From Olive Fruits to Olive Oil: Phenolic Compound Transfer in Six Different Olive Cultivars Grown under the Same Agronomical Conditions
Abstract
:1. Introduction
2. Results and Discussion
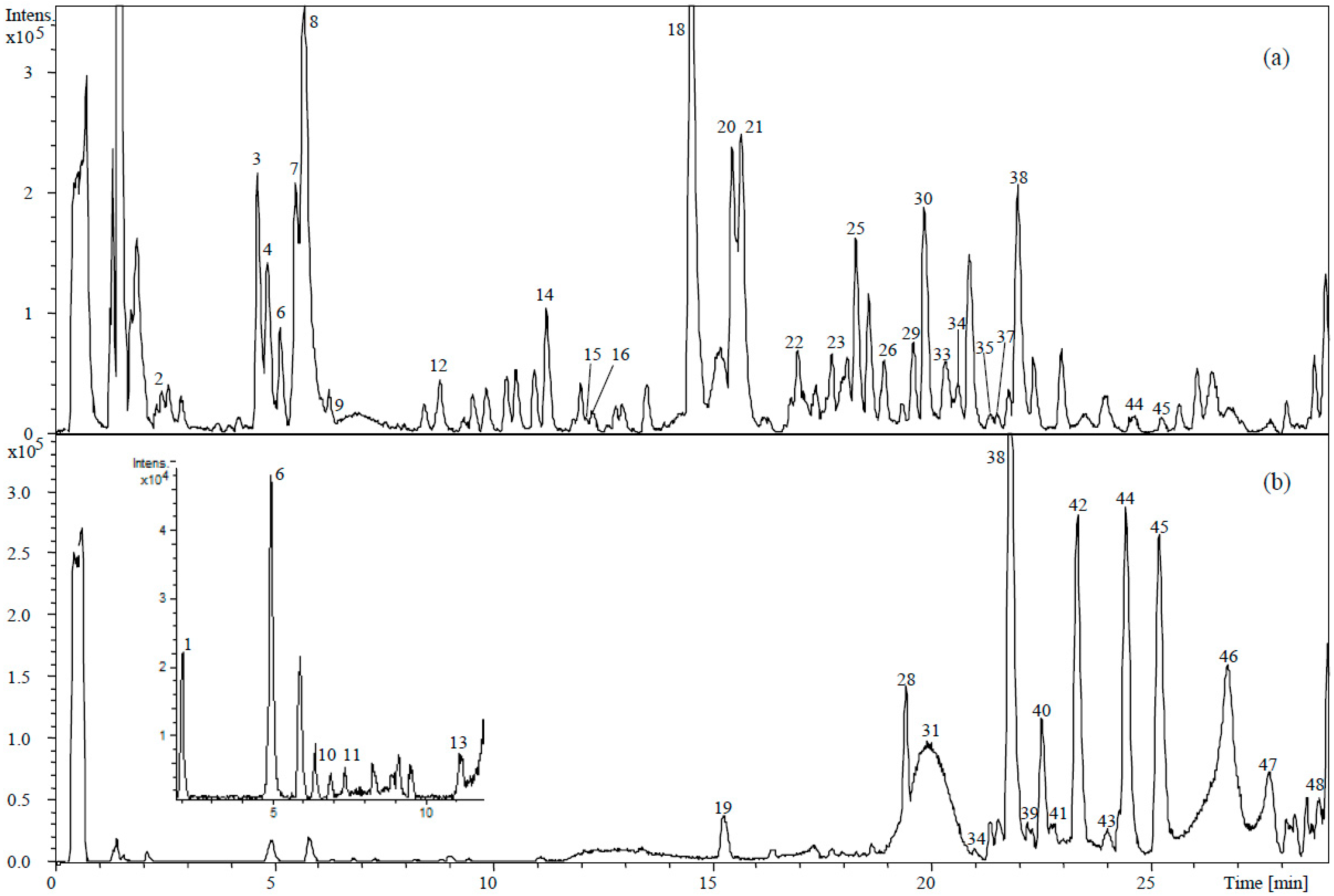
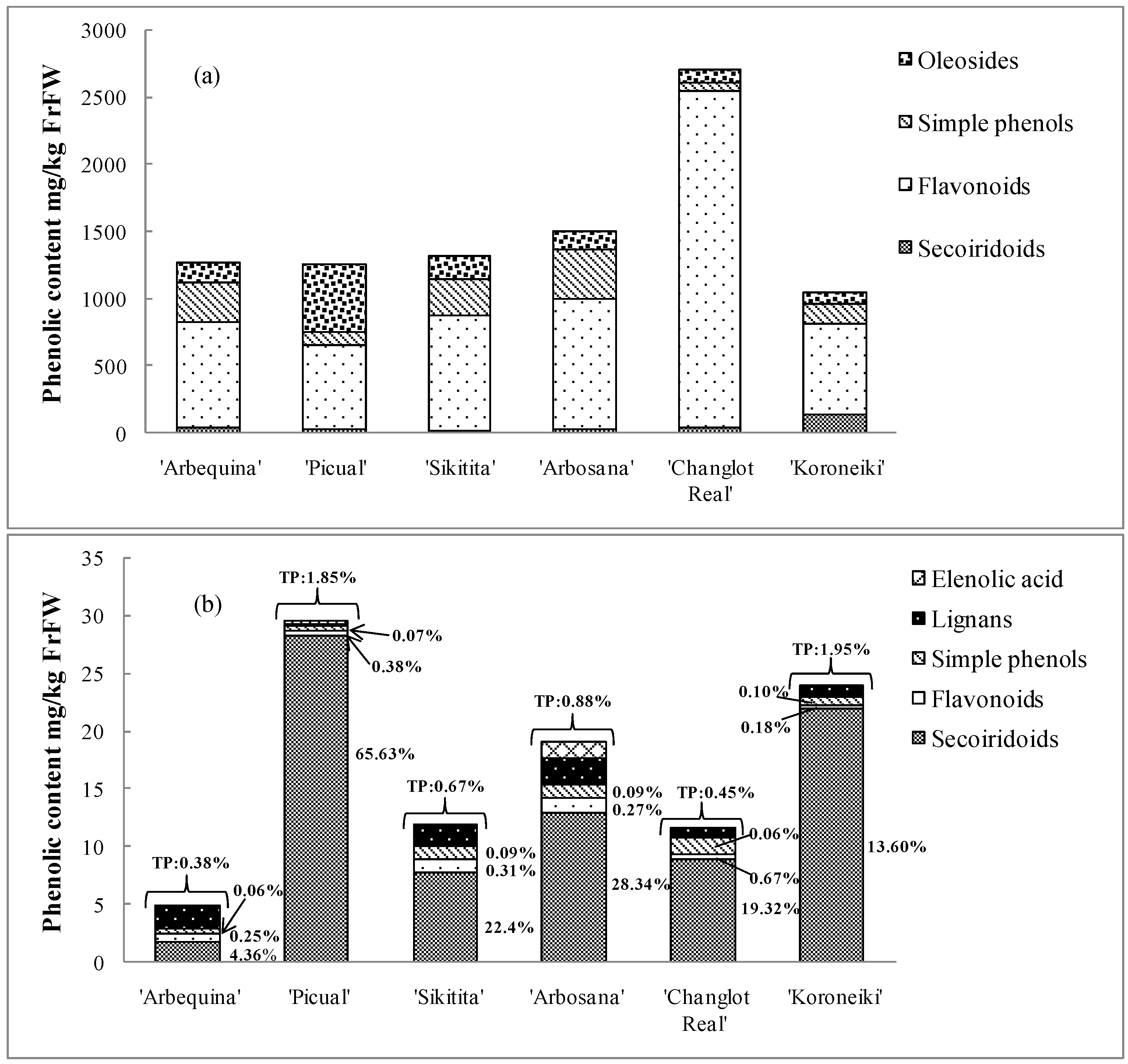
2.1. Quantitative Characterization of Phenolic Compounds
2.2. Transfer of Phenolic Compounds from Fruits to Oil
2.2.1. Qualitative Changes
2.2.2. Quantitative Changes
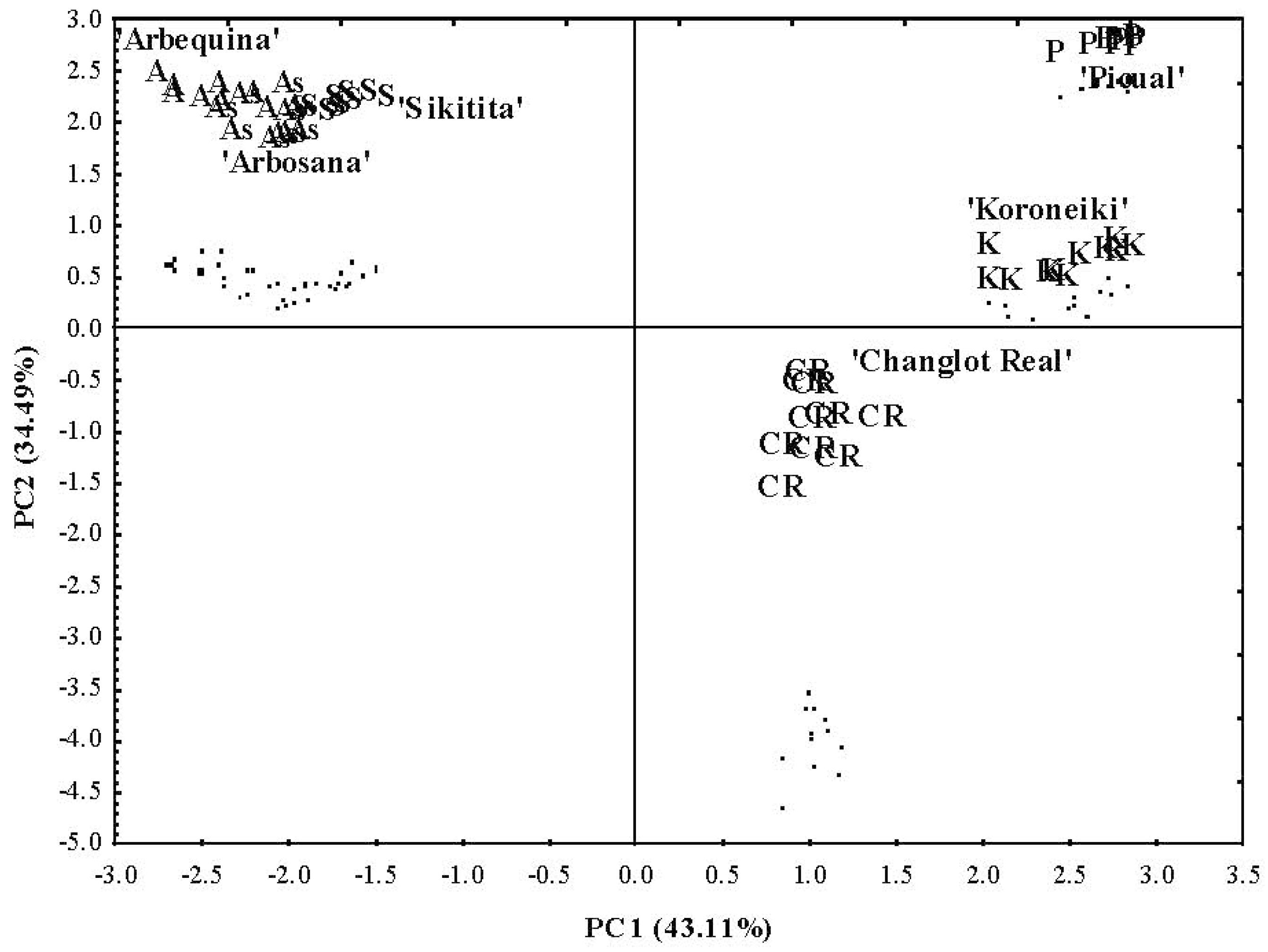
2.3. Chemometric Analysis
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Samples
3.3. Extraction of Phenolic Compounds from Olive Fruits and Oils
3.4. Determination of Phenolic Compounds by HPLC-DAD-TOF-MS
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Servili, M.; Sordini, B.; Esposto, S.; Urbani, S.; Veneziani, G.; di Maio, I.; Selvaggini, R.; Taticchi, A. Biological activities of phenolic compounds of extra virgin olive oil. Antioxidants 2014, 3, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Boskou, D.; Blekas, G.; Tsimidou, M. Phenolic compounds in olive oil and olives. Curr. Top. Nutraceutical Res. 2005, 3, 125–136. [Google Scholar]
- Clodoveo, M.L.; Durante, V.; La Notte, D. Working towards the development of innovative ultrasound equipment for the extraction of virgin olive oil. Ultrason. Sonochem. 2013, 20, 1261–1270. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Brenes, M.; García, P.; Romero, C.; Garrido, A. Phenolic content of commercial olive oils. Eur. Food Res. Technol. 2003, 216, 520–525. [Google Scholar]
- Bakhouche, A.; Lozano-Sánchez, J.; Beltrán-Debón, R.; Joven, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phenolic characterization and geographical classification of commercial Arbequina extra-virgin olive oils produced in southern Catalonia. Food Res. Int. 2013, 50, 401–408. [Google Scholar] [CrossRef]
- Dabbou, S.; Rjiba, I.; Nakbi, A.; Gazzah, N.; Issaoui, M.; Hammami, M. Compositional quality of virgin olive oils from cultivars introduced in Tunisian arid zones in comparison to Chemlali cultivars. Sci. Hortic. 2010, 124, 122–127. [Google Scholar] [CrossRef]
- Taamalli, A.; Arráez Román, D.; Zarrouk, M.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Classification of ‘Chemlali’ accessions according to the geographical area using chemometric methods of phenolic profiles analysed by HPLC-ESI-TOF-MS. Food Chem. 2012, 132, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Taamalli, A.; Gomez-Caravaca, A.M.; Zarrouk, M.; Segura-Carretero, A.; Fernandez-Gutierrez, A. Determination of apolar and minor polar compounds and other chemical parameters for the discrimination of six different varieties of Tunisian extra-virgin olive oil cultivated in their traditional growing area. Eur. Food Res. Technol. 2010, 231, 965–975. [Google Scholar] [CrossRef]
- Caruso, G.; Gucci, R.; Urbani, S.; Esposto, S.; Taticchi, A.; di Maio, I.; Selvaggini, R.; Servili, M. Effect of different irrigation volumes during fruit development on quality of virgin olive oil of cv. Frantoio. Agric. Water Manag. 2014, 134, 94–103. [Google Scholar] [CrossRef]
- Farinelli, D.; Tombesi, S. Performance and oil quality of ‘Arbequina’ and four Italian olive cultivars under super high density hedgerow planting system cultivated in central Italy. Sci. Hortic. 2015, 192, 97–107. [Google Scholar] [CrossRef]
- García, J.M.; Cuevas, M.V.; Fernández, J.E. Production and oil quality in ‘Arbequina’ olive (Olea europaea, L.) trees under two deficit irrigation strategies. Irrig. Sci. 2013, 31, 359–370. [Google Scholar] [CrossRef]
- Baccouri, B.; Zarrouk, W.; Baccouri, O.; Guerfel, M.; Nouairi, I.; Krichene, D.; Daoud, D.; Zarrouk, M. Composition, quality and oxidative stability of virgin olive oils from some selected wild olives (Olea europaea L. subsp. oleaster). Grasas y Aceites 2008, 59, 346–351. [Google Scholar] [CrossRef]
- Servili, M.; Esposto, S.; Fabiani, R.; Urbani, S.; Taticchi, A.; Mariucci, F.; Selvaggini, R.; Montedoro, G.F. Phenolic compounds in olive oil: Antioxidant, health and organoleptic activities according to their chemical structure. Inflammopharmacology 2009, 17, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Cerretani, L.; Bendini, A.; Fernández-Gutiérrez, A. Use of capillary electrophoresis with UV detection to compare the phenolic profiles of extra-virgin olive oils belonging to Spanish and Italian PDOs and their relation to sensorial properties. J. Sci. Food Agric. 2009, 89, 2144–2155. [Google Scholar] [CrossRef]
- Alagna, F.; Mariotti, R.; Panara, F.; Caporali, S.; Urbani, S.; Veneziani, G.; Esposto, S.; Taticchi, A.; Rosati, A.; Rao, R.; et al. Olive phenolic compounds: Metabolic and transcriptional profiling during fruit development. BMC Plant Biol. 2012, 12. [Google Scholar] [CrossRef] [PubMed]
- Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Lercker, G. Phenolic molecules in virgin olive oils: a survey of their sensory properties, health effects, antioxidant activity and analytical methods. An overview of the last decade. Molecules 2007, 12, 1679–1719. [Google Scholar] [CrossRef] [PubMed]
- Jerman Klen, T.; Mozetič Vodopivec, B. The fate of olive fruit phenols during commercial olive oil processing: Traditional press versus continuous two-and three-phase centrifuge. LWT-Food Sci. Technol. 2012, 49, 267–274. [Google Scholar] [CrossRef]
- Jerman Klen, T.; Wondra, A.G.; Vrhovšek, U.; Sivilotti, P.; Mozetič Vodopivec, B. Olive fruit phenols transfer, transformation, and partition trail during laboratory-scale olive oil processing. J. Agric. Food Chem. 2015, 63, 4570–4579. [Google Scholar] [CrossRef] [PubMed]
- Tura, D.; Gigliotti, C.; Pedò, S.; Failla, O.; Bassi, D.; Serraiocco, A. Influence of cultivar and site of cultivation on levels of lipophilic and hydrophilic antioxidants in virgin olive oils (Olea Europaea L.) and correlations with oxidative stability. Sci. Hortic. 2007, 112, 108–119. [Google Scholar] [CrossRef]
- Dağdelen, A.; Tümen, G.; Ozcan, M.M.; Dündar, E. Phenolics profiles of olive fruits (Olea europaea L.) and oils from ‘Ayvalık’, ‘Domat’ and ‘Gemlik’ varieties at different ripening stages. Food Chem. 2013, 136, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.; Robards, K. Phenolic compounds in olives. Analyst 1998, 123, 31R–44R. [Google Scholar] [CrossRef]
- Talhaoui, N.; Gómez-Caravaca, A.M.; Leon, L.; de la Rosa, R.; Fernandez-Gutierrez, A.; Segura-Carretero, A. Pattern of variation of fruit traits and phenol content in olive fruits from six different cultivars. J. Agric. Food Chem. 2015, 63, 10466–10476. [Google Scholar] [CrossRef] [PubMed]
- Artajo, L.-S.; Romero, M.-P.; Suárez, M.; Motilva, M.-J. Partition of phenolic compounds during the virgin olive oil industrial extraction process. Eur. Food Res. Technol. 2007, 225, 617–625. [Google Scholar] [CrossRef]
- Frankel, E.; Bakhouche, A.; Lozano-Sánchez, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Literature review on production process to obtain extra virgin olive oil enriched in bioactive compounds. Potential use of byproducts as alternative sources of polyphenols. J. Agric. Food Chem. 2013, 61, 5179–5188. [Google Scholar] [CrossRef] [PubMed]
- Sánchez de Medina, V.; Priego-Capote, F.; de Castro, M.D.L. Characterization of monovarietal virgin olive oils by phenols profiling. Talanta 2015, 132, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Servili, M.; Selvaggini, R.; Esposto, S.; Taticchi, A.; Montedoro, G.; Morozzi, G. Health and sensory properties of virgin olive oil hydrophilic phenols: Agronomic and technological aspects of production that affect their occurrence in the oil. J. Chromatogr. A 2004, 1054, 113–127. [Google Scholar] [CrossRef]
- Goulas, V.; Charisiadis, P.; Gerothanassis, I.; Manganaris, G. Classification, biotransformation and antioxidant activity of olive fruit biophenols: A review. Curr. Bioact. Compd. 2012, 8, 232–239. [Google Scholar] [CrossRef]
- Obied, H.K.; Karuso, P.; Prenzler, P.D.; Robards, K. Novel secoiridoids with antioxidant activity from Australian olive mill waste. J. Agric. Food Chem. 2007, 55, 2848–2853. [Google Scholar] [CrossRef] [PubMed]
- Brenes, M.; Hidalgo, F.J.; García, A.; Rios, J.J.; García, P.; Zamora, R.; Garrido, A. Pinoresinol and 1-acetoxypinoresinol, two new phenolic compounds identified in olive oil. J. Am. Oil Chem. Soc. 2000, 77, 715–720. [Google Scholar] [CrossRef]
- Oliveras López, M.J.; Innocenti, M.; Ieri, F.; Giaccherini, C.; Romani, A.; Mulinacci, N. HPLC/DAD/ESI/MS detection of lignans from Spanish and Italian Olea europaea L. fruits. J. Food Compos. Anal. 2008, 21, 62–70. [Google Scholar] [CrossRef]
- Benito, M.; Lasa, J.M.; Gracia, P.; Oria, R.; Abenoza, M.; Varona, L.; Sánchez-Gimeno, A.C. Olive oil quality and ripening in super-high-density ‘Arbequina’ orchard. J. Sci. Food Agric. 2013, 93, 2207–2220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodis, P.S.; Karathanos, V.T.; Mantzavinou, A. Partitioning of olive oil antioxidants between oil and water phases. J. Agric. Food Chem. 2002, 50, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, M.B. Water Use in Crop Production; Kirkham, M.B., Ed.; The Haworth Press: Philadelphia, PA, USA, 1999. [Google Scholar]
- Artajo, L.-S.; Romero, M.P.; Motilva, M.J. Transfer of phenolic compounds during olive oil extraction in relation to ripening stage of the fruit. J. Sci. Food Agric. 2006, 86, 518–527. [Google Scholar] [CrossRef]
- Bouaziz, M.; Jemai, H.; Khabou, W.; Sayadi, S. Oil content, phenolic profiling and antioxidant potential of Tunisian olive drupes. J. Sci. Food Agric. 2010, 90, 1750–1758. [Google Scholar] [CrossRef] [PubMed]
- Morelló, J.-R.; Romero, M.-P.; Motilva, M.-J. Effect of the maturation process of the olive fruit on the phenolic fraction of drupes and oils from ‘Arbequina’, ‘Farga’, and ‘Morrut’ Cultivars. J. Agric. Food Chem. 2004, 52, 6002–6009. [Google Scholar] [CrossRef] [PubMed]
- Rivas, A.; Sanchez-Ortiz, A.; Jimenez, B.; García-Moyano, J.; Lorenzo, M.L. Phenolic acid content and sensory properties of two Spanish monovarietal virgin olive oils. Eur. J. Lipid Sci. Technol. 2013, 115, 621–630. [Google Scholar] [CrossRef]
- Rotondi, A.; Alfei, B.; Magli, M.; Pannelli, G. Influence of genetic matrix and crop year on chemical and sensory profiles of Italian monovarietal extra-virgin olive oils. J. Sci. Food Agric. 2010, 90, 2641–2648. [Google Scholar] [CrossRef] [PubMed]
- Rallo, L.; Barranco, D.; de la Rosa, R.; León, L. ‘Chiquitita’ Olive. HortScience 2008, 43, 529–531. [Google Scholar]
- Garcia-Gonzalez, D.; Tena, N.; Aparicio, R. Quality characterization of the new virgin olive oil var. ‘Sikitita’ by phenols and volatile compounds. J. Agric. Food Chem. 2010, 58, 8357–8364. [Google Scholar] [CrossRef] [PubMed]
- Rallo, L.; Barranco, D.; Caballero, J.M.; del Río, C.; Martín, A.; Tous, J.; Trujillo, I. Variedades de Olivo en España; Rallo, L., Barranco, D., Caballero, J.M., del Río, C., Martín, A., Tous, J., Trujillo, I., Eds.; Mundi-Pren.; Junta de Andalucía, MAPA: Madrid, Spain, 2005. [Google Scholar]
- Talhaoui, N.; Gómez-Caravaca, A.M.; León, L.; de la Rosa, R.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Determination of phenolic compounds of ‘Sikitita’ olive leaves by HPLC-DAD-TOF-MS. Comparison with its parents ‘Arbequina’ and ‘Picual’ olive leaves. LWT-Food Sci. Technol. 2014, 58, 28–34. [Google Scholar] [CrossRef]

| Compounds a | Rt min | m/z | Formula | Phenolic Content (mg/kg FrFW or mg/kg oil) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ‘Arbequina’ Fruit Oil | ‘Picual’ Fruit Oil | ‘Sikitita’ Fruit Oil | ‘Arbosana’ Fruit Oil | ‘Changlot Real’ Fruit Oil | ‘Koroneiki’ Fruit Oil | |||||||||||
| 1 | Vanillin isomer a 3 | 2.09 | 151 | C8H8O3 | n.i. | 0.10 (0.01) | n.i. | 0.18 (0.01) | n.i. | 0.21 (0.02) | n.i. | 0.40 (0.01) | n.i. | 0.11 (0.01) | n.i. | 0.12 (0.01) |
| 2 | Hydroxytyrosol glucoside isomer a 3 | 2.31 | 315 | C14H20O8 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | 91.74 (7.53) | n.i. | n.i. | n.i. |
| 3 | Hydroxytyrosol glucoside isomer b 3 | 4.58 | 315 | C14H20O8 | n.i. | n.i. | n.i. | n.i. | 212.34 (23.07) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 4 | Hydroxytyrosol glucoside isomer c 3 | 4.8 | 315 | C14H20O8 | n.i. | n.i. | 79.68 (7.89) | n.i. | 56.37 (2.04) | n.i. | n.i. | n.i. | 440.58 (40.67) | n.i. | 96.80 (7.09) | n.i. |
| 5 | Hydroxytyrosol glucoside isomer d 3 | 4.82 | 315 | C14H20O8 | 257.39 (22.06) | n.i. | 58.32 (5.01) | n.i. | n.i. | n.i. | 276.71 (16.71) | n.i. | 433.30 (35.44) | n.i. | 114.74 (7.39) | n.i. |
| 6 | Hydroxytyrosol 3 | 5.14 | 153 | C8H10O3 | 62.91 (4.93) | 0.29 (0.01) | 107.97 (7.40) | 1.12 (0.04) | 85.16 (8.26) | 0.83 (0.07) | 73.41 (6.08) | 1.39 (0.07) | 73.53 (7.59) | 1.63 (0.713) | 61.61 (5.43) | 1.57 (0.11) |
| 7 | Oleoside derivative isomer a 4 | 5.67 | 407 | C17H28O11 | 20.22 (1.17) | n.i. | 12.26 (0.98) | n.i. | 19.80 (0.76) | n.i. | 24.33 (1.18) | n.i. | 23.36 (1.95) | n.i. | 17.67 (1.87) | n.i. |
| 8 | Oleoside derivative isomer b 4 | 6.26 | 407 | C17H28O11 | 57.93 (3.00) | n.i. | 231.21 (22.43) | n.i. | 72.07 (3.41) | n.i. | 71.31 (6.25) | n.i. | 63.80 (4.94) | n.i. | 52.48 (4.32) | n.i. |
| 9 | Tyrosol glucoside 3 | 6.59 | 299 | C14H20O7 | n.i. | n.i. | n.i. | n.i. | 46.27 (3.88) | n.i. | n.i. | n.i. | 615.18 (49.57) | n.i. | n.i. | n.i. |
| 10 | Vanillin isomer b 3 | 6.81 | 151 | C8H8O3 | n.i. | 0.022 (0.001) | n.i. | n.i. | n.i. | 0.040 (0.004) | n.i. | 0.028 (0.002) | n.i. | n.i. | n.i. | n.i. |
| 11 | Tyrosol 3 | 7.28 | 137 | C8H10O2 | n.i. | n.i. | n.i. | 1.99 (0.12) | n.i. | 2.28 (0.24) | n.i. | 2.06 (0.15) | n.i. | 10.25 (0.96) | n.i. | 3.46 (0.12) |
| 12 | p-coumaric acid 3 | 8.73 | 163 | C8H8O3 | 27.88 (2.35) | n.i. | 27.51 (2.40) | n.i. | 61.71 (4.57) | - | 79.01 (5.59) | n.i. | 21.09 (2.03) | n.i. | 53.50 (3.19) | n.i. |
| 13 | Vanillin isomer c 3 | 11.06 | 151 | C8H8O3 | n.i. | 0.27 (0.03) | n.i. | n.i. | n.i. | 0.18 (0.02) | n.i. | 0.28 (0.02) | n.i. | n.i. | n.i. | n.i. |
| 14 | Oleuropein aglycone derivative 1 | 11.44 | 377 | C16H26O10 | 12.70 (0.42) | n.i. | 18.47 (1.12) | n.i. | 11.72 (0.87) | - | 21.01 (1.80) | - | 24.55 (2.38) | n.i. | 106.25 (9.67) | n.i. |
| 15 | β-hydroxy-verbascoside isomer a 3 | 12.06 | 639 | C29H36O16 | n.i. | n.i. | 5.22 (0.08) | n.i. | 11.40 (0.72) | - | 10.99 (0.54) | - | 16.96 (1.65) | n.i. | 5.99 (0.42) | n.i. |
| 16 | β-hydroxy-verbascoside isomer b 3 | 12.21 | 639 | C29H36O16 | n.i. | n.i. | 2.36 (0.07) | n.i. | 10.19 (0.79) | - | 14.02 (1.32) | - | 15.07 (0.77) | n.i. | 4.02 (0.40) | n.i. |
| 17 | Demethyloleuropein 1 | 13.95 | 525 | C24H30O13 | 9.75 (0.94) | n.i. | 23.68 (2.15) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 18 | Rutin 2 | 14.48 | 609 | C27H30O16 | 110.16 (6.09) | n.i. | 19.48 (1.79) | n.i. | 114.34 (10.88) | - | 189.44 (18.01) | - | 20.27 (0.45) | n.i. | 113.53 (10.67) | n.i. |
| 19 | Hydroxytyrosol acetate/3,4-DHPEA-AC 3 | 15.22 | 195 | C10H12O4 | n.i. | 2.67 (0.20) | n.i. | n.i. | n.i. | 2.06 (0.14) | n.i. | 2.37 (0.08) | n.i. | n.i. | n.i. | n.i. |
| 20 | Luteolin glucoside isomer 2 | 15.4 | 447 | C21H20O11 | 149.29 (10.75) | n.i. | 18.87 (0.70) | n.i. | 69.84 (3.29) | n.i. | 129.79 (12.63) | n.i. | 24.94 (2.42) | n.i. | 14.92 (0.74) | n.i. |
| 21 | Verbascoside isomer a 3 | 15.61 | 623 | C29H36O15 | 340.74 (33.43) | n.i. | 307.91 (25.61) | n.i. | 308.55 (13.75) | n.i. | 406.40 (28.24) | n.i. | 731.26 (59.36) | n.i. | 292.94 (27.97) | n.i. |
| 22 | Verbascoside isomer b 3 | 16.96 | 623 | C29H36O15 | 103.94 (8.40) | n.i. | 37.58 (3.75) | n.i. | 69.32 (4.01) | n.i. | 101.78 (8.34) | n.i. | 58.20 (4.96) | n.i. | 46.35 (4.59) | n.i. |
| 23 | Apigenin rutinoside 2 | 17.95 | 577 | C27H30O14 | 5.94 (0.39) | n.i. | 4.32 (0.41) | n.i. | 6.14 (0.50) | n.i. | 7.81 (0.70) | n.i. | n.i. | n.i. | 2.12 (0.23) | n.i. |
| 24 | Oleuropein glucoside 1 | 18.05 | 701 | C31H42O18 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | 24.98 (2.43) | n.i. | 3.50 (0.30) | n.i. | n.i. | n.i. |
| 25 | Caffeoyl-6-oleoside 4 | 18.48 | 551 | C25H28O14 | n.i. | n.i. | 87.76 (8.34) | n.i. | 24.98 (2.42) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 26 | Oleuropein isomer a 1 | 18.87 | 539 | C25H32O13 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | 7.82 (0.77) | n.i. | n.i. | n.i. | n.i. | n.i. |
| 27 | Oleuropein isomer b 1 | 19.07 | 539 | C25H32O13 | n.i. | n.i. | 3.93 (0.28) | n.i. | n.i. | n.i. | 4.38 (0.23) | n.i. | 5.47 (0.59) | n.i. | n.i. | n.i. |
| 28 | 10-Hydroxyoleuropein aglycone 1 | 19.38 | 335 | C17H20O7 | n.i. | 0.71 (0.05) | - | 0.62 (0.04) | n.i. | 7.91 (0.86) | n.i. | 3.20 (0.27) | n.i. | 0.23 (0.02) | n.i. | 0.22 (0.02) |
| 29 | Oleuropein isomer c 1 | 19.53 | 539 | C25H32O13 | 1.80 (0.09) | n.i. | 1.46 (0.14) | n.i. | n.i. | n.i. | n.i. | n.i. | 5.37 (0.18) | n.i. | 29.05 (2.21) | n.i. |
| 30 | 6-p-Coumaroyl secologanoside isomer a 4 | 19.80 | 535 | C25H28O13 | 58.93 (1.82) | n.i. | 176.42 (13.78) | n.i. | 50.38 (4.10) | n.i. | 45.13 (3.61) | n.i. | 9.89 (0.84) | n.i. | 14.29 (1.39) | n.i. |
| 31 | Deacetoxyoleuropein aglycone isomer a 1 | 19.87 | 319 | C17H20O6 | n.i. | 7.77 (0.62) | n.i. | 1.26 (0.09) | n.i. | 12.14 (1.20) | n.i. | 29.86 (2.29) | n.i. | 2.14 (0.22) | n.i. | 2.90 (0.09) |
| 32 | Oleuropein isomer d 1 | 20.15 | 539 | C25H32O13 | n.i. | n.i. | n.i. | n.i. | 2.32 (0.17) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 33 | Oleuropein isomer e 1 | 20.47 | 539 | C25H32O13 | 4.18 (0.17) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 34 | Oleuropein aglycone isomer a 1 | 20.59 | 377 | C19H22O8 | n.i. | n.i. | 4.29 (0.22) | n.i. | n.i. | n.i. | n.i. | n.i. | 9.52 (0.87) | n.i. | n.i. | 12.22 (1.01) |
| 35 | 6-p-Coumaroyl secologanoside isomer b 4 | 20.72 | 535 | C25H28O13 | 2.63 (0.26) | n.i. | 1.43 (0.11) | n.i. | n.i. | n.i. | 2.07 (0.19) | n.i. | n.i. | n.i. | n.i. | n.i. |
| 36 | Oleuropein aglycone isomer b 1 | 20.86 | 377 | C19H22O8 | n.i. | n.i. | 9.89 (0.82) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 37 | Oleuropein isomer f 1 | 21.22 | 539 | C25H32O13 | n.i. | n.i. | 3.86 (0.29) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| 38 | Luteolin 2 | 21.94 | 285 | C15H10O6 | 25.42 (1.93) | 3.51 (0.31) | 47.50 (2.81) | 1.93 (0.08) | 68.53 (6.57) | 3.19 (0.20) | 33.01 (3.24) | 3.65 (0.38) | 18.60 (1.34) | 2.20 (0.04) | 18.44 (1.01) | 1.31 (0.08) |
| 39 | Deacetoxyoleuropein aglycone isomer b 1 | 22.29 | 319 | C17H20O6 | n.i. | n.q. | n.i. | 1.09 (0.09) | n.i. | 0.11 (0.01) | n.i. | 0.71 (0.03) | n.i. | 0.05 (0.01) | n.i. | n.q. |
| 40 | Oleuropein aglycone c 1 | 22.48 | 377 | C19H22O8 | n.i. | 0.68 (0.06) | n.i. | 1.76 (0.10) | n.i. | 1.02 (0.07) | n.i. | 0.32 (0.03) | n.i. | 0.94 (0.08) | n.i. | 1.98 (0.15) |
| 41 | Elenolic acid methyl ester 5 | 22.61 | 255 | C12H16O6 | n.i. | 0.18 (0.01) | n.i. | 0.21 (0.02) | n.i. | 0.12 (0.01) | n.i. | 1.55 (0.15) | n.i. | n.i. | n.i. | - |
| 42 | Acetoxypinoresinol 6 | 23.3 | 415 | C22H24O8 | n.i. | 13.04 (1.39) | n.i. | 0.13 (0.01) | n.i. | 8.27 (0.80) | n.i. | 11.70 (0.76) | n.i. | 7.00 (0.47) | n.i. | 5.88 (0.55) |
| 43 | Pinoresinol 6 | 23.93 | 357 | C20H22O6 | n.i. | 0.46 (0.04) | n.i. | n.i. | n.i. | 0.42 (0.03) | n.i. | 0.81 (0.08) | n.i. | n.i. | n.i. | n.i. |
| 44 | Apigenin 2 | 24.62 | 269 | C15H10O5 | 0.49 (0.03) | 1.42 (0.04) | 1.52 (0.09) | 0.73 (0.02) | 1.69 (0.17) | 1.06 (0.06) | 8.22 (0.92) | 3.73 (0.21) | 1.38 (0.04) | 1.15 (0.02) | 1.09 (0.07) | 0.72 (0.05) |
| 45 | Diosmetin 2 | 25.54 | 299 | C16H12O6 | 0.53 (0.05) | 0.55 (0.03) | n.i. | n.q. | 2.64 (0.26) | 1.91 (0.08) | n.i. | 0.12 (0.01) | n.i. | n.q. | n.i. | n.q. |
| 46 | Oleuropein aglycone d 1 | 26.73 | 377 | C19H22O8 | n.i. | 2.74 (0.19) | n.i. | 89.63 (7.49) | n.i. | 17.49 (1.76) | n.i. | 21.46 (2.92) | n.i. | 57.21 (4.83) | n.i. | 118.39 (16.09) |
| 47 | Oleuropein aglycone c 1 | 27.79 | 377 | C19H22O8 | n.i. | 0.96 (0.07) | n.i. | 36.13 (3.41) | n.i. | 6.62 (0.35) | n.i. | 14.28 (1.11) | n.i. | 4.49 (0.22) | n.i. | 3.43 (0.30) |
| 48 | Ligstroside aglycone 1 | 28.76 | 361 | C19H22O7 | n.i. | 0.54 (0.05) | n.i. | 32.08 (2.22) | n.i. | 0.45 (0.05) | n.i. | 1.20 (0.08) | n.i. | 3.72 (0.17) | n.i. | 3.56 (0.20) |
| Total | 1265.33 (55.34) | 35.92 (1.68) | 1249.35 (53.14) | 173.13 (8.66) | 1318.03 (48.95) | 66.33 (2.16) | 1508.95 (43.54) | 99.20 (4.55) | 2699.89 (200.56) | 155.76 (16.69) | 1066.84 (47.69) | 169.56 (7.65) | ||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talhaoui, N.; Gómez-Caravaca, A.M.; León, L.; De la Rosa, R.; Fernández-Gutiérrez, A.; Segura-Carretero, A. From Olive Fruits to Olive Oil: Phenolic Compound Transfer in Six Different Olive Cultivars Grown under the Same Agronomical Conditions. Int. J. Mol. Sci. 2016, 17, 337. https://doi.org/10.3390/ijms17030337
Talhaoui N, Gómez-Caravaca AM, León L, De la Rosa R, Fernández-Gutiérrez A, Segura-Carretero A. From Olive Fruits to Olive Oil: Phenolic Compound Transfer in Six Different Olive Cultivars Grown under the Same Agronomical Conditions. International Journal of Molecular Sciences. 2016; 17(3):337. https://doi.org/10.3390/ijms17030337
Chicago/Turabian StyleTalhaoui, Nassima, Ana María Gómez-Caravaca, Lorenzo León, Raúl De la Rosa, Alberto Fernández-Gutiérrez, and Antonio Segura-Carretero. 2016. "From Olive Fruits to Olive Oil: Phenolic Compound Transfer in Six Different Olive Cultivars Grown under the Same Agronomical Conditions" International Journal of Molecular Sciences 17, no. 3: 337. https://doi.org/10.3390/ijms17030337









