Coinfection with Epstein–Barr Virus (EBV), Human Papilloma Virus (HPV) and Polyoma BK Virus (BKPyV) in Laryngeal, Oropharyngeal and Oral Cavity Cancer
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. DNA Extraction from Fresh Frozen Tumour Tissue; Detection of EBV DNA
4.3. HPV Detection and Genotyping
4.4. Detection of BKV
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fitzmaurice, C.; Allen, C.; Barber, R.M.; Barregard, L.; Bhutta, Z.A.; Brenner, H.; Dicker, D.J.; Chimed-Orchir, O.; Dandona, R.; Dandona, L.; et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2017, 3, 524–548. Available online: https://jamanetwork.com/journals/jamaoncology/fullarticle/2588797 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gatta, G.; Botta, L.; Sánchez, M.J.; Anderson, L.A.; Pierannunzio, D.; Licitra, L.; EUROCARE Working Group. Prognoses and improvement for head and neck cancers diagnosed in Europe in early 2000s: The EUROCARE-5 population-based study. Eur. J. Cancer 2015, 51, 2130–2143. Available online: http://www.ejcancer.com/article/S0959-804900749-2/fulltext (accessed on 7 November 2017). [CrossRef] [PubMed]
- Wojciechowska, U.; Olasem, P.; Czauderna, K.; Didkowska, J. Cancer in Poland in 2014; Ministerstwo Zdrowia: Warszawa, Poland, 2016; pp. 42–45.
- Fakhry, C.; Westra, W.H.; Li, S.; Cmelak, A.; Ridge, J.A.; Pinto, H.; Forastiere, A.; Gillison, M.L. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J. Natl. Cancer Inst. 2008, 100, 261–269. Available online: https://academic.oup.com/jnci/article-lookup/doi/10.1093/jnci/djn011 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gillison, M.L.; Koch, W.M.; Capone, R.B.; Spafford, M.; Westra, W.H.; Wu, L.; Zahurak, M.L.; Daniel, R.W.; Viglione, M.; Symer, D.E.; et al. Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J. Natl. Cancer Inst. 2000, 92, 709–720. Available online: https://academic.oup.com/jnci/article/92/9/709/2906131/Evidence-for-a-Causal-Association-Between-Human (accessed on 7 November 2017). [CrossRef] [PubMed]
- Hillbertz, N.S.; Hirsch, J.M.; Jalouli, J.; Jalouli, M.M.; Sand, L. Viral and molecular aspects of oral cancer. Anticancer Res. 2012, 32, 4201–4212. Available online: http://ar.iiarjournals.org/content/32/10/4201.long (accessed on 7 November 2017). [PubMed]
- Scully, C. Oral cancer aetiopathogenesis; past, present and future aspects. Medicina Oral Patologia Oral Y Cirugia Bucal 2011, 16, e306–e311. Available online: https://www.researchgate.net/publication/50868315_Oral_cancer_aetiopathogenesis_past_present_and_future_aspects (accessed on 7 November 2017). [CrossRef]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer: Lyon, France, 2007; pp. 222–230. ISBN 978-92-832-1290-4.
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. A Review of Human Carcinogens. Biological Agents; International Agency for Research on Cancer: Lyon, France, 2012; p. 255. ISBN 978-92-832-1319-2.
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Epstein-Barr virus and Kaposi’ Sarcoma Herpesvirus/Human Herpesvirus 8; International Agency for Research on Cancer: Lyon, France, 1997; pp. 47–262. ISBN 92832-12703.
- Acharya, S.; Ekalaksananan, T.; Vatanasapt, P.; Loyha, K.; Phusingha, P.; Promthet, S.; Kongyingyoes, B.; Pientong, C. Association of Epstein-Barr virus infection with oral squamous cell carcinoma in a case-control study. J. Oral Pathol. Med. 2015, 44, 252–257. Available online: http://onlinelibrary.wiley.com/doi/10.1111/jop.12231/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Jalouli, J.; Ibrahim, S.O.; Mehrotra, R.; Jalouli, M.M.; Sapkota, D.; Larson, P.A.; Hirsch, J.M. Prevalence of viral (HPV, EBV, HSV) infections in oral submucous fibrosis and oral cancer from India. Acta Oto-Laryngol. 2010, 130, 1306–1311. Available online: http://www.tandfonline.com/doi/full/10.3109/00016481003782041?needAccess=true (accessed on 7 November 2017). [CrossRef] [PubMed]
- Jalouli, J.; Jalouli, M.M.; Sapkota, D.; Ibrahim, S.O.; Larson, P.A.; Sand, L. Human papilloma virus, herpes simplex and Epstein-Barr virus in oral squamous cell carcinoma from eight different countries. Anticancer Res. 2012, 32, 571–580. Available online: http://ar.iiarjournals.org/content/32/2/571.long (accessed on 7 November 2017). [PubMed]
- Kis, A.; Feher, K.; Gall, T.; Tar, I.; Boda, R.; Toth, E.D.; Méhes, G.; Gergely, L.; Szarka, K. Epstein-Barr virus prevalence in oral squamous cell cancer and potentially malignant oral disorders in an eastern Hungarian population. Eur. J. Oral Sci. 2009, 117, 537–540. Available online: http://onlinelibrary.wiley.com/wol1/doi/10.1111/j.1600-0722.2009.00660.x/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Bennett, S.; Broekema, N.; Imperiale, M. BK polyomavirus: Emerging pathogen. Microbes Infect. 2012, 14, 672–683. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3568954/pdf/nihms-360495.pdf (accessed on 7 November 2017). [CrossRef] [PubMed]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Malaria and Some Polyomaviruses (SV40, BK, JC, and Merkel Cell Viruses); IARC Monographs: Lyon, France, 2014; Volume 104, pp. 215–251. ISBN 978-92-832-0142-7. [Google Scholar]
- Burger-Calderon, R.; Webster-Cyriaque, J. Human BK Polyomavirus—The Potential for Head and Neck Malignancy and Disease. Cancers 2015, 7, 1244–1270. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4586768/pdf/cancers-07-00835.pdf (accessed on 7 November 2017). [CrossRef] [PubMed]
- Neirynck, V.; Claes, K.; Naesens, M.; De Wever, L.; Pirenne, J.; Kuypers, D.; Vanrenterghem, Y.; van Poppel, H.; Kabanda, A.; Lerut, E. Renal cell carcinoma in the allograft: What is the role of Polyomavirus? Case Rep. Nephrol. Urol. 2012, 2, 125–134. Available online: https://www.karger.com/Article/Pdf/341917 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Schowalter, R.; Reinhold, W.; Buck, C. Entry tropism of BK and Merkel cell polyomaviruses in cell culture. PLoS ONE 2012, 7, e42181. Available online: http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0042181 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Raeesi, N.; Gheissari, A.; Akrami, M.; Moghim, S. Urinary BK virus excretion in children newly diagnosed with acute lymphoblastic leukemia. Int. J. Prev. Med. 2012, 6, 402–407. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3389437/ (accessed on 7 November 2017).
- Konietzny, R.; Fischer, R.; Ternette, N.; Wright, C.; Turney, B.; Chakera, A. Detection of BK virus in urine from renal transplant subjects my mass spectrometry. Clin. Proteomics 2012, 9, 4–13. Available online: https://clinicalproteomicsjournal.biomedcentral.com/articles/10.1186/1559-0275-9-4 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Tognon, M.; Corallini, A.; Martini, F.; Negrini, M.; Barbanti-Brodano, G. Oncogenic transformation by BK virus and association with human tumors. Oncogene 2003, 22, 5192–5200. Available online: http://www.nature.com/onc/journal/v22/n33/full/1206550a.html (accessed on 7 November 2017). [CrossRef] [PubMed]
- Jeffers, L.K.; Madden, V.; Webster-Cyriaque, J. BK virus has tropism for human salivary gland cells in vitro: Implications for transmission. Virology 2009, 394, 183–193. Available online: http://www.sciencedirect.com/science/article/pii/S0042682209004267?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Kenan, D.J.; Mieczkowski, P.A.; Latulippe, E.; Côté, I.; Singh, H.K.; Nickeleit, V. BK Polyomavirus Genomic Integration and Large T Antigen Expression: Evolving Paradigms in Human Oncogenesis. Am. J. Transplant. 2017, 17, 1674–1680. Available online: http://onlinelibrary.wiley.com/doi/10.1002/path.4584/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Dalianis, T.; Hirsch, H.H. Human polyomaviruses in disease and cancer. Virology 2013, 437, 63–72. Available online: http://www.sciencedirect.com/science/article/pii/S0042682213000044?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Polz, D.; Morshed, K.; Jarzyński, A.; Polz-Dacewicz, M. Prevalence of Polyoma BKVirus (BKPyV), Epstein-Barr Virus (EBV) and Human Papilloma Virus (HPV) in Oropharyngeal Cancer. Pol. J. Microbiol. 2015, 64, 323–328. Available online: http://www.pjm.microbiology.pl/archive/vol6442015323.pdf (accessed on 7 November 2017). [CrossRef]
- Vedham, V.; Divi, R.L.; Starks, V.L.; Verma, M. Multiple infections and cancer: Implications in epidemiology. Technol. Cancer Res. Treat. 2014, 13, 177–194. Available online: http://journals.sagepub.com/doi/abs/10.7785/tcrt.2012.500366 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Sand, L.; Wallström, M.; Hirsch, J.M. Smokeless tobacco, viruses and oral cancer. Oral Health Dent. Manag. 2014, 13, 372–378. Available online: https://www.omicsonline.org/open-access/smokeless-tobacco-viruses-and-oral-cancer-2247-2452.1000594.pdf (accessed on 7 November 2017). [PubMed]
- Metgud, R.; Astekar, M.; Verma, M.; Sharma, A. Role of viruses in oral squamous cell carcinoma. Oncol. Rev. 2012, 6, 164–170. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4419625/ (accessed on 7 November 2017). [CrossRef] [PubMed]
- Sathish, N.; Wang, X.; Yuan, Y. Human Papillomavirus (HPV)-associated oral cancers and treatment strategies. J. Dent. Res. 2014, 93, 29S–36S. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4107541/ (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gillison, M.L. Current topics in the epidemiology of oral cavity and oropharyngeal cancers. Head Neck 2007, 29, 779–792. Available online: http://onlinelibrary.wiley.com/wol1/doi/10.1002/hed.20573/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gillison, M.L.; Castellsagué, X.; Chaturvedi, A.; Goodman, M.T.; Snijders, P.; Tommasino, M.; Arbyn, M.; Franceschi, S. Eurogin Roadmap: Comparative epidemiology of HPV infection and associated cancers of the head and neck and cervix. Int. J. Cancer 2014, 134, 497–507. Available online: http://onlinelibrary.wiley.com/doi/10.1002/ijc.28201/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Syrjänen, K.; Syrjänen, S. Detection of human papillomavirus in sinonasal carcinoma: Systematic review and meta-analysis. Hum. Pathol. 2013, 44, 983–991. Available online: http://www.humanpathol.com/article/S0046-817700320-6/fulltext (accessed on 7 November 2017). [CrossRef] [PubMed]
- Syrjänen, K.J.; Syrjänen, S.M.; Lamberg, M.A.; Pyrhönen, S. Human papillomavirus (HPV) involvement in squamous cell lesions of the oral cavity. Proc. Finn. Dent. Soc. 1983, 79, 1–8. [Google Scholar] [PubMed]
- Anantharaman, D.; Gheit, T.; Waterboer, T.; Abedi-Ardekani, B.; Carreira, C.; McKay-Chopin, S.; Gaborieau, V.; Marron, M.; Lagiou, P.; Ahrens, W.; et al. Human Papillomavirus infections and upper aero-digestive tract cancers. The ARCAGE study. J. Natl. Cancer Int. 2013, 105, 536–545. Available online: https://academic.oup.com/jnci/article-lookup/doi/10.1093/jnci/djt053 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Fakhry, C.; D’Souza, G. Discussing the diagnosis of HPV-OSCC: Common questions and answers. Oral Oncol. 2013, 49, 863–871. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4264664/pdf/nihms647259.pdf (accessed on 7 November 2017). [CrossRef] [PubMed]
- Fakhry, C.; Psyrri, A.; Chaturvedi, A. HPV and head and neck cancers: State-of-the-science. Oral Oncol. 2014, 50, 353–355. Available online: http://www.sciencedirect.com/science/article/pii/S1368837514001006?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Benson, E.; Li, R.; Eisele, D.; Fakhry, C. The clinical impast of HPV tumor status upon head and neck squamous cell carcinomas. Oral Oncol. 2014, 50, 565–574. Available online: http://www.sciencedirect.com/science/article/pii/S136883751300691X?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Haukioja, A.; Asunta, M.; Söderling, E.; Syrjänen, S. Persistent oral human papillomavirus infection is associated with smoking and elevated salivary immunoglobulin G concentration. J. Clin. Virol. 2014, 61, 101–106. Available online: http://www.journalofclinicalvirology.com/article/S1386-653200243-1/fulltext (accessed on 7 November 2017). [CrossRef] [PubMed]
- Rautava, J.; Syrjänen, S. Human papillomavirus infections in the oral mucosa. J. Am. Dent. Assoc. 2011, 142, 905–914. Available online: http://jada.ada.org/article/S0002-817762065-1/fulltext (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gillison, M.L.; Zhang, Q.; Jordan, R.; Xiao, W.; Westra, W.H.; Trotti, A.; Spencer, S.; Harris, J.; Chung, C.H.; Ang, K.K. Tobacco smoking and increased risk of death and progression for patients with p16 posotive and p16-negative oropharyngeal cancer. J. Clin. Oncol. 2012, 30, 2102–2111. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3397696/ and http://ascopubs.org/doi/full/10.1200/JCO.2011.38.4099 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Zur Hausen, H. Biochemical approaches to detection of Epstein-Barr virus in human tumors. Cancer Res. 1976, 36, 678–680. Available online: https://pdfs.semanticscholar.org/5f61/2ed773454019a9eb509e542e82f4f3a2a93e.pdf (accessed on 7 November 2017). [PubMed]
- Jalouli, J.; Ibrahim, S.; Sapkota, D.; Jalouli, M.M.; Vasstrand, E.N.; Hirsch, J.M.; Larson, P.A. Presence of human papilloma virus, herpes simplex virus and Epstein-Barr virus DNA in oral biopsies from Sudanese patients with regard to toombak use. J Oral Pathol. Med. 2011, 19, 599–604. Available online: http://onlinelibrary.wiley.com/wol1/doi/10.1111/j.1600-0714.2010.00910.x/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Senyuta, N.; Yakovleva, L.; Goncharova, E.; Scherback, L.; Diduk, S.; Smirova, K.; Maksimovich, D.; Gurtsevitch, V. Epstein-Barr virus latent membrane protein 1 polymorphism in nasopharyngeal carcinoma and other oral cavity tumors in Russia. J. Med. Virol. 2013, 86, 290–300. Available online: http://onlinelibrary.wiley.com/wol1/doi/10.1002/jmv.23729/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gulley, M.L.; Tang, W. Laboratory Assays for Epstein-Barr Virus-Related Disease. J. Mol. Diagn. 2008, 10, 279–292. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2438195/ (accessed on 7 November 2017). [CrossRef] [PubMed]
- Al Moustafa, A.E.; Chen, D.; Ghabreau, L.; Akil, N. Association between human papillomavirus and Epstein-Barr virus infections in human oral carcinogenesis. Med. Hypotheses 2009, 73, 184–186. Available online: http://www.sciencedirect.com/science/article/pii/S0306987709001686?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Jiang, R.; Ekshyyan, O.; Moore-Medlin, T.; Rong, X.; Nathan, S.; Gu, X.; Abreo, F.; Rosenthal, E.L.; Shi, M.; Guidry, J.T.; et al. Association between human papilloma virus/Epstein-Barr virus coinfection and oral carcinogenesis. J. Oral Pathol. Med. 2015, 44, 28–36. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4286485/ and http://onlinelibrary.wiley.com/wol1/doi/10.1111/jop.12221/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Sand, L.; Jalouli, J. Viruses and oral cancer. Is there a link? Microbes Infect. 2014, 16, 371–378. Available online: http://www.sciencedirect.com/science/article/pii/S1286457914000239?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Deng, Z.; Uehara, T.; Maeda, H.; Hasegawa, M.; Matayoshi, S.; Kiyuna, A.; Agena, S.; Pan, X.; Zhang, C.; Yamashita, Y.; et al. Epstein-Barr virus and human papillomavirus infections and genotype distribution in head and neck cancers. PLoS ONE 2014, 9, e113702. Available online: http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0113702 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Ying, S.; Song-Ling, P.; Li-Fang, Y.; Xue, C.; Yong-Guang, T.; Ya, C. Co-infection of Epstein-Barr virus and human papillomavirus in human tumorigenesis. Chin. J. Cancer 2016, 35, 16. Available online: https://cjcjournal.biomedcentral.com/articles/10.1186/s40880-016-0079-1 (accessed on 7 November 2017). [CrossRef]
- Makielski, K.R.; Lee, D.; Lorenz, L.D.; Nawandar, D.M.; Chiu, Y.F.; Kenney, S.C.; Lambert, P.F. Human papillomavirus promotes Epstein–Barr virus maintenance and lytic reactivation in immortalized oral keratinocytes. Virology 2016, 495, 52–62. Available online: http://www.sciencedirect.com/science/article/pii/S0042682216301052?via%3Dihub and https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4912861/ (accessed on 7 November 2017). [CrossRef] [PubMed]
- Guidry, J.T.; Scott, T.S. The interaction between human papillmavirus and other viruses. Virus Res. 2017, 231, 130–147. Available online: http://www.sciencedirect.com/science/article/pii/S0168170216306633?via%3Dihub (accessed on 7 November 2017). [CrossRef] [PubMed]
- Hachana, M.; Amara, K.; Ziadi, S.; Gacem, R.B.; Korbi, S.; Trimeche, M. Investigation of human JC and BK polyomaviruses in breast carcinomas. Breast Cancer Res. Treat. 2012, 133, 969–977. Available online: https://link.springer.com/article/10.1007%2Fs10549-011-1876-5 (accessed on 7 November 2017). [CrossRef] [PubMed]
- Comar, M.; Bonifacio, D.; Zanconati, F.; Di Napoli, M.; Isidoro, E.; Martini, F.; Torelli, L.; Tognon, M. High prevalence of BK poliyomavirus sequences in Human papillomavirus-16-positive precancerous cervical lesions. J. Med. Virol. 2011, 83, 1770–1776. Available online: http://onlinelibrary.wiley.com/doi/10.1002/jmv.22184/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Burger-Calderon, R.; Madden, V.; Hallett, R.A.; Gingerich, A.D.; Nickeleit, V.; Webster-Cyriaque, J. Replication of oral BK virus in human salivary gland cells. J. Virol. 2014, 88, 559–573. Available online: http://jvi.asm.org/content/88/1/559.full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Moens, U.; van Ghule, M.; Ehlers, B. Are human polyomaviruses co-factors for cancers induced by other oncoviruses? Rev. Med. Virol. 2014, 24, 343–360. Available online: http://onlinelibrary.wiley.com/wol1/doi/10.1002/rmv.1798/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Fraase, K.; Hart, J.; Wu, H.; Pang, X.; Ma, L.; Grant, F.; Li, A.; Lennon, A.; Hu, P.C.; Dong, J. BK virus as a potential co-factor for HPV in the development of cervical neoplasia. Ann. Clin. Lab. Sci. 2012, 42, 130–134. Available online: http://www.annclinlabsci.org/content/42/2/130.long (accessed on 7 November 2017). [PubMed]
- Delbue, S.; Ferrante, P.; Provenzano, M. Polyomavirus BK and prostate cancer: An unworthy scientific effort? Oncoscience 2014, 1, 296–303. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4278296/ (accessed on 7 November 2017). [CrossRef] [PubMed]
- Gonzalez-Moles, M.; Gutiérrez, J.; Ruiz, I.; Fernández, J.A.; Rodriguez, M.; Aneiros, J. Epstein-Barr virus and oral squamous cell carcinoma in patients without HIV infection: Viral detection by polymerase chain reaction. Microbios 1998, 96, 23–31. [Google Scholar] [PubMed]
- Guidry, J.T.; Birdwell, C.E.; Scott, R.S. Epstein-Barr virus in the pathogenesis of oral cancers. Oral Dis. 2017. Available online: http://onlinelibrary.wiley.com/doi/10.1111/odi.12656/full (accessed on 7 November 2017). [CrossRef] [PubMed]
- Fathallah, I.; Parroche, P.; Gruffat, H.; Zannetti, C.; Johansson, H.; Yue, J.; Manet, E.; Tommasino, M.; Sylla, B.S.; Hasan, U.A. EBV latent membrane protein 1 is a negative regulator of TLR9. J. Immunol. 2010, 185, 6439–6447. Available online: http://www.jimmunol.org/content/185/11/6439.long (accessed on 7 November 2017). [CrossRef] [PubMed]
- Shahzad, N.; Shuda, M.; Gheit, T.; Kwun, H.J.; Cornet, I.; Saidj, D.; Zannetti, C.; Hasan, U.; Chang, Y.; Moore, P.S.; et al. The T Antigen Locus of Merkel Cell Polyomavirus Downregulates Human Toll-Like Receptor 9 Expression. J. Virol. 2013, 87, 13009–13019. Available online: http://jvi.asm.org/content/87/23/13009 (accessed on 30 November 2017). [CrossRef] [PubMed]
- Sobin, L.H.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours, 7th ed.; Wiley-Blackwell: Washington, DC, USA, 2009; pp. 22–45. ISBN 978-1-4443-3241-4. [Google Scholar]
- Cardesa, A.; Gale, N.; Nadal, A.; Zidor, N. Squamous cell carcinoma. In World Health Organization Classifiation of Tumours. Pathology and Genetics of Head and Neck Tumours; Barnes, L., Eveson, J.W., Reichart, P., Sidransky, D., Eds.; IARC Press: Lyon, France, 2005; pp. 118–121. ISBN 92-832-2417-5. [Google Scholar]
- Polz-Dacewicz, M.; Strycharz-Dudziak, M.; Dworzański, J.; Stec, A.; Kocot, J. Salivary and serum IL-10, TNF-α, TGF-β, VEGF levels in oropharyngeal squamous cell carcinoma and correlation with HPV and EBV infection. Infect. Agent. Cancer 2016, 11, 45–53. Available online: https://infectagentscancer.biomedcentral.com/articles/10.1186/s13027-017-0141-x (accessed on 7 November 2017). [CrossRef] [PubMed]
- Arthur, R.R.; Dagostin, S.; Shah, K.V. Detection of BK virus and JC virus in urine and brain tissue by the polymerase chain reaction. J. Clin. Microbiol. 1989, 27, 1174–1179. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC267522/pdf/jcm00066-0050.pdf (accessed on 7 November 2017). [PubMed]
| Total n = 146 | |||
|---|---|---|---|
| n | % | ||
| Sex | Female | 18 | 12.3 |
| Male | 128 | 87.7 | |
| Age | <50 | 27 | 18.5 |
| 50–69 | 92 | 63.0 | |
| ≥70 | 27 | 18.5 | |
| Place of residence | Urban | 95 | 65.1 |
| Rural | 51 | 34.9 | |
| Smoking | Yes | 103 | 70.6 |
| No | 43 | 29.4 | |
| Alcohol abuse | Yes | 87 | 59.6 |
| No | 59 | 40.4 | |
| Histological grading | G1 | 28 | 19.2 |
| G2 | 94 | 64.4 | |
| G3 | 24 | 16.4 | |
| T stage | T1 | 29 | 19.9 |
| T2 | 53 | 36.3 | |
| T3 | 27 | 18.5 | |
| T4 | 37 | 25.3 | |
| N stage | N1 | 67 | 45.8 |
| N2 | 33 | 22.6 | |
| N3 | 23 | 15.8 | |
| N4 | 23 | 15.8 | |
| M stage | M0 | 146 | 100.0 |
| M1 | 0 | 0 | |
| Location of cancer | Oropharynx | 53 | 36.3 |
| Larynx | 40 | 27.4 | |
| Oral cavity | 53 | 36.3 | |
| Single Infection | ||||
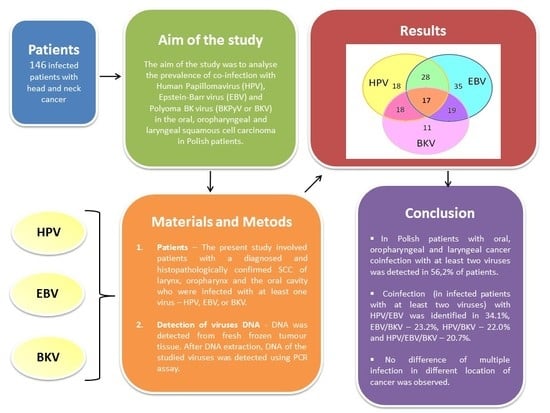
| HPV | EBV | BKV | 64 (43.8%) | |
| 18 (28.1%) | 35 (54.7%) | 11 (17.2) | ||
| Multiple Infection | ||||
| HPV + EBV | HPV + BKV | EBV + BKV | HPV + EBV + BKV | 82 (56.2%) |
| 28 (34.1%) | 18 (22%) | 19 (23.2%) | 17 (20.7%) | |
| Single Infection | |||||||
| Location of Cancer | HPV | EBV | BKV | Total | p | ||
| Oropharynx | 4 (22.2%) | 20 (57.1%) | 4 (36.4%) | 28 | 43.7% | 0.0102 * | |
| Larynx | 6 (33.3%) | 12 (34.3%) | 4 (36.4%) | 22 | 34.4% | ||
| Oral cavity | 8 (44.5%) | 3 (8.6%) | 3 (27.2%) | 14 | 21.9% | ||
| Total | 18 | 35 | 11 | 64 | 100% | ||
| Multiple Infection | |||||||
| Location of Cancer | HPV + EBV | HPV + BKV | EBV + BKV | HPV + EBV + BKV | Total | p | |
| Oropharynx | 8 (28.6%) | 8 (44.5%) | 6 (31.6%) | 3 (17.7%) | 25 | 30.5% | 0.7123 |
| Larynx | 6 (21.4%) | 4 (22.2%) | 4 (21.0%) | 4 (23.5%) | 18 | 21.9% | |
| Oral cavity | 14 (50%) | 6 (33.3%) | 9 (47.4%) | 10 (58.8%) | 39 | 47.6% | |
| Total | 28 | 18 | 19 | 17 | 82 | 100% | |
| HPV + EBV n = 28 | HPV + BKV n = 18 | EBV + BKV n = 19 | HPV + EBV + BKV n = 17 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | ||
| Sex | female | 2 | 7.1 | 2 | 11.1 | 2 | 10.5 | 1 | 5.9 |
| male | 26 | 92.9 | 16 | 88.9 | 17 | 89.5 | 16 | 94.1 | |
| p | 0.9030 | 0.9010 | 0.9402 | 0.6601 | |||||
| Age | <50 | 4 | 14.3 | 2 | 11.1 | 3 | 15.8 | 1 | 5.9 |
| 50–69 | 19 | 67.8 | 14 | 77.8 | 13 | 68.4 | 14 | 82.3 | |
| ≥70 | 5 | 17.9 | 2 | 11.1 | 3 | 15.8 | 2 | 11.8 | |
| p | 0.9211 | 0.6831 | 0.9880 | 0.5910 | |||||
| Place of residence | Urban | 23 | 82.1 | 15 | 83.3 | 16 | 84.2 | 16 | 94.1 |
| Rural | 5 | 17.9 | 3 | 16.7 | 3 | 15.8 | 1 | 5.9 | |
| p | 0.0400 * | 0.0640 | 0.0330 * | 0.0440 * | |||||
| Smoking | Yes | 22 | 78.6 | 13 | 72.2 | 15 | 78.9 | 13 | 76.5 |
| No | 6 | 21.4 | 5 | 27.8 | 4 | 21.1 | 4 | 23.5 | |
| p | 0.6010 | 0.7280 | 0.5691 | 0.7581 | |||||
| Alcohol abuse | Yes | 26 | 92.9 | 15 | 83.3 | 16 | 84.2 | 15 | 88.2 |
| No | 2 | 7.1 | 3 | 16.7 | 3 | 15.8 | 2 | 11.8 | |
| p | 0.0004 * | 0.0890 | 0.0420 * | 0.0800 | |||||
| Histological grading | G1–G2 | 12 | 42.8 | 5 | 27.8 | 7 | 36.8 | 3 | 17.7 |
| G3 | 16 | 57.2 | 13 | 72.2 | 12 | 63.2 | 14 | 82.3 | |
| p | 10−4 * | 10−4 * | 2 × 10−4 * | 10−4 * | |||||
| T stage | T1–T2 | 20 | 71.4 | 14 | 77.8 | 13 | 68.4 | 15 | 88.2 |
| T3–T4 | 8 | 28.6 | 4 | 22.2 | 6 | 31.6 | 2 | 11.8 | |
| p | 0.3310 | 0.4260 | 0.7180 | 0.0430 * | |||||
| N stage | N1–N2 | 22 | 78.6 | 16 | 88.9 | 16 | 84.2 | 17 | 100.0 |
| N3–N4 | 6 | 21.4 | 2 | 11.1 | 3 | 15.8 | 0 | 0 | |
| p | 0.0349 * | 0.0235 * | 0.0274 * | 0.0009 * | |||||
| OR (95% CI) | OR (95% CI) | OR (95% CI) | |
|---|---|---|---|
| Variable | HPV/HPV + EBV | HPV/HPV + BKV | HPV/HPV + EBV + BKV |
| G1–G2 | 0.8 (0.14–4.46) | 0.61 (0.18–2.06) | 0.4 (0.1–1.69) |
| G3 | 1.45 (0.54–3.87) | 2.46 (0.73–8.25) | 3.7 (0.88–15.27) |
| p | 0.7580 | 0.2250 | 0.1440 |
| T1–T2 | 0.91 (0.59–4.23) | 1.7 (0.48–6.13) | 2.46 (0.65–9.32) |
| T3–T4 | 1.57 (0.59–4.2) | 0.6 (0.12–3.1) | 0.44 (0.05–3.1) |
| p | 0.7800 | 0.7950 | 0.4320 |
| N1–N2 | 0.65 (0.21–2.12) | 2.86 (0.81–10.24) | 0.23 (0.03–1.93) |
| N3–N4 | 1.67 (0.03–2.25) | 0.80 (0.08–7.79) | 11.45 (1.37–95.6) |
| p | 0.4330 | 0.4120 | 0.0067 * |
| Variable | EBV/HPV + EBV | EBV/EBV + BKV | EBV/HPV + EBV + BKV |
| G1–G2 | 0.54 (0.11–2.73) | 0.40 (0.14–1.21) | 0.19 (0.05–0.78) |
| G3 | 4.14 (1.52–11.23) | 5.0 (1.61-15.54) | 10.5 (2.49–44.11) |
| p | 0.0160 * | 0.0080 * | 0.0010 * |
| T1–T2 | 1.12 (0.39–3.59) | 1.78 (0.52–5.98) | 3.04 (0.82–11.23) |
| T3–T4 | 0.61 (0.16–2.37) | 0.91 (0.23–3.68) | 0.31 (0.04–2.66) |
| p | 0.8700 | 0.8280 | 0.3060 |
| N1–N2 | 1.33 (0.52–3.42) | 1.92 (0.61–6.09) | 0.35 (0.04–2.96) |
| N3–N4 | 1.14 (0.27–4.84) | 0.5 (0.06–4.36) | 8.8 (1.07–72.34) |
| p | 0.7060 | 0.6930 | 0.0160 * |
| Variable | BKV/HPV + BKV | BKV/EBV + BKV | BKV/HPV + EBV + BKV |
| G1–G2 | 0.38 (0.11–1.39) | 0.53 (0.16–1.79) | 0.25 (0.06–1.13) |
| G3 | 3 (0.82–10.9) | 2.2 (0.64–7.19) | 4.5 (1.01–20.1) |
| p | 0.2160 | 0.3870 | 0.0427 * |
| T1–T2 | 2.5 (0.59–10.55) | 2.1 (0.5–8.6) | 1.43 (0.36–5.65) |
| T3–T4 | 0.36 (0.06–1.93) | 0.8 (0.18–2.17) | 3.5 (0.8–15.9) |
| p | 0.4300 | 0.6780 | 0.0457 * |
| N1–N2 | 3.59 (0.9–13.9) | 3.1 (0.88–11.61) | 0.3 (0.03–2.74) |
| N3–N4 | 0.28 (0.03–2.62) | 0.5 (0.09–2.99) | 14.4 (1.6–126.1) |
| p | 0.2920 | 0.8950 | 0.0204 * |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drop, B.; Strycharz-Dudziak, M.; Kliszczewska, E.; Polz-Dacewicz, M. Coinfection with Epstein–Barr Virus (EBV), Human Papilloma Virus (HPV) and Polyoma BK Virus (BKPyV) in Laryngeal, Oropharyngeal and Oral Cavity Cancer. Int. J. Mol. Sci. 2017, 18, 2752. https://doi.org/10.3390/ijms18122752
Drop B, Strycharz-Dudziak M, Kliszczewska E, Polz-Dacewicz M. Coinfection with Epstein–Barr Virus (EBV), Human Papilloma Virus (HPV) and Polyoma BK Virus (BKPyV) in Laryngeal, Oropharyngeal and Oral Cavity Cancer. International Journal of Molecular Sciences. 2017; 18(12):2752. https://doi.org/10.3390/ijms18122752
Chicago/Turabian StyleDrop, Bartłomiej, Małgorzata Strycharz-Dudziak, Ewa Kliszczewska, and Małgorzata Polz-Dacewicz. 2017. "Coinfection with Epstein–Barr Virus (EBV), Human Papilloma Virus (HPV) and Polyoma BK Virus (BKPyV) in Laryngeal, Oropharyngeal and Oral Cavity Cancer" International Journal of Molecular Sciences 18, no. 12: 2752. https://doi.org/10.3390/ijms18122752
APA StyleDrop, B., Strycharz-Dudziak, M., Kliszczewska, E., & Polz-Dacewicz, M. (2017). Coinfection with Epstein–Barr Virus (EBV), Human Papilloma Virus (HPV) and Polyoma BK Virus (BKPyV) in Laryngeal, Oropharyngeal and Oral Cavity Cancer. International Journal of Molecular Sciences, 18(12), 2752. https://doi.org/10.3390/ijms18122752