Human Saliva Collection Devices for Proteomics: An Update
Abstract
:1. Introduction
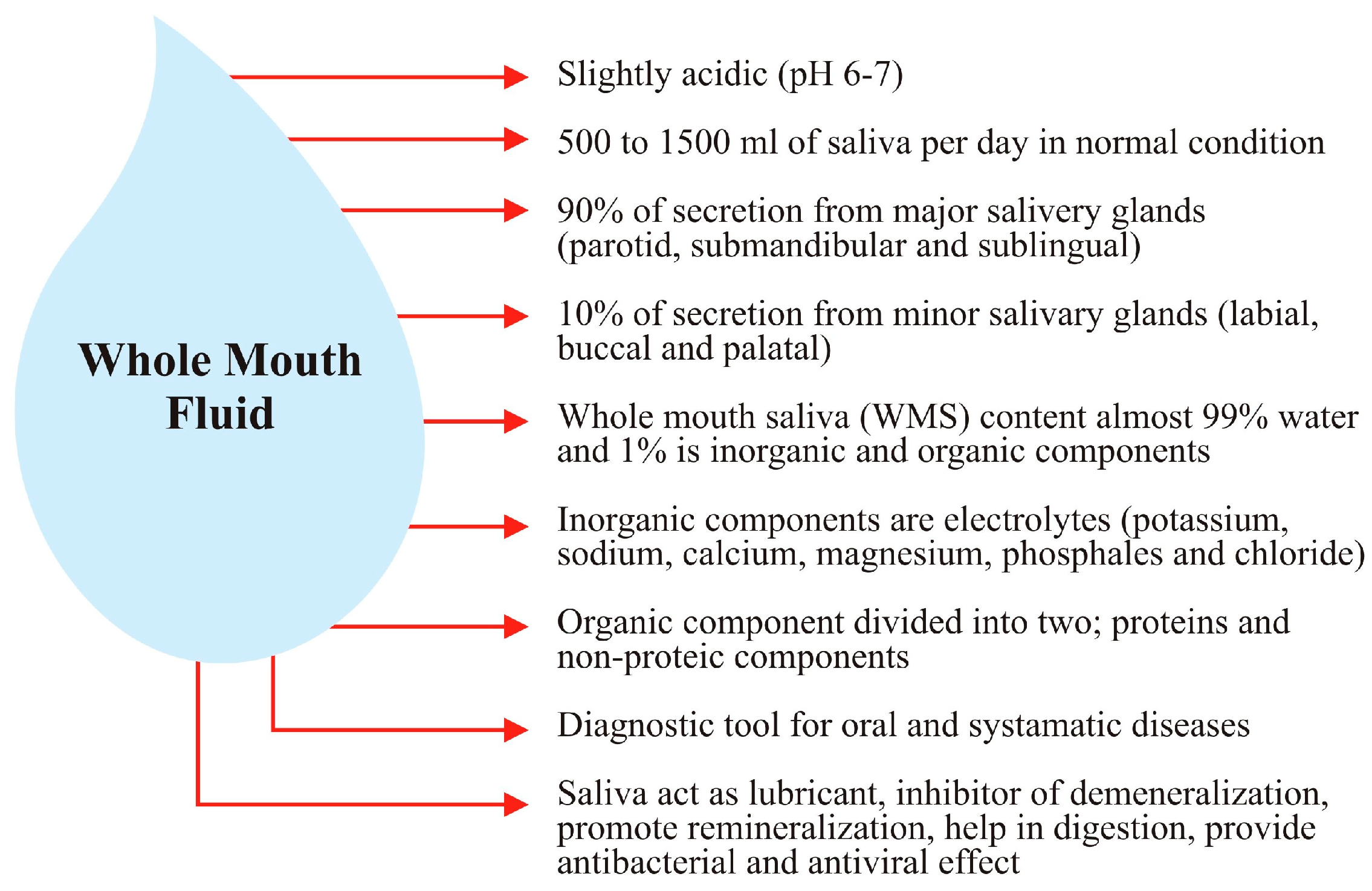
2. Human Salivary Proteomics
- Non-invasive
- Simple collection protocols
- Non-infectious sample
- Easily disposal
- Easily transportable
- Cost effective
- Not subject to cultural and religious “taboos”
- Safe and effective
- Higher patient compliance.
3. Human Saliva Collection Devices
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Su, S.-B.; Chuen, T.; Poon, W.; Thongboonkerd, V. Human Body Fluid. BioMed Res. Int. 2013, 2013, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Gstaiger, M.; Aebersold, R. Applying mass spectrometry-based proteomics to genetics, genomics and network biology. Nat. Rev. Genet. 2009, 10, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Horgan, R.; Kenny, L. “Omic”technologies: Genomics, transcriptomics, proteomics and metabolomics. Obstet. Gynaecol. 2011, 13, 189–195. [Google Scholar] [CrossRef]
- Ai, J.; Smith, B.; Wong, D.T. Saliva ontology: An ontology-based framework for a Salivaomics Knowledge Base. BMC Bioinform. 2010, 11. [Google Scholar] [CrossRef] [PubMed]
- Viet, C.T.; Schmidt, B.L. Methylation array analysis of preoperative and postoperative saliva DNA in oral cancer patients. Cancer Epidemiol. Biomark. Prev. 2008, 17, 3603–3611. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhou, X.; John, M.A.R.S.; Wong, D.T.W. RNA profiling of cell-free saliva using microarray technology. J. Dent. Res. 2004, 83, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xiao, H.; Zhou, H.; Santiago, S.; Lee, J.M.; Garon, E.B.; Yang, J.; Brinkmann, O.; Yan, X.; Akin, D.; et al. Development of transcriptomic biomarker signature in human saliva to detect lung cancer. Cell. Mol. Life Sci. 2012, 69, 3341–3350. [Google Scholar] [CrossRef] [PubMed]
- Yakob, M.; Fuentes, L.; Wang, M.B.; Abemayor, E.; Wong, D.T.W. Salivary biomarkers for detection of oral squamous cell carcinoma: current state and recent advances. Curr. Oral Health Rep. 2014, 1, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-H.; Kim, J.H.; Zhou, H.; Kim, B.W.; Wong, D.T. Salivary transcriptomic biomarkers for detection of ovarian cancer: For serous papillary adenocarcinoma. J. Mol. Med. 2012, 90, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Gao, P.; Cheng, F.; Wang, X.; Duan, Y. Measurement of salivary metabolite biomarkers for early monitoring of oral cancer with ultra performance liquid chromatography–mass spectrometry. Talanta 2014, 119, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Gao, P.; Wang, X.; Duan, Y. Investigation and identification of potential biomarkers in human saliva for the early diagnosis of oral squamous cell carcinoma. Clin. Chim. Acta 2014, 427, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.; Abdallah, M.; Hamdan, N. Barrier Membranes for Periodontal Guided Tissue Regeneration Applications. In Handbook of Oral Biomaterials; Pan Stanford Publishing: Singapore, 2014; Volume 6, pp. 605–636. [Google Scholar]
- Khurshid, Z.; Zafar, M.S.; Najeeb, S.; Zohaib, S. Human saliva: A future diagnostic tool. Dent. Sci. 2015, 2, 260–265. [Google Scholar]
- Khurshid, Z.; Naseem, M.; Sheikh, Z.; Najeeb, S.; Shahab, S.; Zafar, M.S. Oral antimicrobial peptides: Types and role in the oral cavity. Saudi Pharm. J. 2015. [Google Scholar] [CrossRef]
- Zafar, M.S.; Khurshid, Z.; Almas, K. Oral tissue engineering progress and challenges. Tissue Eng. Regen. Med. 2015, 12, 387–397. [Google Scholar] [CrossRef]
- Garg, K.; Chandra, S.; Raj, V.; Fareed, W.; Zafar, M. Molecular and genetic aspects of odontogenic tumors: A review. Iran. J. Basic Med. Sci. 2015, 18, 529–536. [Google Scholar] [PubMed]
- Khurshid, Z.; Zohaib, S.; Najeeb, S.; Zafar, M.S.; Rehman, R.; Rehman, I.U. Advances of proteomic sciences in dentistry. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Latterich, M.; Abramovitz, M.; Leyland-Jones, B. Proteomics: New technologies and clinical applications. Eur. J. Cancer 2008, 44, 2737–2741. [Google Scholar] [CrossRef] [PubMed]
- Reymond, M.A. Proteomics of Human Bodyfluids: Principles, Methods, and Applications; Thongboonkerd, V., Ed.; Springer Science & Business Media: New York, NY, USA, 2008; Volume 2, 286p. [Google Scholar]
- Edgar, W.M. Saliva: Its secretion, composition and functions. Br. Dent. J. 1992, 172, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, W.L.; Moffa, E.B.; Mussi, M.C.M.; Machado, M.A. Zika virus infection spread through saliva—A truth or myth? Braz. Oral Res. 2016, 30. [Google Scholar] [CrossRef] [PubMed]
- Streckfus, C.F.; Bigler, L.R. Saliva as a diagnostic fluid. Oral Dis. 2002, 8, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Punyadeera, C.; Slowey, P.D. Saliva as an emerging biofluid for clinical diagnosis and applications of MEMS/NEMS in salivary diagnostics. Nanobiomater. Clin. Dent. 2012, 453–473. [Google Scholar] [CrossRef]
- Malamud, D. Saliva as a diagnostic fluid. Dent. Clin. N. Am. 2011, 55, 159–178. [Google Scholar] [CrossRef] [PubMed]
- Caswell, S.V.; Cortes, N.; Mitchell, K.; Liotta, L.; Petricoin, E.F. Development of Nanoparticle-Enabled Protein Biomarker Discovery: Implementation for Saliva-Based Traumatic Brain Injury Detection. In Advances in Salivary Diagnostics; Springer Berlin Heidelberg: Berlin, Germany; Heidelberg, Germany, 2015; pp. 121–129. [Google Scholar]
- Farnaud, S.J.C.; Kosti, O.; Getting, S.J.; Renshaw, D. Saliva: Physiology and diagnostic potential in health and disease. Sci. World J. 2010, 10, 434–456. [Google Scholar] [CrossRef] [PubMed]
- Sreebny, L.M. Saliva in health and disease: An appraisal and update. Int. Dent. J. 2000, 50, 140–161. [Google Scholar] [CrossRef] [PubMed]
- Aps, J.K.M.; Martens, L.C. Review: The physiology of saliva and transfer of drugs into saliva. Forensic Sci. Int. 2005, 150, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Dodds, M.W.J.; Johnson, D.A.; Yeh, C.-K. Health benefits of saliva: A review. J. Dent. 2005, 33, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Najeeb, S.; Mali, M.; Moin, S.F.; Raza, S.Q.; Zohaib, S.; Sefat, F.; Zafar, M.S. Histatin peptides: Pharmacological functions and its applications in dentistry. Saudi Pharm. J. 2016. [Google Scholar] [CrossRef]
- Huang, C.-M. Comparative proteomic analysis of human whole saliva. Arch. Oral Biol. 2004, 49, 951–962. [Google Scholar] [CrossRef] [PubMed]
- Amado, F.M.L.; Ferreira, R.P.; Vitorino, R. One decade of salivary proteomics: Current approaches and outstanding challenges. Clin. Biochem. 2013, 46, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Musso, D.; Roche, C.; Nhan, T.-X.; Robin, E.; Teissier, A.; Cao-Lormeau, V.-M. Detection of Zika virus in saliva. J. Clin. Virol. 2015, 68, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Besnard, M.; Lastère, S.; Teissier, A.; Cao-Lormeau, V.; Musso, D. Evidence of perinatal transmission of Zika virus, French Polynesia, December 2013 and February 2014. Eurosurveillance 2014, 19. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, J.; Lin, C.-C.; Abemayor, E.; Wang, M.B.; Wong, D.T.W. The emerging landscape of salivary diagnostics. Periodontology 2000 2016, 70, 38–52. [Google Scholar] [CrossRef] [PubMed]
- Bassim, C.W.; Ambatipudi, K.S.; Mays, J.W.; Edwards, D.A.; Swatkoski, S.; Fassil, H.; Baird, K.; Gucek, M.; Melvin, J.E.; Pavletic, S.Z. Quantitative salivary proteomic differences in oral chronic graft-versus-host disease. J. Clin. Immunol. 2012, 32, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Zafar, M.S.; Zohaib, S.; Najeeb, S.; Naseem, M. Green tea (Camellia Sinensis): Chemistry and oral health. Open Dent. J. 2016, 10. [Google Scholar] [CrossRef]
- Aebersold, R.; Mann, M. Mass spectrometry-based proteomics. Nature 2003, 422, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Rangé, H.; Léger, T.; Huchon, C.; Ciangura, C.; Diallo, D.; Poitou, C.; Meilhac, O.; Bouchard, P.; Chaussain, C. Salivary proteome modifications associated with periodontitis in obese patients. J. Clin. Periodontol. 2012, 39, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Huynh, A.H.S.; Veith, P.D.; McGregor, N.R.; Adams, G.G.; Chen, D.; Reynolds, E.C.; Ngo, L.H.; Darby, I.B. Gingival crevicular fluid proteomes in health, gingivitis and chronic periodontitis. J. Periodontal Res. 2015, 50, 637–649. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yu, Q.; Lin, Q.; Duan, Y. Emerging salivary biomarkers by mass spectrometry. Clin. Chim. Acta 2015, 438, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, W.W. Human saliva II. A technical procedure for calcium analysis. J. Dent. Res. 1934, 14, 425–434. [Google Scholar] [CrossRef]
- Chiappin, S.; Antonelli, G.; Gatti, R.; de Palo, E.F. Saliva specimen: A new laboratory tool for diagnostic and basic investigation. Clin. Chim. Acta 2007, 383, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Henson, B.S.; Wong, D.T. Collection, storage, and processing of saliva samples for downstream molecular applications. Methods Mol. Biol. 2010, 666, 21–30. [Google Scholar] [PubMed]
- Michishige, F.; Kanno, K.; Yoshinaga, S.; Hinode, D.; Takehisa, Y.; Yasuoka, S. Effect of saliva collection method on the concentration of protein components in saliva. J. Med. Investig. 2006, 53, 140–146. [Google Scholar] [CrossRef]
- Gröschl, M.; Rauh, M. Influence of commercial collection devices for saliva on the reliability of salivary steroids analysis. Steroids 2006, 71, 1097–1100. [Google Scholar] [CrossRef] [PubMed]
- Neyraud, E.; Sayd, T.; Morzel, M.; Dransfield, E. Proteomic analysis of human whole and parotid salivas following stimulation by different tastes. J. Proteome Res. 2006, 5, 2474–2480. [Google Scholar] [CrossRef] [PubMed]
- Carlson, A.J.; Crittenden, A.L. The relation of ptyalin concentration to the diet and to the rate of salivary secretion. Exp. Biol. Med. 1909, 7, 52–54. [Google Scholar] [CrossRef]
- Truelove, E.L.; Bixler, D.; Merritt, A.D. Simplified method for collection of pure submandibular saliva in large volumes. J. Dent. Res. 1967, 46, 1400–1403. [Google Scholar] [CrossRef] [PubMed]
- Kutscher, A.H.; Mandel, I.D.; Zegarelli, E.V.; Denning, C.; Eriv, A.; Ruiz, L.; Ellegood, K.; Phalen, J. A technique for collecting the secretion of minor salivary glands: I. Use of capillary tubes. J. Oral Ther. Pharmacol. 1967, 3, 391–392. [Google Scholar] [PubMed]
- Gröschl, M.; Köhler, H.; Topf, H.-G.; Rupprecht, T.; Rauh, M. Evaluation of saliva collection devices for the analysis of steroids, peptides and therapeutic drugs. J. Pharm. Biomed. Anal. 2008, 47, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Topkas, E.; Keith, P.; Dimeski, G.; Cooper-White, J.; Punyadeera, C. Evaluation of saliva collection devices for the analysis of proteins. Clin. Chim. Acta 2012, 413, 1066–1070. [Google Scholar] [CrossRef] [PubMed]
- Speicher, D.J.; Wanzala, P.; D’Lima, M.; Johnson, K.E.; Johnson, N.W. Detecting DNA viruses in oral fluids: Evaluation of collection and storage methods. Diagn. Microbiol. Infect. Dis. 2015, 82, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Slowey, P.; Miocevic, O.; Buck, R.; Laughlin, M.; Shirtcliff, E.A.; Rai, B. Salivary Biomarkers for Parkinson’s Disease: Statistical Analysis for Panel Selection. In Proceedings of the Second North American Saliva Symposium, Seattle, WA, USA, 5 December 2015.
- Sorsa, T.; Gursoy, U.K.; Nwhator, S.; Hernandez, M.; Tervahartiala, T.; Leppilahti, J.; Gursoy, M.; Könönen, E.; Emingil, G.; Pussinen, P.J.; et al. Analysis of matrix metalloproteinases, especially MMP-8, in gingival creviclular fluid, mouthrinse and saliva for monitoring periodontal diseases. Periodontol. 2000 2016, 70, 142–163. [Google Scholar] [CrossRef] [PubMed]
- Streckfus, C.F. Advances in Salivary Diagnostics; Springer Berlin Heidelberg: Berlin, Germany; Heidelberg, Germany, 2015. [Google Scholar]
- Mohamed, R.; Campbell, J.-L.; Cooper-White, J.; Dimeski, G.; Punyadeera, C. The impact of saliva collection and processing methods on CRP, IgE, and Myoglobin immunoassays. Clin. Transl. Med. 2012, 1, 19. [Google Scholar] [CrossRef] [PubMed]
- Shirtcliff, E.A.; Buck, R.L.; Laughlin, M.J.; Hart, T.; Cole, C.R.; Slowey, P.D. Salivary cortisol results obtainable within minutes of sample collection correspond with traditional immunoassays. Clin. Ther. 2015, 37, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Broadhurst, M.J.; Kelly, J.D.; Miller, A.; Semper, A.; Bailey, D.; Groppelli, E.; Simpson, A.; Brooks, T.; Hula, S.; Nyoni, W.; et al. ReEBOV antigen rapid test kit for point-of-care and laboratory-based testing for ebola virus disease: A field validation study. Lancet 2015, 386. [Google Scholar] [CrossRef]
- Elachola, H.; Gozzer, E.; Zhuo, J.; Memish, Z.A. A crucial time for public health preparedness: Zika virus and the 2016 Olympics, Umrah, and Hajj. Lancet 2016, 387, 630–632. [Google Scholar] [CrossRef]
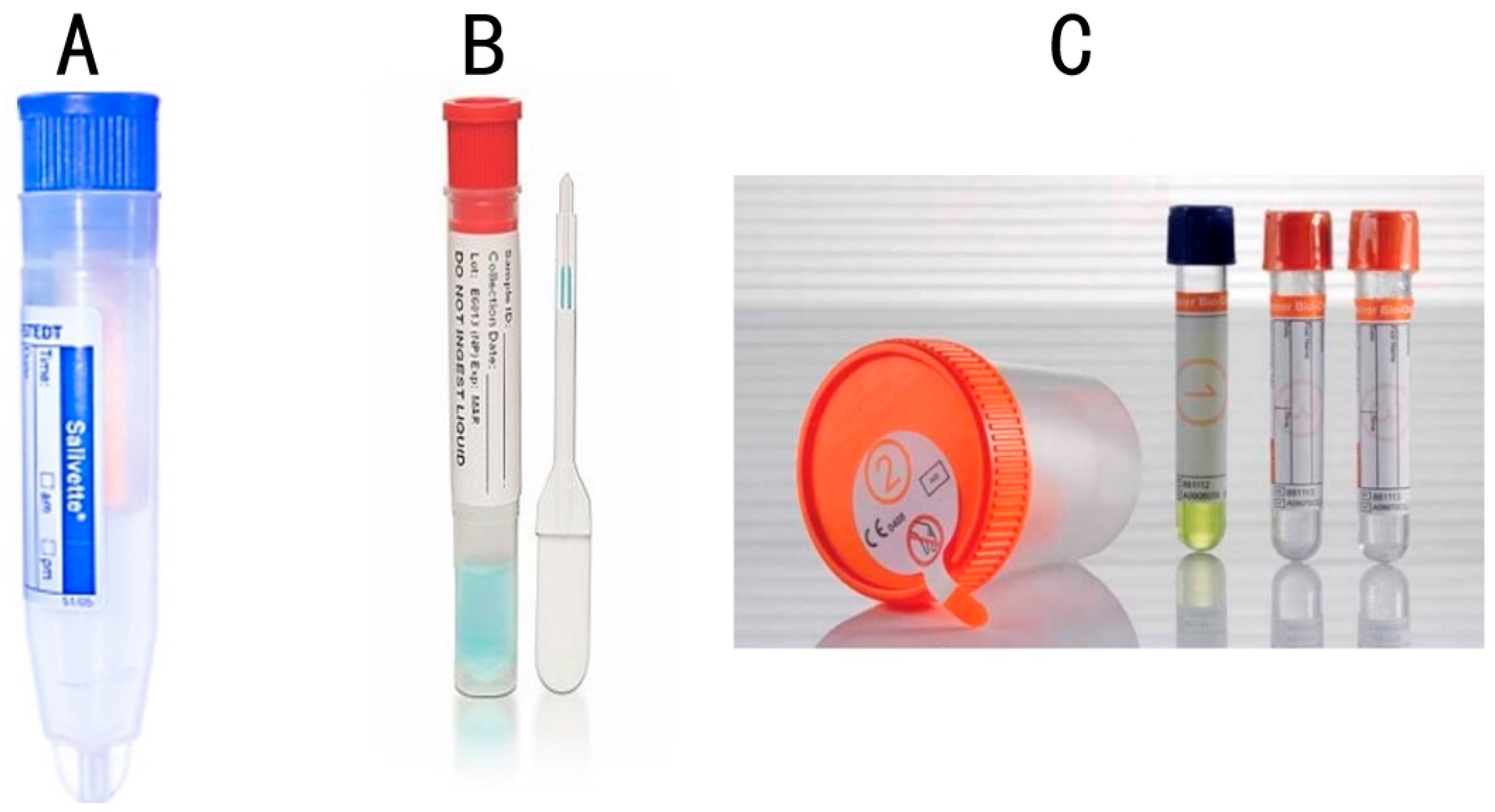
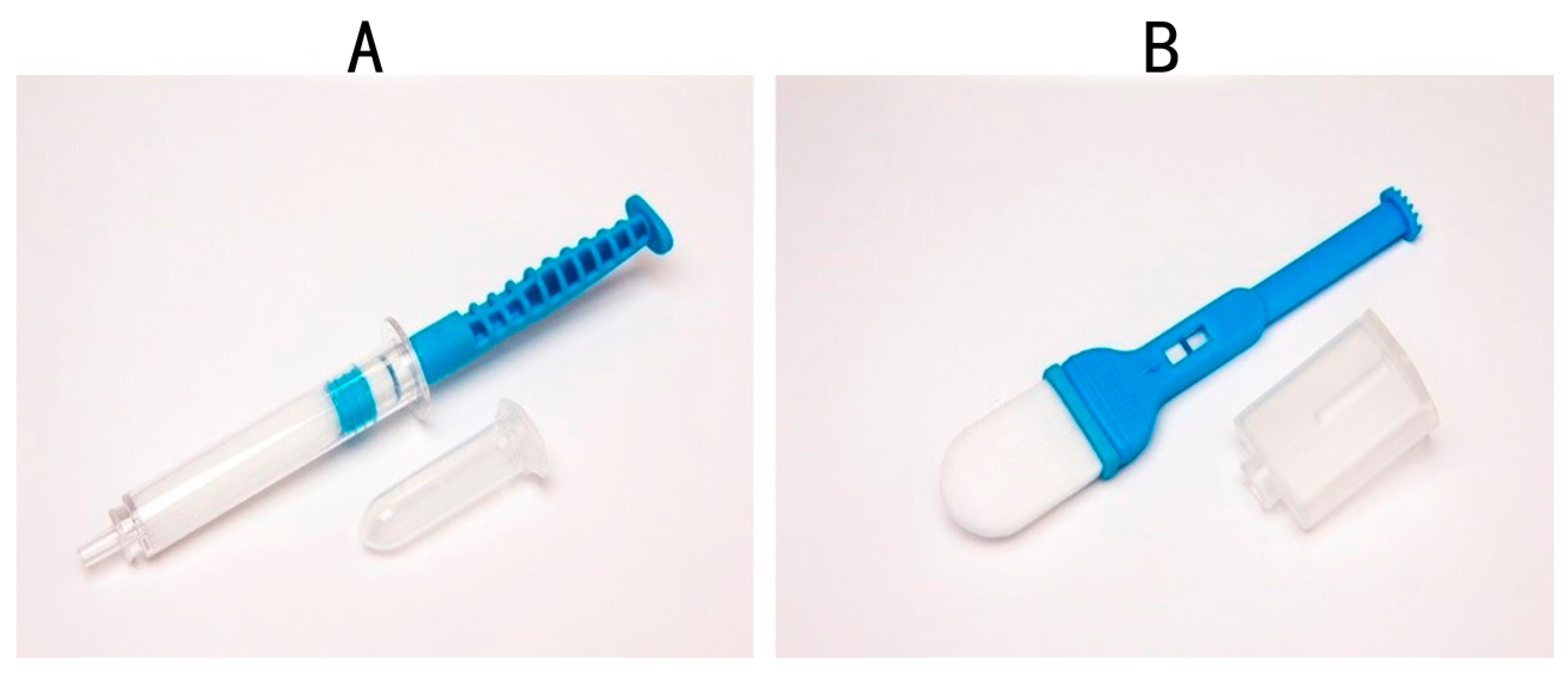
| Type of Whole Mouth Fluid | Method of Collection and Type of Collection Device | References |
|---|---|---|
| Whole Saliva (WS) | Patients should refrain from eating, drinking, and oral hygiene procedures for at least 1 h before saliva collection. (Optimum collection time is 8–10 a.m.). Before collection perform a 1 min oral rinse with distilled water and then after 5 min collect ~5 mL of saliva. Collected sample must be processed in the laboratory within 1 h. | [44] |
| Unstimulated Whole Saliva (USWS) | Passive drooling: In this method restrict oral movement and drain saliva from the lower lip into a plastic vial. | [45] |
| Spitting method: Instruct subject to spit into a collection vial. In this method 14 times more bacterial contamination is introduced into the sample. | ||
| Stimulated Whole Saliva (SWS) | For the stimulation of glands, chewing different things like natural gum, a piece of paraffin wax, citric acids, and powdered drink crystals have been used. | [46,47] |
| Parotid Gland | Method introduced by Carlson and Crittenden (1910). In this method a double chambered metallic cup with two outlet tubes is used. One end holds the cup in place using vacuum suction. The second half acts as a collection vehicle for saliva. Specimen collection can be enhanced by smearing citric acid (10%; 1 mL) on the dorsum of tongue every 30 s. Discard the first 1.5 mL of saliva prior to sample collection. | [48] |
| Submandibular/Sublingual Gland | Truelove, Bixler, and Merrit (1967) used a “V”-shaped collector. This method is similar to that for parotid gland collection, but in this case the initial 2 mL is discarded. | [49] |
| Minor Glands | Kutscher et al. (1967) used capillary tubes for collecting saliva from minor glands located at the everted surface of the lower lips. | [50] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khurshid, Z.; Zohaib, S.; Najeeb, S.; Zafar, M.S.; Slowey, P.D.; Almas, K. Human Saliva Collection Devices for Proteomics: An Update. Int. J. Mol. Sci. 2016, 17, 846. https://doi.org/10.3390/ijms17060846
Khurshid Z, Zohaib S, Najeeb S, Zafar MS, Slowey PD, Almas K. Human Saliva Collection Devices for Proteomics: An Update. International Journal of Molecular Sciences. 2016; 17(6):846. https://doi.org/10.3390/ijms17060846
Chicago/Turabian StyleKhurshid, Zohaib, Sana Zohaib, Shariq Najeeb, Muhammad Sohail Zafar, Paul D. Slowey, and Khalid Almas. 2016. "Human Saliva Collection Devices for Proteomics: An Update" International Journal of Molecular Sciences 17, no. 6: 846. https://doi.org/10.3390/ijms17060846
APA StyleKhurshid, Z., Zohaib, S., Najeeb, S., Zafar, M. S., Slowey, P. D., & Almas, K. (2016). Human Saliva Collection Devices for Proteomics: An Update. International Journal of Molecular Sciences, 17(6), 846. https://doi.org/10.3390/ijms17060846









