ω-3 Fatty Acids and Cardiovascular Diseases: Effects, Mechanisms and Dietary Relevance
Abstract
:1. Introduction
2. Cardiovascular Diseases
3. ω-3 Fatty Acids and Their Metabolites
3.1. General
3.2. Eicosanoids and Specialized Pro-Resolving Mediators (SPM)
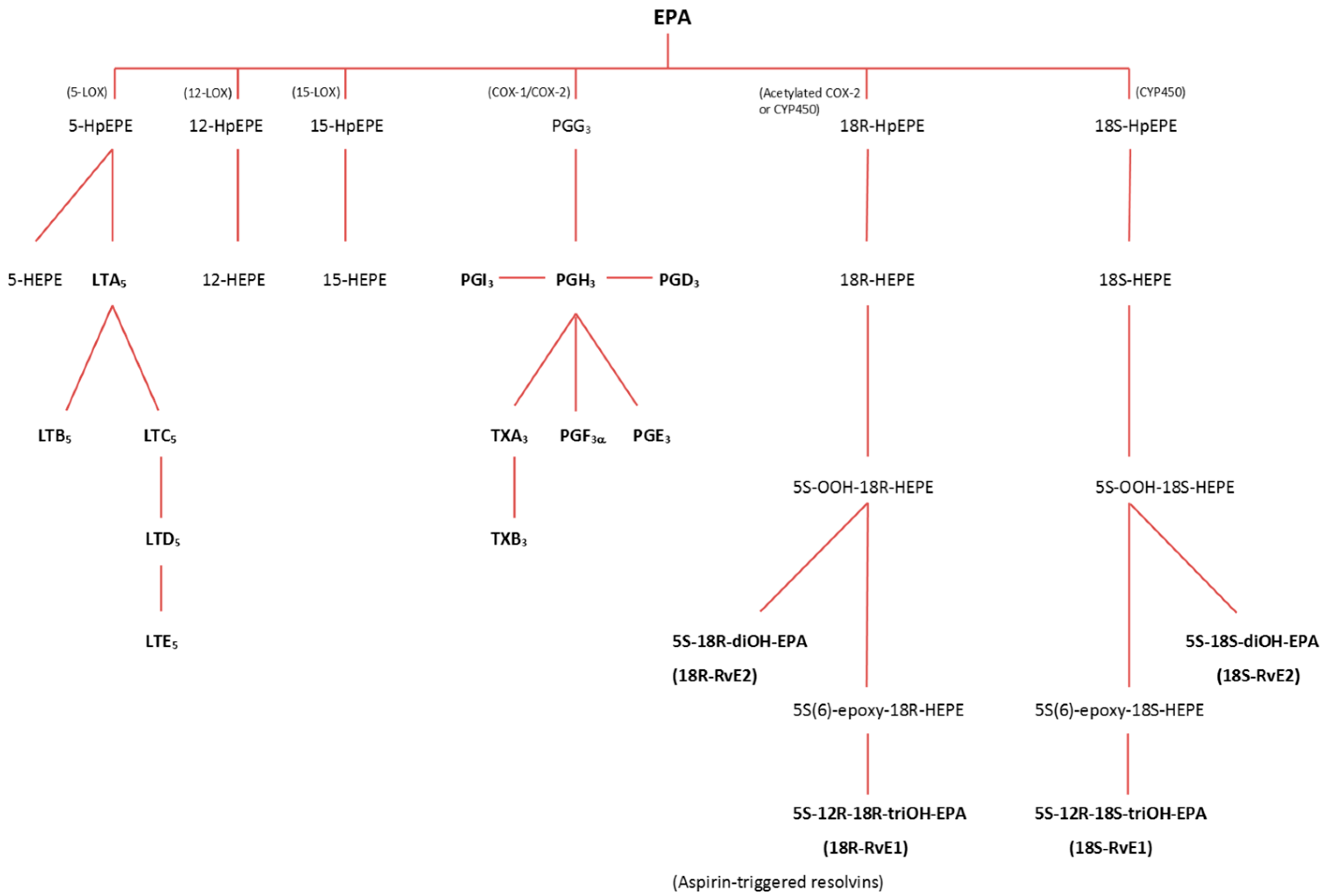
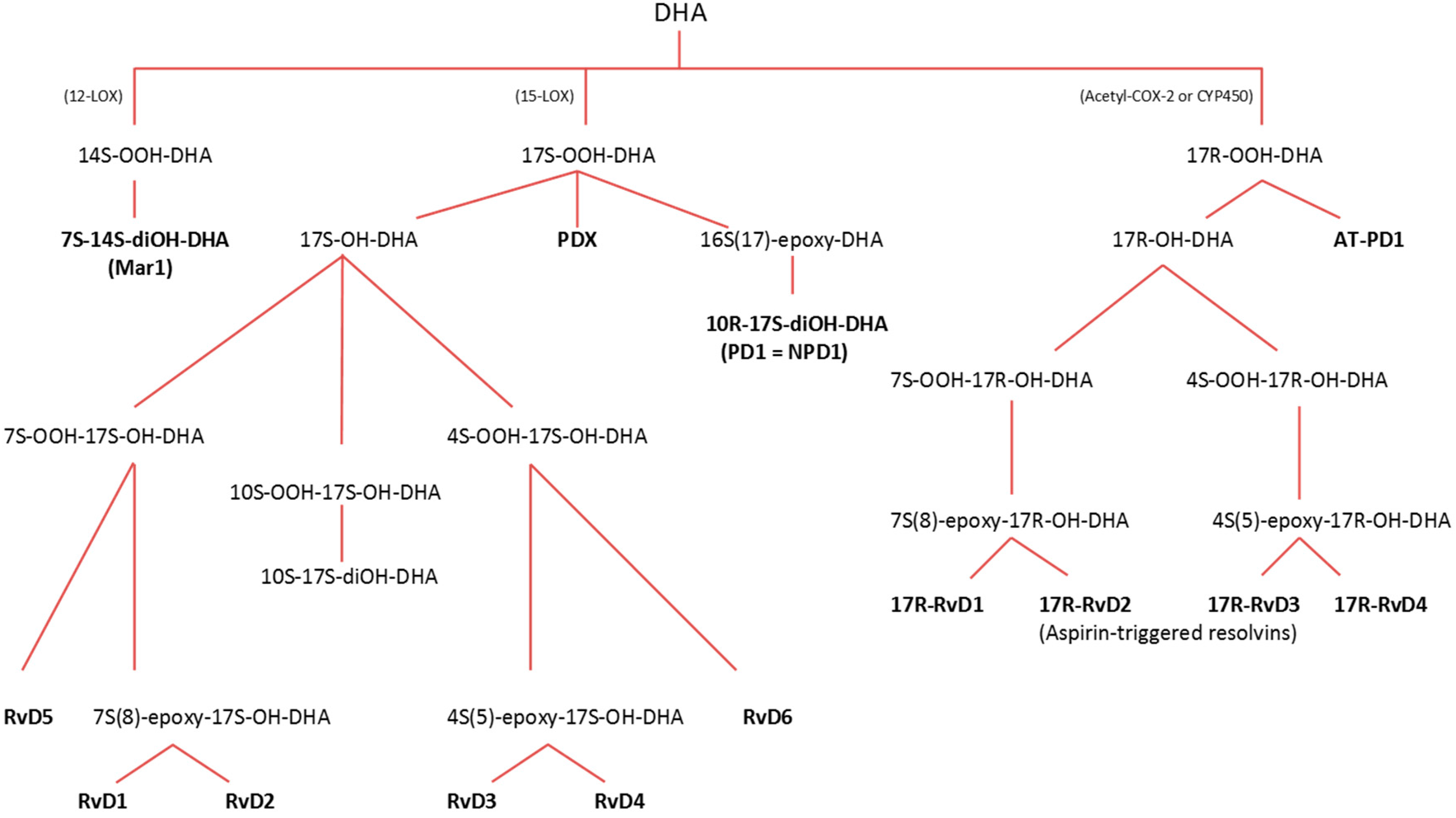
3.2.1. Lipoxygenase (LOX) Pathway
3.2.2. Cyclooxygenase (COX) Pathway
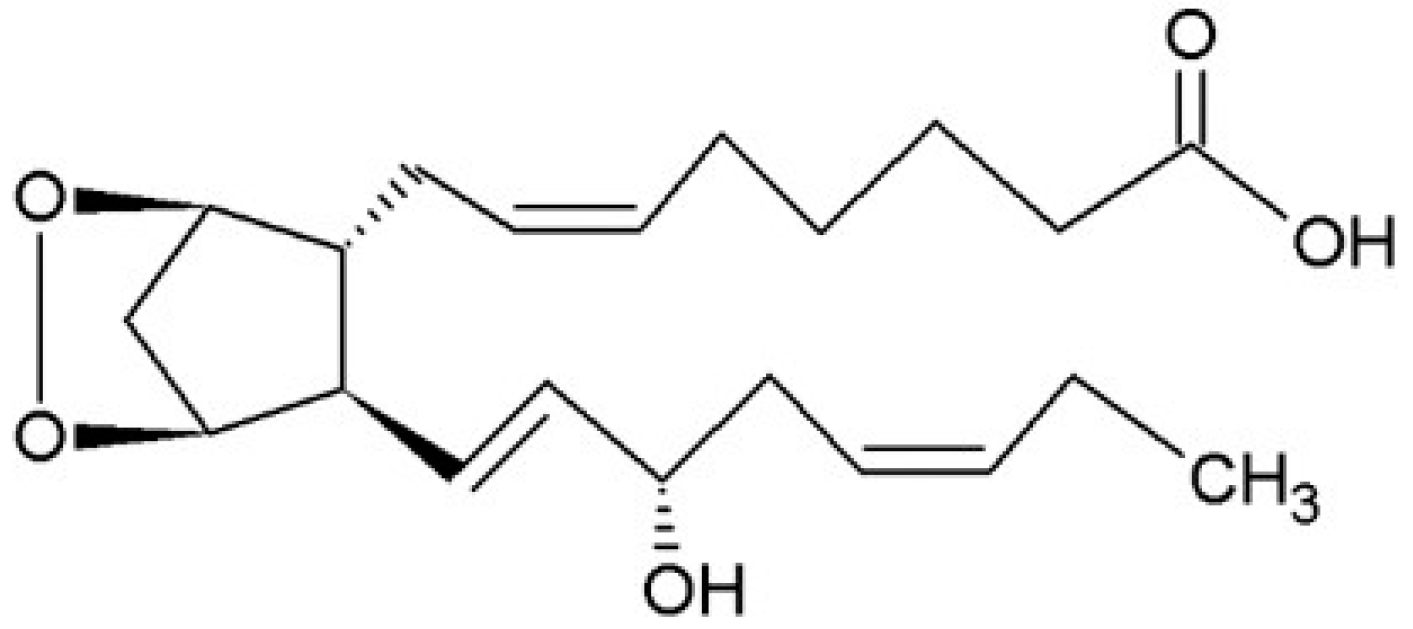
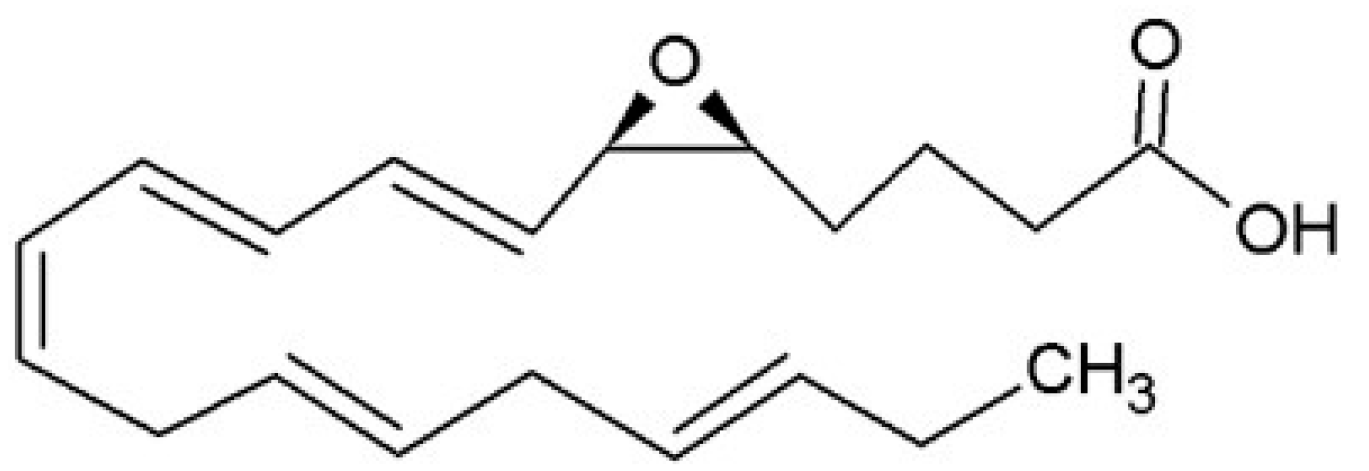
3.2.3. Cytochrome P-450 (CYP) Pathway
3.2.4. Eicosanoids, SPM and Cellular Effects on Atherosclerosis
3.3. Lipid Peroxidation and Its Products
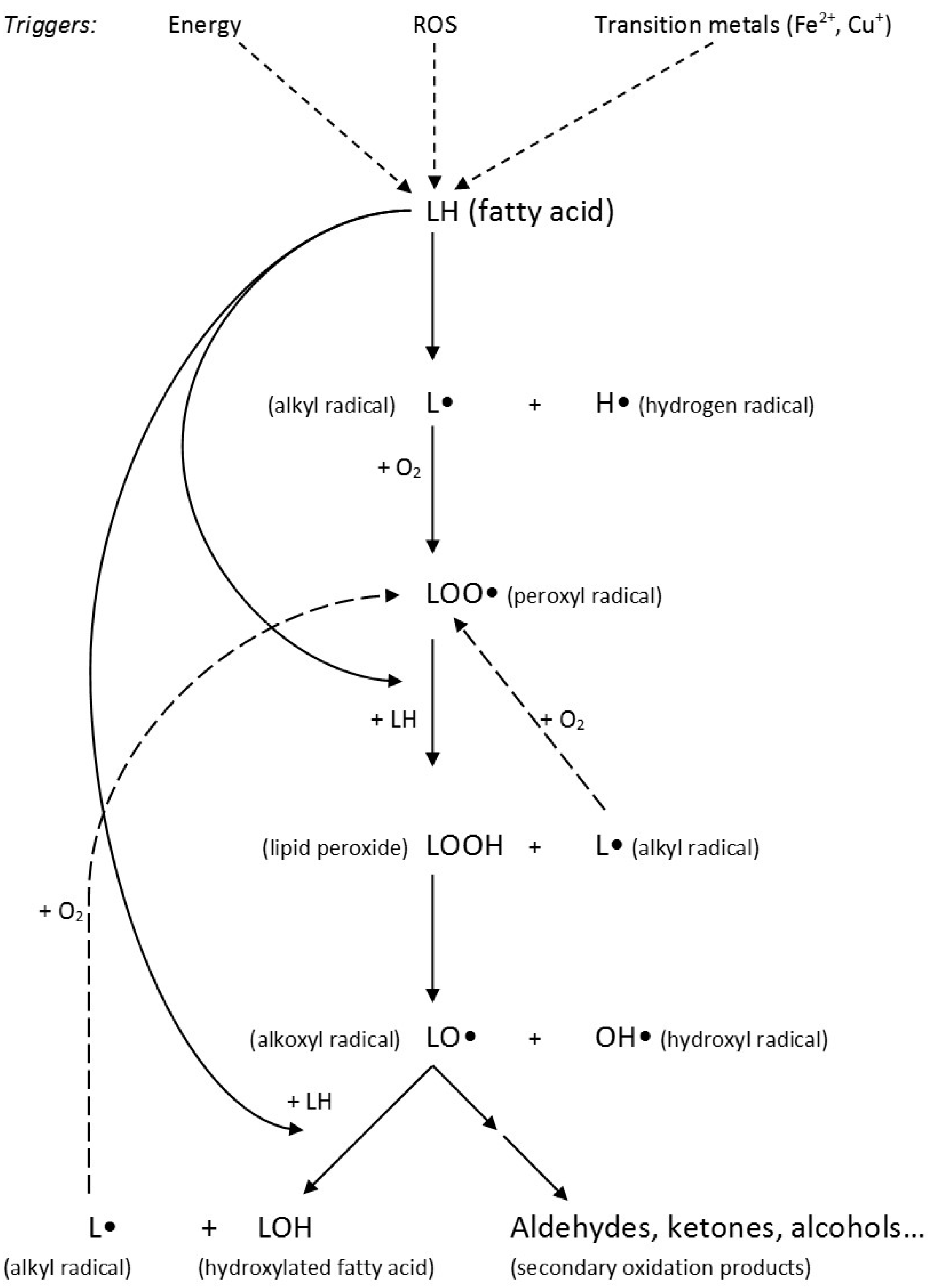
Lipid Peroxidation Products and Cellular Effects on Atherosclerosis
3.4. Clinical Effects of n-3 Intake on Atherosclerosis
Dietary Intake of Oxidized Lipids
4. Seafood or n-3 Supplements?
5. Epidemiological Studies on Consumption of Fish and n-3 Supplements
| Reference | Year | Main Conclusions |
|---|---|---|
| Zheng et al. [108] | 2012 | Included 315,812 participants; average follow up period 15.9 years. Low (1 serving/week) or moderate fish consumption (2–4 servings/week) had a prevented of CHD mortality significantly. Difficult to conclude for high fish consumption (>5 servings/week) due to the limited amount of studies of high fish consumption. |
| Djousse et al. [110] | 2012 | Included 176,441 participants; average follow up period 13.3 years. The authors concluded that there was a linear and inverse association between consumption of fish, as well as marine ω-3 fatty acids, and the risk of heart failure (HF). |
| Chowdhury et al. [111] | 2012 | Included 794,000 participants from 26 prospective cohort-studies and 12 randomized controlled trials; Higher (especially fatty) fish consumption (1 fish meal per week vs. 2 or more weekly meals) is moderately but significantly associated with a reduced risk of cerebrovascular disease. No association with marine n-3 fatty acids and cerebrovascular disease. Effect attributed to other components in fish? |
| Li et al. [112] | 2013 | Included 170,231 participants; average follow up period 9.7 years. Reported a dose-dependent inverse relationship between fish consumption and incidence of HF. Weekly fish consumption (once or more) could reduce HF incidence. |
| Xun et al. [113] | 2012 | Included 402,127 individuals with an average 12.8 years of follow-up. Observed a modest inverse association between fish intake and ischemic stroke. |
| Yinko et al. [114] | 2014 | Included 408,305 participants from 11 prospective cohort and 8 case-control studies and reported an inverse, dose-related association between fish consumption and the risk of acute coronary syndrome. |
| Reference | Year | Main Conclusions |
|---|---|---|
| Musa-Veloso et al. [115] | 2011 | Included 214,426 subjects and concluded that a daily supplementation with 250 mg EPA and DHA (or more) significantly reduced the risk of sudden cardiac death, whereas the risks of total fatal coronary events and non-fatal myocardial infarction was not significantly reduced. |
| Delgado-Lista et al. [116] | 2012 | Included 46,737 subjects with high cardiovascular risk. The marine fatty acids EPA and DHA decreased the risk of cardiovascular events, cardiac death and coronary events, especially in patients with high cardiovascular risk, but no reduced risk of all-cause mortality. |
| Rizos et al. [117] | 2013 | Included 68,680 patients. The authors concluded that there was no association between EPA and DHA supplementation and lowered risk of all-cause mortality, cardiac death, myocardial infarction and stroke. |
| Casula et al. [118] | 2013 | Including 15,348 patients with a history of cardiovascular disease. Long-term supplementation of high dose n-3 PUFA may be beneficial for the onset of cardiac death, sudden death and myocardial infarction among patients with a history of CVD. |
| Zheng et al. [119] | 2014 | Included 24,788 patients with impaired glucose metabolism (IGM). EPA and DHA had no protective effect on cardiovascular mortality, major cardiovascular events or all-cause mortality in IGM patients. |
| Wen et al. [120] | 2014 | Included 32,656 patients with coronary heart disease (CHD). Supplement of n-3 PUFA in patients with CHD is associated with a reduction in death from cardiac causes, sudden cardiac death and death from all causes, whereas no association where found on major cardiovascular events. |
| Enns et al. [121] | 2014 | Included 396 patients with peripheral arterial disease. Not sufficient evidence to indicate a beneficial effect of n-3 PUFA supplementation on the incidence of cardiovascular events and related serous complications in adults with peripheral arterial disease. |
6. Dietary Recommendations
| Authority/Organization | Country/Region | Year | Recommendation | Reference |
|---|---|---|---|---|
| The American Heart Association | USA | 2015 | A variety of (preferably fatty) fish at least twice a week | [137] |
| The Norwegian Directorate of Health/VKM | Norway | 2014 | Fish as dinner at least 2–3 times per week | [91] |
| Food and Agricultural Organization of the United Nations (FAO)/World Health Organization (WHO) | World | 2011 | At least 1–2 100 g servings of fatty fish per week | [136] |
| European Food Safety Association (EFSA) | Europe | 2010 | 250 mg EPA + DHA daily | [138] |
| Scientific Advisory Committee for Nutrition (SACN) | UK | 2004 | 450 mg EPA + DHA daily | [139] |
| International Society for the Study of Fatty Acids and Lipids (ISSFAL) | UK/Europe | 2004 | 500 mg EPA + DHA daily | [140] |
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bang, H.O.; Dyerberg, J.; Hjorne, N. Composition of food consumed by greenland eskimos. Acta Med. Scand. 1976, 200, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.O.; Dyerberg, J.; Nielsen, A.B. Plasma lipid and lipoprotein pattern in Greenlandic West-coast Eskimos. Lancet 1971, 1, 1143–1145. [Google Scholar] [CrossRef]
- Dyerberg, J.; Bang, H.O. Hemostatic function and platelet poly-unsaturated fatty acids in eskimos. Lancet 1979, 2, 433–435. [Google Scholar] [CrossRef]
- Dyerberg, J.; Bang, H.O.; Stoffersen, E.; Moncada, S.; Vane, J.R. Eicosapentaenoic acid and prevention of thrombosis and atherosclerosis? Lancet 1978, 2, 117–119. [Google Scholar] [CrossRef]
- Bjerregaard, P.; Young, T.K.; Hegele, R.A. Low incidence of cardiovascular disease among the inuit-what is the evidence? Atherosclerosis 2003, 166, 351–357. [Google Scholar] [CrossRef]
- Fodor, J.G.; Helis, E.; Yazdekhasti, N.; Vohnout, B. “Fishing” for the origins of the “Eskimos and heart disease” Story: Facts or wishful thinking? Can. J. Cardiol. 2014, 30, 864–868. [Google Scholar] [CrossRef] [PubMed]
- Cardiovascular diseases (CVDs): Fact sheet no. 317. World Health Organization: Geneva, Switzerland; Available online: http://www.who.int/mediacentre/factsheets/fs317/en/index.html (accessed on 15 August 2015).
- Mendis, S.; Puska, P.; Norrving, B. Global Atlas on Cardiovascular Disease Prevention and Control; Mendis, S., Puska, P., Eds.; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Yusuf, H.R.; Giles, W.H.; Croft, J.B.; Anda, R.F.; Casper, M.L. Impact of multiple risk factor profiles on determining cardiovascular disease risk. Prev. Med. 1998, 27, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Galley, H.F.; Webster, N.R. Physiology of the endothelium. Br. J. Anaesth. 2004, 93, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, P.O.; Lerman, L.O.; Lerman, A. Endothelial dysfunction: A marker of atherosclerotic risk. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 168–175. [Google Scholar] [CrossRef] [PubMed]
- McLaren, J.E.; Michael, D.R.; Ashlin, T.G.; Ramji, D.P. Cytokines, macrophage lipid metabolism and foam cells: Implications for cardiovascular disease therapy. Prog. Lipid Res. 2011, 50, 331–347. [Google Scholar] [CrossRef] [PubMed]
- Szmitko, P.E.; Wang, C.H.; Weisel, R.D.; de Almeida, J.R.; Anderson, T.J.; Verma, S. New markers of inflammation and endothelial cell activation: Part I. Circulation 2003, 108, 1917–1923. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.L.; Murphy, R.C. The eicosanoids: Cyclooxygenase, lipoxygenase, and epoxygenase pathways. In Biochemistry of Lipids, Lipoproteins and Membranes, 5th ed.; Vance, D.E., Vance, J.E., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Bannenberg, G.; Serhan, C.N. Specialized pro-resolving lipid mediators in the inflammatory response: An update. Biochim. Biophys. Acta 2010, 1801, 1260–1273. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Kumlin, M.; Ingelman-Sundberg, M.; Wolk, A. Dietary long-chain n-3 fatty acids for the prevention of cancer: A review of potential mechanisms. Am. J. Clin. Nutr. 2004, 79, 935–945. [Google Scholar] [PubMed]
- Calder, P.C. Marine ω-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N. Resolution phase of inflammation: Novel endogenous anti-inflammatory and proresolving lipid mediators and pathways. Annu. Rev. Immunol. 2007, 25, 101–137. [Google Scholar] [CrossRef] [PubMed]
- Stables, M.J.; Gilroy, D.W. Old and new generation lipid mediators in acute inflammation and resolution. Prog. Lipid Res. 2011, 50, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Morita, I. Distinct functions of COX-1 and COX-2. Prostaglandins Other Lipid Mediat. 2002, 68–69, 165–175. [Google Scholar] [CrossRef]
- Smith, W.L.; Langenbach, R. Why there are two cyclooxygenase isozymes. J. Clin. Investig. 2001, 107, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.L. Variants of cyclooxygenase-1 and their roles in medicine. Thromb. Res. 2003, 110, 265–268. [Google Scholar] [CrossRef]
- Barbosa-Sicard, E.; Markovic, M.; Honeck, H.; Christ, B.; Muller, D.N.; Schunck, W.H. Eicosapentaenoic acid metabolism by cytochrome P450 enzymes of the CYP2C subfamily. Biochem. Biophys. Res. Commun. 2005, 329, 1275–1281. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C.; Konkel, A.; Fischer, R.; Schunck, W.H. Cytochrome P450-dependent metabolism of ω-6 and ω-3 long-chain polyunsaturated fatty acids. Pharmacol. Rep. 2010, 62, 536–547. [Google Scholar] [CrossRef]
- Pratico, D.; Dogne, J.M. Vascular biology of eicosanoids and atherogenesis. Expert Rev. Cardiovasc. Ther. 2009, 7, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the ω-6/ω-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, E.B.; Tager, A.M.; Liu, E.; Pettersson, A.; Owman, C.; Munn, L.; Luster, A.D.; Gerszten, R.E. Mechanisms of leukotriene B-4-triggered monocyte adhesion. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1761–1767. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhao, A.; Wong, F.; Ayala, J.M.; Struthers, M.; Ujjainwalla, F.; Wright, S.D.; Springer, M.S.; Evans, J.; Cui, J.S. Leukotriene B-4 strongly increases monocyte chemoattractant protein-1 in human monocytes. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1783–1788. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.J. Differential effects of arachidonic and eicosapentaenoic acid-derived eicosanoids on polymorphonuclear transmigration across endothelial cell cultures. J. Pharmacol. Exp. Ther. 2009, 331, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Yates, C.M.; Calder, P.C.; Rainger, G.E. Pharmacology and therapeutics of ω-3 polyunsaturated fatty acids in chronic inflammatory disease. Pharmacol. Ther. 2014, 141, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Oliver, E.; McGillicuddy, F.C.; Harford, K.A.; Reynolds, C.M.; Phillips, C.M.; Ferguson, J.F.; Roche, H.M. Docosahexaenoic acid attenuates macrophage-induced inflammation and improves insulin sensitivity in adipocytes-specific differential effects between LC n-3 PUFA. J. Nutr. Biochem. 2012, 23, 1192–1200. [Google Scholar] [CrossRef] [PubMed]
- Yagi, S.; Aihara, K.; Fukuda, D.; Takashima, A.; Hara, T.; Hotchi, J.; Ise, T.; Yamaguchi, K.; Tobiume, T.; Iwase, T.; et al. Effects of docosahexaenoic acid on the endothelial function in patients with coronary artery disease. J. Atheroscler. Thromb. 2015, 22, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Qu, Q.; Xuan, W.; Fan, G.H. Roles of resolvins in the resolution of acute inflammation. Cell Biol. Int. 2015, 39, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Munoz, M.F.; Arguelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014, 2014. [Google Scholar] [CrossRef]
- Esterbauer, H.; Schaur, R.J.; Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic. Biol. Med. 1991, 11, 81–128. [Google Scholar] [CrossRef]
- Riahi, Y.; Cohen, G.; Shamni, O.; Sasson, S. Signaling and cytotoxic functions of 4-hydroxyalkenals. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E879–E886. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, R.; Haenen, G.R.M.M.; van den Berg, H.; Bast, A. Transcription factor NF-κB as a potential biomarker for oxidative stress. Br. J. Nutr. 2001, 86, S121–S127. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H. Cytotoxicity and genotoxicity of lipid-oxidation products. Am. J. Clin. Nutr. 1993, 57, 779–786. [Google Scholar]
- Long, E.K.; Murphy, T.C.; Leiphon, L.J.; Watt, J.; Morrow, J.D.; Milne, G.L.; Howard, J.R.; Picklo, M.J., Sr. Trans-4-hydroxy-2-hexenal is a neurotoxic product of docosahexaenoic (22:6; n-3) acid oxidation. J. Neurochem. 2008, 105, 714–724. [Google Scholar] [CrossRef] [PubMed]
- Je, J.H.; Lee, J.Y.; Jung, K.J.; Sung, B.; Go, E.K.; Yu, B.P.; Chung, H.Y. NF-κB activation mechanism of 4-hydroxyhexenal via NIK/IKK and p38 MAPK pathway. FEBS Lett. 2004, 566, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Ishikado, A.; Nishio, Y.; Morino, K.; Ugi, S.; Kondo, H.; Makino, T.; Kashiwagi, A.; Maegawa, H. Low concentration of 4-hydroxy hexenal increases heme oxygenase-1 expression through activation of Nrf2 and antioxidative activity in vascular endothelial cells. Biochem. Biophys. Res. Commun. 2010, 402, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Uchida, K. Role of reactive aldehyde in cardiovascular diseases. Free Radic. Biol. Med. 2000, 28, 1685–1696. [Google Scholar] [CrossRef]
- Yamada, S.; Funada, T.; Shibata, N.; Kobayashi, M.; Kawai, Y.; Tatsuda, E.; Furuhata, A.; Uchida, K. Protein-bound 4-hydroxy-2-hexenal as a marker of oxidized n-3 polyunsaturated fatty acids. J. Lipid Res. 2004, 45, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Flock, M.R.; Skulas-Ray, A.C.; Harris, W.S.; Etherton, T.D.; Fleming, J.A.; Kris-Etherton, P.M. Determinants of erythrocyte ω-3 fatty acid content in response to fish oil supplementation: A dose-response randomized controlled trial. J. Am. Heart Assoc. 2013, 2, e000513. [Google Scholar] [CrossRef] [PubMed]
- Keenan, A.H.; Pedersen, T.L.; Fillaus, K.; Larson, M.K.; Shearer, G.C.; Newman, J.W. Basal ω-3 fatty acid status affects fatty acid and oxylipin responses to high-dose n-3-HUFA in healthy volunteers. J. Lipid Res. 2012, 53, 1662–1669. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; von Schacky, C. The ω-3 index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Von Schacky, C. ω-3 index and cardiovascular health. Nutrients 2014, 6, 799–814. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S. The ω-3 index: From biomarker to risk marker to risk factor. Curr. Atheroscler. Rep. 2009, 11, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Maaloe, T.; Schmidt, E.B.; Svensson, M.; Aardestrup, I.V.; Christensen, J.H. The effect of n-3 polyunsaturated fatty acids on leukotriene B4 and leukotriene B5 production from stimulated neutrophil granulocytes in patients with chronic kidney disease. Prostaglandins Leukot. Essent. Fatty Acids 2011, 85, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Raatz, S.K.; Young, L.R.; Picklo, M.J., Sr.; Sauter, E.R.; Qin, W.; Kurzer, M.S. Total dietary fat and fatty acid content modifies plasma phospholipid fatty acids, desaturase activity indices, and urinary prostaglandin E in women. Nutr. Res. 2012, 32, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Stanke-Labesque, F.; Moliere, P.; Bessard, J.; Laville, M.; Vericel, E.; Lagarde, M. Effect of dietary supplementation with increasing doses of docosahexaenoic acid on neutrophil lipid composition and leukotriene production in human healthy volunteers. Br. J. Nutr. 2008, 100, 829–833. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.; Konkel, A.; Mehling, H.; Blossey, K.; Gapelyuk, A.; Wessel, N.; von Schacky, C.; Dechend, R.; Muller, D.N.; Rothe, M.; et al. Dietary ω-3 fatty acids modulate the eicosanoid profile in man primarily via the CYP-epoxygenase pathway. J. Lipid Res. 2014, 55, 1150–1164. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C.; Markovic, M.; Blossey, K.; Wallukat, G.; Fischer, R.; Dechend, R.; Konkel, A.; von Schacky, C.; Luft, F.C.; Muller, D.N.; et al. Arachidonic acid-metabolizing cytochrome P450 enzymes are targets of ω3 fatty acids. J. Biol. Chem. 2010, 285, 32720–32733. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Watanabe, Y.; Kumagai, M.; Fujimoto, K. Administration of dietary fish oil capsules in healthy middle-aged Japanese men with a high level of fish consumption. Int. J. Food Sci. Nutr. 2009, 60 (Suppl. 5), 136–142. [Google Scholar] [PubMed]
- Ciubotaru, I.; Lee, Y.S.; Wander, R.C. Dietary fish oil decreases C-reactive protein, interleukin-6, and triacylglycerol to HDL-cholesterol ratio in postmenopausal women on HRT. J. Nutr. Biochem. 2003, 14, 513–521. [Google Scholar] [CrossRef]
- Kiecolt-Glaser, J.K.; Belury, M.A.; Andridge, R.; Malarkey, W.B.; Glaser, R. ω-3 supplementation lowers inflammation and anxiety in medical students: A randomized controlled trial. Brain Behav. Immun. 2011, 25, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Belury, M.A.; Andridge, R.; Malarkey, W.B.; Hwang, B.S.; Glaser, R. ω-3 supplementation lowers inflammation in healthy middle-aged and older adults: A randomized controlled trial. Brain Behav. Immun. 2012, 26, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Moertl, D.; Hammer, A.; Steiner, S.; Hutuleac, R.; Vonbank, K.; Berger, R. Dose-dependent effects of ω-3-polyunsaturated fatty acids on systolic left ventricular function, endothelial function, and markers of inflammation in chronic heart failure of nonischemic origin: A double-blind, placebo-controlled, 3-arm study. Am. Heart J. 2011, 161. [Google Scholar] [CrossRef] [PubMed]
- Nodari, S.; Triggiani, M.; Campia, U.; Manerba, A.; Milesi, G.; Cesana, B.M.; Gheorghiade, M.; Cas, L.D. Effects of n-3 polyunsaturated fatty acids on left ventricular function and functional capacity in patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 2011, 57, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.T.; Shao, L.; Teng, L.L.; Hu, B.; Luo, Y.; Yu, X.; Zhang, D.F.; Zhang, H. Effects of n-3 polyunsaturated fatty acid therapy on plasma inflammatory markers and N-terminal pro-brain natriuretic peptide in elderly patients with chronic heart failure. J. Int. Med. Res. 2009, 37, 1831–1841. [Google Scholar] [CrossRef] [PubMed]
- Elvevoll, E.O.; Eilertsen, K.E.; Brox, J.; Dragnes, B.T.; Falkenberg, P.; Olsen, J.O.; Kirkhus, B.; Lamglait, A.; Osterud, B. Seafood diets: Hypolipidemic and antiatherogenic effects of taurine and n-3 fatty acids. Atherosclerosis 2008, 200, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Juarez-Lopez, C.; Klunder-Klunder, M.; Madrigal-Azcarate, A.; Flores-Huerta, S. ω-3 polyunsaturated fatty acids reduce insulin resistance and triglycerides in obese children and adolescents. Pediatr. Diabetes 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, C.; Li, L.; Man, Q.; Meng, L.; Song, P.; Froyland, L.; Du, Z.Y. Dietary inclusion of salmon, herring and pompano as oily fish reduces CVD risk markers in dyslipidaemic middle-aged and elderly Chinese women. Br. J. Nutr. 2012, 108, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, C.; Li, L.; Man, Q.; Song, P.; Meng, L.; Du, Z.Y.; Froyland, L. Inclusion of atlantic salmon in the Chinese diet reduces cardiovascular disease risk markers in dyslipidemic adult men. Nutr. Res. 2010, 30, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Baldassarre, D.; Amato, M.; Eligini, S.; Barbieri, S.S.; Mussoni, L.; Frigerio, B.; Kozakova, M.; Tremoli, E.; Sirtori, C.R.; Colli, S. Effect of n-3 fatty acids on carotid atherosclerosis and haemostasis in patients with combined hyperlipoproteinemia: A double-blind pilot study in primary prevention. Ann. Med. 2006, 38, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Ramprasath, V.R.; Eyal, I.; Zchut, S.; Jones, P.J. Enhanced increase of ω-3 index in healthy individuals with response to 4-week n-3 fatty acid supplementation from krill oil vs. fish oil. Lipids Health Dis. 2013, 12. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Ohta, M.; Nakakuki, M.; Kami, H.; Uchiyama, R.; Kawano, H.; Notsu, T.; Imada, K.; Shimano, H. Distinct regulation of plasma LDL cholesterol by eicosapentaenoic acid and docosahexaenoic acid in high fat diet-fed hamsters: Participation of cholesterol ester transfer protein and LDL receptor. Prostaglandins Leukot. Essent. Fatty Acids 2013, 88, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Root, M.; Collier, S.R.; Zwetsloot, K.A.; West, K.L.; McGinn, M.C. A randomized trial of fish oil ω-3 fatty acids on arterial health, inflammation, and metabolic syndrome in a young healthy population. Nutr. J. 2013, 12. [Google Scholar] [CrossRef]
- Garaiova, I.; Muchova, J.; Nagyova, Z.; Mislanova, C.; Oravec, S.; Dukat, A.; Wang, D.; Plummer, S.F.; Durackova, Z. Effect of a plant sterol, fish oil and B vitamin combination on cardiovascular risk factors in hypercholesterolemic children and adolescents: A pilot study. Nutr. J. 2013, 12. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Moro, T.; Bosco, G.; Bianco, A.; Grimaldi, K.A.; Camporesi, E.; Mangar, D. Effects of n-3 polyunsaturated fatty acids (ω-3) supplementation on some cardiovascular risk factors with a ketogenic Mediterranean diet. Mar. Drugs 2015, 13, 996–1009. [Google Scholar] [PubMed]
- Iketani, T.; Takazawa, K.; Yamashina, A. Effect of eicosapentaenoic acid on central systolic blood pressure. Prostaglandins Leukot. Essent. Fatty Acids 2013, 88, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Ramel, A.; Martinez, J.A.; Kiely, M.; Bandarra, N.M.; Thorsdottir, I. Moderate consumption of fatty fish reduces diastolic blood pressure in overweight and obese European young adults during energy restriction. Nutrition 2010, 26, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, B.M.; Vessby, B.; Uusitupa, M.; Berglund, L.; Pedersen, E.; Riccardi, G.; Rivellese, A.A.; Tapsell, L.; Hermansen, K.; Group, K.S. Effects of dietary saturated, monounsaturated, and n-3 fatty acids on blood pressure in healthy subjects. Am. J. Clin. Nutr. 2006, 83, 221–226. [Google Scholar] [PubMed]
- Campbell, F.; Dickinson, H.O.; Critchley, J.A.; Ford, G.A.; Bradburn, M. A systematic review of fish-oil supplements for the prevention and treatment of hypertension. Eur. J. Prev. Cardiol. 2013, 20, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.E.; van Elswyk, M.; Alexander, D.D. Long-chain ω-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: A meta-analysis of randomized controlled trials. Am. J. Hypertens. 2014, 27, 885–896. [Google Scholar] [CrossRef] [PubMed]
- Awada, M.; Soulage, C.O.; Meynier, A.; Debard, C.; Plaisancie, P.; Benoit, B.; Picard, G.; Loizon, E.; Chauvin, M.A.; Estienne, M.; et al. Dietary oxidized n-3 PUFA induce oxidative stress and inflammation: Role of intestinal absorption of 4-HHE and reactivity in intestinal cells. J. Lipid Res. 2012, 53, 2069–2080. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Harper, A.F.; Zhao, J.; Dalloul, R.A. Effects of a dietary antioxidant blend and vitamin E on growth performance, oxidative status, and meat quality in broiler chickens fed a diet high in oxidants. Poult. Sci. 2014, 93, 1649–1657. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Harper, A.F.; Zhao, J.; Estienne, M.J.; Dalloul, R.A. Supplementing antioxidants to pigs fed diets high in oxidants: I. Effects on growth performance, liver function, and oxidative status. J. Anim. Sci. 2014, 92, 5455–5463. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.P.; Liu, H.L.; Wang, C.G.; Yang, P.; Sun, C.B.; Chan, S.M. Effect of oxidized fish oil on growth performance and oxidative stress of Litopenaeus vannamei. Aquac. Nutr. 2015, 21, 121–127. [Google Scholar] [CrossRef]
- Eilertsen, K.E.; Maehre, H.K.; Cludts, K.; Olsen, J.O.; Hoylaerts, M.F. Dietary enrichment of apolipoprotein E-deficient mice with extra virgin olive oil in combination with seal oil inhibits atherogenesis. Lipids Health Dis. 2011, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Eilertsen, K.E.; Maehre, H.K.; Jensen, I.J.; Devold, H.; Olsen, J.O.; Lie, R.K.; Brox, J.; Berg, V.; Elvevoll, E.O.; Osterud, B. A wax ester and astaxanthin-rich extract from the marine copepod Calanus finmarchicus attenuates atherogenesis in female apolipoprotein E-deficient mice. J. Nutr. 2012, 142, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Ottestad, I.; Vogt, G.; Retterstol, K.; Myhrstad, M.C.; Haugen, J.E.; Nilsson, A.; Ravn-Haren, G.; Nordvi, B.; Bronner, K.W.; Andersen, L.F.; et al. Oxidised fish oil does not influence established markers of oxidative stress in healthy human subjects: A randomised controlled trial. Br. J. Nutr. 2012, 108, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Hernandez, V.M.; Gallar, M.; Sanchez-Soriano, J.; Micol, V.; Roche, E.; Garcia-Garcia, E. Effect of ω-3 dietary supplements with different oxidation levels in the lipidic profile of women: A randomized controlled trial. Int. J. Food Sci. Nutr. 2013, 64, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Asghari, L.; Zeynali, F.; Sahari, M.A. Effects of boiling, deep-frying, and microwave treatment on the proximate composition of rainbow trout fillets: Changes in fatty acids, total protein, and minerals. J. Appl. Ichthyol. 2013, 29, 847–853. [Google Scholar] [CrossRef]
- Mierke-Klemeyer, S.; Larsen, R.; Oehlenschlager, J.; Maehre, H.; Elvevoll, E.O.; Bandarra, N.M.; Parreira, R.; Andrade, A.M.; Nunes, M.L.; Schram, E.; et al. Retention of health-related beneficial components during household preparation of selenium-enriched African catfish (Clarias gariepinus) fillets. Eur. Food Res. Technol. 2008, 227, 827–833. [Google Scholar] [CrossRef]
- Perez-Palacios, T.; Petisca, C.; Casal, S.; Ferreira, I.M.P.L.V.O. Changes in chemical composition of frozen coated fish products during deep-frying. Int. J. Food Sci. Nutr. 2014, 65, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Pirini, M.; Testi, S.; Ventrella, V.; Pagliarani, A.; Badiani, A. Blue-back fish: Fatty acid profile in selected seasons and retention upon baking. Food Chem. 2010, 123, 306–314. [Google Scholar] [CrossRef]
- Weichselbaum, E.; Coe, S.; Buttriss, J.; Stanner, S. Fish in the diet: A review. Nutr. Bull. 2013, 38, 128–177. [Google Scholar] [CrossRef]
- Larsen, R.; Eilertsen, K.E.; Maehre, H.K.; Jensen, I.J.; Elvevoll, E.O. Taurine content in marine foods—Beneficial health effects. In Bioactive Compounds from Marine Foods: Plant and Animal Sources; Hernández-Ledesma, B., Herrero, M., Eds.; Wiley-Blackwell Oxford: Chichester, UK, 2014; pp. 249–268. [Google Scholar]
- Jensen, I.J.; Abrahamsen, H.; Maehre, H.K.; Elvevoll, E.O. Changes in antioxidative capacity of saithe (Pollachius virens) and Shrimp (Pandalus borealis) during in vitro digestion. J. Agric. Food Chem. 2009, 57, 10928–10932. [Google Scholar] [CrossRef] [PubMed]
- Skåre, J.U.; Brantsæter, A.L.; Frøyland, L.; Hemre, G.L.; Knutsen, H.K.; Lillegaard, I.T.L.; Torstensen, B. Benefit-risk assessment of fish and fish products in the Norwegian diet—An update. Eur. J. Nutr. Food Saf. 2015, 5, 260–266. [Google Scholar] [CrossRef]
- Oterhals, A.; Kvamme, B.; Berntssen, M.H. Modeling of a short-path distillation process to remove persistent organic pollutants in fish oil based on process parameters and quantitative structure properties relationships. Chemosphere 2010, 80, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Oterhals, A.; Berntssen, M.H. Effects of refining and removal of persistent organic pollutants by short-path distillation on nutritional quality and oxidative stability of fish oil. J. Agric. Food Chem. 2010, 58, 12250–12259. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, F.; Yang, L.; Zu, Y.G.; Wang, H.; Qu, S.Z.; Zhang, Y. Oxidative stability of fish oil supplemented with carnosic acid compared with synthetic antioxidants during long-term storage. Food Chem. 2011, 128, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Zuta, P.C.; Simpson, B.K.; Zhao, X.; Leclerc, L. The effect of α-tocopherol on the oxidation of mackerel oil. Food Chem. 2007, 100, 800–807. [Google Scholar] [CrossRef]
- Ahn, J.H.; Kim, Y.P.; Seo, E.M.; Choi, Y.K.; Kim, H.S. Antioxidant effect of natural plant extracts on the microencapsulated high oleic sunflower oil. J. Food Eng. 2008, 84, 327–334. [Google Scholar] [CrossRef]
- Kagami, Y.; Sugimura, S.; Fujishima, N.; Matsuda, K.; Kometani, T.; Matsumura, Y. Oxidative stability, structure, and physical characteristics of microcapsules formed by spray drying of fish oil with protein and dextrin wall materials. J. Food Sci. 2003, 68, 2248–2255. [Google Scholar] [CrossRef]
- Bottcher, S.; Steinhauser, U.; Drusch, S. Off-flavour masking of secondary lipid oxidation products by pea dextrin. Food Chem. 2015, 169, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Kralovec, J.A.; Zhang, S.C.; Zhang, W.; Barrow, C.J. A review of the progress in enzymatic concentration and microencapsulation of ω-3 rich oil from fish and microbial sources. Food Chem. 2012, 131, 639–644. [Google Scholar] [CrossRef]
- Ritter, J.C.S.; Budge, S.M.; Jovica, F.; Reid, A.J.M. Oxidation rates of triacylglycerol and ethyl ester fish oils. J. Am. Oil Chem. Soc. 2015, 92, 561–569. [Google Scholar] [CrossRef]
- Bosch, J.; Gerstein, H.C.; Dagenais, G.R.; Diaz, R.; Dyal, L.; Jung, H.; Maggiono, A.P.; Probstfield, J.; Ramachandran, A.; Riddle, M.C.; et al. n-3 fatty acids and cardiovascular outcomes in patients with dysglycemia. N. Engl. J. Med. 2012, 367, 309–318. [Google Scholar] [PubMed]
- Chrysohoou, C.; Panagiotakos, D.B.; Pitsavos, C.; Skoumas, J.; Krinos, X.; Chloptsios, Y.; Nikolaou, V.; Stefanadis, C. Long-term fish consumption is associated with protection against arrhythmia in healthy persons in a Mediterranean region—The ATTICA study. Am. J. Clin. Nutr. 2007, 85, 1385–1391. [Google Scholar] [PubMed]
- Mozaffarian, D.; Marchioli, R.; Macchia, A.; Silletta, M.G.; Ferrazzi, P.; Gardner, T.J.; Latini, R.; Libby, P.; Lombardi, F.; O’Gara, P.T.; et al. Fish oil and postoperative atrial fibrillation the ω-3 fatty acids for prevention of post-operative atrial fibrillation (OPERA) randomized trial. JAMA 2012, 308, 2001–2011. [Google Scholar] [CrossRef] [PubMed]
- Roncaglioni, M.C.; Tombesi, M.; Avanzini, F.; Barlera, S.; Caimi, V.; Longoni, P.; Marzona, I.; Milani, V.; Silletta, M.G.; Tognoni, G.; et al. n-3 fatty acids in patients with multiple cardiovascular risk factors. N. Engl. J. Med. 2013, 368, 1800–1808. [Google Scholar] [PubMed]
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R.; Barlera, S.; Franzosi, M.G.; Latini, R.; Lucci, D.; Nicolosi, G.L.; Porcu, M.; Tognoni, G.; et al. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar] [PubMed]
- Virtanen, J.K.; Mozaffarian, D.; Chiuve, S.E.; Rimm, E.B. Fish consumption and risk of major chronic disease in men. Am. J. Clin. Nutr. 2008, 88, 1618–1625. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised openlabel, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Zheng, J.; Huang, T.; Yu, Y.; Hu, X.; Yang, B.; Li, D. Fish consumption and CHD mortality: An updated meta-analysis of seventeen cohort studies. Public. Health Nutr. 2012, 15, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, A.M.; Sun, Q.; Hu, F.B.; Stampfer, M.J.; Manson, J.E.; Willett, W.C. Major dietary protein sources and risk of coronary heart disease in women. Circulation 2010, 122, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Djousse, L.; Akinkuolie, A.O.; Wu, J.H.Y.; Ding, E.L.; Gaziano, J.M. Fish consumption, ω-3 fatty acids and risk of heart failure: A meta-analysis. Clin. Nutr. 2012, 31, 846–853. [Google Scholar] [PubMed]
- Chowdhury, R.; Stevens, S.; Gorman, D.; Pan, A.; Warnakula, S.; Chowdhury, S.; Ward, H.; Johnson, L.; Crowe, F.; Hu, F.B.; et al. Association between fish consumption, long chain ω-3 fatty acids, and risk of cerebrovascular disease: Systematic review and meta-analysis. BMJ 2012, 345, e6698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.H.; Zhou, C.H.; Pei, H.J.; Zhou, X.L.; Li, L.H.; Wu, Y.J.; Hui, R.T. Fish consumption and incidence of heart failure: A meta-analysis of prospective cohort studies. Chin. Med. J. 2013, 126, 942–948. [Google Scholar] [PubMed]
- Xun, P.; Qin, B.; Song, Y.; Nakamura, Y.; Kurth, T.; Yaemsiri, S.; Djousse, L.; He, K. Fish consumption and risk of stroke and its subtypes: Accumulative evidence from a meta-analysis of prospective cohort studies. Eur. J. Clin. Nutr. 2012, 66, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Yinko, S.S.L.L.; Stark, K.D.; Thanassoulis, G.; Pilote, L. Fish consumption and acute coronary syndrome: A meta-analysis. Am. J. Med. 2014, 127, 848–852. [Google Scholar] [CrossRef] [PubMed]
- Musa-Veloso, K.; Binns, M.A.; Kocenas, A.; Chung, C.; Rice, H.; Oppedal-Olsen, H.; Lloyd, H.; Lemke, S. Impact of low v. moderate intakes of long-chain n-3 fatty acids on risk of coronary heart disease. Br. J. Nutr. 2011, 106, 1129–1141. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Lista, J.; Perez-Martinez, P.; Lopez-Miranda, J.; Perez-Jimenez, F. Long chain ω-3 fatty acids and cardiovascular disease: A systematic review. Br. J. Nutr. 2012, 107 (Suppl. 2), S201–S213. [Google Scholar] [CrossRef] [PubMed]
- Rizos, E.C.; Ntzani, E.E.; Bika, E.; Kostapanos, M.S.; Elisaf, M.S. Association between ω-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Casula, M.; Soranna, D.; Catapano, A.L.; Corrao, G. Long-term effect of high dose ω-3 fatty acid supplementation for secondary prevention of cardiovascular outcomes: A meta-analysis of randomized, placebo controlled trials. Atheroscler. Suppl. 2013, 14, 243–251. [Google Scholar] [CrossRef]
- Zheng, T.; Zhao, J.; Wang, Y.; Liu, W.; Wang, Z.; Shang, Y.; Zhang, W.; Zhang, Y.; Zhong, M. The limited effect of ω-3 polyunsaturated fatty acids on cardiovascular risk in patients with impaired glucose metabolism: A meta-analysis. Clin. Biochem. 2014, 47, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.T.; Dai, J.H.; Gao, Q. Effects of ω-3 fatty acid on major cardiovascular events and mortality in patients with coronary heart disease: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Enns, J.E.; Yeganeh, A.; Zarychanski, R.; Abou-Setta, A.M.; Friesen, C.; Zahradka, P.; Taylor, C.G. The impact of ω-3 polyunsaturated fatty acid supplementation on the incidence of cardiovascular events and complications in peripheral arterial disease: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Burr, M.L.; Gilbert, J.F.; Holliday, R.M.; Elwood, P.C.; Fehily, A.M.; Rogers, S.; Sweetnam, P.M.; Deadman, N.M. Effects of changes in fat, fish, and fiber intakes on death and myocardial reinfarction—Diet and reinfarction trial (DART). Lancet 1989, 2, 757–761. [Google Scholar] [PubMed]
- Valagussa, F.; Franzosi, M.G.; Geraci, E.; Mininni, N.; Nicolosi, G.L.; Santini, M.; Tavazzi, L.; Vecchio, C.; Marchioli, R.; Bomba, E.; et al. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-Prevenzione trial. Lancet 1999, 354, 447–455. [Google Scholar]
- FAO (2014). The state of world fisheries and aquaculture 2014 (Online). The Food and Agriculture Organization of the United Nations: Rome, Italy; Available online: http://www.fao.org/3/a-i3720e.pdf (accessed on 10 August 2015).
- Mozaffarian, D.; Rimm, E.B. Fish intake, contaminants, and human health—Evaluating the risks and the benefits. JAMA 2006, 296, 1885–1899. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, C.D.; Cramer, H.; Michalsen, A.; Kessler, C.; Steckhan, N.; Choi, K.; Dobos, G. Effects of high phenolic olive oil on cardiovascular risk factors: A systematic review and meta-analysis. Phytomedicine 2015, 22, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Albert, B.B.; Cameron-Smith, D.; Hofman, P.L.; Cutfield, W.S. Oxidation of marine ω-3 supplements and human health. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Albert, B.B.; Derraik, J.G.B.; Cameron-Smith, D.; Hofman, P.L.; Tumanov, S.; Villas-Boas, S.G.; Garg, M.L.; Cutfield, W.S. Fish oil supplements in New Zealand are highly oxidised and do not meet label content of n-3 PUFA. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Halvorsen, B.L.; Blomhoff, R. Determination of lipid oxidation products in vegetable oils and marineω-3 supplements. Food Nutr. Res. 2011, 55. [Google Scholar] [CrossRef]
- Opperman, M.; Benade, S. Analysis of the ω-3 fatty acid content of South African fish oil supplements: A follow-up study. Cardiovasc. J. Afr. 2013, 24, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, A.C.; Cladis, D.P.; Santerre, C.R. A comparison of actual vs. stated label amounts of EPA and DHA in commercial ω-3 dietary supplements in the United States. J. Sci. Food Agric. 2015, 95, 1260–1267. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Evolutionary aspects of diet: The ω-6/ω-3 ratio and the brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the ratio of ω-6/ω-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am. J. Clin. Nutr. 2006, 83, 1505S–1519S. [Google Scholar] [PubMed]
- Patterson, E.; Wall, R.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. Health implications of high dietary ω-6 polyunsaturated Fatty acids. J. Nutr. Metab. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Report of the Joint FAO/WHO Expert Consultation on the Risks and Benefits of Fish Consumption. Food and Agriculture Organization of the United Nations; World Health Organization: Rome, Italy; Geneva, Switzerland, 2011. [Google Scholar]
- AHA (2015). AHA recommendation (Online). The American Heart Association: Dallas, TX, USA; Available online: http://www.heart.org/HEARTORG/GettingHealthy/NutritionCenter/Fish-101_UCM_305986_Article.jsp#aha_recommendation (accessed on 10 August 2015).
- EFSA (2010). EFSA sets European dietary reference values for nutrient intakes (Online). European Food Safety Authority: Parma, Italy; Available online: http://www.efsa.europa.eu/en/press/news/nda100326.htm (accessed on 15 August 2015).
- UK SACN. Advice on Fish Consumption: Benefits and Risks. In EDID Collection, Proceedings of the Scientific Advisory Committee on Nutrition, Norwich, UK, 18 June 2004.
- ISSFAL (2004). Recommendations for Intake of Polyunsaturated Fatty Acids in Healthy Adults (Online). In Proceedings of the International Society for the Study of Fatty Acids and Lipids, Brighton, UK; Available online: http://www.issfal.org/statements/pufa-recommendations/statement-3 (accessed on 10 August 2015).
- Elvevoll, E.O.; Barstad, H.; Breimo, E.S.; Brox, J.; Eilertsen, K.E.; Lund, T.; Olsen, J.O.; Osterud, B. Enhanced incorporation of n-3 fatty acids from fish compared with fish oils. Lipids 2006, 41, 1109–1114. [Google Scholar] [CrossRef] [PubMed]
- Galli, C.; Marangoni, F. n-3 fatty acids in the Mediterranean diet. Prostaglandins Leukot. Essent. Fatty Acids 2006, 75, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Rise, P.; Barassi, M.C.; Marangoni, F.; Galli, C. Dietary intake of fish vs. formulations leads to higher plasma concentrations of n-3 fatty acids. Lipids 2003, 38, 415–418. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maehre, H.K.; Jensen, I.-J.; Elvevoll, E.O.; Eilertsen, K.-E. ω-3 Fatty Acids and Cardiovascular Diseases: Effects, Mechanisms and Dietary Relevance. Int. J. Mol. Sci. 2015, 16, 22636-22661. https://doi.org/10.3390/ijms160922636
Maehre HK, Jensen I-J, Elvevoll EO, Eilertsen K-E. ω-3 Fatty Acids and Cardiovascular Diseases: Effects, Mechanisms and Dietary Relevance. International Journal of Molecular Sciences. 2015; 16(9):22636-22661. https://doi.org/10.3390/ijms160922636
Chicago/Turabian StyleMaehre, Hanne K., Ida-Johanne Jensen, Edel O. Elvevoll, and Karl-Erik Eilertsen. 2015. "ω-3 Fatty Acids and Cardiovascular Diseases: Effects, Mechanisms and Dietary Relevance" International Journal of Molecular Sciences 16, no. 9: 22636-22661. https://doi.org/10.3390/ijms160922636
APA StyleMaehre, H. K., Jensen, I.-J., Elvevoll, E. O., & Eilertsen, K.-E. (2015). ω-3 Fatty Acids and Cardiovascular Diseases: Effects, Mechanisms and Dietary Relevance. International Journal of Molecular Sciences, 16(9), 22636-22661. https://doi.org/10.3390/ijms160922636






