Cytotoxicity of 11-epi-Sinulariolide Acetate Isolated from Cultured Soft Corals on HA22T Cells through the Endoplasmic Reticulum Stress Pathway and Mitochondrial Dysfunction
Abstract
:1. Introduction
2. Results
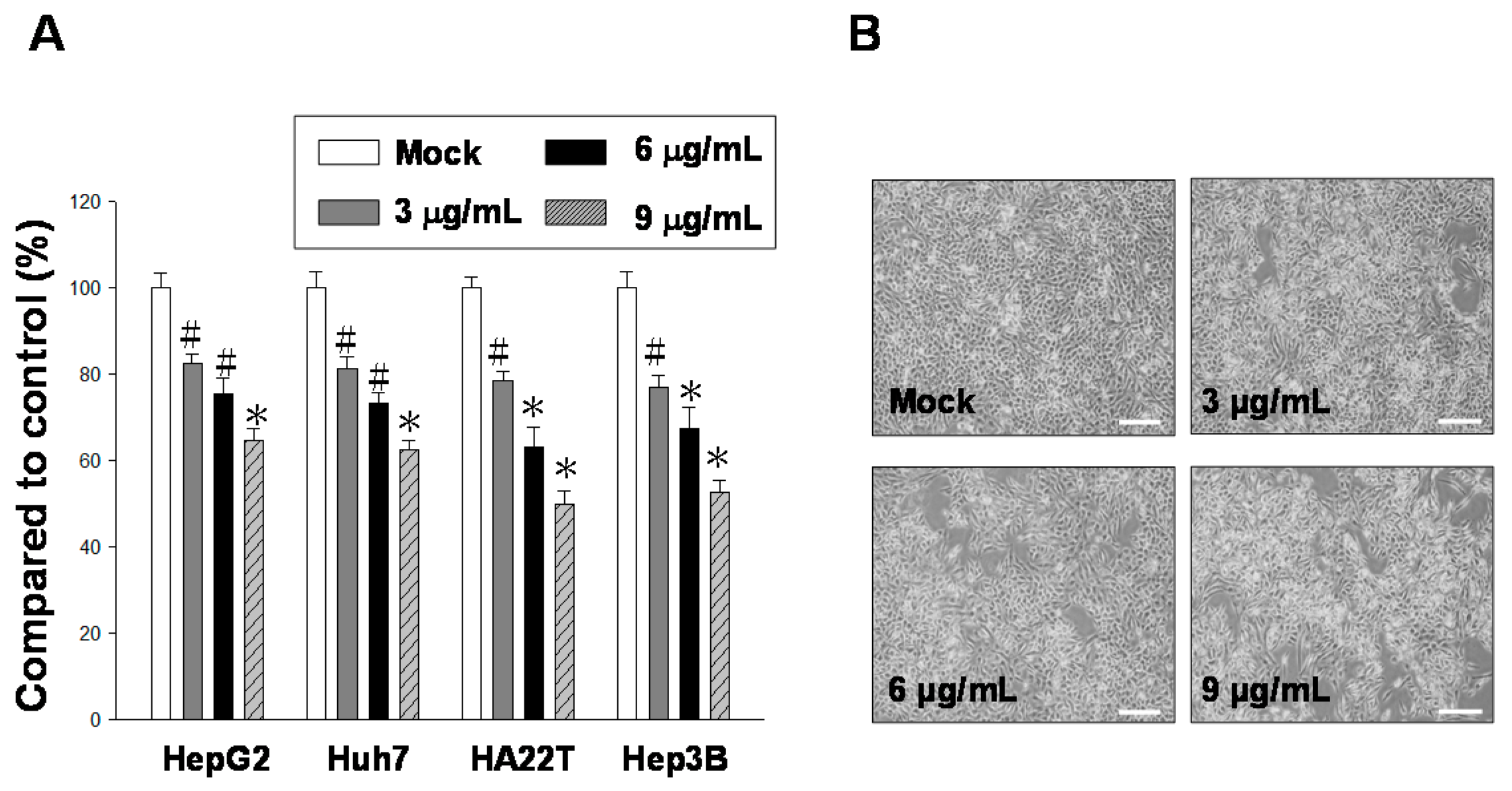
2.1. 11-epi-SA Is Cytotoxic toward Several HCC Cell Lines
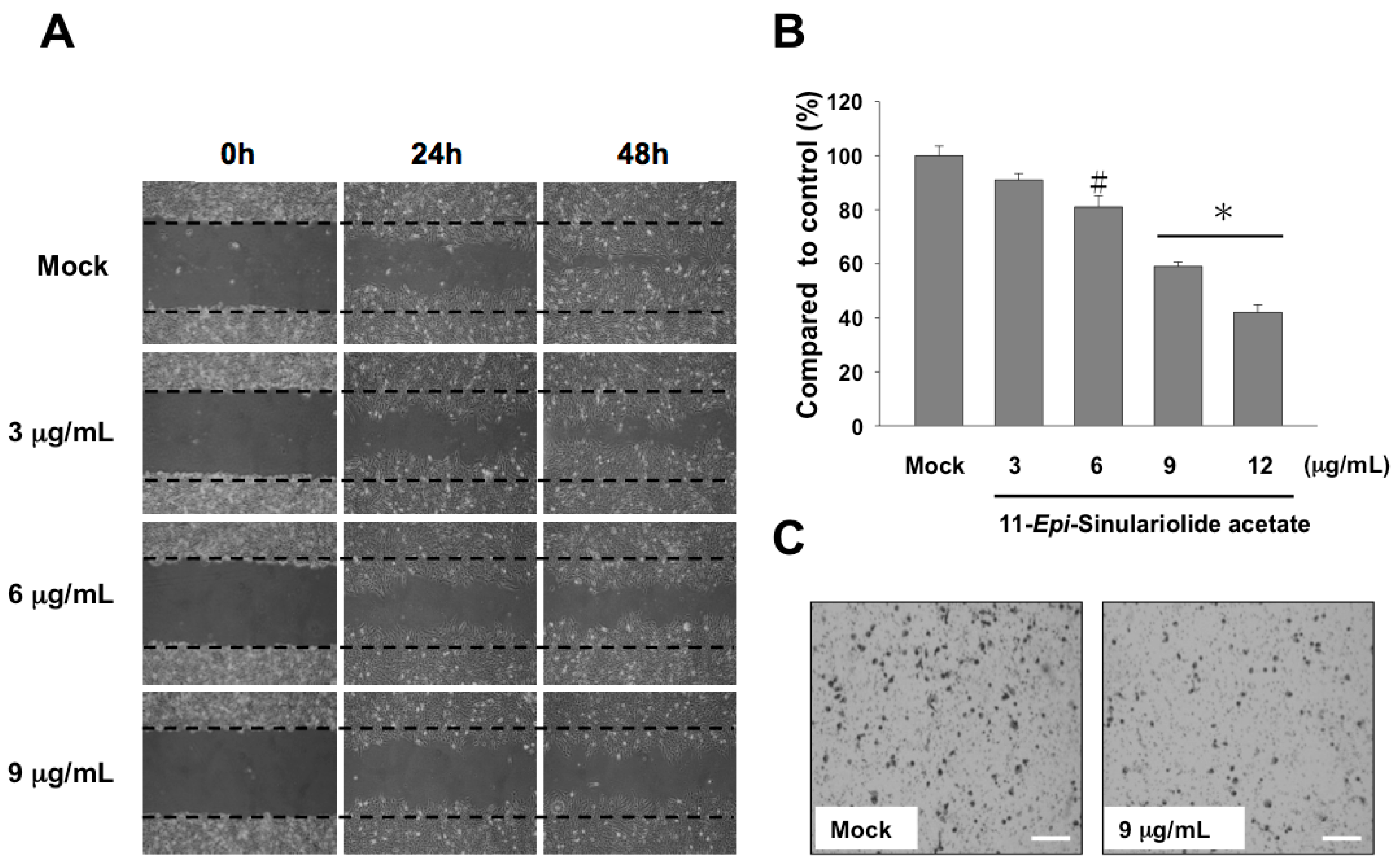
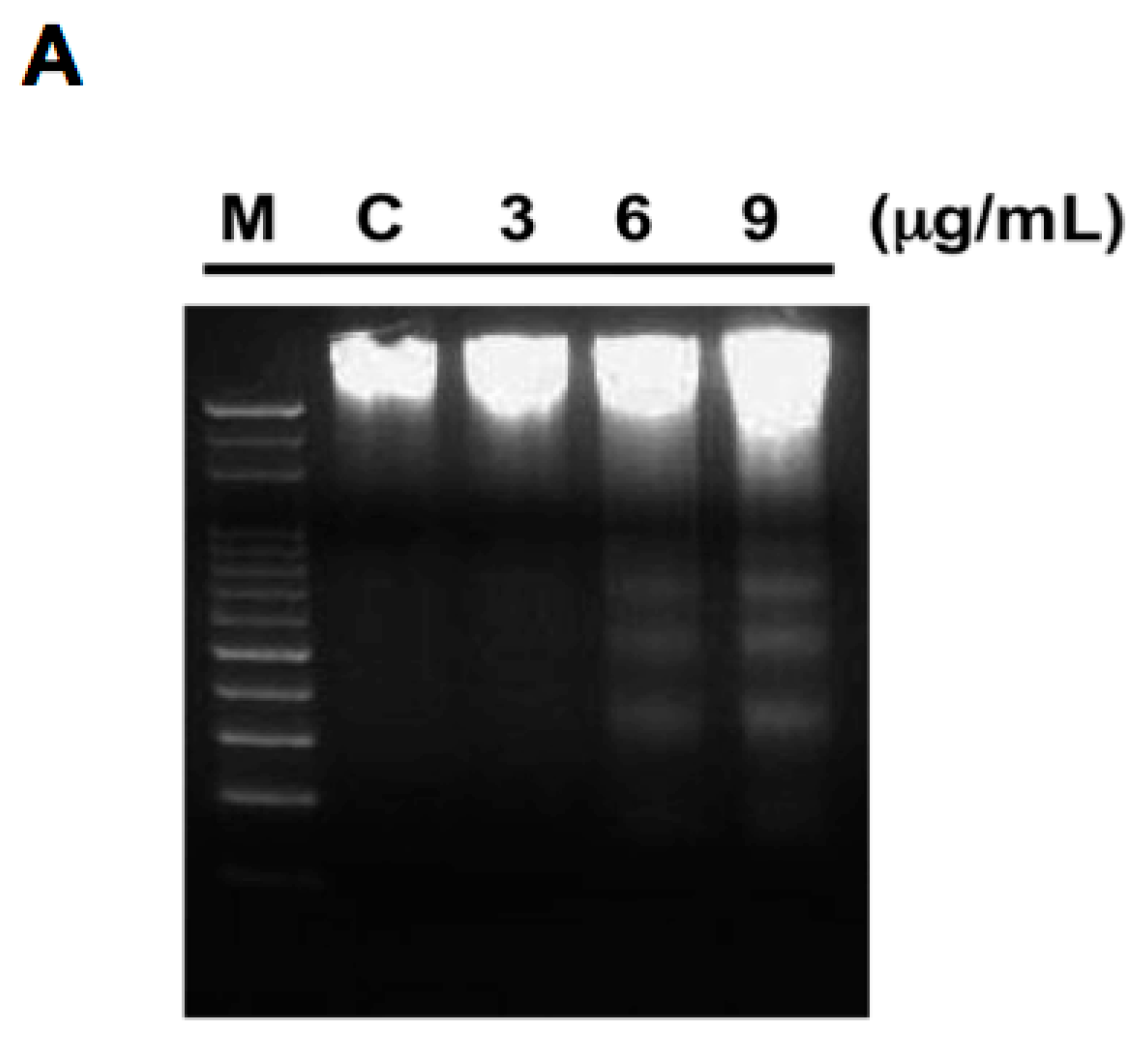
2.2. HA22T Cells Show Apoptotic Characteristics on 11-epi-SA Treatment
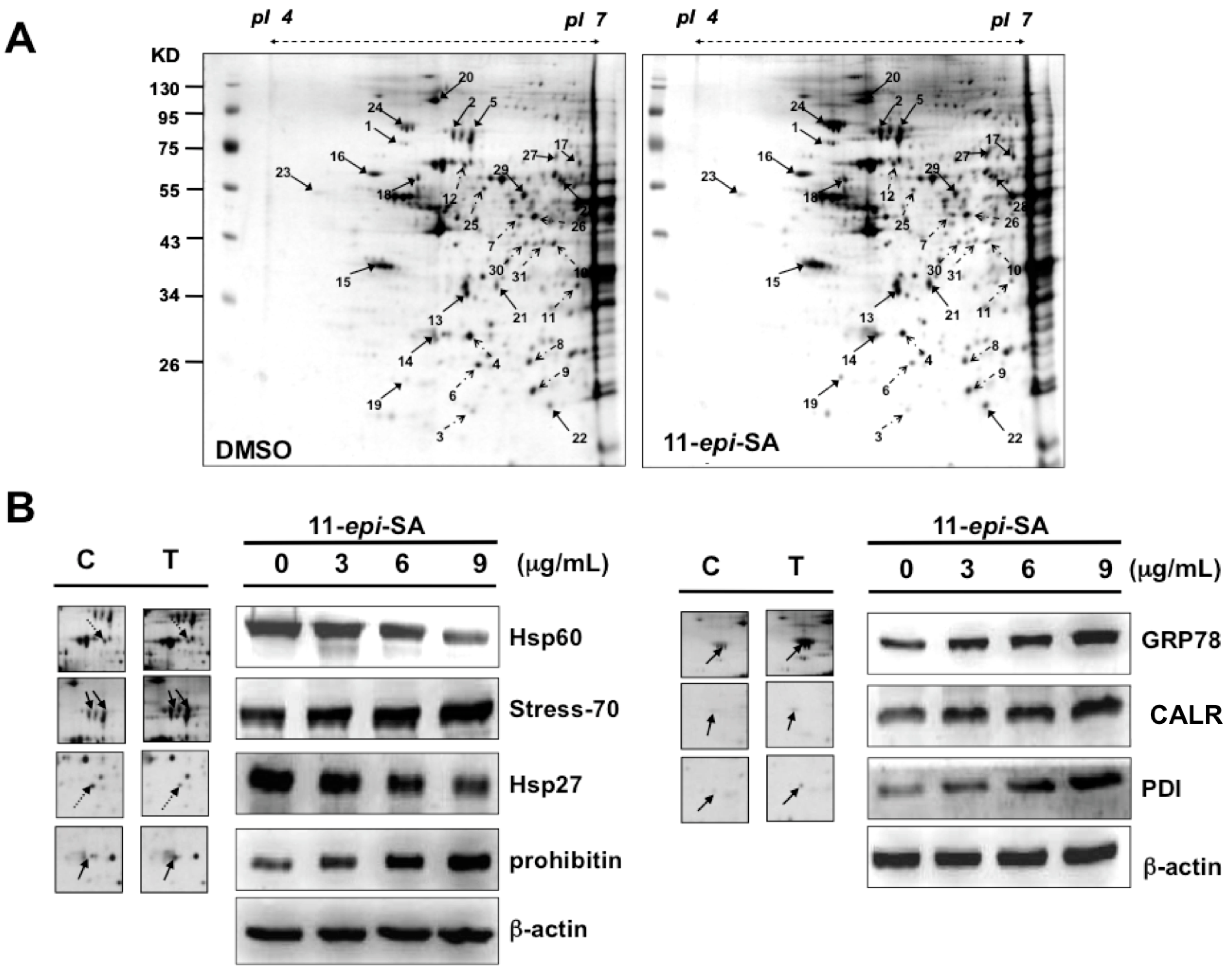
2.3. Global Proteomic Profiling of 11-epi-SA-Treated HA22T Cells
2.4. 11-epi-SA Causes Mitochondrial Dysfunction
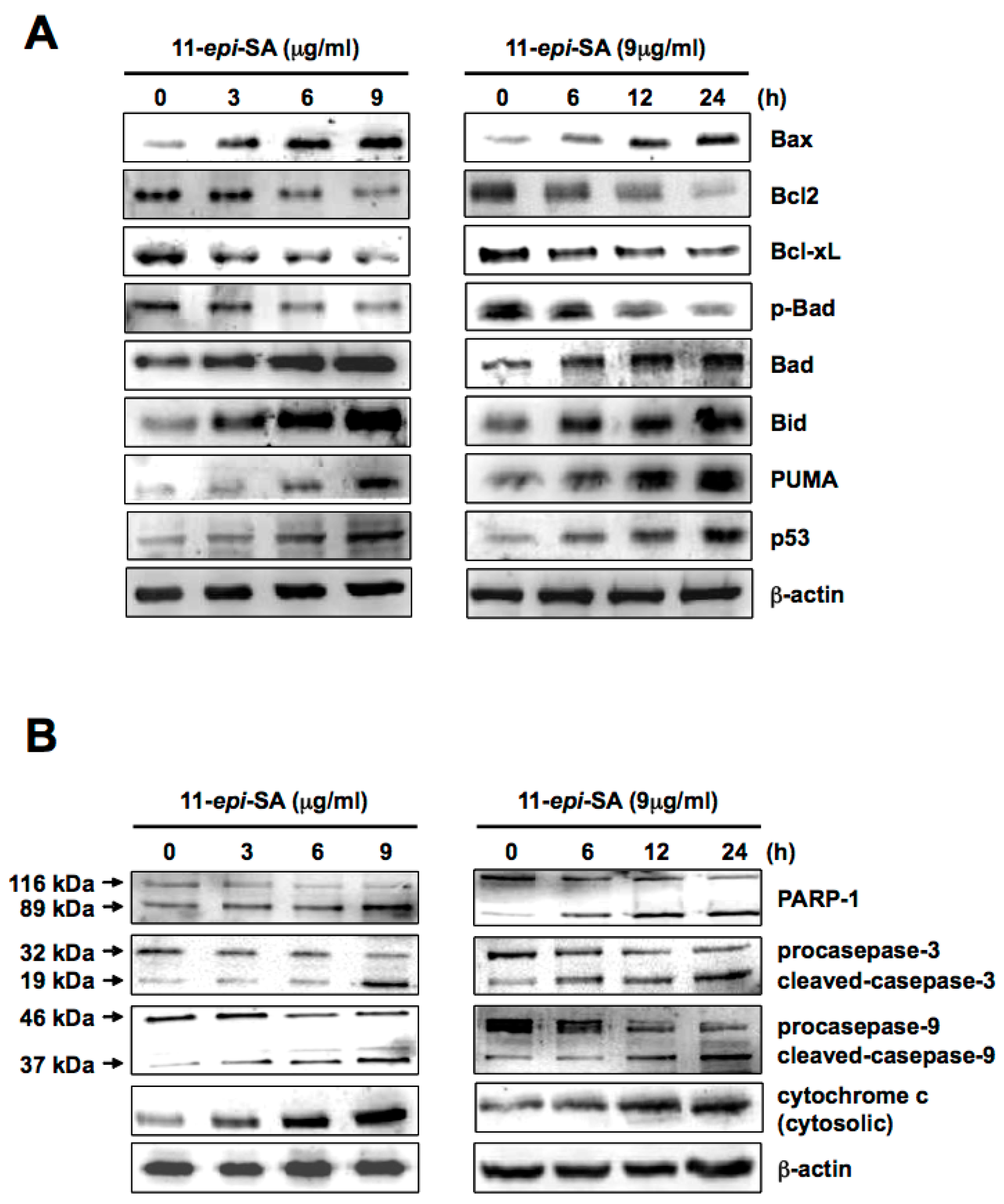
2.5. The Mitochondria-Related Pathway Is Activated by 11-epi-SA in a Dose and Time-Dependent Manner
2.6. The Caspase-Dependent Pathway Is Activated in 11-epi-SA-Treated HA22T Cells
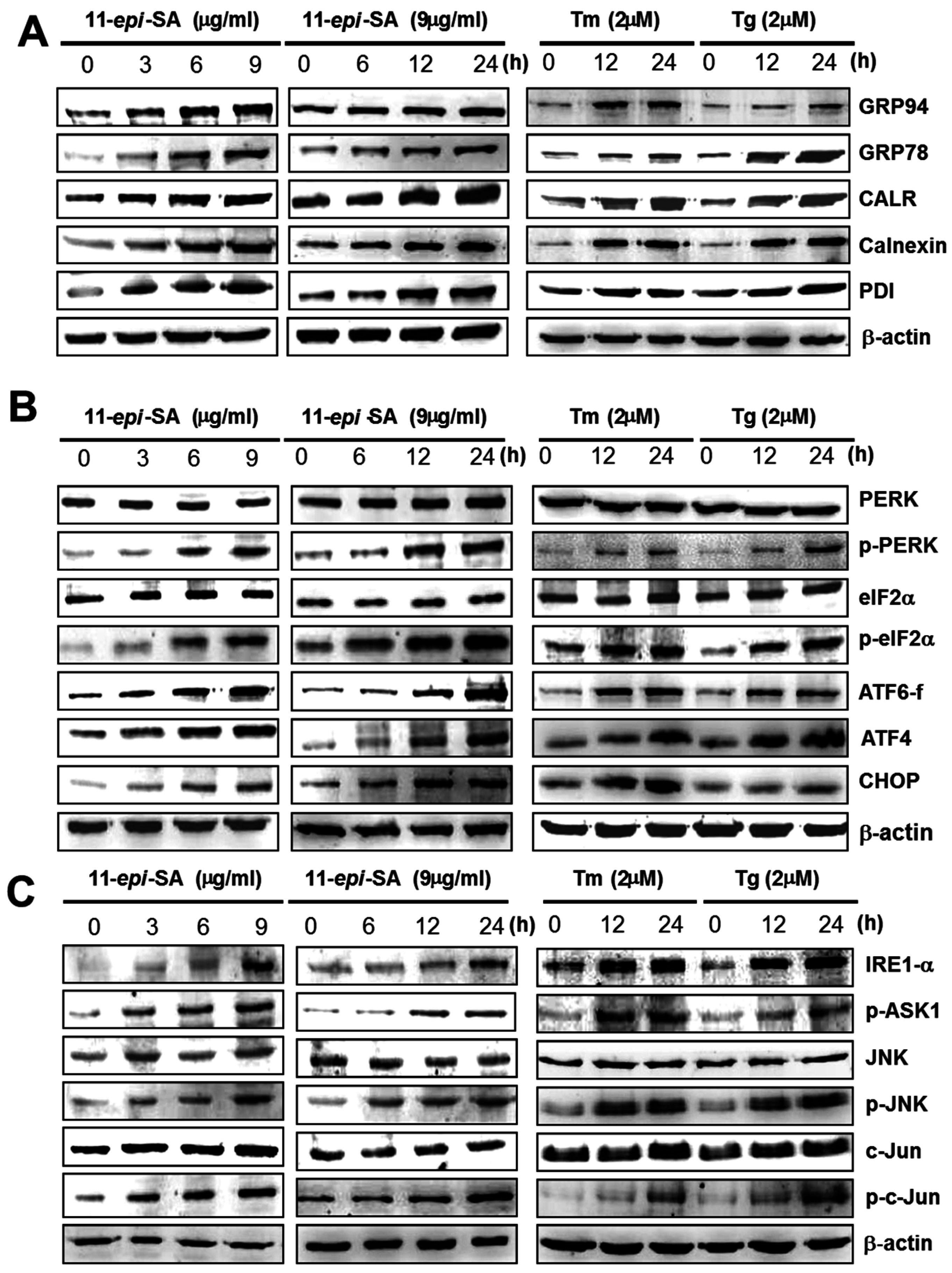
2.7. 11-epi-SA Induces the Activation of the ER Stress-Induced Pathway
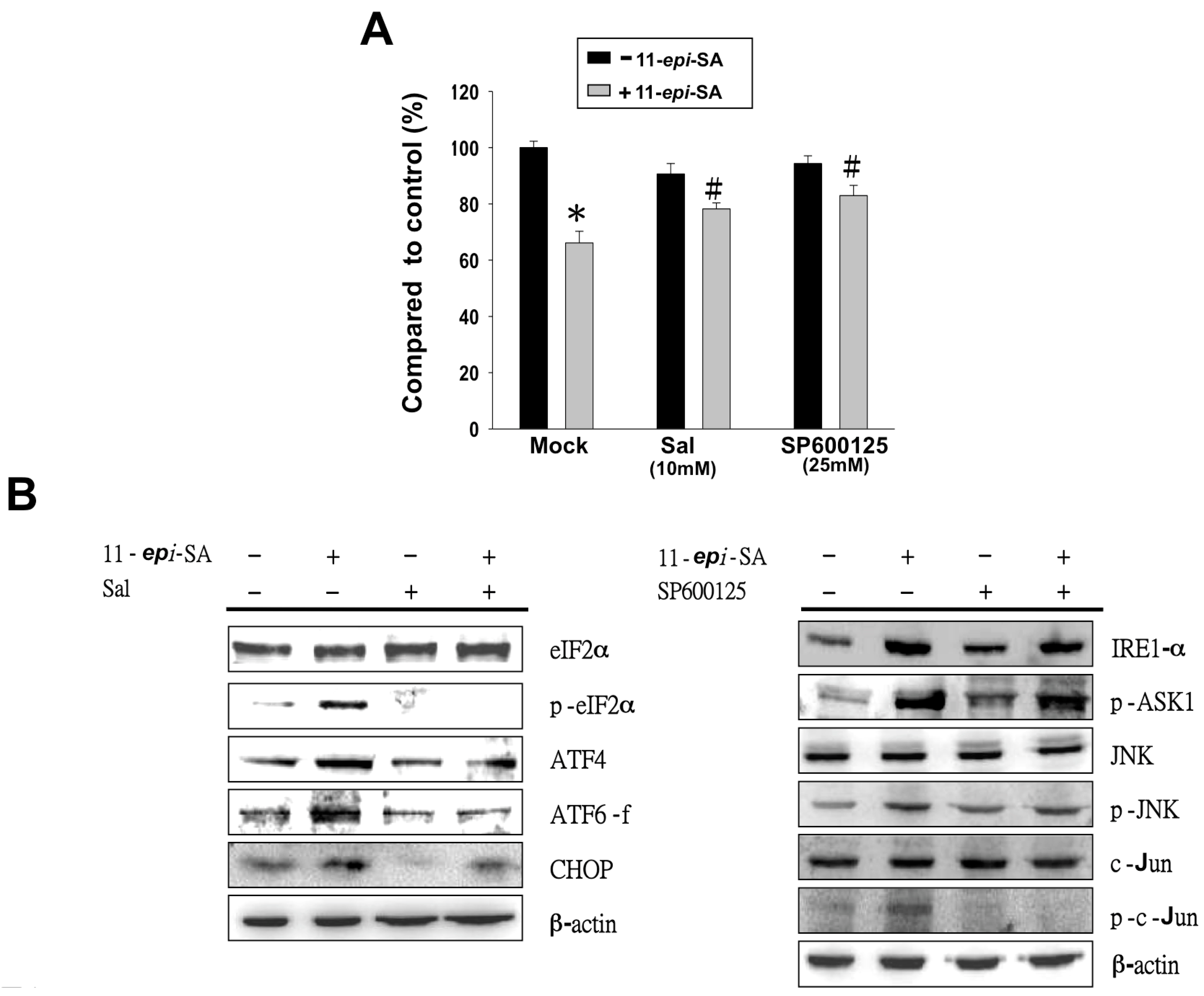
2.8. Inhibition of ER Stress-Related Pathways Rescues the 11-epi-SA-Induced Cytotoxicity of HA22T Cells
3. Discussion
3.1. 11-epi-SA Induces Apoptosis in HCC Cells
3.2. Mitochondria and ER Are Involved in 11-epi-SA-Induced Cytotoxicity
3.3. Mitochondrial Dysfunction Is a Crucial Event in 11-epi-SA-Related Cell Cytotoxicity
3.4. ER Stress Partially Initiates 11-epi-SA-Induced Cytotoxicity in HA22T Cells
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture and Drug Treatment
4.3. Cell Viability Assay
4.4. Wound Healing and Trans-Well Migration Assays
4.5. Flow Cytometric Assessment of Apoptosis
4.6. DNA Fragmentation Assay
4.7. Mitochondrial Transmembrane Potential Analysis
4.8. Protein Preparation and Measurement
4.9. Two-Dimensional Gel Electrophoresis and Protein Identification by Liquid Chromatography–Tandem Mass Spectrometry
4.10. Immunofluorescence Microscopy
4.11. Western Blotting
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Perz, J.F.; Armstrong, G.L.; Farrington, L.A.; Hutin, Y.J.; Bell, B.P. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J. Hepatol. 2006, 45, 529–538. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, A.; Llovet, J.M. Targeted therapies for hepatocellular carcinoma. Gastroenterology 2011, 140, 1410–1426. [Google Scholar] [CrossRef] [PubMed]
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Blum, H.E.; Spangenberg, H.C. Hepatocellular carcinoma: An update. Arch. Iran. Med. 2007, 10, 361–371. [Google Scholar] [PubMed]
- Masuda, T.; Beppu, T.; Ishiko, T.; Horino, K.; Baba, Y.; Mizumoto, T.; Hayashi, H.; Okabe, H.; Horlad, H.; Doi, K.; et al. Intrahepatic dissemination of hepatocellular carcinoma after local ablation therapy. J. Hepato-Biliary-Pancreat. 2008, 15, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Poon, D.; Anderson, B.O.; Chen, L.T.; Tanaka, K.; Lau, W.Y.; van Cutsem, E.; Singh, H.; Chow, W.C.; Ooi, L.L.; Chow, P.; et al. Management of hepatocellular carcinoma in Asia: Consensus statement from the Asian Oncology Summit 2009. Lancet Oncol. 2009, 10, 1111–1118. [Google Scholar] [CrossRef]
- Rampone, B.; Schiavone, B.; Martino, A.; Viviano, C.; Confuorto, G. Current management strategy of hepatocellular carcinoma. World J. Gastroenterol. 2009, 15, 3210–3216. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 2001, 18, 1–49. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 2002, 19, 1–48. [Google Scholar] [PubMed]
- Ojika, M.; Islam, M.K.; Shintani, T.; Zhang, Y.; Okamoto, T.; Sakagami, Y. Three new cytotoxic acylspermidines from the soft coral, Sinularia sp. Biosci. Biotechnol. Biochem. 2003, 67, 1410–1412. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.J.; Su, J.H.; Tsai, C.C.; Chen, Y.J.; Liao, M.H.; Wu, Y.J. 11-epi-Sinulariolide acetate reduces cell migration and invasion of human hepatocellular carcinoma by reducing the activation of ERK1/2, p38MAPK and FAK/PI3K/AKT/mTOR signaling pathways. Mar. Drugs 2014, 12, 4783–4798. [Google Scholar] [CrossRef] [PubMed]
- Poza, J.J.; Fernández, R.; Reyes, F.; Rodríguez, J.; Jiménez, C. Isolation, biological significance, synthesis, and cytotoxic evaluation of new natural parathiosteroids A–C and analogues from the soft coral Paragorgia sp. J. Org. Chem. 2008, 73, 7978–7984. [Google Scholar] [CrossRef] [PubMed]
- Chiang, P.C.; Chien, C.L.; Pan, S.L.; Chen, W.P.; Teng, C.M.; Shen, Y.C.; Guh, J.H. Induction of endoplasmic reticulum stress and apoptosis by a marine prostanoid in human hepatocellular carcinoma. J. Hepatol. 2005, 43, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Kamel, H.N.; Ferreira, D.; Garcia-Fernandez, L.F.; Slattery, M. Cytotoxic diterpenoids from the hybrid soft coral Sinularia maxima x Sinularia polydactyla. J. Natl. Prod. 2007, 70, 1223–1227. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.M.; Khanfar, M.A.; Elnagar, A.Y.; Mohammed, R.; Shaala, L.A.; Youssef, D.T.; Hifnawy, M.S.; El Sayed, K.A. Pachycladins A–E, prostate cancer invasion and migration inhibitory eunicellin-based diterpenoids from the red sea soft coral Cladiella pachyclados. J. Natl. Prod. 2010, 73, 848–853. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.I.; Chen, C.C.; Chen, J.C.; Su, J.H.; Huang, H.H.; Chen, J.Y.; Wu, Y.J. Proteomic analysis of anti-tumor effects of 11-dehydrosinulariolide on CAL-27 cells. Mar. Drugs 2011, 9, 1254–1272. [Google Scholar] [CrossRef] [PubMed]
- Neoh, C.A.; Wang, R.Y.; Din, Z.H.; Su, J.H.; Chen, Y.K.; Tsai, F.J.; Weng, S.H.; Wu, Y.J. Induction of apoptosis by sinulariolide from soft coral through mitochondrial-related and p38MAPK pathways on human bladder carcinoma cells. Mar. Drugs 2012, 10, 2893–2911. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.S.; Michael, A.; Butler-Manuel, S.A. Advances in the treatment of ovarian cancer—A potential role of antiinflammatory phytochemicals. Discov. Med. 2012, 13, 7–17. [Google Scholar] [PubMed]
- Slee, E.A.; Martin, S.J. Regulation of caspase activation in apoptosis: Implications for transformation and drug resistance. Cytotechnology 1998, 27, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Hensley, P.; Mishra, M.; Kyprianou, N. Targeting caspases in cancer therapeutics. Biol. Chem. 2013, 394, 831–843. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Albrecht, A.M.; Ni, X.; Yang, J.; Li, M. Glioblastoma tumor initiating cells: Therapeutic strategies targeting apoptosis and microRNA pathways. Curr. Mol. Med. 2013, 13, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Fiandalo, M.V.; Kyprianou, N. Caspase control: Protagonists of cancer cell apoptosis. Exp. Oncol. 2012, 34, 165–175. [Google Scholar] [PubMed]
- Shimizu, S.; Tsujimoto, Y. Proapoptotic BH3-only Bcl-2 family members induce cytochrome c release, but not mitochondrial membrane potential loss, and do not directly modulate voltage-dependent anion channel activity. Proc. Natl. Acad. Sci. USA 2000, 97, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Wajant, H. The Fas signaling pathway: More than a paradigm. Science 2002, 296, 1635–1636. [Google Scholar] [CrossRef] [PubMed]
- Denicourt, C.; Dowdy, S.F. Medicine. Targeting apoptotic pathways in cancer cells. Science 2004, 305, 1411–1413. [Google Scholar] [CrossRef] [PubMed]
- Matthews, G.M.; Newbold, A.; Johnstone, R.W. Intrinsic and extrinsic apoptotic pathway signaling as determinants of histone deacetylase inhibitor antitumor activity. Adv. Cancer Res. 2012, 116, 165–197. [Google Scholar] [PubMed]
- Ron, D.; Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 2007, 8, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Zielinski, R.R.; Eigl, B.J.; Chi, K.N. Targeting the apoptosis pathway in prostate cancer. Cancer J. 2013, 19, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Green, D.R.; Reed, J.C. Mitochondria and apoptosis. Science 1998, 281, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Ballarin, L.; Burighel, P.; Cima, F. A tale of death and life: Natural apoptosis in the colonial ascidian Botryllus schlosseri (Urochordata, Ascidiacea). Curr. Pharm. Des. 2008, 14, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Basanez, G.; Soane, L.; Hardwick, J.M. A new view of the lethal apoptotic pore. PLoS Biol. 2012, 10, e1001399. [Google Scholar] [CrossRef] [PubMed]
- González-Gironès, D.M.; Moncunill-Massaguer, C.; Iglesias-Serret, D.; Cosialls, A.M.; Pérez-Perarnau, A.; Palmeri, C.M.; Rubio-Patiño, C.; Villunger, A.; Pons, G.; Gil, J. AICAR induces Bax/Bak-dependent apoptosis through upregulation of the BH3-only proteins BIM and NOXA in mouse embryonic fibroblasts. Apoptosis 2013, 18, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Kaparou, M.; Choumerianou, D.; Perdikogianni, C.; Martimianaki, G.; Kalmanti, M.; Stiakaki, E. Enhanced levels of the apoptotic Bax/Bcl-2 ratio in children with acute lymphoblastic leukemia and high-risk features. Genet. Mol. Biol. 2013, 36, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Hoshyar, R.; Bathaie, S.Z.; Sadeghizadeh, M. Crocin triggers the apoptosis through increasing the Bax/Bcl-2 ratio and caspase activation in human gastric adenocarcinoma (AGS) cells. DNA Cell Biol. 2013, 32, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Gotoh, M.; Sano-Maeda, K.; Murofushi, H.; Murakami-Murofushi, K. Protection of neuroblastoma Neuro2a cells from hypoxia-induced apoptosis by cyclic phosphatidic acid (cPA). PLoS ONE 2012, 7, e51093. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, D.W.; Thornberry, N.A. Apoptosis. Life and death decisions. Science 2003, 299, 214–215. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.V.; Ellerby, H.M.; Bredesen, D.E. Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ. 2004, 11, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Boyce, M.; Yuan, J. Cellular response to endoplasmic reticulum stress: A matter of life or death. Cell Death Differ. 2006, 13, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Moenner, M.; Pluquet, O.; Bouchecareilh, M.; Chevet, E. Integrated endoplasmic reticulum stress responses in cancer. Cancer Res. 2007, 67, 10631–10634. [Google Scholar] [CrossRef] [PubMed]
- Su, T.R.; Lin, J.J.; Chiu, C.C.; Chen, J.Y.; Su, J.H.; Cheng, Z.J.; Hwang, W.I.; Huang, H.H.; Wu, Y.J. Proteomic investigation of anti-tumor activities exerted by sinularin against A2058 melanoma cells. Electrophoresis 2012, 33, 1139–1152. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.Y.; Chiu, C.C.; Haung, R.W.; Yeh, C.C.; Huang, K.J.; Chang, K.F.; Hseu, Y.C.; Chang, F.R.; Chang, H.W.; Wu, Y.C. Antiproliferative effects of goniothalamin on Ca9-22 oral cancer cells through apoptosis, DNA damage and ROS induction. Mutat. Res. 2012, 747, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.I.; Wang, R.Y.; Lin, J.J.; Su, J.H.; Chiu, C.C.; Chen, J.C.; Chen, J.Y.; Wu, Y.J. Proteomic profiling of the 11-dehydrosinulariolide-treated oral carcinoma cells Ca9-22: Effects on the cell apoptosis through mitochondrial-related and ER stress pathway. J. Proteom. 2012, 75, 5578–5589. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Che, C.M.; Chiu, J.F.; He, Q.Y. Dioscin (saponin)-induced generation of reactive oxygen species through mitochondria dysfunction: A proteomic-based study. J. Proteome Res. 2007, 6, 4703–4710. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.B. Apoptosis in the pathogenesis and treatment of disease. Science 1995, 267, 1456–1462. [Google Scholar] [CrossRef] [PubMed]
- Zi, X.; Simoneau, A.R. Flavokawain A, a novel chalcone from Kava extract, induces apoptosis in bladder cancer cells by involvement of Bax protein-dependent and mitochondria-dependent apoptotic pathway and suppresses tumor growth in mice. Cancer Res. 2005, 65, 3479–3486. [Google Scholar] [PubMed]
- Imberti, R.; Nieminen, A.L.; Herman, B.; Lemasters, J.J. Mitochondrial and glycolytic dysfunction in lethal injury to hepatocytes by t-butylhydroperoxide: Protection by fructose, cyclosporin A and trifluoperazine. J. Pharmacol. Exp. Ther. 1993, 265, 392–400. [Google Scholar] [PubMed]
- Seya, T.; Shime, H.; Takaki, H.; Azuma, M.; Oshiumi, H.; Matsumoto, M. TLR3/TICAM-1 signaling in tumor cell RIP3-dependent necroptosis. Oncoimmunology 2012, 1, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Green, D.R.; Oberst, A.; Dillon, C.P.; Weinlich, R.; Salvesen, G.S. RIPK-dependent necrosis and its regulation by caspases: A mystery in five acts. Mol. Cell 2011, 44, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Putchala, M.C.; Ramani, P.; Sherlin, H.J.; Premkumar, P.; Natesan, A. Ascorbic acid and its pro-oxidant activity as a therapy for tumours of oral cavity—A systematic review. Arch. Oral Biol. 2013, 58, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Leanza, L.; Henry, B.; Sassi, N.; Zoratti, M.; Chandy, K.G.; Gulbins, E.; Szabo, I. Inhibitors of mitochondrial Kv1.3 channels induce Bax/Bak-independent death of cancer cells. EMBO Mol. Med. 2012, 4, 577–593. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhang, S.; Ong, C.N.; Shen, H.M. Critical role of pro-apoptotic Bcl-2 family members in andrographolide-induced apoptosis in human cancer cells. Biochem. Pharmacol. 2006, 72, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.C.; Cullen, S.P.; Martin, S.J. Apoptosis: Controlled demolition at the cellular level. Nat. Rev. Mol. Cell Biol. 2008, 9, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, Q.Y.; Sun, R.W.; Che, C.M.; Chiu, J.F. GoldIII porphyrin 1a induced apoptosis by mitochondrial death pathways related to reactive oxygen species. Cancer Res. 2005, 65, 11553–11564. [Google Scholar] [CrossRef] [PubMed]
- Chipuk, J.E.; Kuwana, T.; Bouchier-Hayes, L.; Droin, N.M.; Newmeyer, D.D.; Schuler, M.; Green, D.R. Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 2004, 303, 1010–1014. [Google Scholar] [CrossRef] [PubMed]
- Reubold, T.F.; Eschenburg, S. A molecular view on signal transduction by the apoptosome. Cell Signal. 2012, 24, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Wurstle, M.L.; Laussmann, M.A.; Rehm, M. The central role of initiator caspase-9 in apoptosis signal transduction and the regulation of its activation and activity on the apoptosome. Exp. Cell Res. 2012, 318, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Bratton, S.B.; Salvesen, G.S. Regulation of the Apaf-1-caspase-9 apoptosome. J. Cell Sci. 2010, 123, 3209–3214. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.A.; Schnellmann, R.G. Calpains, mitochondria, and apoptosis. Cardiovasc. Res. 2012, 96, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Snigdha, S.; Smith, E.D.; Prieto, G.A.; Cotman, C.W. Caspase-3 activation as a bifurcation point between plasticity and cell death. Neurosci. Bull. 2012, 28, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Chaitanya, G.V.; Steven, A.J.; Babu, P.P. PARP-1 cleavage fragments: Signatures of cell-death proteases in neurodegeneration. Cell Commun. Signal. CCS 2010, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.Y.; Bailly-Maitre, B.; Reed, J.C. Endoplasmic reticulum stress: Cell life and death decisions. J. Clin. Investig. 2005, 115, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Xu, W.J.; Reed, J.C. Cell death and endoplasmic reticulum stress: Disease relevance and therapeutic opportunities. Nat. Rev. Drug Discov. 2008, 7, 1013–1030. [Google Scholar] [CrossRef] [PubMed]
- Kouroku, Y.; Fujita, E.; Tanida, I.; Ueno, T.; Isoai, A.; Kumagai, H.; Ogawa, S.; Kaufman, R.J.; Kominami, E.; Momoi, T. ER stress (PERK/eIF2alpha phosphorylation) mediates the polyglutamine-induced LC3 conversion, an essential step for autophagy formation. Cell Death Differ. 2007, 14, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Oyadomari, S.; Mori, M. Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ. 2004, 11, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Hendershot, L.M. Herp is dually regulated by both the endoplasmic reticulum stress-specific branch of the unfolded protein response and a branch that is shared with other cellular stress pathways. J. Biol. Chem. 2004, 279, 13792–13799. [Google Scholar] [CrossRef] [PubMed]
- Araki, K.; Nagata, K. Protein folding and quality control in the ER. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Frank, C.L.; Korth, M.J.; Sopher, B.L.; Novoa, I.; Ron, D.; Katze, M.G. Control of perk eIF2α kinase activity by the endoplasmic reticulum stress-induced molecular chaperone p58IPK. Proc. Natl. Acad. Sci. USA 2002, 99, 15920–15925. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Ochiai, K.; Kondo, S.; Tsumagari, K.; Murakami, T.; Cavener, D.R.; Imaizumi, K. Endoplasmic reticulum stress response mediated by the PERK-eIF2(alpha)-ATF4 pathway is involved in osteoblast differentiation induced by BMP2. J. Biol. Chem. 2011, 286, 4809–4818. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Baumeister, P.; Yang, S.; Abcouwer, S.F.; Lee, A.S. Induction of Grp78/BiP by translational block: Activation of the Grp78 promoter by ATF4 through and upstream ATF/CRE site independent of the endoplasmic reticulum stress elements. J. Biol. Chem. 2003, 278, 37375–37385. [Google Scholar] [CrossRef] [PubMed]
- Lenna, S.; Chrobak, I.; Farina, G.A.; Rodriguez-Pascual, F.; Lamas, S.; Lafyatis, R.; Scorza, R.; Trojanowska, M. HLA-B35 and dsRNA induce endothelin-1 via activation of ATF4 in human microvascular endothelial cells. PLoS ONE 2013, 8, e56123. [Google Scholar] [CrossRef] [PubMed]
- Haze, K.; Yoshida, H.; Yanagi, H.; Yura, T.; Mori, K. Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol. Biol. Cell 1999, 10, 3787–3799. [Google Scholar] [CrossRef] [PubMed]
- Puthalakath, H.; O’Reilly, L.A.; Gunn, P.; Lee, L.; Kelly, P.N.; Huntington, N.D.; Hughes, P.D.; Michalak, E.M.; McKimm-Breschkin, J.; Motoyama, N.; et al. ER stress triggers apoptosis by activating BH3-only protein Bim. Cell 2007, 129, 1337–1349. [Google Scholar] [CrossRef] [PubMed]
- Wagner, E.F.; Nebreda, A.R. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat. Rev. Cancer 2009, 9, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Ichijo, H.; Nishida, E.; Irie, K.; ten Dijke, P.; Saitoh, M.; Moriguchi, T.; Takagi, M.; Matsumoto, K.; Miyazono, K.; Gotoh, Y. Induction of apoptosis by ASK1, a mammalian MAPKKK that activates SAPK/JNK and p38 signaling pathways. Science 1997, 275, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Huigsloot, M.; Tijdens, I.B.; Mulder, G.J.; van de Water, B. Differential regulation of doxorubicin-induced mitochondrial dysfunction and apoptosis by Bcl-2 in mammary adenocarcinoma (MTLn3) cells. J. Biol. Chem. 2002, 277, 35869–35879. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Srivastava, R.K. Bax and Bak genes are essential for maximum apoptotic response by curcumin, a polyphenolic compound and cancer chemopreventive agent derived from turmeric, Curcuma longa. Carcinogenesis 2007, 28, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Fusaro, G.; Padmanabhan, J.; Chellappan, S.P. Prohibitin co-localizes with Rb in the nucleus and recruits N-CoR and HDAC1 for transcriptional repression. Oncogene 2002, 21, 8388–8396. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, B.; Faller, D.V. Prohibitin requires Brg-1 and Brm for the repression of E2F and cell growth. EMBO J. 2002, 21, 3019–3028. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, B.; Faller, D.V. Brg1/Brm and prohibitin are required for growth suppression by estrogen antagonists. EMBO J. 2004, 23, 2293–2303. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Chambers, K.J.; Faller, D.V.; Wang, S. Reprogramming of the SWI/SNF complex for co-activation or co-repression in prohibitin-mediated estrogen receptor regulation. Oncogene 2007, 26, 7153–7157. [Google Scholar] [CrossRef] [PubMed]
- Fusaro, G.; Wang, S.; Chellappan, S. Differential regulation of Rb family proteins and prohibitin during camptothecin-induced apoptosis. Oncogene 2002, 21, 4539–4548. [Google Scholar] [CrossRef] [PubMed]
- Fusaro, G.; Dasgupta, P.; Rastogi, S.; Joshi, B.; Chellappan, S. Prohibitin induces the transcriptional activity of p53 and is exported from the nucleus upon apoptotic signaling. J. Biol. Chem. 2003, 278, 47853–47861. [Google Scholar] [CrossRef] [PubMed]
- Joshi, B.; Rastogi, S.; Morris, M.; Carastro, L.M.; DeCook, C.; Seto, E.; Chellappan, S.P. Differential regulation of human YY1 and caspase 7 promoters by prohibitin through E2F1 and p53 binding sites. Biochem. J. 2007, 401, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes, A.J.; Frases, S.; Gomez, F.J.; Zancope-Oliveira, R.M.; Nosanchuk, J.D. Monoclonal antibodies to heat shock protein 60 alter the pathogenesis of Histoplasma capsulatum. Infect. Immun. 2009, 77, 1357–1367. [Google Scholar] [CrossRef] [PubMed]
- Kirchhoff, S.R.; Gupta, S.; Knowlton, A.A. Cytosolic heat shock protein 60, apoptosis, and myocardial injury. Circulation 2002, 105, 2899–2904. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Knowlton, A.A. HSP60, Bax, apoptosis and the heart. J. Cell. Mol. Med. 2005, 9, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, J.C.; Dohi, T.; Kang, B.H.; Altieri, D.C. Hsp60 regulation of tumor cell apoptosis. J. Biol. Chem. 2008, 283, 5188–5194. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, D.T.; Kaufman, R.J. A trip to the ER: Coping with stress. Trends Cell Biol. 2004, 14, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Kuwana, T.; Newmeyer, D.D. Bcl-2-family proteins and the role of mitochondria in apoptosis. Curr. Opin. Cell Biol. 2003, 15, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.; Zha, J.; Jockel, J.; Boise, L.H.; Thompson, C.B.; Korsmeyer, S.J. Bad, a heterodimeric partner for Bcl-xL and Bcl-2, displaces Bax and promotes cell death. Cell 1995, 80, 285–291. [Google Scholar] [CrossRef]
- Zha, J.; Harada, H.; Yang, E.; Jockel, J.; Korsmeyer, S.J. Serine phosphorylation of death agonist BAD in response to survival factor results in binding to 14-3-3 not BCL-XL. Cell 1996, 87, 619–628. [Google Scholar] [CrossRef]
- Igney, F.H.; Krammer, P.H. Death and anti-death: Tumour resistance to apoptosis. Nat. Rev. Cancer 2002, 2, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.M.; Wu, Y.J.; Chen, C.C.; Hsu, J.L.; Chen, J.C.; Chen, J.Y.; Huang, C.H.; Ko, Y.C. Identification of low-abundance proteins via fractionation of the urine proteome with weak anion exchange chromatography. Proteome Sci. 2011, 9, 17. [Google Scholar] [CrossRef] [PubMed]




| Spot No. | Protein Name | Accession No. | Calculated MW/pI | Peptide Matched | Sequence Covered % | MASCOT Score | Regulation (Fold Change) |
|---|---|---|---|---|---|---|---|
| 1 | Lamin-A/C | P02545 | 74.09/6.57 | 20 | 22 | 126 | +3.3 |
| 2 | Stress-70 | P38646 | 73.65/5.87 | 54 | 46 | 570 | +2.5 |
| 3 | Peroxiredoxin-2 | P32119 | 21.87/5.66 | 31 | 43 | 316 | −2.3 |
| 4 | Isopentenyl-diphosphate delta-isomerase 2 | Q9BXS1 | 26.7/6.01 | 1 | 5 | 75 | −2.5 |
| 5 | Stress-70 | P38646 | 73.63/5.87 | 100 | 63 | 1233 | +3.1 |
| 6 | Heat-shock protein 27 | P04792 | 22.76/5.98 | 52 | 71 | 753 | −3.9 |
| 7 | Cytoplasmic protein NCK1 | P16333 | 42.83/6.06 | 6 | 9 | 78 | −2.8 |
| 8 | Heat-shock protein 27 | P04792 | 22.76/5.98 | 90 | 72 | 945 | −2.4 |
| 9 | Thioredoxin-dependent peroxide reductase | P30048 | 27.67/7.67 | 20 | 35 | 209 | −3.0 |
| 10 | Heterogeneous nuclear ribonucleoprotein A/B | Q99729 | 36.59/9.04 | 3 | 9 | 72 | −3.2 |
| 11 | Heterogeneous nuclear ribonucleoprotein H3 | P31942 | 36.91/6.37 | 14 | 30 | 173 | −2.9 |
| 12 | 60 kDa heat-shock protein | P10809 | 61.01/5.7 | 13 | 11 | 70 | −1.8 |
| 13 | Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta 2 | P62879 | 37.32/5.6 | 13 | 22 | 52 | +3.1 |
| 14 | Prohibitin | P35232 | 29.78/5.57 | 385 | 88 | 10,621 | +2.2 |
| 15 | Nucleophosmin | P06748 | 32.55/4.64 | 6 | 22 | 107 | +2.1 |
| 16 | Protein disulfide-isomerase | P07237 | 57.08/4.76 | 51 | 52 | 314 | +2.7 |
| 17 | T-complex protein 1 subunit zeta | P40227 | 57.98/6.23 | 27 | 37 | 278 | +1.8 |
| 18 | Vimentin | P08670 | 53.61/5.06 | 52 | 52 | 507 | +2.1 |
| 19 | Rho GDP-dissociation inhibitor 1 | P52565 | 23.19/5.02 | 6 | 15 | 67 | +2.6 |
| 20 | Transitional endoplasmic reticulum ATPase | P55072 | 89.22/5.14 | 55 | 47 | 594 | +4.4 |
| 21 | l-lactate dehydrogenase B chain | P07195 | 36.61/5.71 | 16 | 32 | 147 | +3.1 |
| 22 | Protein DJ-1 | Q99497 | 19.87/6.33 | 58 | 62 | 368 | +2.0 |
| 23 | Calreticulin | P27797 | 48.11/4.29 | 28 | 43 | 193 | +1.9 |
| 24 | 78 kDa glucose-regulated protein precursor | P11021 | 72.28/5.07 | 61 | 56 | 900 | +5.2 |
| 25 | Tubulin alpha-ubiquitous chain | P68363 | 50.12/4.94 | 23 | 35 | 251 | −1.8 |
| 26 | Ornithine aminotransferase | P04181 | 48.85/6.57 | 21 | 56 | 1065 | −1.9 |
| 27 | T-complex protein 1 subunit beta | P78371 | 57.45/6.01 | 33 | 40 | 386 | +2.1 |
| 28 | Succinyl-CoA:3-ketoacid-coenzyme A transferase 1 | P55809 | 56.12/7.14 | 14 | 12 | 234 | −2.4 |
| 29 | Heterogeneous nuclear ribonucleoprotein H | P31943 | 49.19/5.89 | 45 | 32 | 626 | +1.7 |
| 30 | Macrophage capping protein | P40121 | 38.49/5.88 | 17 | 29 | 283 | −2.3 |
| 31 | Macrophage capping protein | P40121 | 38.49/5.88 | 3 | 6 | 72 | −1.9 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, J.-J.; Wang, R.Y.L.; Chen, J.-C.; Chiu, C.-C.; Liao, M.-H.; Wu, Y.-J. Cytotoxicity of 11-epi-Sinulariolide Acetate Isolated from Cultured Soft Corals on HA22T Cells through the Endoplasmic Reticulum Stress Pathway and Mitochondrial Dysfunction. Int. J. Mol. Sci. 2016, 17, 1787. https://doi.org/10.3390/ijms17111787
Lin J-J, Wang RYL, Chen J-C, Chiu C-C, Liao M-H, Wu Y-J. Cytotoxicity of 11-epi-Sinulariolide Acetate Isolated from Cultured Soft Corals on HA22T Cells through the Endoplasmic Reticulum Stress Pathway and Mitochondrial Dysfunction. International Journal of Molecular Sciences. 2016; 17(11):1787. https://doi.org/10.3390/ijms17111787
Chicago/Turabian StyleLin, Jen-Jie, Robert Y. L. Wang, Jiing-Chuan Chen, Chien-Chih Chiu, Ming-Hui Liao, and Yu-Jen Wu. 2016. "Cytotoxicity of 11-epi-Sinulariolide Acetate Isolated from Cultured Soft Corals on HA22T Cells through the Endoplasmic Reticulum Stress Pathway and Mitochondrial Dysfunction" International Journal of Molecular Sciences 17, no. 11: 1787. https://doi.org/10.3390/ijms17111787
APA StyleLin, J.-J., Wang, R. Y. L., Chen, J.-C., Chiu, C.-C., Liao, M.-H., & Wu, Y.-J. (2016). Cytotoxicity of 11-epi-Sinulariolide Acetate Isolated from Cultured Soft Corals on HA22T Cells through the Endoplasmic Reticulum Stress Pathway and Mitochondrial Dysfunction. International Journal of Molecular Sciences, 17(11), 1787. https://doi.org/10.3390/ijms17111787






