Down-Regulation of Transglutaminase 2 Stimulates Redifferentiation of Dedifferentiated Chondrocytes through Enhancing Glucose Metabolism
Abstract
:1. Introduction
2. Results
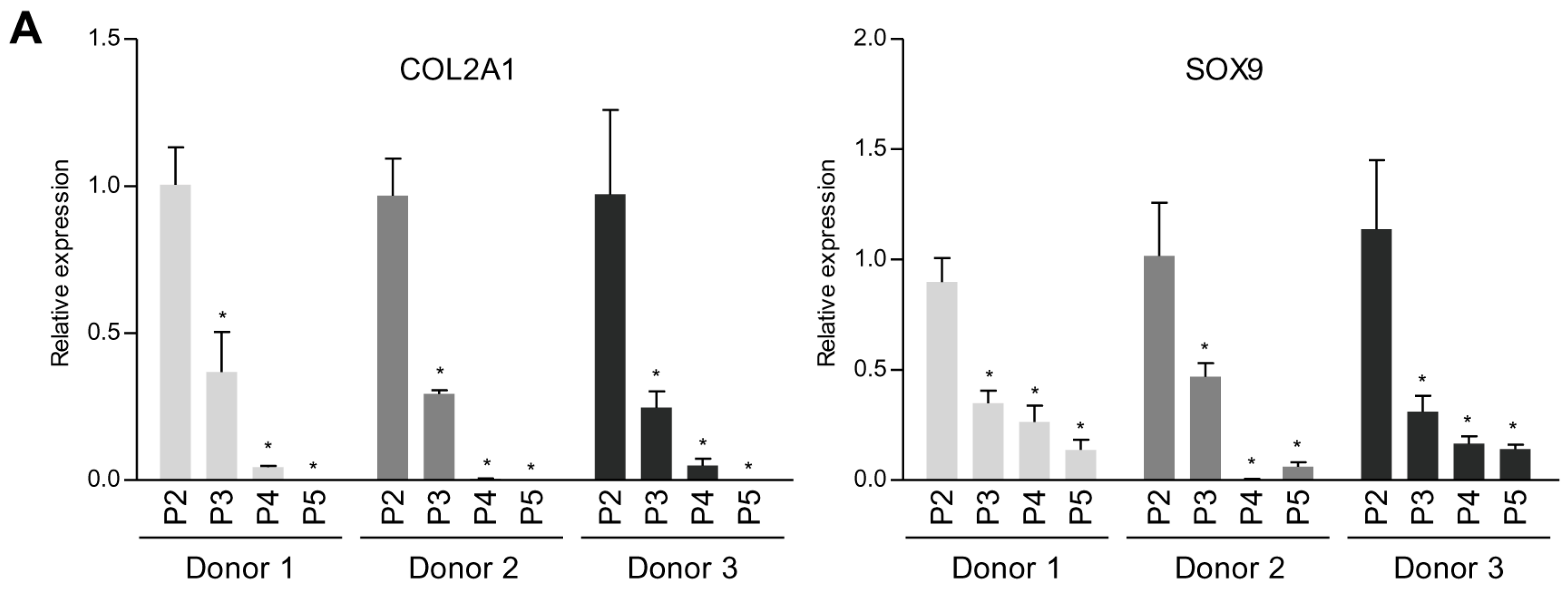
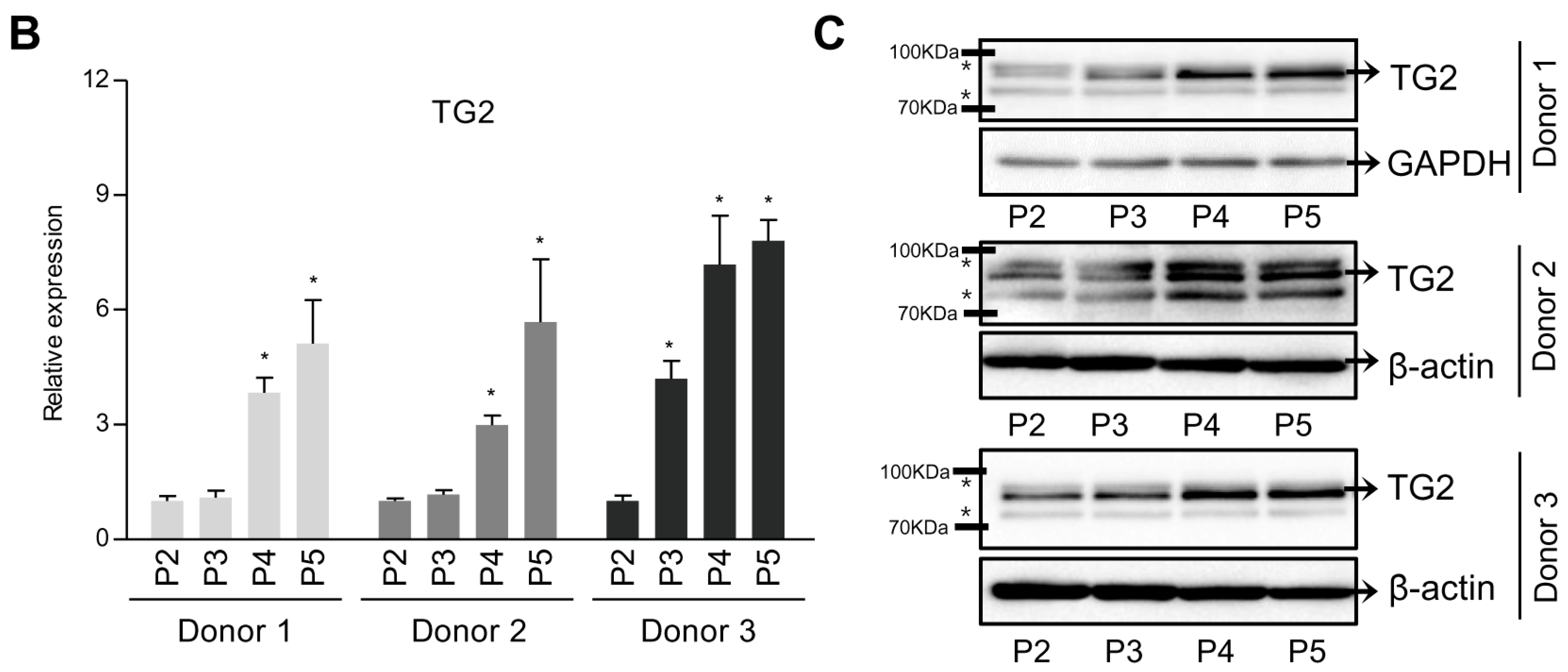
2.1. TG2 Expression is Associated With Dedifferentiation of Chondrocytes
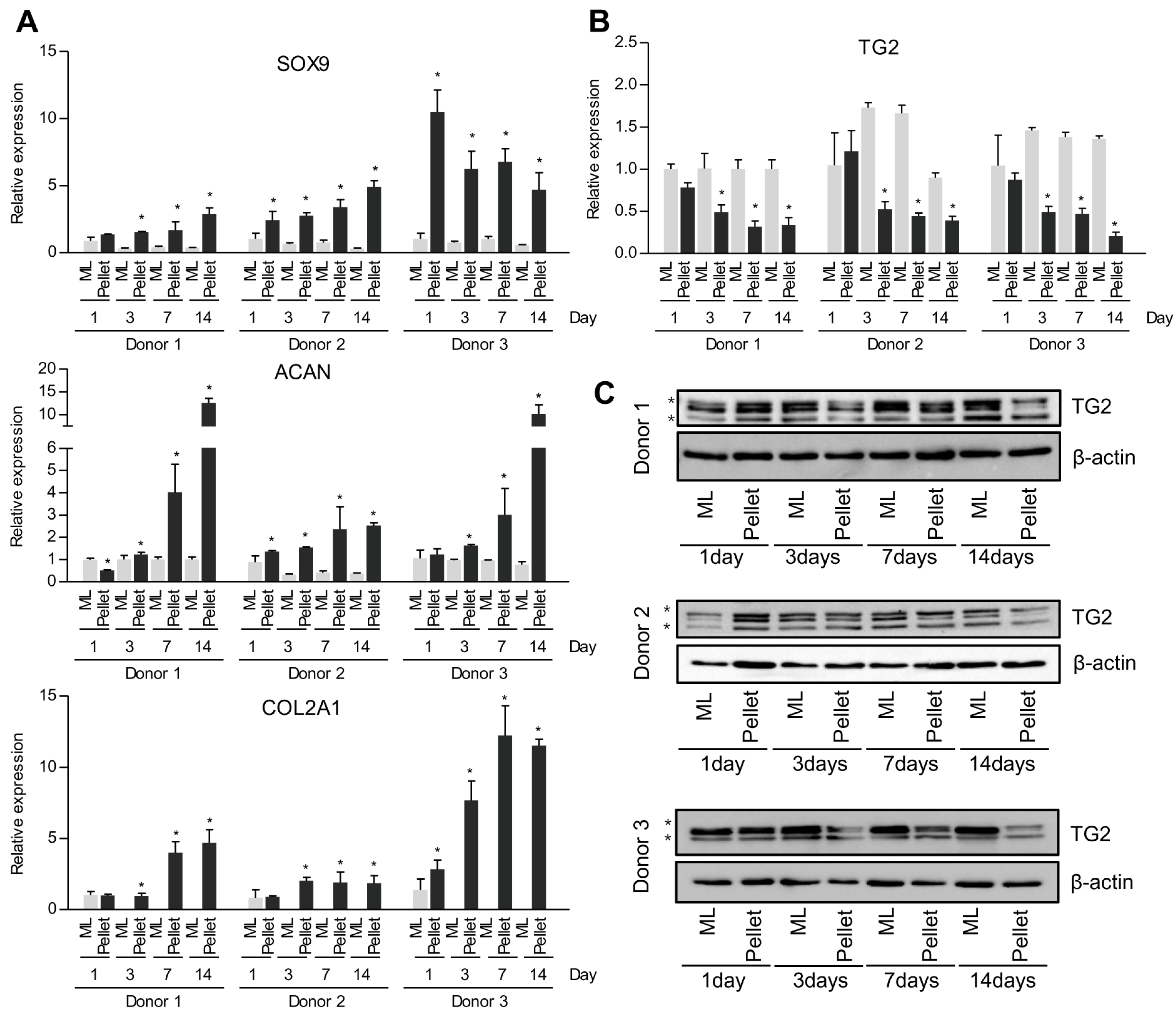
2.2. Alteration of Energy Metabolism in Pellet Culture System
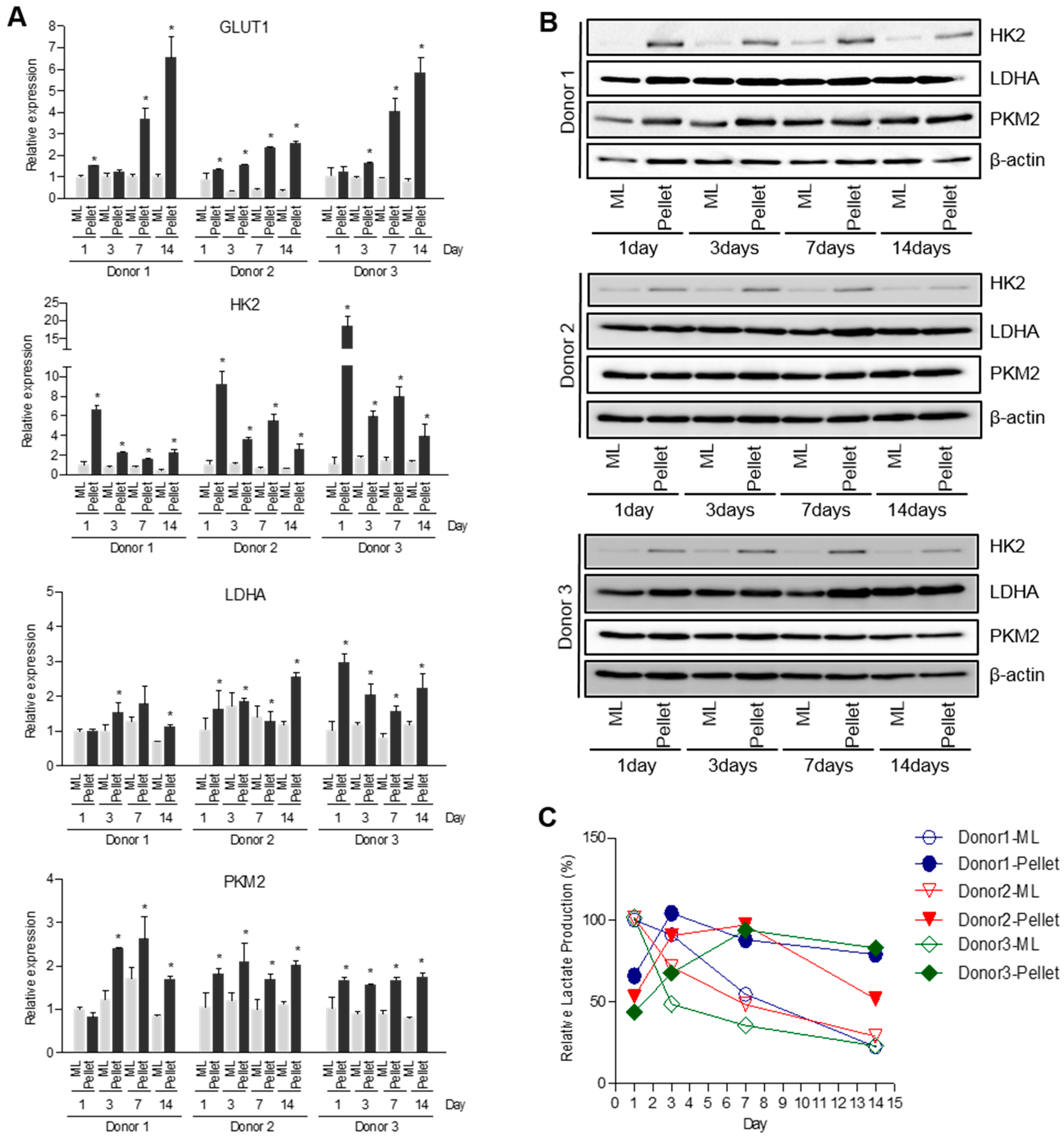
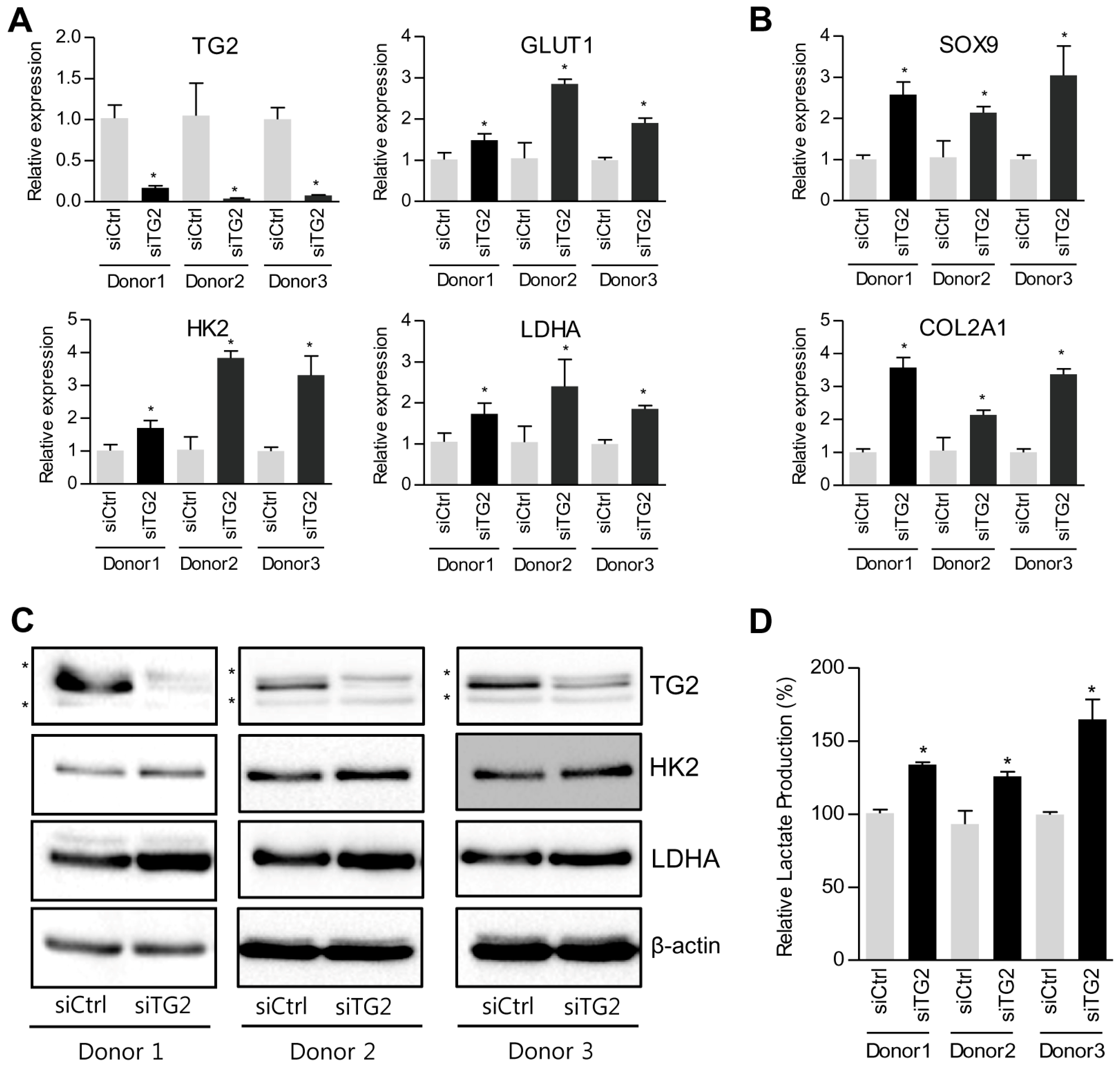
2.3. Down-Regulation of TG2 Enhances Glycolytic Pathway in Chondrocytes
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Chondrocyte Isolation
4.3. Pellet Preparation and Cultivation
4.4. Western Blot Analysis
4.5. RNA Isolation and Real Time Quantitative PCR
4.6. Lactate Assay
4.7. Knockdown Experiment
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 3D culture system | Three-Dimensional Culture System |
| ACI | Autologous Chondrocyte Implantation |
| ECM | Extracellular Matrix |
| GAG | Glycosaminoglycan |
| TG2 | Transglutaminase 2 |
| SOX9 | SRY (Sex Determining Region Y)-Box 9 (SOX9) |
| COL2A1 | Collagen Type II |
| ACAN | Aggrecan |
| GLUT1 | Glucose Transporter 1 |
| HK2 | Hexokinase 2 |
| LDHA | Lactate Dehydrogenase A |
| PKM2 | Pyruvate Kinase Muscle isozyme M2 |
| TGF-β1 | Transforming Growth Factor Beta 1 |
References
- Buckwalter, J. Articular cartilage. AAOS Instr. Course Lect. 1982, 32, 349–370. [Google Scholar]
- Poole, A.R.; Kojima, T.; Yasuda, T.; Mwale, F.; Kobayashi, M.; Laverty, S. Composition and structure of articular cartilage: A template for tissue repair. Clin. Orthop. Relat. Res. 2001, 391, S26–S33. [Google Scholar] [CrossRef]
- Temenoff, J.S.; Mikos, A.G. Tissue engineering for regeneration of articular cartilage. Biomaterials 2000, 21, 431–440. [Google Scholar] [CrossRef]
- Lane, J.; Brighton, C.; Menkowitz, B. Anaerobic and aerobic metabolism in articular cartilage. J. Rheumatol. 1976, 4, 334–342. [Google Scholar]
- Mobasheri, A.; Vannucci, S.; Bondy, C.; Carter, S.; Innes, J.; Arteaga, M.; Trujillo, E.; Ferraz, I.; Shakibaei, M.; Martín-Vasallo, P. Glucose transport and metabolism in chondrocytes: A key to understanding chondrogenesis, skeletal development and cartilage degradation in osteoarthritis. Histol. Histopathol. 2002, 17, 1239–1267. [Google Scholar] [PubMed]
- Stockwell, R. Metabolism of cartilage. Cartilage 2012, 1, 253–273. [Google Scholar]
- URBAN, J. Evidence for a negative Pasteur effect in articular cartilage. Biochem. J. 1997, 321, 95–102. [Google Scholar]
- Nishida, T.; Kubota, S.; Aoyama, E.; Takigawa, M. Impaired glycolytic metabolism causes chondrocyte hypertrophy-like changes via promotion of phospho-Smad1/5/8 translocation into nucleus. Osteoarthr. Cartil. 2013, 21, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.-M.; Kim, H.A.; Kim, S.-J. 2-Deoxy-D-glucose regulates dedifferentiation through β-catenin pathway in rabbit articular chondrocytes. Exp. Mol. Med. 2010, 42, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.D.; Siston, R.A.; Pan, X.; Flanigan, D.C. Autologous Chondrocyte Implantation. J. Bone Joint Surg. 2010, 92, 2220–2233. [Google Scholar] [CrossRef] [PubMed]
- Knutsen, G.; Engebretsen, L.; Ludvigsen, T.C.; Drogset, J.O.; Grøntvedt, T.; Solheim, E.; Strand, T.; Roberts, S.; Isaksen, V.; Johansen, O. Autologous chondrocyte implantation compared with microfracture in the knee: A randomized trail. J. Bone Joint Surg. 2004, 86, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Benya, P.D.; Padilla, S.R.; Nimni, M.E. Independent regulation of collagen types by chondrocytes during the loss of differentiated function in culture. Cell 1978, 15, 1313–1321. [Google Scholar] [CrossRef]
- Von Der Mark, K.; Gauss, V.; Von Der Mark, H.; Müller, P. Relationship between cell shape and type of collagen synthesised as chondrocytes lose their cartilage phenotype in culture. Nature 1977, 267, 531–532. [Google Scholar] [CrossRef] [PubMed]
- Stoppoloni, D.; Politi, L.; Leopizzi, M.; Gaetani, S.; Guazzo, R.; Basciani, S.; Moreschini, O.; de Santi, M.; Scandurra, R.; d’Abusco, A.S. Effect of glucosamine and its peptidyl-derivative on the production of extracellular matrix components by human primary chondrocytes. Osteoarthr. Cartil. 2015, 23, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Chua, K.; Aminuddin, B.; Fuzina, N.; Ruszymah, B. Insulin-transferrin-selenium prevent human chondrocyte dedifferentiation and promote the formation of high quality tissue engineered human hyaline cartilage. Eur. Cell Mater. 2005, 9, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Martin, I.; Vunjak-Novakovic, G.; Yang, J.; Langer, R.; Freed, L. Mammalian chondrocytes expanded in the presence of fibroblast growth factor 2 maintain the ability to differentiate and regenerate three-dimensional cartilaginous tissue. Exp. Cell Res. 1999, 253, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Narcisi, R.; Signorile, L.; Verhaar, J.; Giannoni, P.; Van Osch, G. TGFβ inhibition during expansion phase increases the chondrogenic re-differentiation capacity of human articular chondrocytes. Osteoarthr. Cartil. 2012, 20, 1152–1160. [Google Scholar] [CrossRef] [PubMed]
- Pei, M.; Seidel, J.; Vunjak-Novakovic, G.; Freed, L. Growth factors for sequential cellular de-and re-differentiation in tissue engineering. Biochem. Biophys. Res. Commun. 2002, 294, 149–154. [Google Scholar] [CrossRef]
- Stewart, M.C.; Saunders, K.M.; Burton-Wurster, N.; Macleod, J.N. Phenotypic stability of articular chondrocytes in vitro: The effects of culture models, bone morphogenetic protein 2, and serum supplementation. J. Bone Miner. Res. 2000, 15, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Hicks, D.L.; Sage, A.B.; Schumacher, B.L.; Sah, R.L.; Watson, D. Growth and phenotype of low-density nasal septal chondrocyte monolayers. Otolaryngol.-Head Neck Surg. 2005, 133, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Mandl, E.W.; Van Der Veen, S.W.; Verhaar, J.A.; Van Osch, G.J. Multiplication of human chondrocytes with low seeding densities accelerates cell yield without losing redifferentiation capacity. Tissue Eng. 2004, 10, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Tanzil, G.; de Souza, P.; Castrejon, H.V.; John, T.; Merker, H.-J.; Scheid, A.; Shakibaei, M. Redifferentiation of dedifferentiated human chondrocytes in high-density cultures. Cell Tissue Res. 2002, 308, 371–379. [Google Scholar] [PubMed]
- Watt, F.M. Effect of seeding density on stability of the differentiated phenotype of pig articular chondrocytes in culture. J. Cell Sci. 1988, 89, 373–378. [Google Scholar] [PubMed]
- Lafont, J.E. Lack of oxygen in articular cartilage: Consequences for chondrocyte biology. Int. J. Exp. Pathol. 2010, 91, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.L.; Thoms, B.L.; Vaghjiani, R.J.; Lafont, J.E. HIF-mediated articular chondrocyte function: Prospects for cartilage repair. Arthritis Res. Ther. 2009, 11, 213. [Google Scholar] [CrossRef] [PubMed]
- Sanz-Ramos, P.; Mora, G.; Ripalda, P.; Vicente-Pascual, M.; Izal-Azcárate, I. Identification of signalling pathways triggered by changes in the mechanical environment in rat chondrocytes. Osteoarthr. Cartil. 2012, 20, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.; Dong, M.; Corbeil, D.; Gelinsky, M.; Günther, K.P.; Fickert, S. Pellet culture elicits superior chondrogenic redifferentiation than alginate-based systems. Biotechnol. Prog. 2009, 25, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-J.; Bhang, S.H.; La, W.-G.; Yang, H.S.; Seong, J.Y.; Lee, H.; Im, G.-I.; Lee, S.-H.; Kim, B.-S. Spinner-flask culture induces redifferentiation of de-differentiated chondrocytes. Biotechnol. Lett. 2011, 33, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; McCaffery, J.M.; Spencer, R.G.; Francomano, C.A. Hyaline cartilage engineered by chondrocytes in pellet culture: Histological, immunohistochemical and ultrastructural analysis in comparison with cartilage explants. J. Anat. 2004, 205, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Eckert, R.L.; Kaartinen, M.T.; Nurminskaya, M.; Belkin, A.M.; Colak, G.; Johnson, G.V.; Mehta, K. Transglutaminase regulation of cell function. Physiol. Rev. 2014, 94, 383–417. [Google Scholar] [CrossRef] [PubMed]
- Gundemir, S.; Colak, G.; Tucholski, J.; Johnson, G.V. Transglutaminase 2: A molecular Swiss army knife. BBA Mol. Cell Res. 2012, 1823, 406–419. [Google Scholar] [CrossRef] [PubMed]
- Gaudry, C.; Verderio, E.; Jones, R.; Smith, C.; Griffin, M. Tissue transglutaminase is an important player at the surface of human endothelial cells: Evidence for its externalization and its colocalization with the β 1 integrin. Exp. Cell Res. 1999, 252, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Gaudry, C.A.; Verderio, E.; Aeschlimann, D.; Cox, A.; Smith, C.; Griffin, M. Cell surface localization of tissue transglutaminase is dependent on a fibronectin-binding site in its n-terminal β-sandwich domain. J. Biol. Chem. 1999, 274, 30707–30714. [Google Scholar] [CrossRef] [PubMed]
- Gentile, V.; Thomazy, V.; Piacentini, M.; Fesus, L.; Davies, P. Expression of tissue transglutaminase in Balb-C 3T3 fibroblasts: Effects on cellular morphology and adhesion. J. Cell Biol. 1992, 119, 463–474. [Google Scholar] [CrossRef] [PubMed]
- Nadalutti, C.; Viiri, K.; Kaukinen, K.; Mäki, M.; Lindfors, K. Extracellular transglutaminase 2 has a role in cell adhesion, whereas intracellular transglutaminase 2 is involved in regulation of endothelial cell proliferation and apoptosis. Cell Prolif. 2011, 44, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Xu, J.; Brady, S.; Gao, H.; Yu, D.; Reuben, J.; Mehta, K. Tissue transglutaminase promotes drug resistance and invasion by inducing mesenchymal transition in mammary epithelial cells. PLoS ONE 2010, 5, e13390. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Mehta, K. Tissue transglutaminase-mediated chemoresistance in cancer cells. Drug Resist. Updat. 2007, 10, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Akar, U.; Ozpolat, B.; Mehta, K.; Fok, J.; Kondo, Y.; Lopez-Berestein, G. Tissue transglutaminase inhibits autophagy in pancreatic cancer cells. Mol. Cancer Res. 2007, 5, 241–249. [Google Scholar] [CrossRef] [PubMed]
- D’Eletto, M.; Farrace, M.; Rossin, F.; Strappazzon, F.; di Giacomo, G.; Cecconi, F.; Melino, G.; Sepe, S.; Moreno, S.; Fimia, G.; et al. Type 2 transglutaminase is involved in the autophagy-dependent clearance of ubiquitinated proteins. Cell Death Differ. 2012, 19, 1228–1238. [Google Scholar] [CrossRef] [PubMed]
- Altuntas, S.; D’Eletto, M.; Rossin, F.; Hidalgo, L.D.; Farrace, M.G.; Falasca, L.; Piredda, L.; Cocco, S.; Mastroberardino, P.G.; Piacentini, M.; et al. Type 2 transglutaminase, mitochondria and huntington’s disease: Menage a trois. Mitochondrion 2014, 19, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Rossin, F.; D’Eletto, M.; Falasca, L.; Sepe, S.; Cocco, S.; Fimia, G.; Campanella, M.; Mastroberardino, P.; Farrace, M.; Piacentini, M. Transglutaminase 2 ablation leads to mitophagy impairment associated with a metabolic shift towards aerobic glycolysis. Cell Death Differ. 2015, 22, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Van der Kraan, P.; van den Berg, W. Chondrocyte hypertrophy and osteoarthritis: Role in initiation and progression of cartilage degeneration? Osteoarthr. Cartil. 2012, 20, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Huebner, J.L.; Johnson, K.A.; Kraus, V.B.; Terkeltaub, R.A. Transglutaminase 2 is a marker of chondrocyte hypertrophy and osteoarthritis severity in the Hartley guinea pig model of knee OA. Osteoarthr. Cartil. 2009, 17, 1056–1064. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Donti, T.R.; Agnihotri, N.; Mehta, K. Transglutaminase 2 reprogramming of glucose metabolism in mammary epithelial cells via activation of inflammatory signaling pathways. Int. J. Cancer 2014, 134, 2798–2807. [Google Scholar] [CrossRef] [PubMed]
- Csosz, E.; Bagossi, P.; Nagy, Z.; Dosztanyi, Z.; Simon, I.; Fesus, L. Substrate preference of transglutaminase 2 revealed by logistic regression analysis and intrinsic disorder examination. J. Mol. Biol. 2008, 383, 390–402. [Google Scholar] [CrossRef] [PubMed]
- Facchiano, A.; Facchiano, F. Transglutaminases and their substrates in biology and human diseases: 50 years of growing. Amino Acids 2009, 36, 599–614. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; van Etten, D.; Nanda, N.; Graham, R.M.; Terkeltaub, R.A. Distinct transglutaminase 2-independent and transglutaminase 2-dependent pathways mediate articular chondrocyte hypertrophy. J. Biol. Chem. 2003, 278, 18824–18832. [Google Scholar] [CrossRef] [PubMed]
- Nurminsky, D.; Shanmugasundaram, S.; Deasey, S.; Michaud, C.; Allen, S.; Hendig, D.; Dastjerdi, A.; Francis-West, P.; Nurminskaya, M. Transglutaminase 2 regulates early chondrogenesis and glycosaminoglycan synthesis. Mech. Dev. 2011, 128, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Bernassola, F.; Federici, M.; Corazzari, M.; Terrinoni, A.; Hribal, M.L.; de Laurenzi, V.; Ranalli, M.; Massa, O.; Sesti, G.; McLean, W.I.; et al. Role of transglutaminase 2 in glucose tolerance: Knockout mice studies and a putative mutation in a MODY patient. FASEB J. 2002, 16, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Heywood, H.; Lee, D. Monolayer expansion induces an oxidative metabolism and ros in chondrocytes. Biochem. Biophys. Res. Commun. 2008, 373, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.M.; Shan, Z.Z.; Nakamura, H.; Masuko-Hongo, K.; Kato, T.; Nishioka, K.; Yudoh, K. Catabolic stress induces features of chondrocyte senescence through overexpression of caveolin 1: Possible involvement of caveolin 1–induced down-regulation of articular chondrocytes in the pathogenesis of osteoarthritis. Arthritis Rheum. 2006, 54, 818–831. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.-M.; Kim, S.-J. The thymoquinone-induced production of reactive oxygen species promotes dedifferentiation through the ERK pathway and inflammation through the p38 and PI3K pathways in rabbit articular chondrocytes. Int. J. Mol. Med. 2015, 35, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Griffin, M.; Casadio, R.; Bergamini, C. Transglutaminases: Nature’s biological glues. Biochem. J. 2002, 368, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Gao, B.; Huang, C.; Olsen, B.; Rotundo, R.F.; Blumenstock, F.; Saba, T.M. Transglutaminase-mediated fibronectin multimerization in lung endothelial matrix in response to TNF-α. Am. J. Physiol. 2000, 279, L161–L174. [Google Scholar]
- Kuncio, G.S.; Tsyganskaya, M.; Zhu, J.; Liu, S.-L.; Nagy, L.; Thomazy, V.; Davies, P.J.; Zern, M.A. TNF-α modulates expression of the tissue transglutaminase gene in liver cells. Am. J. Physiol. 1998, 274, G240–G245. [Google Scholar] [PubMed]
- Johnson, K.; Hashimoto, S.; Lotz, M.; Pritzker, K.; Terkeltaub, R. Interleukin-1 induces pro-mineralizing activity of cartilage tissue transglutaminase and factor XIIIa. Am. J. Pathol. 2001, 159, 149–163. [Google Scholar] [CrossRef]
- Suto, N.; Ikura, K.; Sasaki, R. Expression induced by interleukin-6 of tissue-type transglutaminase in human hepatoblastoma hepg2 cells. J. Biol. Chem. 1993, 268, 7469–7473. [Google Scholar] [PubMed]
- Murakami, S.; Lefebvre, V.; de Crombrugghe, B. Potent inhibition of the master chondrogenic factor Sox9 gene by interleukin-1 and tumor necrosis factor-α. J. Biol. Chem. 2000, 275, 3687–3692. [Google Scholar] [CrossRef] [PubMed]
- Hunt, N.C.; Shelton, R.M.; Grover, L.M. Reversible mitotic and metabolic inhibition following the encapsulation of fibroblasts in alginate hydrogels. Biomaterials 2009, 30, 6435–6443. [Google Scholar] [CrossRef] [PubMed]
- Mastroberardino, P.G.; Farrace, M.G.; Viti, I.; Pavone, F.; Fimia, G.M.; Melino, G.; Rodolfo, C.; Piacentini, M. “Tissue” transglutaminase contributes to the formation of disulphide bridges in proteins of mitochondrial respiratory complexes. Biochimi. Biophys.Acta 2006, 1757, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, R.A.; Paggi, M.G.; Scott, L.J.; Pedersen, P.L. Purification and characterization of a bindable form of mitochondrial bound hexokinase from the highly glycolytic AS-30D rat hepatoma cell line. Cancer Res. 1988, 48, 913–919. [Google Scholar] [PubMed]
- Christofk, H.R.; Vander Heiden, M.G.; Harris, M.H.; Ramanathan, A.; Gerszten, R.E.; Wei, R.; Fleming, M.D.; Schreiber, S.L.; Cantley, L.C. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature 2008, 452, 230–233. [Google Scholar] [CrossRef] [PubMed]
| Gene | Human Primer Sequence | |
|---|---|---|
| TG2 | Sense | 5′-GGCACCAAGTACCTGCTCA-3′ |
| Antisense | 5′-AGAGGATGCAAAGAGGAACG-3′ | |
| ACAN | Sense | 5′-GCCTGCGCTCCAATGACT-3′ |
| Antisense | 5′-ATGGAACACGATGCCTTTCAC-3′ | |
| COL2A1 | Sense | 5′-CACGTACACTGCCCTGAAGGA-3′ |
| Antisense | 5′-CGATAACAGTCTTGCCCCACTT-3′ | |
| SOX9 | Sense | 5′-CCCCAACAGATCGCCTACAG-3′ |
| Antisense | 5′-GAGTTCTGGTCGGTGTAGTC-3′ | |
| GLUT1 | Sense | 5′-GGTTGTGCCATACTCATGACC-3′ |
| Antisense | 5′-CAGATAGGACATCCAGGGTAGC-3′ | |
| HK2 | Sense | 5′-TCCCCTGCCACCAGACTA-3′ |
| Antisense | 5′-TGGACTTGAATCCCTTGGTC-3′ | |
| PKM2 | Sense | 5′-CGTCTGAACTTCTCTCATGGAA-3′ |
| Antisense | 5′-ATGGGGTCAGAAGCAAAGC-3′ | |
| LDHA | Sense | 5′-GCAGATTTGGCAGAGAGTATAATG-3′ |
| Antisense | 5′-GACATCATCCTTTATTCCGTAAAGA-3′ | |
| 18S rRNA | Sense | 5′-CTTCCACAGGAGGCCTACAC-3′ |
| Antisense | 5′-CGCAAAATATGCTGGAACTTT-3′ | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, K.-W.; Choi, B.; Park, S.; Arai, Y.; Choi, W.C.; Lee, J.-M.; Bae, H.; Han, I.-B.; Lee, S.-H. Down-Regulation of Transglutaminase 2 Stimulates Redifferentiation of Dedifferentiated Chondrocytes through Enhancing Glucose Metabolism. Int. J. Mol. Sci. 2017, 18, 2359. https://doi.org/10.3390/ijms18112359
Ko K-W, Choi B, Park S, Arai Y, Choi WC, Lee J-M, Bae H, Han I-B, Lee S-H. Down-Regulation of Transglutaminase 2 Stimulates Redifferentiation of Dedifferentiated Chondrocytes through Enhancing Glucose Metabolism. International Journal of Molecular Sciences. 2017; 18(11):2359. https://doi.org/10.3390/ijms18112359
Chicago/Turabian StyleKo, Kyoung-Won, Bogyu Choi, Sunghyun Park, Yoshie Arai, Won Chul Choi, Joong-Myung Lee, Hojae Bae, In-Bo Han, and Soo-Hong Lee. 2017. "Down-Regulation of Transglutaminase 2 Stimulates Redifferentiation of Dedifferentiated Chondrocytes through Enhancing Glucose Metabolism" International Journal of Molecular Sciences 18, no. 11: 2359. https://doi.org/10.3390/ijms18112359
APA StyleKo, K.-W., Choi, B., Park, S., Arai, Y., Choi, W. C., Lee, J.-M., Bae, H., Han, I.-B., & Lee, S.-H. (2017). Down-Regulation of Transglutaminase 2 Stimulates Redifferentiation of Dedifferentiated Chondrocytes through Enhancing Glucose Metabolism. International Journal of Molecular Sciences, 18(11), 2359. https://doi.org/10.3390/ijms18112359








