Galectins and Carcinogenesis: Their Role in Head and Neck Carcinomas and Thyroid Carcinomas
Abstract
1. Introduction
2. Galectins in Head and Neck Cancer and Thyroid Cancer
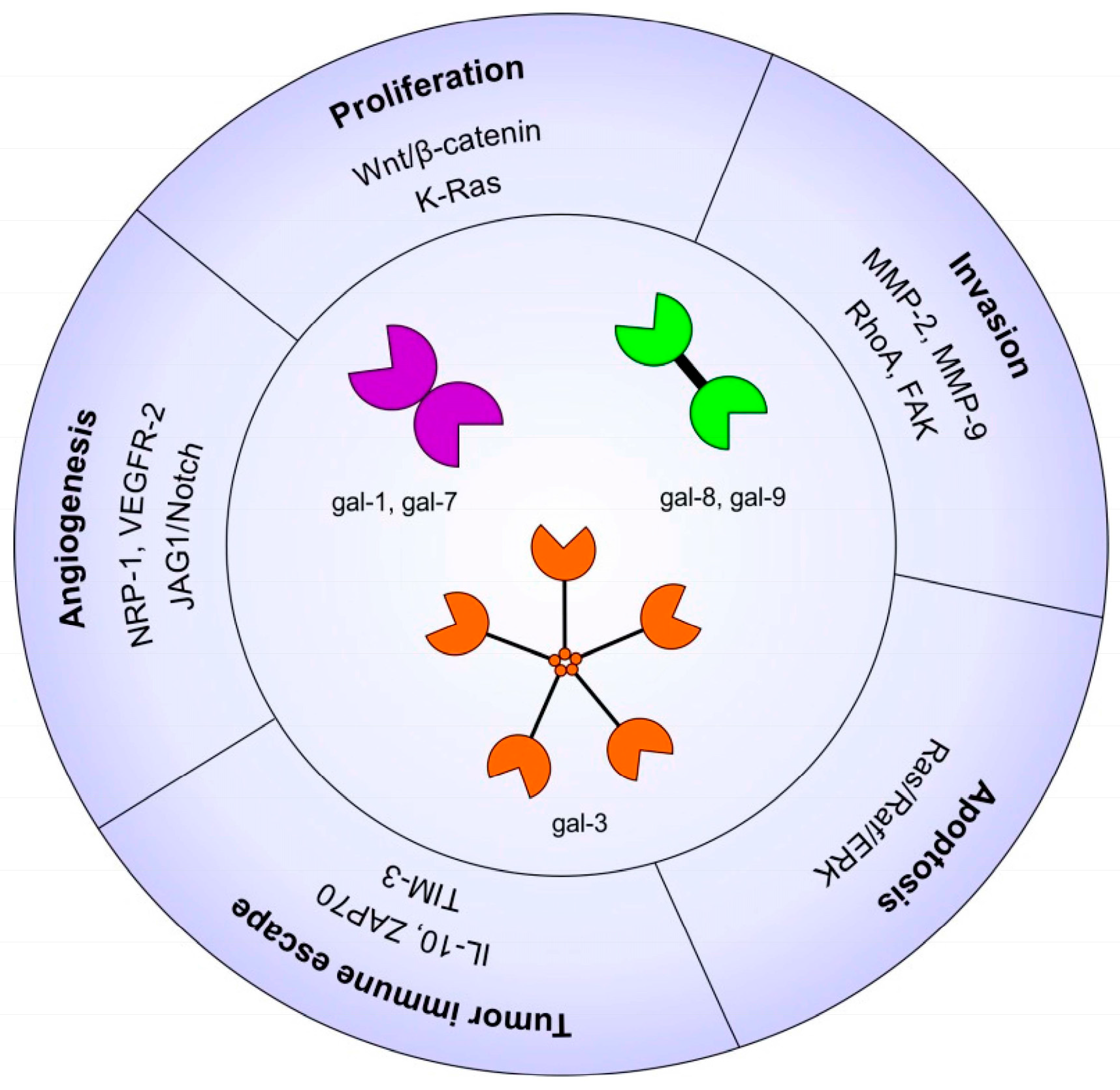
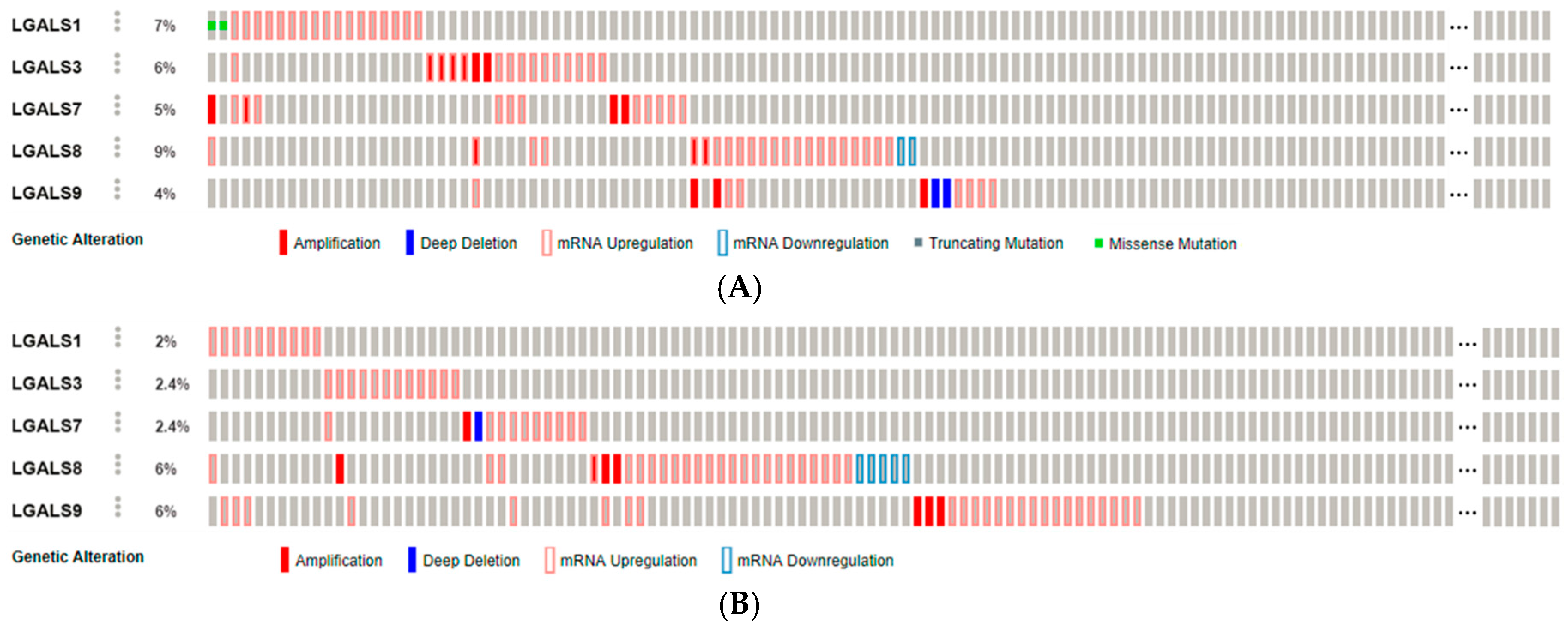
2.1. Galectins in Carcinogenesis
2.2. Galectins and Patient Prognosis
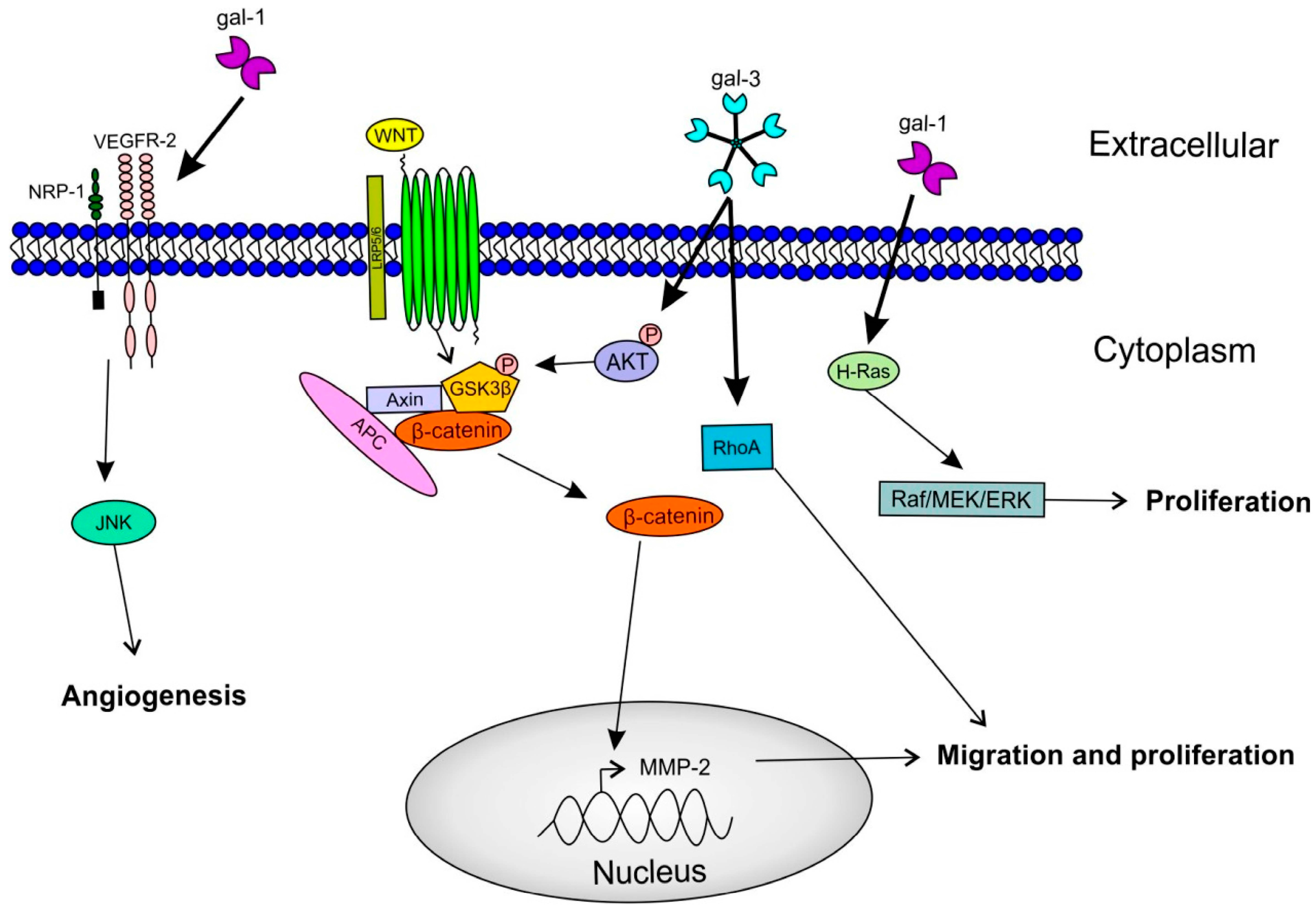
2.3. Galectins and Cell Proliferation/Survival
2.4. Galectins and Cell Migration/Invasion
2.5. Galectins and Angiogenesis
2.6. Galectins and Tumor Immune Escape
2.7. Clinical Potential of Galectin Inhibition
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bose, P.; Brockton, N.T.; Dort, J.C. Head and neck cancer: From anatomy to biology. Int. J. Cancer 2013, 133, 2013–2023. [Google Scholar] [CrossRef] [PubMed]
- Ang, K.K.; Harris, J.; Wheeler, R.; Weber, R.; Rosenthal, D.I.; Nguyen-Tân, P.F.; Westra, W.H.; Chung, C.H.; Jordan, R.C.; Lu, C.; et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 2010, 363, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Duray, A.; Descamps, G.; Decaestecker, C.; Remmelink, M.; Sirtaine, N.; Lechien, J.; Ernoux-Neufcoeur, P.; Bletard, N.; Somja, J.; Depuydt, C.E.; et al. Human papillomavirus DNA strongly correlates with a poorer prognosis in oral cavity carcinoma. Laryngoscope 2012, 122, 1558–1565. [Google Scholar] [CrossRef] [PubMed]
- Buckley, L.; Gupta, R.; Ashford, B.; Jabbour, J.; Clark, J.R. Oropharyngeal cancer and human papilloma virus: Evolving diagnostic and management paradigms. ANZ J. Surg. 2015. [Google Scholar] [CrossRef] [PubMed]
- Descamps, G.; Karaca, Y.; Lechien, J.R.; Kindt, N.; Decaestecker, C.; Remmelink, M.; Larsimont, D.; Andry, G.; Hassid, S.; Rodriguez, A.; et al. Classical risk factors, but not HPV status, predict survival after chemoradiotherapy in advanced head and neck cancer patients. J. Cancer Res. Clin. Oncol. 2016, 142, 2185–2196. [Google Scholar] [CrossRef] [PubMed]
- Sipos, J.A.; Mazzaferri, E.L. Thyroid cancer epidemiology and prognostic variables. Clin. Oncol. R. Coll. Radiol. 2010, 22, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Pazaitou-Panayiotou, K.; Polyzos, S.A.; Mantzoros, C.S. Obesity and thyroid cancer: Epidemiologic associations and underlying mechanisms. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2013, 14, 1006–1022. [Google Scholar] [CrossRef] [PubMed]
- Paz, A.; Haklai, R.; Elad-Sfadia, G.; Ballan, E.; Kloog, Y. Galectin-1 binds oncogenic H-Ras to mediate Ras membrane anchorage and cell transformation. Oncogene 2001, 20, 7486–7493. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.; Grafi-Cohen, M.; Kraiem, Z.; Kloog, Y. Galectin-3 promotes chronic activation of K-Ras and differentiation block in malignant thyroid carcinomas. Mol. Cancer Ther. 2010, 9, 2208–2219. [Google Scholar] [CrossRef] [PubMed]
- Funasaka, T.; Raz, A.; Nangia-Makker, P. Galectin-3 in angiogenesis and metastasis. Glycobiology 2014, 24, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Thijssen, V.L.; Heusschen, R.; Caers, J.; Griffioen, A.W. Galectin expression in cancer diagnosis and prognosis: A systematic review. Biochim. Biophys. Acta 2015, 1855, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.-T.; Patterson, R.J.; Wang, J.L. Intracellular functions of galectins. Biochim. Biophys. Acta 2002, 1572, 263–273. [Google Scholar] [CrossRef]
- Tang, W.; Huang, C.; Tang, C.; Xu, J.; Wang, H. Galectin-3 may serve as a potential marker for diagnosis and prognosis in papillary thyroid carcinoma: A meta-analysis. OncoTargets Ther. 2016, 9, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Arcolia, V.; Journe, F.; Wattier, A.; Leteurtre, E.; Renaud, F.; Gabius, H.-J.; Remmelink, M.; Decaestecker, C.; Rodriguez, A.; Boutry, S.; et al. Galectin-1 is a diagnostic marker involved in thyroid cancer progression. Int. J. Oncol. 2017, 51, 760–770. [Google Scholar] [CrossRef] [PubMed]
- cBioPortal for Cancer Genomics. Available online: http://www.cbioportal.org/ (accessed on 15 December 2017).
- Cancer Genome Atlas Network Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 2015, 517, 576–582. [CrossRef]
- Agrawal, N.; Akbani, R.; Aksoy, B.A.; Ally, A.; Arachchi, H.; Asa, S.L.; Auman, J.T.; Balasundaram, M.; Balu, S.; Baylin, S.B.; et al. Integrated Genomic Characterization of Papillary Thyroid Carcinoma. Cell 2014, 159, 676–690. [Google Scholar] [CrossRef] [PubMed]
- Landa, I.; Ibrahimpasic, T.; Boucai, L.; Sinha, R.; Knauf, J.A.; Shah, R.H.; Dogan, S.; Ricarte-Filho, J.C.; Krishnamoorthy, G.P.; Xu, B.; et al. Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J. Clin. Investig. 2016, 126, 1052–1066. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.C.; Sola Gallego, J.J.; Lotan, R.; El-Naggar, A.K. Differential expression of galectin-1 and galectin-3 in benign and malignant salivary gland neoplasms. Int. J. Oncol. 2000, 17, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Remmelink, M.; de Leval, L.; Decaestecker, C.; Duray, A.; Crompot, E.; Sirtaine, N.; André, S.; Kaltner, H.; Leroy, X.; Gabius, H.-J.; Saussez, S. Quantitative immunohistochemical fingerprinting of adhesion/growth-regulatory galectins in salivary gland tumours: Divergent profiles with diagnostic potential. Histopathology 2011, 58, 543–556. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.-M.; Dong, J.-H.; Chen, L.-L.; Zhang, H.-D. Increased expression of galectin-1 is associated with human oral squamous cell carcinoma development. Oncol. Rep. 2009, 21, 983–987. [Google Scholar] [PubMed]
- De Vasconcelos Carvalho, M.; Pereira, J.D.S.; Alves, P.M.; da Silveira, E.J.D.; de Souza, L.B.; Queiroz, L.M.G. Alterations in the immunoexpression of galectins-1, -3 and -7 between different grades of oral epithelial dysplasia. J. Oral Pathol. Med. 2013, 42, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Sharma, S.C.; Das, S.N. Galectin-1 and galectin-3: Plausible tumour markers for oral squamous cell carcinoma and suitable targets for screening high-risk population. Clin. Chim. Acta Int. J. Clin. Chem. 2015, 442, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-P.; Chen, S.-W.; Zhuang, S.-M.; Li, H.; Song, M. Galectin-3 accelerates the progression of oral tongue squamous cell carcinoma via a Wnt/β-catenin-dependent pathway. Pathol. Oncol. Res. 2013, 19, 461–474. [Google Scholar] [CrossRef] [PubMed]
- Saussez, S.; Decaestecker, C.; Lorfevre, F.; Chevalier, D.; Mortuaire, G.; Kaltner, H.; André, S.; Toubeau, G.; Gabius, H.-J.; Leroy, X. Increased expression and altered intracellular distribution of adhesion/growth-regulatory lectins galectins-1 and -7 during tumour progression in hypopharyngeal and laryngeal squamous cell carcinomas. Histopathology 2008, 52, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.-E.; Tan, T.; Li, C.; Chen, Z.-C.; Ruan, L.; Wang, H.-H.; Su, T.; Zhang, P.-F.; Xiao, Z.-Q. Identification of Galectin-1 as a novel biomarker in nasopharyngeal carcinoma by proteomic analysis. Oncol. Rep. 2010, 24, 495–500. [Google Scholar] [PubMed]
- Duray, A.; De Maesschalck, T.; Decaestecker, C.; Remmelink, M.; Chantrain, G.; Neiveyans, J.; Horoi, M.; Leroy, X.; Gabius, H.-J.; Saussez, S. Galectin fingerprinting in naso-sinusal diseases. Oncol. Rep. 2014, 32, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Bartolazzi, A.; Gasbarri, A.; Papotti, M.; Bussolati, G.; Lucante, T.; Khan, A.; Inohara, H.; Marandino, F.; Orlandi, F.; Nardi, F.; et al. Thyroid Cancer Study Group Application of an immunodiagnostic method for improving preoperative diagnosis of nodular thyroid lesions. Lancet 2001, 357, 1644–1650. [Google Scholar] [CrossRef]
- Bartolazzi, A.; Orlandi, F.; Saggiorato, E.; Volante, M.; Arecco, F.; Rossetto, R.; Palestini, N.; Ghigo, E.; Papotti, M.; Bussolati, G.; et al. Italian Thyroid Cancer Study Group (ITCSG) Galectin-3-expression analysis in the surgical selection of follicular thyroid nodules with indeterminate fine-needle aspiration cytology: A prospective multicentre study. Lancet Oncol. 2008, 9, 543–549. [Google Scholar] [CrossRef]
- Than, T.H.; Swethadri, G.K.; Wong, J.; Ahmad, T.; Jamil, D.; Maganlal, R.K.; Hamdi, M.M.; Abdullah, M.S. Expression of Galectin-3 and Galectin-7 in thyroid malignancy as potential diagnostic indicators. Singap. Med. J. 2008, 49, 333–338. [Google Scholar] [PubMed]
- Saussez, S.; Glinoer, D.; Chantrain, G.; Pattou, F.; Carnaille, B.; André, S.; Gabius, H.-J.; Laurent, G. Serum galectin-1 and galectin-3 levels in benign and malignant nodular thyroid disease. Thyroid Off. J. Am. Thyroid Assoc. 2008, 18, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Le, Q.-T.; Shi, G.; Cao, H.; Nelson, D.W.; Wang, Y.; Chen, E.Y.; Zhao, S.; Kong, C.; Richardson, D.; O’Byrne, K.J.; et al. Galectin-1: A link between tumor hypoxia and tumor immune privilege. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2005, 23, 8932–8941. [Google Scholar] [CrossRef] [PubMed]
- Saussez, S.; Decaestecker, C.; Lorfevre, F.; Cucu, D.-R.; Mortuaire, G.; Chevalier, D.; Wacreniez, A.; Kaltner, H.; André, S.; Toubeau, G.; Camby, I.; et al. High level of galectin-1 expression is a negative prognostic predictor of recurrence in laryngeal squamous cell carcinomas. Int. J. Oncol. 2007, 30, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-L.; Li, C.-F.; Lin, C.; Lin, Y.-S. Galectin-1 overexpression in nasopharyngeal carcinoma: Effect on survival. Acta Otolaryngol. (Stockh.) 2014, 134, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Noda, Y.; Kishino, M.; Sato, S.; Hirose, K.; Sakai, M.; Fukuda, Y.; Murakami, S.; Toyosawa, S. Galectin-1 expression is associated with tumour immunity and prognosis in gingival squamous cell carcinoma. J. Clin. Pathol. 2017, 70, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Honjo, Y.; Inohara, H.; Akahani, S.; Yoshii, T.; Takenaka, Y.; Yoshida, J.; Hattori, K.; Tomiyama, Y.; Raz, A.; Kubo, T. Expression of cytoplasmic galectin-3 as a prognostic marker in tongue carcinoma. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2000, 6, 4635–4640. [Google Scholar]
- Teymoortash, A.; Pientka, A.; Schrader, C.; Tiemann, M.; Werner, J.A. Expression of galectin-3 in adenoid cystic carcinoma of the head and neck and its relationship with distant metastasis. J. Cancer Res. Clin. Oncol. 2006, 132, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Saussez, S.; Lorfevre, F.; Lequeux, T.; Laurent, G.; Chantrain, G.; Vertongen, F.; Toubeau, G.; Decaestecker, C.; Kiss, R. The determination of the levels of circulating galectin-1 and -3 in HNSCC patients could be used to monitor tumor progression and/or responses to therapy. Oral Oncol. 2008, 44, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Saussez, S.; Cucu, D.-R.; Decaestecker, C.; Chevalier, D.; Kaltner, H.; André, S.; Wacreniez, A.; Toubeau, G.; Camby, I.; Gabius, H.-J.; Kiss, R. Galectin 7 (p53-induced gene 1): A new prognostic predictor of recurrence and survival in stage IV hypopharyngeal cancer. Ann. Surg. Oncol. 2006, 13, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Piantelli, M.; Iacobelli, S.; Almadori, G.; Iezzi, M.; Tinari, N.; Natoli, C.; Cadoni, G.; Lauriola, L.; Ranelletti, F.O. Lack of expression of galectin-3 is associated with a poor outcome in node-negative patients with laryngeal squamous-cell carcinoma. J. Clin. Oncol. 2002, 20, 3850–3856. [Google Scholar] [CrossRef] [PubMed]
- Plzák, J.; Betka, J.; Smetana, K.; Chovanec, M.; Kaltner, H.; André, S.; Kodet, R.; Gabius, H.-J. Galectin-3—An emerging prognostic indicator in advanced head and neck carcinoma. Eur. J. Cancer 2004, 40, 2324–2330. [Google Scholar] [CrossRef] [PubMed]
- Thyroid Cancer Survival Rates, by Type and Stage. Available online: https://www.cancer.org/cancer/thyroid-cancer/detection-diagnosis-staging/survival-rates.html (accessed on 27 October 2017).
- Kim, E.S.; Lim, D.J.; Lee, K.; Jung, C.K.; Bae, J.S.; Jung, S.L.; Baek, K.H.; Lee, J.M.; Moon, S.D.; Kang, M.I.; et al. Absence of galectin-3 immunostaining in fine-needle aspiration cytology specimens from papillary thyroid carcinoma is associated with favorable pathological indices. Thyroid Off. J. Am. Thyroid Assoc. 2012, 22, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Lee, J.B. Prognostic value of epidermal growth factor receptor, p53 and galectin-3 expression in papillary thyroid carcinoma. J. Int. Med. Res. 2013, 41, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Carpi, A.; Rossi, G.; Coscio, G.D.; Iervasi, G.; Nicolini, A.; Carpi, F.; Mechanick, J.I.; Bartolazzi, A. Galectin-3 detection on large-needle aspiration biopsy improves preoperative selection of thyroid nodules: A prospective cohort study. Ann. Med. 2010, 42, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Sumana, B.S.; Shashidhar, S.; Shivarudrappa, A.S. Galectin-3 Immunohistochemical Expression in Thyroid Neoplasms. J. Clin. Diagn. Res. 2015, 9, EC07-11. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandria, C.; Braesch-Andersen, S.; Bejo, K.; Reder, S.; Blechert, B.; Schwaiger, M.; Bartolazzi, A. Noninvasive In Vivo Imaging and Biologic Characterization of Thyroid Tumors by ImmunoPET Targeting of Galectin-3. Cancer Res. 2016, 76, 3583–3592. [Google Scholar] [CrossRef] [PubMed]
- Arcolia, V.; Journe, F.; Renaud, F.; Leteurtre, E.; Gabius, H.-J.; Remmelink, M.; Saussez, S. Combination of galectin-3, CK19 and HBME-1 immunostaining improves the diagnosis of thyroid cancer. Oncol. Lett. 2017, 14, 4183–4189. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.-T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Matarrese, P.; Tinari, N.; Semeraro, M.L.; Natoli, C.; Iacobelli, S.; Malorni, W. Galectin-3 overexpression protects from cell damage and death by influencing mitochondrial homeostasis. FEBS Lett. 2000, 473, 311–315. [Google Scholar] [CrossRef]
- Yoshii, T.; Fukumori, T.; Honjo, Y.; Inohara, H.; Kim, H.-R.C.; Raz, A. Galectin-3 phosphorylation is required for its anti-apoptotic function and cell cycle arrest. J. Biol. Chem. 2002, 277, 6852–6857. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Liang, N.; Xie, J.; Luo, H.; Zhang, J.; Deng, G.; Li, Y.; Zhang, J. Gene silencing of galectin-3 changes the biological behavior of Eca109 human esophageal cancer cells. Mol. Med. Rep. 2016, 13, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, C.; Sun, J.; Li, J.; Gu, X.; Xu, W. Antitumor effects of galectin-3 inhibition in human renal carcinoma cells. Exp. Biol. Med. 2016, 241, 1365–1373. [Google Scholar] [CrossRef] [PubMed]
- Harazono, Y.; Kho, D.H.; Balan, V.; Nakajima, K.; Zhang, T.; Hogan, V.; Raz, A. Galectin-3 leads to attenuation of apoptosis through Bax heterodimerization in human thyroid carcinoma cells. Oncotarget 2014, 5, 9992–10001. [Google Scholar] [CrossRef] [PubMed]
- Elad-Sfadia, G.; Haklai, R.; Balan, E.; Kloog, Y. Galectin-3 augments K-Ras activation and triggers a Ras signal that attenuates ERK but not phosphoinositide 3-kinase activity. J. Biol. Chem. 2004, 279, 34922–34930. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-I.; Whang, E.E.; Donner, D.B.; Jiang, X.; Price, B.D.; Carothers, A.M.; Delaine, T.; Leffler, H.; Nilsson, U.J.; Nose, V.; et al. Galectin-3 targeted therapy with a small molecule inhibitor activates apoptosis and enhances both chemosensitivity and radiosensitivity in papillary thyroid cancer. Mol. Cancer Res. 2009, 7, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Menachem, A.; Bodner, O.; Pastor, J.; Raz, A.; Kloog, Y. Inhibition of malignant thyroid carcinoma cell proliferation by Ras and galectin-3 inhibitors. Cell Death Discov. 2015, 1, 15047. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhang, P.; Shi, B.; Zhou, M.; Jiang, H.; Zhang, H.; Pan, X.; Gao, H.; Sun, H.; Li, Z. Galectin-1 overexpression promotes progression and chemoresistance to cisplatin in epithelial ovarian cancer. Cell Death Dis. 2014, 5, e991. [Google Scholar] [CrossRef] [PubMed]
- Xin, M.; Dong, X.-W.; Guo, X.-L. Role of the interaction between galectin-3 and cell adhesion molecules in cancer metastasis. Biomed. Pharmacother. 2015, 69, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Shimura, T.; Yajima, T.; Kubo, N.; Araki, K.; Tsutsumi, S.; Suzuki, H.; Kuwano, H.; Raz, A. Transient gene silencing of galectin-3 suppresses pancreatic cancer cell migration and invasion through degradation of β-catenin. Int. J. Cancer 2011, 129, 2775–2786. [Google Scholar] [CrossRef] [PubMed]
- Díaz, J.; Mendoza, P.; Silva, P.; Quest, A.F.; Torres, V.A. A novel caveolin-1/p85α/Rab5/Tiam1/Rac1 signaling axis in tumor cell migration and invasion. Commun. Integr. Biol. 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Shankar, J.; Wiseman, S.M.; Meng, F.; Kasaian, K.; Strugnell, S.; Mofid, A.; Gown, A.; Jones, S.J.M.; Nabi, I.R. Coordinated expression of galectin-3 and caveolin-1 in thyroid cancer. J. Pathol. 2012, 228, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.; Zou, R.; Chen, Z.; Fan, B.; Wang, Z.; Wang, Y.; Yin, X.; Zhang, D.; Tong, L.; Yang, F.; et al. Silencing RhoA inhibits migration and invasion through Wnt/β-catenin pathway and growth through cell cycle regulation in human tongue cancer. Acta Biochim. Biophys. Sin. 2014, 46, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wang, K.; Zhang, K.; Xu, F.; Yin, Y.; Zhu, L.; Zhou, F. Galectin-1 knockdown in carcinoma-associated fibroblasts inhibits migration and invasion of human MDA-MB-231 breast cancer cells by modulating MMP-9 expression. Acta Biochim. Biophys. Sin. 2016, 48, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-H.; Hong, H.-C.; Hong, T.-M.; Chiang, W.-F.; Jin, Y.-T.; Chen, Y.-L. Targeting galectin-1 in carcinoma-associated fibroblasts inhibits oral squamous cell carcinoma metastasis by downregulating MCP-1/CCL2 expression. Clin. Cancer Res. 2011, 17, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-H.; Hong, T.-M.; Cheng, H.-W.; Pan, S.-H.; Liang, Y.-R.; Hong, H.-C.; Chiang, W.-F.; Wong, T.-Y.; Shieh, D.-B.; Shiau, A.-L.; et al. Galectin-1-mediated tumor invasion and metastasis, up-regulated matrix metalloproteinase expression, and reorganized actin cytoskeletons. Mol. Cancer Res. 2009, 7, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Rizqiawan, A.; Tobiume, K.; Okui, G.; Yamamoto, K.; Shigeishi, H.; Ono, S.; Shimasue, H.; Takechi, M.; Higashikawa, K.; Kamata, N. Autocrine galectin-1 promotes collective cell migration of squamous cell carcinoma cells through up-regulation of distinct integrins. Biochem. Biophys. Res. Commun. 2013, 441, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Baum, L.G.; Seilhamer, J.J.; Pang, M.; Levine, W.B.; Beynon, D.; Berliner, J.A. Synthesis of an endogeneous lectin, galectin-1, by human endothelial cells is up-regulated by endothelial cell activation. Glycoconj. J. 1995, 12, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Thijssen, V.L.J.L.; Postel, R.; Brandwijk, R.J.M.G.E.; Dings, R.P.M.; Nesmelova, I.; Satijn, S.; Verhofstad, N.; Nakabeppu, Y.; Baum, L.G.; Bakkers, J.; et al. Galectin-1 is essential in tumor angiogenesis and is a target for antiangiogenesis therapy. Proc. Natl. Acad. Sci. USA 2006, 103, 15975–15980. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.H.; Ying, N.W.; Wu, M.H.; Chiang, W.F.; Hsu, C.L.; Wong, T.Y.; Jin, Y.T.; Hong, T.M.; Chen, Y.L. Galectin-1, a novel ligand of neuropilin-1, activates VEGFR-2 signaling and modulates the migration of vascular endothelial cells. Oncogene 2008, 27, 3746–3753. [Google Scholar] [CrossRef] [PubMed]
- D’Haene, N.; Sauvage, S.; Maris, C.; Adanja, I.; Le Mercier, M.; Decaestecker, C.; Baum, L.; Salmon, I. VEGFR1 and VEGFR2 involvement in extracellular galectin-1- and galectin-3-induced angiogenesis. PLoS ONE 2013, 8, e67029. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Duckworth, C.A.; Zhao, Q.; Pritchard, D.M.; Rhodes, J.M.; Yu, L.-G. Increased circulation of galectin-3 in cancer induces secretion of metastasis-promoting cytokines from blood vascular endothelium. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2013, 19, 1693–1704. [Google Scholar] [CrossRef] [PubMed]
- Markowska, A.I.; Liu, F.-T.; Panjwani, N. Galectin-3 is an important mediator of VEGF- and bFGF-mediated angiogenic response. J. Exp. Med. 2010, 207, 1981–1993. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, S.N.; Sheldon, H.; Pereira, J.X.; Paluch, C.; Bridges, E.M.; El-Cheikh, M.C.; Harris, A.L.; Bernardes, E.S. Galectin-3 acts as an angiogenic switch to induce tumor angiogenesis via Jagged-1/Notch activation. Oncotarget 2017, 8, 49484–49501. [Google Scholar] [CrossRef] [PubMed]
- Rabinovich, G.A.; Ramhorst, R.E.; Rubinstein, N.; Corigliano, A.; Daroqui, M.C.; Kier-Joffé, E.B.; Fainboim, L. Induction of allogenic T-cell hyporesponsiveness by galectin-1-mediated apoptotic and non-apoptotic mechanisms. Cell Death Differ. 2002, 9, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Camby, I.; Le Mercier, M.; Lefranc, F.; Kiss, R. Galectin-1: A small protein with major functions. Glycobiology 2006, 16, 137R–157R. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, N.; Alvarez, M.; Zwirner, N.W.; Toscano, M.A.; Ilarregui, J.M.; Bravo, A.; Mordoh, J.; Fainboim, L.; Podhajcer, O.L.; Rabinovich, G.A. Targeted inhibition of galectin-1 gene expression in tumor cells results in heightened T cell-mediated rejection; A potential mechanism of tumor-immune privilege. Cancer Cell 2004, 5, 241–251. [Google Scholar] [CrossRef]
- Kovács-Sólyom, F.; Blaskó, A.; Fajka-Boja, R.; Katona, R.L.; Végh, L.; Novák, J.; Szebeni, G.J.; Krenács, L.; Uher, F.; Tubak, V.; Kiss, R.; Monostori, E. Mechanism of tumor cell-induced T-cell apoptosis mediated by galectin-1. Immunol. Lett. 2010, 127, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Cedeno-Laurent, F.; Opperman, M.J.; Barthel, S.R.; Hays, D.; Schatton, T.; Zhan, Q.; He, X.; Matta, K.L.; Supko, J.G.; Frank, M.H.; et al. Metabolic inhibition of galectin-1-binding carbohydrates accentuates antitumor immunity. J. Investig. Dermatol. 2012, 132, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Fukumori, T.; Takenaka, Y.; Yoshii, T.; Kim, H.-R.C.; Hogan, V.; Inohara, H.; Kagawa, S.; Raz, A. CD29 and CD7 mediate galectin-3-induced type II T-cell apoptosis. Cancer Res. 2003, 63, 8302–8311. [Google Scholar] [PubMed]
- Peng, W.; Wang, H.Y.; Miyahara, Y.; Peng, G.; Wang, R.-F. Tumor-associated galectin-3 modulates the function of tumor-reactive T cells. Cancer Res. 2008, 68, 7228–7236. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Sutoh, M.; Hatakeyama, S.; Mori, K.; Yamamoto, H.; Koie, T.; Saitoh, H.; Yamaya, K.; Funyu, T.; Habuchi, T.; et al. MUC1 carrying core 2 O-glycans functions as a molecular shield against NK cell attack, promoting bladder tumor metastasis. Int. J. Oncol. 2012, 40, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Guo, H.; Geng, J.; Zheng, X.; Wei, H.; Sun, R.; Tian, Z. Tumor-released Galectin-3, a soluble inhibitory ligand of human NKp30, plays an important role in tumor escape from NK cell attack. J. Biol. Chem. 2014, 289, 33311–33319. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Anderson, A.C.; Schubart, A.; Xiong, H.; Imitola, J.; Khoury, S.J.; Zheng, X.X.; Strom, T.B.; Kuchroo, V.K. The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat. Immunol. 2005, 6, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Seki, M.; Oomizu, S.; Sakata, K.-M.; Sakata, A.; Arikawa, T.; Watanabe, K.; Ito, K.; Takeshita, K.; Niki, T.; Saita, N.; et al. Galectin-9 suppresses the generation of Th17, promotes the induction of regulatory T cells, and regulates experimental autoimmune arthritis. Clin. Immunol. 2008, 127, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Severson, J.J.; Serracino, H.S.; Mateescu, V.; Raeburn, C.D.; McIntyre, R.C.; Sams, S.B.; Haugen, B.R.; French, J.D. PD-1+Tim-3+ CD8+ T Lymphocytes Display Varied Degrees of Functional Exhaustion in Patients with Regionally Metastatic Differentiated Thyroid Cancer. Cancer Immunol. Res. 2015, 3, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Klyosov, A.; Zomer, E.; Platt, D. DAVANAT® (GM-CT-01) and Colon Cancer: Preclinical and Clinical (Phase I and II) Studies. In Glycobiology and Drug Design; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2012; Volume 1102, pp. 89–130. ISBN 978-0-8412-2765-1. [Google Scholar]
- Demotte, N.; Bigirimana, R.; Wieërs, G.; Stroobant, V.; Squifflet, J.-L.; Carrasco, J.; Thielemans, K.; Baurain, J.-F.; Van Der Smissen, P.; Courtoy, P.J.; van der Bruggen, P. A short treatment with galactomannan GM-CT-01 corrects the functions of freshly isolated human tumor-infiltrating lymphocytes. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2014, 20, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Redmond, W. Immunotherapy plus a galectin-3 inhibitor improves anti-tumor immunity: Insights from mice and a first-in-human phase I clinical trial. In Proceedings of the GTCbio 9th Immunotherapeutics & Immunomonitoring Conference, San Diego, CA, USA, 2 February 2017. [Google Scholar]
| Type of Tissues | Gal-1 | Gal-3 | Gal-7 | Gal-8 | Gal-9 | Ref. |
|---|---|---|---|---|---|---|
| Salivary gland carcinomas | *  | **  |  |  | [19,20] | |
| Oral cavity carcinoma |  |  | [21,22,23,24] | |||
| Laryngeal carcinoma |  |  | [25] | |||
| Hypopharyngeal carcinoma |  |  | [25] | |||
| Naso-pharyngeal carcinoma |  |  |  | [26,27] | ||
| Thyroid carcinomas |  |  | No change | [14,28,29,30,31] |
| Gal-1 | Gal-3 | Gal-9 | ||||
|---|---|---|---|---|---|---|
| HNSCC | TC | HNSCC | TC | HNSCC | TC | |
| Cell proliferation | [22] | [14] | [23] | [54,56] | ||
| Apoptosis | [53,55,56] | |||||
| Cell migration/invasion | [64,65] | [14] | [23,62] | [61] | ||
| Angiogenesis | [69] | |||||
| Tumor immune escape | [34] | [85] | ||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kindt, N.; Journe, F.; Ghanem, G.E.; Saussez, S. Galectins and Carcinogenesis: Their Role in Head and Neck Carcinomas and Thyroid Carcinomas. Int. J. Mol. Sci. 2017, 18, 2745. https://doi.org/10.3390/ijms18122745
Kindt N, Journe F, Ghanem GE, Saussez S. Galectins and Carcinogenesis: Their Role in Head and Neck Carcinomas and Thyroid Carcinomas. International Journal of Molecular Sciences. 2017; 18(12):2745. https://doi.org/10.3390/ijms18122745
Chicago/Turabian StyleKindt, Nadège, Fabrice Journe, Ghanem E. Ghanem, and Sven Saussez. 2017. "Galectins and Carcinogenesis: Their Role in Head and Neck Carcinomas and Thyroid Carcinomas" International Journal of Molecular Sciences 18, no. 12: 2745. https://doi.org/10.3390/ijms18122745
APA StyleKindt, N., Journe, F., Ghanem, G. E., & Saussez, S. (2017). Galectins and Carcinogenesis: Their Role in Head and Neck Carcinomas and Thyroid Carcinomas. International Journal of Molecular Sciences, 18(12), 2745. https://doi.org/10.3390/ijms18122745





