Intracellular Delivery of Molecular Cargo Using Cell-Penetrating Peptides and the Combination Strategies
Abstract
:1. Introduction
| Name | Sequence | References |
|---|---|---|
| Poly-R peptides | Rx * | [19,20,2122] |
| TAT (47–57) | YGRKKRRQRRR | [23] |
| TAT (49–60) | RKKRRQRRRPPQ | [21] |
| PTD4 | YARAAARQARA | [24] |
| PTD5 | RRQRRTSKLMKRGG | [25] |
| TP10 | AGYLLGKINLKALAALAKKIL | [26] |
| M918 | MVTVLFRRLRIRRACGPPRVRV | [27] |
| pAntp (43–58) [Penetratin] | RQIKIWFQNRRMKWKK | [21] |
| KNO | KQINNWFINQRKRHWK | [28] |
| Hph-1 | YARVRRRGPRR | [29] |
| HIV-1 Rev (34–50) ANP (43–58) [Antennapedia] | TRQARRNRRRRWRERQR-GC RQIKIWFQNRRMKWKK-GC-CO | [30] |
| POD | GGG [ARKKAAKA] 4 | [31] |
| S413-PV | ALWKTLLKKVLKAPKKKRKV | [32] |
| S41 | CVQWSLLRGYQPC | [33] |
| pVEC | LLIILRRRIRKQAHAHSK | [34] |

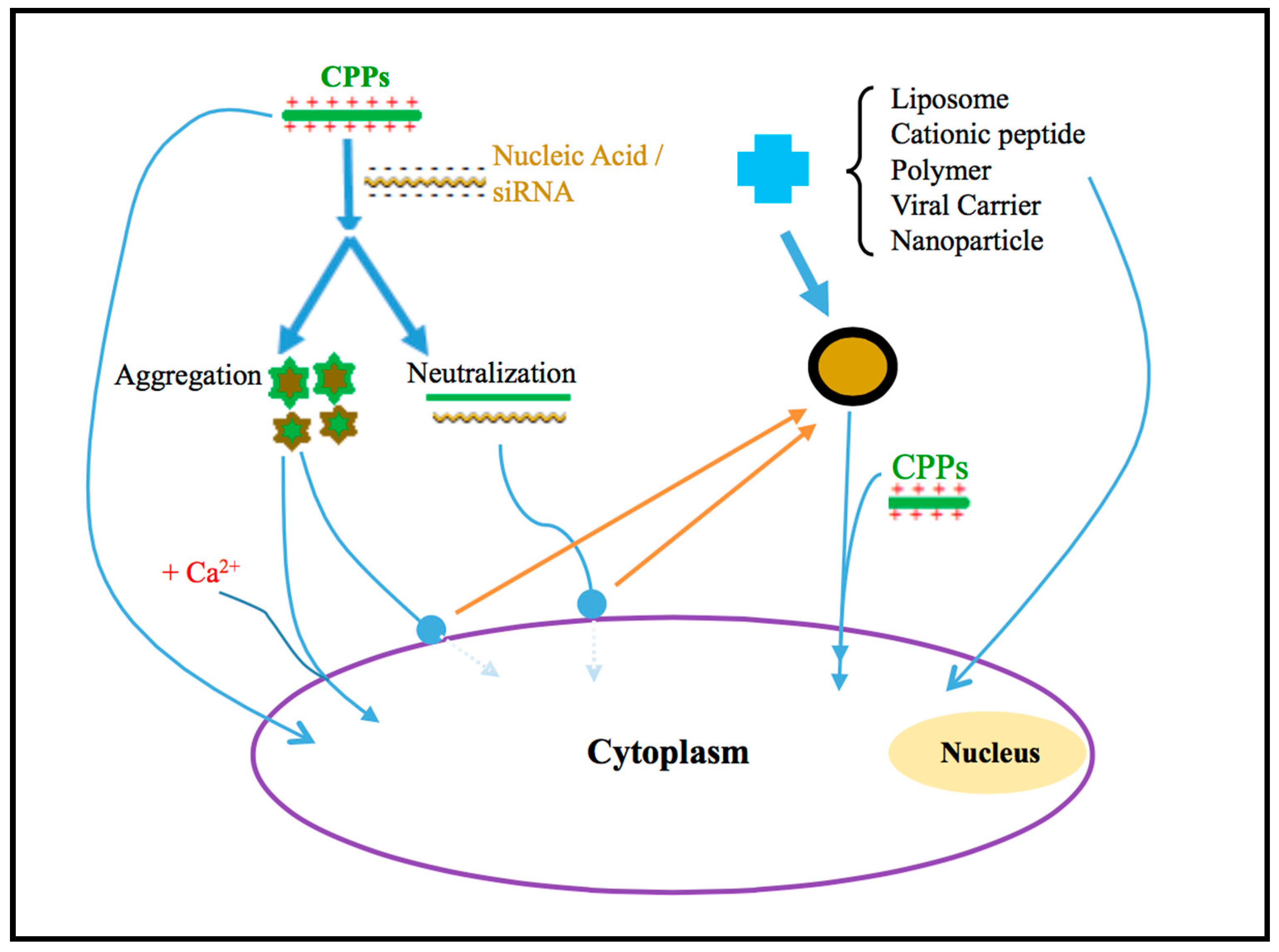
2. CPPs-Modified Liposome
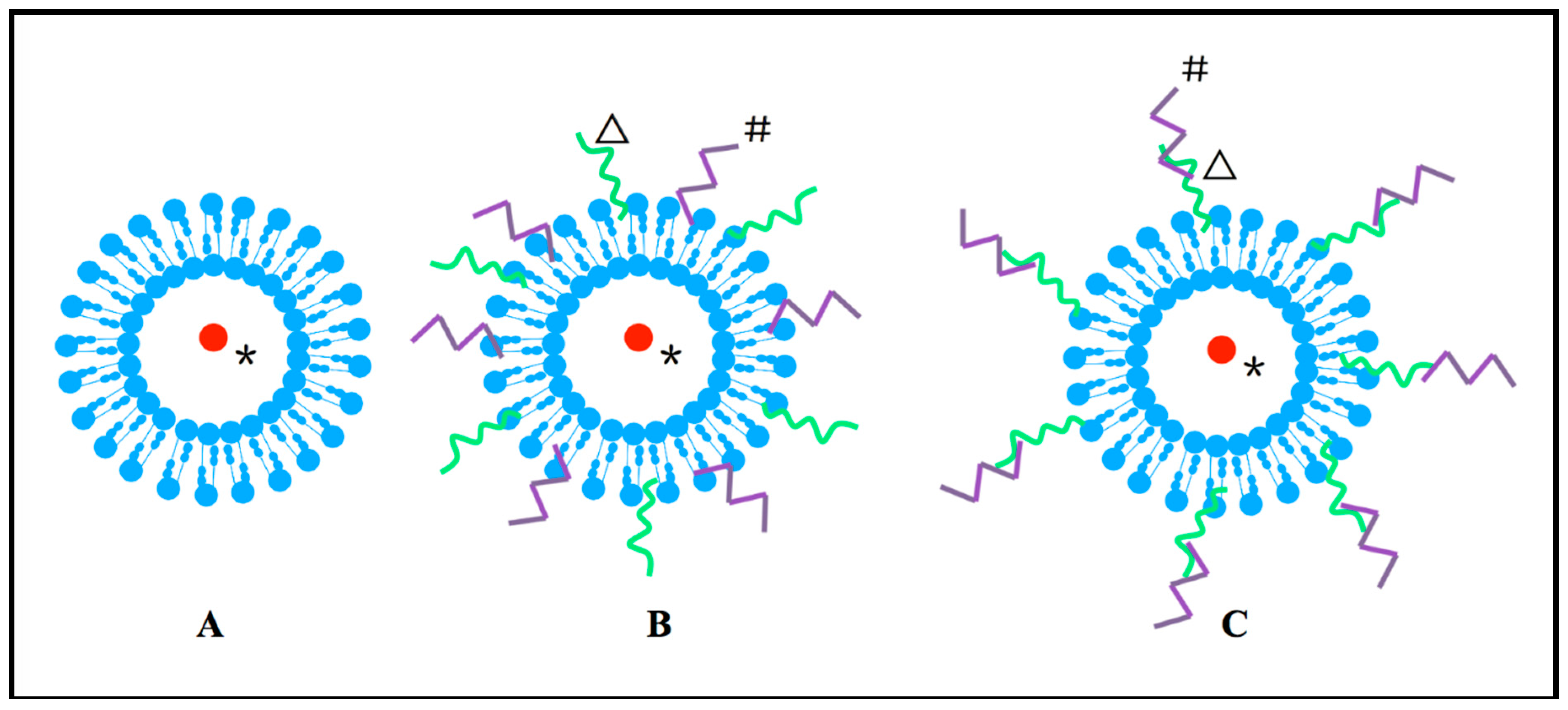
3. Combination of CPPs with Polymer
4. Combination of CPPs with Cationic Peptide
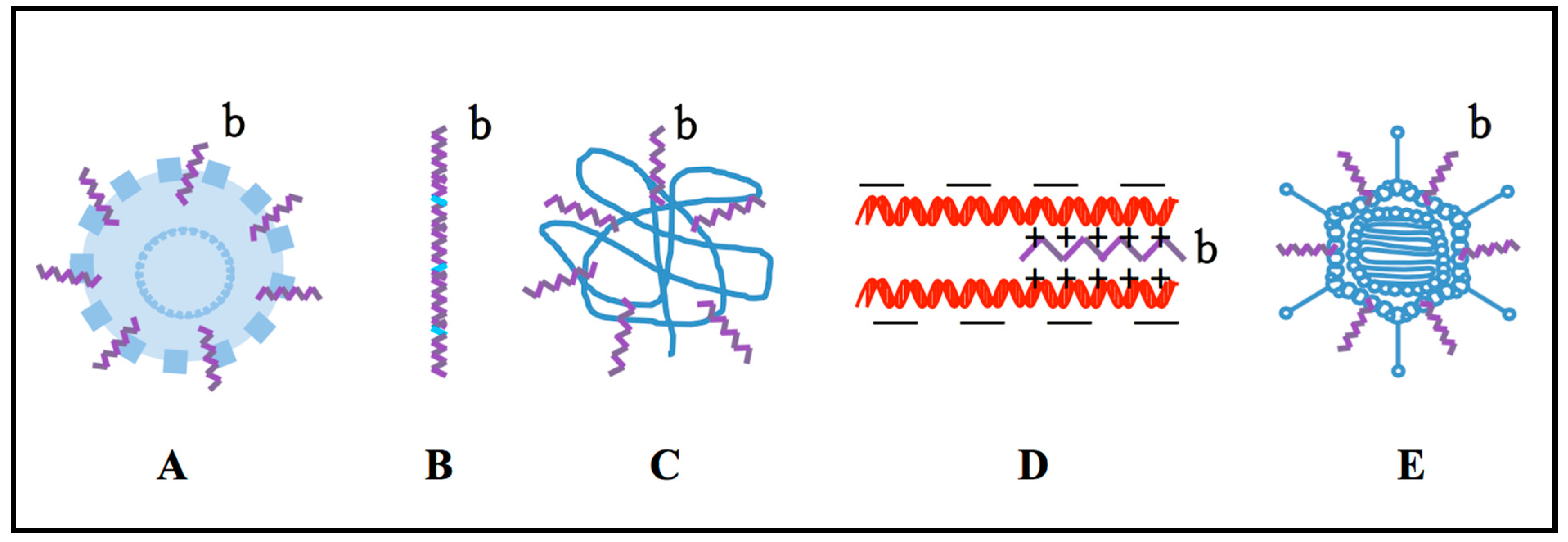
5. Binding of CPPs with the Viral Carrier
6. Calcium and CPPs
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| Abbreviation | Full Name |
| Arg | Arginine |
| Asp | Aspartic acid |
| AVV | Adeno-associated virus |
| BBB | Blood brain barrier |
| C | Cysteine |
| CAR | Coxsackievirus and adenovirus receptor |
| CBA-DAH | Cystamine bisacrylamide-diaminohexane |
| CNS | Central nervous system diseases |
| CPP | Cell penetrating peptide |
| dsRBD/DRBD | Doubled RNA binding domain |
| G | Glycine |
| Glu | Glutamic acid |
| H | Histidine |
| HA2 | hemagglutinin peptide |
| HIV | Human immunodeficiency virus |
| L | Leucine |
| LMWP | Cell penetrating peptide of LMWP |
| Lys/K | Lysine |
| MAP | Model amphipathic peptide |
| MNPs | Magnetic nanoparticles |
| MPEG | Methoxy poly (ethylene glycol) |
| P | Proline |
| PCL | Poly(epsilon-caprolactone) |
| PEG | Polyethylenglycol |
| PLL | Poly-L-Lysine |
| pLys | Polylysine |
| PTD | Protein transduction domain |
| PTD-DRBD | CPP of PTD modified dsRNA binding domain |
| Q | Glutamine |
| R | Arginine |
| R8 | Polyarginine8 |
| RNAi | RNA interference |
| shRNA | Short hairpin RNA |
| siRNA | Small interfering RNA |
| TAT | Cell penetrating peptide of TAT |
| TAT-PEG-MPLs | TAT-conjugated PEGlated Magnetic polymeric liposome |
| TAT-U1A dsRBD | TAT conjugated with U1A dsRBD |
| TP 10 | Transportan10 |
| U1A | U1 small nuclear ribonucleoprotein A |
| V | Valine |
References
- Eguchi, A.; Akuta, T.; Okuyama, H.; Senda, T.; Yokoi, H.; Inokuchi, H.; Fujita, S.; Hayakawa, T.; Takeda, K.; Hasegawa, M.; et al. Protein transduction domain of HIV-1 Tat protein promotes efficient delivery of DNA into mammalian cells. J. Biol. Chem. 2001, 276, 26204–26210. [Google Scholar] [CrossRef] [PubMed]
- Lindgren, M.; Hallbrink, M.; Prochiantz, A.; Langel, U. Cell-penetrating peptides. Trends Pharmacol. Sci. 2000, 21, 99–103. [Google Scholar] [CrossRef]
- Derossi, D.; Chassaing, G.; Prochiantz, A. Trojan peptides: The penetratin system for intracellular delivery. Trends Cell Biol. 1998, 8, 84–87. [Google Scholar] [CrossRef]
- Futaki, S. Arginine-rich peptides: Potential for intracellular delivery of macromolecules and the mystery of the translocation mechanisms. Int. J. Pharm. 2002, 245, 1–7. [Google Scholar] [CrossRef]
- De Coupade, C.; Fittipaldi, A.; Chagnas, V.; Michel, M.; Carlier, S.; Tasciotti, E.; Darmon, A.; Ravel, D.; Kearsey, J.; Giacca, M.; et al. Novel human-derived cell-penetrating peptides for specific subcellular delivery of therapeutic biomolecules. Biochem. J. 2005, 390, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Gump, J.M.; Dowdy, S.F. TAT transduction: The molecular mechanism and therapeutic prospects. Trends Mol. Med. 2007, 13, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Ter-Avetisyan, G.; Tunnemann, G.; Nowak, D.; Nitschke, M.; Herrmann, A.; Drab, M.; Cardoso, M.C. Cell entry of arginine-rich peptides is independent of endocytosis. J. Biol. Chem. 2009, 284, 3370–3378. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Lai, G.H.; Schmidt, N.W.; Sun, V.Z.; Rodriguez, A.R.; Tong, R.; Tang, L.; Cheng, J.; Deming, T.J.; Kamei, D.T.; et al. Translocation of HIV TAT peptide and analogues induced by multiplexed membrane and cytoskeletal interactions. Proc. Natl. Acad. Sci. USA 2011, 108, 16883–16888. [Google Scholar] [CrossRef] [PubMed]
- Li, G.H.; Li, W.; Mumper, R.J.; Nath, A. Molecular mechanisms in the dramatic enhancement of HIV-1 Tat transduction by cationic liposomes. FASEB J. 2012, 26, 2824–2834. [Google Scholar] [CrossRef] [PubMed]
- Brasseur, R.; Divita, G. Happy birthday cell penetrating peptides: Already 20 years. Biochim. Biophys. Acta 2010, 1798, 2177–2181. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.C.; Deshayes, S.; Heitz, F.; Divita, G. Cell-penetrating peptides: From molecular mechanisms to therapeutics. Biol. Cell 2008, 100, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Heitz, F.; Morris, M.C.; Divita, G. Twenty years of cell-penetrating peptides: From molecular mechanisms to therapeutics. Br. J. Pharmacol. 2009, 157, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Frankel, A.D.; Pabo, C.O. Cellular uptake of the tat protein from human immunodeficiency virus. Cell 1988, 55, 1189–1193. [Google Scholar] [CrossRef]
- Gautam, A.; Singh, H.; Tyagi, A.; Chaudhary, K.; Kumar, R.; Kapoor, P.; Raghava, G.P. CPPsite: A curated database of cell penetrating peptides. Database 2012, 2012, bas015. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.P.; Melikov, K.; Vives, E.; Ramos, C.; Verbeure, B.; Gait, M.J.; Chernomordik, L.V.; Lebleu, B. Cell-penetrating peptides. A reevaluation of the mechanism of cellular uptake. J. Biol. Chem. 2003, 278, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Fotin-Mleczek, M.; Fischer, R.; Brock, R. Endocytosis and cationic cell-penetrating peptides—A merger of concepts and methods. Curr. Pharm. Des. 2005, 11, 3613–3628. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.T.; Sayers, E.J. Cell entry of cell penetrating peptides: Tales of tails wagging dogs. J. Control. Release 2012, 161, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Lorents, A.; Kodavali, P.K.; Oskolkov, N.; Langel, U.; Hallbrink, M.; Pooga, M. Cell-penetrating peptides split into two groups based on modulation of intracellular calcium concentration. J. Biol. Chem. 2012, 287, 16880–16889. [Google Scholar] [CrossRef] [PubMed]
- Furuhata, M.; Kawakami, H.; Toma, K.; Hattori, Y.; Maitani, Y. Intracellular delivery of proteins in complexes with oligoarginine-modified liposomes and the effect of oligoarginine length. Bioconj. Chem. 2006, 17, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Takayama, K.; Tadokoro, A.; Pujals, S.; Nakase, I.; Giralt, E.; Futaki, S. Novel system to achieve one-pot modification of cargo molecules with oligoarginine vectors for intracellular delivery. Bioconj. Chem. 2009, 20, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, N.; Burster, T.; Sommandas, V.; Herrmann, T.; Boehm, B.O.; Driessen, C.; Voelter, W.; Kalbacher, H. A novel cell penetrating aspartic protease inhibitor blocks processing and presentation of tetanus toxoid more efficiently than pepstatin A. Biochem. Biophys. Res. Commun. 2007, 364, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Ono, S.; Ichikawa, T.; Arimitsu, S.; Onoda, K.; Tokunaga, K.; Sugiu, K.; Tomizawa, K.; Matsui, H.; Date, I. Novel protein transduction method by using 11R: An effective new drug delivery system for the treatment of cerebrovascular diseases. Stroke 2007, 38, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Foged, C.; Franzyk, H.; Bahrami, S.; Frokjaer, S.; Jaroszewski, J.W.; Nielsen, H.M.; Olsen, C.A. Cellular uptake and membrane-destabilising properties of alpha-peptide/beta-peptoid chimeras: Lessons for the design of new cell-penetrating peptides. Biochim. Biophys. Acta 2008, 1778, 2487–2495. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Yan, Y.; Wang, X.T.; Liu, X.W.; Peng, D.J.; Wang, M.; Tian, J.; Zong, Y.Q.; Zhang, Y.H.; Noteborn, M.H.; et al. PTD4-apoptin protein therapy inhibits tumor growth in vivo. Int. J. Cancer 2009, 124, 2973–2981. [Google Scholar] [CrossRef] [PubMed]
- Mai, J.C.; Mi, Z.; Kim, S.H.; Ng, B.; Robbins, P.D. A proapoptotic peptide for the treatment of solid tumors. Cancer Res. 2001, 61, 7709–7712. [Google Scholar] [PubMed]
- Arrighi, R.B.; Ebikeme, C.; Jiang, Y.; Ranford-Cartwright, L.; Barrett, M.P.; Langel, U.; Faye, I. Cell-penetrating peptide TP10 shows broad-spectrum activity against both Plasmodium falciparum and Trypanosoma brucei brucei. Antimicrob. Agents Chemother. 2008, 52, 3414–3417. [Google Scholar] [CrossRef] [PubMed]
- El-Andaloussi, S.; Johansson, H.J.; Holm, T.; Langel, U. A novel cell-penetrating peptide, M918, for efficient delivery of proteins and peptide nucleic acids. Mol. Ther. 2007, 15, 1820–1826. [Google Scholar] [CrossRef] [PubMed]
- Aussedat, B.; Sagan, S.; Chassaing, G.; Bolbach, G.; Burlina, F. Quantification of the efficiency of cargo delivery by peptidic and pseudo-peptidic Trojan carriers using MALDI-TOF mass spectrometry. Biochim. Biophys. Acta 2006, 1758, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.M.; Ahn, M.H.; Chae, W.J.; Jung, Y.G.; Park, J.C.; Song, H.M.; Kim, Y.E.; Shin, J.A.; Park, C.S.; Park, J.W.; et al. Intranasal delivery of the cytoplasmic domain of CTLA-4 using a novel protein transduction domain prevents allergic inflammation. Nat. Med. 2006, 12, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Kameyama, S.; Horie, M.; Kikuchi, T.; Omura, T.; Takeuchi, T.; Nakase, I.; Sugiura, Y.; Futaki, S. Effects of cell-permeating peptide binding on the distribution of 125I-labeled Fab fragment in rats. Bioconj. Chem. 2006, 17, 597–602. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.N.; Cashman, S.M.; Read, S.P.; Kumar-Singh, R. Cell penetrating peptide POD mediates delivery of recombinant proteins to retina, cornea and skin. Vis. Res. 2010, 50, 686–697. [Google Scholar] [CrossRef] [PubMed]
- Mano, M.; Teodosio, C.; Paiva, A.; Simoes, S.; Pedroso de Lima, M.C. On the mechanisms of the internalization of S4(13)-PV cell-penetrating peptide. Biochem. J. 2005, 390, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Sheng, J.; Oyler, G.; Zhou, B.; Janda, K.; Shoemaker, C.B. Identification and characterization of a novel cell-penetrating peptide. Biochem. Biophys. Res. Commun. 2009, 382, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Elmquist, A.; Hansen, M.; Langel, U. Structure-activity relationship study of the cell-penetrating peptide pVEC. Biochim. Biophys. Acta 2006, 1758, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Li, M.; Zhang, Z.; Gong, T.; Sun, X. Cell-penetrating peptides as delivery enhancers for vaccine. Curr. Pharm. Biotechnol. 2014, 15, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Funhoff, A.M.; van Nostrum, C.F.; Lok, M.C.; Fretz, M.M.; Crommelin, D.J.; Hennink, W.E. Poly(3-guanidinopropyl methacrylate): A novel cationic polymer for gene delivery. Bioconj. Chem. 2004, 15, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Tsui, T. Six-cell penetrating peptide-based fusion proteins for siRNA delivery. Drug Deliv. 2015, 22, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Jo, J.; Hong, S.; Choi, W.Y.; Lee, D.R. Cell-penetrating peptide (CPP)-conjugated proteins is an efficient tool for manipulation of human mesenchymal stromal cells. Sci. Rep. 2014, 4, 4378. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.R.; Liou, J.S.; Huang, Y.W.; Aronstam, R.S.; Lee, H.J. Intracellular delivery of nanoparticles and DNAs by IR9 cell-penetrating peptides. PLoS ONE 2013, 8, e64205. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, A.M.; Trabulo, S.; Cardoso, A.L.; Maia, S.; Gomes, P.; Jurado, A.S.; Pedroso de Lima, M.C. Comparison of the efficiency of complexes based on S4(13)-PV cell-penetrating peptides in plasmid DNA and siRNA delivery. Mol. Pharm. 2013, 10, 2653–2666. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zheng, X.; Koren, V.; Vashist, Y.K.; Tsui, T.Y. Highly efficient delivery of siRNA to a heart transplant model by a novel cell penetrating peptide-dsRNA binding domain. Int. J. Pharm. 2014, 469, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Farkhani, S.M.; Valizadeh, A.; Karami, H.; Mohammadi, S.; Sohrabi, N.; Badrzadeh, F. Cell penetrating peptides: Efficient vectors for delivery of nanoparticles, nanocarriers, therapeutic and diagnostic molecules. Peptides 2014, 57, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Modgil, A.; Zhong, T.; Sun, C.; Singh, J. Influence of short-chain cell-penetrating peptides on transport of doxorubicin encapsulating receptor-targeted liposomes across brain endothelial barrier. Pharm. Res. 2014, 31, 1194–1209. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Modgil, A.; Layek, B.; Arora, K.; Sun, C.; Law, B.; Singh, J. Cell penetrating peptide tethered bi-ligand liposomes for delivery to brain in vivo: Biodistribution and transfection. J. Control. Release: Off. J. Control. Release Soc. 2013, 167, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, W.; Pong, R.C.; Hao, G.; Sun, X.; Hsieh, J.T. Analysis of oligo-arginine cell-permeable peptides uptake by prostate cells. Amino Acids 2012, 42, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Patel, L.N.; Wang, J.; Kim, K.J.; Borok, Z.; Crandall, E.D.; Shen, W.C. Conjugation with cationic cell-penetrating peptide increases pulmonary absorption of insulin. Mol. Pharm. 2009, 6, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Endoh, T.; Ohtsuki, T. Cellular siRNA delivery using cell-penetrating peptides modified for endosomal escape. Adv. Drug Deliv. Rev. 2009, 61, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Meade, B.R.; Dowdy, S.F. Enhancing the cellular uptake of siRNA duplexes following noncovalent packaging with protein transduction domain peptides. Adv. Drug Deliv. Rev. 2008, 60, 530–536. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.J.; Jones, S.; Fabani, M.M.; Ivanova, G.; Arzumanov, A.A.; Gait, M.J. RNA targeting with peptide conjugates of oligonucleotides, siRNA and PNA. Blood Cells Mol. Dis. 2007, 38, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P. Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 2005, 4, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Tang, J.; Fu, L.; Ran, R.; Liu, Y.; Yuan, M.; He, Q. A pH-responsive alpha-helical cell penetrating peptide-mediated liposomal delivery system. Biomaterials 2013, 34, 7980–7993. [Google Scholar] [CrossRef] [PubMed]
- Marty, C.; Meylan, C.; Schott, H.; Ballmer-Hofer, K.; Schwendener, R.A. Enhanced heparan sulfate proteoglycan-mediated uptake of cell-penetrating peptide-modified liposomes. Cell. Mol. Life Sci. 2004, 61, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Fretz, M.M.; Koning, G.A.; Mastrobattista, E.; Jiskoot, W.; Storm, G. OVCAR-3 cells internalize TAT-peptide modified liposomes by endocytosis. Biochim. Biophys. Acta 2004, 1665, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Cryan, S.A.; Devocelle, M.; Moran, P.J.; Hickey, A.J.; Kelly, J.G. Increased intracellular targeting to airway cells using octaarginine-coated liposomes: In vitro assessment of their suitability for inhalation. Mol. Pharm. 2006, 3, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Tang, N.; Liu, X.; Liang, W.; Xu, W.; Torchilin, V.P. siRNA-containing liposomes modified with polyarginine effectively silence the targeted gene. J. Control. Release 2006, 112, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Moriguchi, R.; Kogure, K.; Shastri, N.; Harashima, H. Efficient MHC class I presentation by controlled intracellular trafficking of antigens in octaarginine-modified liposomes. Mol. Ther. 2008, 16, 1507–1514. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P.; Rammohan, R.; Weissig, V.; Levchenko, T.S. TAT peptide on the surface of liposomes affords their efficient intracellular delivery even at low temperature and in the presence of metabolic inhibitors. Proc. Natl. Acad. Sci. USA 2001, 98, 8786–8791. [Google Scholar] [CrossRef] [PubMed]
- Saw, P.E.; Ko, Y.T.; Jon, S. Efficient Liposomal Nanocarrier-mediated Oligodeoxynucleotide Delivery Involving Dual Use of a Cell-Penetrating Peptide as a Packaging and Intracellular Delivery Agent. Macromol. Rapid Commun. 2010, 31, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Koren, E.; Apte, A.; Jani, A.; Torchilin, V.P. Multifunctional PEGylated 2C5-immunoliposomes containing pH-sensitive bonds and TAT peptide for enhanced tumor cell internalization and cytotoxicity. J. Control. Release 2012, 160, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Chen, H.; Yuan, W.; Kuai, R.; Zhang, Q.; Xie, F.; Zhang, L.; Zhang, Z.; Liu, J.; He, Q. Liposome formulated with TAT-modified cholesterol for enhancing the brain delivery. Int. J. Pharm. 2011, 419, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Chen, H.; Zhang, Q.; Wang, X.; Yuan, W.; Kuai, R.; Tang, J.; Zhang, L.; Zhang, Z.; Zhang, Q.; et al. Liposome formulated with TAT-modified cholesterol for improving brain delivery and therapeutic efficacy on brain glioma in animals. Int. J. Pharm. 2011, 420, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, S.; Liao, Z.; Wang, C.; Liu, Y.; Feng, S.; Jiang, X.; Chang, J. PEGlated magnetic polymeric liposome anchored with TAT for delivery of drugs across the blood-spinal cord barrier. Biomaterials 2010, 31, 6589–6596. [Google Scholar] [CrossRef] [PubMed]
- Crombez, L.; Divita, G. A non-covalent peptide-based strategy for siRNA delivery. Methods Mol. Biol. 2011, 683, 349–360. [Google Scholar] [PubMed]
- Walker, L.; Perkins, E.; Kratz, F.; Raucher, D. Cell penetrating peptides fused to a thermally targeted biopolymer drug carrier improve the delivery and antitumor efficacy of an acid-sensitive doxorubicin derivative. Int. J. Pharm. 2012, 436, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Gao, X.; Gu, G.; Liu, Z.; Hu, Q.; Tu, Y.; Song, Q.; Yao, L.; Pang, Z.; Jiang, X.; et al. Penetratin-functionalized PEG-PLA nanoparticles for brain drug delivery. Int. J. Pharm. 2012, 436, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Taki, H.; Tanaka, K.; Takashima, Y.; Okada, H. Cell-penetrating peptide-modified block copolymer micelles promote direct brain delivery via intranasal administration. Pharm. Res. 2011, 28, 2130–2139. [Google Scholar] [CrossRef] [PubMed]
- Nam, H.Y.; Kim, J.; Kim, S.; Yockman, J.W.; Kim, S.W.; Bull, D.A. Cell penetrating peptide conjugated bioreducible polymer for siRNA delivery. Biomaterials 2011, 32, 5213–5222. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, S.; Suita, M.; Yamamoto, T.; Masaoka, Y.; Kataoka, M.; Yamashita, S.; Nakajima, N.; Shinkai, N.; Yamauchi, H.; Hiwatari, K.; et al. Performance of cell-penetrating peptide-linked polymers physically mixed with poorly membrane-permeable molecules on cell membranes. Eur. J. Pharm. Biopharm. 2012, 81, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Hashida, H.; Miyamoto, M.; Cho, Y.; Hida, Y.; Kato, K.; Kurokawa, T.; Okushiba, S.; Kondo, S.; Dosaka-Akita, H.; Katoh, H. Fusion of HIV-1 Tat protein transduction domain to poly-lysine as a new DNA delivery tool. Br. J. Cancer 2004, 90, 1252–1258. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, S.; Suita, M.; Masaoka, Y.; Kataoka, M.; Nakajima, N.; Shinkai, N.; Yamauchi, H.; Hiwatari, K.; Tachikawa, H.; Kimura, R.; et al. Oligoarginine-linked polymers as a new class of penetration enhancers. J. Control. Release 2010, 148, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Sugawara, K.; Tanaka, K.; Horiuchi, S.; Takashima, Y.; Okada, H. Suppression of tumor growth by systemic delivery of anti-VEGF siRNA with cell-penetrating peptide-modified MPEG-PCL nanomicelles. Eur. J. Pharm. Biopharm. 2012, 81, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Bae, K.H.; Kim, J.S.; Nam, Y.S.; Park, T.G. Intracellular delivery of paclitaxel using oil-free, shell cross-linked HSA--multi-armed PEG nanocapsules. Biomaterials 2011, 32, 8635–8644. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, R.; Shen, Y.; Pollack, K.A.; Taylor, J.S.; Wooley, K.L. Dual peptide nucleic acid- and peptide-functionalized shell cross-linked nanoparticles designed to target mRNA toward the diagnosis and treatment of acute lung injury. Bioconj. Chem. 2012, 23, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Kanazawa, T.; Shibata, Y.; Suda, Y.; Fukuda, T.; Takashima, Y.; Okada, H. Development of cell-penetrating peptide-modified MPEG-PCL diblock copolymeric nanoparticles for systemic gene delivery. Int. J. Pharm. 2010, 396, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, C.; Schillinger, U.; Ortiz, A.; Tabatt, K.; Plank, C.; Muller, R.H.; Rosenecker, J. Application of novel solid lipid nanoparticle (SLN)-gene vector formulations based on a dimeric HIV-1 TAT-peptide in vitro and in vivo. Pharm. Res. 2004, 21, 1662–1669. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, T.; Goto, M.; Kodera, K.; Kanazawa, H.; Murakami, Y.; Mizushima, Y.; Higaki, M. Intracellular delivery of siRNA by cell-penetrating peptides modified with cationic oligopeptides. Drug Deliv. 2009, 16, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Mo, R.H.; Zaro, J.L.; Shen, W.C. Comparison of cationic and amphipathic cell penetrating peptides for siRNA delivery and efficacy. Mol. Pharm. 2012, 9, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Palm-Apergi, C.; Eguchi, A.; Dowdy, S.F. PTD-DRBD siRNA delivery. Methods Mol. Biol. 2011, 683, 339–347. [Google Scholar] [PubMed]
- Eguchi, A.; Meade, B.R.; Chang, Y.C.; Fredrickson, C.T.; Willert, K.; Puri, N.; Dowdy, S.F. Efficient siRNA delivery into primary cells by a peptide transduction domain-dsRNA binding domain fusion protein. Nat. Biotechnol. 2009, 27, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Endoh, T.; Sisido, M.; Ohtsuki, T. Cellular siRNA delivery mediated by a cell-permeant RNA-binding protein and photoinduced RNA interference. Bioconj. Chem. 2008, 19, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Cheetham, A.G.; Lin, Y.A.; Cui, H. Self-assembled Tat nanofibers as effective drug carrier and transporter. ACS Nano 2013, 7, 5965–5977. [Google Scholar] [CrossRef] [PubMed]
- Asai, T.; Tsuzuku, T.; Takahashi, S.; Okamoto, A.; Dewa, T.; Nango, M.; Hyodo, K.; Ishihara, H.; Kikuchi, H.; Oku, N. Cell-penetrating peptide-conjugated lipid nanoparticles for siRNA delivery. Biochem. Biophys. Res. Commun. 2014, 444, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Akiyama, F.; Kakizaki, S.; Takashima, Y.; Seta, Y. Delivery of siRNA to the brain using a combination of nose-to-brain delivery and cell-penetrating peptide-modified nano-micelles. Biomaterials 2013, 34, 9220–9226. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Guo, K.; Lu, J.; Venkatraman, S.S.; Luo, D.; Ng, K.C.; Ling, E.A.; Moochhala, S.; Yang, Y.Y. Biologically active core/shell nanoparticles self-assembled from cholesterol-terminated PEG-TAT for drug delivery across the blood-brain barrier. Biomaterials 2008, 29, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.M.; Eroglu, E.; Bawage, S.S.; Vig, K.; Miller, M.E.; Pillai, S.; Dennis, V.A.; Singh, S.R. Enhanced intracellular translocation and biodistribution of gold nanoparticles functionalized with a cell-penetrating peptide (VG-21) from vesicular stomatitis virus. Biomaterials 2014, 35, 9484–9494. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Gao, X.; Gu, G.; Liu, Z.; Zeng, N.; Hu, Q.; Song, Q.; Yao, L.; Pang, Z.; Jiang, X.; et al. Low molecular weight protamine-functionalized nanoparticles for drug delivery to the brain after intranasal administration. Biomaterials 2011, 32, 9888–9898. [Google Scholar] [CrossRef] [PubMed]
- Susumu, K.; Oh, E.; Delehanty, J.B.; Blanco-Canosa, J.B.; Johnson, B.J.; Jain, V.; Hervey, W.J.T.; Algar, W.R.; Boeneman, K.; Dawson, P.E.; et al. Multifunctional compact zwitterionic ligands for preparing robust biocompatible semiconductor quantum dots and gold nanoparticles. J. Am. Chem. Soc. 2011, 133, 9480–9496. [Google Scholar] [CrossRef] [PubMed]
- Veiseh, O.; Kievit, F.M.; Mok, H.; Ayesh, J.; Clark, C.; Fang, C.; Leung, M.; Arami, H.; Park, J.O.; Zhang, M. Cell transcytosing poly-arginine coated magnetic nanovector for safe and effective siRNA delivery. Biomaterials 2011, 32, 5717–5725. [Google Scholar] [CrossRef] [PubMed]
- Nativo, P.; Prior, I.A.; Brust, M. Uptake and intracellular fate of surface-modified gold nanoparticles. ACS Nano 2008, 2, 1639–1644. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Kogure, K.; Futaki, S.; Harashima, H. Octaarginine-modified multifunctional envelope-type nano device for siRNA. J. Control. Release 2007, 119, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Yamauchi, J.; Khalil, I.A.; Kajimoto, K.; Akita, H.; Harashima, H. Cell penetrating peptide-mediated systemic siRNA delivery to the liver. Int. J. Pharm. 2011, 419, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Kircher, M.F.; Josephson, L.; Weissleder, R. Differential conjugation of tat peptide to superparamagnetic nanoparticles and its effect on cellular uptake. Bioconj. Chem. 2002, 13, 840–844. [Google Scholar] [CrossRef]
- Zhang, K.; Fang, H.; Chen, Z.; Taylor, J.S.; Wooley, K.L. Shape effects of nanoparticles conjugated with cell-penetrating peptides (HIV Tat PTD) on CHO cell uptake. Bioconj. Chem. 2008, 19, 1880–1887. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.Y.; Lai, L.H.; Shen, J.; Wang, Q.Q.; Xu, F.J.; Tang, G.P. Gene delivery to tumor cells by cationic polymeric nanovectors coupled to folic acid and the cell-penetrating peptide octaarginine. Biomaterials 2011, 32, 7253–7262. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.; Delehanty, J.B.; Sapsford, K.E.; Susumu, K.; Goswami, R.; Blanco-Canosa, J.B.; Dawson, P.E.; Granek, J.; Shoff, M.; Zhang, Q.; et al. Cellular uptake and fate of PEGylated gold nanoparticles is dependent on both cell-penetration peptides and particle size. ACS Nano 2011, 5, 6434–6448. [Google Scholar] [CrossRef] [PubMed]
- Benihoud, K.; Yeh, P.; Perricaudet, M. Adenovirus vectors for gene delivery. Curr. Opin. Biotechnol. 1999, 10, 440–447. [Google Scholar] [CrossRef]
- Kovesdi, I.; Brough, D.E.; Bruder, J.T.; Wickham, T.J. Adenoviral vectors for gene transfer. Curr. Opin. Biotechnol. 1997, 8, 583–589. [Google Scholar] [CrossRef]
- Wang, L.; Yao, B.; Li, Q.; Mei, K.; Xu, J.R.; Li, H.X.; Wang, Y.S.; Wen, Y.J.; Wang, X.D.; Yang, H.S.; et al. Gene therapy with recombinant adenovirus encoding endostatin encapsulated in cationic liposome in coxsackievirus and adenovirus receptor-deficient colon carcinoma murine models. Hum. Gene Ther. 2011, 22, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Kolawole, A.O.; Wiltshire, S.M.; Frondorf, K.; Excoffon, K.J. Accessibility of the coxsackievirus and adenovirus receptor and its importance in adenovirus gene transduction efficiency. J. Gen. Virol. 2012, 93 Pt 1, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Kida, S.; Eto, Y.; Maeda, M.; Yoshioka, Y.; Nakagawa, S.; Hojo, K.; Tsuda, Y.; Mayumia, T.; Kawasaki, K. Preparation of a Tat-related transporter peptide for carrying the adenovirus vector into cells. Protein Pept. Lett. 2008, 15, 219–222. [Google Scholar] [PubMed]
- Maeda, M.; Kida, S.; Hojo, K.; Eto, Y.; Gaob, J.Q.; Kurachi, S.; Sekiguchi, F.; Mizuguchi, H.; Hayakawa, T.; Mayumi, T.; et al. Design and synthesis of a peptide-PEG transporter tool for carrying adenovirus vector into cells. Bioorg. Med. Chem. Lett. 2005, 15, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Nigatu, A.S.; Vupputuri, S.; Flynn, N.; Ramsey, J.D. Effects of cell-penetrating peptides on transduction efficiency of PEGylated adenovirus. Biomed. Pharmacother. 2015, 71, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Kurachi, S.; Tashiro, K.; Sakurai, F.; Sakurai, H.; Kawabata, K.; Yayama, K.; Okamoto, H.; Nakagawa, S.; Mizuguchi, H. Fiber-modified adenovirus vectors containing the TAT peptide derived from HIV-1 in the fiber knob have efficient gene transfer activity. Gene Ther. 2007, 14, 1160–1165. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Jin, C.; Leja, J.; Majdalani, N.; Nilsson, B.; Eriksson, F.; Essand, M. Adenovirus with hexon Tat-protein transduction domain modification exhibits increased therapeutic effect in experimental neuroblastoma and neuroendocrine tumors. J. Virol. 2011, 85, 13114–13123. [Google Scholar] [CrossRef] [PubMed]
- Bottger, M.; Zaitsev, S.V.; Otto, A.; Haberland, A.; Vorob’ev, V.I. Acid nuclear extracts as mediators of gene transfer and expression. Biochim. Biophys. Acta 1998, 1395, 78–87. [Google Scholar] [CrossRef]
- Shiraishi, T.; Pankratova, S.; Nielsen, P.E. Calcium ions effectively enhance the effect of antisense peptide nucleic acids conjugated to cationic tat and oligoarginine peptides. Chem. Biol. 2005, 12, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Haberland, A.; Knaus, T.; Zaitsev, S.V.; Stahn, R.; Mistry, A.R.; Coutelle, C.; Haller, H.; Bottger, M. Calcium ions as efficient cofactor of polycation-mediated gene transfer. Biochim. Biophys. Acta 1999, 1445, 21–30. [Google Scholar] [CrossRef]
- Baoum, A.; Ovcharenko, D.; Berkland, C. Calcium condensed cell penetrating peptide complexes offer highly efficient, low toxicity gene silencing. Int. J. Pharm. 2012, 427, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Khondee, S.; Baoum, A.; Siahaan, T.J.; Berkland, C. Calcium condensed LABL-TAT complexes effectively target gene delivery to ICAM-1 expressing cells. Mol. Pharm. 2011, 8, 788–798. [Google Scholar] [CrossRef] [PubMed]
- Baoum, A.A.; Berkland, C. Calcium condensation of DNA complexed with cell-penetrating peptides offers efficient, noncytotoxic gene delivery. J. Pharm. Sci. 2011, 100, 1637–1642. [Google Scholar] [CrossRef] [PubMed]
- Khosravi-Darani, K.; Mozafari, M.R.; Rashidi, L.; Mohammadi, M. Calcium based non-viral gene delivery: An overview of methodology and applications. Acta Medica Iran 2010, 48, 133–141. [Google Scholar]
- Zaitsev, S.; Buchwalow, I.; Haberland, A.; Tkachuk, S.; Zaitseva, I.; Haller, H.; Bottger, M. Histone H1-mediated transfection: Role of calcium in the cellular uptake and intracellular fate of H1-DNA complexes. Acta Histochem. 2002, 104, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, J.; Neundorf, I. Peptide vectors for the nonviral delivery of nucleic acids. Acc. Chem. Res. 2012, 45, 1048–1056. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.R.; Lin, M.D.; Chiang, H.J.; Lee, H.J. Arginine-rich cell-penetrating peptides deliver gene into living human cells. Gene 2012, 505, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, A.; Baoum, A.; Ohta, N.; Jacquez, S.; Seo, G.M.; Berkland, C.; Tamura, M. Intratracheal administration of a nanoparticle-based therapy with the angiotensin II type 2 receptor gene attenuates lung cancer growth. Cancer Res. 2012, 72, 2057–2067. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, F.D.; Sancey, L.; Pereira, A.; Rome, C.; Oliveira, V.; Oliveira, E.B.; Nader, H.B.; Yamane, T.; Kerkis, I.; Tersariol, I.L.; et al. The natural cell-penetrating peptide crotamine targets tumor tissue in vivo and triggers a lethal calcium-dependent pathway in cultured cells. Mol. Pharm. 2012, 9, 211–221. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Tsui, T.Y.; Ma, W. Intracellular Delivery of Molecular Cargo Using Cell-Penetrating Peptides and the Combination Strategies. Int. J. Mol. Sci. 2015, 16, 19518-19536. https://doi.org/10.3390/ijms160819518
Li H, Tsui TY, Ma W. Intracellular Delivery of Molecular Cargo Using Cell-Penetrating Peptides and the Combination Strategies. International Journal of Molecular Sciences. 2015; 16(8):19518-19536. https://doi.org/10.3390/ijms160819518
Chicago/Turabian StyleLi, Hua, Tung Yu Tsui, and Wenxue Ma. 2015. "Intracellular Delivery of Molecular Cargo Using Cell-Penetrating Peptides and the Combination Strategies" International Journal of Molecular Sciences 16, no. 8: 19518-19536. https://doi.org/10.3390/ijms160819518
APA StyleLi, H., Tsui, T. Y., & Ma, W. (2015). Intracellular Delivery of Molecular Cargo Using Cell-Penetrating Peptides and the Combination Strategies. International Journal of Molecular Sciences, 16(8), 19518-19536. https://doi.org/10.3390/ijms160819518








