Dietary Zinc Acts as a Sleep Modulator
Abstract
1. Introduction
2. Sleep
3. Zinc and the Central Nervous System
4. “Sleep as You Eat” or How Food Can Regulate Sleep
5. Sleep Regulation, an Unexpected Function of Zinc
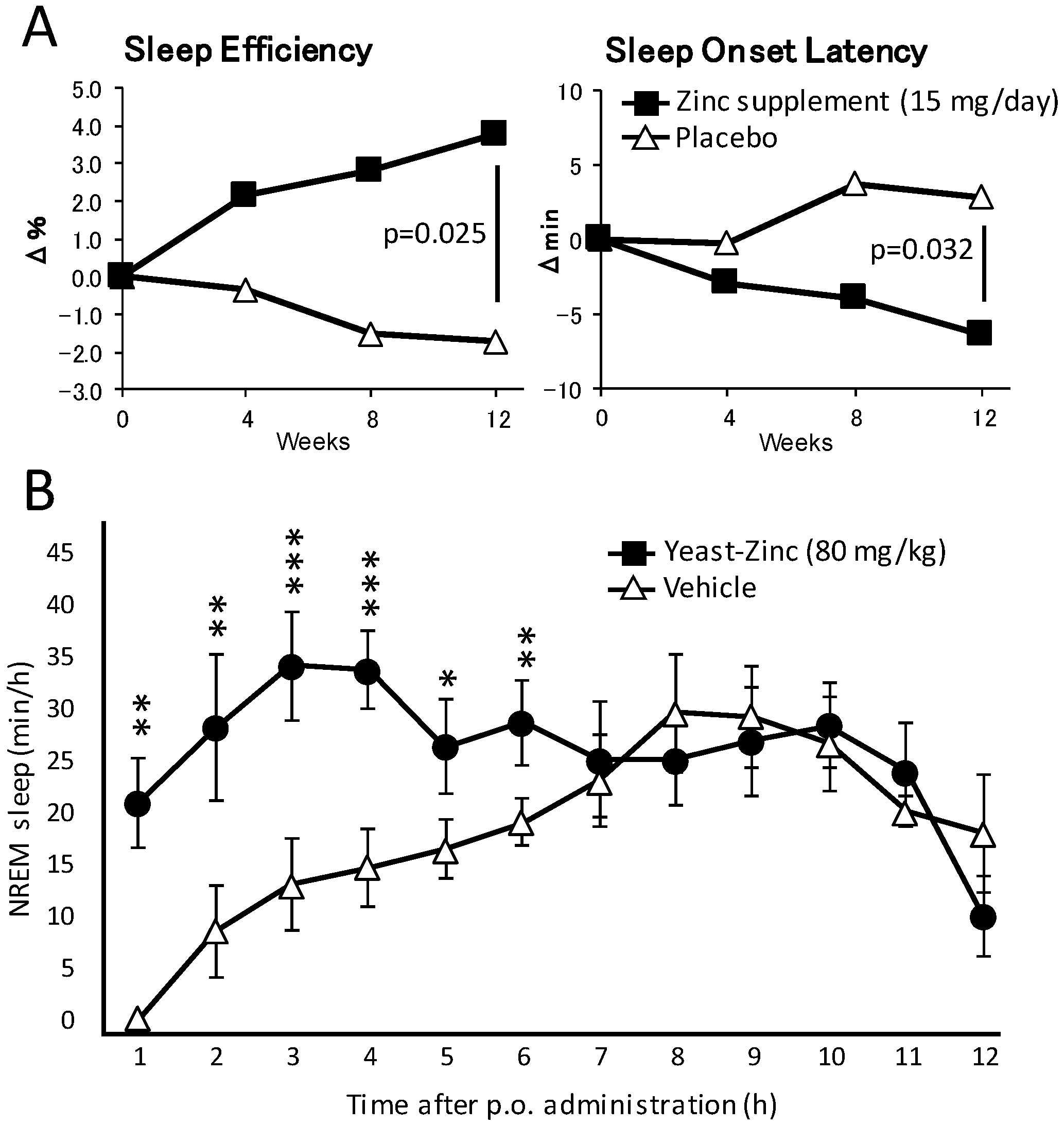
5.1. Clinical Studies
5.2. Experimental Evidence
6. Solving the Mystery of Zinc-Induced Sleep
7. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| CNS | central nervous system |
| EEG | electroencephalogram |
| NREM | non-rapid eye movement |
| REM | rapid eye movement |
| LC | locus coeruleus |
| TMN | tuberomammillary nucleus |
| PB | parabrachial nucleus |
| VLPO | ventrolateral pre-optic area |
| DNA | deoxyribonucleic acid |
| GABA | γ-aminobutyric acid |
| NMDAR | N-methyl-d-aspartate receptor |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor |
| HC | high carbohydrate |
| LF | low fat |
| LC | low carbohydrate |
| HF | high fat |
| CPSQI | Chinese version of the Pittsburgh Sleep Quality Index |
| IDA | iron deficiency anaemia |
| BBB | blood–brain barrier |
| GlyR | glycinergic receptor |
| GPR39 | G protein-coupled receptor 39 |
| p.o. | per os |
References
- Cirelli, C.; Tononi, G. Is sleep essential? PLoS Biol. 2008, 6, e216. [Google Scholar] [CrossRef] [PubMed]
- Bellesi, M.; de Vivo, L.; Chini, M.; Gilli, F.; Tononi, G.; Cirelli, C. Sleep loss promotes astrocytic phagocytosis and microglial activation in mouse cerebral cortex. J. Neurosci. 2017, 37, 5263–5273. [Google Scholar] [CrossRef] [PubMed]
- Kripke, D.F.; Garfinkel, L.; Wingard, D.L.; Klauber, M.R.; Marler, M.R. Mortality associated with sleep duration and insomnia. Arch. Gen. Psychiatry 2002, 59, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Tamakoshi, A.; Ohno, Y. Self-reported sleep duration as a predictor of all-cause mortality: Results from the JACC study, Japan. Sleep 2004, 27, 51–54. [Google Scholar] [PubMed]
- Patel, S.R.; Ayas, N.T.; Malhotra, M.R.; White, D.P.; Schernhammer, E.S.; Speizer, F.E.; Stampfer, M.J.; Hu, F.B. A prospective study of sleep duration and mortality risk in women. Sleep 2004, 27, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Diekelmann, S.; Born, J. The memory function of sleep. Nat. Rev. Neurosci. 2010, 11, 114–126. [Google Scholar] [CrossRef] [PubMed]
- Inoue, S.; Honda, K.; Komoda, Y. Sleep as neuronal detoxification and restitution. Behav. Brain Res. 1995, 69, 91–96. [Google Scholar] [CrossRef]
- Xie, L.; Kang, H.; Xu, Q.; Chen, M.J.; Liao, Y.; Thiyagarajan, M.; O’Donnell, J.; Christensen, D.J.; Nicholson, C.; Iliff, J.J.; et al. Sleep drives metabolite clearance from the adult brain. Science 2013, 342, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Sherin, J.E.; Shiromani, P.J.; McCarley, R.W.; Saper, C.B. Activation of ventrolateral preoptic neurons during sleep. Science 1996, 271, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Saper, C.B.; Fuller, P.M.; Pedersen, N.P.; Lu, J.; Scammell, T.E. Sleep state switching. Neuron 2010, 68, 1023–1042. [Google Scholar] [CrossRef] [PubMed]
- Anaclet, C.; Lin, J.S.; Vetrivelan, R.; Krenzer, M.; Vong, L.; Fuller, P.M.; Lu, J. Identification and characterization of a sleep-active cell group in the rostral medullary brainstem. J. Neurosci. 2012, 32, 17970–17976. [Google Scholar] [CrossRef] [PubMed]
- Anaclet, C.; Ferrari, L.; Arrigoni, E.; Bass, C.E.; Saper, C.B.; Lu, J.; Fuller, P.M. The GABAergic parafacial zone is a medullary slow wave sleep-promoting center. Nat. Neurosci. 2014, 17, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, M.; Shen, H.Y.; Cherasse, Y.; Qu, W.M.; Huang, Z.L.; Bass, C.E.; Winsky-Sommerer, R.; Semba, K.; Fredholm, B.B.; Boison, D.; et al. Arousal effect of caffeine depends on adenosine A2A receptors in the shell of the nucleus accumbens. J. Neurosci. 2011, 31, 10067–10075. [Google Scholar] [CrossRef] [PubMed]
- Oishi, Y.; Xu, Q.; Wang, L.; Zhang, B.J.; Takahashi, K.; Takata, Y.; Luo, Y.J.; Cherasse, Y.; Schiffmann, S.N.; de Kerchove d’Exaerde, A.; et al. Slow-wave sleep is controlled by a subset of nucleus accumbens core neurons in mice. Nat. Commun. 2017, 8, 734. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.S.; Wang, L.; Dong, H.; Qu, W.M.; Yang, S.R.; Cherasse, Y.; Lazarus, M.; Schiffmann, S.N.; d’Exaerde, A.K.; Li, R.X.; et al. Striatal adenosine A2A receptor neurons control active-period sleep via parvalbumin neurons in external globus pallidus. eLife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Discovery of human zinc deficiency: Its impact on human health and disease. Adv. Nutr. 2013, 4, 176–190. [Google Scholar] [CrossRef] [PubMed]
- Klug, A. Zinc finger peptides for the regulation of gene expression. J. Mol. Biol. 1999, 293, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Pauzaite, T.; Thacker, U.; Tollitt, J.; Copeland, N.A. Emerging roles for Ciz1 in cell cycle regulation and as a driver of tumorigenesis. Biomolecules 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Laity, J.H.; Lee, B.M.; Wright, P.E. Zinc finger proteins: New insights into structural and functional diversity. Curr. Opin. Struct. Biol. 2001, 11, 39–46. [Google Scholar] [CrossRef]
- Kim, S.; Yu, N.K.; Kaang, B.K. CTCF as a multifunctional protein in genome regulation and gene expression. Exp. Mol. Med. 2015, 47, e166. [Google Scholar] [CrossRef] [PubMed]
- Loef, M.; von Stillfried, N.; Walach, H. Zinc diet and Alzheimer’s disease: A systematic review. Nutr. Neurosci. 2012, 15, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Hara, T.; Takeda, T.-A.; Takagishi, T.; Fukue, K.; Kambe, T.; Fukada, T. Physiological roles of zinc transporters: Molecular and genetic importance in zinc homeostasis. J. Physiol. Sci. 2017, 67, 283–301. [Google Scholar] [CrossRef] [PubMed]
- Harbison, S.T.; Carbone, M.A.; Ayroles, J.F.; Stone, E.A.; Lyman, R.F.; Mackay, T.F.C. Co-regulated transcriptional networks contribute to natural genetic contribute variation in drosophila sleep. Nat. Genet. 2009, 41, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Groth, C.; Sasamura, T.; Khanna, M.R.; Whitley, M.; Fortini, M.E. Protein trafficking abnormalities in Drosophila tissues with impaired activity of the ZIP7 zinc transporter Catsup. Development 2013, 140, 3018–3027. [Google Scholar] [CrossRef] [PubMed]
- Haug, F.M. Electron microscopical localization of the zinc in hippocampal mossy fibre synapses by a modified sulfide silver procedure. Histochem. Histochem. Histochim. 1967, 8, 355–368. [Google Scholar] [CrossRef]
- Peters, S.; Koh, J.; Choi, D.W. Zinc selectively blocks the action of N-methyl-d-aspartate on cortical neurons. Science 1987, 236, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Westbrook, G.L.; Mayer, M.L. Micromolar concentrations of Zn2+ antagonize NMDA and GABA responses of hippocampal neurons. Nature 1987, 328, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Vogt, K.; Mellor, J.; Tong, G.; Nicoll, R. The actions of synaptically released zinc at hippocampal mossy fiber synapses. Neuron 2000, 26, 187–196. [Google Scholar] [CrossRef]
- Rassendren, F.A.; Lory, P.; Pin, J.P.; Nargeot, J. Zinc has opposite effects on NMDA and non-NMDA receptors expressed in xenopus oocytes. Neuron 1990, 4, 733–740. [Google Scholar] [CrossRef]
- Xie, X.; Gerber, U.; Gahwiler, B.H.; Smart, T.G. Interaction of zinc with ionotropic and metabotropic glutamate receptors in rat hippocampal slices. Neurosci. Lett. 1993, 159, 46–50. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Koh, J.Y.; Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 2005, 6, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Rosati, A.M.; Traversa, U. Mechanisms of inhibitory effects of zinc and cadmium ions on agonist binding to adenosine A1 receptors in rat brain. Biochem. Pharmacol. 1999, 58, 623–632. [Google Scholar] [CrossRef]
- Schetz, J.A.; Sibley, D.R. Zinc allosterically modulates antagonist binding to cloned D1 and D2 dopamine receptors. J. Neurochem. 1997, 68, 1990–1997. [Google Scholar] [CrossRef] [PubMed]
- Gill, C.H.; Peters, J.A.; Lambert, J.J. An electrophysiological investigation of the properties of a murine recombinant 5-HT3 receptor stably expressed in HEK 293 cells. Br. J. Pharmacol. 1995, 114, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, R.J.; Mitrovic, A.D.; Johnston, G.A. Molecular basis for differential inhibition of glutamate transporter subtypes by zinc ions. Mol. Pharmacol. 1998, 54, 189–196. [Google Scholar] [PubMed]
- Richfield, E.K. Zinc modulation of drug binding, cocaine affinity states, and dopamine uptake on the dopamine uptake complex. Mol. Pharmacol. 1993, 43, 100–108. [Google Scholar] [PubMed]
- Busselberg, D.; Evans, M.L.; Rahmann, H.; Carpenter, D.O. Zn2+ blocks the voltage activated calcium current of Aplysia neurons. Neurosci. Lett. 1990, 117, 117–122. [Google Scholar] [CrossRef]
- Erdelyi, L. Zinc blocks the A-type potassium currents in Helix neurons. Acta Physiol. Hung. 1993, 81, 111–120. [Google Scholar] [PubMed]
- Gilly, W.F.; Armstrong, C.M. Slowing of sodium channel opening kinetics in squid axon by extracellular zinc. J. Gen. Physiol. 1982, 79, 935–964. [Google Scholar] [CrossRef] [PubMed]
- Kajita, H.; Whitwell, C.; Brown, P.D. Properties of the inward-rectifying Cl- channel in rat choroid plexus: Regulation by intracellular messengers and inhibition by divalent cations. Pflugers Arch. Eur. J. Physiol. 2000, 440, 933–940. [Google Scholar] [CrossRef]
- Zhang, Y.; Keramidas, A.; Lynch, J.W. The free zinc concentration in the synaptic cleft of artificial glycinergic synapses rises to at least 1 muM. Front. Mol. Neurosci. 2016, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Kodirov, S.A.; Takizawa, S.; Joseph, J.; Kandel, E.R.; Shumyatsky, G.P.; Bolshakov, V.Y. Synaptically released zinc gates long-term potentiation in fear conditioning pathways. Proc. Natl. Acad. Sci. USA 2006, 103, 15218–15223. [Google Scholar] [CrossRef] [PubMed]
- Frederickson, C.J.; Giblin, L.J.; Krezel, A.; McAdoo, D.J.; Mueller, R.N.; Zeng, Y.; Balaji, R.V.; Masalha, R.; Thompson, R.B.; Fierke, C.A.; et al. Concentrations of extracellular free zinc (pZn)e in the central nervous system during simple anesthetization, ischemia and reperfusion. Exp. Neurol. 2006, 198, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Vergnano, A.M.; Rebola, N.; Savtchenko, L.P.; Pinheiro, P.S.; Casado, M.; Kieffer, B.L.; Rusakov, D.A.; Mulle, C.; Paoletti, P. Zinc dynamics and action at excitatory synapses. Neuron 2014, 82, 1101–1114. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hough, C.J.; Suh, S.W.; Sarvey, J.M.; Frederickson, C.J. Rapid translocation of Zn2+ from presynaptic terminals into postsynaptic hippocampal neurons after physiological stimulation. J. Neurophysiol. 2001, 86, 2597–2604. [Google Scholar] [PubMed]
- Hancock, S.M.; Finkelstein, D.I.; Adlard, P.A. Glia and zinc in ageing and Alzheimer’s disease: A mechanism for cognitive decline? Front. Aging Neurosci. 2014, 6, 137. [Google Scholar] [CrossRef] [PubMed]
- Greer, S.M.; Goldstein, A.N.; Walker, M.P. The impact of sleep deprivation on food desire in the human brain. Nat. Commun. 2013, 4, 2259. [Google Scholar] [CrossRef] [PubMed]
- Markwald, R.R.; Melanson, E.L.; Smith, M.R.; Higgins, J.; Perreault, L.; Eckel, R.H.; Wright, K.P., Jr. Impact of insufficient sleep on total daily energy expenditure, food intake, and weight gain. Proc. Natl. Acad. Sci. USA 2013, 110, 5695–5700. [Google Scholar] [CrossRef] [PubMed]
- McEown, K.; Takata, Y.; Cherasse, Y.; Nagata, N.; Aritake, K.; Lazarus, M. Chemogenetic inhibition of the medial prefrontal cortex reverses the effects of REM sleep loss on sucrose consumption. eLife 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Catterson, J.H.; Knowles-Barley, S.; James, K.; Heck, M.M.; Harmar, A.J.; Hartley, P.S. Dietary modulation of Drosophila sleep-wake behaviour. PLoS ONE 2010, 5, e12062. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, T.; Tomita, J.; Hashimoto, R.; Ueno, T.; Kume, S.; Kume, K. Sweetness induces sleep through gustatory signalling independent of nutritional value in a starved fruit fly. Sci. Rep. 2017, 7, 14355. [Google Scholar] [CrossRef] [PubMed]
- Frank, S.; Gonzalez, K.; Lee-Ang, L.; Young, M.C.; Tamez, M.; Mattei, J. Diet and sleep physiology: Public health and clinical implications. Front. Neurol. 2017, 8, 393. [Google Scholar] [CrossRef] [PubMed]
- Phillips, F.; Chen, C.N.; Crisp, A.H.; Koval, J.; McGuinness, B.; Kalucy, R.S.; Kalucy, E.C.; Lacey, J.H. Isocaloric diet changes and electroencephalographic sleep. Lancet 1975, 2, 723–725. [Google Scholar] [CrossRef]
- Lin, H.H.; Tsai, P.S.; Fang, S.C.; Liu, J.F. Effect of kiwifruit consumption on sleep quality in adults with sleep problems. Asia Pac. J. Clin. Nutr. 2011, 20, 169–174. [Google Scholar] [PubMed]
- Masaki, M.; Aritake, K.; Tanaka, H.; Shoyama, Y.; Huang, Z.L.; Urade, Y. Crocin promotes non-rapid eye movement sleep in mice. Mol. Nutr. Food Res. 2012, 56, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Xu, X.H.; Liu, T.Y.; Hong, Z.Y.; Urade, Y.; Huang, Z.L.; Qu, W.M. Safranal enhances non-rapid eye movement sleep in pentobarbital-treated mice. CNS Neurosci. Ther. 2012, 18, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Qu, W.M.; Yue, X.F.; Sun, Y.; Fan, K.; Chen, C.R.; Hou, Y.P.; Urade, Y.; Huang, Z.L. Honokiol promotes non-rapid eye movement sleep via the benzodiazepine site of the GABA(A) receptor in mice. Br. J. Pharmacol. 2012, 167, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.R.; Zhou, X.Z.; Luo, Y.J.; Huang, Z.L.; Urade, Y.; Qu, W.M. Magnolol, a major bioactive constituent of the bark of Magnolia officinalis, induces sleep via the benzodiazepine site of GABA(A) receptor in mice. Neuropharmacology 2012, 63, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Yoon, M.; Pae, A.N.; Jin, Y.H.; Cho, N.C.; Takata, Y.; Urade, Y.; Kim, S.; Kim, J.S.; Yang, H.; et al. Marine polyphenol phlorotannins promote non-rapid eye movement sleep in mice via the benzodiazepine site of the GABAA receptor. Psychopharmacology 2014, 231, 2825–2837. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Midorikawa, T.; Monoi, N.; Kimura, E.; Murata-Matsuno, A.; Sano, T.; Oka, K.; Sugafuji, T.; Uchiyama, A.; Murakoshi, M.; et al. Oral administration of Japanese sake yeast (Saccharomyces cerevisiae sake) promotes non-rapid eye movement sleep in mice via adenosine A2A receptors. J. Sleep Res. 2016, 25, 746–753. [Google Scholar] [CrossRef] [PubMed]
- Monoi, N.; Matsuno, A.; Nagamori, Y.; Kimura, E.; Nakamura, Y.; Oka, K.; Sano, T.; Midorikawa, T.; Sugafuji, T.; Murakoshi, M.; et al. Japanese sake yeast supplementation improves the quality of sleep: A double-blind randomised controlled clinical trial. J. Sleep Res. 2016, 25, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, M.K.; Kaul, S.C.; Wadhwa, R.; Yanagisawa, M.; Urade, Y. Triethylene glycol, an active component of Ashwagandha (Withania somnifera) leaves, is responsible for sleep induction. PLoS ONE 2017, 12, e0172508. [Google Scholar] [CrossRef] [PubMed]
- Grandner, M.A.; Jackson, N.; Gerstner, J.R.; Knutson, K.L. Dietary nutrients associated with short and long sleep duration. Data from a nationally representative sample. Appetite 2013, 64, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Q.; Li, N.; Zhang, Z.; Gao, S.; Yin, H.Y.; Guo, D.M.; Gao, X. Serum zinc, copper, and zinc/copper in healthy residents of Jinan. Biol. Trace Elem. Res. 2009, 131, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Song, C.H.; Kim, Y.H.; Jung, K.I. Associations of zinc and copper levels in serum and hair with sleep duration in adult women. Biol. Trace Elem. Res. 2012, 149, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Liu, J. Associations between blood zinc concentrations and sleep quality in childhood: A cohort study. Nutrients 2015, 7, 5684–5696. [Google Scholar] [CrossRef] [PubMed]
- Kordas, K.; Siegel, E.H.; Olney, D.K.; Katz, J.; Tielsch, J.M.; Kariger, P.K.; Khalfan, S.S.; LeClerq, S.C.; Khatry, S.K.; Stoltzfus, R.J. The effects of iron and/or zinc supplementation on maternal reports of sleep in infants from Nepal and Zanzibar. J. Dev. Behav. Pediatr. 2009, 30, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Opizzi, A.; Monteferrario, F.; Antoniello, N.; Manni, R.; Klersy, C. The effect of melatonin, magnesium, and zinc on primary insomnia in long-term care facility residents in Italy: A double-blind, placebo-controlled clinical trial. J. Am. Geriat. Soc. 2011, 59, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Cherasse, Y.; Suzuki, R.; Mitarai, M.; Ueda, F.; Urade, Y. Zinc-rich oysters as well as zinc-yeast- and astaxanthin-enriched food improved sleep efficiency and sleep onset in a randomized controlled trial of healthy individuals. Mol. Nutr. Food Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Auld, F.; Maschauer, E.L.; Morrison, I.; Skene, D.J.; Riha, R.L. Evidence for the efficacy of melatonin in the treatment of primary adult sleep disorders. Sleep Med. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.J.; Pierce, M.M.; Sehgal, A.; Wu, T.; Skipper, D.C.; Chabba, R. Effect of taurine and caffeine on sleep-wake activity in Drosophila melanogaster. Nat. Sci. Sleep 2010, 2, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Cherasse, Y.; Saito, H.; Nagata, N.; Aritake, K.; Lazarus, M.; Urade, Y. Zinc-containing yeast extract promotes nonrapid eye movement sleep in mice. Mol. Nutr. Food Res. 2015, 59, 2087–2093. [Google Scholar] [CrossRef] [PubMed]
- Blair-West, J.R.; Denton, D.A.; Gibson, A.P.; McKinley, M.J. Opening the blood-brain barrier to zinc. Brain Res. 1990, 507, 6–10. [Google Scholar] [CrossRef]
- Pullen, R.G.; Franklin, P.A.; Hall, G.H. 65zinc uptake from blood into brain and other tissues in the rat. Neurochem. Res. 1990, 15, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Kasarskis, E.J. Zinc metabolism in normal and zinc-deficient rat brain. Exp. Neurol. 1984, 85, 114–127. [Google Scholar] [CrossRef]
- Bobilya, D.J.; Guerin, J.L.; Rowe, D.J. Zinc transport across an in vitro blood-brain barrier model. J. Trace Elem. Exp. Med. 1997, 10, 9–18. [Google Scholar] [CrossRef]
- Markowitz, M.E.; Rosen, J.F.; Mizruchi, M. Circadian variations in serum zinc (Zn) concentrations: Correlation with blood ionized calcium, serum total calcium and phosphate in humans. Am. J. Clin. Nutr. 1985, 41, 689–696. [Google Scholar] [PubMed]
- Ding, F.; O’Donnell, J.; Xu, Q.; Kang, N.; Goldman, N.; Nedergaard, M. Changes in the composition of brain interstitial ions control the sleep-wake cycle. Science 2016, 352, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Tatsuki, F.; Sunagawa, G.A.; Shi, S.; Susaki, E.A.; Yukinaga, H.; Perrin, D.; Sumiyama, K.; Ukai-Tadenuma, M.; Fujishima, H.; Ohno, R.; et al. Involvement of Ca2+-dependent hyperpolarization in sleep duration in mammals. Neuron 2016, 90, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Danscher, G.; Stoltenberg, M. Zinc-specific autometallographic in vivo selenium methods: Tracing of zinc-enriched (ZEN) terminals, ZEN pathways, and pools of zinc ions in a multitude of other ZEN cells. J. Histochem. Cytochem. 2005, 53, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Hondo, M.; Furutani, N.; Yamasaki, M.; Watanabe, M.; Sakurai, T. Orexin neurons receive glycinergic innervations. PLoS ONE 2011, 6, e25076. [Google Scholar] [CrossRef] [PubMed]
- Hirzel, K.; Muller, U.; Latal, A.T.; Hulsmann, S.; Grudzinska, J.; Seeliger, M.W.; Betz, H.; Laube, B. Hyperekplexia phenotype of glycine receptor alpha1 subunit mutant mice identifies Zn2+ as an essential endogenous modulator of glycinergic neurotransmission. Neuron 2006, 52, 679–690. [Google Scholar] [CrossRef] [PubMed]
- McKee, K.K.; Tan, C.P.; Palyha, O.C.; Liu, J.; Feighner, S.D.; Hreniuk, D.L.; Smith, R.G.; Howard, A.D.; van der Ploeg, L.H. Cloning and characterization of two human G protein-coupled receptor genes (GPR38 and GPR39) related to the growth hormone secretagogue and neurotensin receptors. Genomics 1997, 46, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Jackson, V.R.; Nothacker, H.P.; Civelli, O. GPR39 receptor expression in the mouse brain. Neuroreport 2006, 17, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Besser, L.; Chorin, E.; Sekler, I.; Silverman, W.F.; Atkin, S.; Russell, J.T.; Hershfinkel, M. Synaptically released zinc triggers metabotropic signaling via a zinc-sensing receptor in the hippocampus. J. Neurosci. 2009, 29, 2890–2901. [Google Scholar] [CrossRef] [PubMed]
- Mlyniec, K.; Budziszewska, B.; Holst, B.; Ostachowicz, B.; Nowak, G. GPR39 (zinc receptor) knockout mice exhibit depression-like behavior and CREB/BDNF down-regulation in the hippocampus. Int. J. Neuropsychopharmacol. 2014, 18. [Google Scholar] [CrossRef] [PubMed]
- Mlyniec, K.; Doboszewska, U.; Szewczyk, B.; Sowa-Kucma, M.; Misztak, P.; Piekoszewski, W.; Trela, F.; Ostachowicz, B.; Nowak, G. The involvement of the GPR39-Zn(2+)-sensing receptor in the pathophysiology of depression. Studies in rodent models and suicide victims. Neuropharmacology 2014, 79, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.Z. A possible significant role of zinc and GPR39 zinc sensing receptor in Alzheimer disease and epilepsy. Biomed. Pharmacother. 2016, 79, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Burke, S.L.; Maramaldi, P.; Cadet, T.; Kukull, W. Associations between depression, sleep disturbance, and apolipoprotein E in the development of Alzheimer’s disease: Dementia. Int. Psychogeriatr. 2016, 28, 1409–1424. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cherasse, Y.; Urade, Y. Dietary Zinc Acts as a Sleep Modulator. Int. J. Mol. Sci. 2017, 18, 2334. https://doi.org/10.3390/ijms18112334
Cherasse Y, Urade Y. Dietary Zinc Acts as a Sleep Modulator. International Journal of Molecular Sciences. 2017; 18(11):2334. https://doi.org/10.3390/ijms18112334
Chicago/Turabian StyleCherasse, Yoan, and Yoshihiro Urade. 2017. "Dietary Zinc Acts as a Sleep Modulator" International Journal of Molecular Sciences 18, no. 11: 2334. https://doi.org/10.3390/ijms18112334
APA StyleCherasse, Y., & Urade, Y. (2017). Dietary Zinc Acts as a Sleep Modulator. International Journal of Molecular Sciences, 18(11), 2334. https://doi.org/10.3390/ijms18112334





