Identification of Toxic Pyrrolizidine Alkaloids and Their Common Hepatotoxicity Mechanism
Abstract
:1. Introduction
2. Results
2.1. Identifying the Potential Targets of PAs
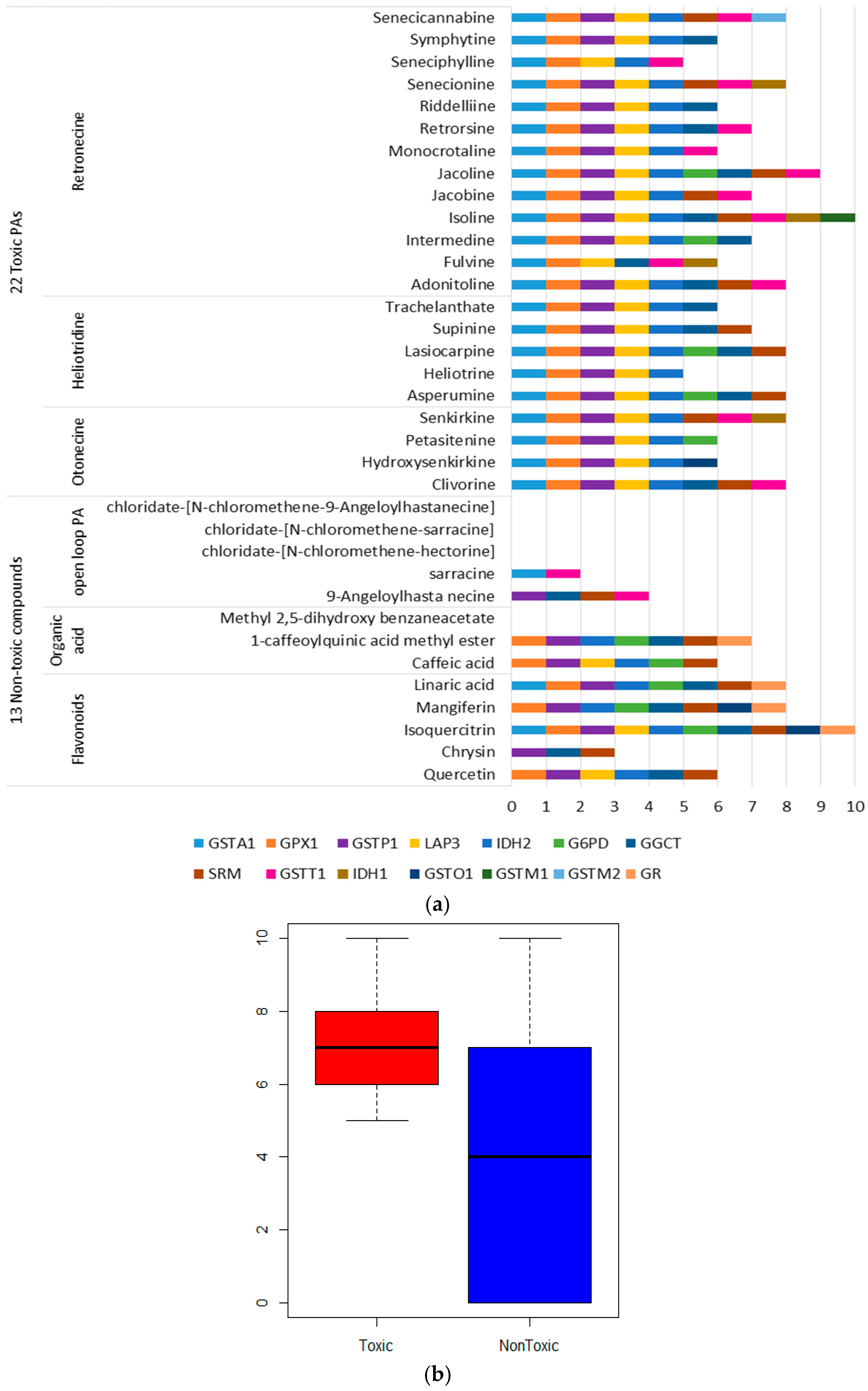
2.2. Targets Enrichment of Toxic PAs
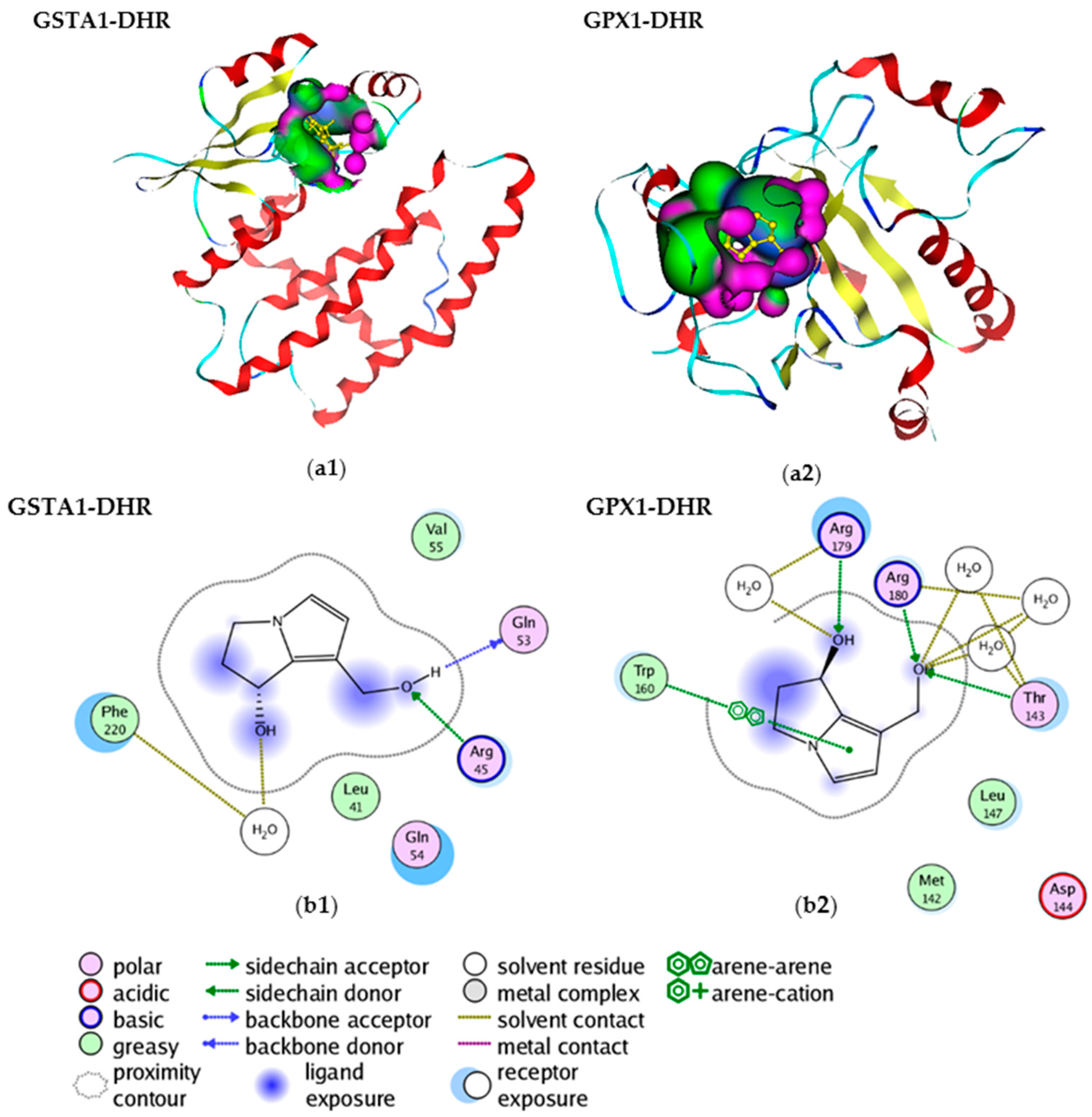
2.3. Molecular Interactions within PAs and GSTA1, GPX1
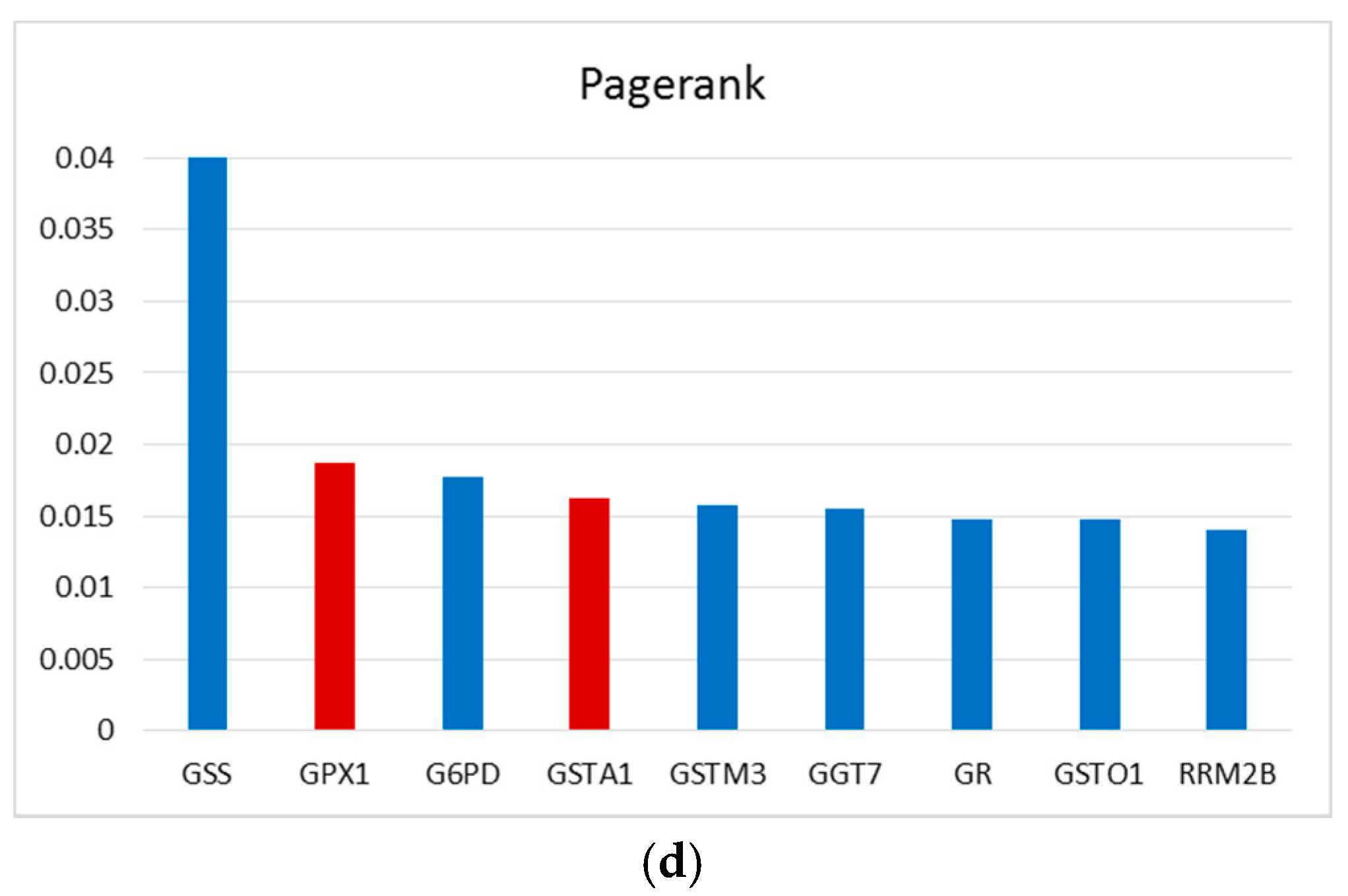
2.4. Importance of the GSTA1 and GPX1 in GSH Metabolism
3. Discussion
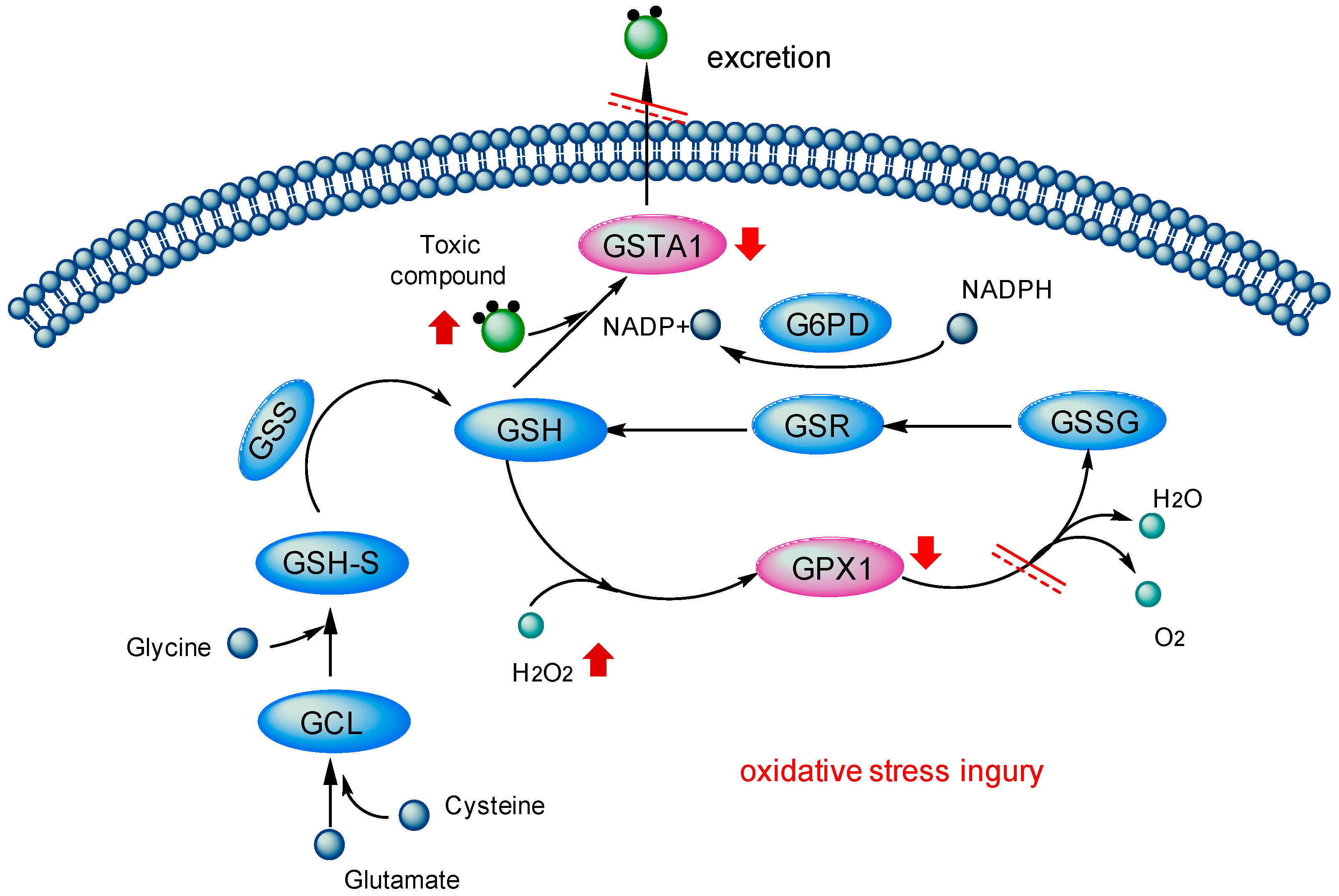
3.1. Toxicity Mechanism Analysis of Toxic PAs Based on GSTA1 and GPX1 Pattern
3.2. Limitations of Reverse Docking
4. Materials and Methods
4.1. Dataset
4.2. Identification of Potential Protein Targets of PAs
4.3. Targets Enrichment of Toxic PAs
4.4. Molecular Interactions within Toxic PAs and GSTA1, GPX1
4.5. Importance of the Targets Pattern in GSH Metabolism
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chou, M.W.; Fu, P.P. Formation of DHP-derived DNA adducts in vivo from dietary supplements and chinese herbal plant extracts containing carcinogenic pyrrolizidine alkaloids. Toxicol. Ind. Health 2006, 22, 321–327. [Google Scholar] [PubMed]
- Roeder, E. Medicinal plants in Europe containing pyrrolizidine alkaloids. Die Pharm. 1995, 50, 83–98. [Google Scholar]
- Roeder, E. Medicinal plants in China containing pyrrolizidine alkaloids. Die Pharm. 2000, 55, 711–726. [Google Scholar]
- Stegelmeier, B.L.; Edgar, J.A.; Colegate, S.M.; Gardner, D.R.; Schoch, T.K.; Coulombe, R.A.; Molyneux, R.J. Pyrrolizidine alkaloid plants, metabolism and toxicity. J. Nat. Toxins 1999, 8, 95–116. [Google Scholar] [PubMed]
- Fu, P.P.; Chou, M.W.; Xia, Q.; Yang, Y.C.; Yan, J.; Doerge, D.R.; Chan, P.C. Genotoxic pyrrolizidine alkaloids and pyrrolizidine alkaloidn-oxides—Mechanisms leading to DNA adduct formation and tumorigenicity. J. Environ. Sci. Health Part C 2001, 19, 353–385. [Google Scholar] [CrossRef]
- Seaman, J.T. Pyrrolizidine alkaloid poisoning of sheep in New South Wales. Aust. Vet. J. 1987, 64, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Green, C.E.; Segall, J.H.; Byard, J.L. Metabolism, cytotoxicity, and genotoxicity of the pyrrolizidine alkaloid senecionine in primary cultures of rat hepatocytes. Toxicol. Appl. Pharmacol. 1981, 60, 176–185. [Google Scholar] [CrossRef]
- Williams, G.M.; Mori, H.; Hirono, I.; Nagao, M. Genotoxicity of pyrrolizidine alkaloids in the hepatocyte primary culture/DNA-repair test. Mutat. Res. 1980, 79, 1–5. [Google Scholar] [CrossRef]
- Ji, L.L.; Zhang, M.; Sheng, Y.C.; Wang, Z.T. Pyrrolizidine alkaloid clivorine induces apoptosis in human normal liver L-02 cells and reduces the expression of p53 protein. Toxicol. In Vitro 2005, 19, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.L.; Zhao, X.G.; Chen, L.; Zhang, M.; Wang, Z.T. Pyrrolizidine alkaloid clivorine inhibits human normal liver L-02 cells growth and activates p38 mitogen-activated protein kinase in L-02 cells. Toxicon 2002, 40, 1685–1690. [Google Scholar] [CrossRef]
- Uhl, M.; Helma, C.; Knasmuller, S. Evaluation of the single cell gel electrophoresis assay with human hepatoma (Hep G2) cells. Mutat. Res. 2000, 468, 213–225. [Google Scholar] [CrossRef]
- Rizo, W.F.; Ferreira, L.E.; Colnaghi, V.; Martins, J.S.; Franchi, L.P.; Takahashi, C.S.; Beleboni, R.O.; Marins, M.; Pereira, P.S.; Fachin, A.L. Cytotoxicity and genotoxicity of coronaridine from Tabernaemontana catharinensis A.DC in a human laryngeal epithelial carcinoma cell line (Hep-2). Genet. Mol. Biol. 2013, 36, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Zuckerman, M.; Steenkamp, V.; Stewart, M.J. Hepatic veno-occlusive disease as a result of a traditional remedy: Confirmation of toxic pyrrolizidine alkaloids as the cause, using an in vitro technique. J. Clin. Pathol. 2002, 55, 676–679. [Google Scholar] [CrossRef] [PubMed]
- Steenkamp, V.; Stewart, M.J.; van der Merwe, S.; Zuckerman, M.; Crowther, N.J. The effect of Senecio latifolius a plant used as a South African traditional medicine, on a human hepatoma cell line. J. Ethnopharmacol. 2001, 78, 51–58. [Google Scholar] [CrossRef]
- Gordon, G.J.; Coleman, W.B.; Grisham, J.W. Induction of cytochrome P450 enzymes in the livers of rats treated with the pyrrolizidine alkaloid retrorsine. Exp. Mol. Pathol. 2000, 69, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Fu, P.P.; Xia, Q.; Lin, G.; Chou, M.W. Pyrrolizidine alkaloids—Genotoxicity, metabolism enzymes, metabolic activation, and mechanisms. Drug Metab. Rev. 2004, 36, 1–55. [Google Scholar] [CrossRef] [PubMed]
- Neuman, M.G.; Cohen, L.; Opris, M.; Nanau, R.M.; Hyunjin, J. Hepatotoxicity of pyrrolizidine alkaloids. J. Pharm. Pharm. Sci. 2015, 18, 825–843. [Google Scholar] [PubMed]
- Ji, L.; Chen, Y.; Wang, Z. Protection of S-adenosyl methionine against the toxicity of clivorine on hepatocytes. Environ. Toxicol. Pharmacol. 2008, 26, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ji, L.; Wang, H.; Wang, Z. Intracellular glutathione plays important roles in pyrrolizidine alkaloids-induced growth inhibition on hepatocytes. Environ. Toxicol. Pharmacol. 2009, 28, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ji, L.; Xiong, A.; Yang, L.; Wang, Z. Involvement of intracellular glutathione in regulating isoline-induced cytotoxicity in human normal liver L-02 cells. Toxicol. Ind. Health 2013, 29, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Sheng, Y.; Jiang, P.; Ji, L.; Xia, Y.; Min, Y.; Wang, Z. The gender-dependent difference of liver GSH antioxidant system in mice and its influence on isoline-induced liver injury. Toxicology 2011, 280, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.Y.; Chen, Y.; Wang, Z.Y.; Ji, L.L.; Wang, Z.T. Pyrrolizidine alkaloid isoline-induced oxidative injury in various mouse tissues. Exp. Toxicol. Pathol. 2010, 62, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Liu, T.; Wang, Z. Pyrrolizidine alkaloid clivorine induced oxidative injury on primary cultured rat hepatocytes. Hum. Exp. Toxicol. 2010, 29, 303–309. [Google Scholar] [PubMed]
- Wang, Z.Y.; Kang, H.; Ji, L.L.; Yang, Y.Q.; Liu, T.Y.; Cao, Z.W.; Morahan, G.; Wang, Z.T. Proteomic characterization of the possible molecular targets of pyrrolizidine alkaloid isoline-induced hepatotoxicity. Environ. Toxicol. Pharmacol. 2012, 34, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Amin, K.A.; Hashem, K.S.; Al-muzafar, H.M.; Taha, E.M. Oxidative hepatotoxicity effects of monocrotaline and its amelioration by lipoic acid, S-adenosyl methionine and vitamin E. J. Complement. Integr. Med. 2014, 11, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Amin, K.A.; Hassan, M.S.; el Awad, S.T.; Hashem, K.S. The protective effects of cerium oxide nanoparticles against hepatic oxidative damage induced by monocrotaline. Int. J. Nanomed. 2011, 6, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M. The KEGG database. Silico Simul. Biol. Process. 2002, 247, 91–103. [Google Scholar]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The protein data bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Park, K. Induction of reactive oxygen species and apoptosis in BEAS-2B cells by mercuric chloride. Toxicol. In Vitro 2007, 21, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T.; Ochiai, M.; Kojima, C.; Ohta, T.; Sakurai, M.H.; Takada, N.O.; Qu, W.; Waalkes, M.P.; Himeno, S.; Fujiwara, K. Preventive mechanism of cellular glutathione in monomethylarsonic acid-induced cytolethality. Toxicol. Appl. Pharmacol. 2005, 206, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Yadav, U.C.; Ramana, K.V.; Awasthi, Y.C.; Srivastava, S.K. Glutathione level regulates HNE-induced genotoxicity in human erythroleukemia cells. Toxicol. Appl. Pharmacol. 2008, 227, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Dukhande, V.V.; Malthankar-Phatak, G.H.; Hugus, J.J.; Daniels, C.K.; Lai, J.C. Manganese-induced neurotoxicity is differentially enhanced by glutathione depletion in astrocytoma and neuroblastoma cells. Neurochem. Res. 2006, 31, 1349–1357. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Pulford, D.J. The glutathione S-transferase supergene family: Regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 445–600. [Google Scholar] [CrossRef] [PubMed]
- Lash, L.H.; Putt, D.A.; Zalups, R.K. Influence of exogenous thiols on inorganic mercury-induced injury in renal proximal and distal tubular cells from normal and uninephrectomized rats. J. Pharmacol. Exp. Ther. 1999, 291, 492–502. [Google Scholar] [PubMed]
- Brigelius-Flohe, R. Tissue-specific functions of individual glutathione peroxidases. Free Radic. Biol. Med. 1999, 27, 951–965. [Google Scholar] [CrossRef]
- Higashi, Y.; Pandey, A.; Goodwin, B.; Delafontaine, P. Insulin-like growth factor-1 regulates glutathione peroxidase expression and activity in vascular endothelial cells: Implications for atheroprotective actions of insulin-like growth factor-1. Biochim. Biophys. Acta 2013, 1832, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Antunes, F.; Han, D.; Cadenas, E. Relative contributions of heart mitochondria glutathione peroxidase and catalase to H2O2 detoxification in in vivo conditions. Free Radic. Biol. Med. 2002, 33, 1260–1267. [Google Scholar] [CrossRef]
- Cohen, G.; Hochstein, P. Glutathione Peroxidase: The Primary agent for the elimination of hydrogen peroxide in erythrocytes. Biochemistry 1963, 2, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Miura, T.; Muraoka, S.; Fujimoto, Y. Lipid peroxidation induced by phenylbutazone radicals. Life Sci. 2002, 70, 2611–2621. [Google Scholar] [CrossRef]
- Sheehan, D.; Meade, G.; Foley, V.M.; Dowd, C.A. Structure, function and evolution of glutathione transferases: Implications for classification of non-mammalian members of an ancient enzyme superfamily. Biochem. J. 2001, 360, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Leaver, M.J.; George, S.G. A piscine glutathione S-transferase which efficiently conjugates the end-products of lipid peroxidation. Mar. Environ. Res. 1998, 46, 71–74. [Google Scholar] [CrossRef]
- Litwack, G.; Ketterer, B.; Arias, I.M. Ligandin: A hepatic protein which binds steroids, bilirubin, carcinogens and a number of exogenous organic anions. Nature 1971, 234, 466–467. [Google Scholar] [CrossRef] [PubMed]
- Douglas, K.T. Mechanism of action of glutathione-dependent enzymes. Adv. Enzymol. Relat. Areas Mol. Biol. 1987, 59, 103–167. [Google Scholar] [PubMed]
- Oakley, A. Glutathione transferases: A structural perspective. Drug Metab. Rev. 2011, 43, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Mannervik, B.; Alin, P.; Guthenberg, C.; Jensson, H.; Tahir, M.K.; Warholm, M.; Jornvall, H. Identification of three classes of cytosolic glutathione transferase common to several mammalian species: Correlation between structural data and enzymatic properties. Proc. Natl. Acad. Sci. USA 1985, 82, 7202–7206. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Oxidative stress: From basic research to clinical application. Am. J. Med. 1991, 91, 31S–38S. [Google Scholar] [CrossRef]
- Pinkus, R.; Weiner, L.M.; Daniel, V. Role of oxidants and antioxidants in the induction of AP-1, NF-κB, and glutathione S-transferase gene expression. J. Biol. Chem. 1996, 271, 13422–13429. [Google Scholar] [PubMed]
- Beckett, G.J.; Chapman, B.J.; Dyson, E.H.; Hayes, J.D. Plasma glutathione S-transferase measurements after paracetamol overdose: Evidence for early hepatocellular damage. Gut 1985, 26, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Hughes, V.F.; Trull, A.K.; Gimson, A.; Friend, P.J.; Jamieson, N.; Duncan, A.; Wight, D.G.D.; Prevost, A.T.; Alexander, G.J.M. Randomized trial to evaluate the clinical benefits of serumα-glutathione S-transferase concentration monitoring after liver transplantation1. Transplantation 1997, 64, 1446–1452. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Caporaso, N.; Tuccillo, C.; Morisco, F.; del Vecchio Blanco, G.; del Vecchio Blanco, C. Alpha-glutathione transferases in HCV-related chronic hepatitis: A new predictive index of response to interferon therapy? J. Hepatol. 1998, 28, 390–395. [Google Scholar] [CrossRef]
- Chen, Y.Z.; Ung, C.Y. Computer automated prediction of potential therapeutic and toxicity protein targets of bioactive compounds from Chinese medicinal plants. Am. J. Chin. Med. 2002, 30, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.W.; Chen, Y.Z. Hydrogen-bond disruption probability in proteins by a modified self-consistent harmonic approach. Biopolymers 2001, 58, 319–328. [Google Scholar] [CrossRef]
- Chen, Y.Z.; Ung, C.Y. Prediction of potential toxicity and side effect protein targets of a small molecule by a ligand-protein inverse docking approach. J. Mol. Graph. Model. 2001, 20, 199–218. [Google Scholar] [CrossRef]
- Rose, P.W.; Bi, C.; Bluhm, W.F.; Christie, C.H.; Dimitropoulos, D.; Dutta, S.; Green, R.K.; Goodsell, D.S.; Prlic, A.; Quesada, M.; et al. The RCSB Protein Data Bank: New resources for research and education. Nucleic Acids Res. 2013, 41, D475–D482. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Z.; Zhi, D.G. Ligand-protein inverse docking and its potential use in the computer search of protein targets of a small molecule. Proteins-Struct. Funct. Genet. 2001, 43, 217–226. [Google Scholar] [CrossRef]
- Prasad, T.S.K.; Kandasamy, K.; Pandey, A. Human protein reference database and human proteinpedia as discovery tools for systems biology. In Reverse Chemical Genetics; Koga, H., Ed.; Humana Press: New York, NY, USA, 2009; Volume 577, pp. 67–79. [Google Scholar]
- Licata, L.; Briganti, L.; Peluso, D.; Perfetto, L.; Iannuccelli, M.; Galeota, E.; Sacco, F.; Palma, A.; Nardozza, A.P.; Santonico, E.; et al. MINT, the molecular interaction database: 2012 update. Nucleic Acids Res. 2012, 40, D857–D861. [Google Scholar] [CrossRef] [PubMed]
- Chatr-Aryamontri, A.; Breitkreutz, B.J.; Heinicke, S.; Boucher, L.; Winter, A.; Stark, C.; Nixon, J.; Ramage, L.; Kolas, N.; O’Donnell, L.; et al. The BioGRID interaction database: 2013 update. Nucleic Acids Res. 2013, 41, D816–D823. [Google Scholar] [CrossRef] [PubMed]
- Orchard, S.; Ammari, M.; Aranda, B.; Breuza, L.; Briganti, L.; Broackes-Carter, F.; Campbell, N.H.; Chavali, G.; Chen, C.; del-Toro, N.; et al. The MIntAct project—IntAct as a common curation platform for 11 molecular interaction databases. Nucleic Acids Res. 2014, 42, D358–D363. [Google Scholar] [CrossRef] [PubMed]
- Salwinski, L.; Miller, C.S.; Smith, A.J.; Pettit, F.K.; Bowie, J.U.; Eisenberg, D. The Database of Interacting Proteins: 2004 update. Nucleic Acids Res. 2004, 32, D449–D451. [Google Scholar] [CrossRef] [PubMed]
- Pagel, P.; Kovac, S.; Oesterheld, M.; Brauner, B.; Dunger-Kaltenbach, I.; Frishman, G.; Montrone, C.; Mark, P.; Stumpflen, V.; Mewes, H.W.; et al. The MIPS mammalian protein-protein interaction database. Bioinformatics 2005, 21, 832–834. [Google Scholar] [CrossRef] [PubMed]

| Coefficients a | ||||||
|---|---|---|---|---|---|---|
| Model | Parameter | Unstandardized Coefficients | Standardized Coefficients | t | Sig. | |
| B | Std. Error | Beta | ||||
| 1 | (Constant) | 8.47 × 10−17 | 0.100 | 0.000 | 1.000 | |
| GSTA1 | 0.846 | 0.117 | 0.777 | 7.207 | 0.000 | |
| 2 | (Constant) | 0.119 | 0.079 | 1.516 | 0.139 | |
| GSTA1 | 0.796 | 0.089 | 0.731 | 8.929 | 0.000 | |
| GR | −0.597 | 0.115 | −0.423 | −5.171 | 0.000 | |
| 3 | (Constant) | −0.067 | 0.067 | −1.005 | 0.322 | |
| GSTA1 | 0.471 | 0.088 | 0.433 | 5.340 | 0.000 | |
| GR | −0.767 | 0.090 | −0.544 | −8.526 | 0.000 | |
| GPX1 | 0.552 | 0.101 | 0.488 | 5.452 | 0.000 | |
| 4 | (Constant) | −0.078 | 0.063 | −1.234 | 0.227 | |
| GSTA1 | 0.546 | 0.089 | 0.502 | 6.107 | 0.000 | |
| GR | −0.962 | 0.121 | −0.683 | −7.964 | 0.000 | |
| GPX1 | 0.858 | 0.166 | 0.697 | 5.183 | 0.000 | |
| LAP3 | −0.364 | 0.161 | −0.334 | −2.261 | 0.031 | |
| Targets Pattern | Toxic Prediction Precision | Non-Toxic Predictive Precision | Total Prediction Precision |
|---|---|---|---|
| GSTA1 | 22/22 | 10/13 | 91.43% |
| GPX1 | 22/22 | 7/13 | 82.86% |
| LAP3 | 0/22 | 4/13 | 11.43% |
| GR | 22/22 | 5/13 | 77.14% |
| LAP3, GR | 0/22 | 2/13 | 5.71% |
| GSTA1, GPX1 | 22/22 | 11/13 | 94.29% |
| GSTA1, GPX1, GR | 22/22 | 0/13 | 62.86% |
| GSTA1, GPX1, LAP3, GR | 0/22 | 0/13 | 0.00% |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, X.; Kang, H.; Feng, J.; Yang, Y.; Tang, K.; Zhu, R.; Yang, L.; Wang, Z.; Cao, Z. Identification of Toxic Pyrrolizidine Alkaloids and Their Common Hepatotoxicity Mechanism. Int. J. Mol. Sci. 2016, 17, 318. https://doi.org/10.3390/ijms17030318
Yan X, Kang H, Feng J, Yang Y, Tang K, Zhu R, Yang L, Wang Z, Cao Z. Identification of Toxic Pyrrolizidine Alkaloids and Their Common Hepatotoxicity Mechanism. International Journal of Molecular Sciences. 2016; 17(3):318. https://doi.org/10.3390/ijms17030318
Chicago/Turabian StyleYan, Xinmiao, Hong Kang, Jun Feng, Yiyan Yang, Kailin Tang, Ruixin Zhu, Li Yang, Zhengtao Wang, and Zhiwei Cao. 2016. "Identification of Toxic Pyrrolizidine Alkaloids and Their Common Hepatotoxicity Mechanism" International Journal of Molecular Sciences 17, no. 3: 318. https://doi.org/10.3390/ijms17030318
APA StyleYan, X., Kang, H., Feng, J., Yang, Y., Tang, K., Zhu, R., Yang, L., Wang, Z., & Cao, Z. (2016). Identification of Toxic Pyrrolizidine Alkaloids and Their Common Hepatotoxicity Mechanism. International Journal of Molecular Sciences, 17(3), 318. https://doi.org/10.3390/ijms17030318







