Composition, Antioxidant, and Antimicrobial Activities of Essential Oils of Endlicheria arenosa (Lauraceae) from the Amazon. Nat. Prod. Commun. 2016, 11, 695–698. [Google Scholar] [PubMed]
- Grecco, S.S.; Martins, E.G.; Girola, N.; de Figueiredo, C.R.; Matsuo, A.L.; Soares, M.G.; Bertoldo, B.C.; Sartorelli, P.; Lago, J.H. Chemical composition and in vitro cytotoxic effects of the essential oil from Nectandra leucantha leaves. Pharm. Biol. 2015, 53, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Salleh, W.M.; Ahmada, F.; Yen, K.H.; Zulkifli, R.M. Chemical Compositions and Biological Activities of Essential Oils of Beilschmiedia glabra. Nat. Prod. Commun. 2015, 10, 1297–1300. [Google Scholar] [PubMed]
- Salleh, W.M.; Ahmada, F.; Yen, K.H. Chemical Compositions and Biological Activities of Essential Oils of Beilschmiedia glabra. Arch. Pharm. Res. 2015, 38, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Telascrea, M.; de Araújo, C.C.; Marques, M.O.M.; Facanali, R.; de Moraes, P.L.R.; Cavalheiro, A.J. Essential oil from leaves of Cryptocarya mandioccana Meisner (Lauraceae): Composition and intraspecific chemical variability. Biochem. Syst. Ecol. 2007, 35, 222–227. [Google Scholar] [CrossRef]
- Pino, J.A.; Fernandes, P.; Marbot, R.; Sá Fontinha, S. Chemical composition of the leaf oil of Ocotea foetens (Alt.) Benth. et Hook. from Madeira. J. Essent. Oil Res. 2004, 16, 131–132. [Google Scholar] [CrossRef]
- Schmidt, J.M.; Noletto, J.A.; Vogler, B.; Setzer, W.N. Abaco bush medicine: Chemical composition of the essential oils of four aromatic medicinal plants from Abaco Island, Bahamas. J. Herbs Spices Med. Plants 2007, 12, 43–65. [Google Scholar] [CrossRef]
- Wright, B.S.; Bansal, A.; Moriarity, D.M.; Takaku, S.; Setzer, W.N. Cytotoxic leaf essential oils from Neotropical Lauraceae: Synergistic effects of essential oil components. Nat. Prod. Commun. 2007, 2, 1241–1244. [Google Scholar]
- Owolabi, M.S.; Ogundajo, A.; Yusuf, K.O.; Lajide, L.; Villanueva, H.E.; Tuten, J.A.; Setzer, W.N. Chemical composition and bioactivity of the essential oil of Chromolaena odorata from Nigeria. Rec. Nat. Prod. 2010, 4, 72–78. [Google Scholar]
- Sobral, M.V.; Xavier, A.L.; Lima, T.C.; de Sousa, D.P. Antitumor activity of monoterpenes found in essential oils. Sci. World J. 2014, 2014, 953451. [Google Scholar] [CrossRef] [PubMed]
- Gautam, N.; Mantha, A.K.; Mittal, S. Essential Oils and Their Constituents as Anticancer Agents: A Mechanistic View. BioMed Res. Int. 2014, 2014, 154106. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, L.; Peña, A.; Velascd, J.; Baptista, J.G.; Rojas, L.; Aparicio, R.; Usubillaga, A. Chemical composition and antibacterial activity of the essential oil of Ruilopezia bracteosa. Nat. Prod. Commun. 2015, 10, 655–656. [Google Scholar] [PubMed]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Da Silva, E.B.P.; Matsuo, A.L.; Figueiredo, C.R.; Chaves, M.H.; Sartorelli, P.; Lago, J.H.G. Chemical constituents and cytotoxic evaluation of essential oils from leaves of Porcelia macrocarpa (Annonaceae). Nat. Prod. Commun. 2013, 8, 277–279. [Google Scholar] [PubMed]
- El Hadri, A.; Del Río, M.A.G.; Sanz, J.; Coloma, A.G.; Idaomar, M.; Ozonas, B.R.; González, J.B.; Reus, M.I.S. Cytotoxic activity of α-humulene and transcaryophyllene from Salvia officinalis in animal and human tumor cells. An. R. Acad. Nac. Farm. 2010, 76, 343–356. [Google Scholar]
- Park, K.R.; Nam, D.; Yun, H.M.; Jang, H.J.; Sethi, G.; Cho, S.K.; Ahn, K.S. β-Caryophyllene oxide inhibits growth and induces apoptosis through the suppression of PI3K/AKT/mTOR/S6K1 pathways and ROS-mediated MAPKs activation. Cancer Lett. 2011, 312, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Santana, J.S.; Sartorelli, P.; Guadagnin, R.C.; Matsuo, A.L.; Figueiredo, C.R.; Soares, M.G.; da Silva, A.M.; Lago, J.H. Essential oils from Schinus terebinthifolius leaves—Chemical composition and in vitro cytotoxicity evaluation. Pharm. Biol. 2012, 50, 1248–1253. [Google Scholar] [CrossRef] [PubMed]
- Hilu, K.W.; Liang, H. The MATK gene: Sequence variation and application in plant systematics. Am. J. Bot. 1997, 84, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Neuhaus, H.; Link, G. The chloroplast tRNALys (UUU) gene from mustard (Sinapis alba) contains a class II intron potentially coding for a maturase-related polypeptide. Curr. Genet. 1987, 11, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Chase, M.W.; Cowan, R.S.; Hollingsworth, P.M.; Berg, C.; Madrinan, S.; Petersen, G.; Seberg, O.; Jorgsensen, T.; Camero, K.M.; Carine, M.; et al. A Proposal for a Standardised Protocol to Barcode All Land Plants. Taxon 2007, 56, 295–299. [Google Scholar]
- Lahaye, R.; van der Bank, M.; Bogarin, D.; Warner, J.; Pupulin, F.; Gigot, G.; Maurin, O.; Duthoit, S.; Barraclough, T.G.; Savolainen, V. DNA barcoding the floras of biodiversity hotspots. Proc. Natl. Acad. Sci. USA 2008, 105, 2923–2928. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007; p. 804. [Google Scholar]
- Da Silva, J.K.R.; Gomes, M.V.S.; Maia, J.G.S.; Dosoky, N.S.; Setzer, W.N. Chemical composition and in vitro biological activities of essential oil chemotypes of Licaria rigida (Kosterm.) Kosterm. (Lauraceae). Int. J. Appl. Res. Nat. Prod. 2016, 9, 1–9. [Google Scholar]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Hygeine 1938, 27, 493–497. [Google Scholar]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
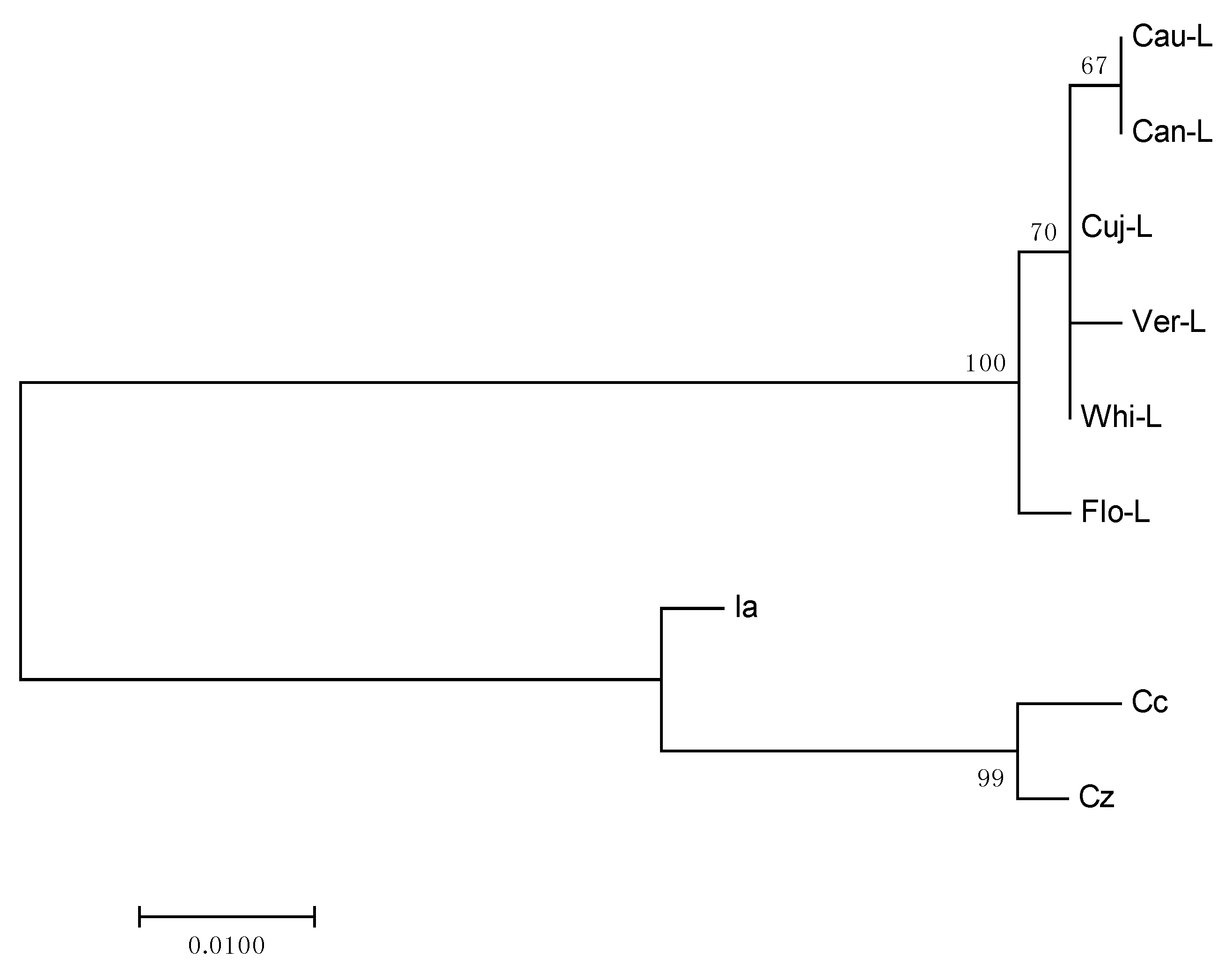
| Species | Geographic Coordinate | Voucher | Plant Material | Sample | Oil Yield (%) |
|---|---|---|---|---|---|
| O. caudata | S 01.0° 44.0′ 18.8′′ | MG 216263 | Leaves | Cau-L | 0.7 |
| W 51.0° 27.0′ 27.4′′ | Branches | Cau-B | 0.1 | ||
| O. cujumary | S 01.0° 44.0′ 14.1′′ | MG 216269 | Leaves | Cuj-L | 0.8 |
| W 51.0° 27.0′ 20.4′′ | Branches | Cuj-B | 0.5 | ||
| O. caniculata | S 01.0° 44.0′ 14.1′′ | MG 216262 | Leaves | Can-L | 0.7 |
| W 51.0° 27.0′ 20.4′′ | Branches | Can-B | 0.2 |
| Constituents | RICalc | RILit | Cau-L | Cau-B | Cuj-L | Cuj-B | Can-L | Can-B | Flo-L * | Ver-L * | Whi-L * |
|---|---|---|---|---|---|---|---|---|---|---|---|
| E-2-Hexenal | 856 | 854 | 1.1 | 0.8 | |||||||
| α-Thujene | 933 | 931 | 0.2 | ||||||||
| α-Pinene | 936 | 932 | 9.8 | 2.1 | 22.5 | 0.7 | 12.7 | ||||
| Camphene | 956 | 953 | 1.7 | 0.1 | 0.3 | ||||||
| Sabinene | 979 | 976 | 0.1 | ||||||||
| β-Pinene | 980 | 974 | 9.7 | 1.8 | 2.2 | 21.3 | 0.3 | 7.3 | |||
| Myrcene | 995 | 991 | 0.7 | 1.1 | 0.5 | ||||||
| α-Phellandrene | 1009 | 1005 | 1 | ||||||||
| p-Cymene | 1028 | 1026 | 19.8 | ||||||||
| Limonene | 1030 | 1024 | 2.1 | 1.8 | 20.5 | 2.7 | 1.1 | ||||
| β-Phellandrene | 1032 | 1031 | 4.0 | ||||||||
| 1,8-Cineole | 1033 | 1033 | 1.3 | ||||||||
| γ-Terpinene | 1064 | 1062 | 0.1 | ||||||||
| α-Terpinolene | 1089 | 1088 | 0.1 | ||||||||
| α-Pinene oxide | 1097 | 1095 | 0.1 | ||||||||
| Linalool | 1104 | 1098 | 1.7 | ||||||||
| Borneol | 1168 | 1165 | 0.1 | ||||||||
| Terpinen-4-ol | 1178 | 1177 | 2.0 | 0.2 | |||||||
| α-Terpineol | 1191 | 1189 | 0.2 | 1.3 | |||||||
| Cuminal | 1238 | 1239 | 0.1 | ||||||||
| 2-Undecanol | 1301 | 1301 | 1.2 | 4.6 | |||||||
| δ-Elemene | 1340 | 1335 | 2.2 | ||||||||
| α-Cubebene | 1351 | 1345 | 2.0 | 2.0 | 0.8 | 2.2 | |||||
| α-Ylangene | 1373 | 1373 | 0.8 | 5.1 | |||||||
| α-Copaene | 1377 | 1374 | 1.0 | 0.1 | 0.1 | 0.8 | |||||
| β-Cubebene | 1392 | 1387 | 1.6 | 0.1 | |||||||
| β-Bourbonene | 1384 | 1387 | 0.7 | 0.1 | 0.1 | ||||||
| δ-Elemene | 1392 | 1389 | 1.5 | 0.4 | 1.1 | 0.8 | 0.3 | 0.7 | 0.3 | ||
| Z-Caryophyllene | 1416 | 1408 | 0.3 | ||||||||
| β-Caryophyllene | 1421 | 1417 | 9.6 | 2.5 | 22.2 | 8.1 | 18.9 | 7.1 | 2.5 | 2.3 | 15.2 |
| 2,5-Dimethoxy-p-cymene | 1424 | 1424 | 0.9 | ||||||||
| β-Copaene | 1428 | 1430 | 0.4 | ||||||||
| β-Gurjunene | 1432 | 1432 | 0.2 | ||||||||
| α-trans-Bergamotene | 1436 | 1432 | 0.9 | 1.9 | |||||||
| γ-Elemene | 1435 | 1434 | 0.8 | 0.1 | |||||||
| α-Guaiene | 1439 | 1439 | 0.1 | ||||||||
| Aromadendrene | 1440 | 1439 | 0.1 | ||||||||
| Z-β-Farnesene | 1443 | 1440 | 0.9 | ||||||||
| Spirolepechinene | 1451 | 1449 | 0.7 | 1.4 | 0.6 | ||||||
| α-Humulene | 1455 | 1452 | 1.8 | 2.4 | 3.8 | 2.5 | 2.5 | 1.7 | 0.3 | 1.7 | 1.7 |
| Sesquisabinene | 1458 | 1457 | 0.9 | ||||||||
| Dehydroaromadendrane | 1460 | 1460 | 0.6 | ||||||||
| E-β-Farnesene | 1462 | 1458 | 0.9 | ||||||||
| allo-Aromadendrene | 1463 | 1461 | 0.2 | ||||||||
| trans-Cadina-1(6),4-diene | 1473 | 1475 | 0.5 | ||||||||
| γ-Selinene | 1477 | 1470 | 0.2 | ||||||||
| γ-Gurjunene | 1477 | 1475 | 0.4 | ||||||||
| γ-Muurolene | 1476 | 1478 | 0.8 | 0.7 | |||||||
| Widdra-2,4(14)-diene | 1483 | 1481 | 6.5 | ||||||||
| Germacrene D | 1484 | 1484 | 19.9 | 8.9 | 0.9 | 5.9 | 1.2 | 0.2 | 5.5 | ||
| β-Selinene | 1485 | 1489 | 2.2 | 20.3 | 12.1 | 0.3 | |||||
| Valencene | 1493 | 1491 | 1.1 | ||||||||
| cis-β-Guaiene | 1493 | 1492 | 8.3 | 3.0 | 5.2 | ||||||
| trans-Muurola-4(14),5-diene | 1491 | 1493 | 1.1 | ||||||||
| 2-Tridecanone | 1497 | 1495 | 7.3 | 30.0 | |||||||
| Viridiflorene | 1493 | 1496 | 9.8 | ||||||||
| Bicyclogermacrene | 1500 | 1500 | 29.6 | 10.4 | 5.3 | ||||||
| α-Muurolene | 1499 | 1500 | 1.7 | 1.0 | 0.1 | ||||||
| Germacrene A | 1504 | 1503 | 0.2 | 2 | |||||||
| β-Bisabolene | 1509 | 1505 | 0.6 | ||||||||
| δ-Amorphene | 1509 | 1511 | 0.8 | 2.0 | |||||||
| E,E-α-Farnesene | 1510 | 1508 | 0.3 | ||||||||
| γ-Cadinene | 1517 | 1513 | 0.9 | 5.9 | 1.6 | 3.1 | 1.7 | 0.1 | 0.4 | 0.7 | |
| 7-epi-α-Selinene | 1517 | 1520 | 4.5 | 14.8 | 9.0 | ||||||
| β-Cadinene | 1519 | 1518 | 0.7 | 0.6 | |||||||
| cis-Calamenene | 1523 | 1521 | 0.6 | ||||||||
| δ-Cadinene | 1526 | 1522 | 1.4 | 13.8 | 6.6 | 4.7 | 0.6 | 3.9 | 0.2 | 0.4 | 3.7 |
| trans-Cadina-1,4-diene | 1532 | 1533 | 0.6 | 0.2 | 0.2 | ||||||
| α-Cadinene | 1537 | 1538 | 0.1 | ||||||||
| α-Calacorene | 1542 | 1544 | 0.9 | 1.5 | 0.2 | ||||||
| Elemol | 1549 | 1549 | 0.1 | ||||||||
| Germacrene B | 1558 | 1559 | 1.6 | 3.3 | 0.8 | 0.4 | 0.7 | ||||
| E-Nerolidol | 1564 | 1561 | 1.4 | 2.5 | 3.9 | ||||||
| β-Calacorene | 1564 | 1564 | 0.7 | 0.5 | |||||||
| γ-Asarone | 1576 | 1572 | 0.4 | ||||||||
| Spathulenol | 1579 | 1577 | 1.4 | 0.5 | 1.0 | 6 | 8.5 | 15.3 | |||
| Caryophyllene oxide | 1585 | 1582 | 1.0 | 2.0 | 12.4 | 0.9 | 2.0 | ||||
| Globulol | 1592 | 1590 | 0.9 | ||||||||
| Viridiflorol | 1597 | 1592 | 1.2 | ||||||||
| Carotol | 1586 | 1594 | 2.7 | ||||||||
| 6-Methoxyelemicin | 1601 | 1595 | 6.3 | ||||||||
| Guaiol | 1597 | 1600 | 1.2 | 4.0 | 5.2 | ||||||
| Humulene epoxide II | 1608 | 1608 | 1.0 | 1.7 | |||||||
| Z-Asarone | 1625 | 1616 | 3.4 | ||||||||
| 1,10-di-epi-Cubenol | 1620 | 1618 | 3.0 | 1.4 | |||||||
| Junenol | 1620 | 1618 | 0.6 | ||||||||
| 1-epi-Cubenol | 1628 | 1627 | 3.8 | 0.4 | 3.3 | ||||||
| Muurola-4,10(14)-dien-1β-ol | 1628 | 1630 | 3.3 | ||||||||
| epi-α-Cadinol | 1642 | 1638 | 2.0 | 0.5 | |||||||
| allo-Aromadendrene epoxide | 1632 | 1639 | 1.4 | ||||||||
| Caryophylla-4(12),8(13)-dien-5β-ol | 1636 | 1639 | 3.2 | ||||||||
| α-Muurolol | 1642 | 1644 | 7.8 | 0.9 | 1.8 | ||||||
| Cubenol | 1646 | 1645 | 2.2 | 1.5 | |||||||
| α-Cadinol | 1652 | 1652 | 2.2 | 1.8 | 1.5 | 1.8 | |||||
| Valerianol | 1654 | 1656 | 6.8 | ||||||||
| Selin-11-en-4α-ol | 1657 | 1658 | 3.1 | 20.6 | |||||||
| 7-epi-α-Eudesmol | 1658 | 1662 | 4.2 | ||||||||
| Bulnesol | 1662 | 1666 | 29.5 | ||||||||
| 14-Hydroxy-(Z)-caryophyllene | 1666 | 1666 | 2.3 | 1.5 | 0.9 | 2.1 | |||||
| E-Asarone | 1683 | 1675 | 3.6 | 8.8 | |||||||
| β-Sinensal | 1699 | 1699 | 0.6 | ||||||||
| E,E-Farnesylacetate | 1843 | 1843 | 10.1 | ||||||||
| Isohibaene | 1922 | 1923 | 0.7 | ||||||||
| Kaurene | 2034 | 2034 | 34.0 |
| Material | MIC (μg·mL−1) | IC50 (μg·mL−1) | ||||
|---|---|---|---|---|---|---|
| P. aer | E. coli | S. epi | S. aur | B. cer | MCF-7 | |
| Can-L | 1250.0 | 19.5 | 625.0 | 625.0 | 625.0 | 63.9 ± 3.7 |
| Cau-L | 1250.0 | 19.5 | 625.0 | 625.0 | 312.5 | 64.0 ± 3.7 |
| Cuj-L | 1250.0 | 19.5 | 312.5 | 625.0 | 312.5 | 67.7 ± 3.7 |
| α-Pinene | 625 | 312 | - | 312 | 625 | 69.5 ± 1.3 |
| β-Pinene | 1250 | 625 | - | 625 | 312 | 71.2 ± 2.0 |
| Limonene | 1250 | 625 | - | 312 | 625 | 77.4 ± 1.2 |
| β-Caryophyllene | 1250 | 312 | - | 312 | 156 | 59.4 ± 5.1 |
| α-Humulene | 1250 | 625 | - | 312 | 312 | 26.5 ± 5.6 |
| Germacrene D | 1250 | 625 | - | 156 | 625 | 69.6 ± 2.5 |
| Caryophyllene oxide | 1250 | 1250 | - | 1250 | 156 | 73.4 ± 3.7 |
| Gentamicin control | 1.22 | 2.44 | <19.5 | 0.61 | 1.22 | - |
| Tingenone control | <19.5 | <19.5 | - | 2.44 | 1.22 | 16.8 ± 1.7 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Silva, J.K.; Da Trindade, R.; Moreira, E.C.; Maia, J.G.S.; Dosoky, N.S.; Miller, R.S.; Cseke, L.J.; Setzer, W.N. Chemical Diversity, Biological Activity, and Genetic Aspects of Three Ocotea Species from the Amazon. Int. J. Mol. Sci. 2017, 18, 1081. https://doi.org/10.3390/ijms18051081
Da Silva JK, Da Trindade R, Moreira EC, Maia JGS, Dosoky NS, Miller RS, Cseke LJ, Setzer WN. Chemical Diversity, Biological Activity, and Genetic Aspects of Three Ocotea Species from the Amazon. International Journal of Molecular Sciences. 2017; 18(5):1081. https://doi.org/10.3390/ijms18051081
Chicago/Turabian StyleDa Silva, Joyce Kelly, Rafaela Da Trindade, Edith Cibelle Moreira, José Guilherme S. Maia, Noura S. Dosoky, Rebecca S. Miller, Leland J. Cseke, and William N. Setzer. 2017. "Chemical Diversity, Biological Activity, and Genetic Aspects of Three Ocotea Species from the Amazon" International Journal of Molecular Sciences 18, no. 5: 1081. https://doi.org/10.3390/ijms18051081
APA StyleDa Silva, J. K., Da Trindade, R., Moreira, E. C., Maia, J. G. S., Dosoky, N. S., Miller, R. S., Cseke, L. J., & Setzer, W. N. (2017). Chemical Diversity, Biological Activity, and Genetic Aspects of Three Ocotea Species from the Amazon. International Journal of Molecular Sciences, 18(5), 1081. https://doi.org/10.3390/ijms18051081