1. Introduction
Matrix-assisted laser desorption ionization mass spectrometry (MALDI MS) has become a suitable technique to explore complex biological materials, such as small organisms (e.g., ticks, flies, mites etc.) [
1,
2,
3], microorganisms [
4,
5,
6], or biological particles (e.g., pollen grains) [
7]. For this purpose, various sample preparation procedures have been modified, which involved grinding of organisms [
8], suspension/extraction methods [
3,
6] or the dissection of organs [
2].
Besides the digestion/extraction step, the fixation of such samples represents a crucial step for the success of the experiment. In contrast to MALDI MS of samples that are deposited on the target plate by being embedded in a matrix, or by covering tissue sections obtained from microtomes with a matrix [
9,
10,
11], these samples have to be fixed by alternative methods. Widely known is the application of surface-enhanced laser desorption ionization time-of-flight mass spectrometry (SELDI-TOF MS) [
12]. There, the target holders incorporate either modified chemical spots (hydrophobic, cationic, anionic, metal ions or hydrophilic), suited for protein expression profiling studies, or pre-activated biological surfaces designed for coupling of biomolecules [
12]. A recent overview of the application of SELDI-TOF MS for the discovery of biomarkers of various forms of human cancer is given by Gopal et al
. [
13]. However, this technique is limited to the investigation of analytes with comparatively low molecular masses.
Another approach is the fixation of samples in adhesives. Kaftan et al. used epoxy glue for the fixation of flies [
1]. Recently, we showed that pollen grains could be fixed in a resin typically used for atomic force microscopy (AFM) applications, without affecting the extraction of analytes or the ionization procedure in MALDI-TOF MS [
14]. In this approach, we showed recently that pollen grains could be fixed without affecting the extraction of analytes or the ionization procedure in MALDI-TOF MS. This approach, however, is time-consuming, since it involves various steps. First, pollen grains are deposited on a resin film that has to be prepared on the target before. Afterwards, the target plate is exposed to solvent vapor that weakens the resin and enables the pollen grains to be fixed in the viscous film. In a subsequent step, the fixed pollen samples are exposed to formic acid atmosphere, with the purpose of extracting the analytes from the pollen grain. Finally, the layer is covered by a matrix. It becomes obvious that this approach is not applicable for fast and automated pollen monitoring. Thus, we focused our efforts on the development of alternative methods for fast pollen grain fixation for MALDI MS probing.
For pollen classification and identification, the applicability of MALDI-TOF MS was proven, combining the mass spectral data with hierarchical cluster analysis (HCA) and principal component analysis (PCA) [
15]. There, almost 200 pollen samples of 74 different species from 11 genera and 2 plant orders were classified according to their taxonomic relationships, including discrimination of species that feature a very high chemical similarity [
15]. The application of multivariate statistics (e.g., PCA) to mass spectral patterns yields an enormous improvement concerning taxonomic classification of the samples compared to common microscopic techniques.
In order to simplify the sample preparation process, we suggest the application of a sticky conductive carbon tape. Conductive tape was used in this context previously for the analysis of methamphetamine incorporated in hair [
16]. A recent application was reported by Kajiwara et al., where spider mites were fixed on a double-sided carbon tape and analyzed by MALDI-TOF MS [
8]. In this article, we demonstrate that the use of conductive carbon tape on a MALDI target facilitates the pollen sample treatment, compared to a conventional sample deposition on a steel target. Different preparation strategies are compared, e.g., regarding the influence of acid concentration in the matrix solution and additional extraction with liquid or gaseous acid. In addition, possible interferences of the formic acid used for extraction with the new target material are examined. The findings show that our approach enhances the information that can be taken from the species-specific mass peak patterns. This results in a clear differentiation in subsequent pattern analysis, which is important when analyzing pollen grains in various real mixtures.
2. Results
Our experiment included pollen grains from four tree species: Scots pine (Pinus sylvestris), Japanese bog birch (Betula tatewakiana), Italian aider (Alnus cordata) and common hazel (Corylus avellana). The first sample represents the plant family of Pinaceae in the order of Coniferales, the other samples belong to the plant family of Betulaceae in the order of Fagales.
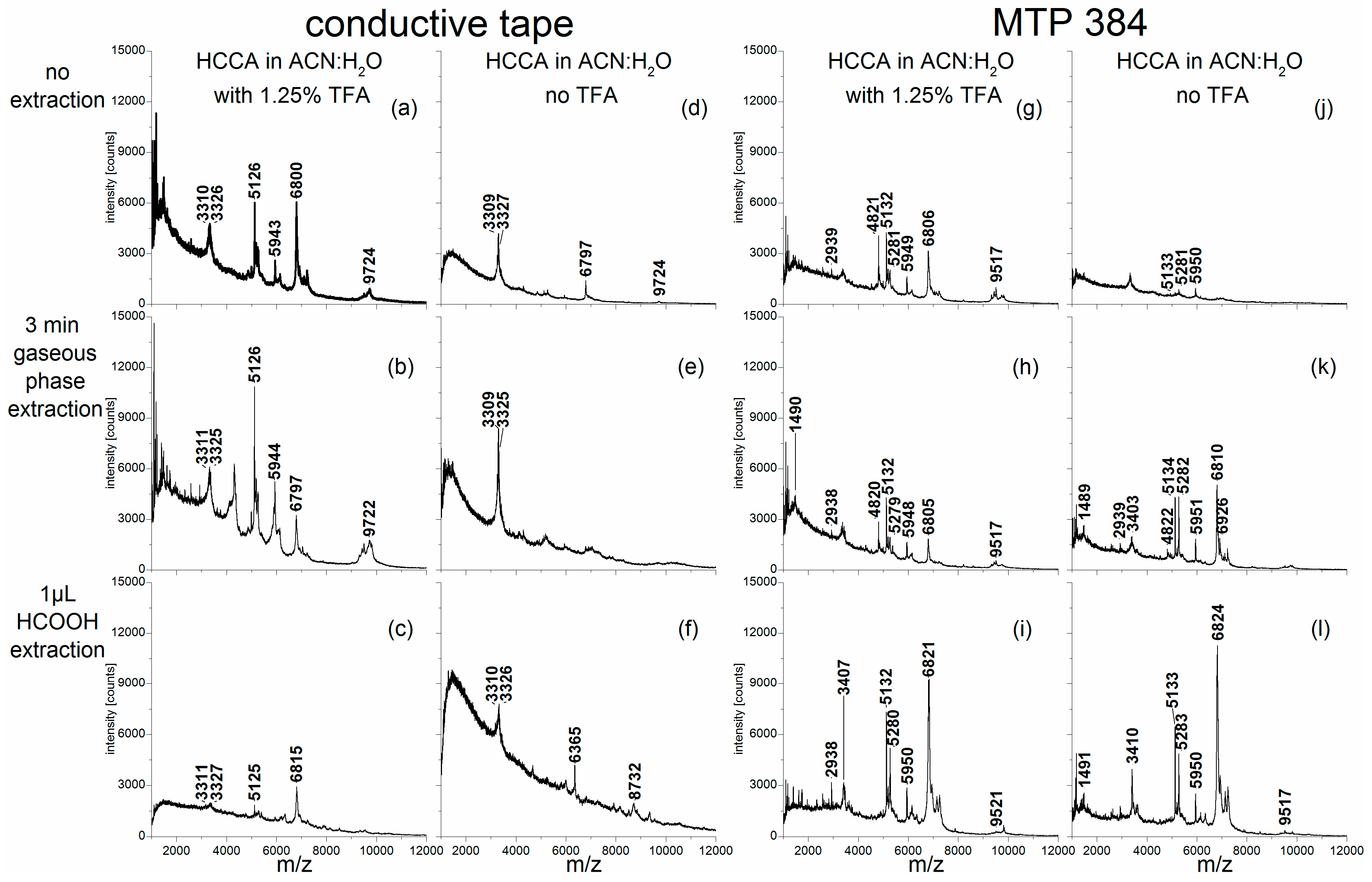
Spectra from all samples were recorded on conductive tape and on a stainless steel target, respectively. In
Figure 1, the average spectra from five repetitive measurements obtained with different sample treatments and target materials are depicted exemplarily for
Alnus cordata. All average spectra showed similar peak patterns, suggesting that these peak patterns are characteristic of this
Alnus cordata sample. However, slight shifts in the accurate masses and differences in signal intensities can be detected using different sample treatments. Spectra, which were recorded on the conductive tape (
Figure 1a–f) generally showed higher absolute signal intensities compared to those with the same treatment strategies measured on the steel target (
Figure 1g–l). Adding trifluoroacetic acid (TFA) to the matrix solution (
Figure 1a–c,g–i) also resulted in increased signal intensities, compared to the respective spectra obtained without TFA addition (
Figure 1d–f,j–l). When the extraction procedure was conducted with an additional droplet deposition of formic acid onto the pollen grains (
Figure 1c,f,i,l), spectra were obtained that feature low signal intensities, even though the individual peaks were clearly distinguishable. In the spectra obtained after formic acid gas phase extraction of the sample (
Figure 1b,e,h,k), the background in the lower mass range as well as the absolute intensities were more intense than in the spectra gained from droplet extraction. Spectra of the pollen samples with no additional extraction step (
Figure 1a,d,g,j) provided signal intensities in the lower mass range that are equal to those of the spectra obtained with gas phase extraction, whereas higher mass peaks (e.g., at
m/
z 6891 and
m/
z 9489) were more intense. The most intense spectrum showing characteristic masses of
Alnus cordata was recorded on conductive tape using a mixture of ACN/H
2O (1.25% TFA) as the matrix solvent and without any additional extraction steps (
Figure 1a).
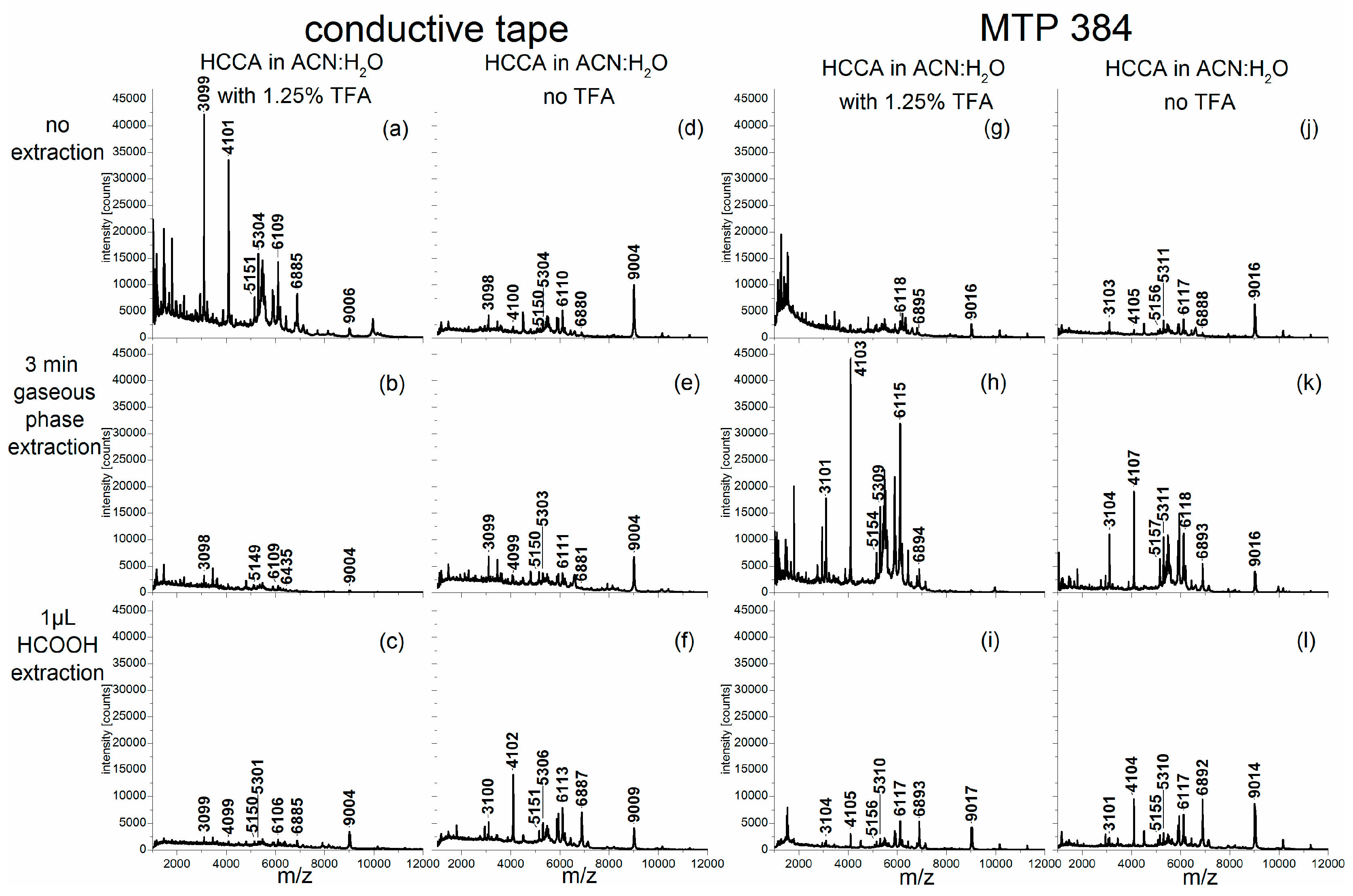
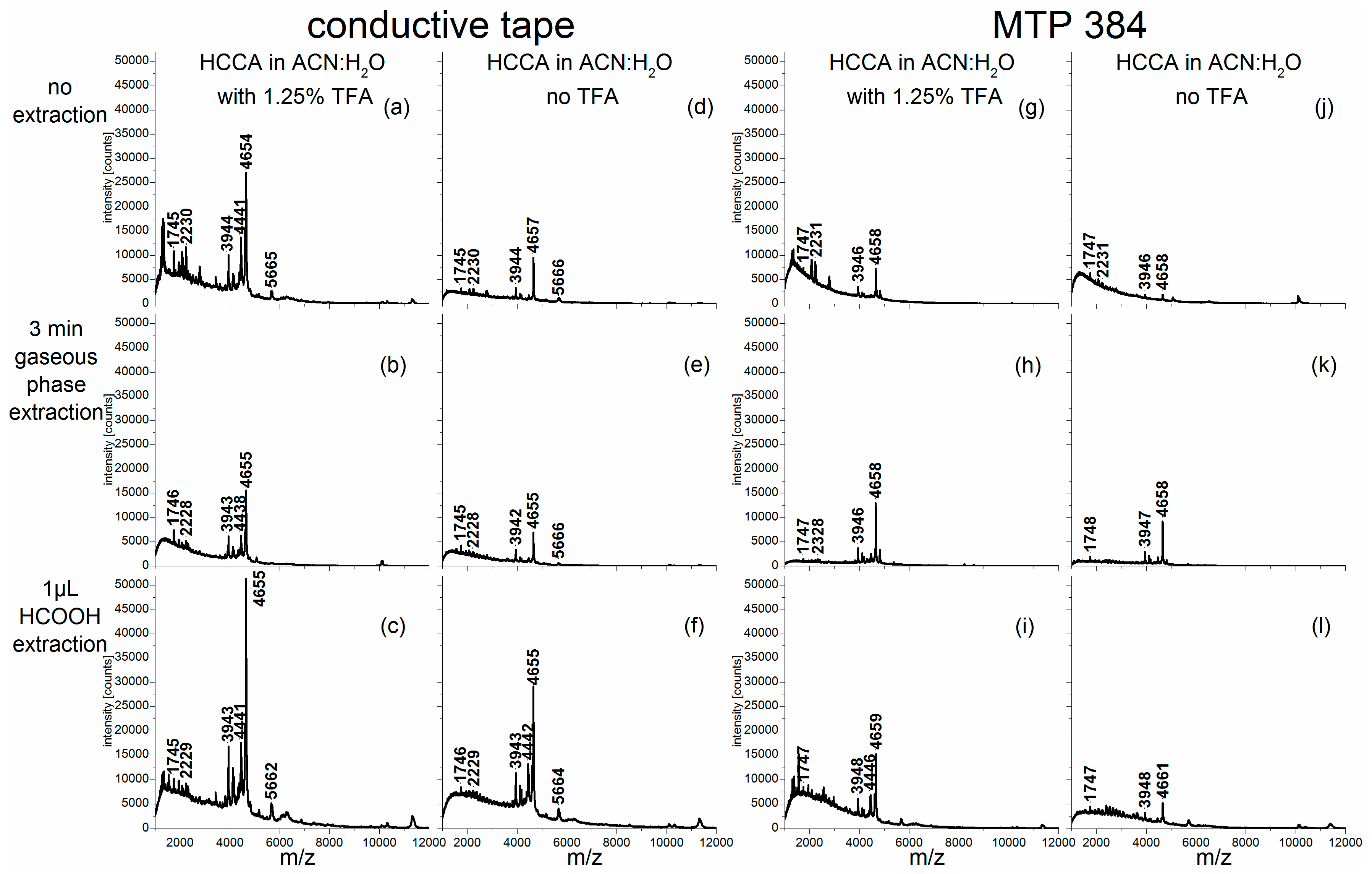
The corresponding mass spectra of the other pollen species (
Betula tatewakiana,
Corylus avellana and
Pinus sylvestris) are shown in the
Appendix (
Figure A1,
Figure A2 and
Figure A3). As indicated by the intensities of the corresponding signals, the use of a matrix solvent containing TFA promoted the extraction of analytes from the exines, i.e., the pollen grains’ outer shells, of most of these pollen samples much more than an additional extraction step using formic acid (for example compare
Figure A2a,h with
Figure A2c,f,i,l; or
Figure A3a–c with
Figure A3b,e,h,k). With the exception of the birch pollen investigations (
Betula tatewakiana) (
Figure A1), our previous findings concerning
Alnus cordata spectra in
Figure 1 were confirmed by the analysis of additional pollen species.
The study of the birch pollen sample with formic acid droplet deposition on the MTP (microtiter plate) steel target holder (
Figure A1i,l) yielded intense spectra that showed a very low background in the lower mass range. In that case, the use of conductive tape in combination with a matrix solvent containing TFA and no additional extraction steps (
Figure A1a) did not provide the spectra with the highest quality. Nevertheless, the spectrum also showed the characteristic peak pattern (as in
Figure A1i,l) that could be analyzed by multivariate tools, as will be discussed below.
At first glance, the spectra of all four pollen species show species-specific peak patterns, which already makes a rough differentiation of species possible by comparing the spectra by eye (
Figure 1 and
Figure A1,
Figure A2 and
Figure A3). In addition to this, an objective differentiation that enables an evaluation of particular sample treatment methods for the purpose of classification and ultimately identification can be obtained by using multivariate data analysis.
Multivariate methods, such as principal component analysis (PCA) can be applied to mass spectrometric data for several reasons, e.g., for quality control, to reduce complexity of the data, and to emphasize differences within a data set that enable classification [
17,
18,
19,
20].
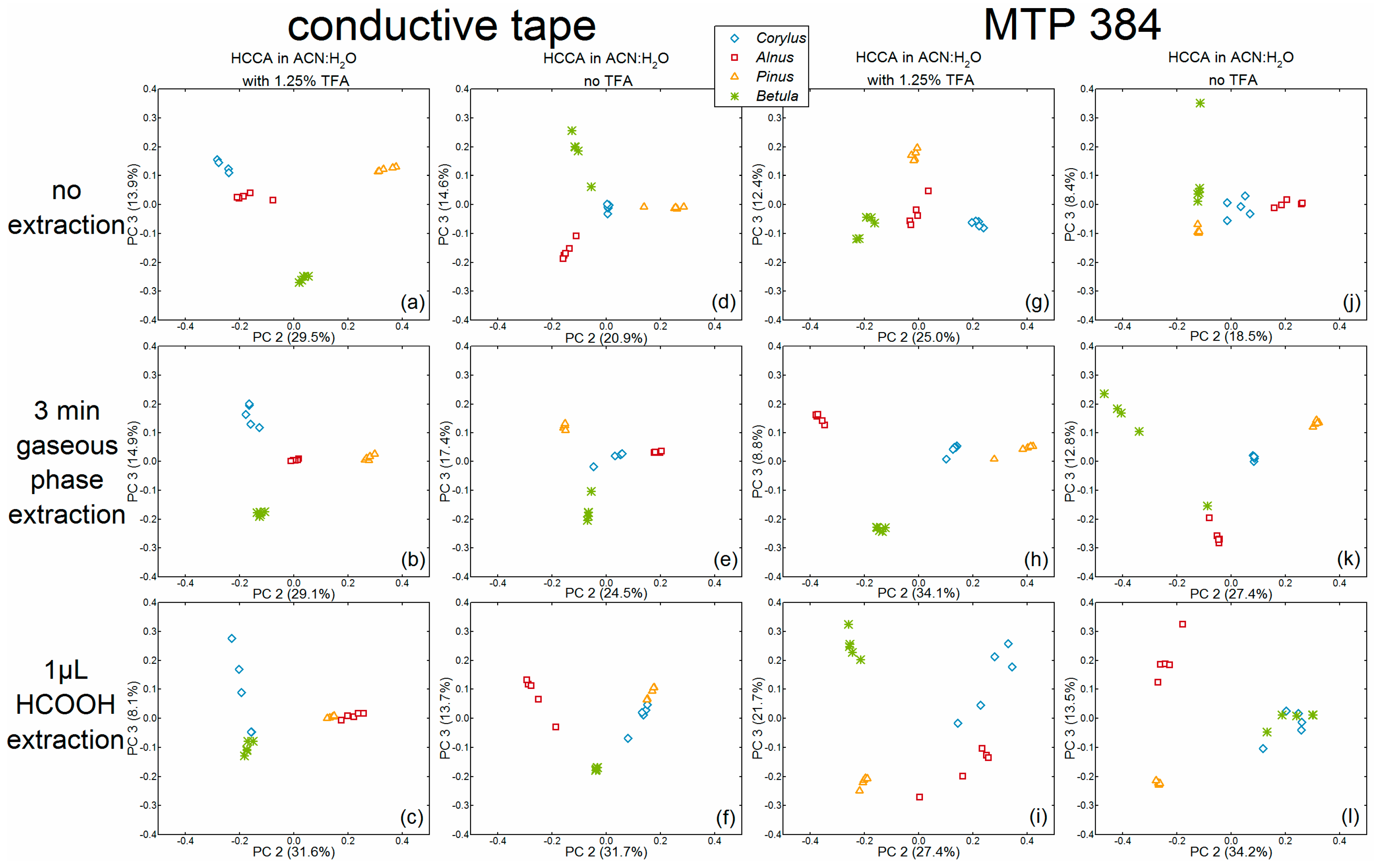
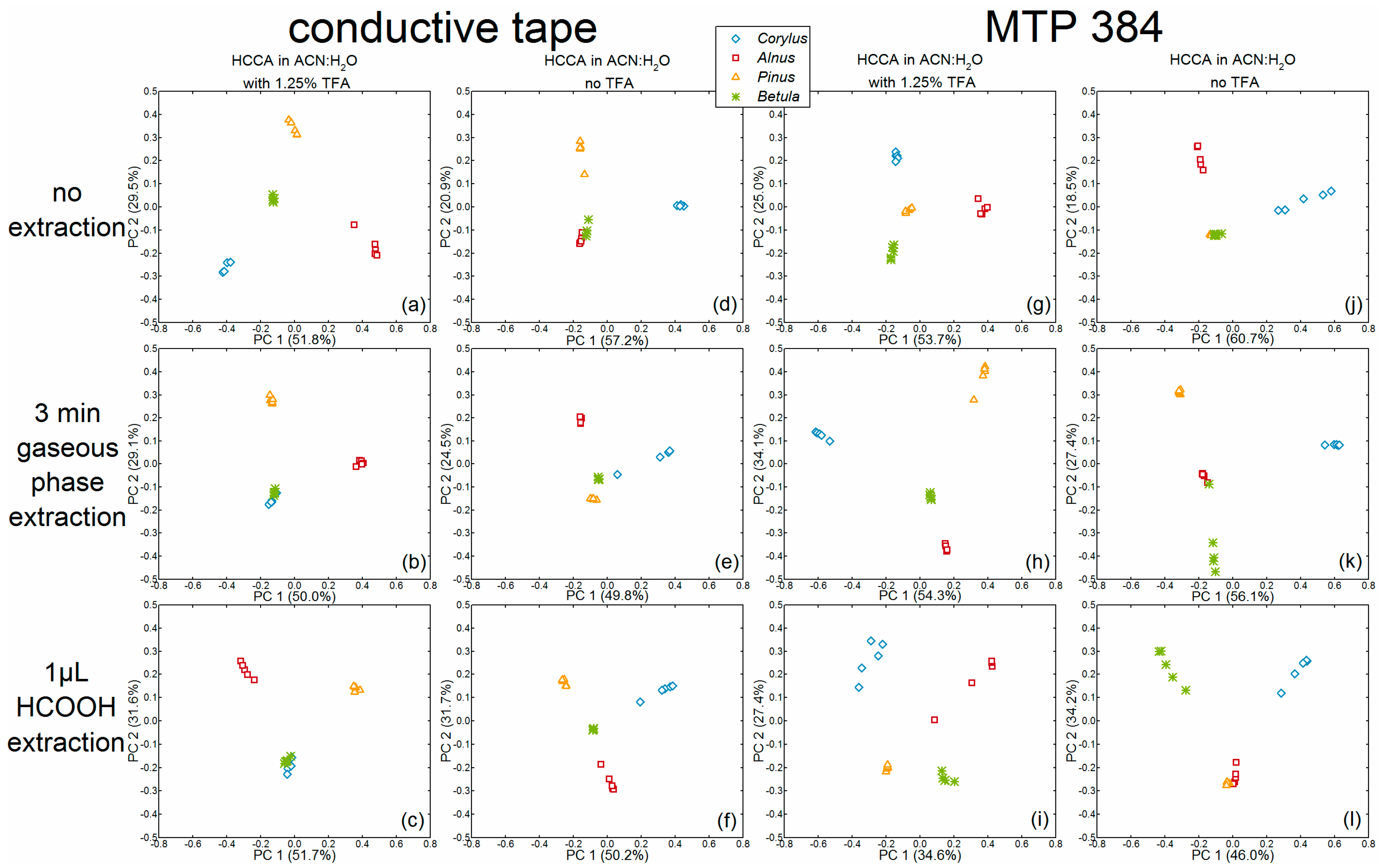
We applied PCA to compare classification of pollen species upon the application of different preparation procedures and to evaluate the preparation setup by finding differences in the chemical composition of the samples. Principal components (PCs) are linear combinations of the original mass spectra and generate a new coordinate system that is based on maximum variance between the spectra [
21]. They are characterized by two matrices: the loadings and the scores, as well as the percentage of original variance they are representing [
22,
23,
24]. The scores of the first and second principal components of the PCA using the respective spectra (
Figure 1 and
Figure A1,
Figure A2 and
Figure A3) after pre-treatment (see
Section 4.4 for further information) are depicted in
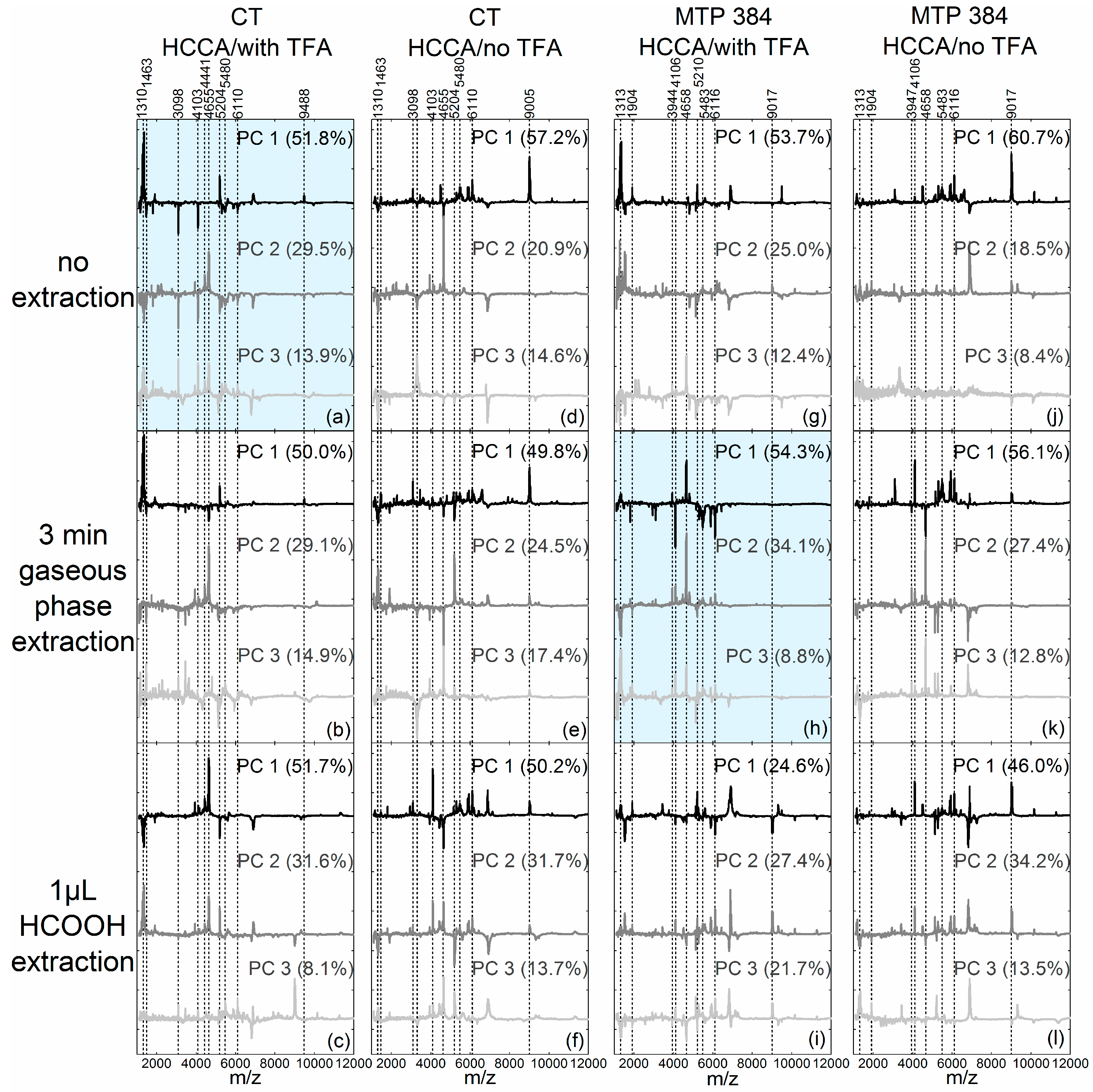
Figure 2. In order to compare the separation of the pollen species, the scores plot of each PCA was evaluated regarding the intra- versus inter-species distances. The loadings, given in
Figure 3, show the connection between the principal components and the original data and can be used for the subsequent interpretation of the scores values (
Figure 2). They indicate which parts of the spectrum are relevant for the respective PC and lead to the separation of the pollen spectra in the scores plots of
Figure 2.
The first parameter to be discussed using the PCA scores plots is the addition of TFA in the matrix solutions: PCAs conducted on spectra obtained without TFA in the matrix solution (
Figure 2d–f,j–l) shared a common trait, that their respective first PC enables a separation of
Corylus avellana spectra (light-blue diamonds) by positive values from negative or zero values of the spectra from all other species. In accord with this, the
Corylus-typical spectral regions, e.g., from
m/
z 5400 to 6117 [
15], yielded positive signals in the loadings of the respective first PCs (see
Figure 3, 2nd and 4th column, black loadings spectra). The other species, however, cannot be differentiated clearly from each other using the scores plots in
Figure 2d,e,j–l. The scores of the second and third PCs of the respective data sets, did not allow clear and accurate differentiation between the species
Alnus cordata,
Pinus sylvestris and
Betula tatewakiana (see
Figure A4) either.
In the PCAs of the data sets that were obtained with the addition of TFA in the matrix solution, a separation of individual pollen species using the first two PCs was possible more frequently, as indicated in the plots illustrated in
Figure 2a,g,h,i. However, in contrast to the plots in
Figure 2a,h, where the individual groups were clearly separated from each other, the distances of the different groups in the plot in
Figure 2g were much smaller. Moreover, considering the homogeneity of each individual group in these four plots, the plot in
Figure 2i especially showed very inhomogeneous distribution of the spectra of
Alnus cordata and
Corylus avellana. Since high intra-species variances are especially noticeable in the bottom row of
Figure 2c,f,i,l they could possibly be explained by the samples treatment with the formic acid droplet method. Different to this observation, the PCAs of data obtained by additional gaseous extraction (
Figure 2b,e,h,k) and by no additional extraction step (
Figure 2a,d,g,j) showed no definite tendency concerning intra-species variances.
Since the scores plots in
Figure 2a,h are suitable for a complete differentiation of pollen species, the underlying PCAs of these data shall be discussed in detail here. In
Figure 2a, the scores of
Corylus avellana spectra feature negative values for the first and second PCs, which result in negative
Corylus-typical peaks [
15] in the respective loadings (e.g., at
m/
z 3098,
m/
z 4103 and the range from
m/
z 5400 to 6110 shown in
Figure 3a, top black spectra and middle gray spectra).
Alnus cordata spectra provide scores values that are positive for the first PC and negative for the second PC (
Figure 2a). The corresponding loadings (
Figure 3a) showed peaks in the lower mass range of
m/
z 1310 to 1463 and peaks around
m/
z 5204 and
m/
z 9488 that can be assigned to
Alnus [
15]. The
Pinus sylvestris spectra were characterized by scores values close to zero for the first PC and very positive score values for the second PC. Consequently, the loadings of the second PC in
Figure 3a showed peaks at
m/
z 4441 and
m/
z 4655 [
15] that do not occur in the loadings of the first PC and that can be identified as
Pinus-typical (compare with
Figure A3a). The
Betula tatewakiana scores values in
Figure 2a were all close to zero, therefore the loadings showed no peaks that can be assigned to
Betula pollen spectra.
In
Figure 2h,
Corylus avellana spectra yield negative scores values for the first PC and positive scores values for the second PC. Hence, the respective loadings (
Figure 3h) showed negative and positive
Corylus-typical peaks in the top black and the middle gray loadings, respectively (e.g., at
m/
z 4106 and the mass range from
m/
z 5400 to 6116). Furthermore, the
Alnus cordata spectra in
Figure 2h had zero values for the first PC and negative values for the second PC. Thus, in the loadings of the second PC in
Figure 3h, negative values for the
Alnus-typical peak pattern in the lower mass range (
m/
z 1313 to 1400 and peaks around
m/
z 1904) can be found (compare with
Figure 1h). The
Pinus sylvestris spectra featured positive scores values for the first and second PCs (
Figure 2h), and
Pinus-typical peaks at
m/
z 3944 and
m/
z 4658 can be observed in the corresponding loadings in
Figure 3h, top black and middle gray loadings).
Betula tatewakiana spectra in the PCA of
Figure 2h, again, were characterized by scores values around zero, which result in loadings that did not show
Betula-typical characteristics.
3. Discussion
In order to compare different preparation techniques for MALDI-TOF MS classification of pollen, we systematically varied the extraction procedure, the target material and the matrix solution. Our results demonstrated that higher absolute signal intensities could be recorded in the spectra obtained on conductive tape compared to those that were received with the same treatment strategies on the steel target (compare a–f of
Figure 1 and
Figure A1,
Figure A2 and
Figure A3 with g–l of the corresponding figure). Nevertheless, conducting PCA on data sets measured on a steel target was still expedient (
Figure 2g–l). Kajiwara et al. [
8] observed a slightly higher mass shift and a missing of some lower mass range peaks when mass spectra are obtained on conductive tape. These findings could not be confirmed by our experiments, where the usage of conductive tape as a sample holder facilitated the collection of mass spectra with intense peaks and less noise (a–f of
Figure 1 and
Figure A1,
Figure A2 and
Figure A3). We assume that the use of the sticky conductive tape could be favorable in the extraction by keeping the extracted analytes closer to the pollen grains, and therefore, enabling higher analyte concentrations that lead to more intense signals. The increased sensitivity could be especially important for the detection of single pollen grains in future applications. As shown in a previous investigation based on the MTP approach, MALDI MS is capable of detecting a few, down to ~3 pollen grains at best [
7].
The treatment with acid seems to be indispensable for a sufficient extraction of analytes from the pollen grains (
Figure 1 and
Figure A1,
Figure A2 and
Figure A3). When no acid (formic acid or TFA, see d,j of
Figure 1 and
Figure A1,
Figure A2 and
Figure A3) is applied, the MALDI mass spectra exhibited less intense peaks and the different pollen species cannot be differentiated clearly in the PCA (
Figure 2d,j). When a matrix solution without TFA was used, the PCA is dominated by the separation of
Corylus avellana spectra from the spectra of the other species, most likely due to less noise and lower background in the
Corylus avellana spectra. The other pollen species did not yield spectra of sufficient quality (see d–f,i–l of
Figure 1,
Figure A1 and
Figure A3), leading to higher inner-class variances in the PCA. The addition of 1.25% TFA to the matrix solution led to an improvement in signal intensity and hence to higher quality spectra (a–c,g–i of
Figure 1 and
Figure A1,
Figure A2 and
Figure A3). As a consequence, the PCA of the data sets that were obtained with a matrix solution containing TFA most often enabled species-specific classification. The formic acid gas phase treatment resulted in intense signals from the birch pollen (
Figure A1b), reasonable intensities for
Pinus and
Alnus pollen (
Figure A3b and
Figure 1b) and insufficient data for
Corylus pollen (
Figure A2b). Nevertheless, the PCAs of this dataset showed a clear intra-species homogeneity, even though the scores values of
Corylus and
Betula-spectra were almost alike. The extraction procedure conducted with formic acid droplet deposition obtained spectra with various intensities for each respective species resulting in large intra-group variance in the PCA. This might be traced back to the non-reproducible evaporation of the formic acid, caused by different droplet spot sizes.