Magnetically Bioprinted Human Myometrial 3D Cell Rings as A Model for Uterine Contractility
Abstract
:1. Introduction
2. Results
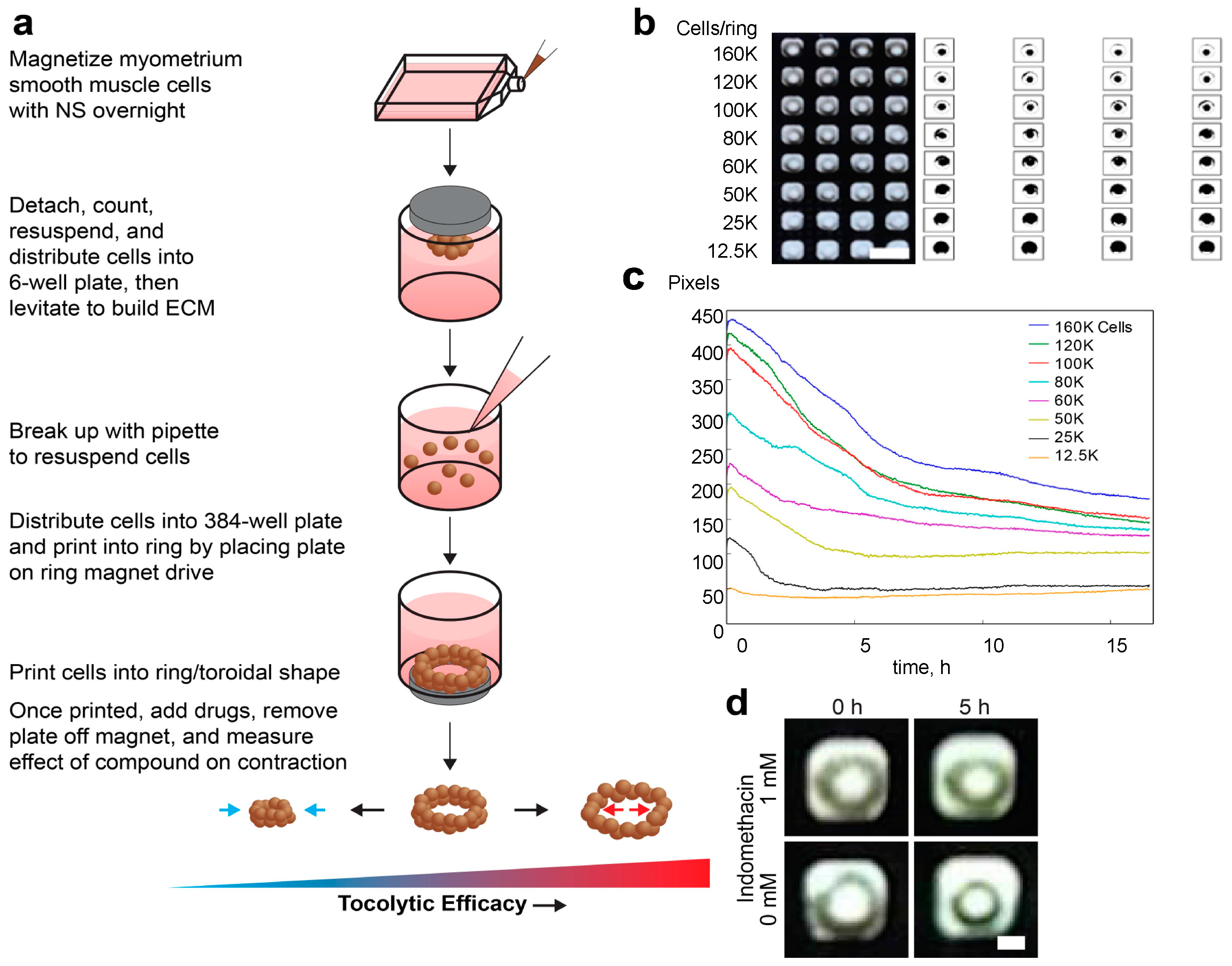
2.1. Bioprinting Commercially Available Human Myometrium Cells
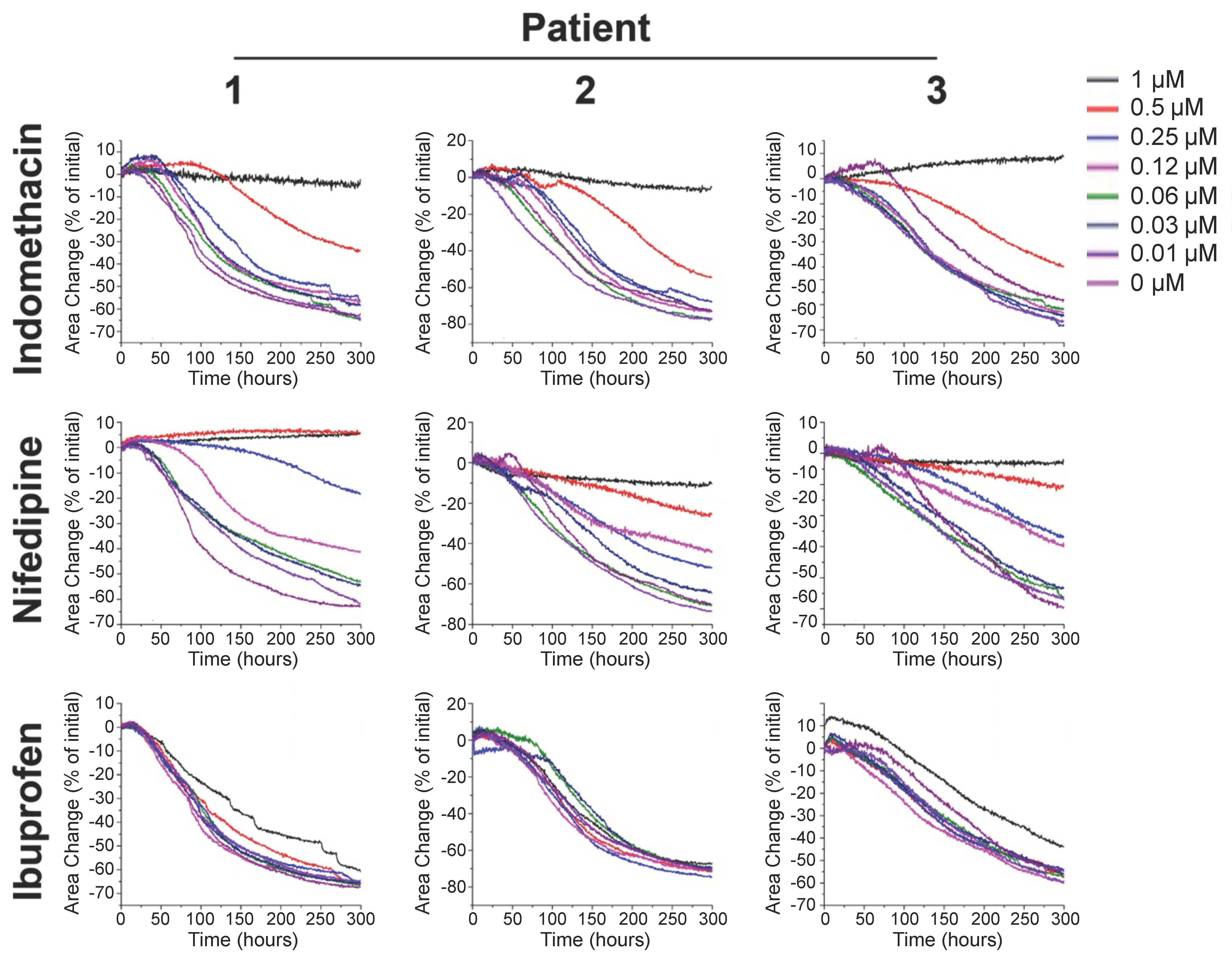
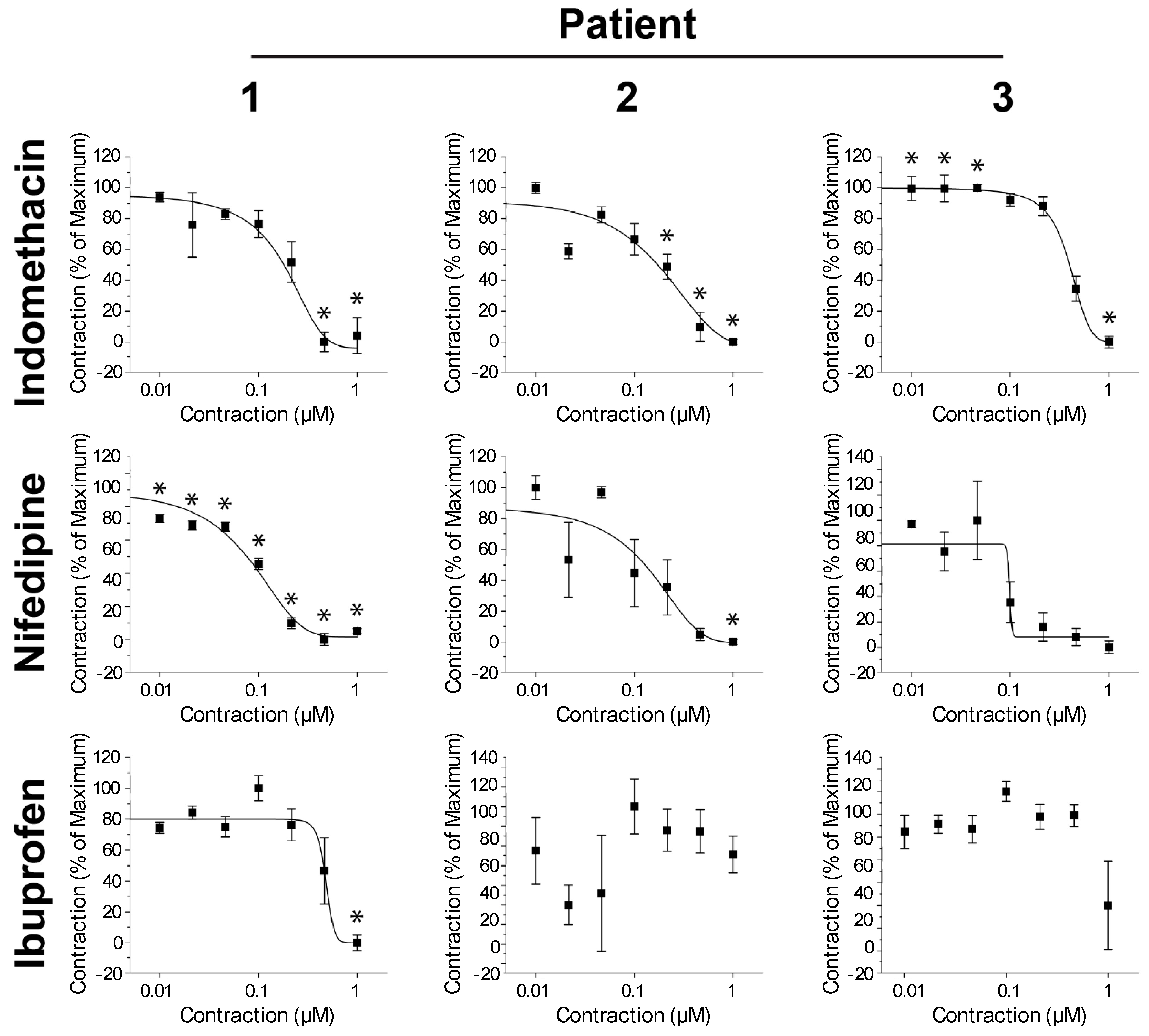
2.2. Bioprinting Freshly Excised and Cryopreserved Patient-Derived Myometrium Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.1.1. Commercially Available Human Uterine Smooth Muscle Cells
4.1.2. Patients-Derived Human Uterine Myometrial Cells
4.2. Cryopreservation of Cells from Uterine Samples from Patients
4.3. Magnetic 3D Bioprinting of Human Myometrial Cells
4.4. Myometrial Smooth Muscle Cell Ring Contractility Assay
4.5. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 2D | two-dimensional |
| 3D | three-dimensional |
| ECM | extracellular matrix |
| SMC | smooth muscle cells |
| NS | nano-shuttle |
| IC50 | inhibitory concentration 50, concentration at which the contractility is 50% of the maximum |
References
- Noe, M.; Kunz, G.; Herbertz, M.; Mall, G.; Leyendecker, G. The cyclic pattern of the immunocytochemical expression of oestrogen and progesterone receptors in human myometrial and endometrial layers: Characterization of the endometrial-subendometrial unit. Hum. Reprod. 1999, 14, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Blanks, A.M.; Shmygol, A.; Thornton, S. Preterm labour. Myometrial function in prematurity. Best Pract. Res. Clin. Obstet. Gynecol. 2007, 21, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Togashi, K.; Nakai, A.; Sugimura, K. Anatomy and physiology of the female pelvis: MR imaging revisited. J. Magn. Reson. Imaging 2001, 13, 842–849. [Google Scholar] [CrossRef] [PubMed]
- Lyons, E.A.; Taylor, P.J.; Zheng, X.H.; Ballard, G.; Levi, C.S.; Kredentser, J.V. Characterization of subendometrial myometrial contractions throughout the menstrual cycle in normal fertile women. Fertil. Steril. 1991, 55, 771–774. [Google Scholar] [CrossRef]
- De Vries, K.; Lyons, E.A.; Ballard, G.; Levi, C.S.; Lindsay, D.J. Contractions of the inner third of the myometrium. Am. J. Obstet. Gynecol. 1990, 162, 679–682. [Google Scholar] [CrossRef]
- Kunz, G.; Beil, D.; Deiniger, H.; Einspanier, A.; Mall, G.; Leyendecker, G. The uterine peristaltic pump. Normal and impeded sperm transport within the female genital tract. Adv. Exp. Med. Biol. 1997, 424, 267–277. [Google Scholar] [PubMed]
- Leyendecker, G.; Kunz, G.; Wildt, L.; Beil, D.; Deininger, H. Uterine hyperperistalsis and dysperistalsis as dysfunctions of the mechanism of rapid sperm transport in patients with endometriosis and infertility. Hum. Reprod. 1996, 11, 1542–1551. [Google Scholar] [CrossRef] [PubMed]
- Brosens, J.J.; Barker, F.G.; De Souza, N.M. Myometrial zonal differentiation and uterine junctional zone hyperplasia in the non-pregnant uterus. Hum. Reprod. Update 1998, 4, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Wray, S. Insights from physiology into myometrial function and dysfunction. Exp. Physiol. 2015, 100, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Huszar, G.; Naftolin, F. The myometrium and uterine cervix in normal and preterm labor. N. Engl. J. Med. 1984, 311, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Elovitz, M.A.; Mrinalini, C. Animal models of preterm birth. Trends Endocrinol. Metab. 2004, 15, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Knight, A. Animal experiments scrutinised: Systematic reviews demonstrate poor human clinical and toxicological utility. ALTEX 2007, 24, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Sawdy, R.; Knock, G.A.; Bennett, P.R.; Poston, L.; Aaronson, P.I. Effect of nimesulide and indomethacin on contractility and the Ca2+ channel current in myometrial smooth muscle from pregnant women. Br. J. Pharmacol. 1998, 125, 1212–1217. [Google Scholar] [CrossRef] [PubMed]
- Quaas, L.; Goppinger, A.; Zahradnik, H.P. The effect of acetylsalicylic acid and indomethacin on the catecholamine-and oxytocin-induced contractility and prostaglandin (6-keto-PGF1 alpha, PGF2 alpha)-production of human pregnant myometrial strips. Prostaglandins 1987, 34, 257–269. [Google Scholar] [CrossRef]
- Sawdy, R.J.; Sullivan, M.H.; Bennett, P.R. The effects of non-steroidal anti-inflammatory compounds on human myometrial contractility. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 109, 33–40. [Google Scholar] [CrossRef]
- Zhang, S. Beyond the Petri dish. Nat. Biotechnol. 2004, 22, 151–152. [Google Scholar] [CrossRef] [PubMed]
- Cukierman, E.; Pankov, R.; Stevens, D.R.; Yamada, K.M. Taking cell-matrix adhesions to the third dimension. Science 2001, 294, 1708–1712. [Google Scholar] [CrossRef] [PubMed]
- Pampaloni, F.; Reynaud, E.G.; Stelzer, E.H. The third dimension bridges the gap between cell culture and live tissue. Nat. Rev. Mol. Cell Biol. 2007, 8, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Bell, E.; Ivarsson, B.; Merrill, C. Production of a tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potential in vitro. Proc. Natl. Acad. Sci. USA 1979, 76, 1274–1278. [Google Scholar] [CrossRef] [PubMed]
- Cuchiara, M.P.; Allen, A.C.; Chen, T.M.; Miller, J.S.; West, J.L. Multilayer microfluidic PEGDA hydrogels. Biomaterials 2010, 31, 5491–5497. [Google Scholar] [CrossRef] [PubMed]
- Ware, M.J.; Colbert, K.; Keshishian, V.; Ho, J.; Corr, S.J.; Curley, S.A.; Biana, G. Generation of homogenous three-dimensional pancreatic cancer cell spheroids using an improved hanging drop technique. Tissue Eng. Part C Methods 2016, 22, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Ware, M.J.; Keshishian, V.; Law, J.J.; Ho, J.C.; Favela, C.A.; Rees, P.; Smith, B.; Mohammad, S.; Hwang, R.F.; Rajapakshe, K.; et al. Generation of an in vitro 3D PDAC stroma rich spheroid model. Biomaterials 2016, 108, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, D.P.; Parker, T.L.; Walker, D.A.; Alexander, C.; Ashford, M.B.; Gellert, P.R.; Garnett, M.C. Multiplexing spheroid volume, resazurin and acid phosphatase viability assays for high-throughput screening of tumour spheroids and stem cell neurospheres. PLoS ONE 2014, 9, e103817. [Google Scholar] [CrossRef] [PubMed]
- Timm, D.M.; Chen, J.; Sing, D.; Gage, J.A.; Haisler, W.L.; Neeley, S.K.; Raphael, R.M.; Dehghani, M.; Rosenblatt, K.P.; Rosenblatt, T.C.; et al. A high-throughput three-dimensional cell migration assay for toxicity screening with mobile device-based macroscopic image analysis. Sci. Rep. 2013, 3, 3000. [Google Scholar] [CrossRef] [PubMed]
- Tseng, H.; Gage, J.A.; Raphael, R.M.; Moore, R.H.; Killian, T.C.; Grande-Allen, K.J.; Souza, G.R. Assembly of a three-dimensional multitype bronchiole coculture model using magnetic levitation. Tissue Eng. Part C Methods 2013, 19, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Shen, T.; Gage, G.A.; Haisler, W.L.; Neeley, S.K.; Shen, T.; Hebel, C.; Barthlow, H.; Wagoner, M.; Souza, G.R. A high-throughput in vitro ring assay for vasoactivity using magnetic 3D. Sci. Rep. 2016, 6, 30640. [Google Scholar]
- Abramovici, A.; Cantu, J.; Jenkins, S.M. Tocolytic therapy for acute preterm labor. Obstet. Gynecol. Clin. N. Am. 2012, 39, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Daquinag, A.C.; Souza, G.R.; Kolonin, M.G. Adipose tissue engineering in three-dimensional levitation tissue culture system based on magnetic nanoparticles. Tissue Eng. Part C Methods 2013, 19, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Jaganathan, H.; Gage, J.; Leonard, F.; Srinivasan, S.; Souza, G.R.; Dave, B.; Godin, B. Three-dimensional in vitro co-culture model of breast tumor using magnetic levitation. Sci. Rep. 2014, 4, 6468. [Google Scholar] [CrossRef] [PubMed]
- Souza, G.R.; Godin, B.; Longo, M.; Refuerzo, J.S. (Inventors) Assignees: Nano3D Biosciences Inc., Methodist Hospital and the University of Texas Health Science Center. Contractility Assay. PCT Application #15374897, 10 December 2016. [Google Scholar]
- Souza, G.R.; Molina, J.R.; Raphael, R.M.; Ozawa, M.G.; Stark, D.J.; Levin, C.S.; Bronk, L.F.; Ananta, J.S.; Mandelin, J.; Georgescu, M.-M.; et al. Three-dimensional tissue culture based on magnetic cell levitation. Nat. Nanotechnol. 2010, 5, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Tseng, H.; Gage, J.A.; Shen, T.; Haisler, W.L.; Neeley, S.K.; Shiao, S.; Chen, J.; Desai, P.K.; Liao, A.; Hebel, C.; et al. A spheroid toxicity assay using magnetic 3D bioprinting and real-time mobile device-based imaging. Sci. Rep. 2015, 5, 13987. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Dhanani, N.; Tseng, H.; Souza, G.R.; Wang, G.; Cao, Y.; Ko, T.C.; Jiang, H.; Wang, R. Nanoparticle improved stem cell therapy for erectile dysfunction in a rat model of cavernous nerve injury. J. Urol. 2016, 195, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Monga, M.; Ku, C.Y.; Dodge, K.; Sanborn, B.M. Oxytocin-stimulated responses in a pregnant human immortalized myometrial cell line. Biol. Reprod. 1996, 55, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Haisler, W.L.; Timm, D.M.; Gage, J.A.; Tseng, H.; Killian, T.C.; Souza, G.R. Three-dimensional cell culturing by magnetic levitation. Nat. Protocol. 2013, 8, 1940–1949. [Google Scholar] [CrossRef] [PubMed]
| Drug | Patient 1 | Patient 2 | Patient 3 |
|---|---|---|---|
| Indomethacin | 0.18 | 0.17 | 0.40 |
| Nifedipine | 0.08 | 0.13 | 0.10 |
| Ibuprofen | 0.46 | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souza, G.R.; Tseng, H.; Gage, J.A.; Mani, A.; Desai, P.; Leonard, F.; Liao, A.; Longo, M.; Refuerzo, J.S.; Godin, B. Magnetically Bioprinted Human Myometrial 3D Cell Rings as A Model for Uterine Contractility. Int. J. Mol. Sci. 2017, 18, 683. https://doi.org/10.3390/ijms18040683
Souza GR, Tseng H, Gage JA, Mani A, Desai P, Leonard F, Liao A, Longo M, Refuerzo JS, Godin B. Magnetically Bioprinted Human Myometrial 3D Cell Rings as A Model for Uterine Contractility. International Journal of Molecular Sciences. 2017; 18(4):683. https://doi.org/10.3390/ijms18040683
Chicago/Turabian StyleSouza, Glauco R., Hubert Tseng, Jacob A. Gage, Arunmani Mani, Pujan Desai, Fransisca Leonard, Angela Liao, Monica Longo, Jerrie S. Refuerzo, and Biana Godin. 2017. "Magnetically Bioprinted Human Myometrial 3D Cell Rings as A Model for Uterine Contractility" International Journal of Molecular Sciences 18, no. 4: 683. https://doi.org/10.3390/ijms18040683
APA StyleSouza, G. R., Tseng, H., Gage, J. A., Mani, A., Desai, P., Leonard, F., Liao, A., Longo, M., Refuerzo, J. S., & Godin, B. (2017). Magnetically Bioprinted Human Myometrial 3D Cell Rings as A Model for Uterine Contractility. International Journal of Molecular Sciences, 18(4), 683. https://doi.org/10.3390/ijms18040683







