Galectin-7 in Epithelial Homeostasis and Carcinomas
Abstract
1. Introduction
1.1. Galectins
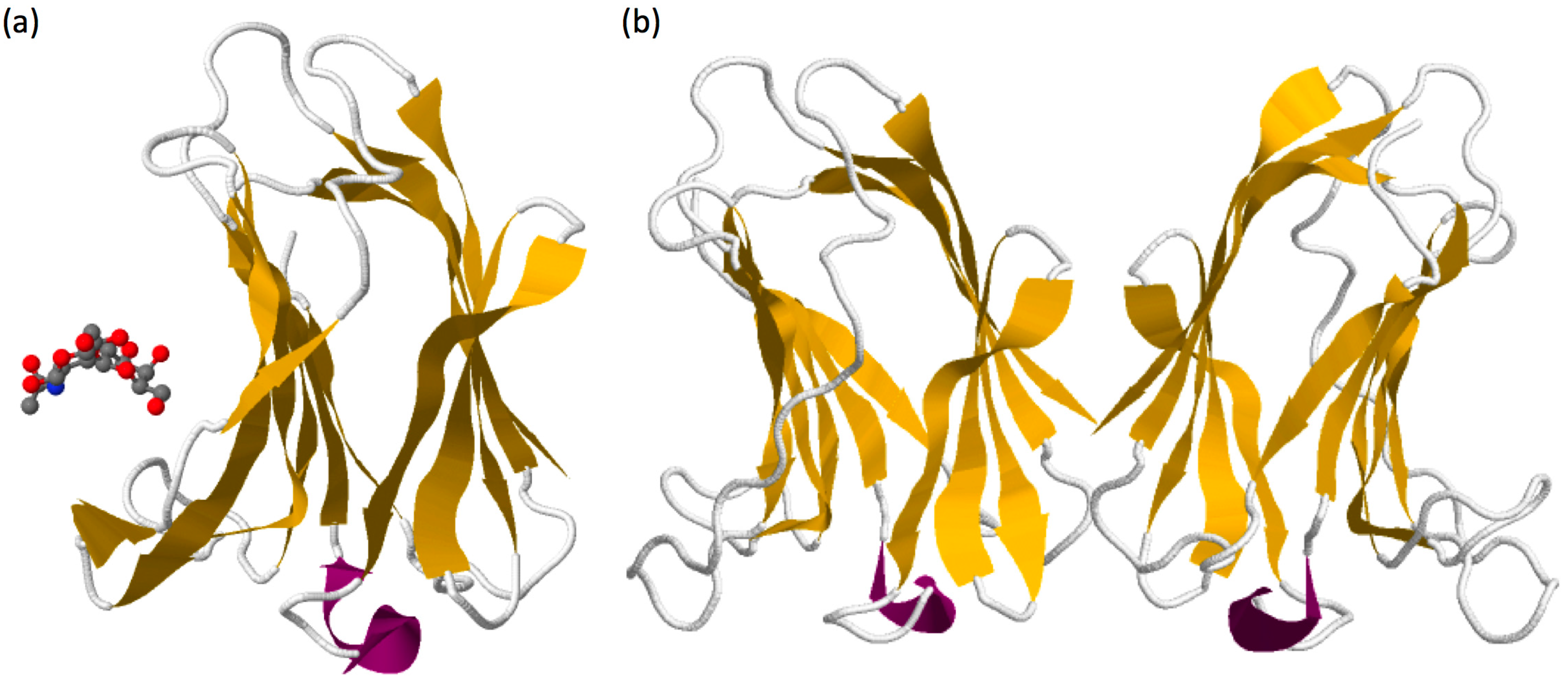
1.2. Galectin-7
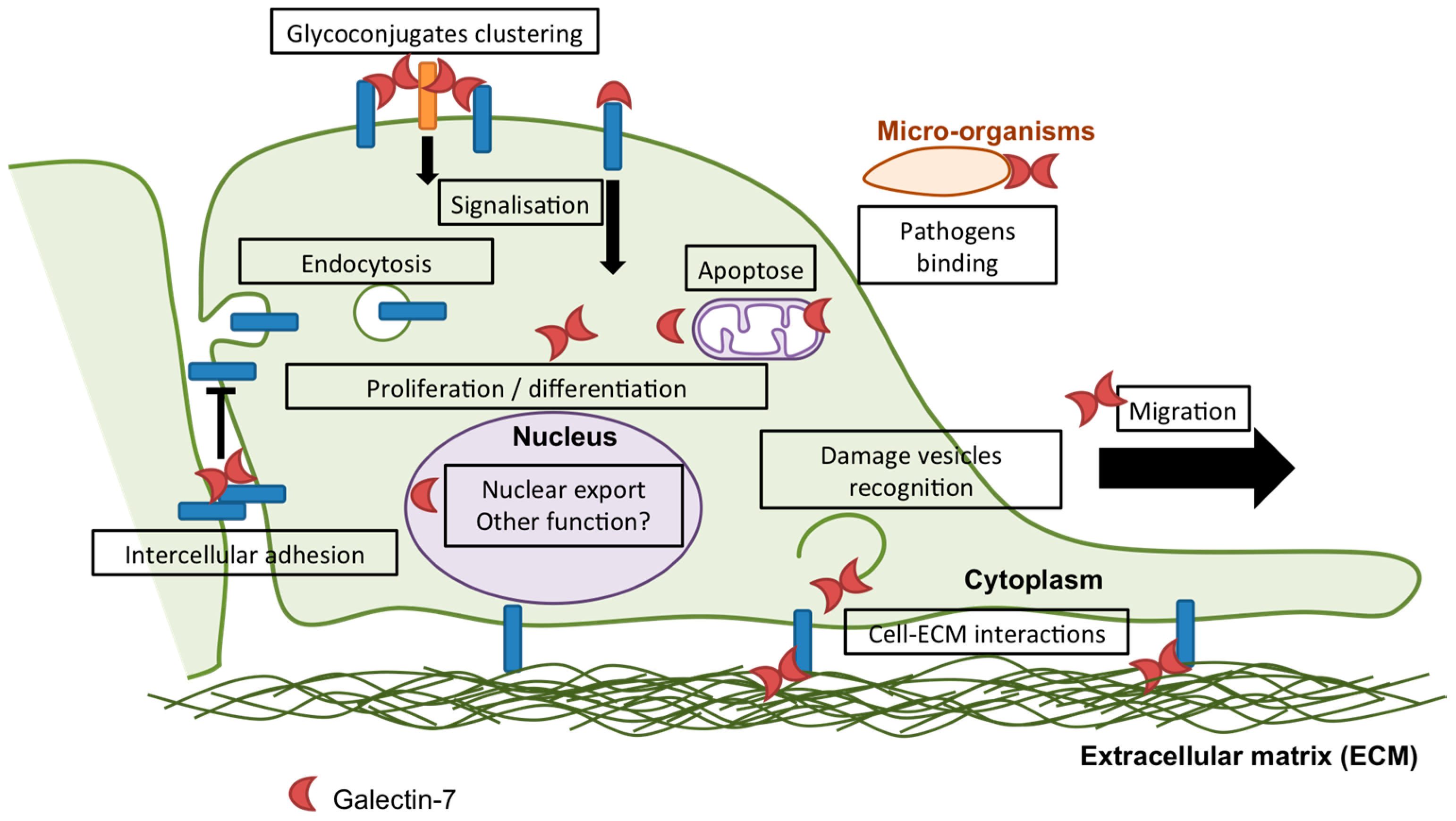
2. Galectin-7 in Epithelial Homeostasis
2.1. Apoptosis
2.2. Proliferation
2.3. Differentiation
3. Galectin-7 in Adhesion and Migration
3.1. Adhesion
3.2. Migration
4. Galectin-7 and Carcinomas
4.1.Galectin-7 as a Tumour Progression Marker
4.2. Galectin-7 as a Therapeutic Tool
5. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| BCR | B-Cell Receptor |
| CRD | Carbohydrate Recognition Domain |
| DMSO | Dimethyl Sulfoxide |
| FAF | F. magna Adhesion Factor |
| ECM | Extracellular Matrix |
| HGF | Hepatocyte Growth Factor |
| JNK | c-Jun N-terminal Kinase |
| LacNAc | N-Acetyl-Lactosamine |
| MMP | Matrix Metalloproteinase |
| MMTV-PyMT | Mouse Mammary Tumor Virus – Polyoma Middle T |
| NES | Nuclear Export Signal |
| NF-κB | Nuclear Factor kappa B |
| NLS | Nuclear Localisation Signal |
| OSCC | Oral Squamous Cell Carcinoma |
| SV-40 | Simian Virus-40 |
| TGFβ | Transforming Growth Factor β |
| UVB | Ultraviolet B |
| WT | Wild Type |
References
- Teichberg, V.I.; Silman, I.; Beitsch, D.D.; Resheff, G. A β-d-galactoside binding protein from electric organ tissue of Electrophorus electricus. Proc. Natl. Acad. Sci. USA 1975, 72, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- De Waard, A.; Hickman, S.; Kornfeld, S. Isolation and properties of β-galactoside binding lectins of calf heart and lung. J. Biol. Chem. 1976, 251, 7581–7587. [Google Scholar] [PubMed]
- Drickamer, K. Two distinct classes of carbohydrate-recognition domains in animal lectins. J. Biol. Chem. 1988, 263, 9557–9560. [Google Scholar] [PubMed]
- Barondes, S.H.; Cooper, D.N.; Gitt, M.A.; Leffler, H. Galectins. Structure and function of a large family of animal lectins. J. Biol. Chem. 1994, 269, 20807–20810. [Google Scholar] [PubMed]
- Than, N.G.; Romero, R.; Goodman, M.; Weckle, A.; Xing, J.; Dong, Z.; Xu, Y.; Tarquini, F.; Szilagyi, A.; Gal, P.; et al. A primate subfamily of galectins expressed at the maternal-fetal interface that promote immune cell death. Proc. Natl. Acad. Sci. USA 2009, 106, 9731–9736. [Google Scholar] [CrossRef] [PubMed]
- Than, N.G.; Romero, R.; Xu, Y.; Erez, O.; Xu, Z.; Bhatti, G.; Leavitt, R.; Chung, T.H.; El-Azzamy, H.; LaJeunesse, C.; et al. Evolutionary origins of the placental expression of chromosome 19 cluster galectins and their complex dysregulation in preeclampsia. Placenta 2014, 35, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Cummings, R.D.; Liu, F.-T.; Vasta, G.R. Galectins. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2015. [Google Scholar]
- Li, S.-Y.; Davidson, P.J.; Lin, N.Y.; Patterson, R.J.; Wang, J.L.; Arnoys, E.J. Transport of galectin-3 between the nucleus and cytoplasm. II. Identification of the signal for nuclear export. Glycobiology 2006, 16, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, S.; Hogan, V.; Inohara, H.; Raz, A. Importin-mediated nuclear translocation of galectin-3. J. Biol. Chem. 2006, 281, 39649–39659. [Google Scholar] [CrossRef] [PubMed]
- Delacour, D.; Koch, A.; Jacob, R. The role of galectins in protein trafficking. Traffic 2009, 10, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Viguier, M.; Advedissian, T.; Delacour, D.; Poirier, F.; Deshayes, F. Galectins in epithelial functions. Tissue Barriers 2014, 2, e29103. [Google Scholar] [CrossRef] [PubMed]
- Villeneuve, C.; Baricault, L.; Canelle, L.; Barboule, N.; Racca, C.; Monsarrat, B.; Magnaldo, T.; Larminat, F. Mitochondrial proteomic approach reveals galectin-7 as a novel BCL-2 binding protein in human cells. Mol. Biol. Cell 2011, 22, 999–1013. [Google Scholar] [CrossRef] [PubMed]
- Oka, T.; Murakami, S.; Arata, Y.; Hirabayashi, J.; Kasai, K.; Wada, Y.; Futai, M. Identification and cloning of rat galectin-2: Expression is predominantly in epithelial cells of the stomach. Arch. Biochem. Biophys. 1999, 361, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Magnaldo, T.; Fowlis, D.; Darmon, M. Galectin-7, a marker of all types of stratified epithelia. Differ. Res. Biol. Divers. 1998, 63, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Rabinovich, G.A. Galectins: An evolutionarily conserved family of animal lectins with multifunctional properties; a trip from the gene to clinical therapy. Cell Death Differ. 1999, 6, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Lobsanov, Y.D.; Gitt, M.A.; Leffler, H.; Barondes, S.H.; Rini, J.M. X-ray crystal structure of the human dimeric S-Lac lectin, L-14-II, in complex with lactose at 2.9-A resolution. J. Biol. Chem. 1993, 268, 27034–27038. [Google Scholar] [PubMed]
- Cooper, D.N.W. Galectinomics: Finding themes in complexity. Biochim. Biophys. Acta 2002, 1572, 209–231. [Google Scholar] [CrossRef]
- Leonidas, D.D.; Vatzaki, E.H.; Vorum, H.; Celis, J.E.; Madsen, P.; Acharya, K.R. Structural basis for the recognition of carbohydrates by human galectin-7. Biochemistry 1998, 37, 13930–13940. [Google Scholar] [CrossRef] [PubMed]
- Di Lella, S.; Sundblad, V.; Cerliani, J.P.; Guardia, C.M.; Estrin, D.A.; Vasta, G.R.; Rabinovich, G.A. When galectins recognize glycans: From biochemistry to physiology and back again. Biochemistry (Moscow) 2011, 50, 7842–7857. [Google Scholar] [CrossRef] [PubMed]
- Houzelstein, D.; Gonçalves, I.R.; Fadden, A.J.; Sidhu, S.S.; Cooper, D.N.W.; Drickamer, K.; Leffler, H.; Poirier, F. Phylogenetic analysis of the vertebrate galectin family. Mol. Biol. Evol. 2004, 21, 1177–1187. [Google Scholar] [CrossRef] [PubMed]
- Rabinovich, G.A.; Toscano, M.A. Turning “sweet” on immunity: Galectin-Glycan interactions in immune tolerance and inflammation. Nat. Rev. Immunol. 2009, 9, 338–352. [Google Scholar] [CrossRef] [PubMed]
- Hirabayashi, J.; Hashidate, T.; Arata, Y.; Nishi, N.; Nakamura, T.; Hirashima, M.; Urashima, T.; Oka, T.; Futai, M.; Muller, W.E.G.; et al. Oligosaccharide specificity of galectins: A search by frontal affinity chromatography. Biochim. Biophys. Acta 2002, 1572, 232–254. [Google Scholar] [CrossRef]
- Stowell, S.R.; Arthur, C.M.; Mehta, P.; Slanina, K.A.; Blixt, O.; Leffler, H.; Smith, D.F.; Cummings, R.D. Galectin-1, -2, and -3 exhibit differential recognition of sialylated glycans and blood group antigens. J. Biol. Chem. 2008, 283, 10109–10123. [Google Scholar] [CrossRef] [PubMed]
- Boscher, C.; Dennis, J.W.; Nabi, I.R. Glycosylation, galectins and cellular signaling. Curr. Opin. Cell Biol. 2011, 23, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Rabinovich, G.A.; Toscano, M.A.; Jackson, S.S.; Vasta, G.R. Functions of cell surface galectin-glycoprotein lattices. Curr. Opin. Struct. Biol. 2007, 17, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Nabi, I.R.; Shankar, J.; Dennis, J.W. The galectin lattice at a glance. J. Cell Sci. 2015, 128, 2213–2219. [Google Scholar] [CrossRef] [PubMed]
- Elantak, L.; Espeli, M.; Boned, A.; Bornet, O.; Bonzi, J.; Gauthier, L.; Feracci, M.; Roche, P.; Guerlesquin, F.; Schiff, C. Structural basis for galectin-1-dependent pre-B cell receptor (pre-BCR) activation. J. Biol. Chem. 2012, 287, 44703–44713. [Google Scholar] [CrossRef] [PubMed]
- Advedissian, T.; Proux-Gillardeaux, V.; Nkosi, R.; Peyret, G.; Nguyen, T.; Poirier, F.; Viguier, M.; Deshayes, F. E-cadherin dynamics is regulated by galectin-7 at epithelial cell surface. Sci. Rep. 2017, 7, 17086. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.-T.; Rabinovich, G.A. Galectins as modulators of tumour progression. Nat. Rev. Cancer 2005, 5, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.-J.; Lin, H.-Y.; Tu, Z.; Huang, B.-S.; Wu, S.-C.; Lin, C.-H. Structural Basis Underlying the Binding Preference of Human Galectins-1, -3 and -7 for Galβ1-3/4GlcNAc. PLoS ONE 2015, 10, e0125946. [Google Scholar] [CrossRef] [PubMed]
- Madsen, P.; Rasmussen, H.H.; Flint, T.; Gromov, P.; Kruse, T.A.; Honoré, B.; Vorum, H.; Celis, J.E. Cloning, expression, and chromosome mapping of human galectin-7. J. Biol. Chem. 1995, 270, 5823–5829. [Google Scholar] [CrossRef] [PubMed]
- Magnaldo, T.; Bernerd, F.; Darmon, M. Galectin-7, a human 14-kDa S-lectin, specifically expressed in keratinocytes and sensitive to retinoic acid. Dev. Biol. 1995, 168, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Nio-Kobayashi, J. Tissue- and cell-specific localization of galectins, β-galactose-binding animal lectins, and their potential functions in health and disease. Anat. Sci. Int. 2017, 92, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Saussez, S.; Kiss, R. Galectin-7. Cell. Mol. Life Sci. 2006, 63, 686–697. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.; Mackay, A.; Grigoriadis, A.; Cossu, A.; Reis-Filho, J.S.; Fulford, L.; Dexter, T.; Davies, S.; Bulmer, K.; Ford, E.; et al. Expression profiling of purified normal human luminal and myoepithelial breast cells: Identification of novel prognostic markers for breast cancer. Cancer Res. 2004, 64, 3037–3045. [Google Scholar] [CrossRef] [PubMed]
- Yaprak, E.; Kasap, M.; Akpınar, G.; Kayaaltı-Yüksek, S.; Sinanoğlu, A.; Guzel, N.; Demirturk Kocasarac, H. The prominent proteins expressed in healthy gingiva: A pilot exploratory tissue proteomics study. Odontology 2017. [Google Scholar] [CrossRef] [PubMed]
- Bernerd, F.; Sarasin, A.; Magnaldo, T. Galectin-7 overexpression is associated with the apoptotic process in UVB-induced sunburn keratinocytes. Proc. Natl. Acad. Sci. USA 1999, 96, 11329–11334. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-L.; Chiang, P.-C.; Lo, C.-H.; Lo, Y.-H.; Hsu, D.K.; Chen, H.-Y.; Liu, F.-T. Galectin-7 Regulates Keratinocyte Proliferation and Differentiation through JNK-miR-203-p63 Signaling. J. Investig. Dermatol. 2016, 136, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Campion, C.G.; Labrie, M.; Lavoie, G.; St-Pierre, Y. Expression of galectin-7 is induced in breast cancer cells by mutant p53. PLoS ONE 2013, 8, e72468. [Google Scholar] [CrossRef] [PubMed]
- Kaltner, H.; Raschta, A.-S.; Manning, J.C.; Gabius, H.-J. Copy-number variation of functional galectin genes: Studying animal galectin-7 (p53-induced gene 1 in man) and tandem-repeat-type galectins-4 and -9. Glycobiology 2013, 23, 1152–1163. [Google Scholar] [CrossRef] [PubMed]
- Vladoiu, M.C.; Labrie, M.; Létourneau, M.; Egesborg, P.; Gagné, D.; Billard, É.; Grosset, A.-A.; Doucet, N.; Chatenet, D.; St-Pierre, Y. Design of a peptidic inhibitor that targets the dimer interface of a prototypic galectin. Oncotarget 2015, 6, 40970–40980. [Google Scholar] [CrossRef] [PubMed]
- Grosset, A.-A.; Labrie, M.; Gagné, D.; Vladoiu, M.-C.; Gaboury, L.; Doucet, N.; St-Pierre, Y. Cytosolic galectin-7 impairs p53 functions and induces chemoresistance in breast cancer cells. BMC Cancer 2014, 14, 801. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, S.; Sleiman, M.H.; Masuyer, G.; Arbez-Gindre, C.; Micha-Screttas, M.; Calogeropoulou, T.; Steele, B.R.; Acharya, K.R. Structural basis of multivalent galactose-based dendrimer recognition by human galectin-7. FEBS J. 2015, 282, 372–387. [Google Scholar] [CrossRef] [PubMed]
- Ermakova, E.; Miller, M.C.; Nesmelova, I.V.; López-Merino, L.; Berbís, M.A.; Nesmelov, Y.; Tkachev, Y.V.; Lagartera, L.; Daragan, V.A.; André, S.; et al. Lactose binding to human galectin-7 (p53-induced gene 1) induces long-range effects through the protein resulting in increased dimer stability and evidence for positive cooperativity. Glycobiology 2013, 23, 508–523. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, Y.; Higashi, K.; Kushida, M.; Hong, Y.Y.; Nakao, S.; Higashiyama, R.; Moro, T.; Itoh, J.; Mikami, T.; Kimura, T.; et al. Hepatocyte growth factor suppresses profibrogenic signal transduction via nuclear export of Smad3 with galectin-7. Gastroenterology 2008, 134, 1180–1190. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.C.; Mörgelin, M.; Reinhardt, D.P.; Olin, A.I.; Björck, L.; Frick, I.-M. Identification of molecular mechanisms used by Finegoldia magna to penetrate and colonize human skin. Mol. Microbiol. 2014, 94, 403–417. [Google Scholar] [CrossRef] [PubMed]
- Gendronneau, G.; Sidhu, S.S.; Delacour, D.; Dang, T.; Calonne, C.; Houzelstein, D.; Magnaldo, T.; Poirier, F. Galectin-7 in the control of epidermal homeostasis after injury. Mol. Biol. Cell 2008, 19, 5541–5549. [Google Scholar] [CrossRef] [PubMed]
- Gendronneau, G.; Sanii, S.; Dang, T.; Deshayes, F.; Delacour, D.; Pichard, E.; Advedissian, T.; Sidhu, S.S.; Viguier, M.; Magnaldo, T.; et al. Overexpression of galectin-7 in mouse epidermis leads to loss of cell junctions and defective skin repair. PLoS ONE 2015, 10, e0119031. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Hiromasa, K.; Kabashima-Kubo, R.; Yoshioka, M.; Nakamura, M. Galectin-7, induced by cis-urocanic acid and ultraviolet B irradiation, down-modulates cytokine production by T lymphocytes. Exp. Dermatol. 2013, 22, 840–842. [Google Scholar] [CrossRef] [PubMed]
- Labrie, M.; Vladoiu, M.C.; Grosset, A.-A.; Gaboury, L.; St-Pierre, Y. Expression and functions of galectin-7 in ovarian cancer. Oncotarget 2014, 5, 7705–7721. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.-Y.; Rabinovich, G.A.; Liu, F.-T. Galectins: Structure, function and therapeutic potential. Expert Rev. Mol. Med. 2008, 10, e17. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.-P.; Li, X.-G. Galectin-7 promotes the invasiveness of human oral squamous cell carcinoma cells via activation of ERK and JNK signaling. Oncol. Lett. 2017, 13, 1919–1924. [Google Scholar] [CrossRef] [PubMed]
- Kopitz, J.; André, S.; von Reitzenstein, C.; Versluis, K.; Kaltner, H.; Pieters, R.J.; Wasano, K.; Kuwabara, I.; Liu, F.-T.; Cantz, M.; et al. Homodimeric galectin-7 (p53-induced gene 1) is a negative growth regulator for human neuroblastoma cells. Oncogene 2003, 22, 6277–6288. [Google Scholar] [CrossRef] [PubMed]
- Kuwabara, I.; Kuwabara, Y.; Yang, R.-Y.; Schuler, M.; Green, D.R.; Zuraw, B.L.; Hsu, D.K.; Liu, F.-T. Galectin-7 (PIG1) exhibits pro-apoptotic function through JNK activation and mitochondrial cytochrome c release. J. Biol. Chem. 2002, 277, 3487–3497. [Google Scholar] [CrossRef] [PubMed]
- Ueda, S.; Kuwabara, I.; Liu, F.-T. Suppression of tumor growth by galectin-7 gene transfer. Cancer Res. 2004, 64, 5672–5676. [Google Scholar] [CrossRef] [PubMed]
- Higareda-Almaraz, J.C.; Ruiz-Moreno, J.S.; Klimentova, J.; Barbieri, D.; Salvador-Gallego, R.; Ly, R.; Valtierra-Gutierrez, I.A.; Dinsart, C.; Rabinovich, G.A.; Stulik, J.; et al. Systems-level effects of ectopic galectin-7 reconstitution in cervical cancer and its microenvironment. BMC Cancer 2016, 16, 680. [Google Scholar] [CrossRef] [PubMed]
- Barkan, B.; Cox, A.D.; Kloog, Y. Ras inhibition boosts galectin-7 at the expense of galectin-1 to sensitize cells to apoptosis. Oncotarget 2013, 4, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Labrie, M.; Vladoiu, M.; Leclerc, B.G.; Grosset, A.-A.; Gaboury, L.; Stagg, J.; St-Pierre, Y. A Mutation in the Carbohydrate Recognition Domain Drives a Phenotypic Switch in the Role of Galectin-7 in Prostate Cancer. PLoS ONE 2015, 10, e0131307. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Wu, T.-C.; Chen, W.-Q.; Zhou, L.-J.; Wu, Y.; Zeng, L.; Pei, H.-P. Roles of galectin-7 and S100A9 in cervical squamous carcinoma: Clinicopathological and in vitro evidence. Int. J. Cancer 2013, 132, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Biron-Pain, K.; Grosset, A.-A.; Poirier, F.; Gaboury, L.; St-Pierre, Y. Expression and functions of galectin-7 in human and murine melanomas. PLoS ONE 2013, 8, e63307. [Google Scholar] [CrossRef] [PubMed]
- Polyak, K.; Xia, Y.; Zweier, J.L.; Kinzler, K.W.; Vogelstein, B. A model for p53-induced apoptosis. Nature 1997, 389, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Sarafian, V.; Jans, R.; Poumay, Y. Expression of lysosome-associated membrane protein 1 (Lamp-1) and galectins in human keratinocytes is regulated by differentiation. Arch. Dermatol. Res. 2006, 298, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, M.; Rasmussen, H.H.; Nielsen, H.V.; Vorum, H.; Orntoft, T.F.; Wolf, H.; Celis, J.E. Proteome profiling of bladder squamous cell carcinomas: Identification of markers that define their degree of differentiation. Cancer Res. 1997, 57, 4111–4117. [Google Scholar] [PubMed]
- Jiang, Y.; Tian, R.; Yu, S.; Zhao, Y.I.; Chen, Y.; Li, H.; Qiao, Y.; Wu, X. Clinical significance of galectin-7 in vulvar squamous cell carcinoma. Oncol. Lett. 2015, 10, 3826–3831. [Google Scholar] [CrossRef] [PubMed]
- Truong, A.B.; Khavari, P.A. Control of keratinocyte proliferation and differentiation by p63. Cell Cycle 2007, 6, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Elola, M.T.; Wolfenstein-Todel, C.; Troncoso, M.F.; Vasta, G.R.; Rabinovich, G.A. Galectins: Matricellular glycan-binding proteins linking cell adhesion, migration, and survival. Cell. Mol. Life Sci. 2007, 64, 1679–1700. [Google Scholar] [CrossRef] [PubMed]
- Menkhorst, E.M.; Gamage, T.; Cuman, C.; Kaitu’u-Lino, T.J.; Tong, S.; Dimitriadis, E. Galectin-7 acts as an adhesion molecule during implantation and increased expression is associated with miscarriage. Placenta 2014, 35, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Rondanino, C.; Poland, P.A.; Kinlough, C.L.; Li, H.; Rbaibi, Y.; Myerburg, M.M.; Al-bataineh, M.M.; Kashlan, O.B.; Pastor-Soler, N.M.; Hallows, K.R.; et al. Galectin-7 modulates the length of the primary cilia and wound repair in polarized kidney epithelial cells. Am. J. Physiol. Ren. Physiol. 2011, 301, F622–F633. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.; Yap, J.; Gamage, T.; Salamonsen, L.; Dimitriadis, E.; Menkhorst, E. Galectin-7 is important for normal uterine repair following menstruation. Mol. Hum. Reprod. 2014, 20, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Demers, M.; Magnaldo, T.; St-Pierre, Y. A novel function for galectin-7: Promoting tumorigenesis by up-regulating MMP-9 gene expression. Cancer Res. 2005, 65, 5205–5210. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Chang, W.Y.; Cho, M. Induction of matrix metalloproteinase-9 by galectin-7 through p38 MAPK signaling in HeLa human cervical epithelial adenocarcinoma cells. Oncol. Rep. 2009, 22, 1373–1379. [Google Scholar] [PubMed]
- Saussez, S.; Cludts, S.; Capouillez, A.; Mortuaire, G.; Smetana, K.; Kaltner, H.; André, S.; Leroy, X.; Gabius, H.-J.; Decaestecker, C. Identification of matrix metalloproteinase-9 as an independent prognostic marker in laryngeal and hypopharyngeal cancer with opposite correlations to adhesion/growth-regulatory galectins-1 and -7. Int. J. Oncol. 2009, 34, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Said, N.; Amin, S.; Wu, H.K.; Bruce, A.; Garate, M.; Hsu, D.K.; Kuwabara, I.; Liu, F.-T.; Panjwani, N. Galectins-3 and -7, but not galectin-1, play a role in re-epithelialization of wounds. J. Biol. Chem. 2002, 277, 42299–42305. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Said, N.; Wu, H.K.; Kuwabara, I.; Liu, F.-T.; Panjwani, N. Galectin-7 as a potential mediator of corneal epithelial cell migration. Arch. Ophthalmol. 2003, 121, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Wu, H.K.; Bruce, A.; Wollenberg, K.; Panjwani, N. Detection of differentially expressed genes in healing mouse corneas, using cDNA microarrays. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2897–2904. [Google Scholar]
- Klíma, J.; Lacina, L.; Dvoránková, B.; Herrmann, D.; Carnwath, J.W.; Niemann, H.; Kaltner, H.; André, S.; Motlík, J.; Gabius, H.-J.; et al. Differential regulation of galectin expression/reactivity during wound healing in porcine skin and in cultures of epidermal cells with functional impact on migration. Physiol. Res. 2009, 58, 873–884. [Google Scholar] [PubMed]
- Friedl, P.; Gilmour, D. Collective cell migration in morphogenesis, regeneration and cancer. Nat. Rev. Mol. Cell Biol. 2009, 10, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, J.J.; Rajasekaran, A.K. Reassessing epithelial to mesenchymal transition as a prerequisite for carcinoma invasion and metastasis. Cancer Res. 2006, 66, 8319–8326. [Google Scholar] [CrossRef] [PubMed]
- Vladoiu, M.C.; Labrie, M.; St-Pierre, Y. Intracellular galectins in cancer cells: Potential new targets for therapy (Review). Int. J. Oncol. 2014, 44, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Thijssen, V.L.; Heusschen, R.; Caers, J.; Griffioen, A.W. Galectin expression in cancer diagnosis and prognosis: A systematic review. Biochim. Biophys. Acta 2015, 1855, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Satelli, A.; Rao, P.S.; Gupta, P.K.; Lockman, P.R.; Srivenugopal, K.S.; Rao, U.S. Varied expression and localization of multiple galectins in different cancer cell lines. Oncol. Rep. 2008, 19, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Balan, V.; Nangia-Makker, P.; Raz, A. Galectins as cancer biomarkers. Cancers 2010, 2, 592–610. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, Y.; Campion, C.G.; Grosset, A.-A. A distinctive role for galectin-7 in cancer ? Front. Biosci. 2012, 17, 438–450. [Google Scholar] [CrossRef]
- Kaur, M.; Kaur, T.; Kamboj, S.S.; Singh, J. Roles of Galectin-7 in Cancer. Asian Pac. J. Cancer Prev. 2016, 17, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Moisan, S.; Demers, M.; Mercier, J.; Magnaldo, T.; Potworowski, E.F.; St-Pierre, Y. Upregulation of galectin-7 in murine lymphoma cells is associated with progression toward an aggressive phenotype. Leukemia 2003, 17, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Demers, M.; Biron-Pain, K.; Hébert, J.; Lamarre, A.; Magnaldo, T.; St-Pierre, Y. Galectin-7 in lymphoma: Elevated expression in human lymphoid malignancies and decreased lymphoma dissemination by antisense strategies in experimental model. Cancer Res. 2007, 67, 2824–2829. [Google Scholar] [CrossRef] [PubMed]
- Demers, M.; Couillard, J.; Giglia-Mari, G.; Magnaldo, T.; St-Pierre, Y. Increased galectin-7 gene expression in lymphoma cells is under the control of DNA methylation. Biochem. Biophys. Res. Commun. 2009, 387, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Talantov, D.; Mazumder, A.; Yu, J.X.; Briggs, T.; Jiang, Y.; Backus, J.; Atkins, D.; Wang, Y. Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2005, 11, 7234–7242. [Google Scholar] [CrossRef] [PubMed]
- Cada, Z.; Chovanec, M.; Smetana, K.; Betka, J.; Lacina, L.; Plzák, J.; Kodet, R.; Stork, J.; Lensch, M.; Kaltner, H.; et al. Galectin-7: Will the lectin’s activity establish clinical correlations in head and neck squamous cell and basal cell carcinomas? Histol. Histopathol. 2009, 24, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-J.; Hwang, J.-A.; Ro, J.Y.; Lee, Y.-S.; Chun, K.-H. Galectin-7 is epigenetically-regulated tumor suppressor in gastric cancer. Oncotarget 2013, 4, 1461–1471. [Google Scholar] [CrossRef] [PubMed]
- Matsui, Y.; Ueda, S.; Watanabe, J.; Kuwabara, I.; Ogawa, O.; Nishiyama, H. Sensitizing effect of galectin-7 in urothelial cancer to cisplatin through the accumulation of intracellular reactive oxygen species. Cancer Res. 2007, 67, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Langbein, S.; Brade, J.; Badawi, J.K.; Hatzinger, M.; Kaltner, H.; Lensch, M.; Specht, K.; André, S.; Brinck, U.; Alken, P.; et al. Gene-expression signature of adhesion/growth-regulatory tissue lectins (galectins) in transitional cell cancer and its prognostic relevance. Histopathology 2007, 51, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; He, Q.-Y.; Yuen, A.P.-W.; Chiu, J.-F. Proteomics of buccal squamous cell carcinoma: The involvement of multiple pathways in tumorigenesis. Proteomics 2004, 4, 2465–2475. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Ding, M.; Yu, M.-L.; Feng, M.-X.; Tan, L.-J.; Zhao, F.-K. Identification of galectin-7 as a potential biomarker for esophageal squamous cell carcinoma by proteomic analysis. BMC Cancer 2010, 10, 290. [Google Scholar] [CrossRef] [PubMed]
- Rorive, S.; Eddafali, B.; Fernandez, S.; Decaestecker, C.; André, S.; Kaltner, H.; Kuwabara, I.; Liu, F.-T.; Gabius, H.-J.; Kiss, R.; et al. Changes in galectin-7 and cytokeratin-19 expression during the progression of malignancy in thyroid tumors: Diagnostic and biological implications. Mod. Pathol. 2002, 15, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Pei, H.; Kaeck, M.; Thompson, H.J. Gene expression changes associated with chemically induced rat mammary carcinogenesis. Mol. Carcinog. 1997, 20, 204–215. [Google Scholar] [CrossRef]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Castronovo, V.; van den Brûle, F.A.; Jackers, P.; Clausse, N.; Liu, F.T.; Gillet, C.; Sobel, M.E. Decreased expression of galectin-3 is associated with progression of human breast cancer. J. Pathol. 1996, 179, 43–48. [Google Scholar] [CrossRef]
- Moumen, M.; Chiche, A.; Cagnet, S.; Petit, V.; Raymond, K.; Faraldo, M.M.; Deugnier, M.-A.; Glukhova, M.A. The mammary myoepithelial cell. Int. J. Dev. Biol. 2011, 55, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Prater, M.D.; Petit, V.; Alasdair Russell, I.; Giraddi, R.R.; Shehata, M.; Menon, S.; Schulte, R.; Kalajzic, I.; Rath, N.; Olson, M.F.; et al. Mammary stem cells have myoepithelial cell properties. Nat. Cell Biol. 2014, 16, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Demers, M.; Rose, A.A.N.; Grosset, A.-A.; Biron-Pain, K.; Gaboury, L.; Siegel, P.M.; St-Pierre, Y. Overexpression of galectin-7, a myoepithelial cell marker, enhances spontaneous metastasis of breast cancer cells. Am. J. Pathol. 2010, 176, 3023–3031. [Google Scholar] [CrossRef] [PubMed]
- Grosset, A.-A.; Poirier, F.; Gaboury, L.; St-Pierre, Y. Galectin-7 Expression Potentiates HER-2-Positive Phenotype in Breast Cancer. PLoS ONE 2016, 11, e0166731. [Google Scholar] [CrossRef] [PubMed]
- Eude-Le Parco, I.; Gendronneau, G.; Dang, T.; Delacour, D.; Thijssen, V.L.; Edelmann, W.; Peuchmaur, M.; Poirier, F. Genetic assessment of the importance of galectin-3 in cancer initiation, progression, and dissemination in mice. Glycobiology 2009, 19, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, J.A.; Queiroz, L.M.G.; Silveira, É.J.D.; Gordon-Nunez, M.A.; Godoy, G.P.; Nonaka, C.F.W.; Alves, P.M. Association of immunoexpression of the galectins-3 and -7 with histopathological and clinical parameters in oral squamous cell carcinoma in young patients. Eur. Arch. Oto-Rhino-Laryngol. 2016, 273, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Alves, P.M.; Godoy, G.P.; Gomes, D.Q.; Medeiros, A.M.C.; de Souza, L.B.; da Silveira, E.J.D.; Vasconcelos, M.G.; Queiroz, L.M.G. Significance of galectins-1, -3, -4 and -7 in the progression of squamous cell carcinoma of the tongue. Pathol. Res. Pract. 2011, 207, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Jeon, H.-K.; Lee, J.-K.; Sung, C.O.; Do, I.-G.; Choi, C.H.; Kim, T.-J.; Kim, B.-G.; Bae, D.-S.; Lee, J.-W. Clinical significance of galectin-7 in epithelial ovarian cancer. Anticancer Res. 2013, 33, 1555–1561. [Google Scholar] [PubMed]
- Cagnoni, A.J.; Pérez Sáez, J.M.; Rabinovich, G.A.; Mariño, K.V. Turning-Off Signaling by Siglecs, Selectins, and Galectins: Chemical Inhibition of Glycan-Dependent Interactions in Cancer. Front. Oncol. 2016, 6, 109. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in cancer: Mechanisms and clinical implications. Nat. Rev. Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Stowell, S.R.; Ju, T.; Cummings, R.D. Protein glycosylation in cancer. Annu. Rev. Pathol. 2015, 10, 473–510. [Google Scholar] [CrossRef] [PubMed]
- Ishida, H.; Togayachi, A.; Sakai, T.; Iwai, T.; Hiruma, T.; Sato, T.; Okubo, R.; Inaba, N.; Kudo, T.; Gotoh, M.; et al. A novel β1,3-N-acetylglucosaminyltransferase (β3Gn-T8), which synthesizes poly-N-acetyllactosamine, is dramatically upregulated in colon cancer. FEBS Lett. 2005, 579, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Croci, D.O.; Cerliani, J.P.; Dalotto-Moreno, T.; Méndez-Huergo, S.P.; Mascanfroni, I.D.; Dergan-Dylon, S.; Toscano, M.A.; Caramelo, J.J.; García-Vallejo, J.J.; Ouyang, J.; et al. Glycosylation-dependent lectin-receptor interactions preserve angiogenesis in anti-VEGF refractory tumors. Cell 2014, 156, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Mirandola, L.; Nguyen, D.D.; Rahman, R.L.; Grizzi, F.; Yuefei, Y.; Figueroa, J.A.; Jenkins, M.R.; Cobos, E.; Chiriva-Internati, M. Anti-galectin-3 therapy: A new chance for multiple myeloma and ovarian cancer? Int. Rev. Immunol. 2014, 33, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Thijssen, V.L.; Griffioen, A.W. Galectin-1 and -9 in angiogenesis: A sweet couple. Glycobiology 2014, 24, 915–920. [Google Scholar] [CrossRef] [PubMed]
- Schulz, H.; Schmoeckel, E.; Kuhn, C.; Hofmann, S.; Mayr, D.; Mahner, S.; Jeschke, U. Galectins-1, -3, and -7 Are Prognostic Markers for Survival of Ovarian Cancer Patients. Int. J. Mol. Sci. 2017, 18, 1230. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.J.; Sulman, E.P.; Eifel, P.J.; Jhingran, A.; Allen, P.K.; Deavers, M.T.; Klopp, A.H. Galectin-7 levels predict radiation response in squamous cell carcinoma of the cervix. Gynecol. Oncol. 2013, 131, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, S.; Morita, K.; Negishi, A.; Harada, H.; Nakajima, Y.; Shimamoto, H.; Tomioka, H.; Tanaka, K.; Ono, M.; Yamada, T.; et al. Galectin-7 as a potential predictive marker of chemo- and/or radio-therapy resistance in oral squamous cell carcinoma. Cancer Med. 2014, 3, 349–361. [Google Scholar] [CrossRef] [PubMed]
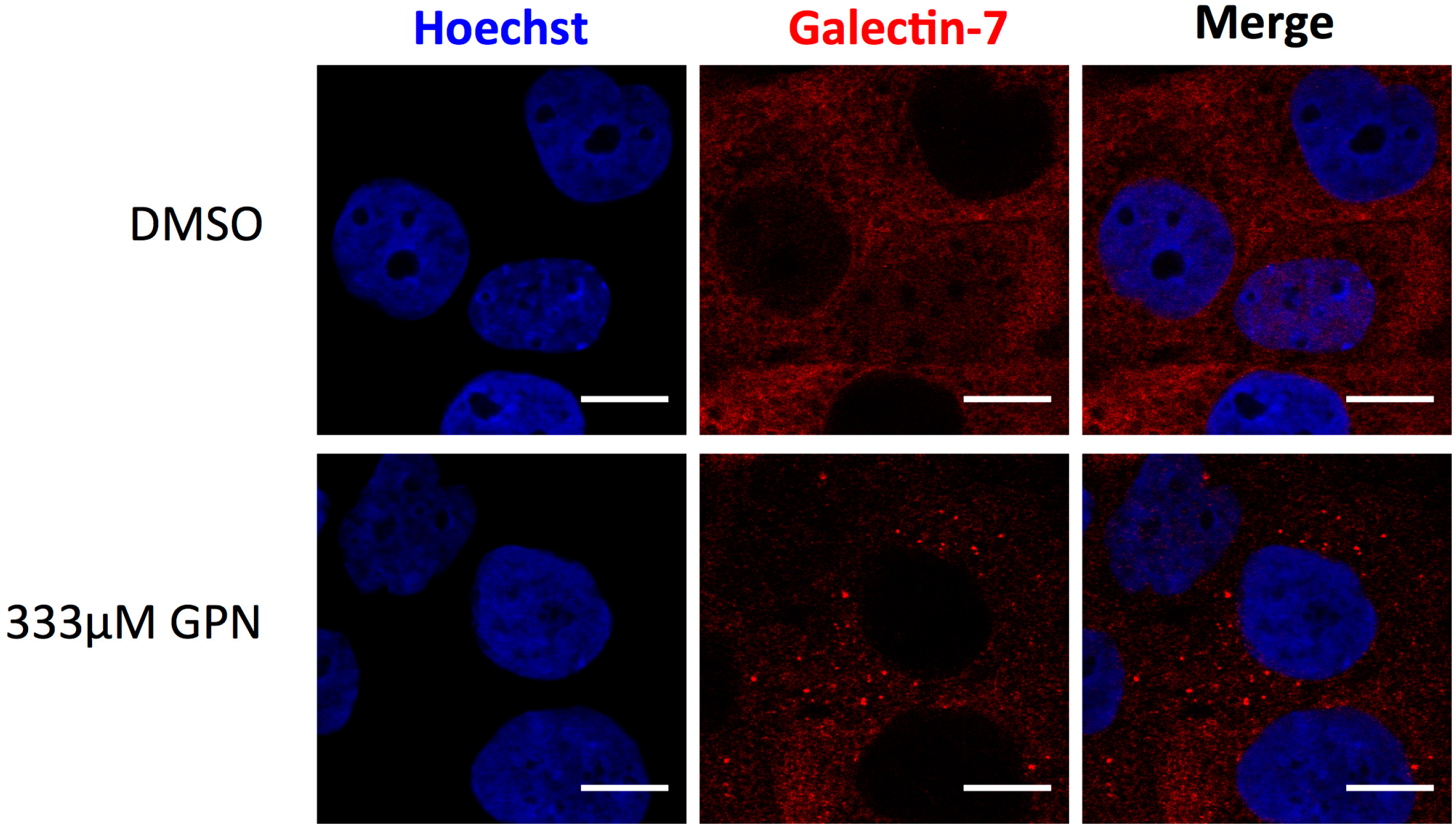
- Bibens-Laulan, N.; St-Pierre, Y. Intracellular galectin-7 expression in cancer cells results from an autocrine transcriptional mechanism and endocytosis of extracellular galectin-7. PLoS ONE 2017, 12, e0187194. [Google Scholar] [CrossRef] [PubMed]
- Advedissian, T.; Deshayes, F.; Poirier, F.; Grandjean, C.; Viguier, M. Galectins, a class of unconventional lectins. Med. Sci. 2015, 31, 499–505. [Google Scholar] [CrossRef]
- Thurston, T.L.M.; Wandel, M.P.; von Muhlinen, N.; Foeglein, A.; Randow, F. Galectin 8 targets damaged vesicles for autophagy to defend cells against bacterial invasion. Nature 2012, 482, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Stegmayr, J.; Lepur, A.; Kahl-Knutson, B.; Aguilar-Moncayo, M.; Klyosov, A.A.; Field, R.A.; Oredsson, S.; Nilsson, U.J.; Leffler, H. Low or No Inhibitory Potency of the Canonical Galectin Carbohydrate-binding Site by Pectins and Galactomannans. J. Biol. Chem. 2016, 291, 13318–13334. [Google Scholar] [CrossRef] [PubMed]
- Dion, J.; Advedissian, T.; Storozhylova, N.; Dahbi, S.; Lambert, A.; Deshayes, F.; Viguier, M.; Tellier, C.; Poirier, F.; Téletchéa, S.; et al. Development of a Sensitive Microarray Platform for the Ranking of Galectin Inhibitors: Identification of a Selective Galectin-3 Inhibitor. ChemBioChem 2017. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Advedissian, T.; Deshayes, F.; Viguier, M. Galectin-7 in Epithelial Homeostasis and Carcinomas. Int. J. Mol. Sci. 2017, 18, 2760. https://doi.org/10.3390/ijms18122760
Advedissian T, Deshayes F, Viguier M. Galectin-7 in Epithelial Homeostasis and Carcinomas. International Journal of Molecular Sciences. 2017; 18(12):2760. https://doi.org/10.3390/ijms18122760
Chicago/Turabian StyleAdvedissian, Tamara, Frédérique Deshayes, and Mireille Viguier. 2017. "Galectin-7 in Epithelial Homeostasis and Carcinomas" International Journal of Molecular Sciences 18, no. 12: 2760. https://doi.org/10.3390/ijms18122760
APA StyleAdvedissian, T., Deshayes, F., & Viguier, M. (2017). Galectin-7 in Epithelial Homeostasis and Carcinomas. International Journal of Molecular Sciences, 18(12), 2760. https://doi.org/10.3390/ijms18122760



