Determination of Free-Form and Peptide Bound Pyrraline in the Commercial Drinks Enriched with Different Protein Hydrolysates
Abstract
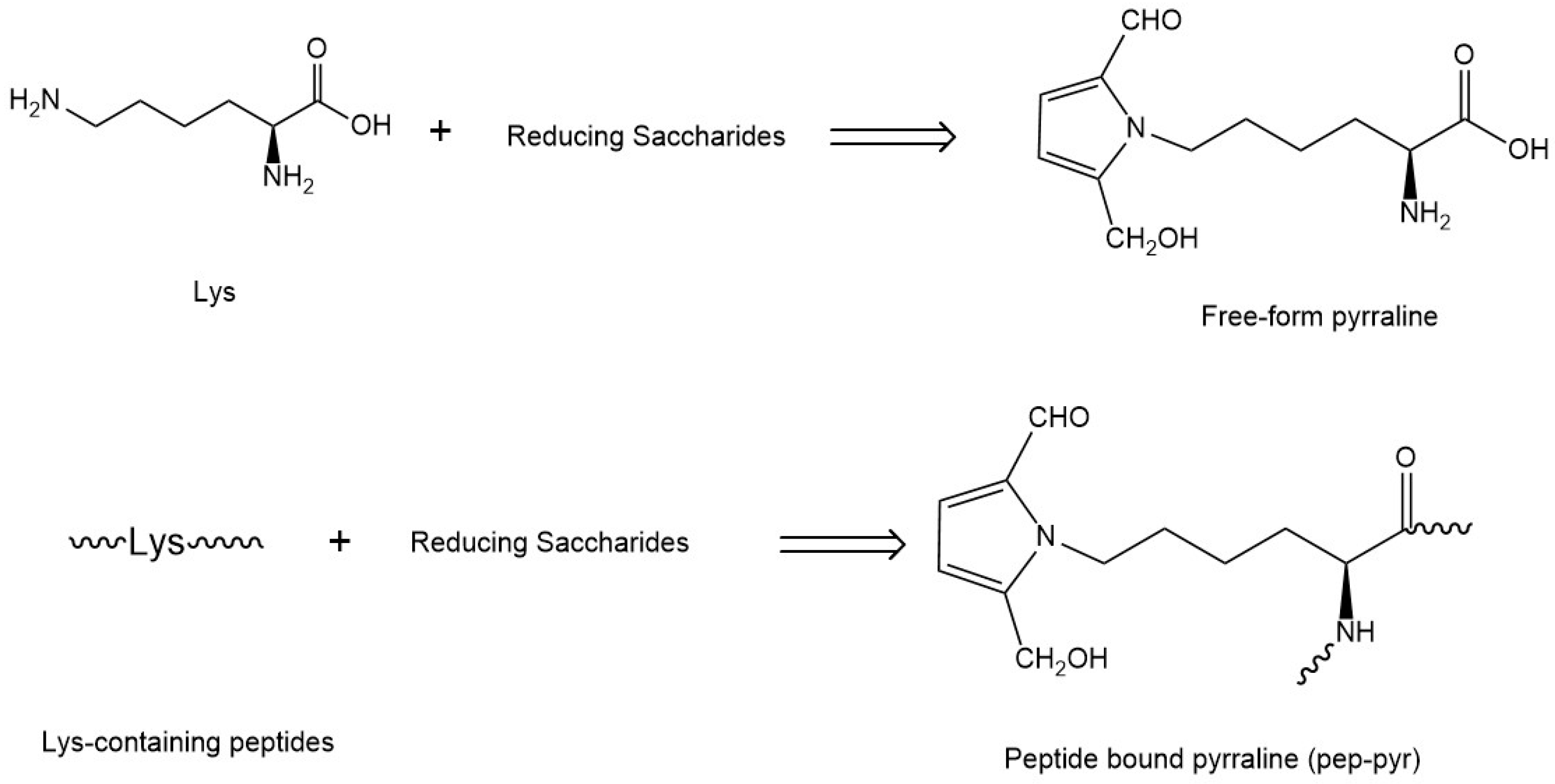
:1. Introduction
2. Results
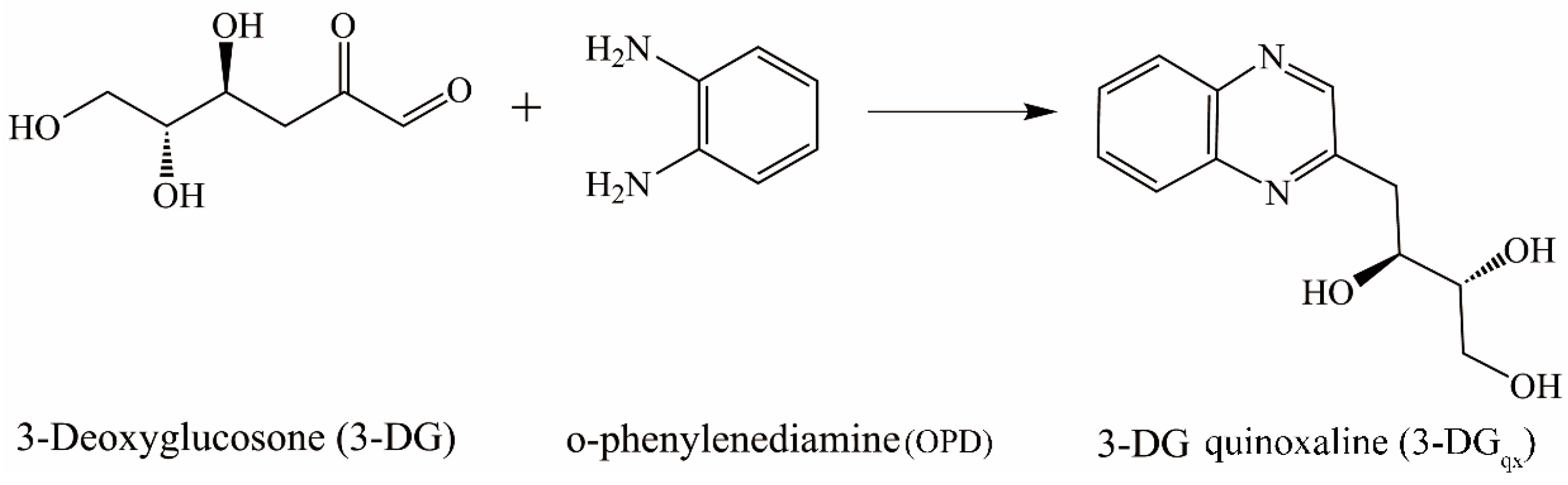
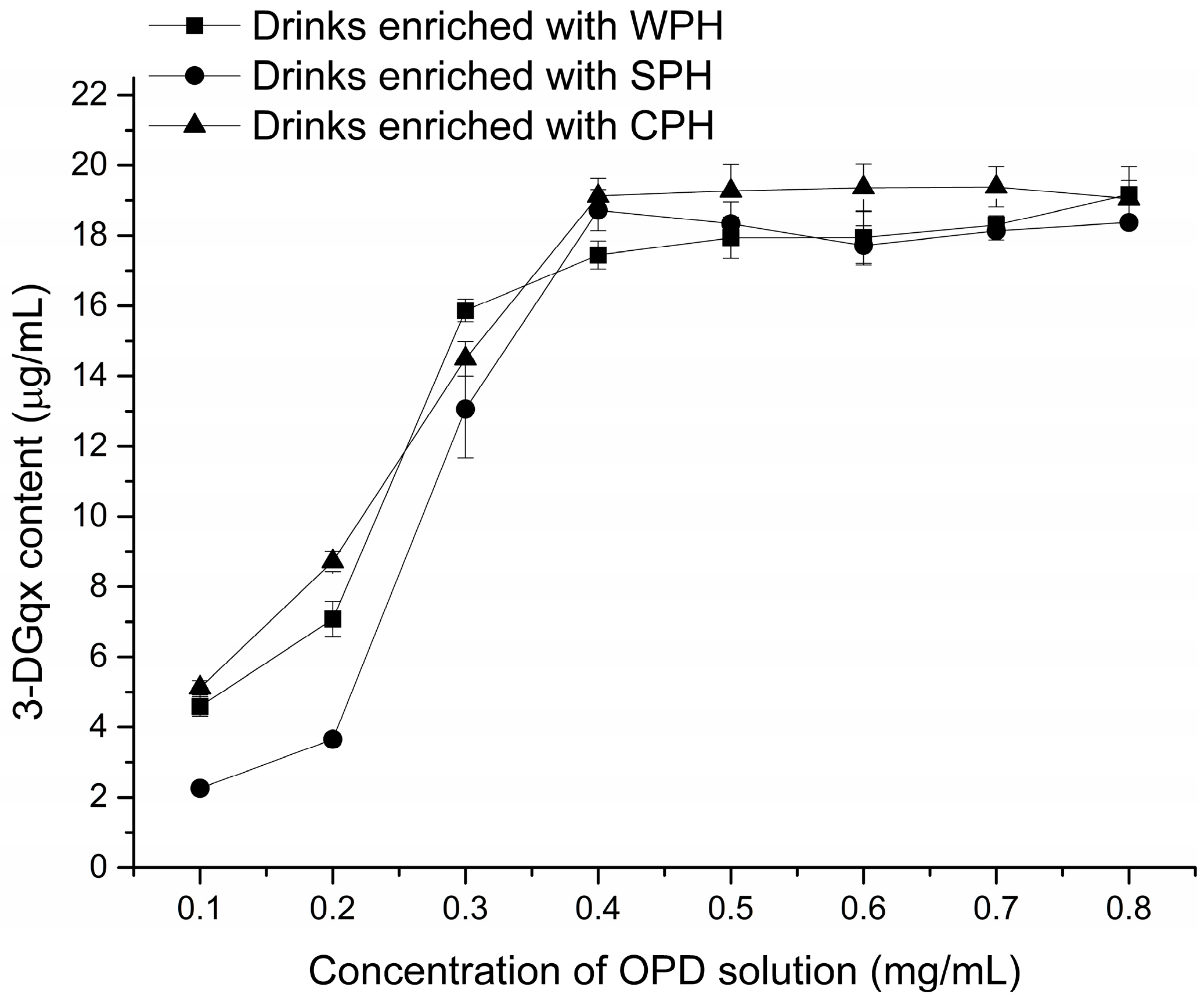
2.1. Optimization of Derivatization Procedure
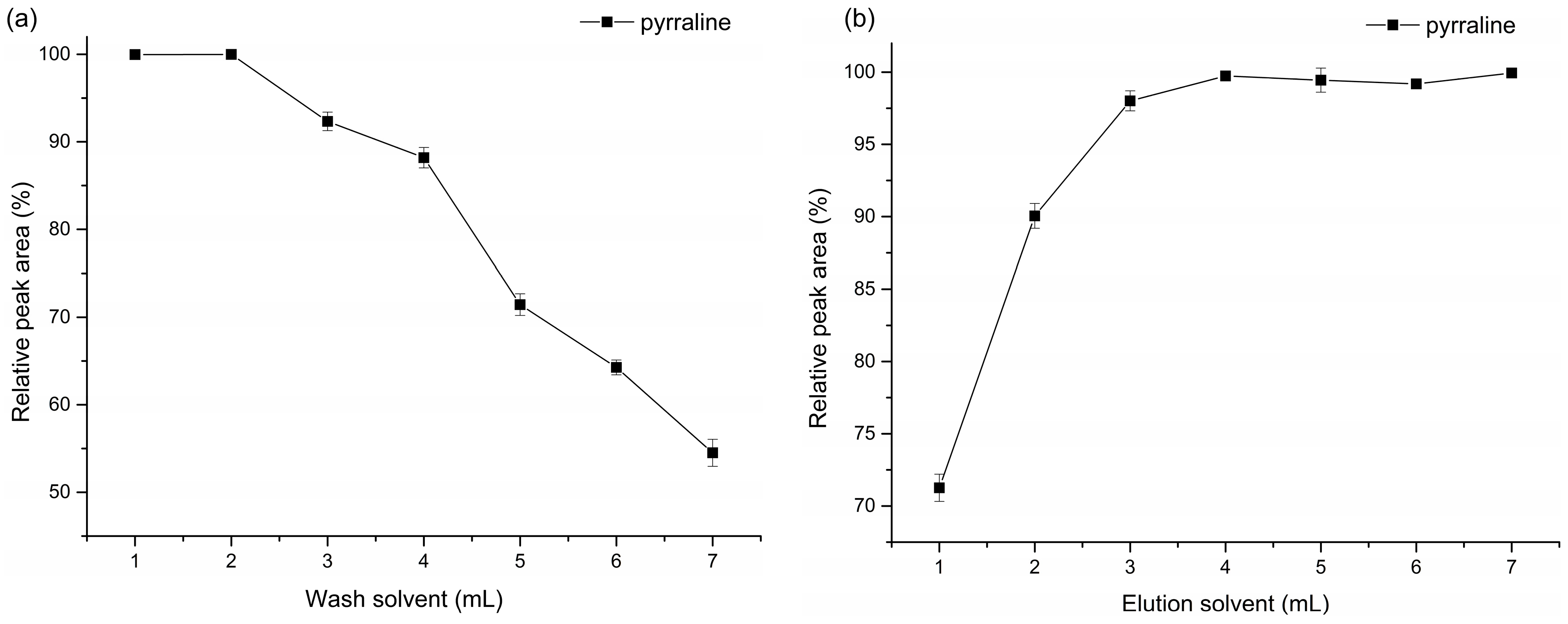
2.2. Optimization of SPE Procedure
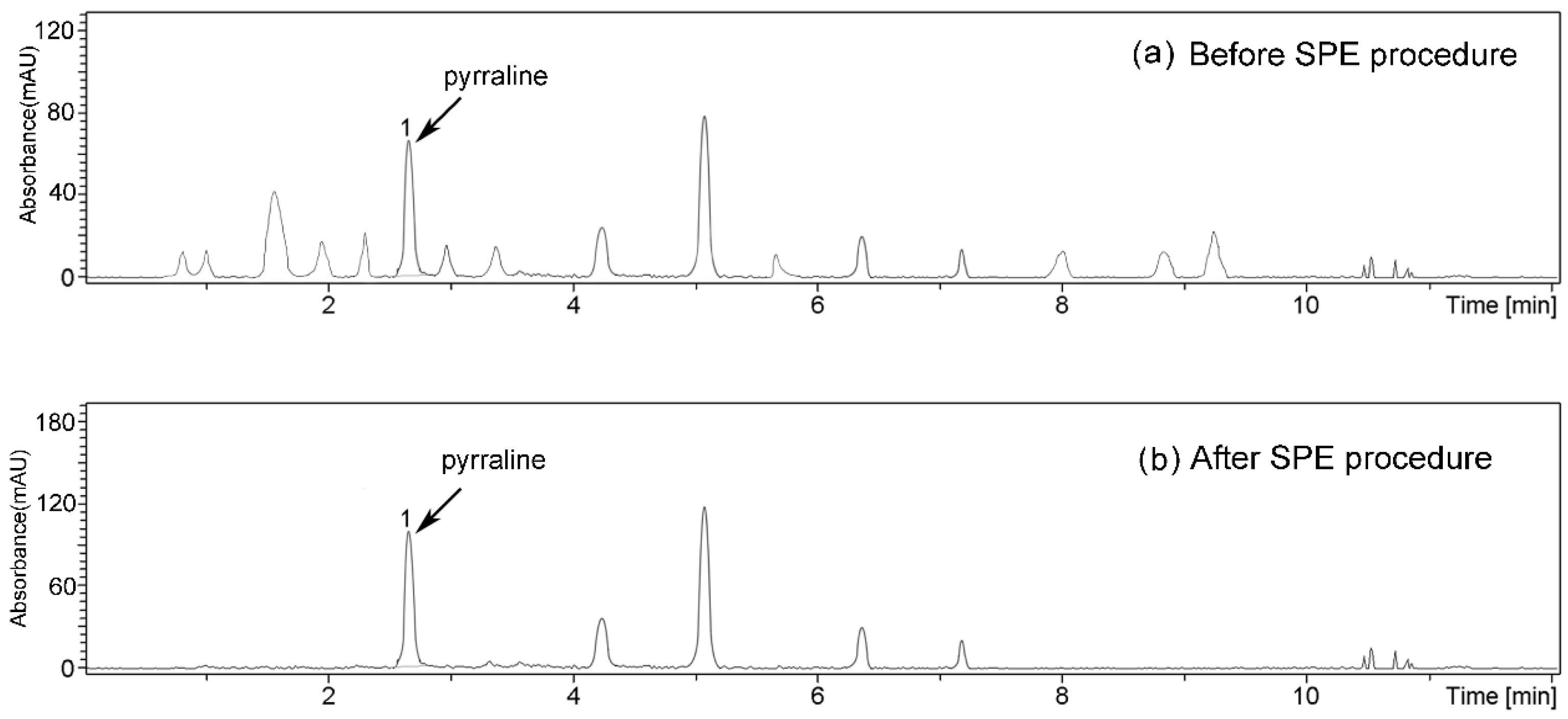
2.3. Purification of PEDs by SPE
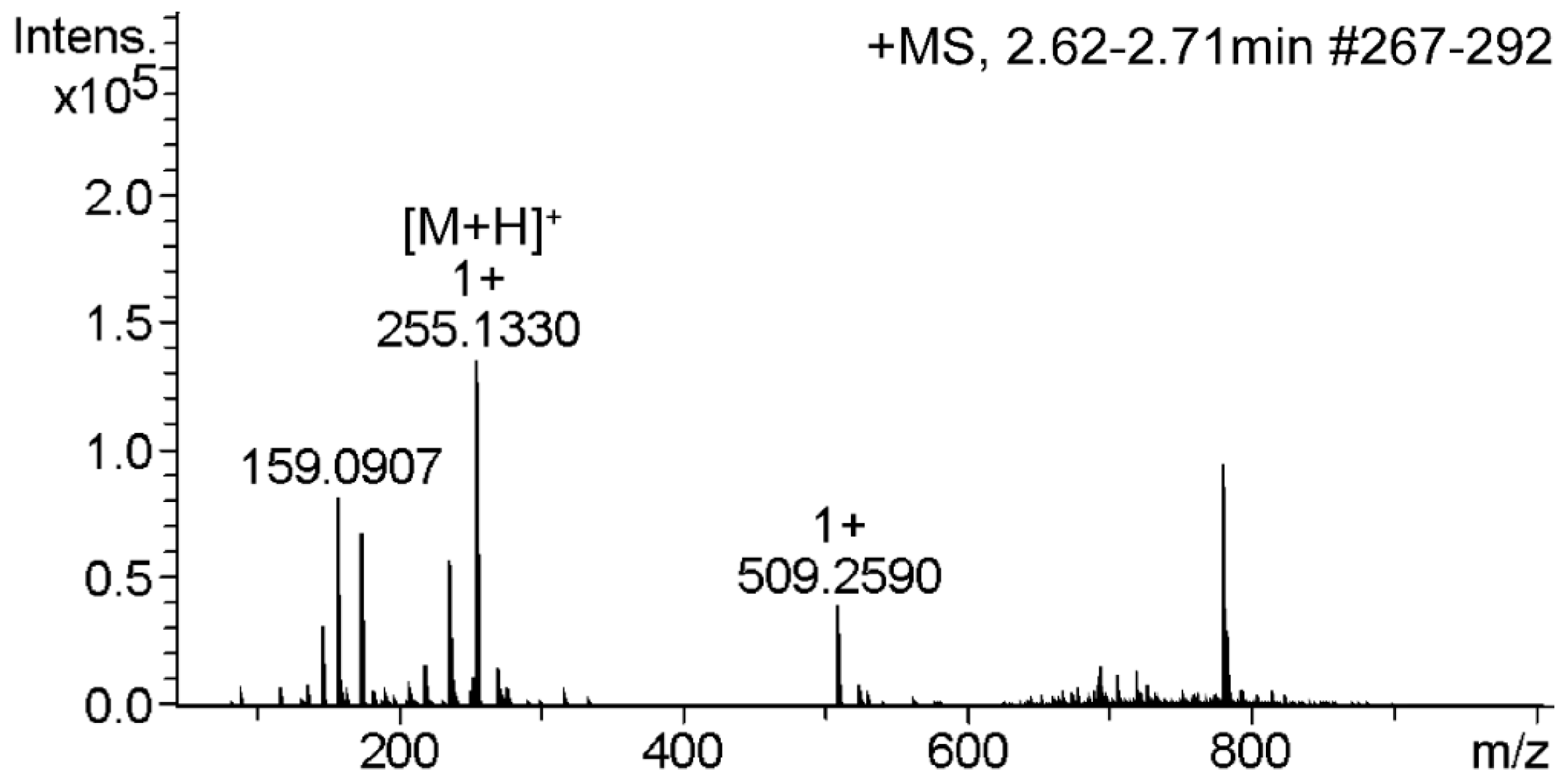
2.4. Validation of the Method
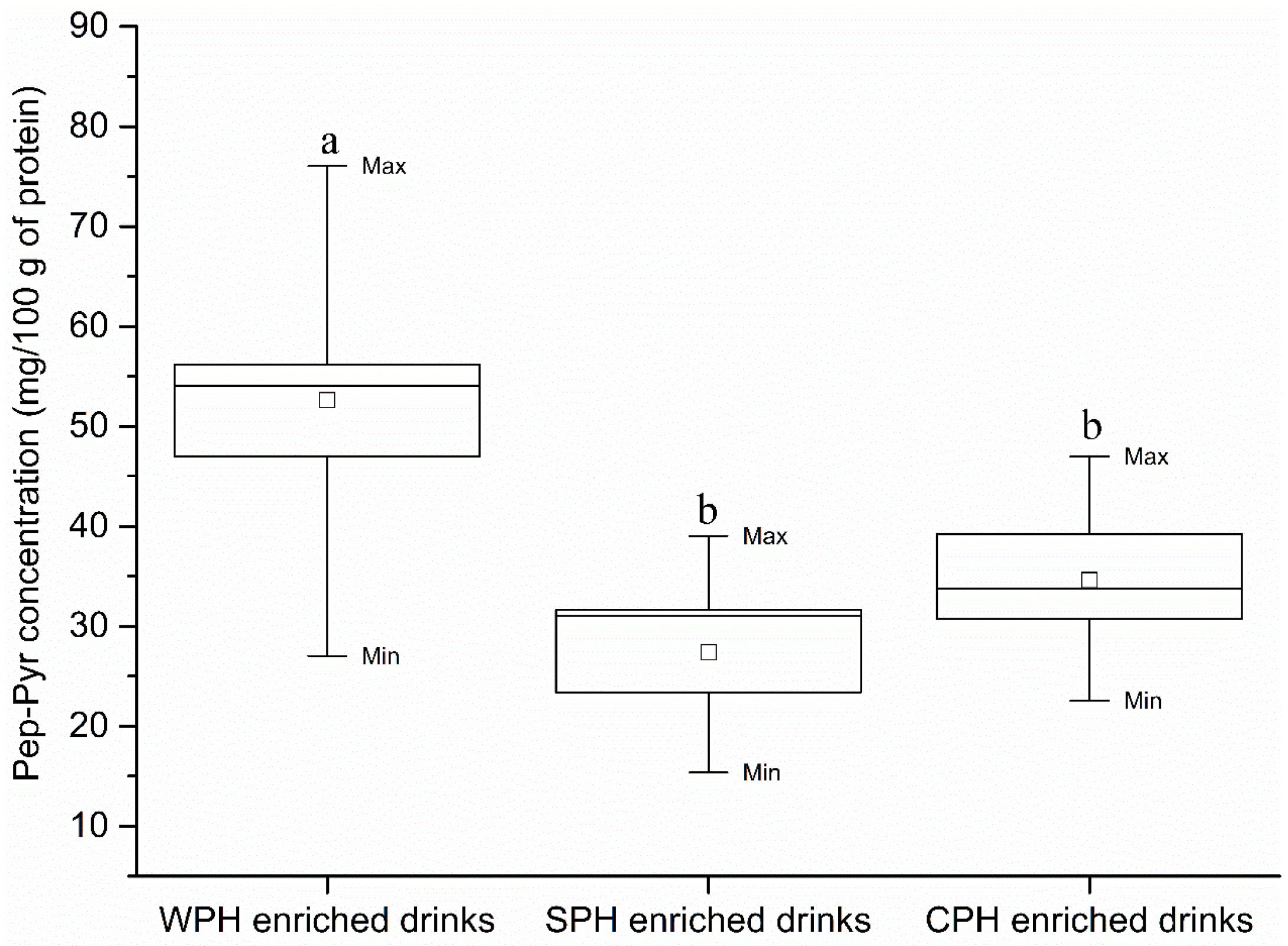
2.5. Quantification of Free-Pyr and Pep-Pyr in Commercial PEDs
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. PED Samples
4.3. Protein Content
4.4. Peptide Lengths Measurement
4.5. Total Reducing Sugar Content Measurement
4.6. Optimization of Derivatization Procedure
4.7. Optimization of SPE Procedure
- (a)
- The cartridge was preconditioned with 4 mL of methanol, and equilibrated with 4 mL of water before loading the sample.
- (b)
- One milliliter of sample was then applied to the cartridge, followed by washing the cartridge with 1 to 7 mL of water.
- (c)
- Finally, the target compounds were eluted from the cartridge with 1 to 7 mL of acetonitrile, and the eluent was evaporated to dryness at 50 °C by pressure blowing concentrator.
- (d)
- The dried residue was then dissolved in 1 mL of HPLC eluent: 0.1% formic acid in water containing 15% (v/v) acetonitrile.
4.8. Quantification of Free-Pyr and Pep-Pyr
4.9. Quantification of 3-DG
4.10. UPLC-UV-MS
4.11. Validation of the Method and Quantification of Pyrraline
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ames, J.M. Evidence against dietary advanced glycation endproducts being a risk to human health. Mol. Nutr. Food Res. 2007, 51, 1085–1090. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.M.; Hori, O.; Brett, J.; Yan, S.D.; Wautier, J.L.; Stern, D. Cellular receptors for advanced glycation end products. Implications for induction of oxidant stress and cellular dysfunction in the pathogenesis of vascular lesions. Arterioscler. Thromb. Vasc. Biol. 1994, 14, 1521–1528. [Google Scholar] [CrossRef]
- Vlassara, H. The age-receptor in the pathogenesis of diabetic complications. Diabetes/Metab. Res. Rev. 2001, 17, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Imani, F.; Horii, Y.; Suthanthiran, M.; Skolnik, E.Y.; Makita, Z.; Sharma, V.; Sehajpal, P.; Vlassara, H. Advanced glycosylation endproduct-specific receptors on human and rat T-lymphocytes mediate synthesis of interferon gamma: Role in tissue remodeling. J. Exp. Med. 1993, 178, 2165–2172. [Google Scholar] [CrossRef] [PubMed]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, S.; Baynes, J. Role of the Maillard reaction in diabetes mellitus and diseases of aging. Drugs Aging 1996, 9, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N. Advanced glycation endproducts—Role in pathology of diabetic complications. Diabetes Res. Clin. Pract. 2005, 67, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Makita, Z.; Radoff, S.; Rayfield, E.J.; Yang, Z.; Skolnik, E.; Delaney, V.; Friedman, E.A.; Cerami, A.; Vlassara, H. Advanced glycosylation end products in patients with diabetic nephropathy. N. Engl. J. Med. 1991, 325, 836–842. [Google Scholar] [CrossRef] [PubMed]
- Miyata, T.; van Ypersele de Strihou, C.; Kurokawa, K.; Baynes, J.W. Alterations in nonenzymatic biochemistry in uremia: Origin and significance of “carbonyl stress” in long-term uremic complications. Kidney Int. 1999, 55, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Luevano-Contreras, C.; Chapman-Novakofski, K. Dietary advanced glycation end products and aging. Nutrients 2010, 2, 1247–1265. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Gao, Q.-D.; Zhu, L.; Peppa, M.; He, C.; Vlassara, H. Oxidative stress-inducing carbonyl compounds from common foods: Novel mediators of cellular dysfunction. Mol. Med. 2002, 8, 337–346. [Google Scholar] [PubMed]
- Koschinsky, T.; He, C.-J.; Mitsuhashi, T.; Bucala, R.; Liu, C.; Buenting, C.; Heitmann, K.; Vlassara, H. Orally absorbed reactive glycation products (glycotoxins): An environmental risk factor in diabetic nephropathy. Proc. Natl. Acad. Sci. USA 1997, 94, 6474–6479. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Cai, W.; Crandall, J.; Goldberg, T.; Oberstein, R.; Dardaine, V.; Peppa, M.; Rayfield, E.J. Inflammatory mediators are induced by dietary glycotoxins, a major risk factor for diabetic angiopathy. Proc. Natl. Acad. Sci. USA 2002, 99, 15596–15601. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Peppa, M.; Cai, W. Restriction of dietary glycotoxins markedly reduces age toxins in renal failure patients. J. Am. Soc. Nephrol. 2003, 14, 728–731. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Peppa, M.; Cai, W.; Goldberg, T.; Lu, M.; Baliga, S.; Vassalotti, J.A.; Vlassara, H. Dietary glycotoxins correlate with circulating advanced glycation end product levels in renal failure patients. Am. J. Kidney Dis. 2003, 42, 532–538. [Google Scholar] [CrossRef]
- Odani, H.; Shinzato, T.; Matsumoto, Y.; Takai, I.; Nakai, S.; Miwa, M.; Iwayama, N.; Amano, I.; Maeda, K. First evidence for accumulation of protein-bound and protein-free pyrraline in human uremic plasma by mass spectrometry. Biochem. Biophys. Res. Commun. 1996, 224, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Henle, T. Maillard reaction of proteins and advanced glycation end products (ages) in food. In Process-Induced Food Toxicants: Occurrence, Formation, Mitigation, and Health Risks; Stadler, R.H., Lineback, D.R., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 215–242. [Google Scholar]
- Nakayama, T.; Hayase, F.; Kato, H. Formation of ε-(2-formyl-5-hydroxy-methyl-pyrrol-1-yl)-l-norleucine in the Maillard reaction between d-glucose and l-lysine. Agric. Biol. Chem. 1980, 44, 1201–1202. [Google Scholar]
- Chiang, G.H. High-performance liquid chromatographic determination of є-pyrrole lysine in processed food. J. Agric. Food Chem. 1988, 36, 506–509. [Google Scholar] [CrossRef]
- Henle, T.; Klostermeyer, H. Determination of protein-bound 2-amino-6-(2-formyl-1-pyrrolyl)-hexanoic acid (“pyrraline”) by ion exchange chromatography and photodiode array detection. Z. Lebensm. Unters. Forch. 1993, 196, 1–4. [Google Scholar] [CrossRef]
- Sengl, M.; Ledl, F.; Severin, T. Maillard-reaktion von rinderserumalbumin MIT glucose hochleistung-flüssigkeitschromatographischer nachweis des 2-formyl-5-(hydroxymethyl)pyrrol-1-norleucins NACH alkalischer hydrolyse. J. Chromatogr. A 1989, 463, 119–125. [Google Scholar] [CrossRef]
- Förster, A.; Kühne, Y.; Henle, T. Studies on absorption and elimination of dietary Maillard reaction products. Ann. N. Y. Acad. Sci. 2005, 1043, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Hellwig, M.; Geissler, S.; Peto, A.; Knütter, I.; Brandsch, M.; Henle, T. Transport of free and peptide-bound pyrraline at intestinal and renal epithelial cells. J. Agric. Food Chem. 2009, 57, 6474–6480. [Google Scholar] [CrossRef] [PubMed]
- Manninen, A. Protein hydrolysates in sports nutrition. Nutr. Metab. 2009, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Clemente, A. Enzymatic protein hydrolysates in human nutrition. Trends Food Sci. Technol. 2000, 11, 254–262. [Google Scholar] [CrossRef]
- Manninen, A.H. Protein hydrolysates in sports and exercise: A brief review. J. Sports Sci. Med. 2004, 3, 60–63. [Google Scholar] [PubMed]
- Pasquale, D.; Mauro, G. Amino Acids and Proteins for the Athlete: The Anabolic Edge, 2nd ed.; CRC Press: New York, NY, USA, 2007. [Google Scholar]
- Ozuna, C.; Paniagua-Martínez, I.; Castaño-Tostado, E.; Ozimek, L.; Amaya-Llano, S.L. Innovative applications of high-intensity ultrasound in the development of functional food ingredients: Production of protein hydrolysates and bioactive peptides. Food Res. Int. 2015, 77, 685–696. [Google Scholar] [CrossRef]
- Jain, S.; Anal, A.K. Optimization of extraction of functional protein hydrolysates from chicken egg shell membrane (ESM) by ultrasonic assisted extraction (UAE) and enzymatic hydrolysis. LWT Food Sci. Technol. 2016, 69, 295–302. [Google Scholar] [CrossRef]
- Degen, J.; Hellwig, M.; Henle, T. 1,2-dicarbonyl compounds in commonly consumed foods. J. Agric. Food Chem. 2012, 60, 7071–7079. [Google Scholar] [CrossRef] [PubMed]
- Revel, G.D.; Pripis-Nicolau, L.; Barbe, J.-C.; Bertrand, A. The detection of α-dicarbonyl compounds in wine by formation of quinoxaline derivatives. J. Sci. Food Agric. 2000, 80, 102–108. [Google Scholar] [CrossRef]
- Pfeifer, Y.V.; Haase, P.T.; Kroh, L.W. Reactivity of thermally treated α-dicarbonyl compounds. J. Agric. Food Chem. 2013, 61, 3090–3096. [Google Scholar] [CrossRef] [PubMed]
- Kocadağlı, T.; Žilić, S.; Taş, N.G.; Vančetović, J.; Dodig, D.; Gökmen, V. Formation of α-dicarbonyl compounds in cookies made from wheat, hull-less barley and colored corn and its relation with phenolic compounds, free amino acids and sugars. Eur. Food Res. Technol. 2015, 242, 51–60. [Google Scholar] [CrossRef]
- Lucci, P.; Pacetti, D.; Frega, N.G.; Núñez, O. Current trends in sample treatment techniques for environmental and food analysis. In Chromatography—The Most Versatile Method of Chemical Analysis; Calderon, L.D.A., Ed.; InTech: Rijeka, Croatia, 2012; pp. 127–164. [Google Scholar]
- Henle, T.; Walter, A.; Klostermeyer, H. Simultaneous determination of protein-bound Maillard products by ion exchange chromatography and photodiode array detection. In Maillard Reactions in Chemistry, Food, and Health; Labuza, T.P., Monnier, V.M., Baynes, J., O’Brien, J., Baynes, J.W., Eds.; Woodhead Publishing Limited: Cambridge, UK, 1994; pp. 195–200. [Google Scholar]
- Resmini, P.; Pellegrino, L. Occurence of protein-bound lysylpyrrolaldehyde in dried pasta. Cereal Chem. 1994, 71, 254–262. [Google Scholar]
- Wellner, A.; Huettl, C.; Henle, T. Formation of Maillard reaction products during heat treatment of carrots. J. Agric. Food Chem. 2011, 59, 7992–7998. [Google Scholar] [CrossRef] [PubMed]
- Li-Chan, E.C.Y. Bioactive peptides and protein hydrolysates: Research trends and challenges for application as nutraceuticals and functional food ingredients. Curr. Opin. Food Sci. 2015, 1, 28–37. [Google Scholar] [CrossRef]
- Anet, E.F.L.J. Mechanism of formation of 3-deoxyglycosuloses. Tetrahedron Lett. 1968, 9, 3525–3528. [Google Scholar] [CrossRef]
- Anet, E.F.L.J. 3-deoxyglycosuloses (3-deoxyglycosones) and the degradation of carbohydrates. Adv. Carbohydr. Chem. 1963, 19, 181–218. [Google Scholar]
- Smuda, M.; Glomb, M.A. Fragmentation pathways during Maillard-induced carbohydrate degradation. J. Agric. Food Chem. 2013, 61, 10198–10208. [Google Scholar] [CrossRef] [PubMed]
- Thornalley, P.J.; Langborg, A.; Minhas, H.S. Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem. J. 1999, 344, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Rufián-Henares, J.Á.; Guerra-Hernández, E.; García-Villanova, B. Pyrraline content in enteral formula processing and storage and model systems. Eur. Food Res. Technol. 2004, 219, 42–47. [Google Scholar] [CrossRef]
- Zhao, M.-M.; Zeng, X.-F.; Cui, C.; Wang, J.-S. Degradation of chicken protein hydrolysate peptides in Maillard reaction with glucose. J. Sichuan Univ. (Eng. Sci. Ed.) 2007, 39, 77–81. [Google Scholar]
- Nursten, H. The Maillard Reaction: Chemistry, Biochemistry and Implications; The Royal Society of Chemistry: London, UK, 2005; pp. 13–18. [Google Scholar]
- Lan, X.; Liu, P.; Xia, S.; Jia, C.; Mukunzi, D.; Zhang, X.; Xia, W.; Tian, H.; Xiao, Z. Temperature effect on the non-volatile compounds of Maillard reaction products derived from xylose-soybean peptide system: Further insights into thermal degradation and cross-linking. Food Chem. 2010, 120, 967–972. [Google Scholar] [CrossRef]
- De Kok, P.M.; Rosing, E.A. Reactivity of peptides in the Maillard reaction. In Thermally Generated Flavors; Parliment, T.H., Morello, M.J., McGorrin, R.J., Eds.; American Chemical Society: Washington, DC, USA, 1993; pp. 158–179. [Google Scholar]
- Mennella, C.; Visciano, M.; Napolitano, A.; del Castillo, M.D.; Fogliano, V. Glycation of lysine-containing dipeptides. J. Pept. Sci. 2006, 12, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Morales, F.J.; van Boekel, M.A.J.S. Formation of lysylpyrraline in heated sugar-casein solutions. Neth. Milk Dairy J. 1996, 50, 347–370. [Google Scholar]
- Siemensma, A.D.; Weijer, W.J.; Bak, H.J. The importance of peptide lengths in hypoallergenic infant formulae. Trends Food Sci. Technol. 1993, 4, 16–21. [Google Scholar] [CrossRef]
- Hewick, R.M.; Hunkapiller, M.W.; Hood, L.E.; Dreyer, W.J. A gas-liquid solid phase peptide and protein sequenator. J. Biol. Chem. 1981, 256, 7990–7997. [Google Scholar] [PubMed]
- Schuster, R. Determination of amino acids in biological, pharmaceutical, plant and food samples by automated precolumn derivatization and high-performance liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1988, 431, 271–284. [Google Scholar] [CrossRef]
- Schägger, H.; von Jagow, G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 1987, 166, 368–379. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Hellwig, M.; Henle, T. Release of pyrraline in absorbable peptides during simulated digestion of casein glycated by 3-deoxyglucosone. Eur. Food Res. Technol. 2013, 237, 47–55. [Google Scholar] [CrossRef]
| RSD a | RSD a | RSD a | RSD a | ||||||
|---|---|---|---|---|---|---|---|---|---|
| pyrraline | μg/mL b | 63.5 | 34.6 | 10.0 | 0.5 | ||||
| Recovery | 98.5% | 2.5% | 95.4% | 2.3% | 94.1% | 4.8% | 93.2% | 3.4% | |
| 3-DG | μg/mL c | 48.6 | 26.3 | 10.0 | 0.5 | ||||
| Recovery | 99.2% | 5.6% | 96.3% | 6.5% | 95.6% | 5.3% | 94.7% | 6.3% |
| Sample | Peptide Source | Free-Form Pyrraline (mg/100 g of Protein) | Peptide Bound Pyrraline (mg/100 g Protein) | Total Pyrraline (mg/100 g of Protein) |
|---|---|---|---|---|
| Group 1 | ||||
| PED1 | WPH | nd | 45.23 ± 1.95 | 45.23 ± 1.95 |
| PED2 | WPH | 5.60 ± 1.15 | 76.05 ± 0.60 | 81.70 ± 0.38 |
| PED3 | WPH | tr | 52.65 ± 2.30 | 52.65 ± 2.30 |
| PED4 | WPH | nd | 59.15 ± 1.58 | 59.15 ± 1.58 |
| PED5 | WPH | tr | 56.10 ± 0.30 | 56.10 ± 0.30 |
| PED6 | WPH | 7.75 ± 0.75 | 27.03 ± 0.58 | 34.78 ± 1.33 |
| PED7 | WPH | nd | 56.18 ± 0.35 | 56.18 ± 0.35 |
| PED8 | WPH | 3.25 ± 0.23 | 46.95 ± 0.55 | 50.20 ± 2.55 |
| PED9 | WPH | 2.95 ± 0.05 | 54.08 ± 0.40 | 57.08 ± 0.83 |
| Median | 54.08 | 56.10 | ||
| Average Percentage | 3.99% | 96.01% | 100% | |
| Group 2 | ||||
| PED10 | SPH | tr | 39.03 ± 1.80 | 39.03 ± 1.80 |
| PED11 | SPH | nd | 16.23 ± 1.80 | 16.23 ± 1.80 |
| PED12 | SPH | tr | 31.63 ± 1.98 | 31.63 ± 1.98 |
| PED13 | SPH | 3.00 ± 0.20 | 15.35 ± 0.15 | 18.35 ± 1.10 |
| PED14 | SPH | 3.05 ± 0.30 | 23.40 ± 0.53 | 26.53 ± 1.53 |
| PED15 | SPH | nd | 31.00 ± 1.28 | 31.00 ± 1.28 |
| PED16 | SPH | nd | 31.43 ± 1.70 | 31.43 ± 1.70 |
| PED17 | SPH | 2.53 ± 0.10 | 27.08 ± 0.08 | 29.60 ± 2.58 |
| PED18 | SPH | tr | 31.65 ± 0.60 | 31.65 ± 0.60 |
| Median | 31.00 | 31.00 | ||
| Average Percentage | 3.39% | 96.61% | 100% | |
| Group 3 | ||||
| PED19 | CPH | 3.35 ± 0.80 | 31.18 ± 0.60 | 34.58 ± 1.78 |
| PED20 | CPH | tr | 30.70 ± 1.00 | 30.70 ± 1.00 |
| PED21 | CPH | nd | 33.73 ± 1.53 | 33.73 ± 1.53 |
| PED22 | CPH | nd | 35.38 ± 1.30 | 35.38 ± 1.30 |
| PED23 | CPH | nd | 29.65 ± 1.30 | 29.65 ± 1.30 |
| PED24 | CPH | tr | 46.98 ± 1.73 | 46.98 ± 1.73 |
| PED25 | CPH | 6.13 ± 0.28 | 22.55 ± 0.30 | 28.70 ± 0.73 |
| PED26 | CPH | nd | 39.23 ± 2.05 | 39.23 ± 2.05 |
| PED27 | CPH | nd | 42.18 ± 2.65 | 42.18 ± 2.65 |
| Median | 33.73 | 34.58 | ||
| Average Percentage | 2.98% | 97.02% | 100% |
| Group 1 (WPH) | Group 2 (SPH) | Group3 (CPH) | |
|---|---|---|---|
| Protein content (g/100 mL) | 4.0 ± 0.3 a | 3.6 ± 0.6 a | 3.9 ± 0.2 a |
| Total reducing sugar content (g/1000 mL) | 82.2 ± 5.0 a | 85.6 ± 3.6 a | 84.5 ± 6.3 a |
| Distribution of peptide lengths (%) | |||
| 1 (free amino acids) | 4.2 ± 0.3 a | 3.2 ± 1.1 b | 4.8 ± 1.4 a |
| 2–5 | 80.3 ± 1.0 a | 70.0 ± 2.0 b | 76.3 ± 1.5 c |
| 6–10 | 8.2 ± 1.2 a | 12.4 ± 1.3 b | 9.0 ± 0.8 a |
| 11–15 | 5.1 ± 0.9 a | 7.3 ± 5.8 a | 6.7 ± 0.2 a |
| Essential Amino Acid | Whey Protein | Soy Protein | Collagen Protein |
|---|---|---|---|
| Ile | 5.5 | 4.9 | 1.7 |
| Leu | 14.2 | 8.2 | 3.4 |
| Lys | 10.2 | 6.3 | 4.1 |
| Met | 2.4 | 1.3 | 0.5 |
| Phe | 3.8 | 5.2 | 2.0 |
| Thr | 5.5 | 3.8 | 2.1 |
| Trp | 2.3 | 1.3 | na |
| Val | 5.9 | 5.0 | 3.0 |
| Total | 42.7 | 36.0 | 16.8 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Z.; Li, L.; Qi, H.; Zhang, X.; Xu, Z.; Li, B. Determination of Free-Form and Peptide Bound Pyrraline in the Commercial Drinks Enriched with Different Protein Hydrolysates. Int. J. Mol. Sci. 2016, 17, 1053. https://doi.org/10.3390/ijms17071053
Liang Z, Li L, Qi H, Zhang X, Xu Z, Li B. Determination of Free-Form and Peptide Bound Pyrraline in the Commercial Drinks Enriched with Different Protein Hydrolysates. International Journal of Molecular Sciences. 2016; 17(7):1053. https://doi.org/10.3390/ijms17071053
Chicago/Turabian StyleLiang, Zhili, Lin Li, Haiping Qi, Xia Zhang, Zhenbo Xu, and Bing Li. 2016. "Determination of Free-Form and Peptide Bound Pyrraline in the Commercial Drinks Enriched with Different Protein Hydrolysates" International Journal of Molecular Sciences 17, no. 7: 1053. https://doi.org/10.3390/ijms17071053
APA StyleLiang, Z., Li, L., Qi, H., Zhang, X., Xu, Z., & Li, B. (2016). Determination of Free-Form and Peptide Bound Pyrraline in the Commercial Drinks Enriched with Different Protein Hydrolysates. International Journal of Molecular Sciences, 17(7), 1053. https://doi.org/10.3390/ijms17071053







