Identification of Powdery Mildew Responsive Genes in Hevea brasiliensis through mRNA Differential Display
Abstract
:1. Introduction
2. Results
2.1. Comparison of Powdery Mildew Resistance between RRIC52 and Reyan 7-33-97
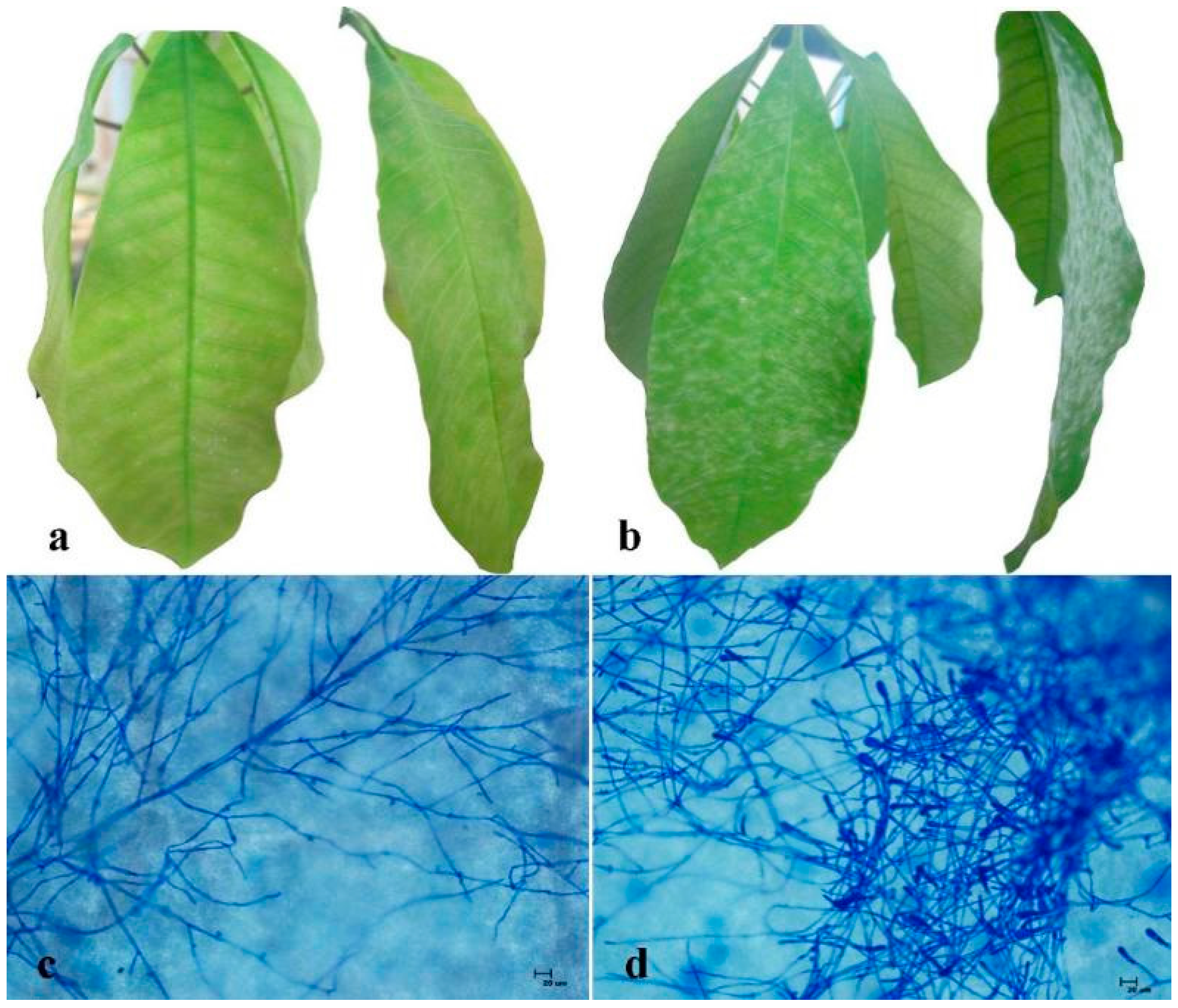
2.2. Identification and Isolation of Differentially Expressed Transcripts
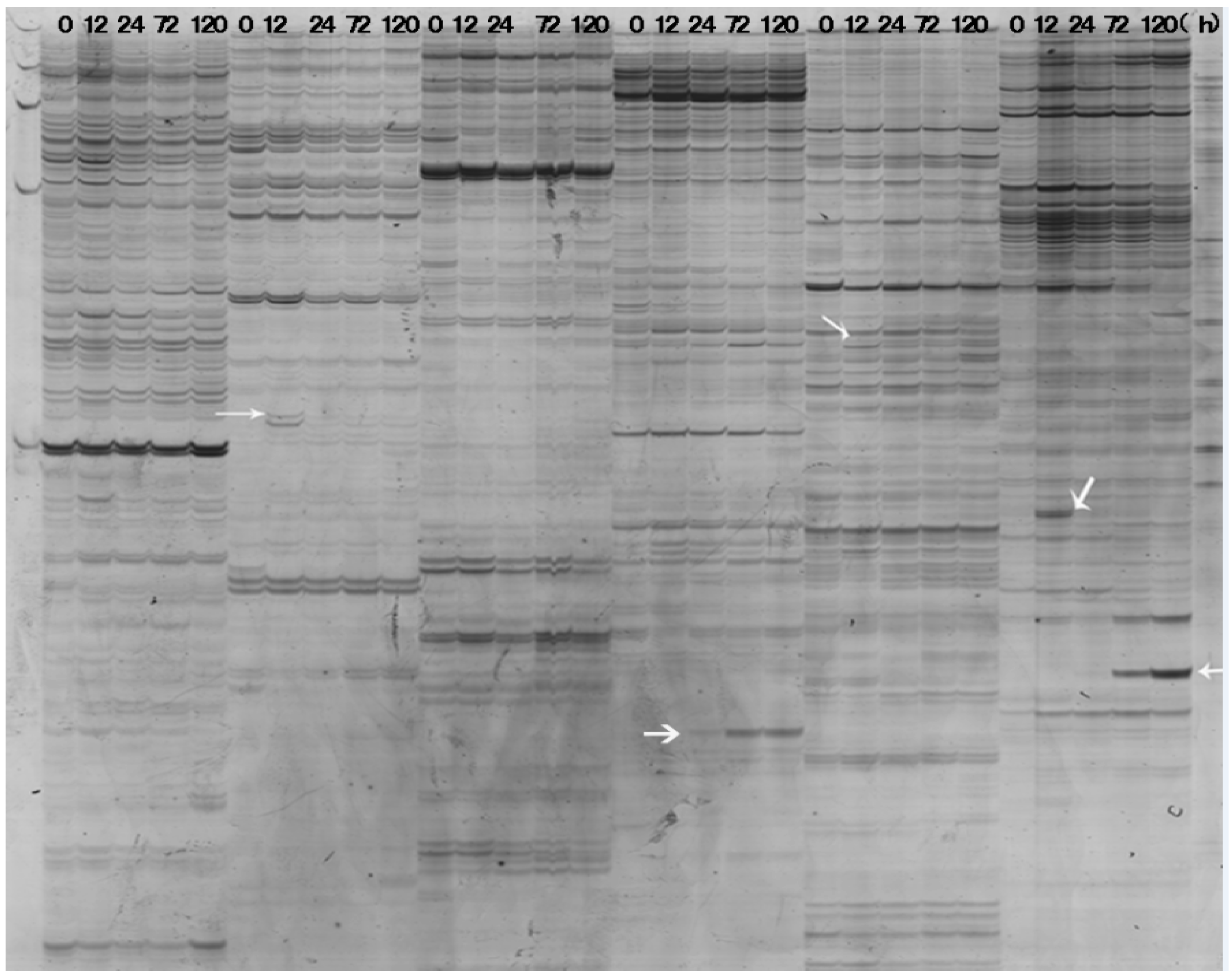


2.3. Sequence Analysis Revealed the Significances of Differentially Expressed RRIC52 Transcripts
2.4. Characterization of the Differentially Expressed ESTs
| ESTs | dbEST_Id | Accession No | Homology Found with | Homologous Species | Identity % | Description | E-Value |
|---|---|---|---|---|---|---|---|
| ESTs in cell wall and membrane pathways | |||||||
| HBOH2 | 79640384 | JZ822682 | XM_002522813.1 | Ricinus communis | 85.47 | pectinesterase-2 precursor, putative | 2 × 10−87 |
| HBOH3 | 79721589 | JZ893198 | U48687.1 | Castanea sativa | 78.07 | endochitinase | 3 × 10−164 |
| HBOH5 | 79721590 | JZ893199 | AB367524.1 | Arabidopsis thaliana | 66 | CERK1 mRNA for chitin elicitor receptor kinase 1 | 1.00 × 10−100 |
| HBOH7 | 79640387 | JZ822685 | XM_002530816.1 | Ricinus communis | 84.47 | glucan endo-1,3-beta-glucosidase precursor, putative | 3 × 10−149 |
| HBOH15 | 79721594 | JZ8931203 | XM_006375808.1 | Populus trichocarpa | 80.33 | germin-like protein 1 | 6 × 10−72 |
| HBOH66 | 79761733 | JZ897003 | XM_006376356.1 | Populus trichocarpa | 81.84 | class IV chitinase family protein | 2 × 10−132 |
| HBOH67 | 79761734 | JZ897004 | XM_002532766.1 | Ricinus communis | 80.94 | chitinase 1 precursor, putative | 3 × 10−118 |
| HBOH68 | 79761735 | JZ897005 | AY973617.1 | Manihot esculenta | 88.24 | germin-like protein GLP partial CdS | 2 × 10−163 |
| ESTs involved in transcription factor and regulatory proteins | |||||||
| HBOH4 | 79640385 | JZ822683 | XM_002526622.1 | Ricinus communis | 86.35 | ferritin, plant, putative | 7 × 10−171 |
| HBOH13 | 79721592 | JZ8931201 | XM_002303816.2 | Populus trichocarpa | 78.24 | WRKY transcription factor 40 family protein | 3 × 10−85 |
| HBOH14 | 79721593 | JZ8931202 | XM_012596852.1 | Gossypium raimondii | 58 | WRKY transcription factor 18-like | 7 × 10−15 |
| HBOH31 | 79761701 | JZ896971 | XM_002534015.1 | Ricinus communis | 87.56 | eukaryotic initiation factor iso-4F subunit p82-34, putative | 3 × 10−62 |
| HBOH33 | 79761703 | JZ896973 | XM_002517578.1 | Ricinus communis | 78.79 | DNA binding protein, putative | 2 × 10−140 |
| HBOH36 | 79761705 | JZ896975 | XM_002527643.1 | Ricinus communis | 84.82 | transcription factor btf3, putative | 0 |
| HBOH55 | 79761724 | JZ896994 | XM_002524212.1 | Ricinus communis | 82.11 | rop guanine nucleotide exchange factor, putative | 0 |
| HBOH60 | 79761727 | JZ896997 | XM_006368449.1 | Populus trichocarpa | 85.21 | WRKY transcription factor 40 family protein | 4 × 10−140 |
| ESTs involved in transporter | |||||||
| HBOH8 | 79640388 | JZ822686 | XM_002521455.1 | Ricinus communis | 85.71 | dimethyladenosine transferase, putative | 1 × 10−53 |
| HBOH28 | 79761698 | JZ896968 | XM_002527741.1 | Ricinus communis | 88.11 | clathrin assembly protein, putative | 2 × 10−144 |
| HBOH38 | 79761707 | JZ896977 | XM_002529119.1 | Ricinus communis | 82.69 | DNA-directed RNA polymerase II, putative | 6 × 10−154 |
| HBOH39 | 79761708 | JZ896977 | XM_002529119.1 | Ricinus communis | 83.15 | DNA-directed RNA polymerase II, putative | 3 × 10−158 |
| HBOH44 | 79761713 | JZ896983 | XM_002269429.2 | Vitis vinifera | 66 | clathrin light chain 2 | 5 × 10−73 |
| HBOH46 | 79761715 | JZ896985 | XM_002531006.1 | Ricinus communis | 76.31 | transferase, transferring glycosyl groups, putative | 4 × 10−90 |
| HBOH52 | 79761721 | JZ896991 | HM461981.1 | Vernicia fordii | 84.48 | glycerol-3-phosphate acyltransferase 9 (GPAT9) gene | 0.00003 |
| HBOH54 | 79761723 | JZ896993 | HQ285842.1 | Hevea brasiliensis | 98.95 | chloroplast, complete genome | 0 |
| HBOH75 | 79761741 | JZ897011 | XM_002524326.1 | Ricinus communis | 72.5 | flavonol 4'-sulfotransferase, putative | 4 × 10−51 |
| HBOH81 | 79761747 | JZ897017 | XM_007017019.1 | Theobroma cacao | 72.69 | non-specific lipid-transfer protein, putative | 2 × 10−26 |
| ESTs involved in signal transduction | |||||||
| HBOH1 | 79640383 | JZ822681 | XM_002516001.1 | Ricinus communis | 68.49 | serine-threonine protein kinase, plant-type, putative | 4 × 10−63 |
| HBOH6 | 79640386 | JZ822684 | XM_002324361.2 | Populus trichocarpa | 87.66 | H+-ATPase family protein mRNA, complete cds | 2 × 10−130 |
| HBOH9 | 79721591 | JZ893120 | XM_003605902.1 | Medicago truncatula | 40 | hypothetical protein mRNA, complete cds | 0.0001 |
| HBOH10 | 79640389 | JZ822687 | XM_007019829.1 | Theobroma cacao | 86.79 | cysteine-rich RLK (RECEPTOR-like protein kinase) 8 | 0.00002 |
| HBOH22 | 79761693 | JZ896963 | XM_002514747.1 | Ricinus communis | 87.7 | 4-nitrophenylphosphatase, putative | 0 |
| HBOH23 | 79761694 | JZ896964 | XM_007019349.1 | Theobroma cacao | 66 | cysteine-rich RLK (RECEPTOR-like protein kinase) 8 | 5 × 10−43 |
| HBOH26 | 79761697 | JZ896967 | XM_002509773.1 | Ricinus communis | 83.05 | ATP binding protein, putative | 3 × 10−170 |
| HBOH41 | 79761710 | JZ896980 | XM_002308527.2 | Populus trichocarpa | 71.47 | kinase family protein | 9 × 10−35 |
| HBOH42 | 79761711 | JZ896981 | AM478326.2 | Vitis vinifera | 84.55 | contig VV78X154662.12, whole genome shotgun sequence | 5 × 10−24 |
| HBOH45 | 79761714 | JZ896984 | XM_007026459.1 | Theobroma cacao | 95.35 | signal transducer and transcription activator isoform 1 | 1 × 10−28 |
| HBOH53 | 79761722 | JZ896992 | XM_002524179.1 | Ricinus communis | 75.7 | kinase, putative | 3 × 10−10 |
| HBOH56 | 79761725 | JZ896995 | XM_002524179.1 | Ricinus communis | 75.26 | kinase, putative | 6 × 10−47 |
| HBOH73 | 79761739 | JZ897009 | XM_002531397.1 | Ricinus communis | 83.52 | serine-threonine protein kinase, plant-type, putative | 1 × 10−74 |
| HBOH74 | 79761740 | JZ897010 | XM_002523439.1 | Ricinus communis | 82.36 | serine-threonine protein kinase, plant-type, putative | 5 × 10−139 |
| HBOH78 | 79761744 | JZ897014 | XM_002530595.1 | Ricinus communis | 69.87 | B-Raf proto-oncogene serine/threonine-protein kinase, putative | 9 × 10−27 |
| HBOH80 | 79761746 | JZ897016 | XM_002513521.1 | Ricinus communis | 88.85 | kinase, putative | 3 × 10−107 |
| ESTs involved in phytoalexin biosynthesis | |||||||
| HBOH48 | 79761717 | JZ896987 | DQ371802.1 | Populus alba | 83.36 | chalcone synthase (CHS) gene, CHS-M2 allele | 5 × 10−159 |
| HBOH61 | 79761728 | JZ896998 | XM_002300417.2 | Populus trichocarpa | 78.07 | senescence-related gene 1 family protein | 5 × 10−180 |
| HBOH62 | 79761729 | JZ896999 | XM_002517028.1 | Ricinus communis | 85.4 | acireductone dioxygenase, putative | 6 × 10−35 |
| HBOH63 | 79761730 | JZ897000 | XM_002519376.1 | Ricinus communis | 84.65 | UDP-glucosyltransferase, putative | 0 |
| HBOH70 | 79761737 | JZ897007 | XM_002533589.1 | Ricinus communis | 83.4 | leucoanthocyanidin dioxygenase, putative | 0 |
| HBOH72 | 79761738 | JZ897008 | XM_002524028.1 | Ricinus communis | 76.42 | short chain alcohol dehydrogenase, putative | 4 × 10−98 |
| HBOH76 | 79761742 | JZ897012 | AY207387.1 | Hevea brasiliensis | 98.12 | 1-aminocyclopropane-1-carboxylate oxidase | 5 × 10−98 |
| HBOH77 | 79761743 | JZ897013 | XM_012216610.1 | Jatropha curcas | 99 | cytochrome P450 82C4-like (LOC105633910), mRNA | 1 × 10−61 |
| HBOH79 | 79761745 | JZ897015 | XM_002311738.2 | Populus trichocarpa | 84.97 | galactinol synthase family protein | 0 |
| HBOH83 | 79761749 | JZ897019 | XM_002531145.1 | Ricinus communis | 81.68 | geranyl geranyl pyrophosphate synthase, putative | 2 × 10−146 |
| HBOH85 | 79761751 | JZ897021 | XM_002531048.1 | Ricinus communis | 100 | cytochrome P450, putative, mRNA | 1 × 10−81 |
| ESTs involved in other metabolism | |||||||
| HBOH11 | 79640390 | JZ822688 | XM_002512371.1 | Ricinus communis | 77.2 | histone h2a, putative | 3 × 10−59 |
| HBOH12 | 79640391 | JZ822689 | XM_002297827.2 | Populus trichocarpa | 84.94 | Humj1 family protein | 2 × 10−124 |
| HBOH18 | 79761690 | JZ896960 | HM363448.1 | Hevea brasiliensis | 94.58 | 60S ribosomal protein L27B (RPL27B) | 3 × 10−143 |
| HBOH19 | 79761691 | JZ896961 | XM_007008726.1 | Theobroma cacao | 85.13 | ubiquitin-specific protease 24 isoform 4 | 1 × 10−53 |
| HBOH29 | 79761699 | JZ896969 | XM_002519479.1 | Ricinus communis | 71.98 | ribonucleoprotein, chloroplast, putative | 8 × 10−28 |
| HBOH30 | 79761700 | JZ896970 | KC533451.1 | Jatropha curcas | 70.7 | SCAR marker SCAR27-740-NT genomic sequence | 3 × 10−16 |
| HBOH32 | 79761702 | JZ896972 | XM_003540551.2 | Glycine max | 76.5 | RNA-binding protein 24-like | 3 × 10−59 |
| HBOH40 | 79761709 | JZ896979 | XM_002520681.1 | Ricinus communis | 85.64 | photosystem I reaction center subunit II, chloroplast precursor, putative | 0 |
| HBOH47 | 79761716 | JZ896986 | AY247789.1 | Hevea brasiliensis | 72.73 | HEV2.1 gene | 1 × 10−31 |
| HBOH49 | 79761718 | JZ896988 | XM_002512371.1 | Ricinus communis | 80.46 | histone h2a, putative | 7 × 10−68 |
| HBOH57 | 79761726 | JZ896996 | M60274.1HEVRBSS | Hevea brasiliensis | 100 | ribulose-1,5-bisphosphate carboxylase small subunit (RbsS) | 7 × 10−89 |
| HBOH64 | 79761731 | JZ897001 | HQ285842.1 | Hevea brasiliensis | 100 | chloroplast, complete genome | 3 × 10−171 |
| HBOH84 | 79761750 | JZ897020 | HG975448.1 | Solanum pennellii | 82.22 | chromosome ch09, complete genome | 4 × 10−28 |
| Unclear ESTs | |||||||
| HBOH17 | 79761689 | JZ896959 | XM_002321005.2 | Populus trichocarpa | 73.09 | hypothetical protein | 2 × 10−24 |
| HBOH21 | 79761692 | JZ896962 | XM_002529035.1 | Ricinus communis | 84.38 | conserved hypothetical protein | 3 × 10−107 |
| HBOH24 | 79761695 | JZ896965 | XM_002514837.1 | Ricinus communis | 73.53 | conserved hypothetical protein | 6 × 10−90 |
| HBOH25 | 79761696 | JZ896966 | CT028466.1 | Poplar | 78.79 | cDNA sequences | 7 × 10−24 |
| HBOH35 | 79761704 | JZ896974 | FN552731.1 | Hevea brasiliensis | 90 | microsatellite DNA, clone mHbCIRA2697 | 2 × 10−51 |
| HBOH37 | 79761706 | JZ896976 | XM_002519498.1 | Ricinus communis | 82.3 | conserved hypothetical protein | 0 |
| HBOH43 | 79761712 | JZ896982 | HQ285842.1 | Hevea brasiliensis | 99.72 | chloroplast, complete genome | 0 |
| HBOH50 | 79761719 | JZ896989 | XM_002517090.1 | Ricinus communis | 86.52 | protein FRIGIDA, putative | 3 × 10−168 |
| HBOH51 | 79761720 | JZ896990 | EF146205.1 | Populus trichocarpa | 69.32 | clone WS0116_D21 unknown mRNA | 2 × 10−12 |
| HBOH65 | 79761732 | JZ897002 | AY439300.1 | Hevea brasiliensis | 83.91 | clone hmct18 microsatellite sequence | 2 × 10−15 |
| HBOH69 | 79761736 | JZ897006 | XM_002519826.1 | Ricinus communis | 87.7 | conserved hypothetical protein | 2 × 10−106 |
| HBOH82 | 79761748 | JZ897018 | AC213413.1 | Populus trichocarpa | 79.66 | clone POP014-E15, complete sequence | 7 × 10−19 |
2.5. Analysis of Selected EST Gene Expression Profiles in RRIC52 and Reyan 7-33-97 after Powdery Mildew Infection Using qRT-PCR
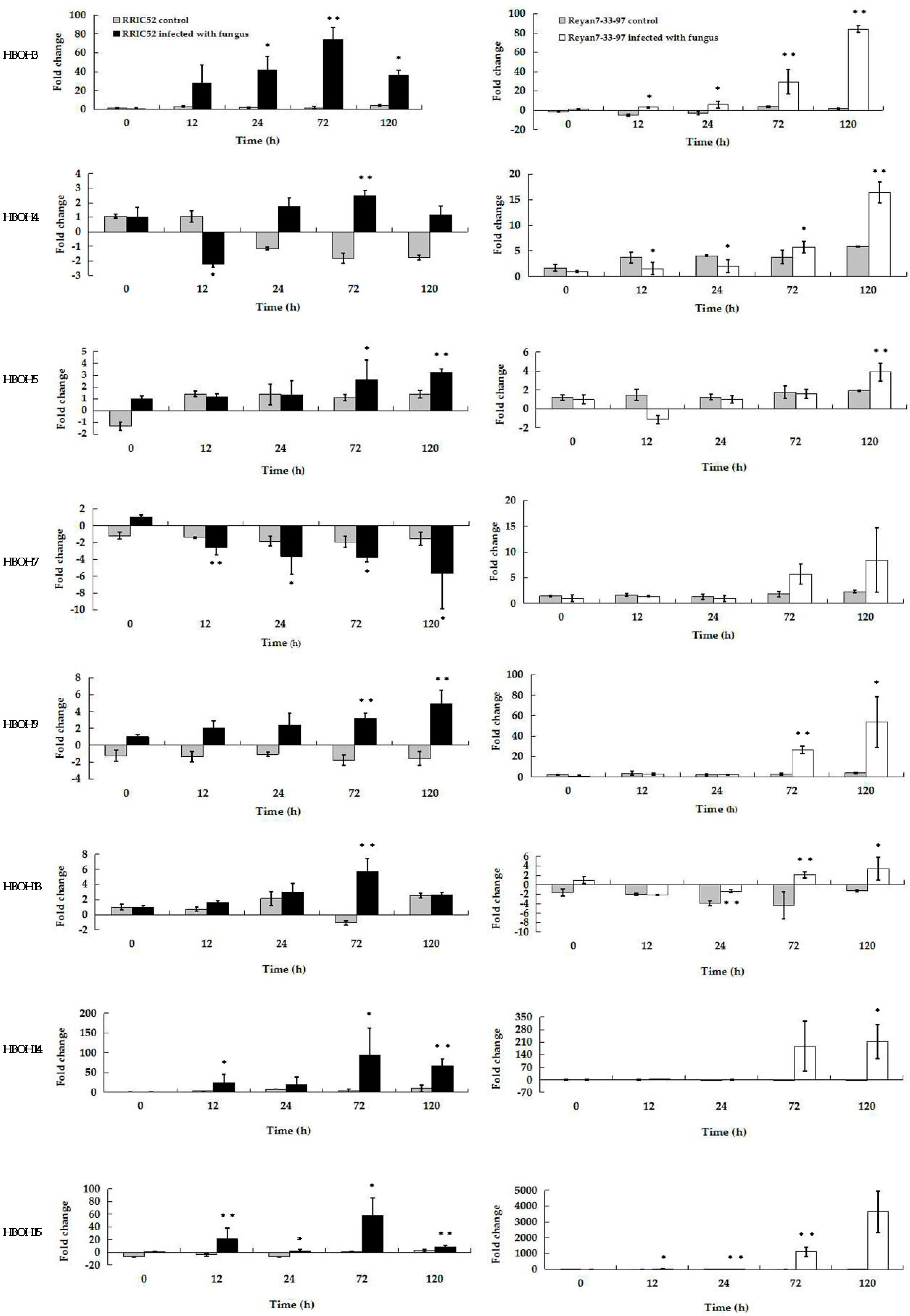
| Clone_ID | Primer Pair Used in DDRT-PCR | Size (bp) | Similar Genes Found in the Database | Functional Relevance | Plant Defense Mechanism Strategies |
|---|---|---|---|---|---|
| HBOH3 | T11A/B0308 | 759 | endochitinase (Castanea sativa) | Chitinases are enzymes that catalyze the hydrolysis of the beta-1,4-N-acetyl-d-glucosamine linkages in chitin polymers | catalyzes the hydrolysis of chitin polymers on fungal cell wall |
| HBOH4 | T11G/B0314 | 685 | ferritin, plant, putative, mRNA (Ricinus communis) | iron ion binding | regulation of cellular iron under oxidative stress |
| HBOH5 | T11A/B0314 | 895 | CERK1 mRNA for chitin elicitor receptor kinase 1 (Arabidopsis thaliana) | involved in receptor and signaling pathways, regulating innate immune responses and inflammation; involved in plant resistance to pathogen infection and herbivore attack | catalyzes the hydrolysis of chitin polymers on fungal cell wall |
| HBOH7 | T11C/B0304 | 494 | Glucan endo-1,3-β-glucosidase precursor, putative (Ricinus communis) | involved in carbohydrate binding | hydrolyses β-1,3 gucans on fungal cell wall |
| HBOH9 | T11G/B0313 | 362 | hypothetical protein mRNA (Medicago truncatula) | a single-pass membrane protein, sphingolipids play important roles in regulating cellular responses, involved in signaling pathways | signal transduction |
| HBOH13 | T11C/B0322 | 525 | WRKY transcription factor 40 family protein (Populus trichocarpa) | involved in the regulation of various physiological programs that are unique to plants, including pathogen defense, senescence and trichome development | regulation of immune effector process |
| HBOH14 | T11G/B0308 | 251 | WRKY transcription factor 18-like (Gossypium raimondii) | involved in the regulation of various physiological programs that are unique to plants, including pathogen defense, senescence and trichome development | regulation of immune effector process |
| HBOH15 | T11A/AP34 | 305 | germin-like protein 1 (Populus trichocarpa) | involved in plant cell wall reinforcement and papillae formation | protected epidermal cells from attack by fungus |
3. Discussion
3.1. Cell Wall Pathway and PR Protein
3.2. Transcription Factor
3.3. Transporter
3.4. Signal Transduction
3.5. Phytoalexin Biosynthesis
3.6. ESTs Involved in Other Metabolic Pathways
4. Materials and Methods
4.1. Rubber Trees and O. heveae
4.2. Inoculation of Rubber Trees with O. heveae HO-73
4.3. RNA Extraction and cDNA Synthesis
4.4. Differential Display Analysis
4.5. Isolation of Differentially Displayed cDNA Bands from Agarose Gel
4.6. Reverse Northern Dot Blot, Cloning, and Sequence Analysis
4.7. Expression Validation by Real-Time Quantitative RT-PCR
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lertpanyasampatha, M.; Viboonjun, U.; Kongsawadworakul, P.; Chrestin, H.; Narangajavana, J. Differential expression of microRNAs and their targets reveals a possible dual role in physiological bark disorder in rubber tree. J. Plant Physiol. 2014, 171, 1117–1126. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Deng, Z.; Chen, C.; Xia, Z.; Wu, M.; He, P.; Chen, S. Identification and characterization of genes associated with tapping panel dryness from hevea brasiliensis latex using suppression subtractive hybridization. BMC Plant Biol. 2010, 10, 140. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Zheng, F.; Zhang, Y. Molecular cloning and characterization of a Mlo gene in rubber tree (Hevea brasiliensis). J. Plant Physiol. 2015, 175, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Limkaisang, S.; Kom-un, S.; Furtado, E.L.; Liew, K.W.; Salleh, B.; Sato, Y.; Takamatsu, S. Molecular phylogenetic and morphological analyses of oidium heveae, a powdery mildew of rubber tree. Mycoscience 2005, 46, 220–226. [Google Scholar] [CrossRef]
- Manickavelu, A.; Kawaura, K.; Oishi, K.; Shin, T.; Kohara, Y.; Yahiaoui, N.; Keller, B.; Suzuki, A.; Yano, K.; Ogihara, Y. Comparative gene expression analysis of susceptible and resistant near-isogenic lines in common wheat infected by Puccinia triticina. DNA Res. 2010, 17, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Pardee, A.B. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 1992, 257, 967–971. [Google Scholar] [CrossRef] [PubMed]
- Liang, P. A decade of differential display. Biotechniques 2002, 33, 338–347. [Google Scholar] [PubMed]
- Ruiyi, Y.Z.W.S.F. Resistant behaviour of popular rubber clones in China to oidium heveae. Chin. J. Trop. Crops 1992, 1, 007. [Google Scholar]
- EST database of GenBank. Available online: http://www.ncbi.nlm.nih.gov/biosample/?term= Library%20of%20differentially%20expressed%20%20%20%20%20%20%20%20%20%20%20%20%20%20%20%20%20transcripts%20in%20Hevea%20brasiliensis%20clone%20RRIC52%20inoculated%20%20%20%20%20%20%20%20%20%20%20%20%20%20%20%20%20with%20powdery%20mildew (accessed on 5 December 2015).
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Elliott, C.; Zhou, F.; Spielmeyer, W.; Panstruga, R.; Schulze-Lefert, P. Functional conservation of wheat and rice Mlo orthologs in defense modulation to the powdery mildew fungus. Mol. Plant-Microbe Interact. 2002, 15, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Barker, C.L.; Donald, T.; Pauquet, J.; Ratnaparkhe, M.; Bouquet, A.; Adam-Blondon, A.-F.; Thomas, M.; Dry, I. Genetic and physical mapping of the grapevine powdery mildew resistance gene, Run1, using a bacterial artificial chromosome library. Theor. Appl. Genet. 2005, 111, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Donald, T.; Pellerone, F.; Adam-Blondon, A.-F.; Bouquet, A.; Thomas, M.; Dry, I. Identification of resistance gene analogs linked to a powdery mildew resistance locus in grapevine. Theor. Appl. Genet. 2002, 104, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Coleman, C.; Copetti, D.; Cipriani, G.; Hoffmann, S.; Kozma, P.; Kovács, L.; Morgante, M.; Testolin, R.; di Gaspero, G. The powdery mildew resistance gene REN1 co-segregates with an NBS-LRR gene cluster in two central Asian grapevines. BMC Genet. 2009, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, S.; di Gaspero, G.; Kovács, L.; Howard, S.; Kiss, E.; Galbács, Z.; Testolin, R.; Kozma, P. Resistance to Erysiphe necator in the grapevine “Kishmish vatkana” is controlled by a single locus through restriction of hyphal growth. Theor. Appl. Genet. 2008, 116, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Ellwood, S.; Calis, O.; Patrick, E.; Li, T.; Coleman, M.; Turner, J.G. Broad-spectrum mildew resistance in arabidopsis thaliana mediated by RPW8. Science 2001, 291, 118–120. [Google Scholar] [CrossRef] [PubMed]
- Dunwell, J.M.; Khuri, S.; Gane, P.J. Microbial relatives of the seed storage proteins of higher plants: Conservation of structure and diversification of function during evolution of the cupin superfamily. Microbiol. Mol. Biol. Rev. 2000, 64, 153–179. [Google Scholar] [CrossRef] [PubMed]
- Christensen, A.B.; Thordal-Christensen, H.; Zimmermann, G.; Gjetting, T.; Lyngkjær, M.F.; Dudler, R.; Schweizer, P. The germinlike protein GLP4 exhibits superoxide dismutase activity and is an important component of quantitative resistance in wheat and barley. Mol. Plant-Microbe Interact. 2004, 17, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhang, Z.; Andersen, C.H.; Schmelzer, E.; Gregersen, P.L.; Collinge, D.B.; Smedegaard-Petersen, V.; Thordal-Christensen, H. An epidermis/papilla-specific oxalate oxidase-like protein in the defence response of barley attacked by the powdery mildew fungus. Plant Mol. Biol. 1998, 36, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Gucciardo, S.; Wisniewski, J.-P.; Brewin, N.J.; Bornemann, S. A germin-like protein with superoxide dismutase activity in pea nodules with high protein sequence identity to a putative rhicadhesin receptor. J. Exp. Bot. 2007, 58, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, P.; Christoffel, A.; Dudler, R. Transient expression of members of the germin-like gene family in epidermal cells of wheat confers disease resistance. Plant J. 1999, 20, 541–552. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, G.; Bäumlein, H.; Mock, H.-P.; Himmelbach, A.; Schweizer, P. The multigene family encoding germin-like proteins of barley. Regulation and function in basal host resistance. Plant Physiol. 2006, 142, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, D.; Able, A.J.; Dry, I.B. Induction of a grapevine germin-like protein (VvGLP3) gene is closely linked to the site of Erysiphe necator infection: A possible role in defense? Mol. Plant-Microbe Interact. 2007, 20, 1112–1125. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L. Pathogenesis-related proteins. Plant Mol. Biol. 1985, 4, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Wolski, E.A.; Maldonado, S.; Daleo, G.R.; Andreu, A.B. A novel α-1,3-glucan elicits plant defense responses in potato and induces protection against rhizoctonia solani AG-3 and Fusarium solani f. Sp. eumartii. Physiol. Mol. Plant Pathol. 2006, 69, 93–103. [Google Scholar] [CrossRef]
- Shibuya, N.; Minami, E. Oligosaccharide signalling for defence responses in plant. Physiol. Mol. Plant Pathol. 2001, 59, 223–233. [Google Scholar] [CrossRef]
- Giannakis, C.; Bucheli, C.; Skene, K.; Robinson, S.; Scott, N.S. Chitinase and β-1,3-glucanase in grapevine leaves: A possible defence against powdery mildew infection. Aust. J. Grape Wine Res. 1998, 4, 14–22. [Google Scholar] [CrossRef]
- Libault, M.; Wan, J.; Czechowski, T.; Udvardi, M.; Stacey, G. Identification of 118 arabidopsis transcription factor and 30 ubiquitin-ligase genes responding to chitin, a plant-defense elicitor. Mol. Plant-Microbe Interact. 2007, 20, 900–911. [Google Scholar] [CrossRef] [PubMed]
- Rushton, P.J.; Somssich, I.E.; Ringler, P.; Shen, Q.J. WRKY transcription factors. Trends Plant Sci. 2010, 15, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.P.; Roccaro, M.; Schön, M.; Logemann, E.; Somssich, I.E. Transcriptional reprogramming regulated by WRKY18 and WRKY40 facilitates powdery mildew infection of arabidopsis. Plant J. 2010, 64, 912–923. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Chen, C.; Fan, B.; Chen, Z. Physical and functional interactions between pathogen-induced arabidopsis WRKY18, WRKY40, and WRKY60 transcription factors. Plant Cell 2006, 18, 1310–1326. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.-H.; Saijo, Y.; Mauch, S.; Biskup, C.; Bieri, S.; Keller, B.; Seki, H.; Ülker, B.; Somssich, I.E.; Schulze-Lefert, P. Nuclear activity of MLA immune receptors links isolate-specific and basal disease-resistance responses. Science 2007, 315, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Šamaj, J.; Baluška, F.; Voigt, B.; Schlicht, M.; Volkmann, D.; Menzel, D. Endocytosis, actin cytoskeleton, and signaling. Plant Physiol. 2004, 135, 1150–1161. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Irani, N.G.; Friml, J. Clathrin-mediated endocytosis: The gateway into plant cells. Curr. Opin. Plant Biol. 2011, 14, 674–682. [Google Scholar] [CrossRef] [PubMed]
- Thoma, S.; Hecht, U.; Kippers, A.; Botella, J.; de Vries, S.; Somerville, C. Tissue-specific expression of a gene encoding a cell wall-localized lipid transfer protein from arabidopsis. Plant Physiol. 1994, 105, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L.; van Strien, E. The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol. Mol. Plant Pathol. 1999, 55, 85–97. [Google Scholar] [CrossRef]
- Roy-Barman, S.; Sautter, C.; Chattoo, B.B. Expression of the lipid transfer protein Ace-AMP1 in transgenic wheat enhances antifungal activity and defense responses. Transgenic Res. 2006, 15, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gasic, K.; Cammue, B.; Broekaert, W.; Korban, S.S. Transgenic rose lines harboring an antimicrobial protein gene, Ace-AMP1, demonstrate enhanced resistance to powdery mildew (Sphaerotheca pannosa). Planta 2003, 218, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Cao, A.; Xing, L.; Wang, X.; Yang, X.; Wang, W.; Sun, Y.; Qian, C.; Ni, J.; Chen, Y.; Liu, D. Serine/threonine kinase gene Stpk-V, a key member of powdery mildew resistance gene Pm21, confers powdery mildew resistance in wheat. Proc. Natl. Acad. Sci. USA 2011, 108, 7727–7732. [Google Scholar] [CrossRef] [PubMed]
- Pedley, K.F.; Martin, G.B. Molecular basis of Pto-mediated resistance to bacterial speck disease in tomato. Annu. Rev. Phytopathol. 2003, 41, 215–243. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Shah, J.; Klessig, D.F. Signal perception and transduction in plant defense responses. Genes Dev. 1997, 11, 1621–1639. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.-S.; Han, M.; Lee, S.-K.; Cho, J.-I.; Ryoo, N.; Heu, S.; Lee, Y.-H.; Bhoo, S.H.; Wang, G.-L.; Hahn, T.-R. A comprehensive expression analysis of the WRKY gene superfamily in rice plants during defense response. Plant Cell Rep. 2006, 25, 836–847. [Google Scholar] [CrossRef] [PubMed]
- Trognitz, F.; Manosalva, P.; Gysin, R.; Niño-Liu, D.; Simon, R.; del Rosario Herrera, M.; Trognitz, B.; Ghislain, M.; Nelson, R. Plant defense genes associated with quantitative resistance to potato late blight in Solanum phureja × Dihaploid S. tuberosum hybrids. Mol. Plant-Microbe Interact. 2002, 15, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Bézier, A.; Lambert, B.; Baillieul, F. Study of defense-related gene expression in grapevine leaves and berries infected with Botrytis cinerea. Eur. J. Plant Pathol. 2002, 108, 111–120. [Google Scholar] [CrossRef]
- Dao, T.; Linthorst, H.; Verpoorte, R. Chalcone synthase and its functions in plant resistance. Phytochem. Rev. 2011, 10, 397–412. [Google Scholar] [CrossRef] [PubMed]
- Mohanta, T.K.; Occhipinti, A.; Zebelo, S.A.; Foti, M.; Fliegmann, J.; Bossi, S.; Maffei, M.E.; Bertea, C.M. Ginkgo biloba responds to herbivory by activating early signaling and direct defenses. PLoS ONE 2012, 7, e32822. [Google Scholar] [CrossRef] [PubMed]
- Pezet, R.; Gindro, K.; Viret, O.; Spring, J.-L. Glycosylation and oxidative dimerization of resveratrol are respectively associated to sensitivity and resistance of grapevine cultivars to downy mildew. Physiol. Mol. Plant Pathol. 2004, 65, 297–303. [Google Scholar] [CrossRef]
- Treutter, D. Significance of flavonoids in plant resistance: A review. Environ. Chem. Lett. 2006, 4, 147–157. [Google Scholar] [CrossRef]
- Persans, M.W.; Wang, J.; Schuler, M.A. Characterization of maize cytochrome p450 monooxygenases induced in response to safeners and bacterial pathogens. Plant Physiol. 2001, 125, 1126–1138. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Bakht, S.; Qin, B.; Leggett, M.; Hemmings, A.; Mellon, F.; Eagles, J.; Werck-Reichhart, D.; Schaller, H.; Lesot, A. A different function for a member of an ancient and highly conserved cytochrome p450 family: From essential sterols to plant defense. Proc. Natl. Acad. Sci. USA 2006, 103, 18848–18853. [Google Scholar] [CrossRef] [PubMed]
- Whitbred, J.M.; Schuler, M.A. Molecular characterization of cyp73a9 andcyp82a1 p450 genes involved in plant defense in pea. Plant Physiol. 2000, 124, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-C.; Kim, S.-Y.; Paek, K.-H.; Choi, D.; Park, J.M. Suppression of cacyp1, a novel cytochrome p450 gene, compromises the basal pathogen defense response of pepper plants. Biochem. Biophys. Res. Commun. 2006, 345, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Narusaka, M.; Seki, M.; Umezawa, T.; Ishida, J.; Nakajima, M.; Enju, A.; Shinozaki, K. Crosstalk in the responses to abiotic and biotic stresses in arabidopsis: Analysis of gene expression in cytochrome p450 gene superfamily by cDNA microarray. Plant Mol. Biol. 2004, 55, 327–342. [Google Scholar] [CrossRef] [PubMed]
- Deák, M.; Horváth, G.V.; Davletova, S.; Török, K.; Sass, L.; Vass, I.; Barna, B.; Király, Z.; Dudits, D. Plants ectopically expressing the ironbinding protein, ferritin, are tolerant to oxidative damage and pathogens. Nat. Biotechnol. 1999, 17, 192–196. [Google Scholar] [PubMed]
- Arnaud, N.; Murgia, I.; Boucherez, J.; Briat, J.-F.; Cellier, F.; Gaymard, F. An iron-induced nitric oxide burst precedes ubiquitin-dependent protein degradation for arabidopsis atfer1 ferritin gene expression. J. Biol. Chem. 2006, 281, 23579–23588. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, S.; Yang, X.; Zhang, M. Chloroplast-located BjFer1 together with anti-oxidative genes alleviate hydrogen peroxide and hydroxyl radical injury in cytoplasmic male-sterile Brassica juncea. Mol. Biol. Rep. 2012, 39, 4169–4176. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Greenshields, D.L.; Sammynaiken, R.; Hirji, R.N.; Selvaraj, G.; Wei, Y. Targeted alterations in iron homeostasis underlie plant defense responses. J. Cell Sci. 2007, 120, 596–605. [Google Scholar] [CrossRef] [PubMed]
- Keogh, R.; Deverall, B.; McLeod, S. Comparison of histological and physiological responses to Phakopsora pachyrhizi in resistant and susceptible soybean. Trans. Br. Mycol. Soc. 1980, 74, 329–333. [Google Scholar] [CrossRef]
- Wolf, G.; Fric, F. A rapid staining method for Erysiphe graminis f. Sp. hordei in and on whole barley leaves with a protein-specific dye. Phytopathology 1981, 71, 596–598. [Google Scholar]
- Xu, J.; Aileni, M.; Abbagani, S.; Zhang, P. A reliable and efficient method for total RNA isolation from various members of spurge family (Euphorbiaceae). Phytochem. Anal. 2010, 21, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Zhu, W.; Zhang, X.; Guo, Z.; O'Connell, R.P.; Averboukh, L.; Wang, F.; Pardee, A.B. Differential display using one-base anchored oligo-dT primers. Nucleic Acids Res. 1994, 22, 5763. [Google Scholar] [CrossRef] [PubMed]
- Minglin, L.; Yuxiu, Z.; Tuanyao, C. Identification of genes up-regulated in response to Cd exposure in Brassica juncea L. Gene 2005, 363, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yin, H.; Wang, W.; Mi, Q.; Liao, X.; Li, X. Identification of Cd-responsive genes of Solanum nigrum seedlings through differential display. Plant Mol. Biol. Rep. 2009, 27, 563–569. [Google Scholar] [CrossRef]
- Rahman, A.Y.A.; Usharraj, A.O.; Misra, B.B.; Thottathil, G.P.; Jayasekaran, K.; Feng, Y.; Hou, S.; Ong, S.Y.; Ng, F.L.; Lee, L.S. Draft genome sequence of the rubber tree Hevea brasiliensis. BMC Genom. 2013, 14, 75. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Qin, Y.; Xiao, X.; Tang, C. Screening of valid reference genes for real-time RT-PCR data normalization in Hevea brasiliensis and expression validation of a sucrose transporter gene HbSUT3. Plant Sci. 2011, 181, 132–139. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Bi, Z.; Di, R.; Liang, P.; He, Q.; Liu, W.; Miao, W.; Zheng, F. Identification of Powdery Mildew Responsive Genes in Hevea brasiliensis through mRNA Differential Display. Int. J. Mol. Sci. 2016, 17, 181. https://doi.org/10.3390/ijms17020181
Li X, Bi Z, Di R, Liang P, He Q, Liu W, Miao W, Zheng F. Identification of Powdery Mildew Responsive Genes in Hevea brasiliensis through mRNA Differential Display. International Journal of Molecular Sciences. 2016; 17(2):181. https://doi.org/10.3390/ijms17020181
Chicago/Turabian StyleLi, Xiang, Zhenghong Bi, Rong Di, Peng Liang, Qiguang He, Wenbo Liu, Weiguo Miao, and Fucong Zheng. 2016. "Identification of Powdery Mildew Responsive Genes in Hevea brasiliensis through mRNA Differential Display" International Journal of Molecular Sciences 17, no. 2: 181. https://doi.org/10.3390/ijms17020181
APA StyleLi, X., Bi, Z., Di, R., Liang, P., He, Q., Liu, W., Miao, W., & Zheng, F. (2016). Identification of Powdery Mildew Responsive Genes in Hevea brasiliensis through mRNA Differential Display. International Journal of Molecular Sciences, 17(2), 181. https://doi.org/10.3390/ijms17020181







