Opportunities for Bio-Based Solvents Created as Petrochemical and Fuel Products Transition towards Renewable Resources
Abstract
:1. The Bio-Based Economy
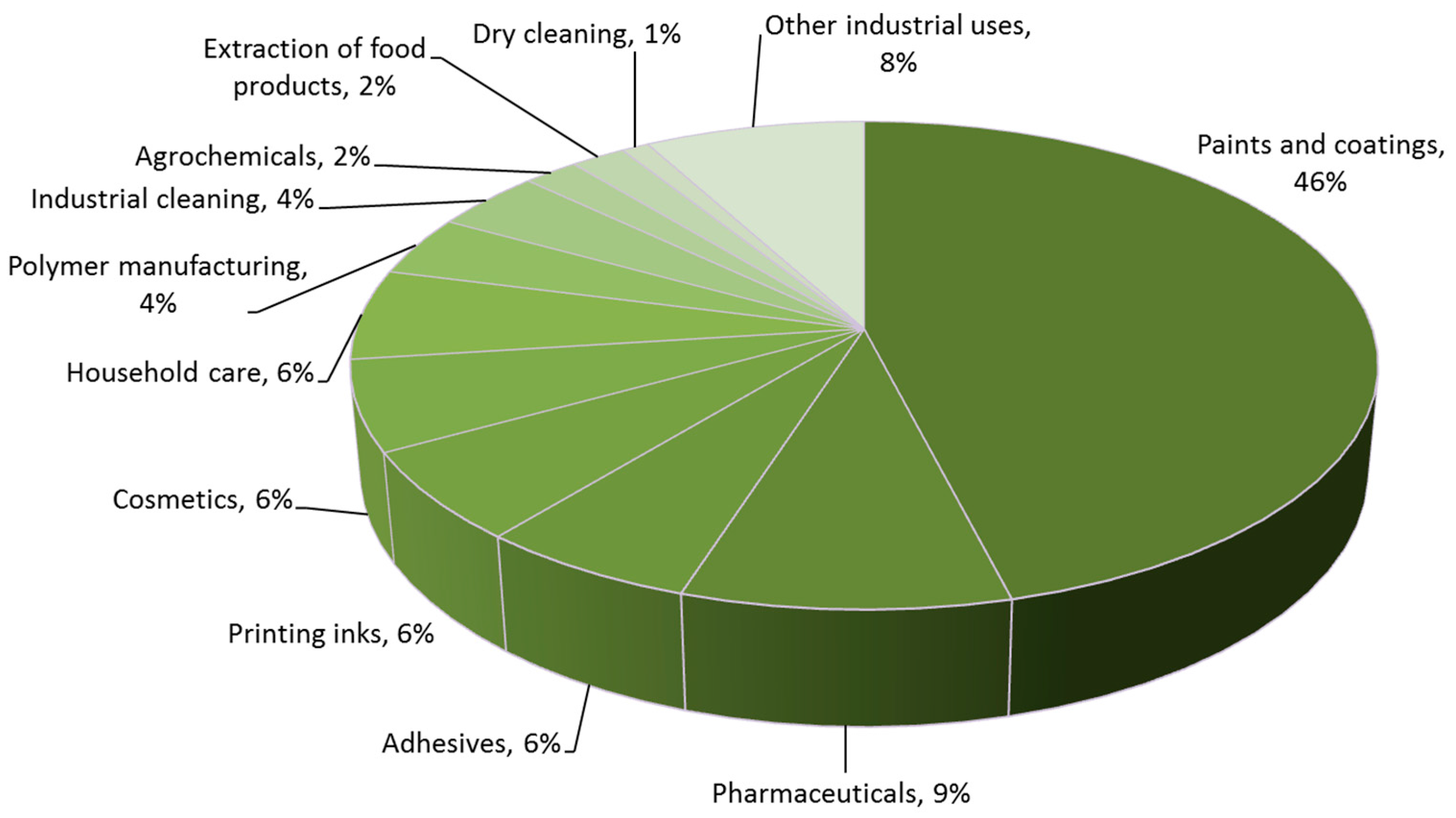
2. Contemporary Solvent Use
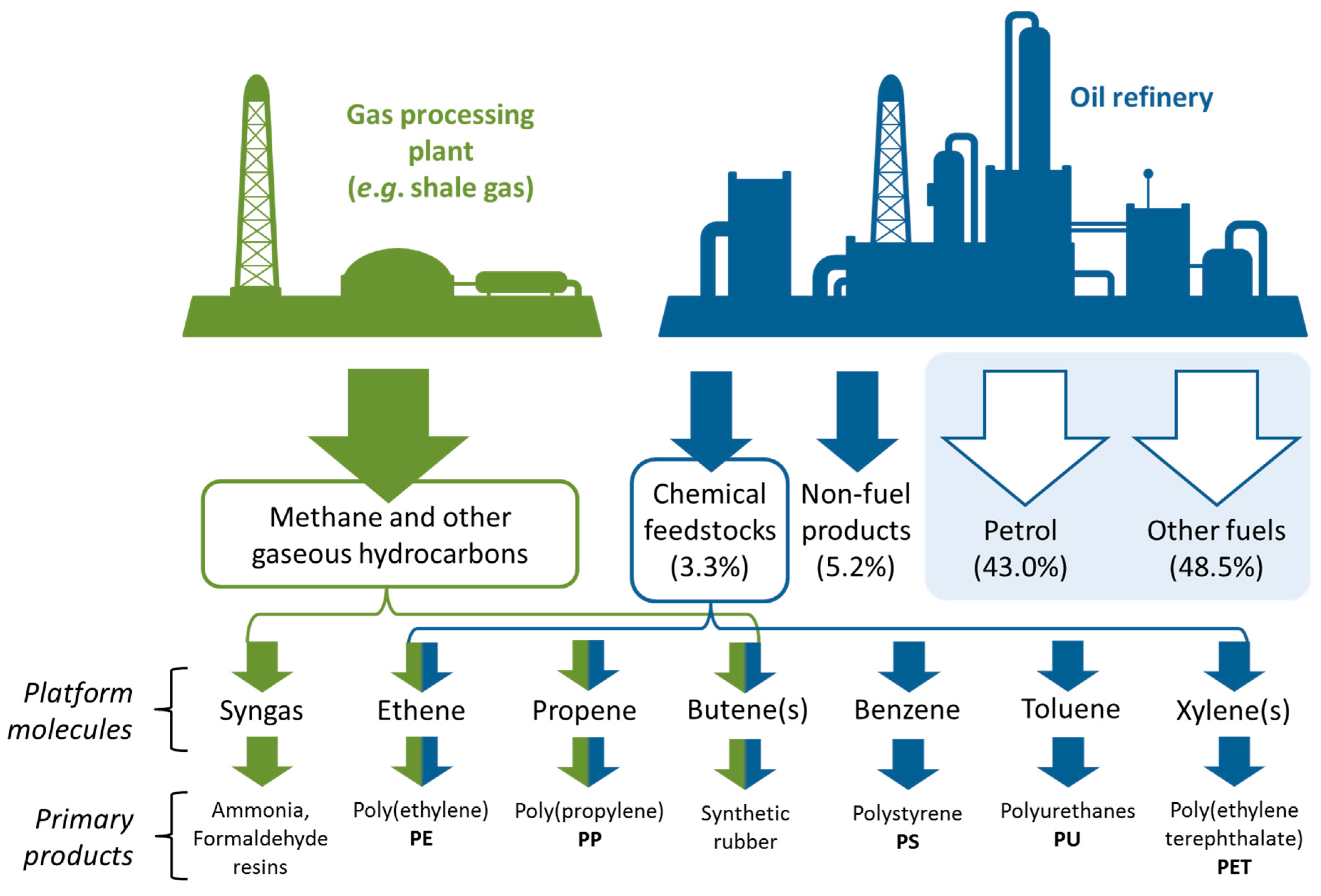
3. The Relationship between the Petrochemical Industry and Biorefineries
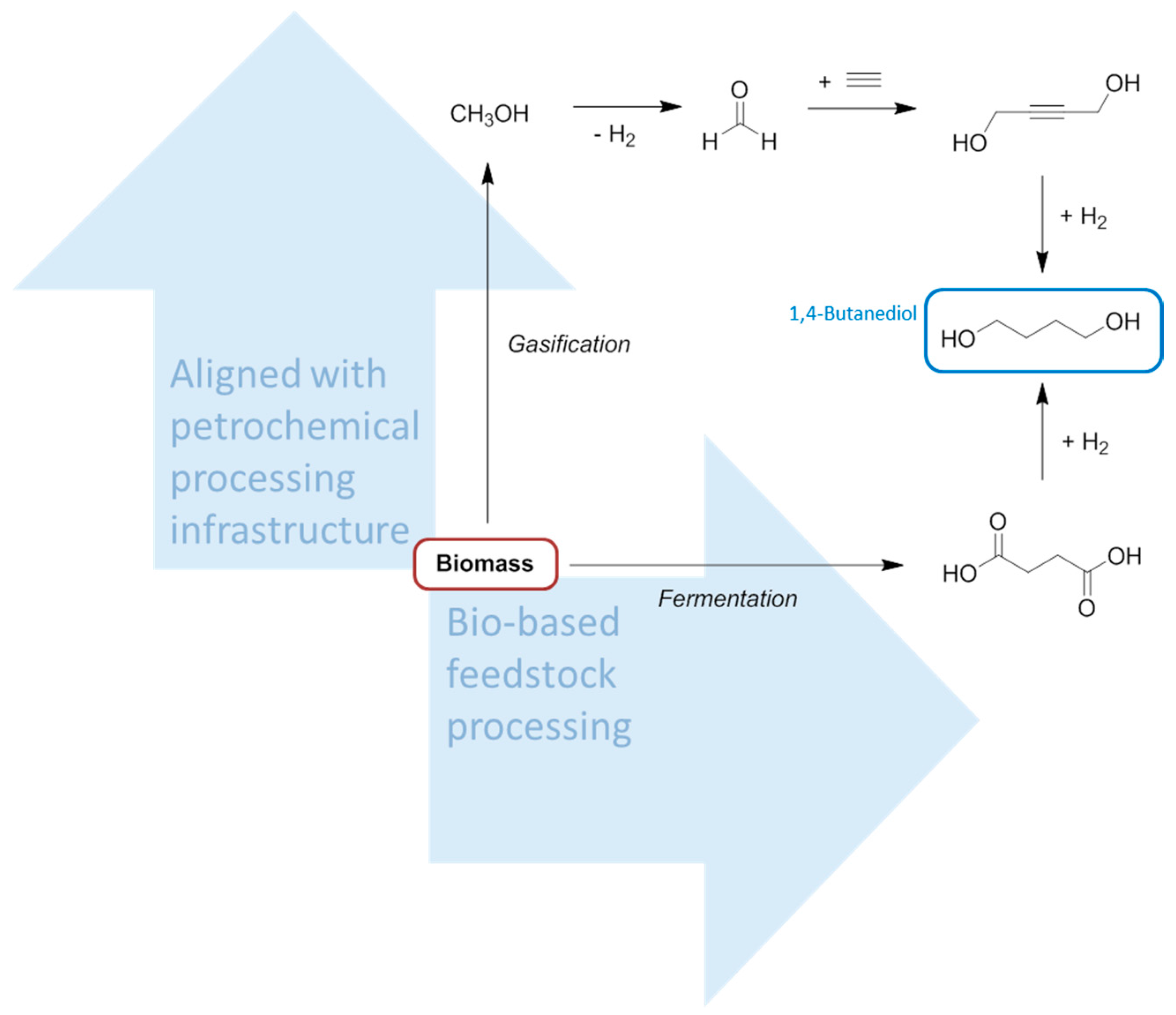
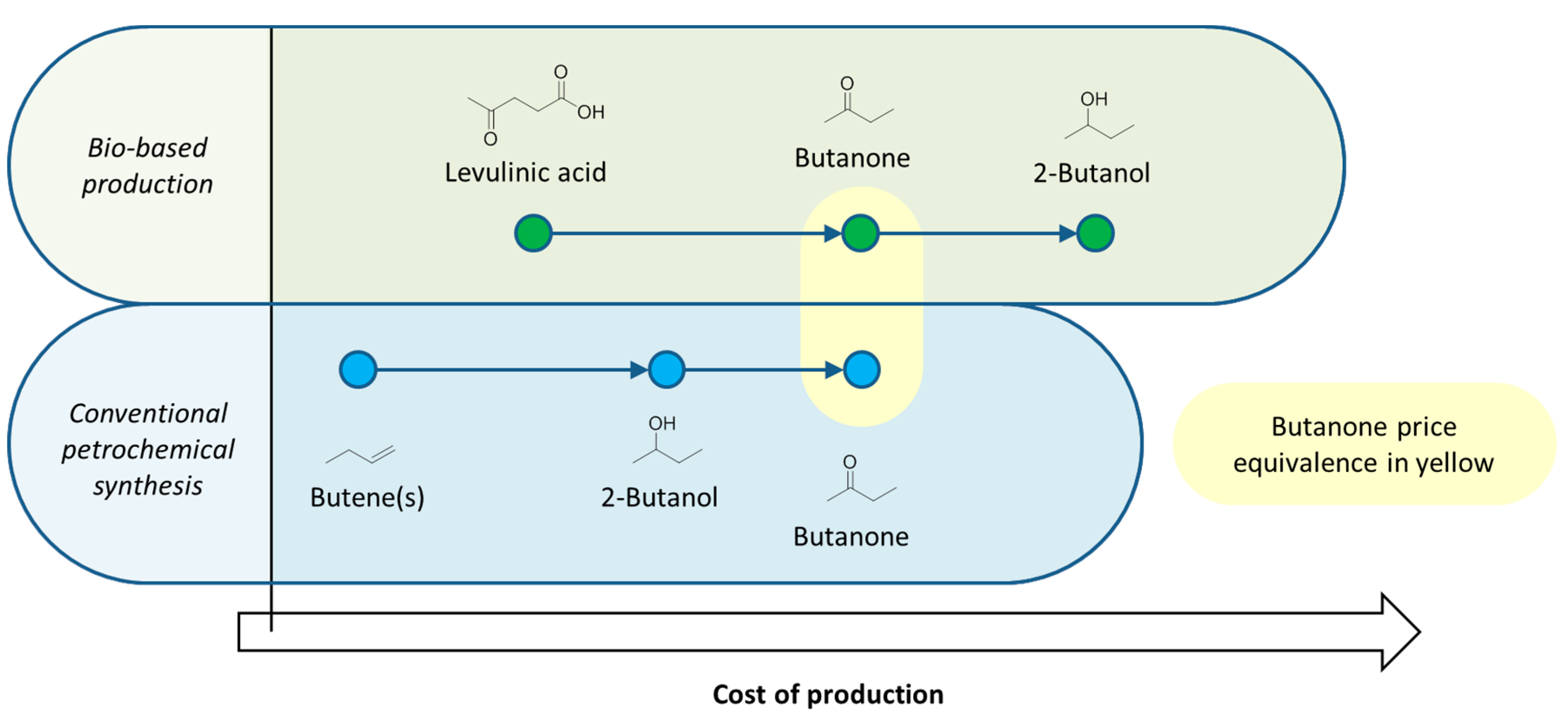
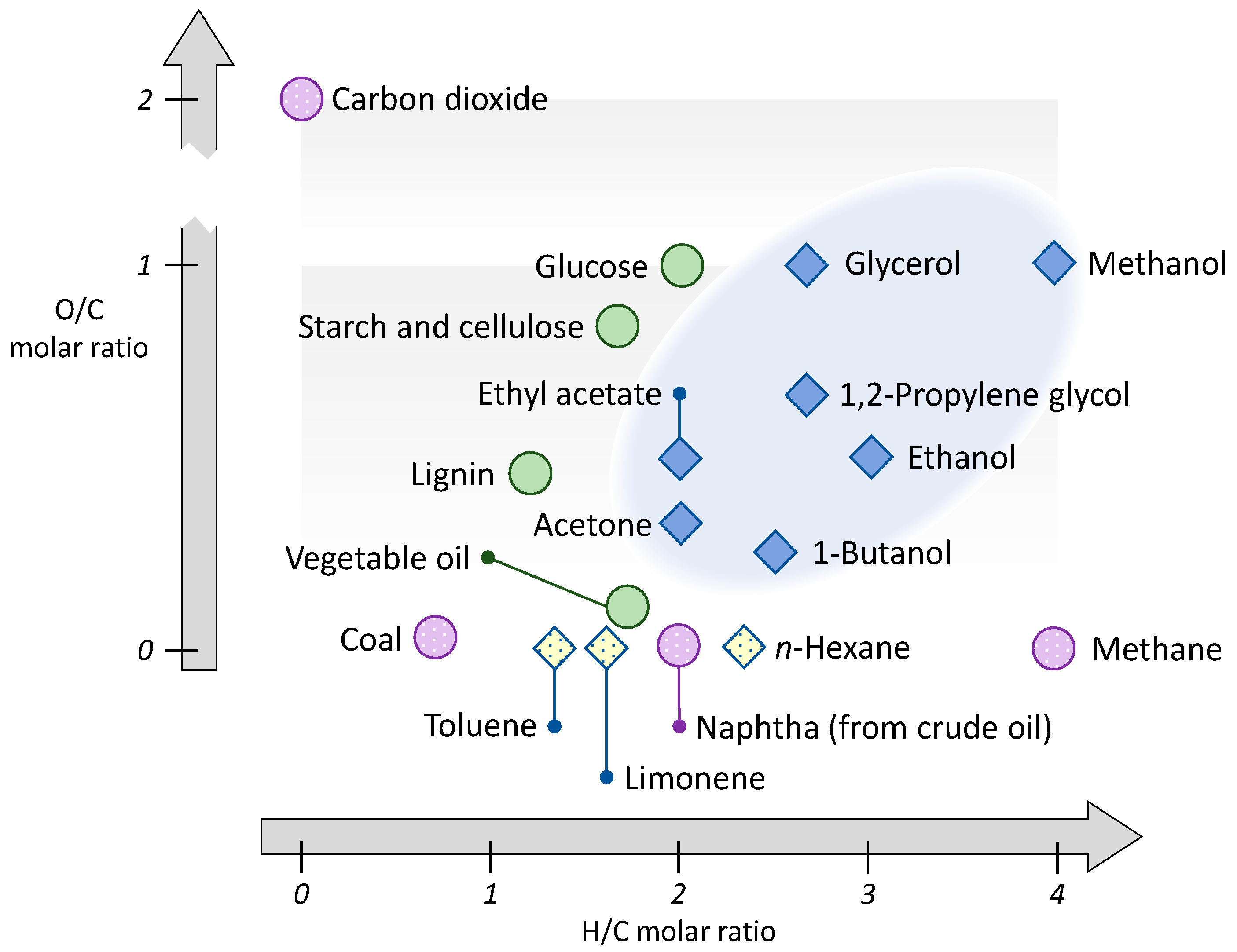
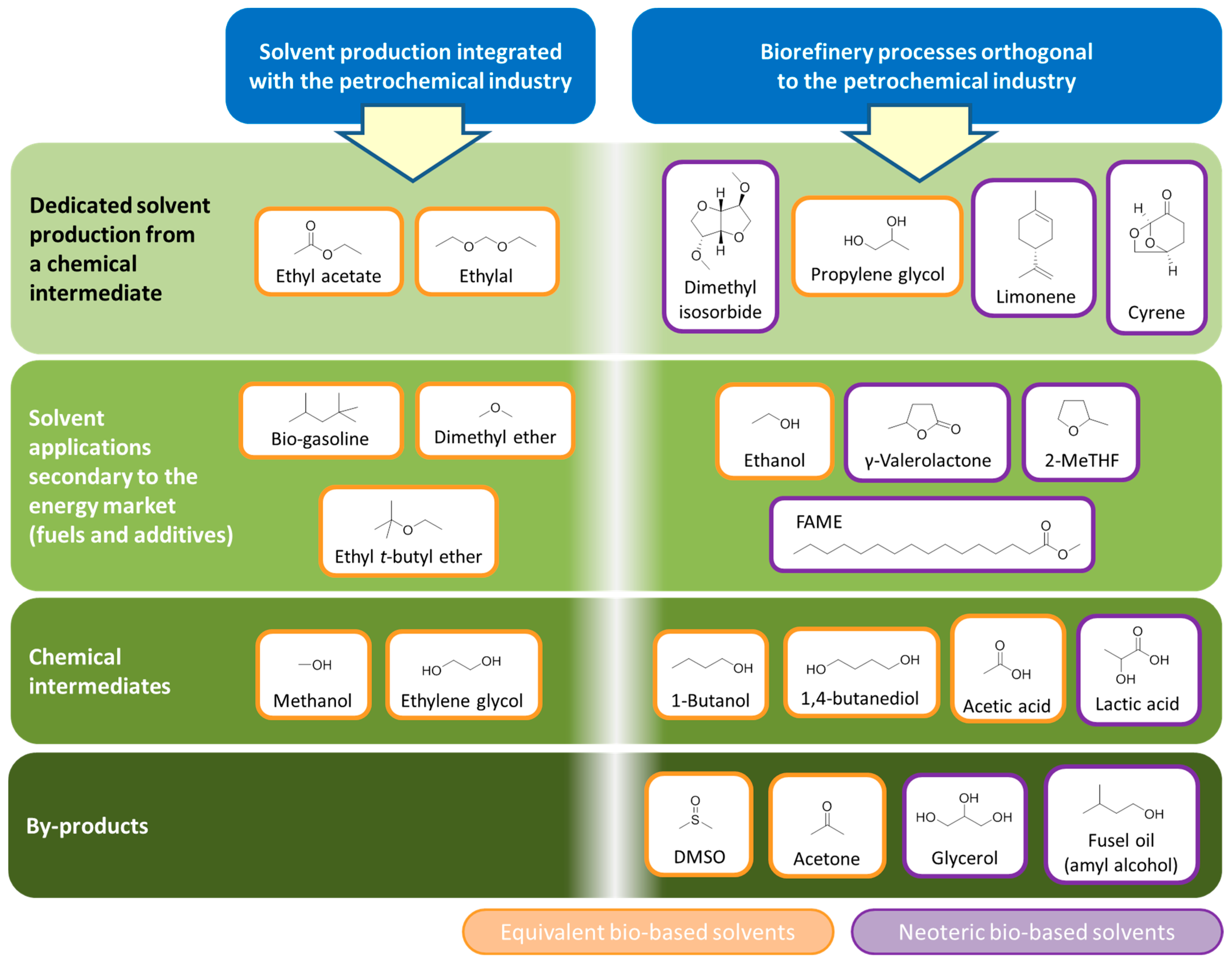
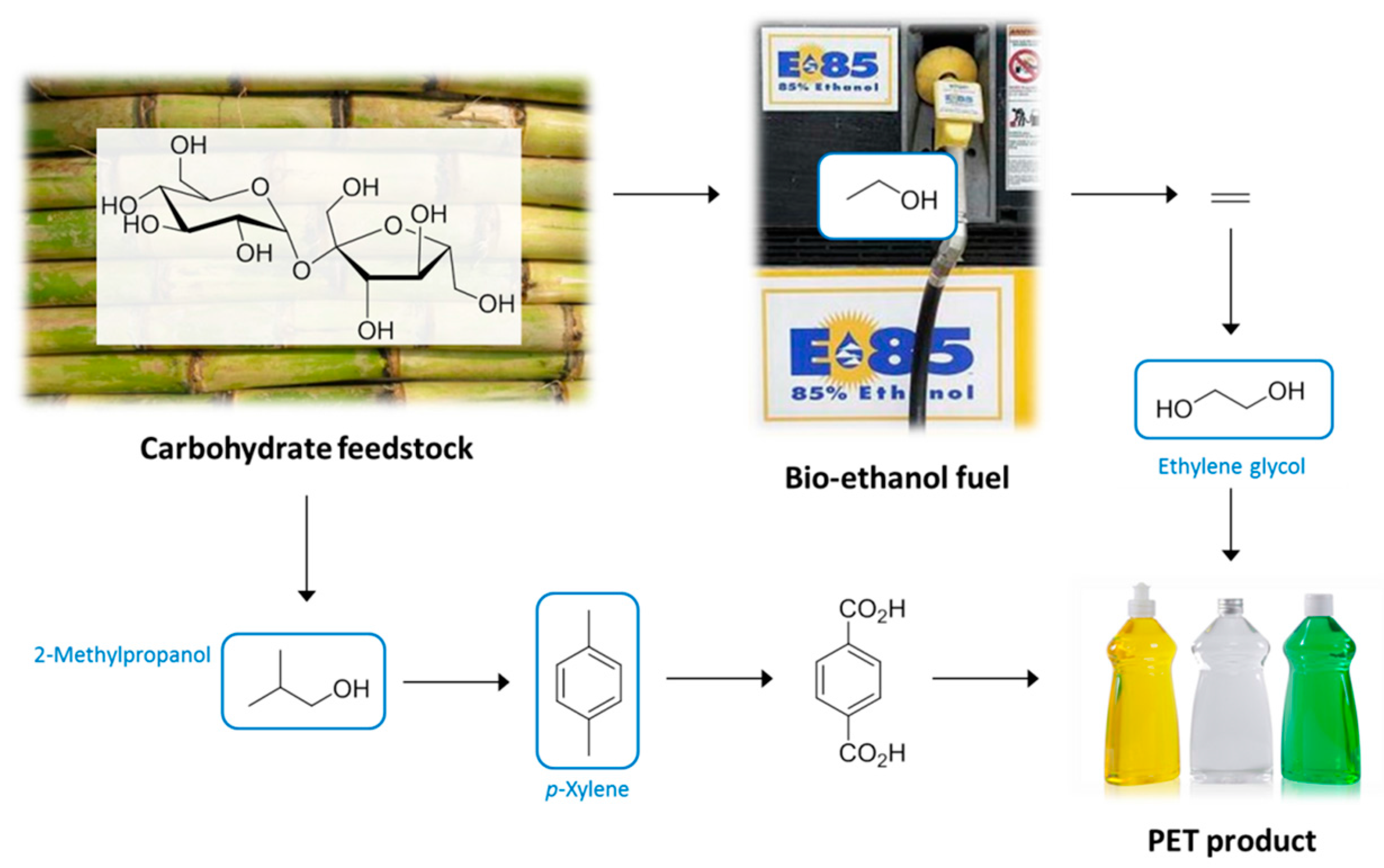
4. Production of Petrochemical Intermediates Relevant to Solvent Manufacturing
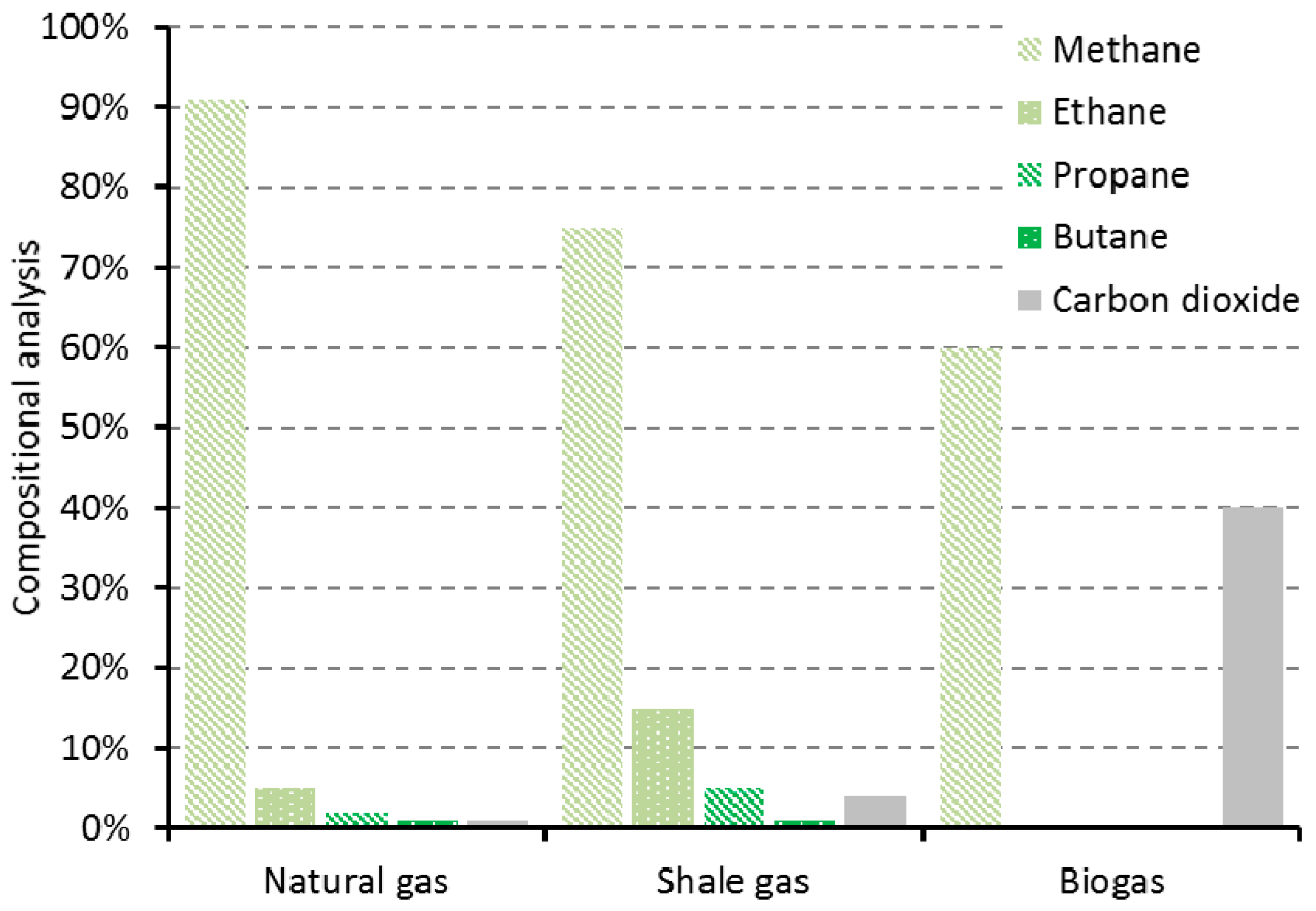
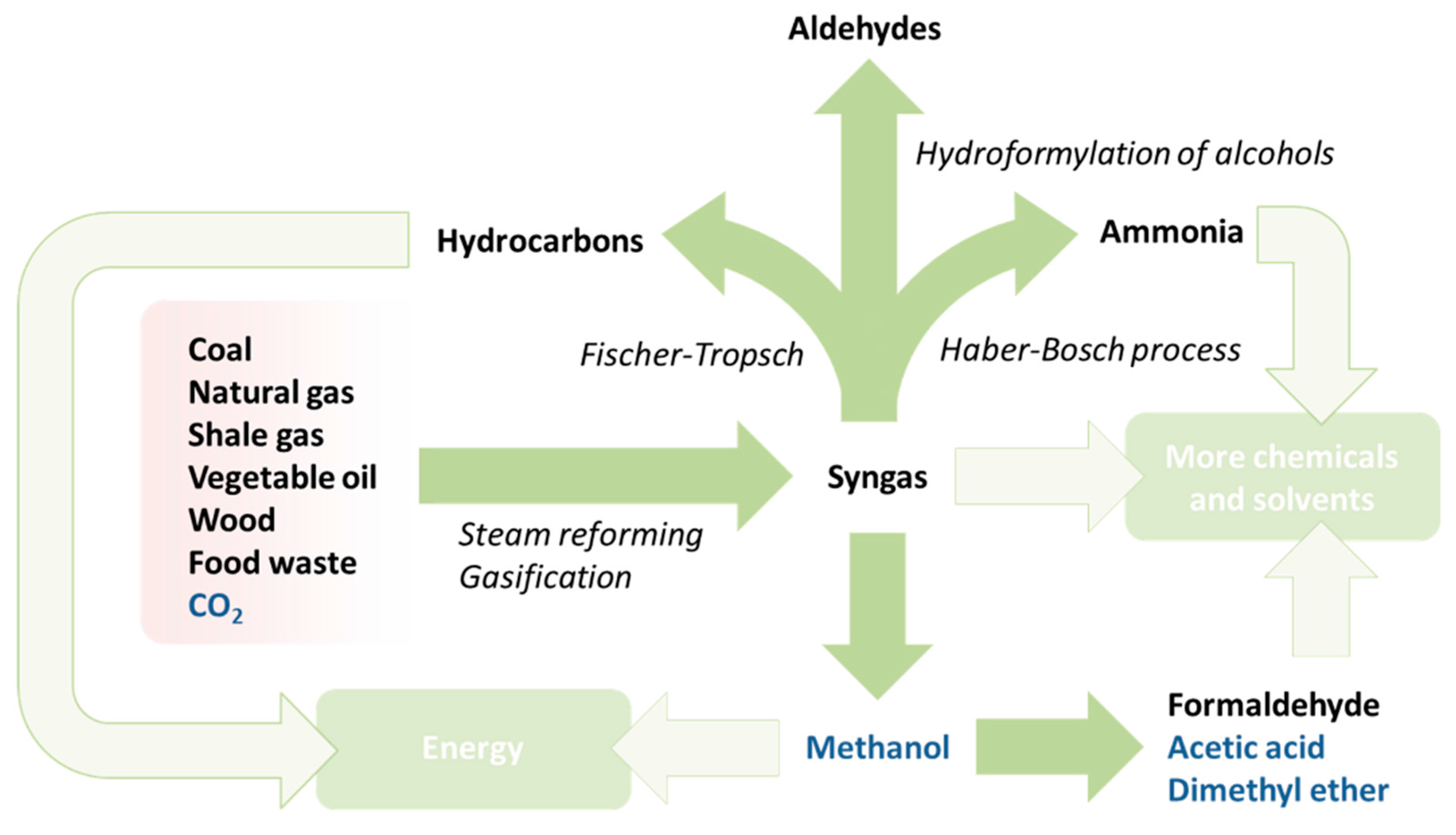
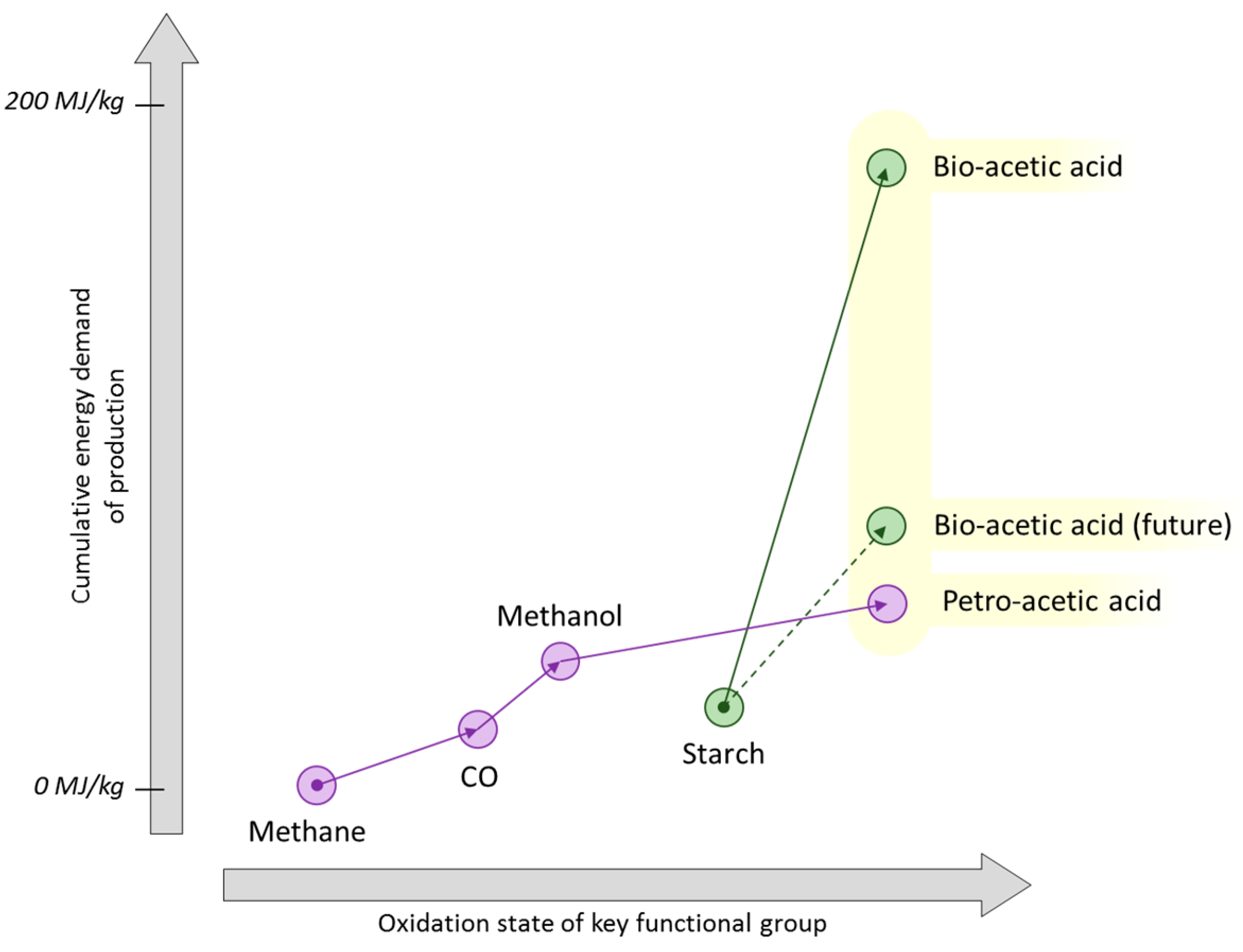
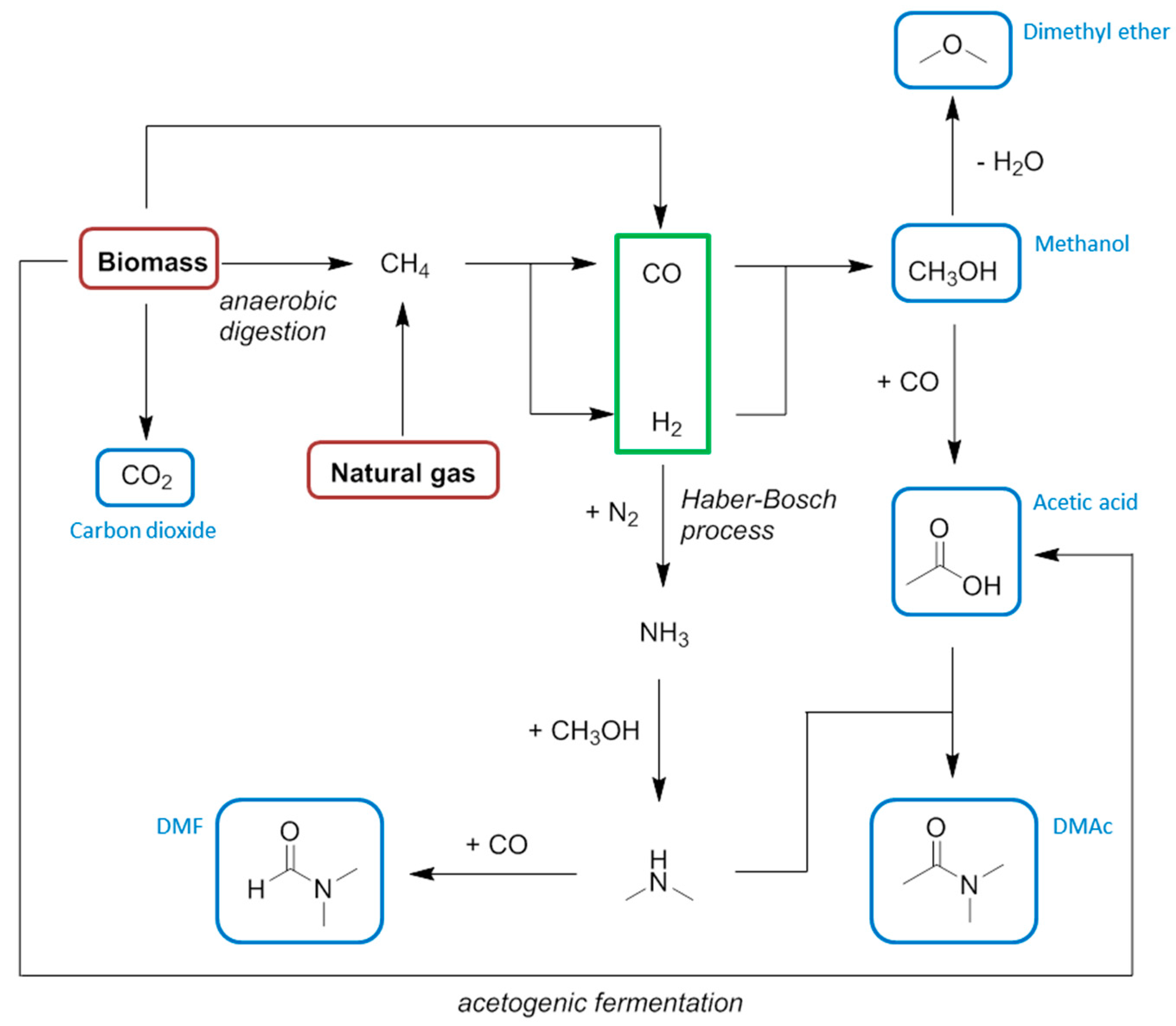
5. Methane and Syngas
| Process | Product | Reference |
|---|---|---|
| Anaerobic digestion | Methane | [105,106] |
| Sludge reactor | Hydrogen | [107] |
| Fermentation | Ethanol | [108] |
| Fermentation | Acetic acid | [109] |
| Fermentation | Lactic acid | [110] |
| Dehydration | 5-hydroxymethylfurfural (HMF) | [111] |
| Catalytic fast pyrolysis | Liquid aromatic hydrocarbons | [102] |

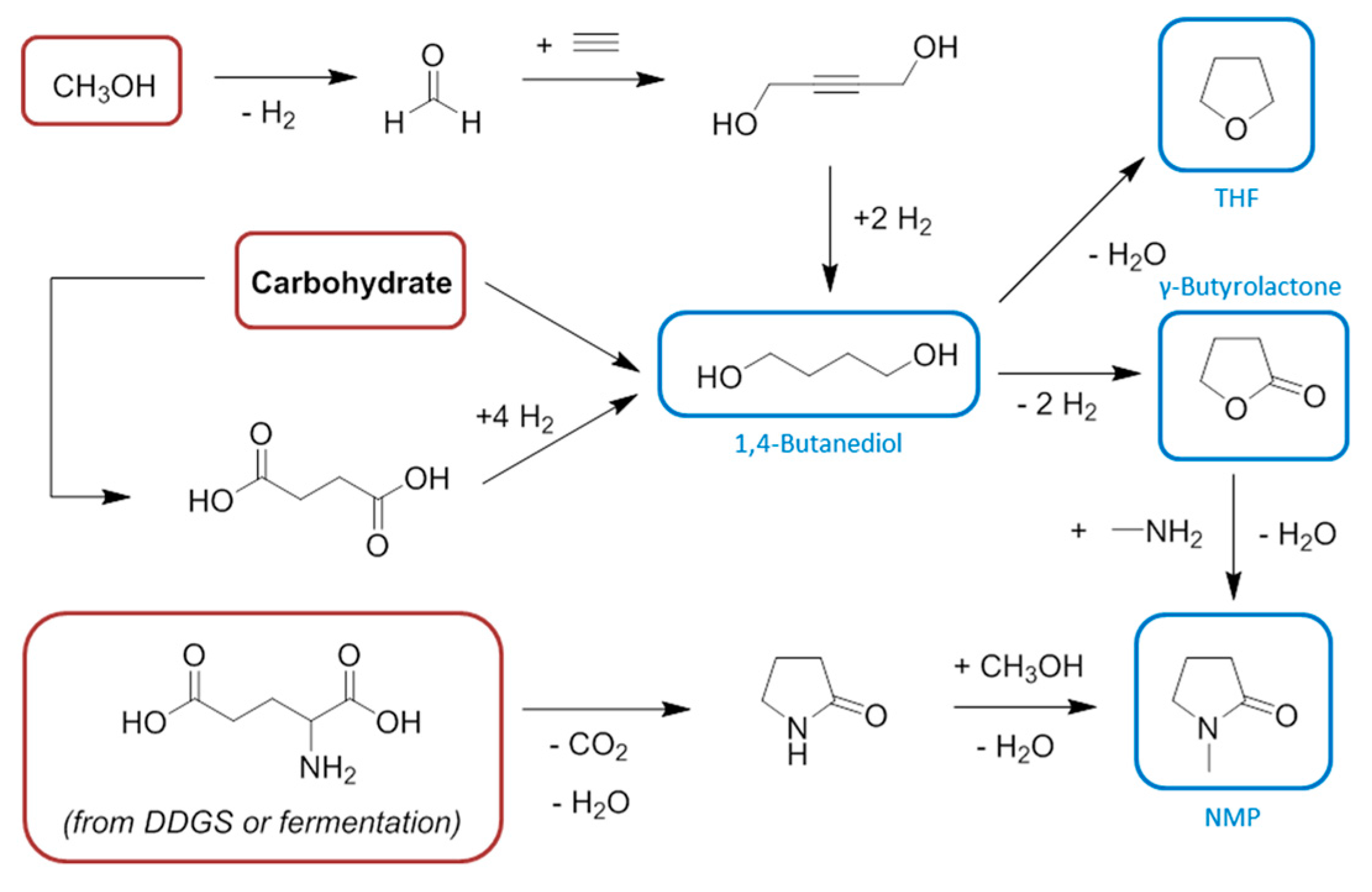
6. Ethylene

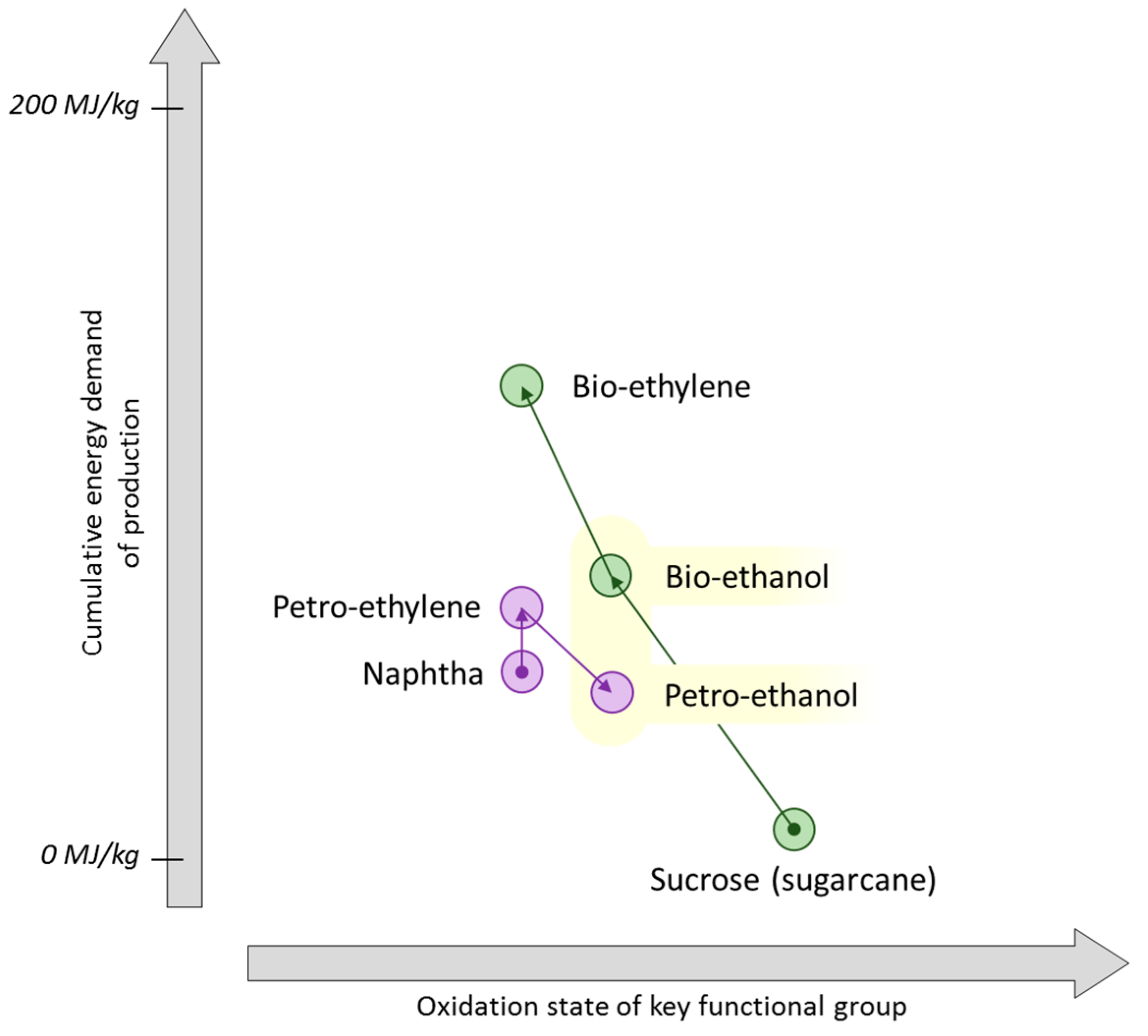
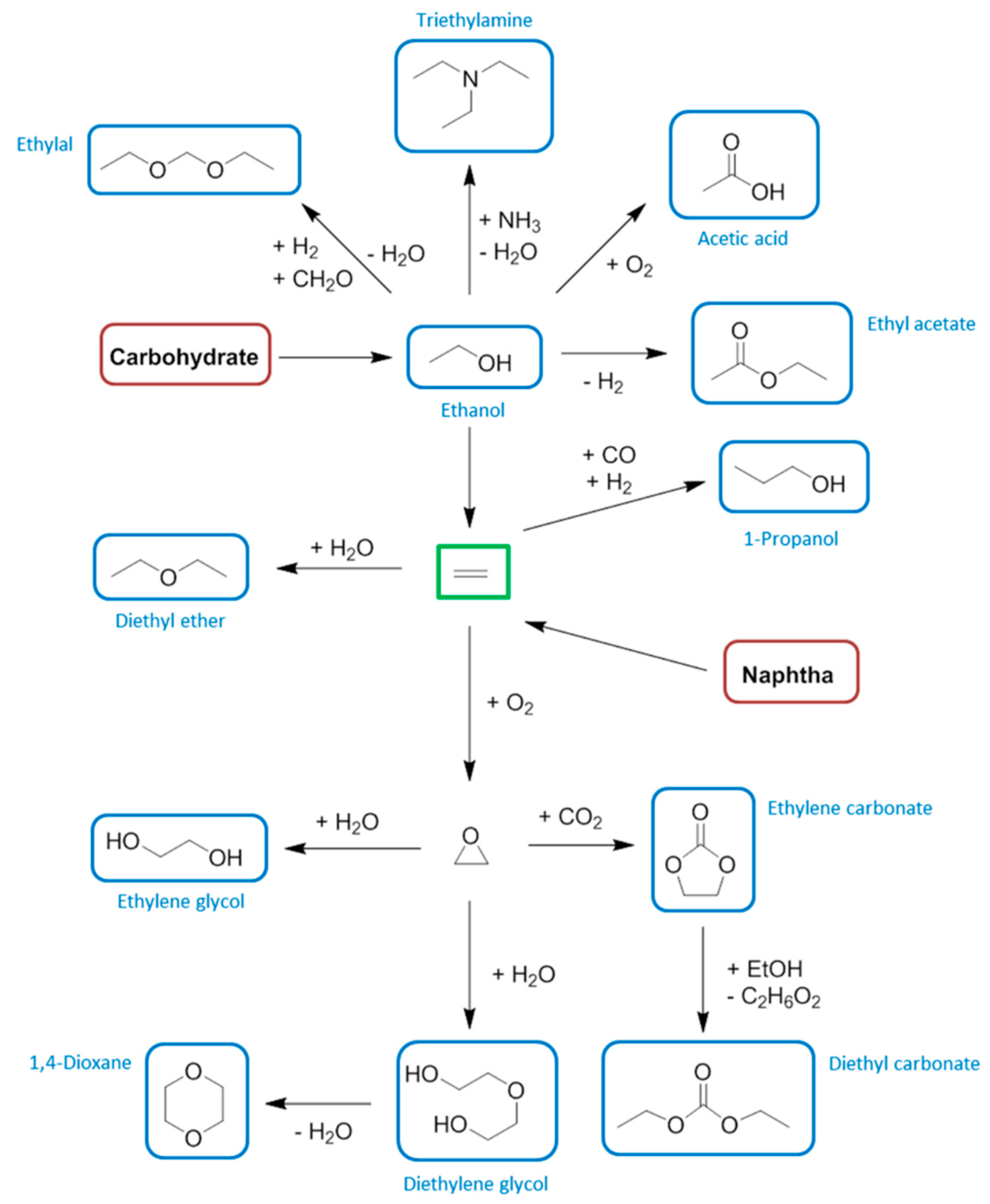
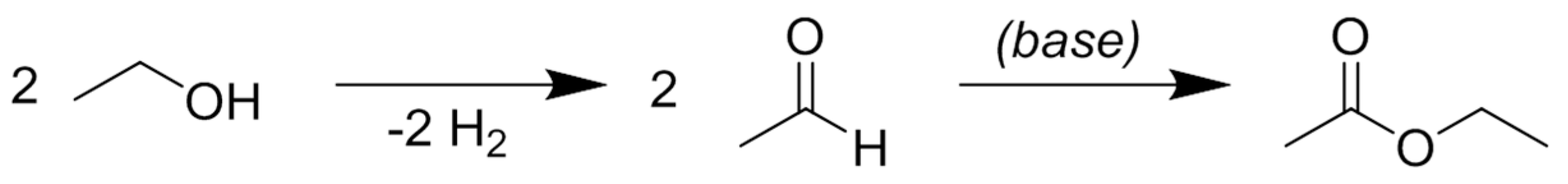
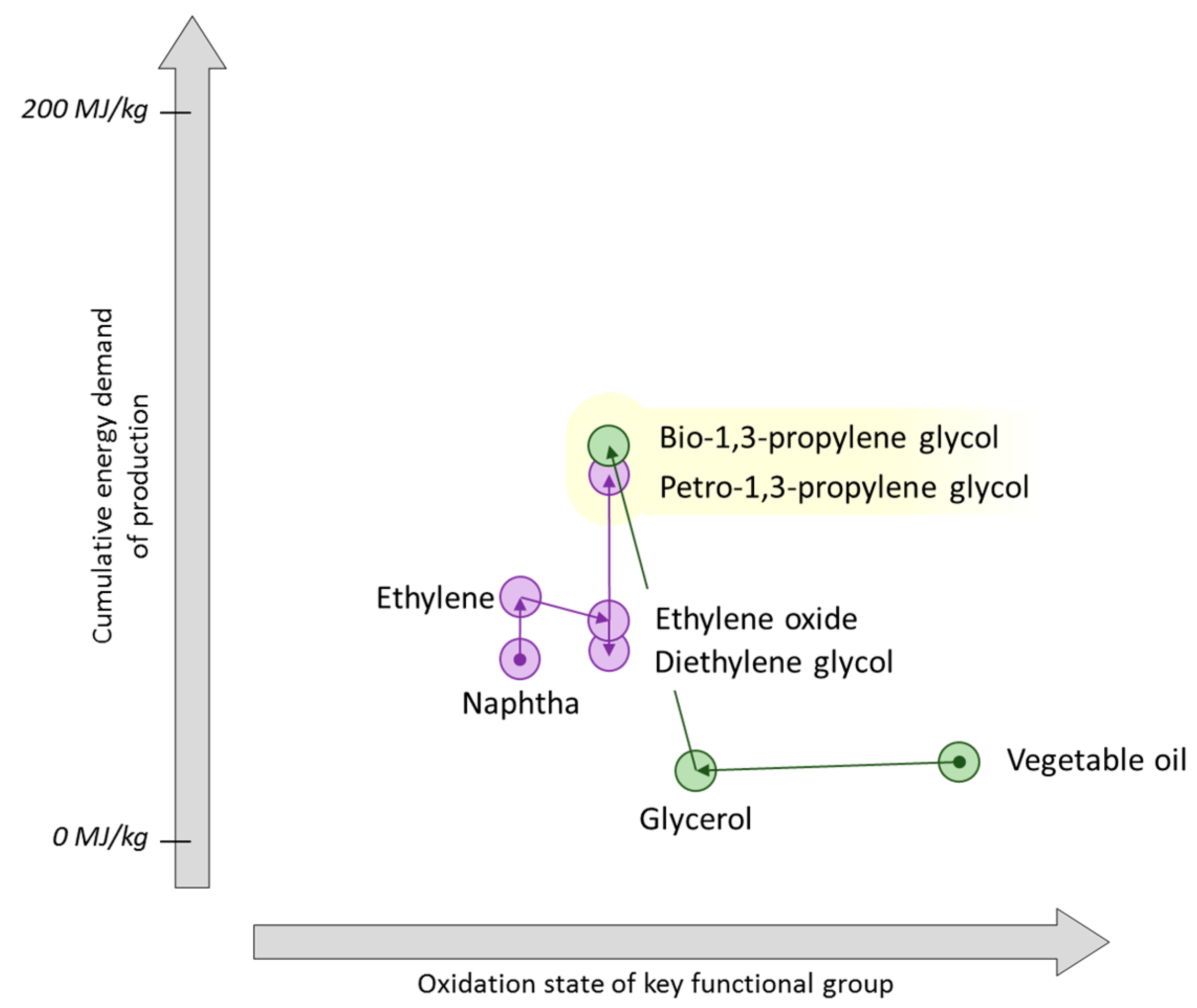
7. Propylene
| Feedstock | Conversion Technology | Readiness Level | Reference |
|---|---|---|---|
| Vegetable oil | Vegetable oil steam cracking | Commercial | [225,226] |
| Glucose | Direct fermentation | Pilot plant | [209] |
| Methanol | Methanol-to-olefins | Commercial with fossil derived feedstock | [229] |
| Ethanol | Metathesis | Pilot plant | [214,215] |
| Direct conversion | Not commercial | [221] | |
| Butanol | Metathesis | Not commercial | [230] |
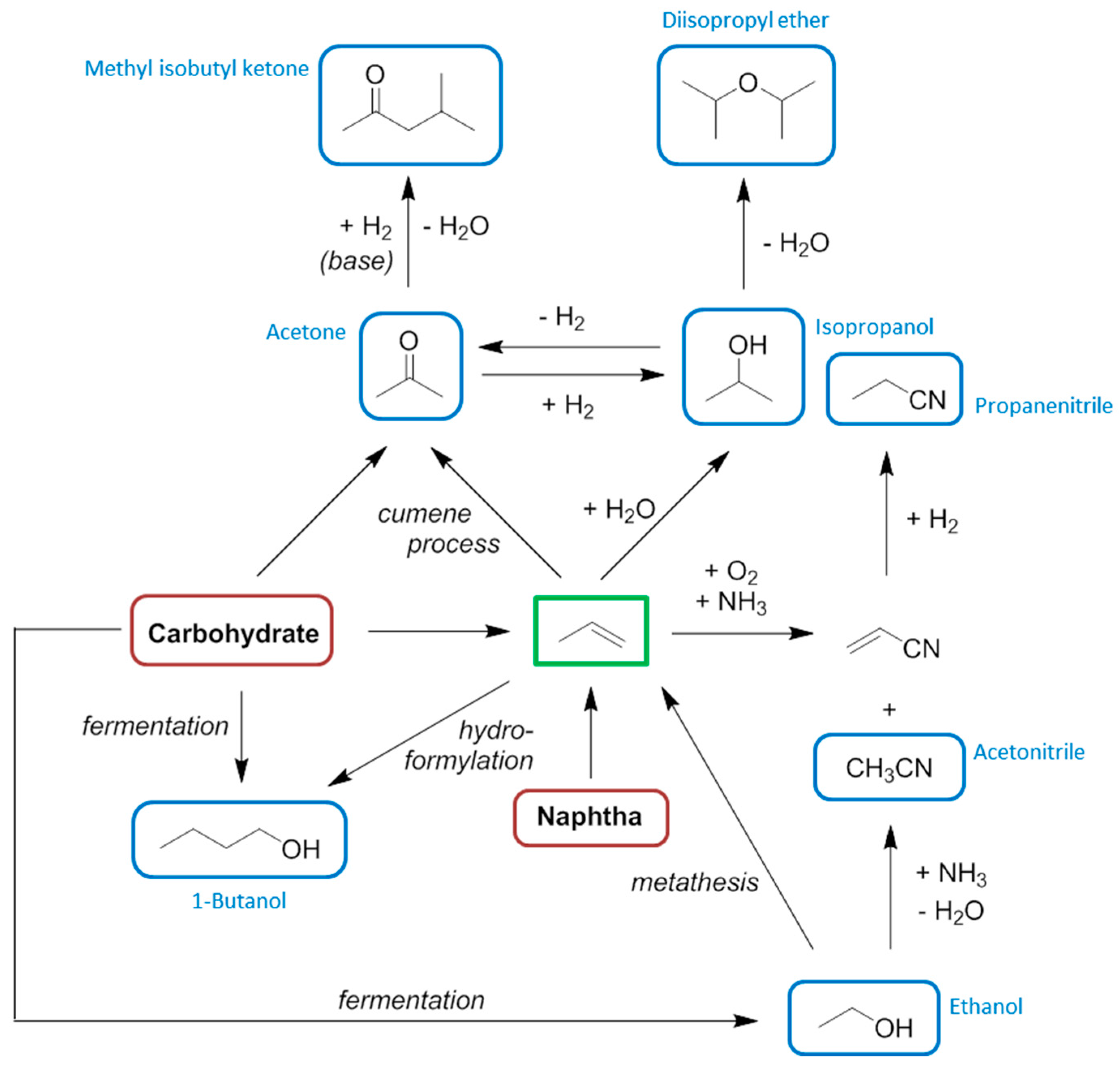
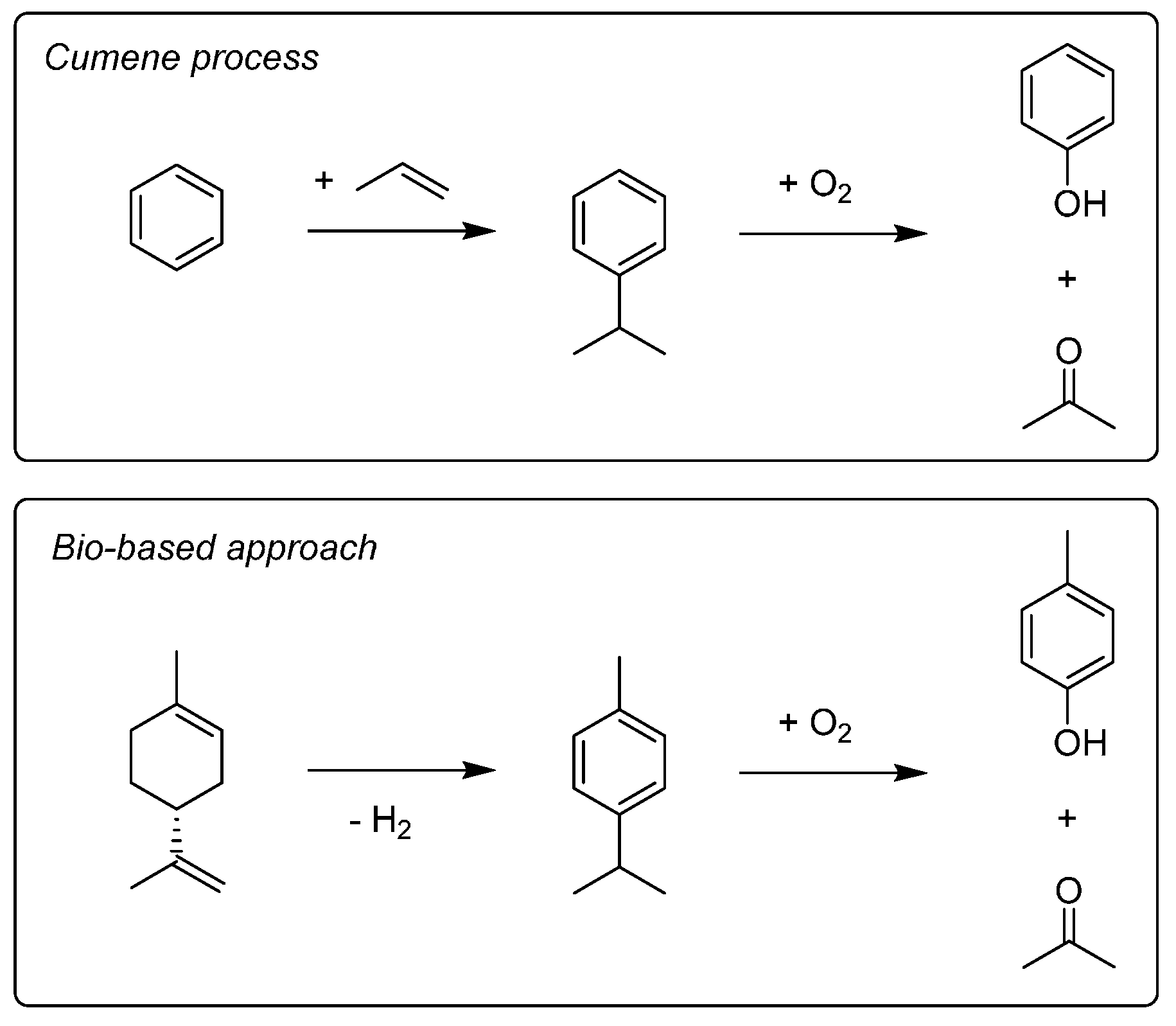
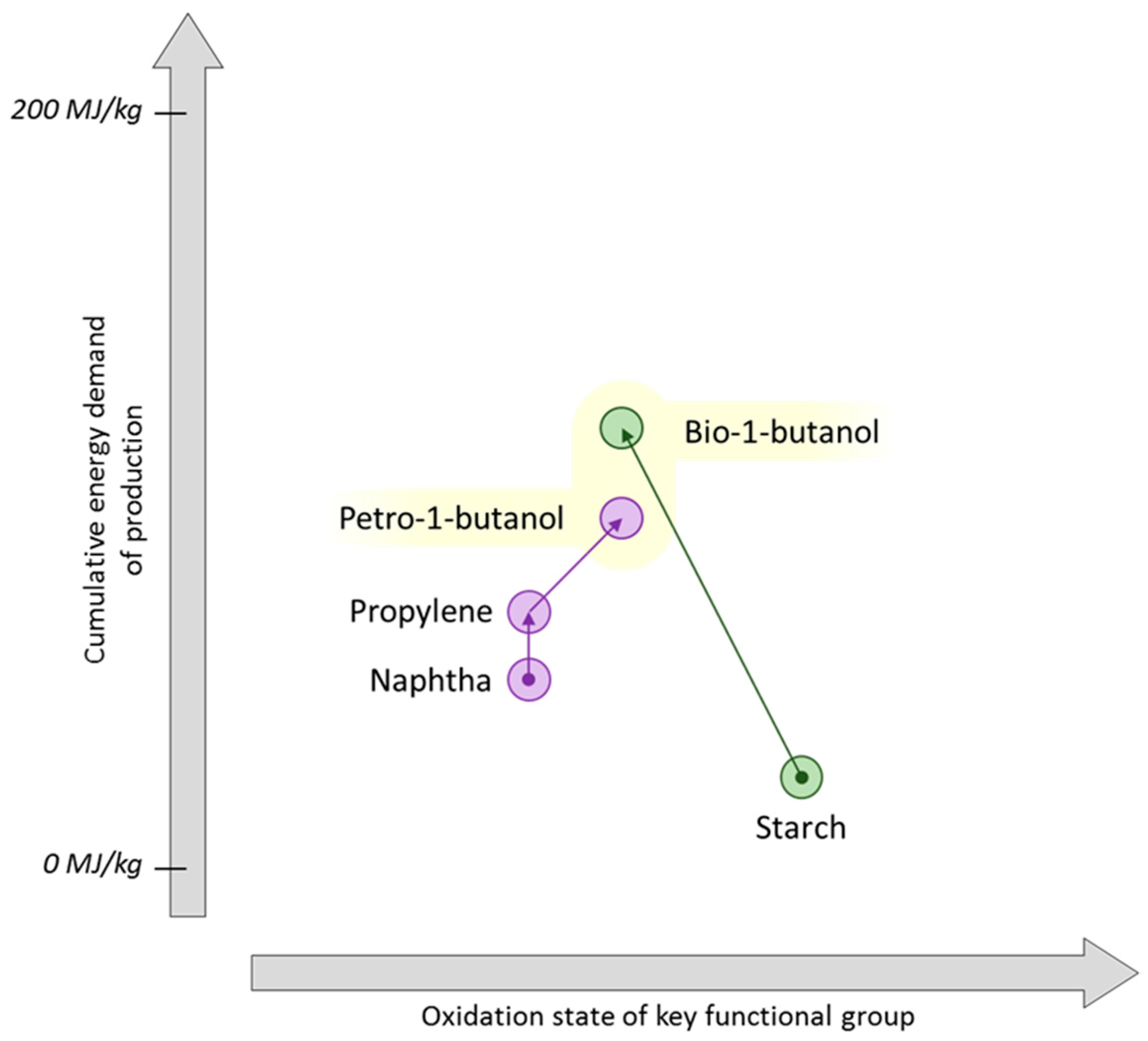
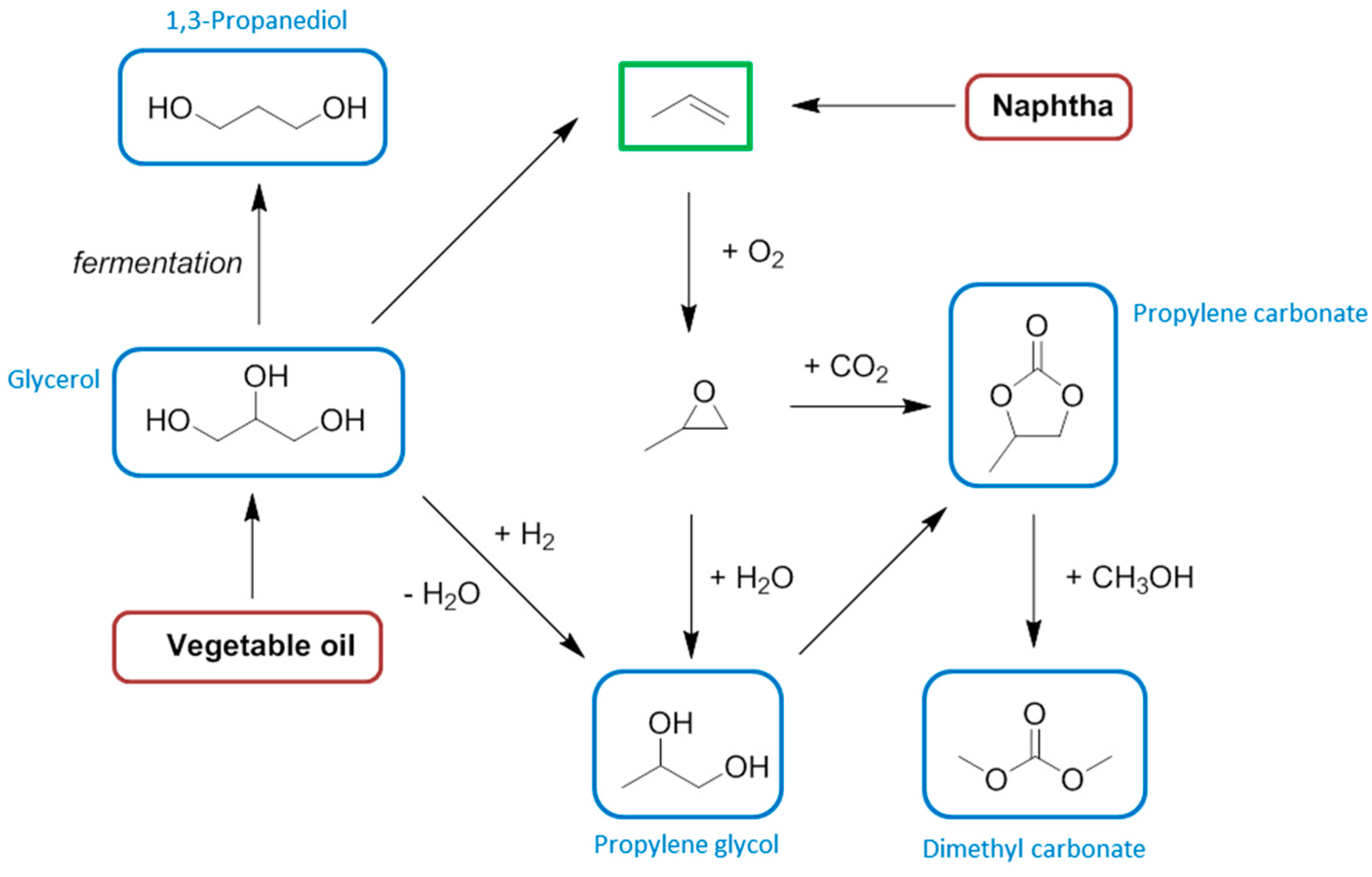
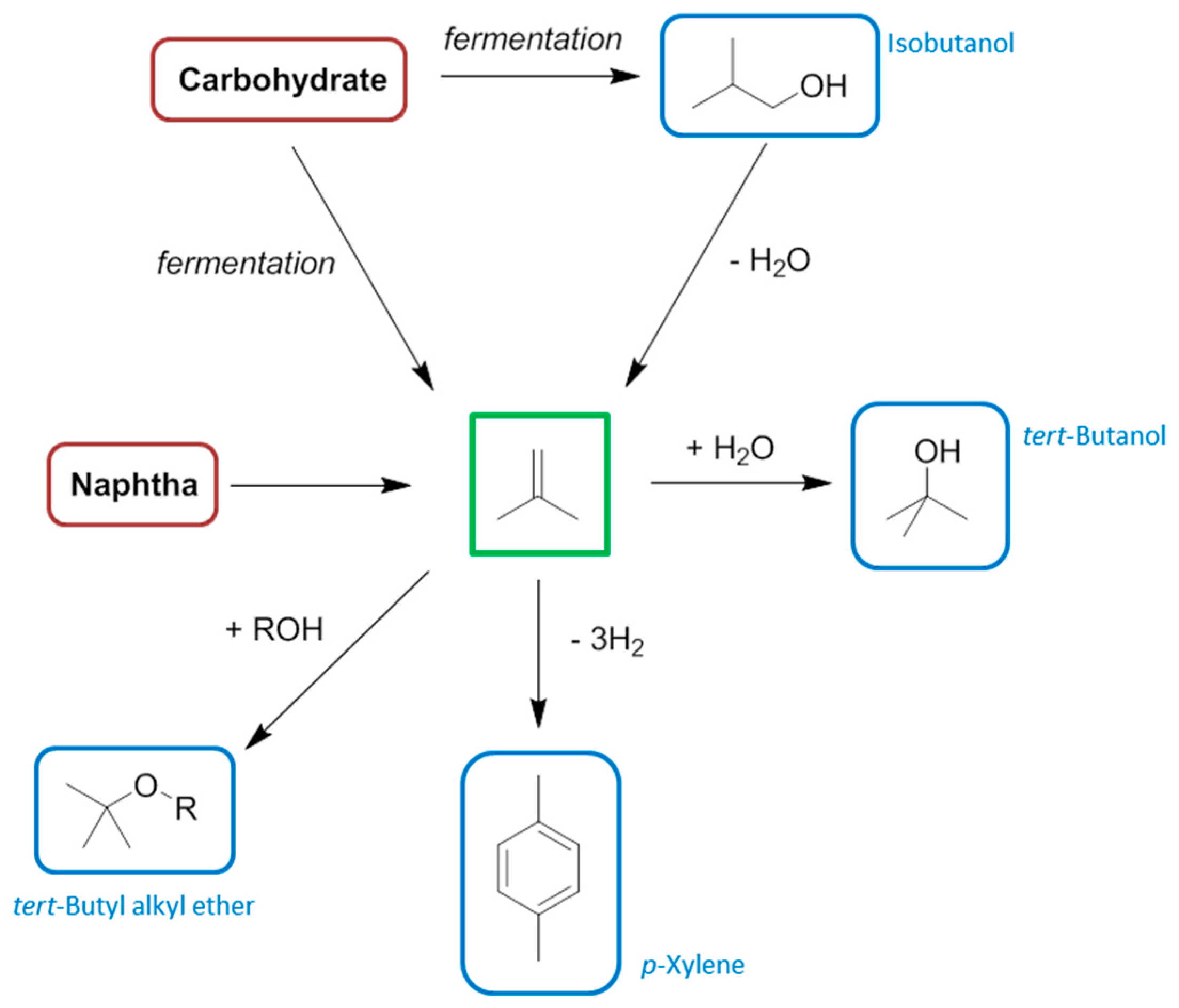
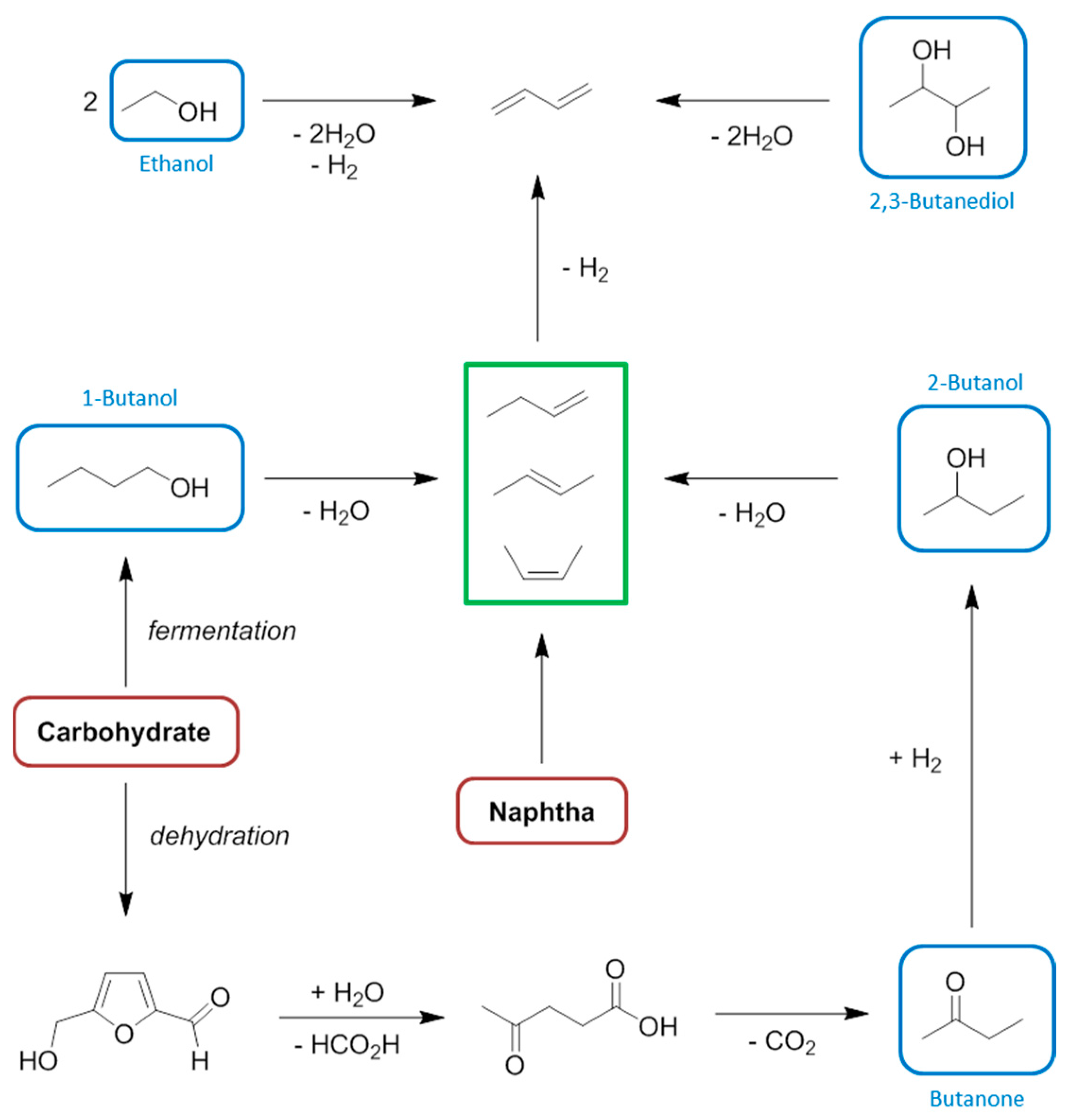
8. Butenes and Butadiene
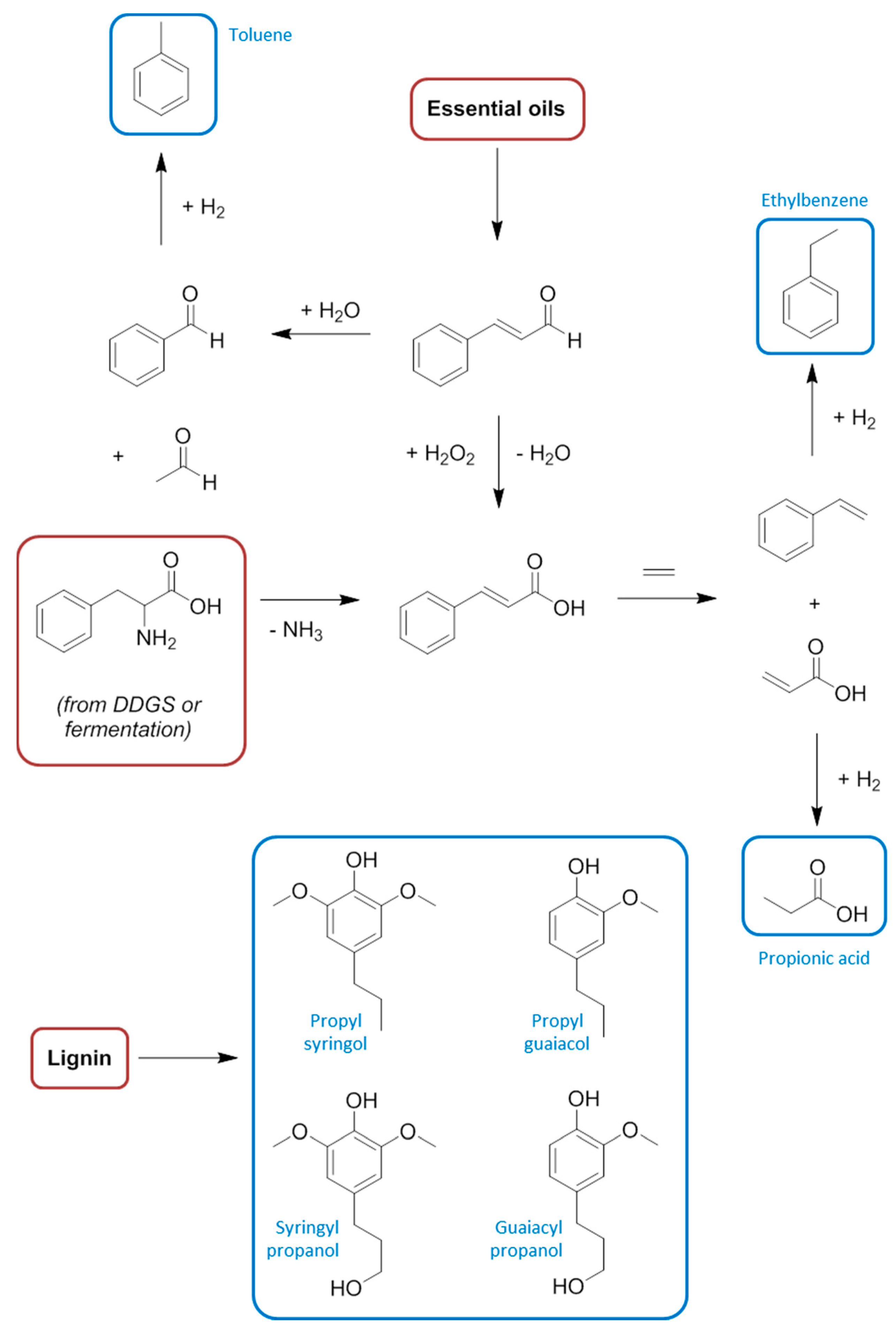
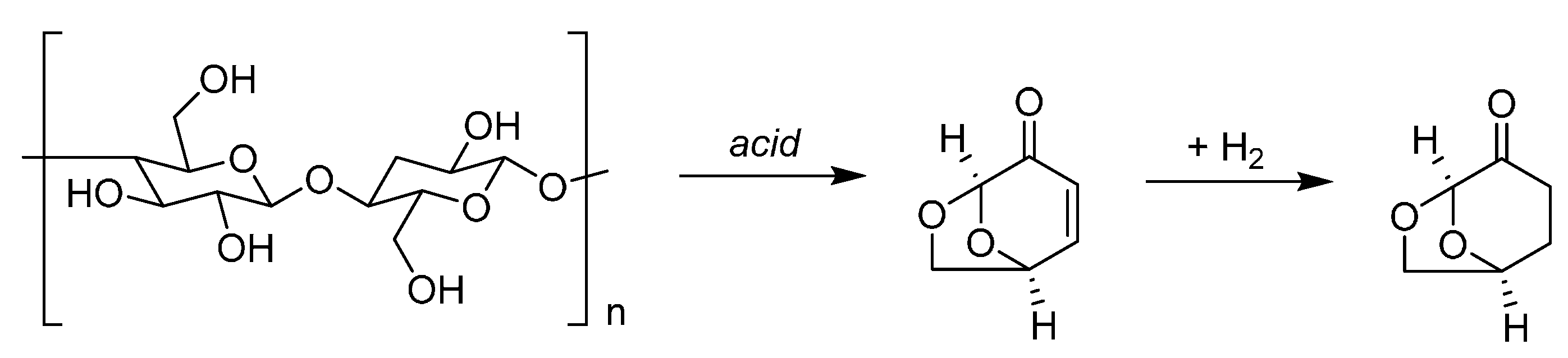
9. Aromatic Base Chemicals
| Solvent | Safety Concerns | Acute Toxicity | Chronic Toxicity | Environmental Issues |
|---|---|---|---|---|
| Benzene | Flammable | Irritant and toxic | Carcinogen and mutagen | HAP |
| Toluene | Flammable | Irritant and toxic | Suspected reproductive toxin | HAP |
| p-Xylene | Flammable | Irritant | Not known | HAP |
| p-Cymene | Flammable | Irritant | Not known | Not known |
| Ethylbenzene | Flammable | Irritant and toxic | Possible carcinogen | Harmful to aquatic life |
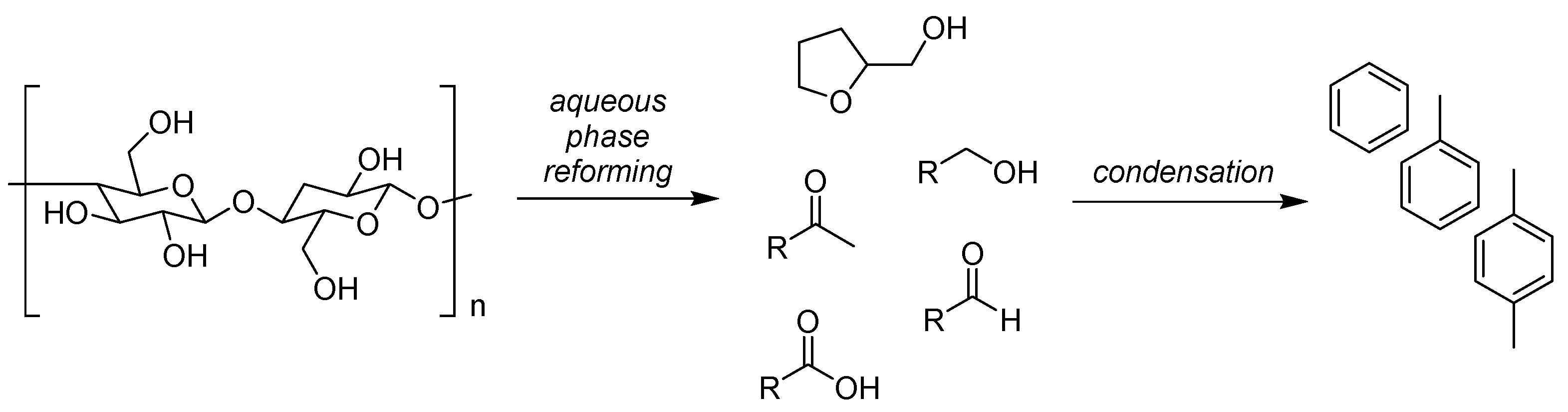
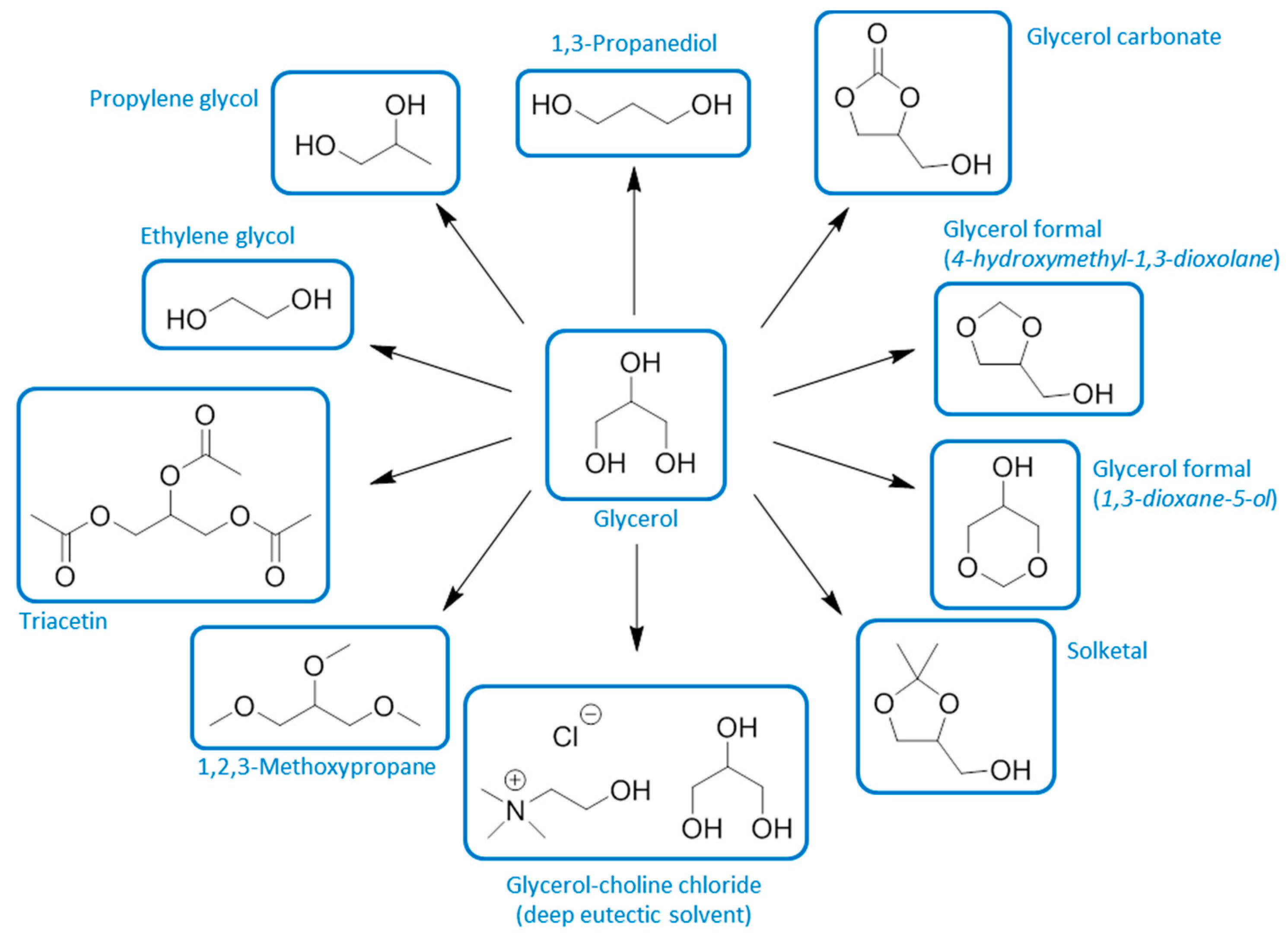
10. Neoteric Bio-Based Solvents
| Solvent | Source | Manufacturer |
|---|---|---|
| Glycerol | Vegetable oil | Cargill [334] |
| Solketal | Vegetable oil | Solvay-Rhodia [335] |
| Glycerol formal | Vegetable oil | Lambiotte & Cie [336] |
| Limonene | Essential oil | Various [337] |
| Lactic acid | Fermentation of carbohydrate | Cellulac [68] |
| Ethyl lactate | Fermentation of carbohydrate | Cellulac [68] |
| Diethyl succinate | Fermentation of carbohydrate | BioAmber [338] |
| Fusel oil | Fermentation of carbohydrate | Various [339] |
| 1,3-Propanediol | Fermentation of carbohydrate | Dupont-Tate & Lyle [261] |
| 1,2-Pentanediol | Reduction of carbohydrate | Pennakem Europa SAS [340] |
| Cyrene | Reduction of carbohydrate | Circa [160] |
| 2-MeTHF | Reduction of carbohydrate | Pennakem Europa SAS [341] |
| γ-Valerolactone | Reduction of carbohydrate | Various [339] |
| Dimethyl isosorbide | Reduction of carbohydrate | Roquette [342,343] |
| Dimethyl ether | Gasification | Chemrec [344] |
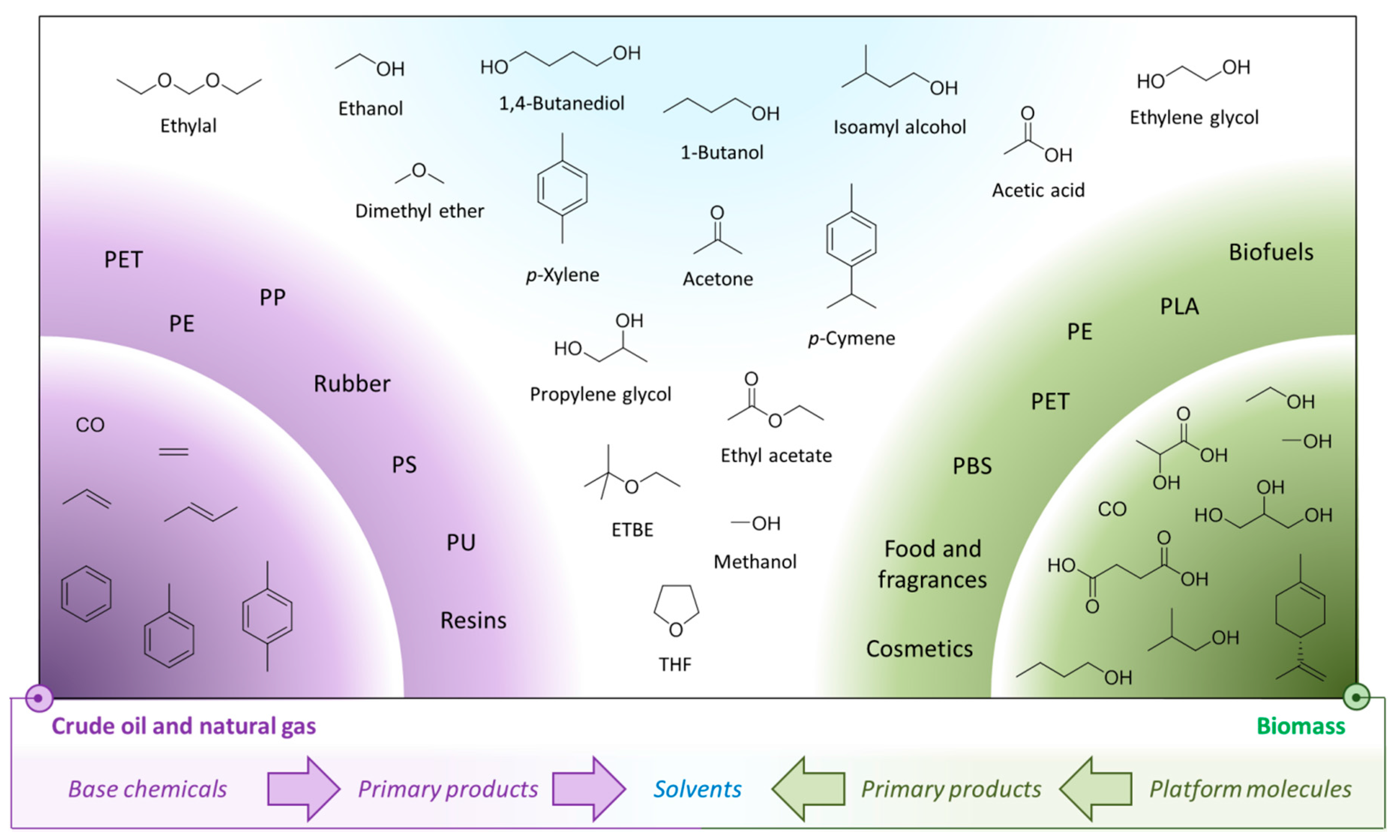
11. Conclusions
| Molecular Size | Fossil Derived Base Chemicals | Bio-Based Platform Molecules |
|---|---|---|
| C1 | Syngas | Bio-syngas |
| Methanol | ||
| C2 | Ethylene | Ethanol |
| Bio-ethylene | ||
| C3 | Propylene | Glycerol |
| Acetone | ||
| Lactic acid | ||
| C4 | Butene(s) | 1-Butanol |
| Butadiene | Succinic acid | |
| Isobutanol | ||
| C5 | n/a | Levulinic acid |
| Furfural | ||
| C6 | Benzene | HMF |
| Levoglucosenone | ||
| C7 | Toluene | n/a |
| C8 | Xylene(s) | n/a |
Author Contributions
Conflicts of Interest
References and Notes
- Nikolau, B.J.; Perera, M.A.D.N.; Brachova, L.; Shanks, B. Platform biochemicals for a biorenewable chemical industry. Plant J. 2008, 54, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Werpy, T.; Petersen, G. Top Value Added Chemicals from Biomass Volume I: Results of Screening for Potential Candidates from Sugars and Synthesis Gas; US Department of Energy: Washington, DC, USA, 2004.
- Bozell, J.J.; Petersen, G.R. Technology development for the production of biobased products from biorefinery carbohydrates—The US Department of Energy’s “Top 10” revisited. Green Chem. 2010, 12, 539–554. [Google Scholar] [CrossRef]
- Sheldon, R.A. Utilisation of biomass for sustainable fuels and chemicals: Molecules, methods and metrics. Catal. Today 2011, 167, 3–13. [Google Scholar] [CrossRef]
- Kobayashi, H.; Fukuoka, A. Synthesis and utilisation of sugar compounds derived from lignocellulosic biomass. Green Chem. 2013, 15, 1740–1763. [Google Scholar] [CrossRef]
- Luterbacher, J.S.; Alonso, D.M.; Dumesic, J.A. Targeted chemical upgrading of lignocellulosic biomass to platform molecules. Green Chem. 2014, 16, 4816–4838. [Google Scholar] [CrossRef]
- Farmer, T.J.; Mascal, M. Platform molecules. In Introduction to Chemicals from Biomass, 2nd ed.; Deswarte, C., Ed.; John Wiley and Sons: Chichester, UK, 2015; pp. 89–156. [Google Scholar]
- Corma, A.; Iborra, S.; Velty, A. Chemical routes for the transformation of biomass into chemicals. Chem. Rev. 2007, 107, 2411–2502. [Google Scholar] [CrossRef] [PubMed]
- Gallezot, P. Conversion of biomass to selected chemical products. Chem. Soc. Rev. 2012, 41, 1538–1558. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.H.; Pfaltzgraff, L.A.; Budarin, V.L.; Hunt, A.J.; Gronnow, M.; Matharu, A.S.; Macquarrie, D.J.; Sherwood, J.R. From waste to wealth using green chemistry. Pure Appl. Chem. 2013, 85, 1625–1631. [Google Scholar] [CrossRef]
- Dodson, J.R.; Cooper, E.C.; Hunt, A.J.; Matharu, A.; Cole, J.; Minihan, A.; Clark, J.H.; Macquarrie, D.J. Alkali silicates and structured mesoporous silicas from biomass power station wastes: The emergence of bio-MCMs. Green Chem. 2013, 15, 1203–1210. [Google Scholar] [CrossRef]
- Christensen, C.H.; Rass-Hansen, J.; Marsden, C.C.; Taarning, E.; Egeblad, K. The renewable chemicals industry. ChemSusChem 2008, 1, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Jérôme, F. Bio-based solvents: An emerging generation of fluids for the design of eco-efficient processes in catalysis and organic chemistry. Chem. Soc. Rev. 2013, 42, 9550–9570. [Google Scholar] [CrossRef] [PubMed]
- Innovating for Sustainable Growth: A Bioeconomy for Europe. Available online: http://ec.europa.eu/research/bioeconomy/pdf/201202_innovating_sustainable_growth_en.pdf (accessed on 15 May 2015).
- M/491 Mandate address to CEN, CENELEC and ETSI for the Development of European Standards and Technical Specifications and/or Technical Reports for Bio-Surfactants and bio-Solvents in Relations to Bio-Based Product Aspects. Available online: http://ec.europa.eu/growth/tools-databases/mandates/index.cfm?fuseaction=search.detail&id=476# (accessed on 18 May 2015).
- Bio-Based Economy. Available online: http://www.biobasedeconomy.eu/standardisation (accessed on 15 May 2015).
- CEN/TC 411/WG 2 Bio-solvents. Available online: http://standards.cen.eu/dyn/www/f?p=204:110:0::::FSP_PROJECT:40174&cs=161BAFBE2B2659903E8B55F9326BA295B (accessed on 18 May 2015).
- New Natural Resource Base in the Chemical Industry only a Matter of Time. Available online: http://www.achema.de/fileadmin/user_upload/Bilder/Presse/ACHEMA2012/Trendberichte/Trendberichte_2012/tb19_en_Biobased_Chemicals.docx (accessed on 16 June 2015).
- Global Solvents Opportunities for Greener Solvents. Available online: http://www.ihs.com/products/chemical-special-reports-global-solvents.html (accessed on 8 June 2015).
- Solvent Market by Type, Application and Source Global Trends and Forecast to 2018. Available online: http://www.marketsandmarkets.com/Market-Reports/solvent-market-1325.html (accessed on 8 June 2015).
- Solvents Market Global Industry Analysis Size Share Growth Trends and Forecast 2012-2018. Available online: http://www.linkedin.com/pulse/20140528072848-339157087-solvents-market-global-industry-analysis-size-share-growth-trends-and-forecast-2012-2018 (accessed on 8 June 2015).
- Solvents Facts and Figures. Available online: http://www.esig.org/en/about-solvents/what-are-solvents/facts-and-figures-the-european-solvents-industry-in-brief (accessed on 8 June 2015).
- ESIG Solvent Fact Sheets. Available online: http://www.esig.org/en/library/publications/fact-sheets (accessed on 8 June 2015).
- Solvents and Ozone ESIG Fact Sheet. Available online: http://www.esig.org/uploads/ModuleXtender/Publications/90/ESIG%20Factsheets%202005%20(EN).pdf (accessed on 8 June 2015).
- BIOCHEM Project D2.3 Report on the Assessment of the Bio-Based Products Market Potential for Innovation. Available online: http://www.biochem-project.eu/download/toolbox/innovation/06/Bio-based%20product%20market%20potential.pdf (accessed on 16 June 2015).
- ESIG Newsletter “Solutions” Spring 1997. Available online: http://www.esig.org/uploads/ModuleXtender/Publications/122/Solutions%201%20(EN).pdf (accessed on 21 July 2015).
- Breeden, S.W.; Clark, J.H.; Macquarrie, D.J.; Sherwood, J.R. Green solvents. In Green Techniques for Organic Synthesis and Medicinal Chemistry; Zhang, C., Ed.; John Wiley and Sons: Chichester, UK, 2012; pp. 241–261. [Google Scholar]
- Directive 2009/32/EC of the European Parliament and of the Council of 23 April 2009 on the Approximation of the Laws of the Member States on Extraction Solvents Used in the Production of Foodstuffs and Food Ingredients. Available online: http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32009L0032 (accessed on 22 May 2015).
- Kerton, F.M.; Marriott, R. Green solvents legislation and certification. In Alternative Solvents for Green Chemistry, 2nd ed.; RSC: Cambridge, UK, 2013; pp. 31–50. [Google Scholar]
- Alfonsi, K.; Colberg, J.; Dunn, P.J.; Fevig, T.; Jennings, S.; Johnson, T.A.; Kleine, H.P.; Knight, C.; Nagy, M.A.; Perry, D.A.; et al. Green chemistry tools to influence a medicinal chemistry and research chemistry based organisation. Green Chem. 2008, 10, 31–36. [Google Scholar] [CrossRef]
- Regulation (EC) 1907/2006 of the European Parliament and of the Council Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemicals Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2007:136:0003:0280:en:PDF (accessed on 16 June 2015).
- ECHA Candidate List of Substances of Very High Concern for Authorisation. Available online: http://echa.europa.eu/candidate-list-table (accessed on 15 May 2015).
- ECHA List of Restrictions. Available online: http://echa.europa.eu/addressing-chemicals-of-concern/restrictions/list-of-restrictions (accessed on 15 May 2015).
- Henderson, R.K.; Jiménez-González, C.; Constable, D.J.C.; Alston, S.R.; Inglis, G.G.A.; Fisher, G.; Sherwood, J.; Binks, S.P.; Curzons, A.D. Expanding GSK’s solvent selection guide—Embedding sustainability into solvent selection starting at medicinal chemistry. Green Chem. 2011, 13, 854–862. [Google Scholar] [CrossRef]
- Prat, D.; Pardigon, O.; Flemming, H.W.; Letestu, S.; Ducandas, V.; Isnard, P.; Guntrum, E.; Senac, T.; Ruisseau, S.; Cruciani, P.; et al. Sanofi’s solvent selection guide: A step toward more sustainable processes. Org. Process Res. Dev. 2013, 17, 1517–1525. [Google Scholar] [CrossRef]
- McKinsey Green Chemicals Survey Key Findings. Available online: http://chemroundtables.com/wp-content/uploads/2012/08/6-20120910-McKinsey-Green-Chemicals-Survey_public-presentation_final.pdf (accessed on 1 June 2015).
- ICH Impurities Guideline for Residual Solvents. Available online: http://www.ich.org/products/guidelines/quality/quality-single/article/impurities-guideline-for-residual-solvents.html (accessed on 1 June 2015).
- Huber, G.W.; Corma, A. Synergies between bio- and oil refineries for the production of fuels from biomass. Angew. Chem. Int. Ed. 2007, 46, 7184–7201. [Google Scholar] [CrossRef] [PubMed]
- Vennestrøm, P.N.R.; Osmundsen, C.M.; Christensen, C.H.; Taarning, E. Beyond petrochemicals: The renewable chemicals industry. Angew. Chem. Int. Ed. 2011, 50, 10502–10509. [Google Scholar] [CrossRef] [PubMed]
- Sherwood, J.; de bruyn, M.; Constantinou, A.; Moity, L.; McElroy, C.R.; Farmer, T.J.; Duncan, T.; Raverty, W.; Hunt, A.J.; Clark, J.H. Dihydrolevoglucosenone (Cyrene) as a bio-based alternative for dipolar aprotic solvents. Chem. Commun. 2014, 50, 9650–9652. [Google Scholar] [CrossRef] [PubMed]
- Ismalaj, E.; Strappaveccia, G.; Ballerini, E.; Elisei, F.; Piermatti, O.; Gelman, D.; Vaccaro, L. γ-Valerolactone as a renewable dipolar aprotic solvent deriving from biomass degradation for the Hiyama reaction. ACS Sustain. Chem. Eng. 2014, 2, 2461–2464. [Google Scholar] [CrossRef]
- Strappaveccia, G.; Ismalaj, E.; Petrucci, C.; Lanari, D.; Marrocchi, A.; Drees, M.; Facchetti, A.; Vaccaro, L. A biomass-derived safe medium to replace toxic dipolar solvents and access cleaner Heck coupling reactions. Green Chem. 2015, 17, 365–372. [Google Scholar] [CrossRef]
- Strappaveccia, G.; Luciani, L.; Bartollini, E.; Marrocchi, A.; Pizzoa, F.; Vaccaro, L. γ-Valerolactone as an alternative biomass-derived medium for the Sonogashira reaction. Green Chem. 2015, 17, 1071–1076. [Google Scholar] [CrossRef]
- Kamm, B.; Kamm, M. Principles of biorefineries. Appl. Microbiol. Biotechnol. 2004, 64, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Dawes, G.J.S.; Scott, E.L.; Le Nôtre, J.; Sanders, J.P.M.; Bitter, J.P.M. Deoxygenation of biobased molecules by decarboxylation and decarbonylation a review on the role of heterogeneous homogeneous and bio-catalysis. Green Chem. 2015, 17, 3231–3250. [Google Scholar] [CrossRef]
- Sheldon, R.A. Green and sustainable manufacture of chemicals from biomass: State of the art. Green Chem. 2014, 16, 950–963. [Google Scholar] [CrossRef]
- An Analysis of European Plastics Production Demand and Waste Data. Available online: http://issuu.com/plasticseuropeebook/docs/final_plastics_the_facts_2014_19122 (accessed on 29 May 2015).
- IHS Report 284, Bio-Based Adipic Acid. Available online: http://www.ihs.com/products/chemical-technology-pep-bio-based-adipic-acid-2012.html (accessed on 15 May 2015).
- Rennovia Product Pipeline. Available online: http://www.rennovia.com/product-pipeline (accessed on 15 May 2015).
- ICIS Green Chemicals, Introducing Rennovia. Available online: http://www.icis.com/blogs/green-chemicals/2010/09/introducing-rennovia (accessed on 15 May 2015).
- Gong, Y.; Lin, L.; Shi, J.; Liu, S. Oxidative decarboxylation of levulinic acid by cupric oxides. Molecules 2010, 15, 7946–7960. [Google Scholar] [CrossRef] [PubMed]
- Biofuels and Sustainability Issues. Available online: http://biofuelstp.eu/sustainability.html (accessed on 16 June 2015).
- Rinaldi, R.; Schüth, F. Design of solid catalysts for the conversion of biomass. Energy Environ. Sci. 2009, 2, 610–626. [Google Scholar] [CrossRef]
- Medium and Long-Term Opportunities and Risk of the Biotechnological Production of Bulk Chemicals from Renewable Resources—The Potential of White Biotechnology. Available online: http://www.bio-economy.net/applications/files/Brew_project_report.pdf (accessed on 16 June 2015).
- FDA Compliance Policy Guide Sec. 562.100 Acetic Acid—Use in Foods—Labeling of Foods in Which Used. Available online: http://www.fda.gov/ICECI/ComplianceManuals/CompliancePolicyGuidanceManual/ucm074577.htm (accessed on 1 June 2015).
- Cheung, H.; Tanke, R.S.; Torrence, G.P. Acetic acid. In Ullmann’s Encyclopedia of Industrial Chemistry; John Wiley and Sons: Hoboken, NJ, USA, 2000. [Google Scholar]
- Pace, V.; Hoyos, P.; Castoldi, L.; Domínguez de María, P.; Alcántara, A.R. 2-Methyltetrahydrofuran (2-MeTHF) a biomass-derived solvent with broad application in organic chemistry. ChemSusChem 2012, 5, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Colley, S.W.; Fawcett, C.R.; Rathmell, C.; Tuck, M.W.M. Process for the Preparation of Ethyl Acetate. US Patent 6809217 B1, 26 October 2004. [Google Scholar]
- SEKAB Ethyl Acetate. Available online: http://www.sekab.com/chemistry/ethyl-acetate (accessed on 25 May 2015).
- Emami-Taba, L.; Irfan, M.F.; Wan Daud, M.A.W.; Chakrabarti, M.H. Fuel blending effects on the co-gasification of coal and biomass a review. Biomass Bioenergy 2013, 57, 249–263. [Google Scholar] [CrossRef]
- Brar, J.S.; Singh, K.; Wang, J.; Kumar, S. Cogasification of coal and biomass a review. Int. J. For. Res. 2012, 2012, 363058. [Google Scholar] [CrossRef]
- Bezergianni, S.; Voutetakis, S.; Kalogianni, A. Catalytic hydrocracking of fresh and used cooking oil. Ind. Eng. Chem. Res. 2009, 48, 8402–8406. [Google Scholar] [CrossRef]
- Furimsky, E. Hydroprocessing challenges in biofuels production. Catal. Today 2013, 217, 13–56. [Google Scholar] [CrossRef]
- Mutlu, H.; Meier, M.A.R. Castor oil as a renewable resource for the chemical industry. Eur. J. Lipid Sci. Technol. 2010, 112, 10–30. [Google Scholar] [CrossRef]
- Morschbacker, A. Bio-Ethanol Based Ethylene. Polym. Rev. 2009, 49, 79–84. [Google Scholar] [CrossRef]
- Fan, D.; Dai, D.J.; Wu, H.S. Ethylene formation by catalytic dehydration of ethanol with industrial considerations. Materials 2013, 6, 101–115. [Google Scholar] [CrossRef]
- Obuchi, S.; Ogawa, S. Packaging and other commercial applications. In Poly(lactic acid) Synthesis Structures Properties Processing and Applications; Auras, L., Selke, T., Eds.; John Wiley & Sons: Hoboken, NJ, USA; pp. 457–467.
- Cellulac Production Processes. Available online: http://cellulac.co.uk/en/main/process-diagram (accessed on 29 May 2015).
- Qureshi, N.; Blaschek, H.P. ABE production from corn: A recent economic evaluation. J. Ind. Microbiol. Biotechnol. 2001, 27, 292–297. [Google Scholar] [CrossRef] [PubMed]
- Green Biologics Acetone. Available online: http://www.greenbiologics.com/acetone.php (accessed on 15 May 2015).
- García, J.I.; García-Marín, H.; Pires, E. Glycerol based solvents: Synthesis, properties and applications. Green Chem. 2014, 16, 1007–1033. [Google Scholar] [CrossRef]
- Ciriminna, R.; Lomeli-Rodriguez, M.; Carà, P.D.; Lopez-Sanchez, J.A.; Pagliaro, M. Limonene: A versatile chemical of the bioeconomy. Chem. Commun. 2014, 50, 15288–15296. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation (WHO) Chemical Assessment of Limonene. Available online: http://www.who.int/ipcs/publications/cicad/en/cicad05.pdf (accessed on 29 May 2015).
- Turpentine Production and Processing. Available online: http://nzic.org.nz/ChemProcesses/forestry/4F.pdf (accessed on 29 May 2015).
- BP Statistical Review of World Energy 2014. Available online: http://www.bp.com/en/global/corporate/about-bp/energy-economics/statistical-review-of-world-energy.html (accessed on 25 May 2015).
- US Energy Information Administration, Shale Gas Provides Largest Share of U.S. Natural Gas Production in 2013. Available online: http://www.eia.gov/todayinenergy/detail.cfm?id=18951 (accessed on 15 May 2015).
- Liu, K.; Cui, Z.; Chen, W.; Zhang, L. Coal and syngas to liquids. In Hydrogen and Syngas Production and Purification Technologies; Liu, S., Subramani, V., Eds.; John Wiley and Sons: Hoboken, NY, USA, 2010; pp. 498–509. [Google Scholar]
- Van Steen, E.; Claeys, M. Fischer-Tropsch catalysts for the biomass-to-liquid (BTL) process. Chem. Eng. Technol. 2008, 31, 655–666. [Google Scholar] [CrossRef]
- Liu, G.; Larson, E.D.; Williams, R.H.; Kreutz, T.G.; Guo, X. Making Fischer-Tropsch fuels and electricity from coal and biomass: Performance and cost analysis. Energy Fuels 2011, 25, 415–437. [Google Scholar] [CrossRef]
- Fischer-Tropsch Fuels from Coal and Biomass. Available online: http://mitei.mit.edu/system/files/kreutz-fischer-tropsch.pdf (accessed on 29 May 2015).
- Fischer-Tropsch Fuels from Coal, Natural Gas, and Biomass Background and Policy. Available online: http://research.policyarchive.org/19952.pdf (accessed on 29 May 2015).
- Wypych, G. Overview of methods of solvent manufacture. In Handbook of Solvents; Wypych, A., Ed.; ChemTec Publishing: Toronto, ON, Canada, 2001; pp. 69–74. [Google Scholar]
- Koutinas, A.A.; Du, C.; Wang, R.H.; Webb, C. Production of chemicals from biomass. In Introduction to Chemicals from Biomass, 1st ed.; Clark, D., Ed.; John Wiley and Sons: Chichester, UK, 2008; p. 78. [Google Scholar]
- Wolf, K.; Yazdani, A.; Yates, P. Chlorinated solvents: Will the alternatives be safer? J. Air Waste Manag. 1991, 41, 1055–1061. [Google Scholar] [CrossRef]
- EPA Report, Alternatives to Chlorinated Solvents for Cleaning and Degreasing. Available online: http://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=30004N9I.TXT (accessed on 29 May 2015).
- EPA Report, Evaluation of Alternatives to Chlorinated Solvent for Metal Cleaning. Available online: http://www.turi.org/content/download/6209/65352/file/1996Report46EllenbeckerandThomas-EvalofAlttoChlorSolvents.pdf (accessed on 29 May 2015).
- Dow Choices and Solutions Newsletter Alternatives to Chlorinated Solvents. Available online: http://www.dow.com/webapps/lit/litorder.asp?filepath=gco/pdfs/noreg/100-06917.pdf&pdf=true (accessed on 29 May 2015).
- Quitmeyer, J. Aqueous cleaners challenge chlorinated solvents. Pollut. Eng. 1991, 23, 88–91. [Google Scholar]
- Solvent Technology for Present and Future Air Quality Regulations. Available online: tttp://www.eastman.com/Literature_Center/M/M310.pdf (accessed on 5 June 2015).
- The Montreal Protocol on Substances that Deplete the Ozone Layer. Available online: http://ozone.unep.org/new_site/en/montreal_protocol.php (accessed on 5 June 2015).
- Virent Makes Gasoline from Cellulosic Biomass. Available online: http://www.virent.com/news/virent-makes-gasoline-from-cellulosic-biomass (accessed on 29 May 2015).
- Chheda, J.N.; Huber, G.W.; Dumesic, J.A. Liquid-phase catalytic processing of biomass-derived oxygenated hydrocarbons to fuels and chemicals. Angew. Chem. Int. Ed. 2007, 46, 7164–7183. [Google Scholar] [CrossRef] [PubMed]
- Davda, R.R.; Shabaker, J.W.; Huber, G.W.; Cortright, R.D.; Dumesic, J.A. A review of catalytic issues and process conditions for renewable hydrogen and alkanes by aqueous-phase reforming of oxygenated hydrocarbons over supported metal catalysts. Appl. Catal. B: Environ. 2005, 56, 171–186. [Google Scholar] [CrossRef]
- NEXBTL Renewable Diesel. Available online: http://www.nesteoil.com/default.asp?path=1,41,11991,22708,22709,22710 (accessed on 25 May 2015).
- Amyris Farnesene. Available online: http://farnesene.net/ (accessed on 25 May 2015).
- Sacia, E.R.; Deaner, M.H.; Louie, Y.; Bell, A.T. Synthesis of biomass-derived methylcyclopentane as a gasoline additive via aldol condensation/hydrodeoxygenation of 2,5-hexanedione. Green Chem. 2015, 17, 2393–2397. [Google Scholar] [CrossRef]
- Kim, J.K.; Han, G.H.; Oh, B.R.; Chun, Y.N.; Eom, C.Y.; Kim, S.W. Volumetric scale-up of a three stage fermentation system for food waste treatment. Bioresour. Technol. 2008, 99, 4394–4399. [Google Scholar] [CrossRef] [PubMed]
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.K.; Pfaltzgraff, L.A.; Herrero-Davila, L.; Mubofu, E.B.; Abderrahim, S.; Clark, J.H.; Koutinas, A.A.; Kopsahelis, N.; Stamatelatou, K.; Dickson, F.; et al. Food waste as a valuable resource for the production of chemicals, materials and fuels current situation and global perspective. Energy Environ. Sci. 2013, 6, 426–464. [Google Scholar] [CrossRef]
- Food Waste. Available online: http://ec.europa.eu/food/safety/food_waste/index_en.htm (accessed on 15 May 2015).
- EU Actions Against Food Waste. Available online: http://ec.europa.eu/food/safety/food_waste/eu_actions/index_en.htm (accessed on 15 May 2015).
- Zhang, B.; Zhong, Z.; Min, M.; Ding, K.; Xie, Q.; Ruan, R. Catalytic fast co-pyrolysis of biomass and food waste to produce aromatics: Analytical Py–GC/MS study. Bioresour. Technol. 2015, 189, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Kondamudi, N.; Mohapatra, S.K.; Misra, M. Spent coffee grounds as a versatile source of green energy. J. Agric. Food Chem. 2008, 56, 11757–11760. [Google Scholar] [CrossRef] [PubMed]
- López, J.A.S.; Li, Q.; Thompson, I.P. Biorefinery of waste orange peel. Crit. Rev. Biotechnol. 2010, 30, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Li, S.; Yuan, H.; Zou, D.; Liu, Y.; Zhu, B.; Chufo, A.; Jaffar, M.; Li, X. Evaluating biomethane production from anaerobic mono- and co-digestion of food waste and floatable oil (FO) skimmed from food waste. Bioresour. Technol. 2015, 185, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Su, H.; Wang, Z.; Tan, T.; Qin, P. Biogas by semi-continuous anaerobic digestion of food waste. Appl. Biochem. Biotechnol. 2015, 175, 3901–3914. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Liu, D.N.; Shi, Y.W.; Tang, J.H.; Li, Y.F.; Ren, N.Q. Biohydrogen production from food waste hydrolysate using continuous mixed immobilized sludge reactors. Bioresour. Technol. 2015, 180, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Qureshi, N.; Chen, M.H.; Liu, W.; Singh, V. Ethanol production from food waste at high solids content with vacuum recovery technology. J. Agric. Food Chem. 2015, 63, 2760–2766. [Google Scholar] [CrossRef] [PubMed]
- Dahiya, S.; Sarkar, O.; Swamy, Y.V.; Mohan, S.V. Acidogenic fermentation of food waste for volatile fatty acid production with co-generation of biohydrogen. Bioresour. Technol. 2015, 182, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, Y.; Zhao, S.; Chen, H.; Zheng, X.; Luo, J.; Liu, Y. Efficient production of optically pure L-lactic acid from food waste at ambient temperature by regulating key enzyme activity. Water Res. 2015, 70, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Parshetti, G.K.; Suryadharma, M.S.; Pham, T.P.T.; Mahmood, R.; Balasubramanian, R. Heterogeneous catalyst-assisted thermochemical conversion of food waste biomass into 5-hydroxymethylfurfural. Bioresour. Technol. 2015, 178, 19–27. [Google Scholar] [CrossRef] [PubMed]
- How Is Gas Extracted from the Ground? Available online: http://www.ukoog.org.uk/knowledge-base/drilling-process-kb/how-is-gas-extracted-from-the-ground (accessed on 16 June 2015).
- Falling oil Prices Reveal America’s Fracking Trap and Saudi Arabia’s Continued Energy Dominance. Available online: http://www.huffingtonpost.com/alexis-crow/america-fracking-saudi-oil_b_6091942.html (accessed on 16 June 2015).
- UK Biogas Plant Map. Available online: http://www.biogas-info.co.uk/maps/index2.htm (accessed on 15 May 2015).
- EurObserv’ER Biogas Barometer 2014. Available online: http://www.eurobserv-er.org/biogas-barometer-2014 (accessed on 15 May 2015).
- Gunaseelan, V.N. Anaerobic digestion of biomass for methane production: A review. Biomass Bioenergy 1997, 13, 83–114. [Google Scholar] [CrossRef]
- An Introduction to Anaerobic Digestion of Organic Wastes. Available online: http://www.biogasmax.co.uk/media/introanaerobicdigestion__073323000_1011_24042007.pdf (accessed on 15 May 2015).
- Subramani, V.; Sharma, P.; Zhang, L.; Liu, K. Catalytic steam reforming technology for the production of hydrogen and syngas. In Hydrogen and Syngas Production and Purification Technologies; Liu, K., Song, C., Subramani, V., Eds.; John Wiley and Sons: Hoboken, NY, USA, 2010; pp. 14–126. [Google Scholar]
- Rostrup-Nielsen, J.; Christiansen, L.J. Concepts in Syngas Manufacture; Imperial College Press: London, UK, 2011; pp. 3–71. [Google Scholar]
- Kamm, B. Production of platform chemicals and synthesis gas from biomass. Angew. Chem. Int. Ed. 2007, 46, 5056–5058. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Li, X.; Fan, J.; Chang, J. Hydrothermal conversion of lignin a review. Renew. Sustain. Energy Rev. 2013, 27, 546–558. [Google Scholar] [CrossRef]
- Chen, T.; Wu, J.; Zhang, J.; Wu, J.; Sun, L. Gasification kinetic analysis of the three pseudocomponents of biomass-cellulose semicellulose and lignin. Bioresour. Technol. 2014, 153, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Consonni, S.; Katofsky, R.E.; Larson, E.D. A gasification-based biorefinery for the pulp and paper industry. Chem. Eng. Res. Des. 2009, 87, 1293–1317. [Google Scholar] [CrossRef]
- Hunt, A.J.; Sin, E.H.K.; Marriott, R.; Clark, J.H. Generation, capture, and utilization of industrial carbon dioxide. ChemSusChem 2010, 3, 306–322. [Google Scholar] [CrossRef] [PubMed]
- Reverchon, E.; de Marco, I. Supercritical fluid extraction and fractionation of natural matter. J. Supercrit. Fluid. 2006, 38, 146–166. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L.; Schlegel, V.L.; Carr, T.P.; Cuppett, S.L. Comparison of supercritical CO2 and hexane extraction of lipids from sorghum distillers grains. Eur. J. Lipid Sci. Technol. 2007, 109, 567–574. [Google Scholar] [CrossRef]
- Jung, G.W.; Kang, H.M.; Chun, B.S. Characterization of wheat bran oil obtained by supercritical carbon dioxide and hexane extraction. J. Ind. Eng. Chem. 2012, 18, 360–363. [Google Scholar] [CrossRef]
- Weissermel, K; Arpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH: Weinheim, Germany, 1993; pp. 13–30. [Google Scholar]
- Weissermel, K; Arpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH: Weinheim, Germany, 1993; p. 27. [Google Scholar]
- A Restriction of Methanol under REACH Has Been Proposed. Available online: http://echa.europa.eu/documents/10162/13641/annex_xv_methanol_en.pdf (accessed on 29 May 2015).
- Production of Bio-Methanol IEA-ETSAP and IRENA© Technology Brief. Available online: https://www.irena.org/DocumentDownloads/Publications/IRENA-ETSAP%20Tech%20Brief%20I08%20Production_of_Bio-methanol.pdf (accessed on 21 July 2015).
- Varişli, D.; Tokay, K.C.; Çiftçi, A.; Doğu, T.; Doğu, G. Methanol dehydration reaction to produce clean diesel alternative dimethylether over mesoporous aluminosilicate-based catalysts. Turk. J. Chem. 2009, 33, 355–366. [Google Scholar]
- Fukaya, M.; Park, Y.S.; Toda, K. Improvement of acetic acid fermentation by molecular breeding and process development. J. Appl. Bacteriol. 1992, 73, 447–454. [Google Scholar] [CrossRef]
- Khan, A.W.; Wall, D.; van den Berg, L. Fermentative bonversion of bellulose to acetic acid and cellulolytic enzyme production by a bacterial mixed culture obtained from sewage sludge. Appl. Environ. Microb. 1981, 41, 1214–1218. [Google Scholar]
- Huijbregts, M.A.J.; Hellweg, S.; Frischknecht, R.; Hendriks, H.W.M.; Hungerbühler, K.; Hendriks, A.J. Cumulative energy demand as predictor for the environmental burden of commodity production. Environ. Sci. Technol. 2010, 44, 2189–2196. [Google Scholar] [CrossRef] [PubMed]
- Capello, C.; Wernet, G.; Sutter, J.; Hellweg, S.; Hungerbühler, K. A comprehensive environmental assessment of petrochemical solvent production. Int. J. Life Cycle Assess. 2009, 14, 467–479. [Google Scholar] [CrossRef]
- The Eco-Profiles for Current and Near-Future NatureWorks® Polylactide (PLA) Production. Available online: http://www.natureworksllc.com/~/media/The_Ingeo_Journey/EcoProfile_LCA/EcoProfile/NTR_Eco_Profile_Industrial_Biotechnology_032007_pdf.pdf?la=en (accessed on 1 June 2015).
- Weissermel, K; Arpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH: Weinheim, Germany, 1993; pp. 177–179. [Google Scholar]
- Sánchez, A.B.; Homs, N.; Miachon, S.; Dalmon, J.A.; Fierrod, J.L.G.; de la Piscina, P.R. Direct transformation of ethanol into ethyl acetate through catalytic membranes containing Pd or Pd-Zn: Comparison with conventional supported catalysts. Green Chem. 2011, 13, 2569–2575. [Google Scholar] [CrossRef]
- Santacesaria, E.; Di, S.M.; Tesser, R.; Carotenuto, G. Process for the Production of Ethyl-Acetate from Ethanol. World Patent 104738 A2, 1 September 2011. [Google Scholar]
- Parker, H.L.; Sherwood, J.; Hunt, A.J.; Clark, J.H. Cyclic carbonates as green alternative solvents for the heck reaction. ACS Sustain. Chem. Eng. 2014, 2, 1739–1742. [Google Scholar] [CrossRef]
- Yim, H.; Haselbeck, R.; Niu, W.; Pujol-Baxley, C.; Burgard, A.; Boldt, J.; Khandurina, J.; Trawick, J.D.; Osterhout, R.E.; Stephen, R.; et al. Metabolic engineering of Escherichia coli for direct production of 1,4-butanediol. Nat. Chem. Biol. 2011, 7, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Kantchev, E.A.B.; Norsten, T.B.; Tan, M.L.Y.; Ng, J.J.Y.; Sullivan, M.B. Thiophene-containing Pechmann dyes and related compounds synthesis and experimental and DFT characterisation. Chem. Eur. J. 2012, 18, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Norsten, T.B.; Kantchev, E.A.B.; Sullivan, M.B. Thiophene-containing Pechmann dye derivatives. Org. Lett. 2010, 12, 4816–4819. [Google Scholar] [CrossRef] [PubMed]
- Prior, T.J.; Rosseinsky, M.J. Chiral direction and interconnection of helical three-connected networks in metal-organic frameworks. Inorg. Chem. 2003, 42, 1564–1575. [Google Scholar] [CrossRef] [PubMed]
- Far Eastern New Century Showcases Apparel with High Renewable Content (Genomatica Press Release). Available online: http://www.genomatica.com/news/press-releases/far-eastern-new-century-showcases-apparel-with-high-renewable-content (accessed on 29 May 2015).
- Toray Succeeds in Production of Bio-Based PBT and Part Samples (Genomatica Press Release). Available online: http://www.genomatica.com/news/press-releases/toray-successfully-produces-bio-based-pbt-and-part-samples (accessed on 29 May 2015).
- BioAmber Succinic Acid. Available online: http://www.bio-amber.com/bioamber/en/products (accessed on 15 May 2015).
- Lammens, T.M.; Franssen, M.C.R.; Scott, E.L.; Sanders, J.P.M. Synthesis of biobased N-methylpyrrolidone by one-pot cyclization and methylation of γ-aminobutyric acid. Green Chem. 2010, 12, 1430–1436. [Google Scholar] [CrossRef]
- Lammens, T.M.; Potting, J.; Sanders, J.P.M.; de Boer, I.J.M. Environmental comparison of biobased chemicals from glutamic acid with their petrochemical equivalents. Environ. Sci. Technol. 2011, 45, 8521–8528. [Google Scholar] [CrossRef] [PubMed]
- Lammens, T.M.; Gangarapu, S.; Franssen, M.C.R.; Scott, E.L.; Sanders, J.P.M. Techno-economic assessment of the production of bio-based chemicals from glutamic acid. Biofuels Bioprod. Biorefining 2012, 6, 177–187. [Google Scholar] [CrossRef]
- De Schouwer, F.; Claes, L.; Claes, N.; Bals, S.; Degrèvec, J.; de Vos, D.E. Pd-catalyzed decarboxylation of glutamic acid and pyroglutamic acid to bio-based 2-pyrrolidone. Green Chem. 2015, 17, 2263–2270. [Google Scholar] [CrossRef]
- Sanders, J.; Scott, E.; Weusthuis, R.; Mooibroek, H. Bio-refinery as the bio-inspired process to bulk chemicals. Macromol. Biosci. 2007, 7, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Mosier, N.S.; Hendrickson, R.; Ezeji, T.; Blaschek, H.; Dien, B.; Cotta, M.; Dale, B.; Ladisch, M.R. Composition of corn dry-grind ethanol by-products DDGS wet cake and thin stillage. Bioresour. Technol. 2008, 99, 5165–5176. [Google Scholar] [CrossRef] [PubMed]
- Metts, L.S.; Rawles, S.D.; Brady, Y.J.; Thompson, K.R.; Gannam, A.L.; Twibell, R.G.; Webster, C.D. Amino acid availability from selected animal and plant derived feedstuffs for market-size sunshine bass (Morone chrysops × Morone saxatilis). Aquac. Nutr. 2011, 17, 123–131. [Google Scholar] [CrossRef]
- Pahm, A.A.; Pedersen, C.; Hoehler, D.; Stein, H.H. Factors affecting the variability in ileal amino acid digestibility in corn distillers dried grains with solubles fed to growing pigs. J. Anim. Sci. 2008, 86, 2180–2189. [Google Scholar] [CrossRef] [PubMed]
- Scott, E.; Peter, F.; Sanders, J. Biomass in the manufacture of industrial products-the use of proteins and amino acids. Appl. Microbiol. Biotechnol. 2007, 75, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Ledoux, A.; Kuigwa, L.S.; Framery, E.; Andrioletti, B. A highly sustainable route to pyrrolidone derivatives—Direct access to biosourced solvents. Green Chem. 2015, 17, 3251–3254. [Google Scholar] [CrossRef]
- TamiSolveNxG. Available online: http://tamisolvenxg.com (accessed on 15 May 2015).
- Circa Cyrene. Available online: http://circagroup.com.au/cyrene (accessed on 15 May 2015).
- Aycock, D.F. Solvent applications of 2-methyltetrahydrofuran in organometallic and biphasic reactions. Org. Process Res. Dev. 2007, 11, 156–159. [Google Scholar] [CrossRef]
- China Steel Green-Lights Commercial-Scale LanzaTech Advanced Biofuels Project. Available online: http://www.biofuelsdigest.com/bdigest/2015/04/22/china-steel-green-lights-46m-for-commercial-scale-lanzatech-advanced-biofuels-project (accessed on 25 May 2015).
- Phillips, J.R.; Atiyeh, H.K.; Tanner, R.S.; Torres, J.R.; Saxena, J.; Wilkins, M.R.; Huhnke, R.L. Butanol and hexanol production in Clostridium carboxidivorans syngas fermentation medium development and culture techniques. Bioresour. Technol. 2015, 190, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.K.; Dandapani, H.; Thiel, K.; Jones, P.R. Microbial production of 1-octanol a naturally excreted biofuel with diesel-like properties. Metab. Eng. Commun. 2015, 2, 1–5. [Google Scholar] [CrossRef]
- Das, D.; Veziroǧlu, T.N. Hydrogen production by biological processes: A survey of literature. Int. J. Hydrog. Energ. 2001, 26, 13–28. [Google Scholar] [CrossRef]
- Melis, A.; Happe, T. Hydrogen production green algae as a source of energy. Plant Physiol. 2001, 127, 740–748. [Google Scholar] [CrossRef] [PubMed]
- Friendly Mutant Algae Could Churn out Sustainable Hydrogen on Your Desktop. Available online: http://cleantechnica.com/2014/02/11/sustainable-hydrogen-from-friendly-mutant-algae (accessed on 29 May 2015).
- Liu, J.; Liu, Y.; Liu, N.; Han, Y.; Zhang, X.; Huang, H.; Lifshitz, Y.; Lee, S.T.; Zhong, J.; Kang, Z. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 2015, 347, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Khadzhiev, S.N.; Kolesnichenko, N.V.; Ezhova, N.N. Manufacturing of lower olefins from natural gas through methanol and its derivatives (review). Pet. Chem. 2008, 48, 325–334. [Google Scholar]
- Lefevere, J.; Mullens, S.; Meynen, V.; van Noyen, J. Structured catalysts for methanol-to-olefins conversion a review. Chem. Pap. 2014, 68, 1143–1153. [Google Scholar] [CrossRef]
- Albo, J.; Alvarez-Guerra, M.; Castaño, P.; Irabien, A. Towards the electrochemical conversion of carbon dioxide into methanol. Green Chem. 2015, 17, 2304–2324. [Google Scholar] [CrossRef]
- Olah, G.A. Beyond oil and gas: The methanol economy. Angew. Chem. Int. Ed. 2005, 44, 2636–2639. [Google Scholar] [CrossRef] [PubMed]
- Olah, G.A.; Goeppert, A.; Prakash, G.K.S. Chemical recycling of carbon dioxide to methanol and dimethyl ether from greenhouse gas to renewable environmentally carbon neutral fuels and synthetic hydrocarbons. J. Org. Chem. 2009, 74, 487–498. [Google Scholar] [CrossRef] [PubMed]
- State of Play on Biofuel Subsidies: Are Policies Ready to Shift? Available online: http:www.iisd.org/gsi/sites/default/files/bf_stateplay_2012.pdf (accessed on 29 May 2015).
- Direct Federal Financial Interventions and Subsidies in Energy in Fiscal Year 2013. Available online: http://www.eia.gov/analysis/requests/subsidy/pdf/subsidy.pdf (accessed on 29 May 2015).
- Tracking Progress: International Cooperation to Reform Fossil-Fuel Subsidies. Available online: http://www.iisd.org/GSI/tracking-progress-g-20-and-apec-commitments-reform (accessed on 29 May 2015).
- India Changes Ethanol Policy Adopts Fixed Price to Reach 5% Blending Target. Available online: http://www.platts.com/latest-news/agriculture/mumbai/india-changes-ethanol-policy-adopts-fixed-price-27923784 (accessed on 8 June 2015).
- The Brazilian Ethanol Programme Impacts on World Ethanol and Sugar Markets. Available online: http://www.fao.org/docrep/006/ad430e/ad430e00.htm (accessed on 8 June 2015).
- European Commission Renewable Energy National Action Plans. Available online: http://ec.europa.eu/energy/en/topics/renewable-energy/national-action-plans (accessed on 8 June 2015).
- Production of Bio-Ethylene IEA-ETSAP and IRENA© Technology Brief. Available online: http://www.irena.org/DocumentDownloads/Publications/IRENA-ETSAP%20Tech%20Brief%20I13%20Production_of_Bio-ethylene.pdf (accessed on 29 May 2015).
- Braskem Green Products. Available online: http://www.braskem.com/site.aspx/green-products-USA (accessed on 15 May 2015).
- Plantbottle. Available online: http://www.coca-colacompany.com/plantbottle-technology/plantbottle (accessed on 15 May 2015).
- Oil (Brent) Price Index. Available online: http://www.quotenet.com/commodities/oil-price (accessed on 29 May 2015).
- Food and Agriculture Organisation of the United Nations Production Data. Available online: http://faostat3.fao.org/browse/Q/*/E (accessed on 29 May 2015).
- Shale Gas Reshaping the US Chemicals Industry. Available online: http://www.pwc.com/en_US/us/industrial-products/publications/assets/pwc-shale-gas-chemicals-industry-potential.pdf (accessed on 29 May 2015).
- Haer, T. Environmental, Social and Economic Sustainability of Biobased Plastics Bio-polyethylene from Brazil and polylactic acid from the U.S. PhD Thesis, University of Groningen, The Netherlands, 2012. [Google Scholar]
- Naphtha (European) Price Index. Available online: http://www.quotenet.com/commodities/naphtha (accessed on 29 May 2015).
- Platts Global Ethylene Price index. Available online: http://www.platts.com/news-feature/2015/petrochemicals/pgpi/ethylene (accessed on 29 May 2015).
- Platts Global Polyethylene Price Index. Available online: http://www.platts.com/news-feature/2015/petrochemicals/pgpi/ldpe (accessed on 29 May 2015).
- The Impact of Falling Crude Oil Prices on Chemical Building Blocks. Available online: http://www.orbichem.com/userfiles/Presentations/ICDC2015_Charles_Fryer_TOC%20SR%20130215.pdf (accessed on 29 May 2015).
- European Bioplastics Market. Available online: http://en.european-bioplastics.org/market (accessed on 29 May 2015).
- Selley, R.C. UK shale gas: The story so far. Mar. Petrol. Geol. 2012, 31, 100–109. [Google Scholar] [CrossRef]
- Weijermars, R. Economic appraisal of shale gas plays in Continental Europe. Appl. Energ. 2013, 106, 100–115. [Google Scholar] [CrossRef]
- Shale Gas Measurement and Associated Issues. Available online: http://www.pipelineandgasjournal.com/shale-gas-measurement-and-associated-issues (accessed on 29 May 2015).
- Platts Special Report, Petrochemicals: Can Shale Gas Save the Naphtha Crackers? Available online: http://www.platts.com/IM.Platts.Content/InsightAnalysis/IndustrySolutionPapers/ShaleGasReport13.pdf (accessed on 15 May 2015).
- Seaborne Ethane a Report into the Commercial Need and Technical Requirements for Very Large Ethane Carriers. Available online: http://www.lr.org/en/_images/213-42424_Seaborne_ethane.pdf (accessed on 8 June 2015).
- PlantBottle™ Packaging. Available online: http://www.coca-cola.co.uk/videos/plant-bottle-packaging-bc1944837204001 (accessed on 29 May 2015).
- Platts Shale Gas Market Report. Available online: http://www.platts.com/IM.Platts.Content/InsightAnalysis/IndustrySolutionPapers/ShaleGasReport13.pdf (accessed on 29 May 2015).
- Van duuren, J.B.J.H.; Brehmer, B.; Mars, A.E.; Eggink, G.; Martins dos Santos, V.A.P.; Sanders, J.P.M. A limited LCA of bio-adipic acid manufacturing the nylon-6,6 precursor adipic acid using the benzoic acid degradation pathway from different feedstocks. Biotechnol. Bioeng. 2011, 108, 1298–1306. [Google Scholar] [CrossRef] [PubMed]
- Cherubini, F.; Jungmeier, G. LCA of a biorefinery concept producing bioethanol, bioenergy, and chemicals from switchgrass. Int. J. Life Cycle Assess. 2010, 15, 53–66. [Google Scholar] [CrossRef]
- Eckert, C.; Xu, W.; Xiong, W.; Lynch, S.; Ungerer, J.; Tao, L.; Gill, R.; Maness, P.C.; Yu, J. Ethylene-forming enzyme and bioethylene production. Biotechnol. Biofuels 2014, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.T.; Yu, K.M.K.; Liao, F.; Young, N.; Nellist, P.; Dent, A.; Kroner, A.; Tsang, S.C.E. A non-syn-gas catalytic route to methanol production. Nat. Commun. 2012, 3, 1050. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.T.; Qu, J.; Elliott, J.; Yu, K.M.K.; Tsang, S.C.E. Hydrogenolysis of ethylene glycol to methanol over modified RANEY® catalysts. Phys. Chem. Chem. Phys. 2013, 15, 9043–9050. [Google Scholar] [CrossRef] [PubMed]
- Aricò, F.; Tundo, P. Dimethyl carbonate: A modern green reagent and solvent. Russ. Chem Rev. 2010, 79, 479–489. [Google Scholar] [CrossRef]
- BASF Diethylamine. Available online: http://product-finder.basf.com/group/corporate/product-finder/en/brand/DIETHYLAMINE_ANHYDROUS (accessed on 1 June 2015).
- Lambiotte&CIE Ethylal. Available online: http://www.agrobiobase.com/en/database/bioproducts/detergency/ethylal (accessed on 1 June 2015).
- Inokuma, K.; Liao, J.C.; Okamoto, M.; Hanai, T. Improvement of isopropanol production by metabolically engineered Escherichia coli using gas stripping. J. Biosci. Bioeng. 2010, 110, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, B.A.; Stowers, C.C.; Pham, V.; Cox, B.M. The production of propionic acid propanol and propylene via sugar fermentation an industrial perspective on the progress, technical challenges and future outlook. Green Chem. 2014, 16, 1066–1076. [Google Scholar] [CrossRef]
- Direct Biological Production of Propylene Third Star on Global Bioenergies’ Race to Renewable Olefins (Press Release). Available online: http://www.global-bioenergies.com/wp-content/uploads/2015/05/141208_pr_en.pdf (accessed on 4 June 2015).
- Global Bioenergies’ Press Release Site. Available online: http://www.global-bioenergies.com/index.php?option=com_content&view=article&id=117&Itemid=200&lang=en (accessed on 19 May 2015).
- Osterhout, R.E.; Burgard, A.P.; Burk, M.J. Microorganisms and Methods for producing 2,4-Pentadienoate, Butadiene, Propylene, 1,3-Butanediol and Related Alcohols. World Patent 028519 A1, 28 February 2013. [Google Scholar]
- Iwamoto, M.; Kosugi, Y. Highly selective conversion of ethene to propene and butenes on nickel ion-loaded mesoporous silica catalysts. J. Phys. Chem. C 2007, 111, 13–15. [Google Scholar] [CrossRef]
- Andrei, R.D.; Popa, M.I.; Fajula, F.; Cammarano, C.; Al Khudhair, A.; Bouchmella, K.; Mutin, P.H.; Hulea, V. Ethylene to propylene by one-pot catalytic cascade reactions. ACS Catal. 2015, 5, 2774–2777. [Google Scholar] [CrossRef]
- Bio-Based Polypropylene; Multiple Synthetic Routes under Investigation. Available online: http://polymerinnovationblog.com/bio-based-polypropylene-multiple-synthetic-routes-under-investigation (accessed on 15 May 2015).
- ICIS, Bioplastics Surge towards Commercialization. Available online: http://www.icis.com/resources/news/2012/07/02/9573828/bioplastics-surge-towards-commercialization (accessed on 15 May 2015).
- Braskem Green Polypropylene. Available online: http://www3.braskem.com.br/upload/rao/2010/en/green-polypropylene.html (accessed on 22 May 2015).
- Braskem to Make Propylene from Ethanol. Available online: http://cenblog.org/the-chemical-notebook/2010/10/brakem-to-make-propylene-from-ethanol/ (accessed on 22 May 2015).
- Mülhaupt, R. Green polymer chemistry and bio-based plastics: Dreams and reality. Macromol. Chem. Phys. 2013, 214, 159–174. [Google Scholar] [CrossRef]
- Braskem Freezes Green Plastics Plans. Available online: http://www.bnamericas.com/news/petrochemicals/braskem-freezes-green-plastics-plans-focuses-elsewhere (accessed on 22 May 2015).
- Iwamoto, M.; Mizuno, S.; Tanaka, M. Direct and selective production of propene from bio-ethanol on Sc-loaded In2O3 catalysts. Chem. Eur. J. 2013, 19, 7214–7220. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, M. Selective catalytic conversion of bio-ethanol to propene: A review of catalysts and reaction pathways. Catal. Today 2015, 242, 243–248. [Google Scholar] [CrossRef]
- Hayashi, F.; Iwamoto, M. Yttrium-modified ceria as a highly durable catalyst for the selective conversion of ethanol to propene and ethene. ACS Catal. 2013, 3, 14–17. [Google Scholar] [CrossRef]
- Mizuno, S.; Kurosawa, M.; Tanak, M.; Iwamoto, M. One-path and selective conversion of ethanol to propene on scandium-modified indium oxide catalysts. Chem. Lett. 2012, 41, 892–894. [Google Scholar] [CrossRef]
- Zhang, Q.; Hu, S.; Zhang, L.; Wu, Z.; Gong, Y.; Dou, T. Facile fabrication of mesopore-containing ZSM-5 zeolite from spent zeolite catalyst for methanol to propylene reaction. Green Chem. 2014, 16, 77–81. [Google Scholar] [CrossRef]
- Neste Oil Biopropane Press Release. Available online: http://www.nesteoil.com/default.asp?path=1,41,540,17988,7906,24702 (accessed on 29 May 2015).
- Neste Oil’s Biopropane Will Be Sold to SHV Energy (Press Release). Available online: http://www.nesteoil.com/default.asp?path=1,41,540,1259,1260,22862,24388 (accessed on 29 May 2015).
- Schweitzer, N.M.; Hu, B.; Das, U.; Kim, H.; Greeley, J.; Curtiss, L.A.; Stair, P.C.; Miller, J.T.; Hock, A.S. Propylene hydrogenation and propane dehydrogenation by a single-site Zn2+ on silica catalyst. ACS Catal. 2014, 4, 1091–1098. [Google Scholar] [CrossRef]
- Dijkmans, T.; Pyl, S.P.; Reyniers, M.F.; Abhari, R.; van Geem, K.M.; Marin, G.B. Production of bio-ethene and propene: Alternatives for bulk chemicals and polymers. Green Chem. 2013, 15, 3064–3076. [Google Scholar] [CrossRef]
- Total Petrochemicals Methanol to Olefins. Available online: http://www.totalrefiningchemicals.com/EN/aboutus/understand_petrochemicals/Pages/Methanol_To_Olefins.aspx (accessed on 8 June 2015).
- Vermeiren, W.; Adam, C.; Minoux, D. Production of Propylene via Simultaneous Dehydration and Skeletal Isomerisation of Isobutanol on Acid Catalysts Followed by Metathesis. World Patent 113836 A1, 22 September 2011. [Google Scholar]
- Solvent Substitution or Replacment Options for MIBK. Available online: http://www.eastman.com/Literature_Center/T/TT127.pdf (accessed on 8 June 2015).
- Zhu, S.; Zhu, Y.; Hao, S.; Zheng, H.; Moa, T.; Li, Y. One-step hydrogenolysis of glycerol to biopropanols over Pt–H4SiW12O40/ZrO2 catalysts. Green Chem. 2012, 14, 2607–2616. [Google Scholar] [CrossRef]
- Martin-Luengo, M.A.; Yates, M.; Rojo, E.S.; Arribas, D.H.; Aguilar, D.; Hitzky, E.R. Sustainable p-cymene and hydrogen from limonene. Appl. Catal. A: Gen. 2010, 387, 141–146. [Google Scholar] [CrossRef]
- Weissermel, K; Arpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH: Weinheim, Germany, 1993; pp. 276–277. [Google Scholar]
- Birkhoff, R.; Bhoomi, R. Integrated Process for Producing Cumene and Purifying Isopropanol. World Patent 028003 A1, 20 February 2014. [Google Scholar]
- Brettschneider, F.; Jankowski, V.; Günthner, T.; Salem, S.; Nierhaus, M.; Schulz, A.; Zidek, W.; Jankowski, J. Replacement of acetonitrile by ethanol as solvent in reversed phase chromatography of biomolecules. J. Chromatogr. B 2010, 878, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Weissermel, K.; Arpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH: Weinheim, Germany, 1993; pp. 301–302. [Google Scholar]
- McConvey, I.F.; Woods, D.; Lewis, M.; Gan, Q.; Nancarrow, P. The importance of acetonitrile in the pharmaceutical industry and opportunities for its recovery from waste. Org. Process Res. Dev. 2012, 16, 612–624. [Google Scholar] [CrossRef]
- Desai, A.M.; Andreae, M.; Mullen, D.G.; Banaszak Holl, M.M.; Baker, J.R., Jr. Acetonitrile shortage: Use of isopropanol as an alternative elution system for ultra/high performance liquid chromatography. Anal. Methods 2011, 3, 56–58. [Google Scholar] [CrossRef] [PubMed]
- Corker, E.C.; Mentzel, U.V.; Mielby, J.; Riisager, A.; Fehrmann, R. An alternative pathway for production of acetonitrile: Ruthenium catalysed aerobic dehydrogenation of ethylamine. Green Chem. 2013, 15, 928–933. [Google Scholar] [CrossRef]
- Kohlpaintner, C.W.; Fischer, R.W.; Cornils, B. Aqueous biphasic catalysis Ruhrchemie/Rhône-Poulenc oxo process. Appl. Catal. A: Gen. 2001, 221, 219–225. [Google Scholar] [CrossRef]
- Cornils, B. Industrial aqueous biphasic catalysis status and directions. Org. Proc. Res. Dev. 1998, 2, 121–127. [Google Scholar] [CrossRef]
- EBTP Biobutanol. Available online: http://biofuelstp.eu/butanol.html (accessed on 1 June 2015).
- Uyttebroek, M.; van Hecke, W.; Vanbroekhoven, K. Sustainability metrics of 1-butanol. Catal. Today 2015, 239, 7–10. [Google Scholar] [CrossRef]
- Butanol’s High Value Markets. Available online: http://www.cobalttech.com/biobutanol.html (accessed on 1 June 2015).
- Ciriminna, R.; Pina, C.D.; Rossi, M.; Pagliaro, M. Understanding the glycerol market. Eur. J. Lipid Sci. Technol. 2014, 116, 1432–1439. [Google Scholar] [CrossRef]
- Zacharopoulou, V.; Vasiliadoua, E.S.; Lemonidou, A.A. One-step propylene formation from bio-glycerol over molybdena-based catalysts. Green Chem. 2015, 17, 903–912. [Google Scholar] [CrossRef]
- Pagliaro, M.; Ciriminna, R.; Kimura, H.; Rossi, M.; Pina, C.D. From glycerol to value-added products. Angew. Chem. Int. Ed. 2007, 46, 4434–4440. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.H.; Beltramini, J.N.; Fan, Y.X.; Lu, G.Q. Chemoselective catalytic conversion of glycerol as a biorenewable source to valuable commodity chemicals. Chem. Soc. Rev. 2008, 37, 527–549. [Google Scholar] [CrossRef] [PubMed]
- Ten Dam, J.; Hanefeld, U. Renewable Chemicals: Dehydroxylation of glycerol and polyols. ChemSusChem 2011, 4, 1017–1034. [Google Scholar] [CrossRef] [PubMed]
- Marinas, A.; Bruijnincx, P.; Ftouni, J.; Urbano, F.J.; Pinel, C. Sustainability metrics for a fossil- and renewable-based route for 1,2-propanediol production: A comparison. Catal. Today 2015, 239, 31–37. [Google Scholar] [CrossRef]
- Posada, J.A.; Rincón, L.E.; Cardona, C.A. Design and analysis of biorefineries based on raw glycerol. Addressing the glycerol problem. Bioresour. Technol. 2012, 111, 282–293. [Google Scholar] [CrossRef] [PubMed]
- 1,3-Propanediol Fermentation Process. Available online: http://www2.dupont.com/Renewably_Sourced_Materials/en_US/proc-buildingblocks.html (accessed on 29 May 2015).
- Comerford, J.W.; Ingram, I.D.V.; North, M.; Wu, X. Sustainable metal-based catalysts for the synthesis of cyclic carbonates containing five-membered rings. Green Chem. 2015, 17, 1966–1987. [Google Scholar] [CrossRef]
- North, M.; Pasquale, R.; Young, C. Synthesis of cyclic carbonates from epoxides and CO2. Green Chem. 2010, 12, 1514–1539. [Google Scholar] [CrossRef]
- Honda, M.; Tamura, M.; Nakao, K.; Suzuki, K.; Nakagawa, Y.; Tomishige, K. Direct cyclic carbonate synthesis from CO2 and diol over carboxylation/hydration cascade catalyst of CeO2 with 2-cyanopyridine. ACS Catal. 2014, 4, 1893–1896. [Google Scholar] [CrossRef]
- Gao, Z.W.; Wang, S.F.; Xia, C.G. Synthesis of propylene carbonate from urea and 1,2-propanediol. Chin. Chem. Lett. 2009, 20, 131–135. [Google Scholar] [CrossRef]
- Chemistry World Podcast Ethylene Glycol. Available online: http://www.rsc.org/chemistryworld/podcast/CIIEcompounds/transcripts/ethyleneglycol.asp (accessed on 1 June 2015).
- Davy Propylene Glycol. Available online: http://www.davyprotech.com/what-we-do/licensed-processes-and-core-technologies/licensed-processes/propylene-glycol/specification (accessed on 1 June 2015).
- Panichelli, L.; Dauriat, A.; Gnansounou, E. Life cycle assessment of soybean-based biodiesel in Argentina for export. Int. J. Life Cycle Assess. 2009, 14, 144–159. [Google Scholar] [CrossRef]
- Susterra® 1,3-Propanediol. Available online: http://www.duponttateandlyle.com/susterra (accessed on 29 May 2015).
- DOW Ethylene versus Propylene Glycol. Available online: http://www.dow.com/heattrans/support/selection/ethylene-vs-propylene.htm (accessed on 4 June 2015).
- Díaz-Álvarez, A.E.; Francos, J.; Lastra-Barreira, B.; Crocheta, P.; Cadierno, V. Glycerol and derived solvents: New sustainable reaction media for organic synthesis. Chem. Commun. 2011, 47, 6208–6227. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Jérôme, F. Glycerol as a sustainable solvent for green chemistry. Green Chem. 2010, 12, 1127–1138. [Google Scholar] [CrossRef]
- Agrobiobase Glycerol Formal. Available online: http://www.agrobiobase.com/en/database/bioproducts/chemistry-formulation-synthesis/glycerol-formal (accessed on 15 May 2015).
- Moity, L.; Benazzouz, A.; Molinier, V.; Nardello-Rataj, V.; Elmkaddem, M.K.; de Caro, P.; Thiébaud-Roux, S.; Gerbaud, V.; Marione, P.; Aubry, J.M. Glycerol acetals and ketals as bio-based solvents: Positioning in Hansen and COSMO-RS spaces, volatility and stability towards hydrolysis and autoxidation. Green Chem. 2015, 17, 1779–1792. [Google Scholar] [CrossRef]
- Fiume, M.Z. Final report on the safety assessment of triacetin. Int. J. Toxicol. 2003, 22, 1–10. [Google Scholar] [PubMed]
- Groot, W.; van Krieken, J.; Sliekersl, O.; de Vos, S. Production and purification of lactic acid and lactide. In Poly(lactic acid) Synthesis Structures Properties Processing and Applications; Auras, L., Selke, T., Eds.; John Wiley & Sons: Hoboken, NJ, USA; pp. 1–18.
- Yang, J.; Tan, J.N.; Gu, Y. Lactic acid as an invaluable bio-based solvent for organic reactions. Green Chem. 2012, 14, 3304–3317. [Google Scholar] [CrossRef]
- Galactic Ethyl Lactate. Available online: http://www.lactic.com/en-us/13_ethyl-lactate/index.html (accessed on 15 May 2015).
- Cellulac Ethyl Lactate. Available online: http://cellulac.co.uk/en/ethyl-lactate (accessed on 15 May 2015).
- Vertec Ethyl Lactate. Available online: http://www.vertecbiosolvents.com/EL.htm (accessed on 1 June 2015).
- Pereira, C.S.M.; Silva, V.M.T.M.; Rodrigues, A.E. Ethyl lactate as a solvent properties applications and production processes a review. Green Chem. 2011, 13, 2658–2671. [Google Scholar] [CrossRef]
- Simonov, M.N.; Simakova, I.L.; Parmon, V.N. Hydrogenation of lactic acid to propylene glycol over copper-containing catalysts. React. Kinet. Catal. Lett. 2009, 97, 157–162. [Google Scholar] [CrossRef]
- Jang, H.; Kim, S.H.; Lee, D.; Shim, S.E.; Baeck, S.H.; Kim, B.S.; Chang, T.S. Hydrogenation of lactic acid to propylene glycol over a carbon-supported ruthenium catalyst. J. Mol. Catal. A: Chem. 2013, 380, 57–60. [Google Scholar] [CrossRef]
- Blombach, B.; Riester, T.; Wieschalka, S.; Ziert, C.; Youn, J.W.; Wendisch, V.F.; Eikmanns, B.J. Corynebacterium glutamicum tailored for efficient isobutanol production. Appl. Environ. Microb. 2011, 77, 3300–3310. [Google Scholar] [CrossRef] [PubMed]
- Peters, M.W.; Taylor, J.D.; Jenni, M.; Manzer, L.E.; Henton, D.E. Integrated Process to Selectivity Convert Renewable Isobutanol to p-Xylene. US Patent 0087000 A1, 14 April 2011. [Google Scholar]
- Van Leeuwen, B.N.M.; van der Wulp, A.M.; Duijnstee, I.; van Maris, A.J.A.; Straathof, A.J.J. Fermentative production of isobutene. Appl. Microbiol. Biotechnol. 2012, 93, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- Global Bioenergies Reports First Isobutene Production from Waste Biomass (Press Release). Available online: http://www.global-bioenergies.com/communiques/20150303_pr_en.pdf (accessed on 19 May 2015).
- Weissermel, K; Arpe, H.J. Industrial Organic Chemistry, 2nd ed.; VCH: Weinheim, Germany, 1993; p. 73. [Google Scholar]
- Johnson, R.; Pankow, J.; Bender, D.; Price, C.; Zogorski, J. MTBE to what extent will past releases contaminate community water supply wells? Environ. Sci. Technol. 2000, 34, 210A–217A. [Google Scholar] [CrossRef] [PubMed]
- Squillace, P.J.; Pankow, J.F.; Korte, N.E.; Zogorski, J.S. Review of the environmental behavior and fate of methyl tert-butyl ether. Environ. Toxicol. Chem. 1997, 16, 1836–1844. [Google Scholar] [CrossRef]
- Braskem Ethyl Tertiary-Butyl Ether. Available online: http://www.braskem.com/site.aspx/Ethyl-Tertiary-Butyl-Ether (accessed on 18 May 2015).
- Ghiaci, P.; Norbeck, J.; Larsson, C. 2-Butanol and butanone production in saccharomyces cerevisiae through combination of a B12 dependent dehydratase and a secondary alcohol dehydrogenase using a TEV-based expression system. PLoS ONE 2014, 9, 102774. [Google Scholar] [CrossRef] [PubMed]
- Green Chemicals Blog, Levulinic Acid Commercialization Expands. Available online: http://greenchemicalsblog.com/2015/03/05/levulinic-acid-commercialization-expands/ (accessed on 18 May 2015).
- Zhao, R.; Cabezas, H.; Nishtala, S.R. The design of technologically effective and environmentally benign solvent substitutes. In Green Chemical Syntheses and Processes; Anastas, P.T., Heine, L.G., Williamson, T.C., Eds.; American Chemical Society: Washington, DC, USA, 2000; pp. 230–243. [Google Scholar]
- Angelici, C.; Weckhuysen, B.M.; Bruijnincx, P.C.A. Chemocatalytic conversion of ethanol into butadiene and other bulk chemicals. ChemSusChem 2013, 6, 1595–1614. [Google Scholar] [CrossRef] [PubMed]
- Posada, J.A.; Patel, A.D.; Roes, A.; Blok, K.; Faaij, A.P.C.; Patel, M.K. Potential of bioethanol as a chemical building block for biorefineries: Preliminary sustainability assessment of 12 bioethanol-based products. Bioresour. Technol. 2013, 135, 490–499. [Google Scholar] [CrossRef] [PubMed]
- BioSyntha and ZuvaChem Announce a Merger and Financing to form ZuvaSyntha Ltd. Available online: http://www.nnfcc.co.uk/news/biosyntha-and-zuvachem-announce-a-merger-and-financing-to-form-zuvasyntha-ltd (accessed on 1 June 2015).
- Marliere, P. Production of Volatile Dienes by Enzymatic Dehydration of Light Alkenols. US Patent 8703455 B2, 22 April 2014. [Google Scholar]
- Köpke, M.; Mihalcea, C.; Liew, F.M.; Tizard, J.H.; Ali, M.S.; Conolly, J.J.; Al-Sinawi, B.; Simpson, S.D. 2,3-Butanediol production by acetogenic bacteria, an alternative route to chemical synthesis, using industrial waste gas. Appl. Environ. Microbiol. 2011, 77, 5467–5475. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Yamada, Y.; Sato, S. Efficient production of 1,3-butadiene in the catalytic dehydration of 2,3-butanediol. Appl. Catal. A: Gen. 2015, 491, 163–169. [Google Scholar] [CrossRef]
- Acrylonitrile-Butadiene-Styrene (ABS). Available online: http://www.plasticseurope.org/what-is-plastic/types-of-plastics-11148/engineering-plastics/abs.aspx (accessed on 1 June 2015).
- Huber, G.W.; Cortright, R.D.; Dumesic, J.A. Renewable alkanes by aqueous-phase reforming of biomass-derived oxygenates. Angew. Chem. Int. Ed. 2004, 43, 1549–1551. [Google Scholar] [CrossRef] [PubMed]
- Chheda, J.N.; Dumesic, J.A. An overview of dehydration aldol-condensation and hydrogenation processes for production of liquid alkanes from biomass-derived carbohydrates. Catal. Today 2007, 123, 59–70. [Google Scholar] [CrossRef]
- Pyl, S.P.; Dijkmans, T.; Antonykutty, J.M.; Reyniers, M.F.; Harlin, A.; van Geem, K.M.; Marin, G.B. Wood-derived olefins by steam cracking of hydrodeoxygenated tall oils. Bioresour. Technol. 2012, 126, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Bielansky, P.; Reichhold, A.; Schönberger, C. Catalytic cracking of rapeseed oil to high octane gasoline and olefins. Chem. Eng. Process. 2010, 49, 873–880. [Google Scholar] [CrossRef]
- Bielansky, P.; Weinert, A.; Schönberger, C.; Reichhold, A. Catalytic conversion of vegetable oils in a continuous FCC pilot plant. Fuel Process. Technol. 2011, 92, 2305–2311. [Google Scholar] [CrossRef]
- Note: Toluene is included on the list of restrictions in Annex XVII to REACH EC regulation 1907/2006, relevant to spray paints.
- Eastman Solvents—Performance Sheet: Suggested Replacements for Toluene. Available online: http://www.eastman.com/Literature_Center/T/TT41.pdf (accessed on 29 May 2015).
- Toluene Uses and Market Data. Available online: http://www.icis.com/resources/news/2007/11/07/9076550/toluene-uses-and-market-data (accessed on 29 May 2015).
- The Clean Air Act Amendments of 1990 List of Hazardous Air Pollutants. Available online: http://www.epa.gov/ttn/atw/orig189.html (accessed on 29 May 2015).
- Subsitution Support Portal (SubsPort) Restricted and Priority Substances Database. Available online: http://www.subsport.eu/listoflists (accessed on 29 May 2015).
- Elkington, J. Cannibals with Forks, 1st ed.; Capstone: Oxford, UK, 1999; p. 281. [Google Scholar]
- Eastman Performance Solvents—Technical Tip. Available online: http://www.eastman.com/Literature_Center/T/TT69.pdf (accessed on 29 May 2015).
- Yadav, G.D.; Fernandes, G.P. Selective synthesis of natural benzaldehyde by hydrolysis of cinnamaldehyde using novel hydrotalcite catalyst. Catal. Today 2013, 207, 162–169. [Google Scholar] [CrossRef]
- Liu, S.; Fan, X.; Yan, X.; Du, X.; Chen, L. Catalytic reduction of benzaldehyde to toluene over Ni/γ-Al2O3 in the presence of aniline and H2. Appl. Catal. A: Gen. 2011, 400, 99–103. [Google Scholar] [CrossRef]
- Spekreijse, J.; Le Nôtre, J.; van Haveren, J.; Scott, E.L.; Sanders, J.P.M. Simultaneous production of biobased styrene and acrylates using ethenolysis. Green Chem. 2012, 14, 2747–2751. [Google Scholar] [CrossRef]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The catalytic valorization of lignin for the production of renewable chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef] [PubMed]
- Azadi, P.; Carrasquillo-Flores, R.; Pagán-Torres, Y.J.; Gurbüz, E.I.; Farnood, R.; Dumesic, J.A. Catalytic conversion of biomass using solvents derived from lignin. Green Chem. 2012, 14, 1573–1576. [Google Scholar] [CrossRef]
- Clark, J.H.; Macquarrie, D.J.; Sherwood, J. A quantitative comparison between conventional and bio-derived solvents from citrus waste in esterification and amidation kinetic studies. Green Chem. 2012, 14, 90–93. [Google Scholar] [CrossRef]
- Clark, J.H.; Macquarrie, D.J.; Sherwood, J. The combined role of catalysis and solvent effects on the biginelli reaction improving efficiency and sustainability. Chem. Eur. J. 2013, 19, 5174–5182. [Google Scholar] [CrossRef] [PubMed]
- Paggiola, G.; Hunt, A.J.; McElroy, C.R.; Sherwood, J.; Clark, J.H. Biocatalysis in bio-derived solvents an improved approach for medium optimisation. Green Chem. 2014, 16, 2107–2110. [Google Scholar] [CrossRef]
- Dávila, J.A.; Rosenberg, M.; Cardona, C.A. Techno-economic and Environmental Assessment of p-cymene and pectin production from orange peel. Waste Biomass Valorization 2015, 6, 253–261. [Google Scholar] [CrossRef]
- Leita, B.A.; Warden, A.C.; Burke, N.; O'Shea, M.S.; Trimm, D. Production of p-cymene and hydrogen from a bio-renewable feedstock–1,8-cineole (eucalyptus oil). Green Chem. 2010, 12, 70–76. [Google Scholar] [CrossRef]
- Berti, C.; Binassi, E.; Colonna, M.; Fiorini, M.; Kannan, G.; Karanam, S.; Mazzacurati, M.; Odeh, I. Bio-Based Terephthalate Polyesters. US Patent 0168461 A1, 1 July 2010. [Google Scholar]
- Colonna, M.; Berti, C.; Fiorini, M.; Binassi, E.; Mazzacurati, M.; Vannini, M.; Karanam, S. Synthesis and radiocarbon evidence of terephthalate polyesters completely prepared from renewable resources. Green Chem. 2011, 13, 2543–2548. [Google Scholar] [CrossRef]
- Liu, C.; Wang, H.; Karim, A.M.; Sun, J.; Wang, Y. Catalytic fast pyrolysis of lignocellulosic biomass. Chem. Soc. Rev. 2014, 43, 7594–7623. [Google Scholar] [CrossRef] [PubMed]
- Kelkar, S.; Saffron, C.M.; Andreassi, K.; Li, Z.; Murkute, A.; Miller, D.J.; Pinnavaia, T.J.; Kriegel, R.M. A survey of catalysts for aromatics from fast pyrolysis of biomass. Appl. Catal. B: Environ. 2015, 174–175, 85–95. [Google Scholar] [CrossRef]
- Bauer, F.; Hulteberg, C. Is there a future in glycerol as a feedstock in the production of biofuels and biochemicals? Biofuels Bioprod. Biorefining 2013, 7, 43–51. [Google Scholar] [CrossRef]
- Anellotech Announces Successful Start-up of Pearl River Pilot Plant with Production of kilogram-Scale BTX (Press Release). Available online: http://anellotech.com/press/anellotech-announces-successful-start-pearl-river-pilot-plant-production-kilogram-scale-btx (accessed on 29 May 2015).
- Carlson, T.R.; Tompsett, G.A.; Conner, W.C.; Huber, G.W. Aromatic production from catalytic fast pyrolysis of biomass-derived feedstocks. Top. Catal. 2009, 52, 241–252. [Google Scholar] [CrossRef]
- Carlson, T.R.; Cheng, Y.T.; Jae, J.; Huber, GW. Production of green aromatics and olefins by catalytic fast pyrolysis of wood sawdust. Energy Environ. Sci. 2011, 4, 145–161. [Google Scholar] [CrossRef]
- Blommel, P.; Held, A.; Goodwin, R.; Cortright, R. Process for Converting Biomass to Aromatic Hydrocarbons. US Patent 0349361 A1, 27 November 2014. [Google Scholar]
- Virent Announces World’s First Demonstration of Full Range Bio-Aromatics Production. Available online: http://www.virent.com/news/virent-announces-worlds-first-demonstration-of-full-range-bio-aromatics-production (accessed on 29 May 2015).
- Virent BioFormPX Paraxylene Used for World’s First PET Plastic Bottle Made Entirely from Plant Based Material. Available online: http://www.virent.com/news/virent-bioformpx-paraxylene-used-for-worlds-first-pet-plastic-bottle-made-entirely-from-plant-based-material/ (accessed on 15 June 2015).
- Coca-Cola Produces 100% Bio-Based PET Bottle. Available online: http://greenchemicalsblog.com/2015/06/09/coca-cola-produces-100-bio-based-pet-bottle (accessed on 15 June 2015).
- Hallett, J.P.; Welton, T. Room-temperature ionic liquids solvents for synthesis and catalysis 2. Chem. Rev. 2011, 111, 3508–3576. [Google Scholar] [CrossRef] [PubMed]
- Beckman, E.J. Supercritical and near-critical CO2 in green chemical synthesis and processing. J. Supercrit. Fluids 2004, 28, 121–191. [Google Scholar] [CrossRef]
- Simon, M.O.; Li, C.J. Green chemistry oriented organic synthesis in water. Chem. Soc. Rev. 2012, 41, 1415–1427. [Google Scholar] [CrossRef] [PubMed]
- Jessop, P.G. Searching for green solvents. Green Chem. 2011, 13, 1391–1398. [Google Scholar] [CrossRef]
- Wolfson, A.; Dlugy, C.; Tavor, D. Glycerol-based solvents in organic synthesis. Trends Organ. Chem. 2011, 15, 41–50. [Google Scholar]
- Abbott, A.P.; Harris, R.C.; Ryder, K.S.; D'Agostino, C.; Gladden, L.F.; Mantle, M.D. Glycerol eutectics as sustainable solvent systems. Green Chem. 2011, 13, 82–90. [Google Scholar] [CrossRef]
- Cargill Glycerine. Available online: http://www.cargill.com/products/industrial/bioindustrial-index/glycerin/index.jsp (accessed on 29 May 2015).
- Augeo™ SL 191 (Solketal). Available online: http://www.rhodia.com.cn/en/binaries/Flyer_AugeoSL191_EN.pdf (accessed on 29 May 2015).
- Glycerol Formal Description. Available online: http://www.lambiotte.com/Glycerol_Formal-product_view.htm?id=69 (accessed on 29 May 2015).
- Florachem d-Limonene. Available online: http://industrial.florachem.com/products/citrus (accessed on 29 May 2015).
- BioAmber Diethyl Succinate. Available online: http://www.ulprospector.com/en/na/PersonalCare/Detail/6387/349249/Bio-DES (accessed on 29 May 2015).
- Advanced Biotech Flavor and Fragance Ingredients. Available online: http://www.adv-bio.com (accessed on 8 June 2015).
- Pennakem 1,2-Pentanediol. Available online: http://www.pennakem.com/products/%20%205343-92-0_1,2-Pentanediol_UPDATE.html (accessed on 29 May 2015).
- Pennakem 2-Methyltetrahydrofuran. Available online: http://www.pennakem.com/products/methyltetrahydrofuran.html (accessed on 29 May 2015).
- French Roquette Builds Isosorbide Plant. Available online: http://www.icis.com/resources/news/2007/09/13/9062102/french-roquette-builds-isosorbide-plant (accessed on 8 June 2015).
- Lotioncrafter Dimethyl Isosorbide. Available online: http://www.lotioncrafter.com/dimethyl-isosorbide-dmi.html (accessed on 8 June 2015).
- Chemrec Dimethyl Ether. Available online: http://www.chemrec.se (accessed on 29 May 2015).
- Soy Solvents a Market Opportunity Study. Available online: http://soynewuses.org/wp-content/uploads/pdf/final_SolventsMarketStudy.pdf (accessed on 5 June 2015).
- Horváth, I.T.; Mehdi, H.; Fábos, V.; Boda, L.; Mika, L.T. γ-Valerolactone a sustainable liquid for energy and carbon-based chemicals. Green Chem. 2008, 10, 238–242. [Google Scholar] [CrossRef]
- Cao, F.; Schwartz, T.J.; McClelland, D.J.; Krishna, S.H.; Dumesic, J.A.; George, W.; Huber, G.W. Dehydration of cellulose to levoglucosenone using polar aprotic solvents. Energy Environ. Sci. 2015, 8, 1808–1815. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, J.H.; Farmer, T.J.; Hunt, A.J.; Sherwood, J. Opportunities for Bio-Based Solvents Created as Petrochemical and Fuel Products Transition towards Renewable Resources. Int. J. Mol. Sci. 2015, 16, 17101-17159. https://doi.org/10.3390/ijms160817101
Clark JH, Farmer TJ, Hunt AJ, Sherwood J. Opportunities for Bio-Based Solvents Created as Petrochemical and Fuel Products Transition towards Renewable Resources. International Journal of Molecular Sciences. 2015; 16(8):17101-17159. https://doi.org/10.3390/ijms160817101
Chicago/Turabian StyleClark, James H., Thomas J. Farmer, Andrew J. Hunt, and James Sherwood. 2015. "Opportunities for Bio-Based Solvents Created as Petrochemical and Fuel Products Transition towards Renewable Resources" International Journal of Molecular Sciences 16, no. 8: 17101-17159. https://doi.org/10.3390/ijms160817101
APA StyleClark, J. H., Farmer, T. J., Hunt, A. J., & Sherwood, J. (2015). Opportunities for Bio-Based Solvents Created as Petrochemical and Fuel Products Transition towards Renewable Resources. International Journal of Molecular Sciences, 16(8), 17101-17159. https://doi.org/10.3390/ijms160817101