Dual Function of Glucosamine in Gelatin/Hyaluronic Acid Cryogel to Modulate Scaffold Mechanical Properties and to Maintain Chondrogenic Phenotype for Cartilage Tissue Engineering
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Cryogels
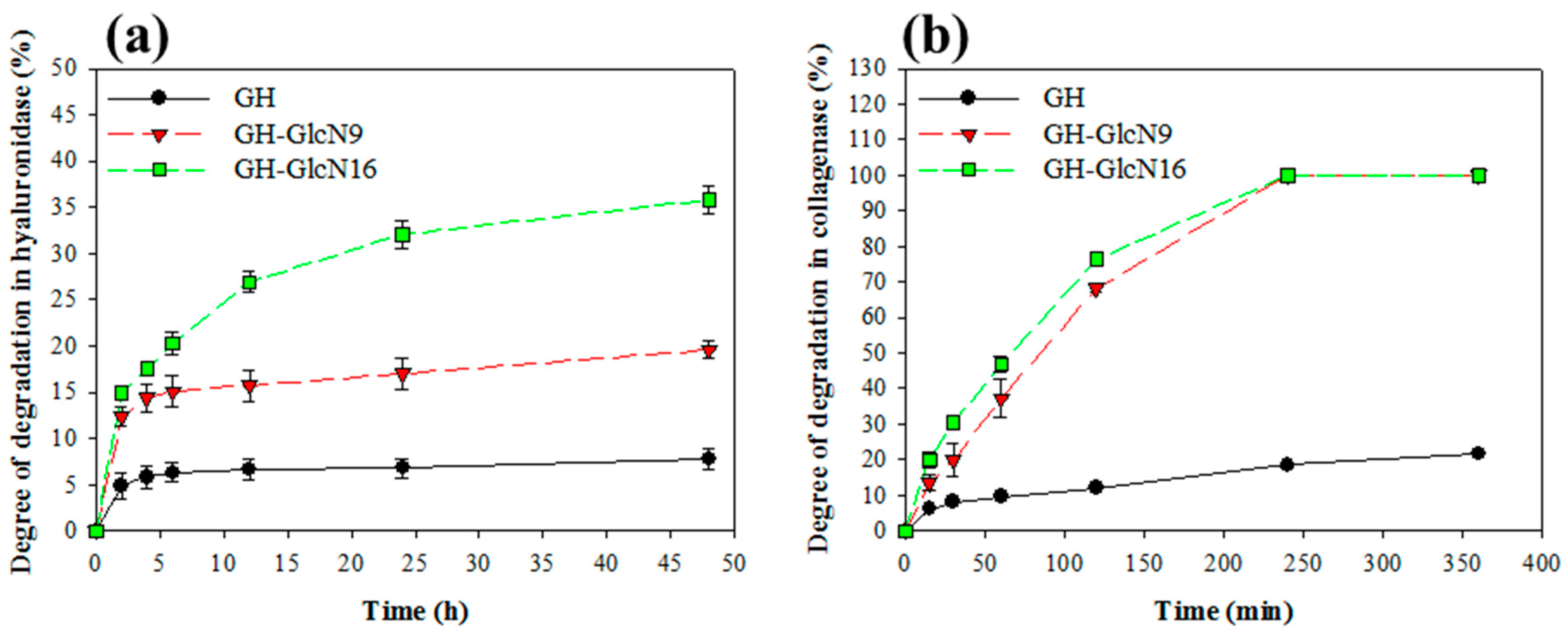
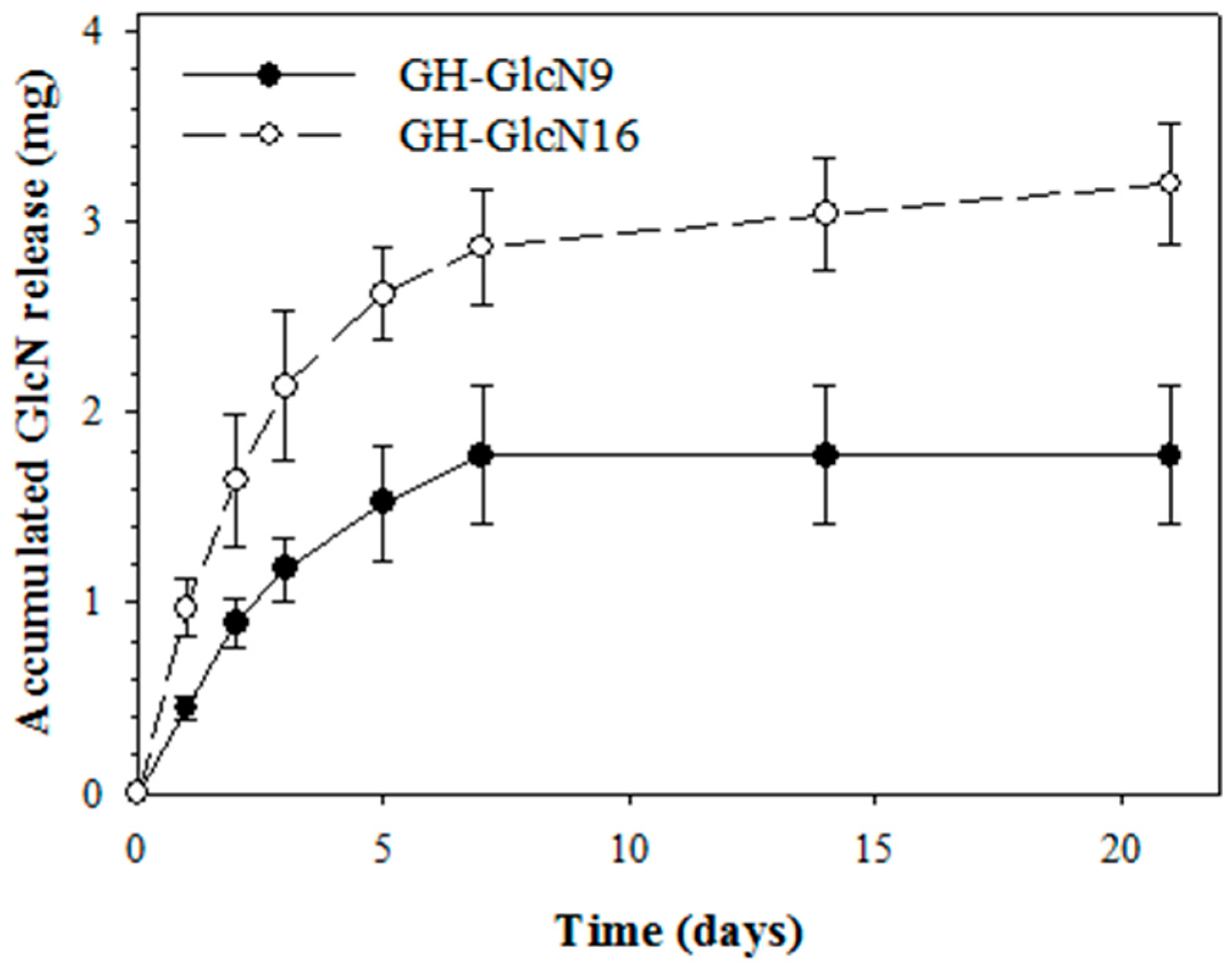
2.2. Cryogel Degradation and GlcN Release
2.3. Mechanical Properties of Cryogels
2.3.1. Compression Mechanical Tests
2.3.2. Stress Relaxation Tests
2.3.3. Dynamic Mechanical Analysis
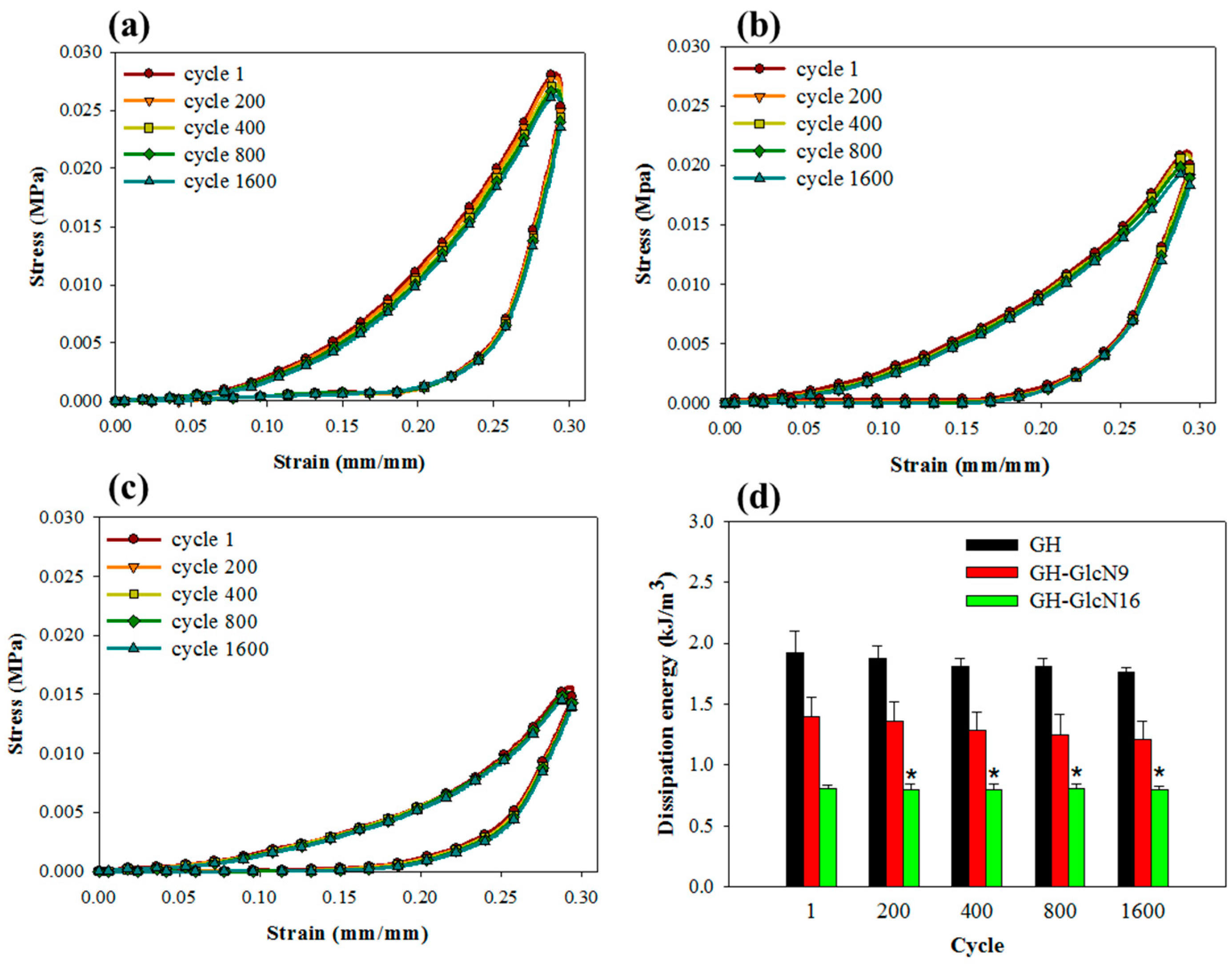
2.3.4. Cyclic Compression Analysis
2.4. In Vitro Experiments
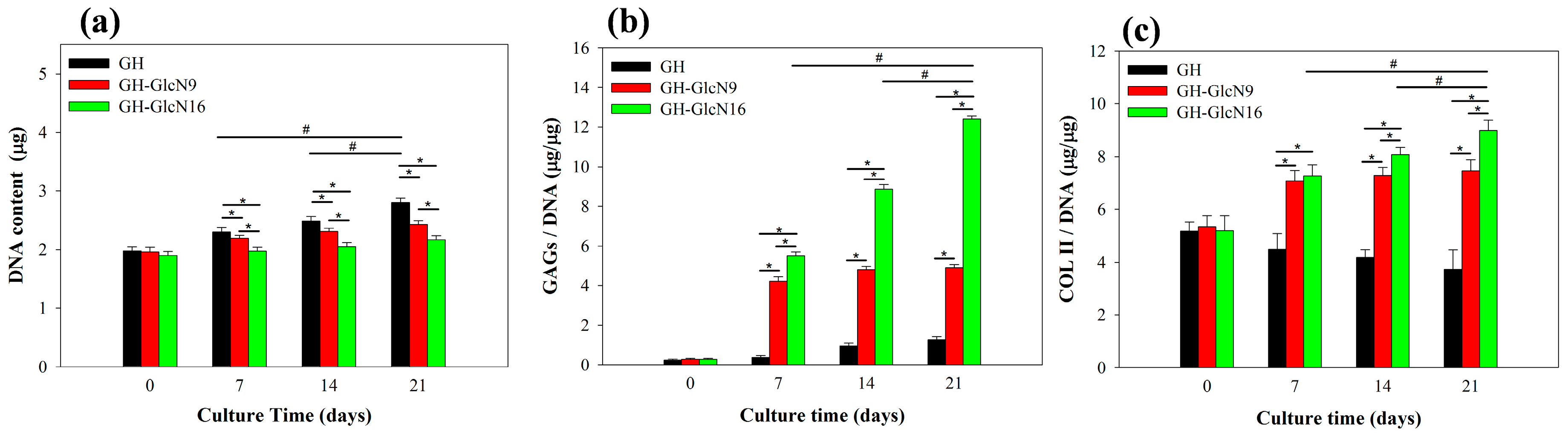
2.4.1. Cell Proliferation
2.4.2. GAGs and COL II Production
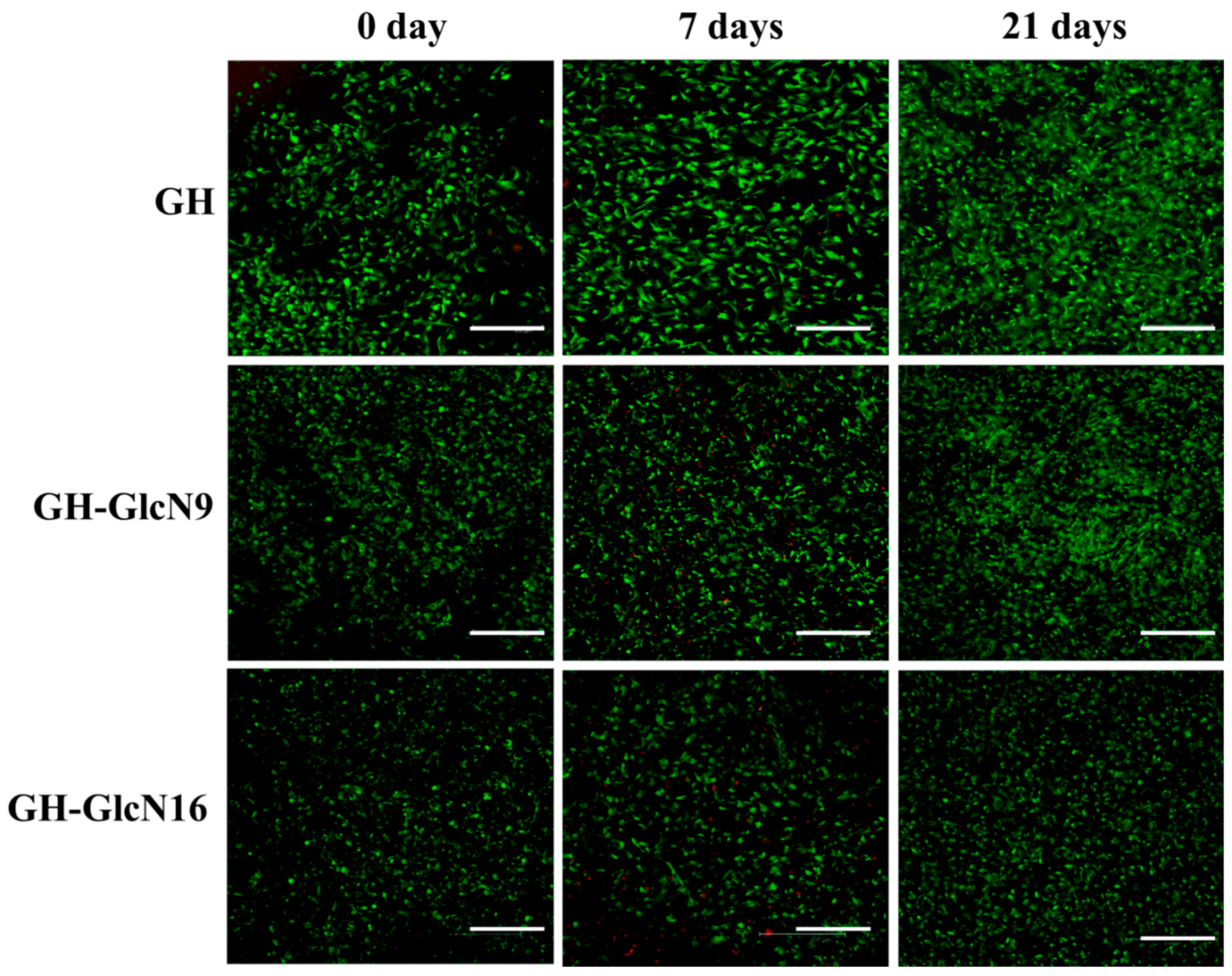
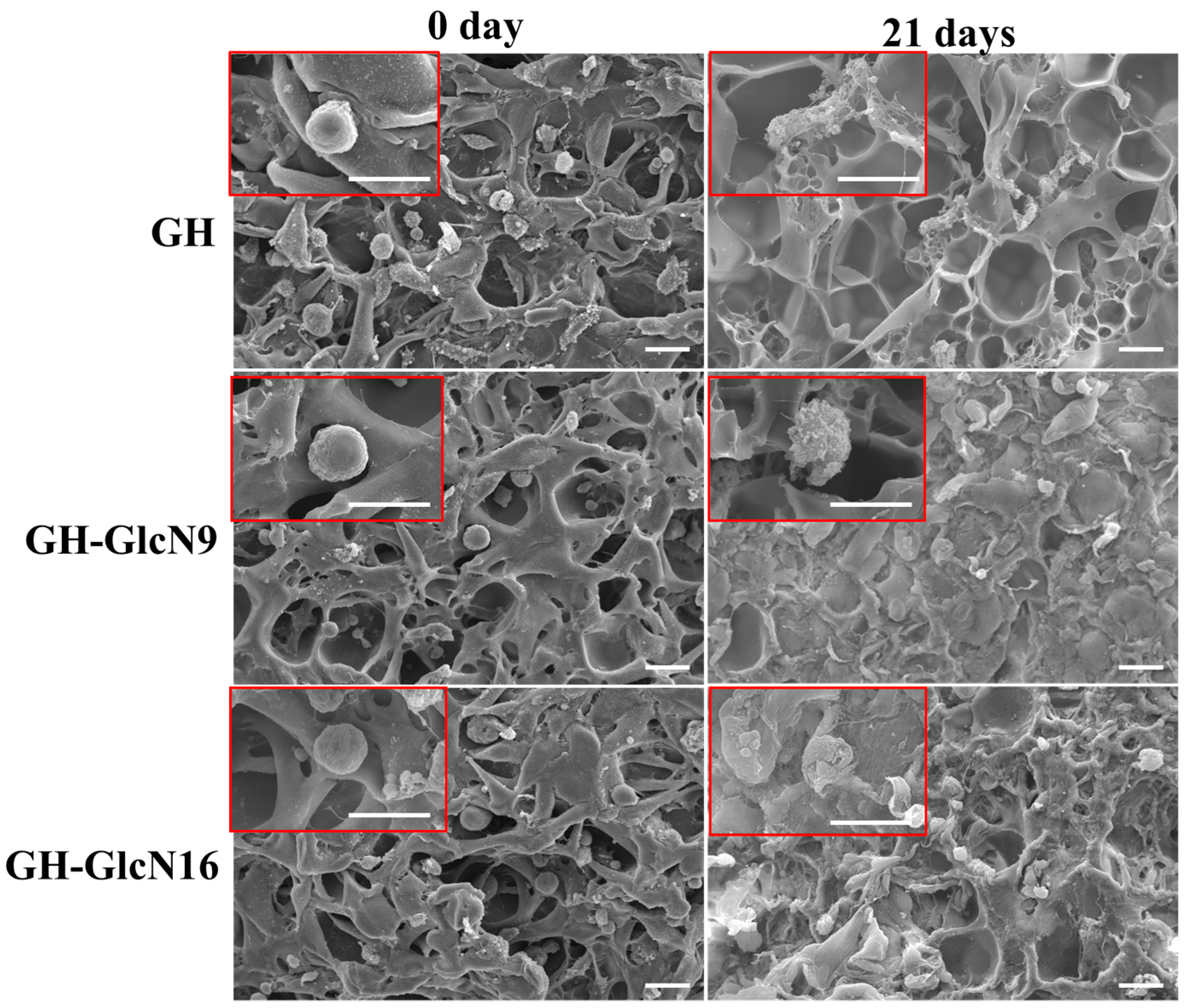
2.4.3. Live/Dead Staining and SEM Observation
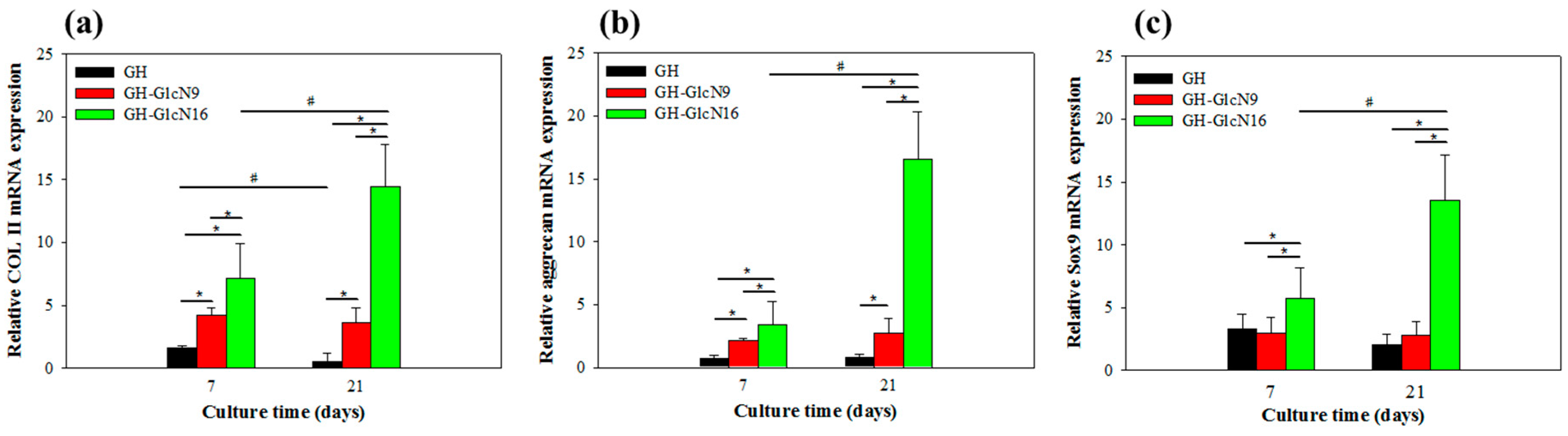
2.4.4. Gene Expression
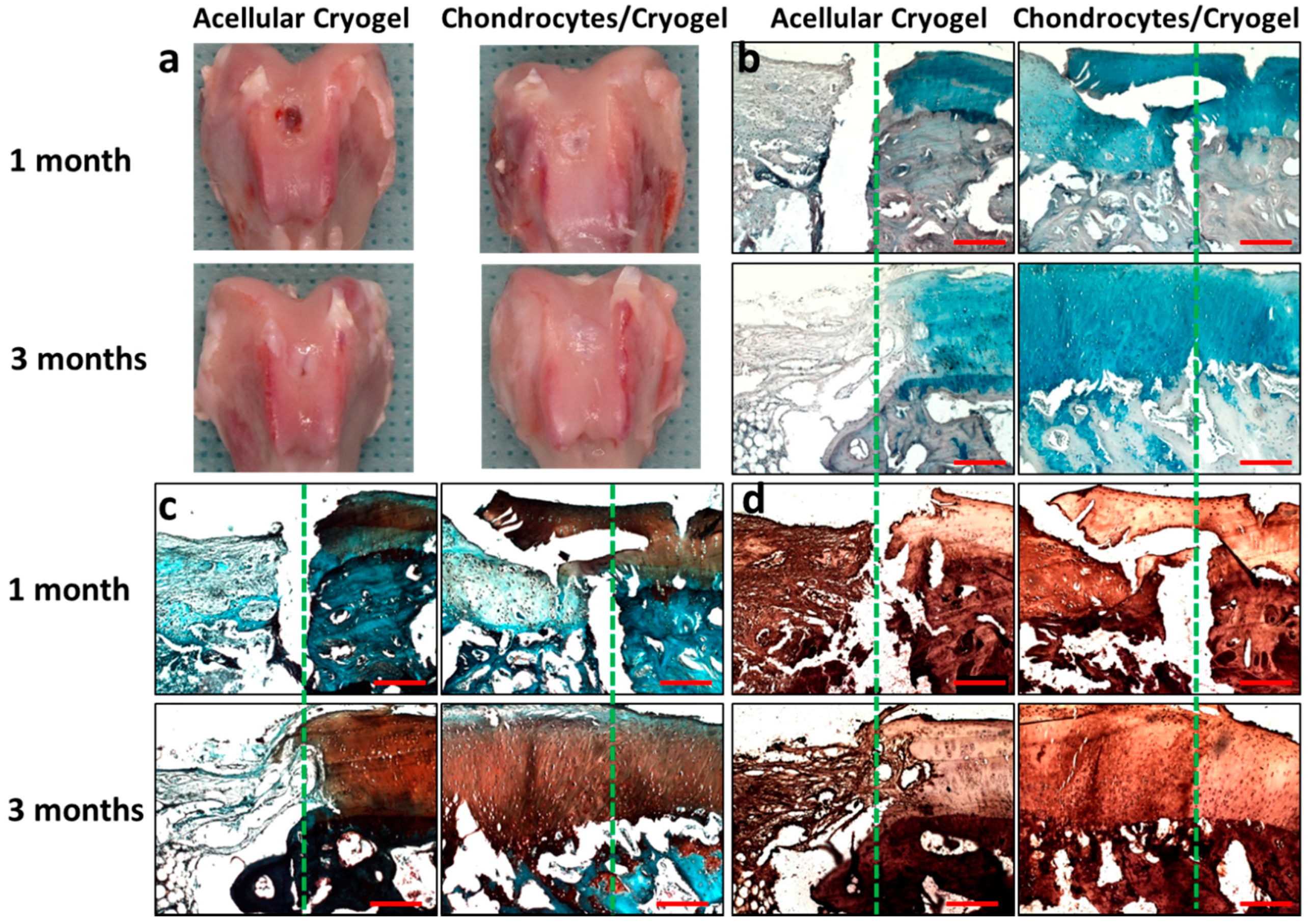
2.5. In Vivo Animal Studies
3. Experimental Section
3.1. Materials
3.2. Preparation of GH, GH-GlcN9, and GH-GlcN16 Cryogels
3.3. Characterization of Cryogels
3.4. In Vitro Cell Culture
3.5. In Vivo Animal Study
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hubka, K.M.; Dahlin, R.L.; Meretoja, V.V.; Kasper, F.K.; Mikos, A.G. Enhancing chondrogenic phenotype for cartilage tissue engineering: Monoculture and coculture of articular chondrocytes and mesenchymal stem cells. Tissue Eng. B Rev. 2014, 20, 641–654. [Google Scholar] [CrossRef] [PubMed]
- Annabi, N.; Nichol, J.W.; Zhong, X.; Ji, C.; Koshy, S.; Khademhosseini, A.; Dehghani, F. Controlling the porosity and microarchitecture of hydrogels for tissue engineering. Tissue Eng. B Rev. 2010, 16, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Plieva, F.M.; Galaev, I.Y.; Mattiasson, B. Macroporous gels prepared at subzero temperatures as novel materials for chromatography of particulate-containing fluids and cell culture applications. J. Sep. Sci. 2007, 30, 1657–1671. [Google Scholar] [CrossRef] [PubMed]
- Lozinsky, V.I.; Plieva, F.M.; Galaev, I.Y.; Mattiasson, B. The potential of polymeric cryogels in bioseparation. Bioseparation 2001, 10, 163–188. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Srivastava, A. Cell separation using cryogel-based affinity chromatography. Nat. Protoc. 2010, 5, 1737–1747. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.H.; Liao, H.T.; Chen, J.P. Preparation and characterization of gelatin/hyaluronic acid cryogels for adipose tissue engineering: In vitro and in vivo studies. Acta Biomater. 2013, 9, 9012–9026. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.T.; Shalumon, K.T.; Chang, K.H.; Sheu, C.; Chen, J.P. Investigation of synergistic effects of inductive and conductive factors in gelatin-based cryogels for bone tissue engineering. J. Mater. Chem. B 2016, 4, 1827–1841. [Google Scholar] [CrossRef]
- Hwang, Y.; Sangaj, N.; Varghese, S. Interconnected macroporous poly(ethylene glycol) cryogels as a cell scaffold for cartilage tissue engineering. Tissue Eng. A 2010, 16, 3033–3041. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Bhat, S.; Nayak, V.; Kumar, A. Efficacy of supermacroporous poly(ethylene glycol)-gelatin cryogel matrix for soft tissue engineering applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 47, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Yin, B.; Ma, P.; Chen, J.; Wang, H.; Wu, G.; Li, B.; Li, Q.; Huang, Z.; Qiu, G.; Wu, Z. Hybrid macro-porous titanium ornamented by degradable 3D Gel/nHA micro-scaffolds for bone tissue regeneration. Int. J. Mol. Sci. 2016, 17, 575. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Liu, H.C.; Lin, C.C.; Chou, C.H.; Lin, F.H. Gelatin-chondroitin-hyaluronan tri-copolymer scaffold for cartilage tissue engineering. Biomaterials 2003, 24, 4853–4858. [Google Scholar] [CrossRef]
- Ichimaru, S.; Nakagawa, S.; Arai, Y.; Kishida, T.; Shin-Ya, M.; Honjo, K.; Tsuchida, S.; Inoue, H.; Fujiwara, H.; Shimomura, S.; et al. Hypoxia potentiates anabolic effects of exogenous hyaluronic acid in rat articular cartilage. Int. J. Mol. Sci. 2016, 17, 1013. [Google Scholar] [CrossRef] [PubMed]
- Mabuchi, K.; Obara, T.; Ikegami, K.; Yamaguchi, T.; Kanayama, T. Molecular weight independence of the effect of additive hyaluronic acid on the lubricating characteristics in synovial joints with experimental deterioration. Clin. Biomech. (Bristol, Avon) 1999, 14, 352–356. [Google Scholar] [CrossRef]
- Suzuki, A.; Tanimoto, K.; Ohno, S.; Nakatani, Y.; Honda, K.; Tanaka, N.; Doi, T.; Ohno-Nakahara, M.; Yoneno, K.; Ueki, M.; et al. The metabolism of hyaluronan in cultured rabbit growth plate chondrocytes during differentiation. Biochim. Biophys. Acta 2005, 1743, 57–63. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yoo, H.S.; Lee, E.A.; Yoon, J.J.; Park, T.G. Hyaluronic acid modified biodegradable scaffolds for cartilage tissue engineering. Biomaterials 2005, 26, 1925–1933. [Google Scholar] [CrossRef] [PubMed]
- Angele, P.; Müller, R.; Schumann, D.; Englert, C.; Zellner, J.; Johnstone, B.; Yoo, J.; Hammer, J.; Fierlbeck, J.; Angele, M.K.; et al. Characterization of esterified hyaluronan-gelatin polymer composites suitable for chondrogenic differentiation of mesenchymal stem cells. J. Biomed. Mater. Res. A 2009, 91A, 416–427. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Wu, J.; Lao, L.; Gao, C. Gelatin/chitosan/hyaluronan scaffold integrated with PLGA microspheres for cartilage tissue engineering. Acta Biomater. 2009, 5, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, C.; Berner, A.; Koch, M.; Krutsch, W.; Kujat, R.; Angele, P.; Nerlich, M.; Zellner, J. Higher ratios of hyaluronic acid enhance chondrogenic differentiation of human mscs in a hyaluronic acid-gelatin composite scaffold. Materials 2016, 9, 381. [Google Scholar] [CrossRef]
- Kuo, C.Y.; Chen, C.H.; Hsiao, C.Y.; Chen, J.P. Incorporation of chitosan in biomimetic gelatin/chondroitin-6-sulfate/hyaluronan cryogel for cartilage tissue engineering. Carbohydr. Polym. 2015, 117, 722–730. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.E.; LaValley, M.P.; Gulin, J.P.; Felson, D.T. Glucosamine and chondroitin for treatment of osteoarthritis: A systematic quality assessment and meta-analysis. J. Am. Med. Assoc. 2000, 283, 1469–1475. [Google Scholar] [CrossRef]
- Rovati, L.C. Clinical research in osteoarthritis: Design and results of short-term and long-term trials with disease-modifying drugs. Int. J. Tissue React. 1992, 14, 243–251. [Google Scholar] [PubMed]
- Morelli, V.; Naquin, C.; Weaver, V. Alternative therapies for traditional disease states: Osteoarthritis. Am. Fam. Phys. 2003, 67, 339–344. [Google Scholar]
- Barclay, T.S.; Tsourounis, C.; McCart, G.M. Glucosamine. Ann. Pharmacother. 1998, 32, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Derfoul, A.; Miyoshi, A.D.; Freeman, D.E.; Tuan, R.S. Glucosamine promotes chondrogenic phenotype in both chondrocytes and mesenchymal stem cells and inhibits MMP-13 expression and matrix degradation. Osteoarthr. Cartil. 2007, 15, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Dalirfardouei, R.; Karimi, G.; Jamialahmadi, K. Molecular mechanisms and biomedical applications of glucosamine as a potential multifunctional therapeutic agent. Life Sci. 2016, 152, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Li, B.; Yang, J.; Ni, Y.; Teng, Y.; Guo, L.; Fan, H.; Fan, Y.; Zhang, X. Effects of composition and mechanical property of injectable collagen I/II composite hydrogels on chondrocyte behaviors. Tissue Eng. A 2016, 22, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Murphy, W.L.; Dennis, R.G.; Kileny, J.L.; Mooney, D.J. Salt fusion: An approach to improve pore interconnectivity within tissue engineering scaffolds. Tissue Eng. 2002, 8, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Park, S.N.; Lee, H.J.; Lee, K.H.; Suh, H. Biological characterization of EDC-crosslinked collagen-hyaluronic acid matrix in dermal tissue restoration. Biomaterials 2003, 24, 1631–1641. [Google Scholar] [CrossRef]
- Yao, C.H.; Liu, B.S.; Chang, C.J.; Hsu, S.H.; Chen, Y.S. Preparation of networks of gelatin and genipin as degradable biomaterials. Mater. Chem. Phys. 2004, 83, 204–208. [Google Scholar] [CrossRef]
- Schuh, E.; Hofmann, S.; Stok, K.; Notbohm, H.; Muller, R.; Rotter, N. Chondrocyte redifferentiation in 3D: The effect of adhesion site density and substrate elasticity. J. Biomed. Mater. Res. A 2012, 100, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, G.D.; Skaalure, S.C.; Bryant, S.J. Gel structure has an impact on pericellular and extracellular matrix deposition, which subsequently alters metabolic activities in chondrocyte-laden peg hydrogels. Acta Biomater. 2011, 7, 492–504. [Google Scholar] [CrossRef] [PubMed]
- Callahan, L.A.; Ganios, A.M.; Childers, E.P.; Weiner, S.D.; Becker, M.L. Primary human chondrocyte extracellular matrix formation and phenotype maintenance using RGD-derivatized PEGDMhydrogels possessing a continuous Young’s modulus gradient. Acta Biomater. 2013, 9, 6095–6104. [Google Scholar] [CrossRef] [PubMed]
- Schuh, E.; Kramer, J.; Rohwedel, J.; Notbohm, H.; Muller, R.; Gutsmann, T.; Rotter, N. Effect of matrix elasticity on the maintenance of the chondrogenic phenotype. Tissue Eng. A 2010, 16, 1281–1290. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Huebsch, N.; Mooney, D.J.; Suo, Z. Stress-relaxation behavior in gels with ionic and covalent crosslinks. J. Appl. Phys. 2010, 107, 063509. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, O.; Gu, L.; Klumpers, D.; Darnell, M.; Bencherif, S.A.; Weaver, J.C.; Huebsch, N.; Lee, H.-P.; Lippens, E.; Duda, G.N.; et al. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat. Mater. 2016, 15, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, S.; Li, J.; Wang, X.; Zhang, J.; Kawazoe, N.; Chen, G. 3D culture of chondrocytes in gelatin hydrogels with different stiffness. Polymers 2016, 8, 269. [Google Scholar] [CrossRef]
- Torres, F.G.; Nazhat, S.N.; Sheikh Md Fadzullah, S.H.; Maquet, V.; Boccaccini, A.R. Mechanical properties and bioactivity of porous PLGA/TiO2 nanoparticle-filled composites for tissue engineering scaffolds. Compos. Sci. Technol. 2007, 67, 1139–1147. [Google Scholar] [CrossRef]
- Chameettachal, S.; Murab, S.; Vaid, R.; Midha, S.; Ghosh, S. Effect of visco-elastic silk-chitosan microcomposite scaffolds on matrix deposition and biomechanical functionality for cartilage tissue engineering. J. Tissue Eng. Regen. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Nicoll, S.B.; Mauck, R.L.; Ateshian, G.A. Cartilage mechanical response under dynamic compression at physiological stress levels following collagenase digestion. Ann. Biomed. Eng. 2008, 36, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.K.; Li, Z.; Woo, S.L. Dynamic behavior of a biphasic cartilage model under cyclic compressive loading. J. Biomech. 1995, 28, 357–364. [Google Scholar] [CrossRef]
- Varga, F.; Drzik, M.; Handl, M.; Chlpik, J.; Kos, P.; Filova, E.; Rampichova, M.; Necas, A.; Trc, T.; Amler, E. Biomechanical characterization of cartilages by a novel approach of blunt impact testing. Physiol. Res. 2007, 56 (Suppl. 1), S61–S68. [Google Scholar] [PubMed]
- Abdel-Sayed, P.; Darwiche, S.E.; Kettenberger, U.; Pioletti, D.P. The role of energy dissipation of polymeric scaffolds in the mechanobiological modulation of chondrogenic expression. Biomaterials 2014, 35, 1890–1897. [Google Scholar] [CrossRef] [PubMed]
- Hwang, N.S.; Varghese, S.; Theprungsirikul, P.; Canver, A.; Elisseeff, J. Enhanced chondrogenic differentiation of murine embryonic stem cells in hydrogels with glucosamine. Biomaterials 2006, 27, 6015–6023. [Google Scholar] [CrossRef] [PubMed]
- Terry, D.E.; Rees-Milton, K.; Smith, P.; Carran, J.; Pezeshki, P.; Woods, C.; Greer, P.; Anastassiades, T.P. N-acylation of glucosamine modulates chondrocyte growth, proteoglycan synthesis, and gene expression. J. Rheumatol. 2005, 32, 1775–1786. [Google Scholar] [PubMed]
- Varghese, S.; Theprungsirikul, P.; Sahani, S.; Hwang, N.; Yarema, K.J.; Elisseeff, J.H. Glucosamine modulates chondrocyte proliferation, matrix synthesis, and gene expression. Osteoarthr. Cartil. 2007, 15, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Terry, D.E.; Rees-Milton, K.; Pruss, C.; Hopwood, J.; Carran, J.; Anastassiades, T.P. Modulation of articular chondrocyte proliferation and anionic glycoconjugate synthesis by glucosamine (GlcN), N-acetyl GlcN (GlcNAc), GlcN sulfate salt (GlcN.S) and covalent glucosamine sulfates (GlcN-SO4). Osteoarthr. Cartil. 2007, 15, 946–956. [Google Scholar] [CrossRef] [PubMed]
- Noyszewski, E.A.; Wroblewski, K.; Dodge, G.R.; Kudchodkar, S.; Beers, J.; Sarma, A.V.; Reddy, R. Preferential incorporation of glucosamine into the galactosamine moieties of chondroitin sulfates in articular cartilage explants. Arthritis Rheumatol. 2001, 44, 1089–1095. [Google Scholar] [CrossRef]
- Mroz, P.J.; Silbert, J.E. Use of 3H-glucosamine and 35S-sulfate with cultured human chondrocytes to determine the effect of glucosamine concentration on formation of chondroitin sulfate. Arthritis Rheumatol. 2004, 50, 3574–3579. [Google Scholar] [CrossRef] [PubMed]
- Hindle, P.; Hall, A.C.; Biant, L.C. Viability of chondrocytes seeded onto a collagen I/III membrane for matrix-induced autologous chondrocyte implantation. J. Orthop. Res. 2014, 32, 1495–1502. [Google Scholar] [CrossRef] [PubMed]
- Glowacki, J.; Trepman, E.; Folkman, J. Cell shape and phenotypic expression in chondrocytes. Proc. Soc. Exp. Biol. Med. 1983, 172, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Cheah, K.S.; Lau, E.T.; Au, P.K.; Tam, P.P. Expression of the mouse alpha 1(II) collagen gene is not restricted to cartilage during development. Development 1991, 111, 945–953. [Google Scholar] [PubMed]
- Lefebvre, V.; Huang, W.; Harley, V.R.; Goodfellow, P.N.; de Crombrugghe, B. Sox9 is a potent activator of the chondrocyte-specific enhancer of the pro alpha1(II) collagen gene. Mol. Cell. Biol. 1997, 17, 2336–2346. [Google Scholar] [CrossRef] [PubMed]
- Stoppoloni, D.; Politi, L.; Leopizzi, M.; Gaetani, S.; Guazzo, R.; Basciani, S.; Moreschini, O.; de Santi, M.; Scandurra, R.; Scotto d’Abusco, A. Effect of glucosamine and its peptidyl-derivative on the production of extracellular matrix components by human primary chondrocytes. Osteoarthr. Cartil. 2015, 23, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.W.; Qin, L.; Tai, J.K.; Lee, S.K.; Leung, K.S.; Chan, K.M. Engineered allogeneic chondrocyte pellet for reconstruction of fibrocartilage zone at bone-tendon junction—A preliminary histological observation. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 70, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Vinatier, C.; Mrugala, D.; Jorgensen, C.; Guicheux, J.; Noel, D. Cartilage engineering: A crucial combination of cells, biomaterials and biofactors. Trends Biotechnol. 2009, 27, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ma, P.X. Poly(alpha-hydroxyl acids)/hydroxyapatite porous composites for bone-tissue engineering. I. Preparation and morphology. J. Biomed. Mater. Res. 1999, 44, 446–455. [Google Scholar] [CrossRef]
- Elson, L.A.; Morgan, W.T.J. A colorimetric method for the determination of glucosamine and chondrosamine. Biochem. J. 1933, 27, 1824–1828. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.L.Y.; Gomez, M.A.; Akeson, W.H. The time and history-dependent viscoelastic properties of the canine medial collateral ligament. J. Biomech. Eng. 1981, 103, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.K.; Campbell, G.; Zhang, Z.F.; Hui, A.J.; Boughner, D.R. Optimizing the tensile properties of polyvinyl alcohol hydrogel for the construction of a bioprosthetic heart valve stent. J. Biomed. Mater. Res. 2002, 63, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.P.; Cheng, T.H. Thermo-responsive chitosan-graft-poly(N-isopropylacrylamide) injectable hydrogel for cultivation of chondrocytes and meniscus cells. Macromol. Biosci. 2006, 6, 1026–1039. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Sah, R.L.; Doong, J.Y.; Grodzinsky, A.J. Fluorometric assay of DNA in cartilage explants using hoechst 33258. Anal. Biochem. 1988, 174, 168–176. [Google Scholar] [CrossRef]
- Enobakhare, B.O.; Bader, D.L.; Lee, D.A. Quantification of sulfated glycosaminoglycans in chondrocyte/alginate cultures, by use of 1,9-dimethylmethylene blue. Anal. Biochem. 1996, 243, 189–191. [Google Scholar] [CrossRef] [PubMed]
- Power, J.; Hernandez, P.; Guehring, H.; Getgood, A.; Henson, F. Intra-articular injection of rhFGF-18 improves the healing in microfracture treated chondral defects in an ovine model. J. Orthop. Res. 2014, 32, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Mainil-Varlet, P.; Aigner, T.; Brittberg, M.; Bullough, P.; Hollander, A.; Hunziker, E.; Kandel, R.; Nehrer, S.; Pritzker, K.; Roberts, S.; et al. Histological assessment of cartilage repair: A report by the histology endpoint committee of the international cartilage repair society (ICRS). J. Bone Jt. Surg. Am. 2003, 85 (Suppl. 2), 45–57. [Google Scholar] [CrossRef]
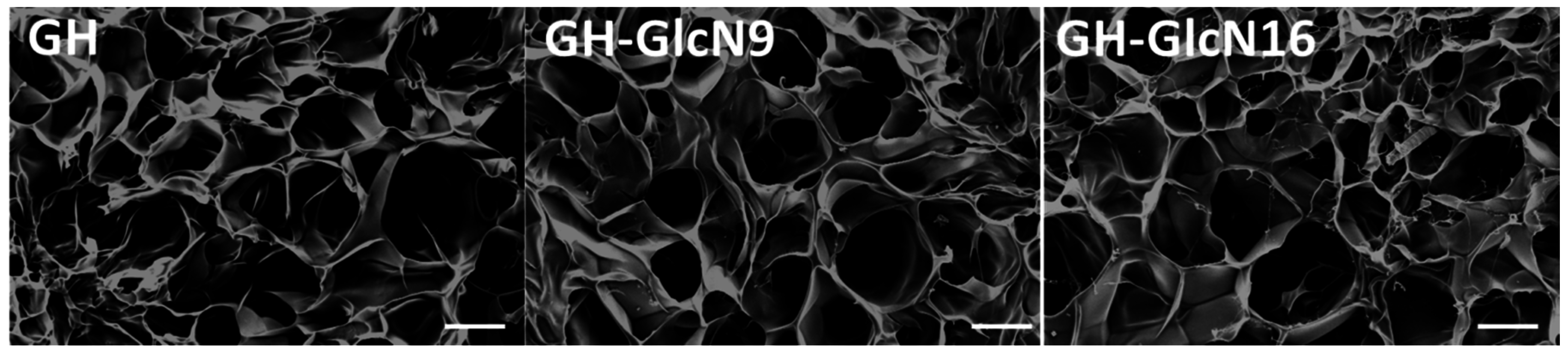

| GH | GH-GlcN9 | GH-GlcN16 | |
|---|---|---|---|
| Pore size (μm) | 103.5 ± 9.4 | 105.6 ± 11.5 | 108.3 ± 13.3 |
| Porosity (%) | 80.9 ± 0.7 | 83.8 ± 1.8 * | 91.1 ± 1.5 *,# |
| Density (mg/cm3) | 134.9 ± 1.2 | 131.5 ± 1.0 * | 127.6 ± 1.3 *,# |
| Swelling ratio | 11.4 ± 0.9 | 19.2 ± 0.6 * | 31.4 ± 1.7 *,# |
| GH | GH-GlcN9 | GH-GlcN16 | |
|---|---|---|---|
| Compressive elastic modulus, ε = 0.1 (kPa) | 45.1 ± 5.3 | 30.3 ± 4.1 * | 24.9 ± 3.6 * |
| Compressive elastic modulus, ε = 0.3 (kPa) | 255.6 ± 77.9 | 156 ± 25.3 | 132.3 ± 19.8 * |
| Compressive strain to failure (%) | 45.7 ± 1.5 | 49 ± 1.0 | 47.7 ± 3.1 |
| Compressive stress to failure (kPa) | 118.6 ± 12.1 | 90.6 ± 8.9 * | 53.3 ± 5.0 *,# |
| Toughness (kJ/m3) | 12.7 ± 1.3 | 10.8 ± 0.8 | 8.2 ± 2.1 * |
| Compression energy (kJ/m3) | 2.5 ± 0.1 | 1.9 ± 0.3 * | 1.3 ± 0.1 *,# |
| Relaxation energy (kJ/m3) | 0.8 ± 0.1 | 0.7 ± 0.2 | 0.5 ± 0.1 * |
| Dissipation energy (kJ/m3) | 1.7 ± 0.1 | 1.2 ± 0.1 * | 0.8 ± 0.1 *,# |
| Percentage of energy dissipation (%) | 68.2 ± 4.4 | 61.3 ± 1.4 * | 59.7 ± 1.6 * |
| Acellular Cryogel | Chondrocytes/Cryogel | |||
|---|---|---|---|---|
| Gross View | Histology | Gross View | Histology | |
| 1 month | 2.83 ± 0.98 | 2.67 ± 0.82 | 5.33 ± 0.82 * | 7.17 ± 1.17 * |
| 3 months | 4.83 ± 0.41 * | 5.50 ± 0.84 * | 8.33 ± 1.03 # | 14.83 ± 2.40 # |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-H.; Kuo, C.-Y.; Wang, Y.-J.; Chen, J.-P. Dual Function of Glucosamine in Gelatin/Hyaluronic Acid Cryogel to Modulate Scaffold Mechanical Properties and to Maintain Chondrogenic Phenotype for Cartilage Tissue Engineering. Int. J. Mol. Sci. 2016, 17, 1957. https://doi.org/10.3390/ijms17111957
Chen C-H, Kuo C-Y, Wang Y-J, Chen J-P. Dual Function of Glucosamine in Gelatin/Hyaluronic Acid Cryogel to Modulate Scaffold Mechanical Properties and to Maintain Chondrogenic Phenotype for Cartilage Tissue Engineering. International Journal of Molecular Sciences. 2016; 17(11):1957. https://doi.org/10.3390/ijms17111957
Chicago/Turabian StyleChen, Chih-Hao, Chang-Yi Kuo, Yan-Jie Wang, and Jyh-Ping Chen. 2016. "Dual Function of Glucosamine in Gelatin/Hyaluronic Acid Cryogel to Modulate Scaffold Mechanical Properties and to Maintain Chondrogenic Phenotype for Cartilage Tissue Engineering" International Journal of Molecular Sciences 17, no. 11: 1957. https://doi.org/10.3390/ijms17111957
APA StyleChen, C.-H., Kuo, C.-Y., Wang, Y.-J., & Chen, J.-P. (2016). Dual Function of Glucosamine in Gelatin/Hyaluronic Acid Cryogel to Modulate Scaffold Mechanical Properties and to Maintain Chondrogenic Phenotype for Cartilage Tissue Engineering. International Journal of Molecular Sciences, 17(11), 1957. https://doi.org/10.3390/ijms17111957







