Effect of Clozapine on DNA Methylation in Peripheral Leukocytes from Patients with Treatment-Resistant Schizophrenia
Abstract
:1. Introduction
2. Results
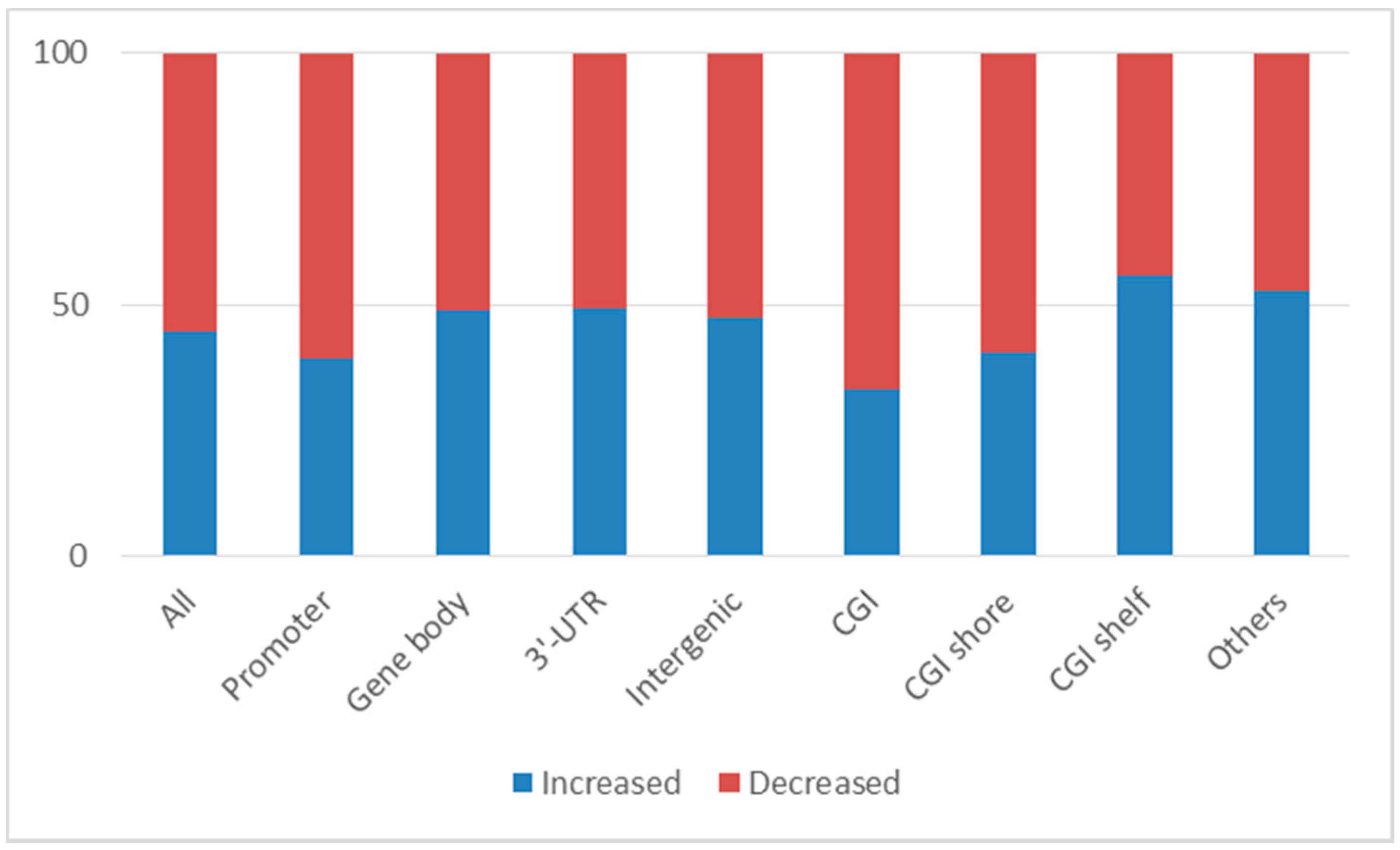
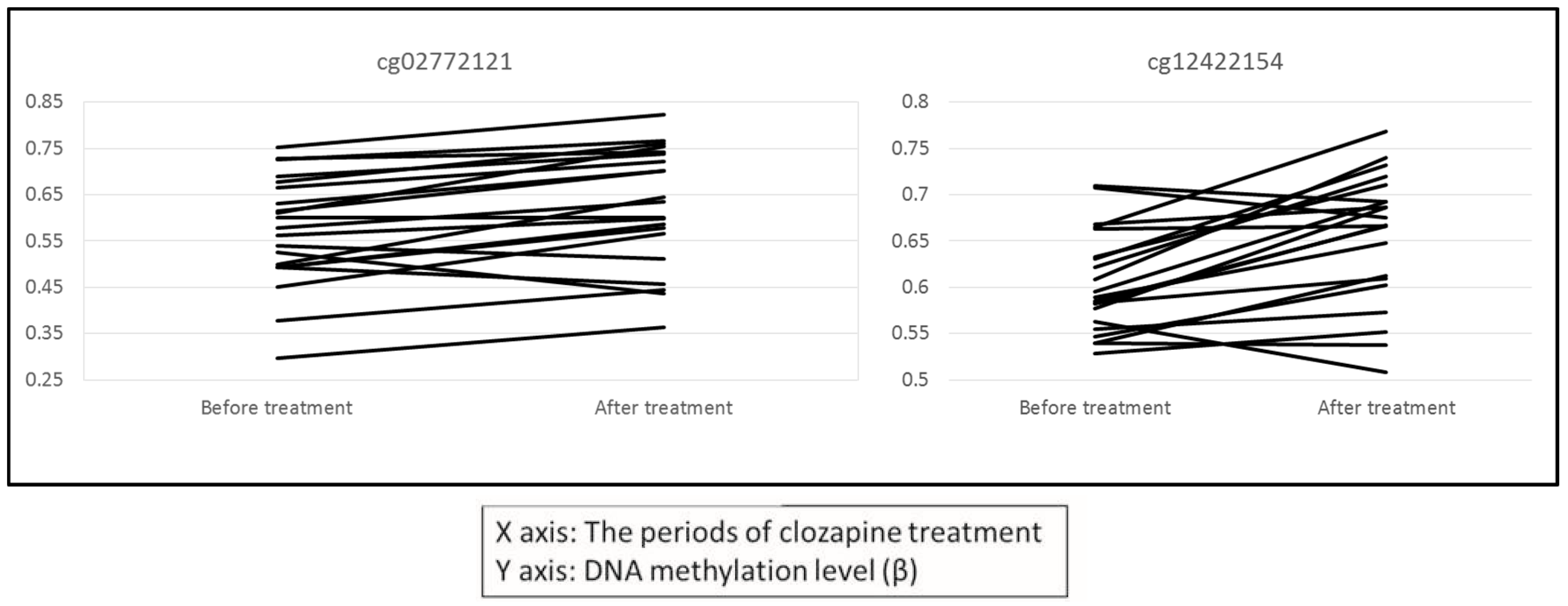
2.1. Changes in DNA Methylation in Leukocytes after Clozapine Treatment
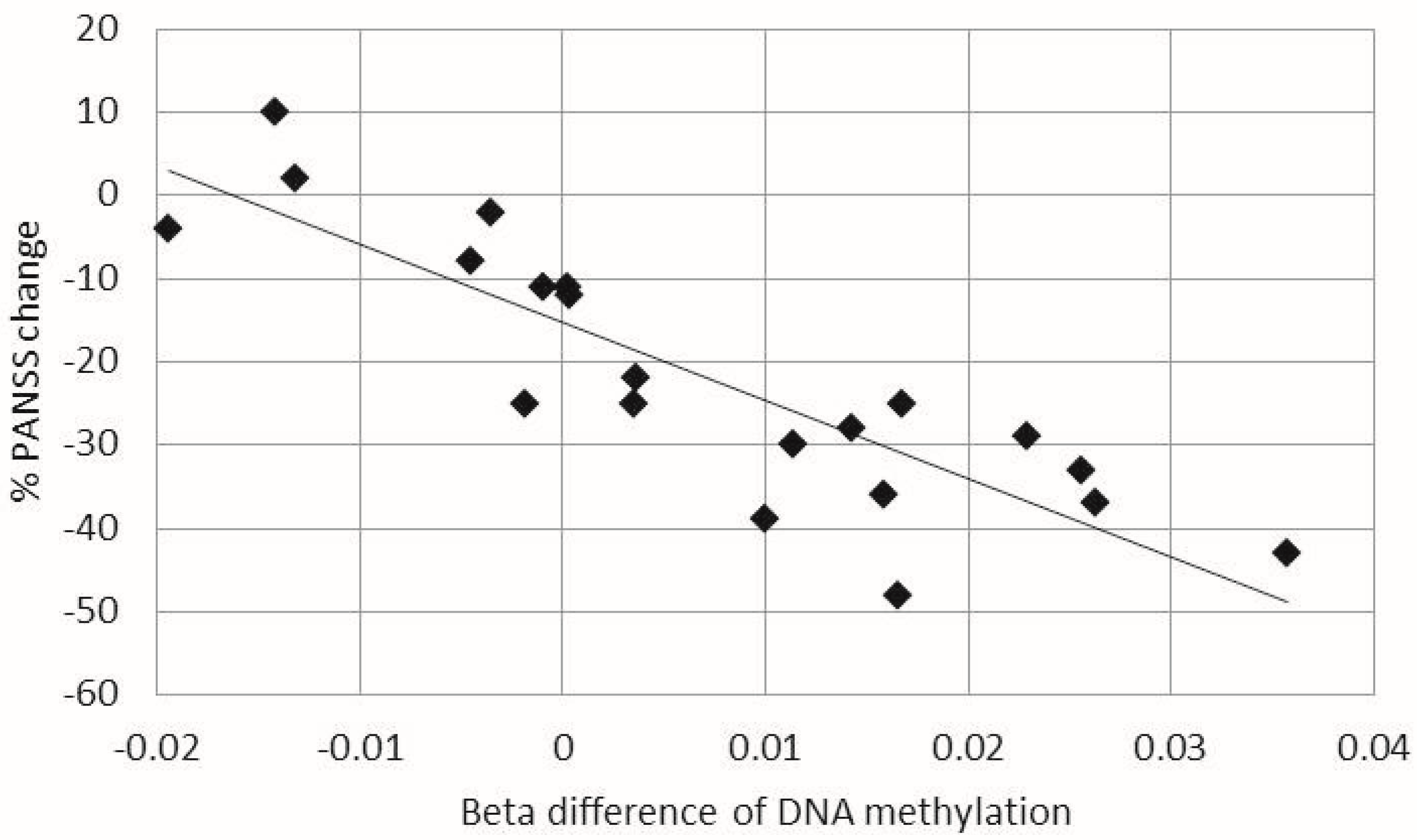
2.2. Correlations between Changes in DNA Methylation in Leukocytes and Clinical Outcomes
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Analysis of DNA Methylation
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gaebel, W.; Zielasek, J. Schizophrenia in 2020: Trends in diagnosis and therapy. Psychiatry Clin. Neurosci. 2015, 69, 661–673. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, H.Y. Treatment-resistant schizophrenia—The role of clozapine. Curr. Med. Res. Opin. 1997, 14, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Kane, J.; Honigfeld, G.; Singer, J.; Meltzer, H. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch. Gen. Psychiatry 1988, 45, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Wahlbeck, K.; Cheine, M.; Essalim, A.; Adams, C. Evidence of Clozapine’s Effectiveness in Schizophrenia: A Systematic Review and Meta-Analysis of Randomized Trials. Am. J. Psychiatry 1999, 156, 990–999. [Google Scholar] [PubMed]
- Corvin, A.P. Neuronal cell adhesion genes: Key players in risk for schizophrenia, bipolar disorder and other neurodevelopmental brain disorders? Cell Adhes. Migr. 2010, 4, 511–514. [Google Scholar] [CrossRef]
- Nakazawa, T.; Kikuchi, M.; Ishikawa, M.; Yamamori, H.; Nagayasu, K.; Matsumoto, T.; Fujimoto, M.; Yasuda, Y.; Fujiwara, M.; Okada, S.; et al. Differential gene expression profiles in neurons generated from lymphoblastoid B-cell line-derived iPS cells from monozygotic twin cases with treatment-resistant schizophrenia and discordant responses to clozapine. Schizophr. Res. 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Tang, B.; He, Y.; Jin, P. DNA methylation dynamics in neurogenesis. Epigenomics 2016, 8, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Tognini, P.; Napoli, D.; Pizzorusso, T. Dynamic DNA methylation in the brain: A new epigenetic mark for experience-dependent plasticity. Front. Cell. Neurosci. 2015, 9, 331. [Google Scholar] [CrossRef] [PubMed]
- Smith, Z.D.; Meissner, A. DNA methylation: Roles in mammalian development. Nat. Rev. Genet. 2013, 14, 204–220. [Google Scholar] [CrossRef] [PubMed]
- Pidsley, R.; Mill, J. Epigenetic studies of psychosis: Current findings, methodological approaches, and implications for postmortem research. Biol. Psychiatry 2011, 69, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, M.; Bundo, M.; Kasai, K.; Iwamoto, K. DNA methylation in schizophrenia: Progress and challenges of epigenetic studies. Genome Med. 2012, 4, 96. [Google Scholar] [CrossRef]
- Kinoshita, M.; Numata, S.; Tajima, A.; Shimodera, S.; Ono, S.; Imamura, A.; Iga, J.; Watanabe, S.; Kikuchi, K.; Kubo, H.; et al. DNA methylation signatures of peripheral leukocytes in schizophrenia. Neuromol. Med. 2013, 15, 95–101. [Google Scholar] [CrossRef]
- Akbarian, S. Epigenetic mechanisms in schizophrenia. Dialogues Clin. Neurosci. 2014, 16, 405–417. [Google Scholar] [PubMed]
- Kinoshita, M.; Numata, S.; Tajima, A.; Ohi, K.; Hashimoto, R.; Shimodera, S.; Imoto, I.; Takeda, M.; Ohmori, T. Aberrant DNA methylation of blood in schizophrenia by adjusting for estimated cellular proportions. Neuromol. Med. 2014, 16, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Numata, S.; Ye, T.; Herman, M.; Lipska, B.K. DNA methylation changes in the postmortem dorsolateral prefrontal cortex of patients with schizophrenia. Front. Genet. 2014, 5, 280. [Google Scholar] [CrossRef] [PubMed]
- Cariaga-Martinez, A.; Saiz-Ruiz, J.; Alelú-Paz, R. From Linkage Studies to Epigenetics: What We Know and What We Need to Know in the Neurobiology of Schizophrenia. Front. Neurosci. 2016, 10, 202. [Google Scholar] [CrossRef] [PubMed]
- Montano, C.; Taub, M.A.; Jaffe, A.; Briem, E.; Feinberg, J.I.; Trygvadottir, R.; Idrizi, A.; Runarsson, A.; Berndsen, B.; Gur, R.C.; et al. Association of DNA Methylation Differences With Schizophrenia in an Epigenome-Wide Association Study. JAMA Psychiatry 2016, 73, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Teroganova, N.; Girshkin, L.; Suter, C.M.; Green, M.J. DNA methylation in peripheral tissue of schizophrenia and bipolar disorder: A systematic review. BMC Genet. 2016, 17, 27. [Google Scholar] [CrossRef] [PubMed]
- Melka, M.G.; Castellani, C.A.; Laufer, B.I.; Rajakumar, R.N.; O’Reilly, R.; Singh, S.M. Olanzapine induced DNA methylation changes support the dopamine hypothesis of psychosis. Mol. Psychiatry 2013, 1, 19. [Google Scholar] [CrossRef] [PubMed]
- Melka, M.G.; Laufer, B.I.; McDonald, P.; Castellani, C.A.; Rajakumar, R.N.; O’Reilly, R.; Singh, S.M. The effects of olanzapine on genome-wide DNA methylation in the hippocampus and cerebellum. Clin. Epigenet. 2014, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Melka, M.G.; Castellani, C.A.; Rajakumar, N.; O’Reilly, R.; Singh, S.M. Olanzapine-induced methylation alters cadherin gene families and associated pathways implicated in psychosis. BMC Neurosci. 2014, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- Murata, Y.; Nishioka, M.; Bundo, M.; Sunaga, F.; Kasai, K.; Iwamoto, K. Comprehensive DNA methylation analysis of human neuroblastoma cells treated with blonanserin. Neurosci. Lett. 2014, 563, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Melka, M.G.; Rajakumar, R.N.; O’Reilly, R.; Singh, S.M. Olanzapine-induced DNA methylation in the hippocampus and cerebellum in genes mapped to human 22q11 and implicated in schizophrenia. Psychiatr. Genet. 2015, 25, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, H.; Bundo, M.; Asai, T.; Sunaga, F.; Ueda, J.; Ishigooka, J.; Kasai, K.; Kato, T.; Iwamoto, K. Effects of quetiapine on DNA methylation in neuroblastoma cells. Prog. Neuropsychopharmacol. Biol. Psychiatry 2015, 56, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Dong, E.; Nelson, M.; Grayson, D.R.; Costa, E.; Guidotti, A. Clozapine and sulpiride but not haloperidol or olanzapine activate brain DNA demethylation. Proc. Natl. Acad. Sci. USA 2008, 105, 13614–13619. [Google Scholar] [CrossRef] [PubMed]
- Dong, E.; Tueting, P.; Matrisciano, F.; Grayson, D.R.; Guidotti, A. Behavioral and molecular neuroepigenetic alterations in prenatally stressed mice: Relevance for the study of chromatin remodeling properties of antipsychotic drugs. Transl. Psychiatry 2016, 6, e711. [Google Scholar] [CrossRef] [PubMed]
- Cherlyn, S.Y.; Woon, P.S.; Liu, J.J.; Ong, W.Y.; Tsai, G.C.; Sim, K. Genetic association studies of glutamate, GABA and related genes in schizophrenia and bipolar disorder: A decade of advance. Neurosci. Biobehav. Rev. 2010, 34, 958–977. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Guha, S.; Ikeda, M.; Iwata, N.; Malhotra, A.K.; Pe’er, I.; Darvasi, A.; Lencz, T. Excess of homozygosity in the major histocompatibility complex in schizophrenia. Hum. Mol. Genet. 2014, 23, 6088–6095. [Google Scholar] [CrossRef] [PubMed]
- Coburn, L.; Lopez, H.; Caldwell, B.J.; Moussa, E.; Yap, C.; Priya, R.; Noppe, A.; Roberts, A.P.; Lobaskin, V.; Yap, A.S.; et al. Contact inhibition of locomotion and mechanical cross-talk between cell–cell and cell–substrate adhesion determine the pattern of junctional tension in epithelial cell aggregates. Mol. Biol. Cell 2016, 27, 3436–3448. [Google Scholar] [CrossRef] [PubMed]
- Berrier, A.L.; Yamada, K.M. Cell-matrix adhesion. J. Cell. Physiol. 2007, 213, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Nagappan-Chettiar, S.; Johnson-Venkatesh, E.M.; Umemori, H. Activity-dependent proteolytic cleavage of cell adhesion molecules regulates excitatory synaptic development and function. Neurosci. Res. 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Bukalo, O.; Fentrop, N.; Lee, A.Y.; Salmen, B.; Law, J.W.; Wotjak, C.T.; Schweizer, M.; Dityatev, A.; Schachner, M. Conditional ablation of the neural cell adhesion molecule reduces precisionof spatial learning, long-term potentiation, and depression in the CA1subfield of mouse hippocampus. J. Neurosci. 2004, 24, 1565–1577. [Google Scholar] [CrossRef] [PubMed]
- Stoenica, L.; Senkov, O.; Gerardy-Schahn, R.; Weinhold, B.; Schachner, M.; Dityatev, A. In vivo synaptic plasticity in the dentate gyrus of mice deficient inthe neural cell adhesion molecule NCAM or its polysialic acid. Eur. J. Neurosci. 2006, 23, 2255–2264. [Google Scholar] [CrossRef] [PubMed]
- O’Dushlaine, C.; Kenny, E.; Heron, E.; Donohoe, G.; Gill, M.; Morris, D.; International Schizophrenia Consortium; Corvin, A. Molecular pathways involved in neuronal cell adhesion and membrane scaffolding contribute to schizophrenia and bipolar disorder susceptibility. Mol. Psychiatry 2011, 16, 286–292. [Google Scholar]
- Piras, F.; Schiff, M.; Chiapponi, C.; Bossù, P.; Mühlenhoff, M.; Caltagirone, C.; Gerardy-Schahn, R.; Hildebrandt, H.; Spalletta, G. Brain structure, cognition and negative symptoms in schizophrenia are associated with serum levels of polysialic acid-modified NCAM. Transl. Psychiatry 2015, 5, e658. [Google Scholar] [CrossRef] [PubMed]
- Aonurm-Helm, A.; Jaako, K.; Jürgenson, M.; Zharkovsky, A. Pharmacological approach for targeting dysfunctional brain plasticity: Focus on neural cell adhesion molecule (NCAM). Pharmacol. Res. 2016, 113 Pt B, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Crisafulli, C.; Chiesa, A.; Han, C.; Lee, S.J.; Shim, D.S.; Balzarro, B.; Andrisano, C.; Sidoti, A.; Patkar, A.A.; Pae, C.U.; et al. Possible influence of CREB1, CREBBP and CREM variants on diagnosis and treatment outcome in patients with schizophrenia. Neurosci. Lett. 2012, 508, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Jia, P.; Wang, L.; Meltzer, H.Y.; Zhao, Z. Common variants conferring risk of schizophrenia: A pathway analysis of GWAS data. Schizophr. Res. 2010, 122, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Kim, J.H.; Song, G.G. Pathway analysis of a genome-wide association study in schizophrenia. Gene 2013, 525, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Need, A.C.; Keefe, R.S.; Ge, D.; Grossman, I.; Dickson, S.; McEvoy, J.P.; Goldstein, D.B. Pharmacogenetics of antipsychotic response in the CATIE trial: A candidate gene analysis. Eur. J. Hum. Genet. 2009, 17, 946–957. [Google Scholar] [CrossRef] [PubMed]
- Yamamori, H.; Hashimoto, R.; Ishima, T.; Kishi, F.; Yasuda, Y.; Ohi, K.; Fujimoto, M.; Umeda-Yano, S.; Ito, A.; Hashimoto, K.; et al. Plasma levels of mature brain-derived neurotrophic factor (BDNF) and matrix metalloproteinase-9 (MMP-9) in treatment-resistant schizophrenia treated with clozapine. Neurosci. Lett. 2013, 556, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Yamamori, H.; Hashimoto, R.; Ishima, T.; Kishi, F.; Yasuda, Y.; Ohi, K.; Fujimoto, M.; Umeda-Yano, S.; Ito, A.; Hashimoto, K.; et al. Changes in plasma d-serine, l-serine, and glycine levels in treatment-resistant schizophrenia before and after clozapine treatment. Neurosci. Lett. 2014, 582, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.A.; Lemire, M.; Choufani, S.; Butcher, D.T.; Grafodatskaya, D.; Zanke, B.W.; Gallinger, S.; Hudson, T.J.; Weksberg, R. Discovery of cross-reactive probes and polymorphic CpGs in the Illumina Infinium HumanMethylation450 microarray. Epigenetics 2013, 8, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
| Probe ID | Average Beta Difference before Treatment of Clozapine | Average Beta Difference after Treatment of Clozapine | Average Beta Difference between Treatment of Clozapine | p-Value | Chromosome | Position * | UCSC RefGene Name | UCSC RefGene Group | Relation to UCSC CpG Island |
|---|---|---|---|---|---|---|---|---|---|
| cg15542713 | 0.427 | 0.496 | 0.070 | 1.1 × 10−4 | 1 | 42385581 | HIVEP3 | Promoter | CGI shore |
| cg02772121 | 0.570 | 0.624 | 0.054 | 3.2 × 10−4 | 6 | 30130881 | TRIM15 | Promoter | Others |
| cg10864200 | 0.608 | 0.557 | −0.050 | 7.3 × 10−4 | 4 | 720809 | PCGF3 | Promoter | CGI shelf |
| cg12422154 | 0.601 | 0.652 | 0.050 | 2.5 × 10−4 | 6 | 30130819 | TRIM15 | Promoter | Others |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinoshita, M.; Numata, S.; Tajima, A.; Yamamori, H.; Yasuda, Y.; Fujimoto, M.; Watanabe, S.; Umehara, H.; Shimodera, S.; Nakazawa, T.; et al. Effect of Clozapine on DNA Methylation in Peripheral Leukocytes from Patients with Treatment-Resistant Schizophrenia. Int. J. Mol. Sci. 2017, 18, 632. https://doi.org/10.3390/ijms18030632
Kinoshita M, Numata S, Tajima A, Yamamori H, Yasuda Y, Fujimoto M, Watanabe S, Umehara H, Shimodera S, Nakazawa T, et al. Effect of Clozapine on DNA Methylation in Peripheral Leukocytes from Patients with Treatment-Resistant Schizophrenia. International Journal of Molecular Sciences. 2017; 18(3):632. https://doi.org/10.3390/ijms18030632
Chicago/Turabian StyleKinoshita, Makoto, Shusuke Numata, Atsushi Tajima, Hidenaga Yamamori, Yuka Yasuda, Michiko Fujimoto, Shinya Watanabe, Hidehiro Umehara, Shinji Shimodera, Takanobu Nakazawa, and et al. 2017. "Effect of Clozapine on DNA Methylation in Peripheral Leukocytes from Patients with Treatment-Resistant Schizophrenia" International Journal of Molecular Sciences 18, no. 3: 632. https://doi.org/10.3390/ijms18030632
APA StyleKinoshita, M., Numata, S., Tajima, A., Yamamori, H., Yasuda, Y., Fujimoto, M., Watanabe, S., Umehara, H., Shimodera, S., Nakazawa, T., Kikuchi, M., Nakaya, A., Hashimoto, H., Imoto, I., Hashimoto, R., & Ohmori, T. (2017). Effect of Clozapine on DNA Methylation in Peripheral Leukocytes from Patients with Treatment-Resistant Schizophrenia. International Journal of Molecular Sciences, 18(3), 632. https://doi.org/10.3390/ijms18030632







