LC-MS/MS Analysis Unravels Deep Oxidation of Manganese Superoxide Dismutase in Kidney Cancer
Abstract
:1. Introduction
2. Results
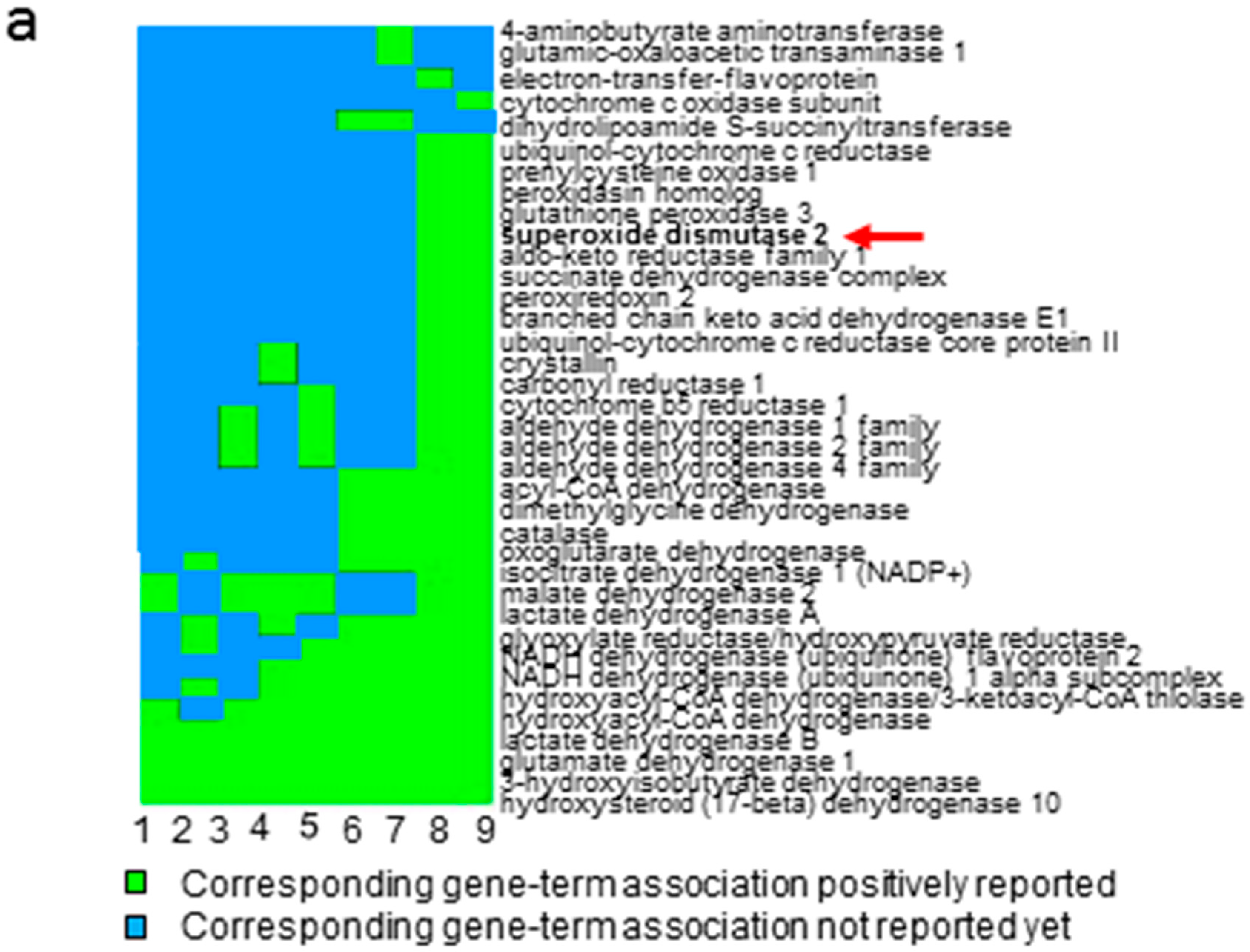
2.1. Quantitative Proteomic Analysis Suggests the Involvement of Anti-Oxidative Stress Pathway in ccRCC
2.2. Oxidative Modification of MNSOD
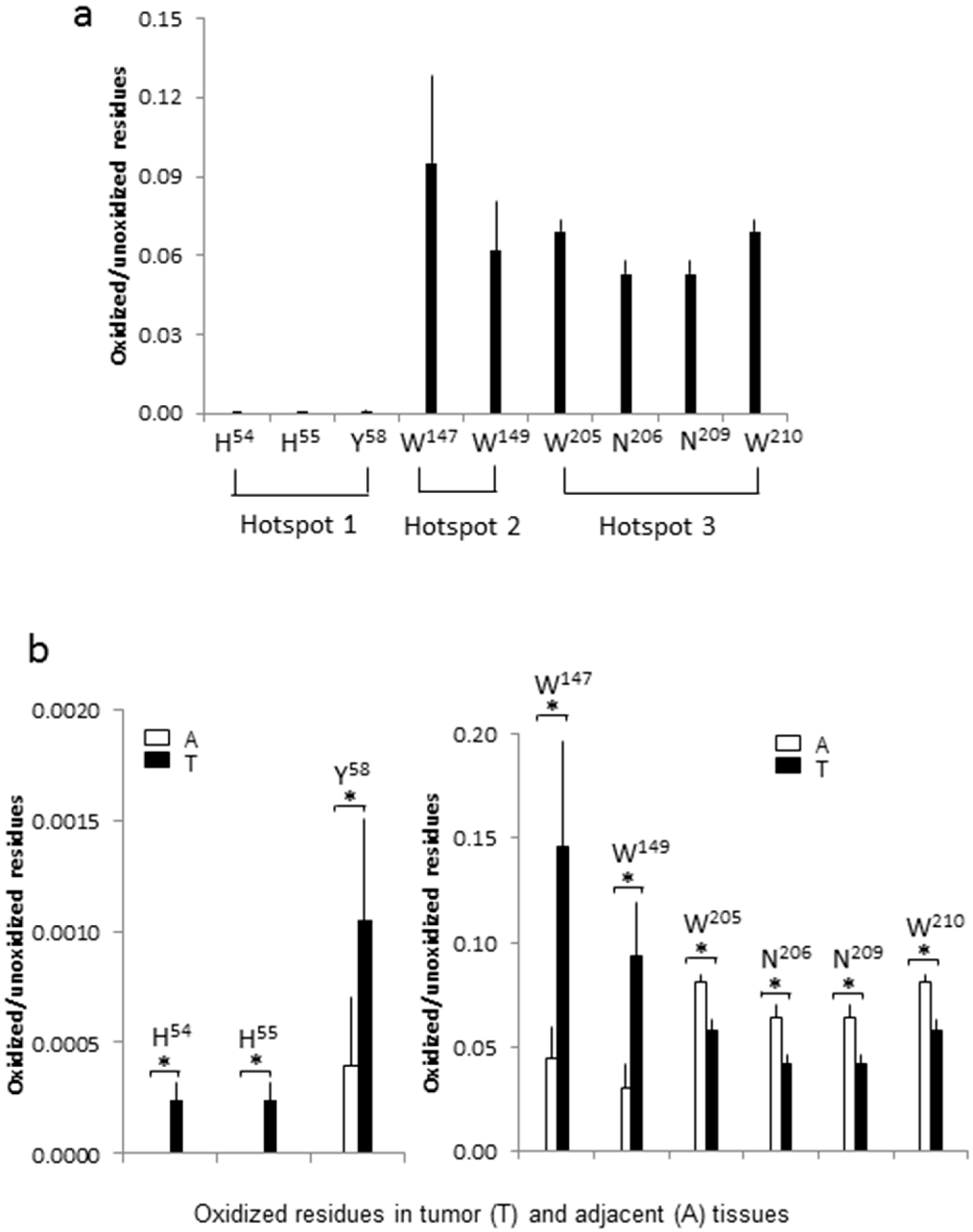
2.3. Tryptophan Oxidation at W147/W149 and W205/W210 of MNSOD
2.4. Asparagine Oxidation at N206 and N209 of MNSOD
2.5. Histidine Oxidation at H54 and H55 of MNSOD
2.6. Tyrosine Oxidation at Y58 of MNSOD
2.7. Oxidation at H54, H55, Y58, W147 and W149 of MNSOD Is Up-Regulated in ccRCC
2.8. The Relative MNSOD Enzymatic Activity Is Attenuated in ccRCC
3. Discussion
4. Materials and Methods
4.1. Sample Preparation and Protein Extraction
4.2. Liquid Chromatography Tandem Mass Spectrometry
4.3. Label-Free Quantification
4.4. Pathway and Network Analyses of Dysregulated Proteins in ccRCC
4.5. Western Blotting (WB) Analysis
4.6. MNSOD Enzymatic Activity Assay
4.7. Data Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dhar, S.K.; St. Clair, D.K. Manganese superoxide dismutase regulation and cancer. Free Radic. Biol. Med. 2012, 52, 2209–2222. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.J.; Shin, D.S.; Getzoff, E.D.; Tainer, J.A. The structural biochemistry of the superoxide dismutases. Biochim. Biophys. Acta 2010, 1804, 245–262. [Google Scholar] [CrossRef] [PubMed]
- Ozden, O.; Park, S.H.; Kim, H.S.; Jiang, H.; Coleman, M.C.; Spitz, D.R.; Gius, D. Acetylation of MNSOD directs enzymatic activity responding to cellular nutrient status or oxidative stress. Aging 2011, 3, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Holley, A.K.; Dhar, S.K.; Xu, Y.; St. Clair, D.K. Manganese superoxide dismutase: Beyond life and death. Amino Acids 2012, 42, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, E.R.; Levine, R.L. Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 2003, 25, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Nagy, P.; Lechte, T.P.; Das, A.B.; Winterbourn, C.C. Conjugation of glutathione to oxidized tyrosine residues in peptides and proteins. J. Biol. Chem. 2012, 287, 26068–26076. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, H.; Nair, J. Potential role of lipid peroxidation derived DNA damage in human colon carcinogenesis: Studies on exocyclic base adducts as stable oxidative stress markers. Cancer Detect. Prev. 2002, 26, 308–312. [Google Scholar] [CrossRef]
- Shacter, E. Quantification and significance of protein oxidation in biological samples. Drug Metab. Rev. 2000, 32, 307–326. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Yan, L.J. Protein oxidative modifications: Beneficial roles in disease and health. J. Biochem. Pharmacol. Res. 2013, 1, 15–26. [Google Scholar] [PubMed]
- Castro, L.; Demicheli, V.; Tortora, V.; Radi, R. Mitochondrial protein tyrosine nitration. Free Radic. Res. 2011, 45, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.J.; Hearn, A.S.; Cabelli, D.E.; Nick, H.S.; Tainer, J.A.; Silverman, D.N. Contribution of human manganese superoxide dismutase tyrosine 34 to structure and catalysis. Biochemistry 2009, 48, 3417–3424. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Wu, F.; Ding, S.; Sun, L.; Liu, Z.; Ding, K.; Lu, J. Label-free quantitative proteomic analysis reveals potential biomarkers and pathways in renal cell carcinoma. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2015, 36, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, J.; Lin, Y.; Lei, Q.; Guan, K.L.; Zhao, S.; Xiong, Y. Tumour suppressor SIRT3 deacetylates and activates manganese superoxide dismutase to scavenge ROS. EMBO Rep. 2011, 12, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Sosa, V.; Moline, T.; Somoza, R.; Paciucci, R.; Kondoh, H.; ME, L.L. Oxidative stress and cancer: An overview. Ageing Res. Rev. 2013, 12, 376–390. [Google Scholar] [CrossRef] [PubMed]
- Marshall, F.F. Differential protein profiling in renal cell carcinoma. J. Urol. 2005, 173, 1114. [Google Scholar] [CrossRef]
- Sverko, A.; Sobocanec, S.; Kusic, B.; Macak-Safranko, Z.; Saric, A.; Lenicek, T.; Kraus, O.; Andrisic, L.; Korolija, M.; Balog, T.; et al. Superoxide dismutase and cytochrome P450 isoenzymes might be associated with higher risk of renal cell carcinoma in male patients. Int. Immunopharmacol. 2011, 11, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Homma-Takeda, S.; Sasaki, A.; Kikushima, M.; Kumagai, Y.; Uchida, K.; Kawai, K.; Akaza, H.; Shimojo, N. Enzyme activity and protein content of superoxide dismutase isozymes in human renal cell carcinoma. Res. Commun. Mol. Pathol. Pharmacol. 2000, 108, 49–55. [Google Scholar] [PubMed]
- Zhang, Y.; Smith, B.J.; Oberley, L.W. Enzymatic activity is necessary for the tumor-suppressive effects of MNSOD. Antioxid. Redox Signal. 2006, 8, 1283–1293. [Google Scholar] [CrossRef] [PubMed]
- Weydert, C.J.; Waugh, T.A.; Ritchie, J.M.; Iyer, K.S.; Smith, J.L.; Li, L.; Spitz, D.R.; Oberley, L.W. Overexpression of manganese or copper-zinc superoxide dismutase inhibits breast cancer growth. Free Radic. Biol. Med. 2006, 41, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Na, S.; Bandeira, N.; Paek, E. Fast multi-blind modification search through tandem mass spectrometry. Mol. Cell. Proteom. MCP 2012. [Google Scholar] [CrossRef] [PubMed]
- John, J.P.; Pollak, A.; Lubec, G. Complete sequencing and oxidative modification of manganese superoxide dismutase in medulloblastoma cells. Electrophoresis 2009, 30, 3006–3016. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Erlandsen, H.; Haavik, J.; Knappskog, P.M.; Stevens, R.C. Three-dimensional structure of human tryptophan hydroxylase and its implications for the biosynthesis of the neurotransmitters serotonin and melatonin. Biochemistry 2002, 41, 12569–12574. [Google Scholar] [CrossRef] [PubMed]
- Stenflo, J.; Ohlin, A.K.; Owen, W.G.; Schneider, W.J. β-Hydroxyaspartic acid or β-hydroxyasparagine in bovine low density lipoprotein receptor and in bovine thrombomodulin. J. Biol. Chem. 1988, 263, 21–24. [Google Scholar] [PubMed]
- Uchida, K.; Kawakishi, S. 2-Oxo-histidine as a novel biological marker for oxidatively modified proteins. FEBS Lett. 1993, 332, 208–210. [Google Scholar] [CrossRef]
- Schulze, W.X.; Usadel, B. Quantitation in mass-spectrometry-based proteomics. Annu. Rev. Plant Biol. 2010, 61, 491–516. [Google Scholar] [CrossRef] [PubMed]
- El Refaey, M.; Watkins, C.P.; Kennedy, E.J.; Chang, A.; Zhong, Q.; Ding, K.H.; Shi, X.M.; Xu, J.; Bollag, W.B.; Hill, W.D.; et al. Oxidation of the aromatic amino acids tryptophan and tyrosine disrupts their anabolic effects on bone marrow mesenchymal stem cells. Mol. Cell. Endocrinol. 2015, 410, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Ge, W.; Chowdhury, R.; Claridge, T.D.; Kramer, H.B.; Schmierer, B.; McDonough, M.A.; Gong, L.; Kessler, B.M.; Ratcliffe, P.J.; et al. Asparagine and aspartate hydroxylation of the cytoskeletal ankyrin family is catalyzed by factor-inhibiting hypoxia-inducible factor. J. Biol. Chem. 2011, 286, 7648–7660. [Google Scholar] [CrossRef] [PubMed]
- Hearn, A.S.; Stroupe, M.E.; Cabelli, D.E.; Ramilo, C.A.; Luba, J.P.; Tainer, J.A.; Nick, H.S.; Silverman, D.N. Catalytic and structural effects of amino acid substitution at histidine 30 in human manganese superoxide dismutase: Insertion of valine Cγ into the substrate access channel. Biochemistry 2003, 42, 2781–2789. [Google Scholar] [CrossRef] [PubMed]
- Cabelli, D.E.; Guan, Y.; Leveque, V.; Hearn, A.S.; Tainer, J.A.; Nick, H.S.; Silverman, D.N. Role of tryptophan 161 in catalysis by human manganese superoxide dismutase. Biochemistry 1999, 38, 11686–11692. [Google Scholar] [CrossRef] [PubMed]
- Quint, P.; Reutzel, R.; Mikulski, R.; McKenna, R.; Silverman, D.N. Crystal structure of nitrated human manganese superoxide dismutase: Mechanism of inactivation. Free Radic. Biol. Med. 2006, 40, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Wagner, U.G.; Pattridge, K.A.; Ludwig, M.L.; Stallings, W.C.; Werber, M.M.; Oefner, C.; Frolow, F.; Sussman, J.L. Comparison of the crystal structures of genetically engineered human manganese superoxide dismutase and manganese superoxide dismutase from Thermus thermophilus: Differences in dimer–dimer interaction. Protein Sci. 1993, 2, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Yamakura, F.; Ikeda, K. Modification of tryptophan and tryptophan residues in proteins by reactive nitrogen species. Nitric Oxide Biol. Chem. 2006, 14, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Quint, P.S.; Domsic, J.F.; Cabelli, D.E.; McKenna, R.; Silverman, D.N. Role of a glutamate bridge spanning the dimeric interface of human manganese superoxide dismutase. Biochemistry 2008, 47, 4621–4628. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.P.; Loo, S.Y.; Shin, S.W.; Tan, T.Z.; Eng, C.B.; Singh, R.; Putti, T.C.; Ong, C.W.; Salto-Tellez, M.; Goh, B.C.; et al. Manganese superoxide dismutase is a promising target for enhancing chemosensitivity of basal-like breast carcinoma. Antioxid. Redox Signal. 2014, 20, 2326–2346. [Google Scholar] [CrossRef] [PubMed]
- Su, N.; Choi, H.P.; Wang, F.; Su, H.; Fei, Z.; Yang, J.H.; Azadzoi, K.M. Quantitative proteomic analysis of differentially expressed proteins and downstream signaling pathways in chronic bladder ischemia. J. Urol. 2016, 195, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Bartolome, S.; Navarro, P.; Martin-Maroto, F.; Lopez-Ferrer, D.; Ramos-Fernandez, A.; Villar, M.; Garcia-Ruiz, J.P.; Vazquez, J. Properties of average score distributions of SEQUEST: The probability ratio method. Mol. Cell. Proteom. MCP 2008, 7, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Liotta, L.A.; Petricoin, E.F. The spectra count label-free quantitation in cancer proteomics. Cancer Genom. Proteom. 2012, 9, 135–142. [Google Scholar]
- Pagel, O.; Loroch, S.; Sickmann, A.; Zahedi, R.P. Current strategies and findings in clinically relevant post-translational modification-specific proteomics. Expert Rev. Proteom. 2015, 12, 235–253. [Google Scholar] [CrossRef] [PubMed]
- Atrih, A.; Mudaliar, M.A.; Zakikhani, P.; Lamont, D.J.; Huang, J.T.; Bray, S.E.; Barton, G.; Fleming, S.; Nabi, G. Quantitative proteomics in resected renal cancer tissue for biomarker discovery and profiling. Br. J. Cancer 2014, 110, 1622–1633. [Google Scholar] [CrossRef] [PubMed]



| Amino Acid | A | C | D | E | F | G | H | I | K | L | M | N | P | Q | R | S | T | V | W | Y | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adjacent | 3 | 0 | 0 | 0 | 0 | 5 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 28 | 4 | 41 |
| Tumor | 4 | 0 | 0 | 0 | 0 | 22 | 14 | 0 | 0 | 0 | 0 | 12 | 0 | 0 | 0 | 0 | 0 | 2 | 53 | 20 | 127 |
| Total | 7 | 0 | 0 | 0 | 0 | 27 | 14 | 0 | 0 | 0 | 0 | 13 | 0 | 0 | 0 | 0 | 0 | 2 | 81 | 24 | 168 |
| m/z b | z | Peptide with Modification c | Amino Acid Modified | Ions Score (XCorr) d | Expectation Value |
|---|---|---|---|---|---|
| 877.93 | 2 | 54H(ox)HAAYVNNLNVTEEK68 | His54 | 80 (4.9) | 4.93 × 10−6 |
| 885.92 | 2 | 54H(ox)H(ox)AAYVNNLNVTEEK68 | His54, His55 | 44 (3.7) | 1.87 × 10−2 |
| 888.44 | 2 | 203AIW(ox)N(ox)VINWENVTER216 | Trp205, Asn206 e | 81 (3.8) | 4.05 × 10−6 |
| 888.44 | 2 | 203AIWNVIN(ox)W(ox)ENVTER216 | Asn209 e, Trp210 | 59 (3.9) | 5.83 × 10−4 |
| 1026.02 | 2 | 135LTAASVGVQGSGW(ox)GWLGFNK154 | Trp147 | 75 (5.4) | 1.61 × 10−5 |
| 1034.02 | 2 | 135LTAASVGVQGSGW(ox)GW(ox)LGFNK154 | Trp147, Trp149 | 68 (4.7) | 1.03 × 10−4 |
| 880.44 | 2 | 203AIW(ox)NVINWENVTER216 | Trp205 | 66 (4.7) | 9.59 × 10−5 |
| 880.44 | 2 | 203AIWNVINW(ox)ENVTER216 | Trp210 | 79 (5.0) | 5.04 × 10−6 |
| 877.93 | 2 | 54HHAAY(ox)VNNLNVTEEK68 | Tyr58 | 96 (5.6) | 1.24 × 10−7 |
| 892.42 | 2 | 54HHAAY(no)VNNLNVTEEK68 | Tyr58 | 27 (3.4) | 4.10 × 10−1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Z.; Azadzoi, K.M.; Choi, H.-P.; Jing, R.; Lu, X.; Li, C.; Wang, F.; Lu, J.; Yang, J.-H. LC-MS/MS Analysis Unravels Deep Oxidation of Manganese Superoxide Dismutase in Kidney Cancer. Int. J. Mol. Sci. 2017, 18, 319. https://doi.org/10.3390/ijms18020319
Zhao Z, Azadzoi KM, Choi H-P, Jing R, Lu X, Li C, Wang F, Lu J, Yang J-H. LC-MS/MS Analysis Unravels Deep Oxidation of Manganese Superoxide Dismutase in Kidney Cancer. International Journal of Molecular Sciences. 2017; 18(2):319. https://doi.org/10.3390/ijms18020319
Chicago/Turabian StyleZhao, Zuohui, Kazem M. Azadzoi, Han-Pil Choi, Ruirui Jing, Xin Lu, Cuiling Li, Fengqin Wang, Jiaju Lu, and Jing-Hua Yang. 2017. "LC-MS/MS Analysis Unravels Deep Oxidation of Manganese Superoxide Dismutase in Kidney Cancer" International Journal of Molecular Sciences 18, no. 2: 319. https://doi.org/10.3390/ijms18020319
APA StyleZhao, Z., Azadzoi, K. M., Choi, H.-P., Jing, R., Lu, X., Li, C., Wang, F., Lu, J., & Yang, J.-H. (2017). LC-MS/MS Analysis Unravels Deep Oxidation of Manganese Superoxide Dismutase in Kidney Cancer. International Journal of Molecular Sciences, 18(2), 319. https://doi.org/10.3390/ijms18020319







