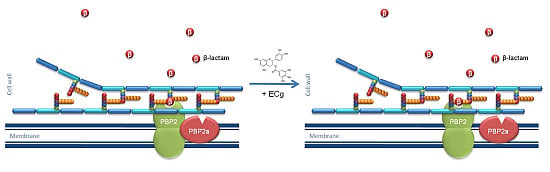
Impact of the β-Lactam Resistance Modifier (−)-Epicatechin Gallate on the Non-Random Distribution of Phospholipids across the Cytoplasmic Membrane of Staphylococcus aureus
Abstract
:1. Introduction
2. Results
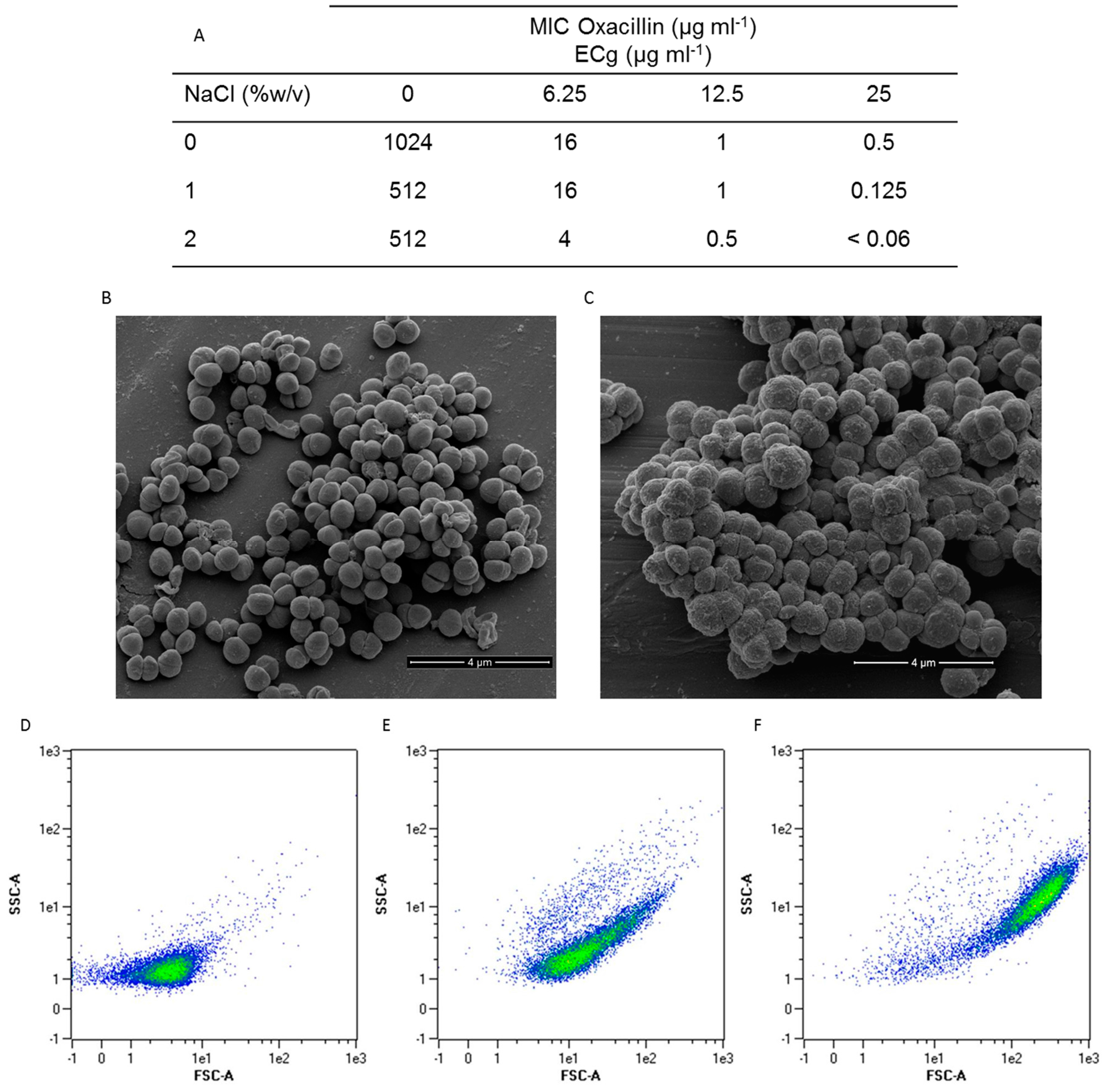
2.1. Intercalation of ECg into the EMRSA-16 CM Induces Major Changes to the Bacterial Phenotype
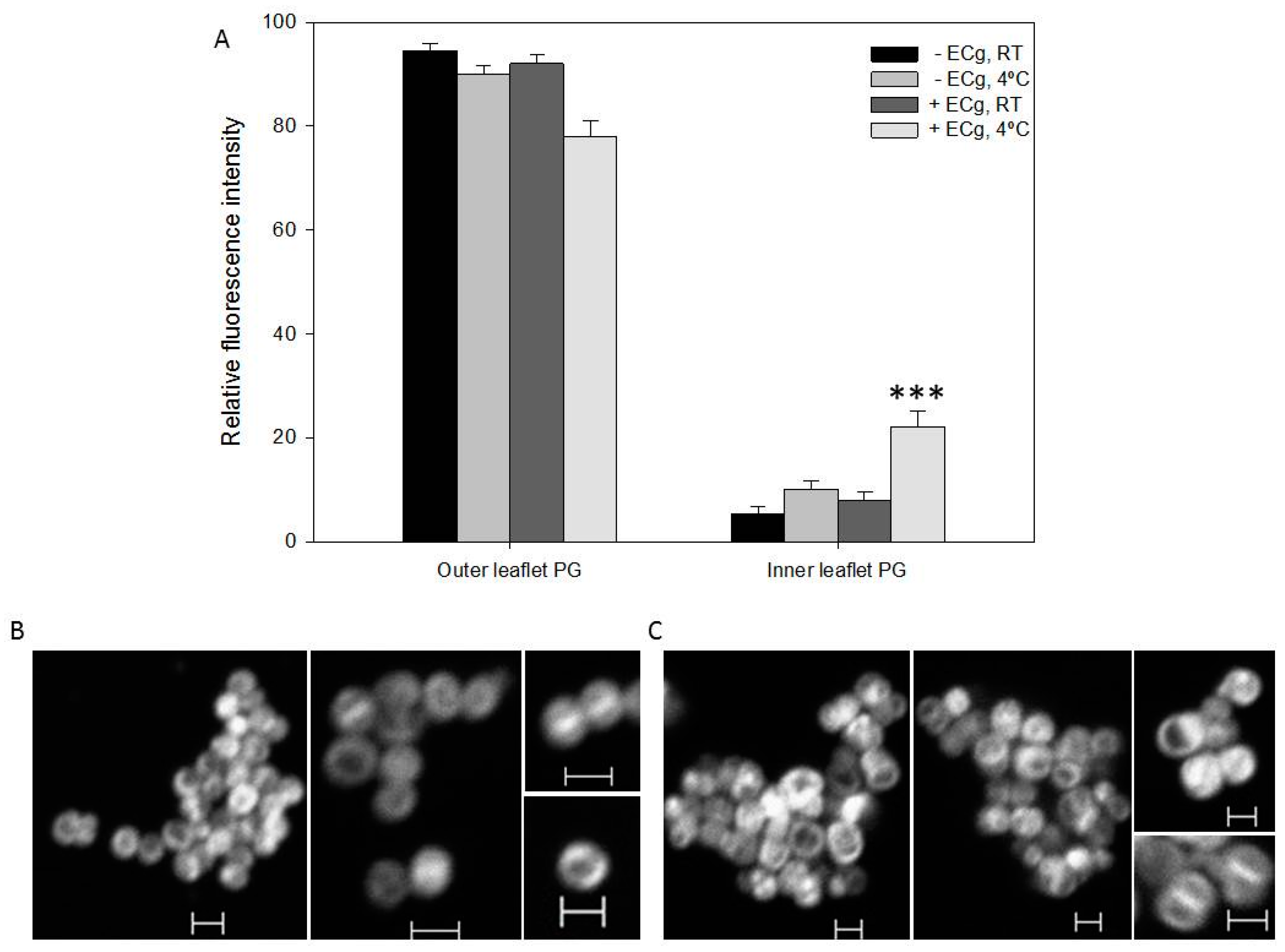
2.2. Intercalation of ECg into the EMRSA-16 CM Induces Small but Statistically Significant Changes in Membrane Lipid Symmetry
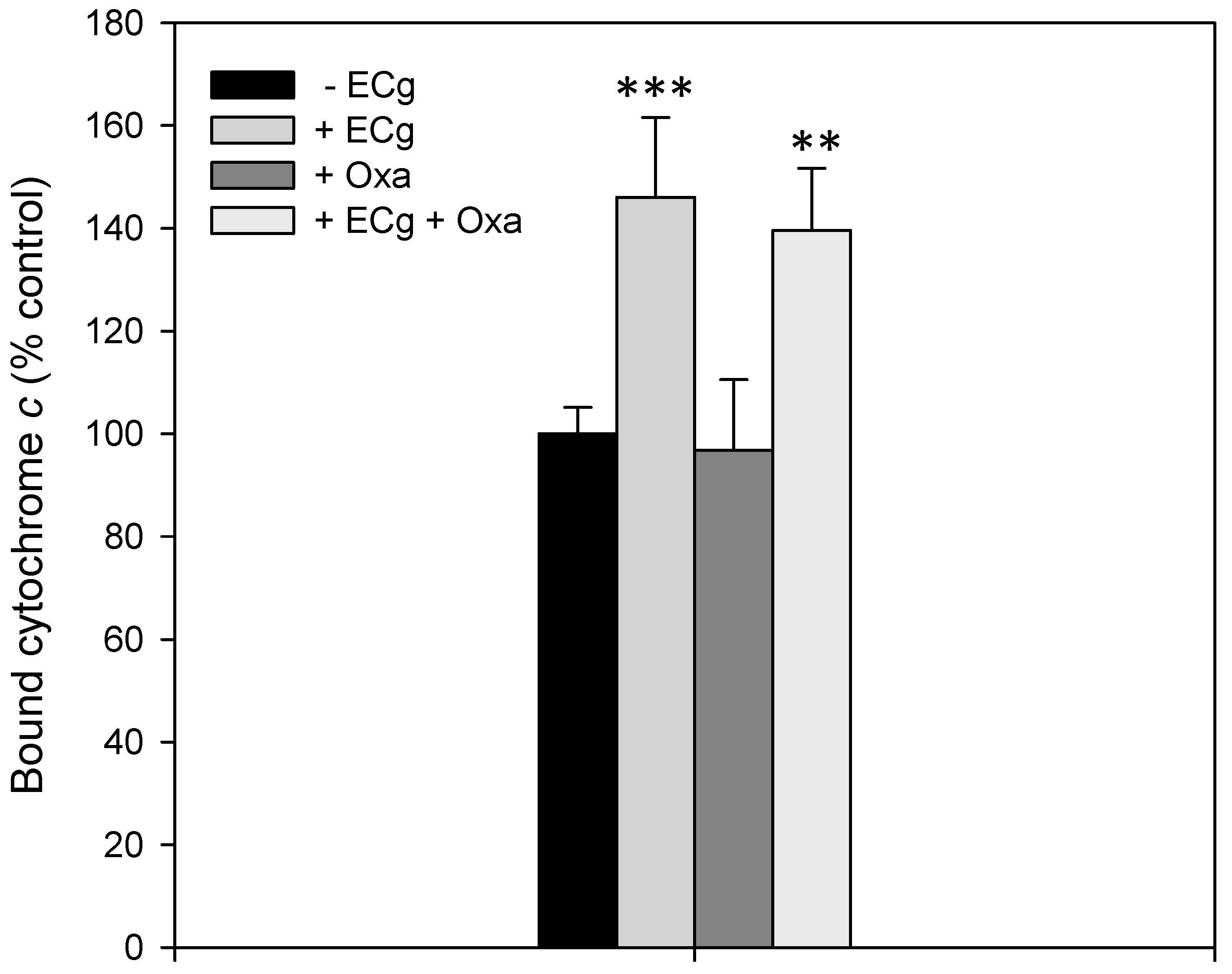
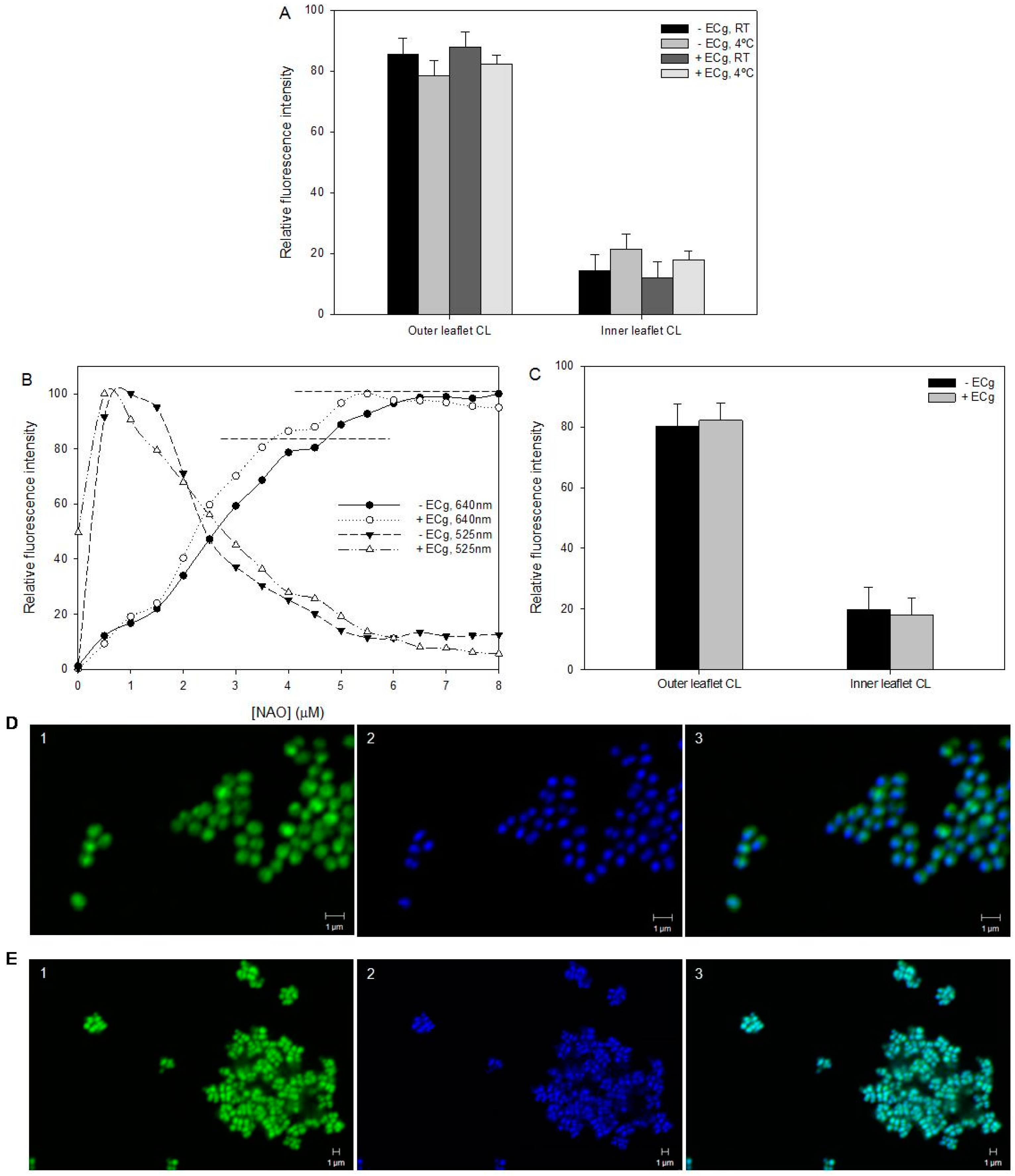
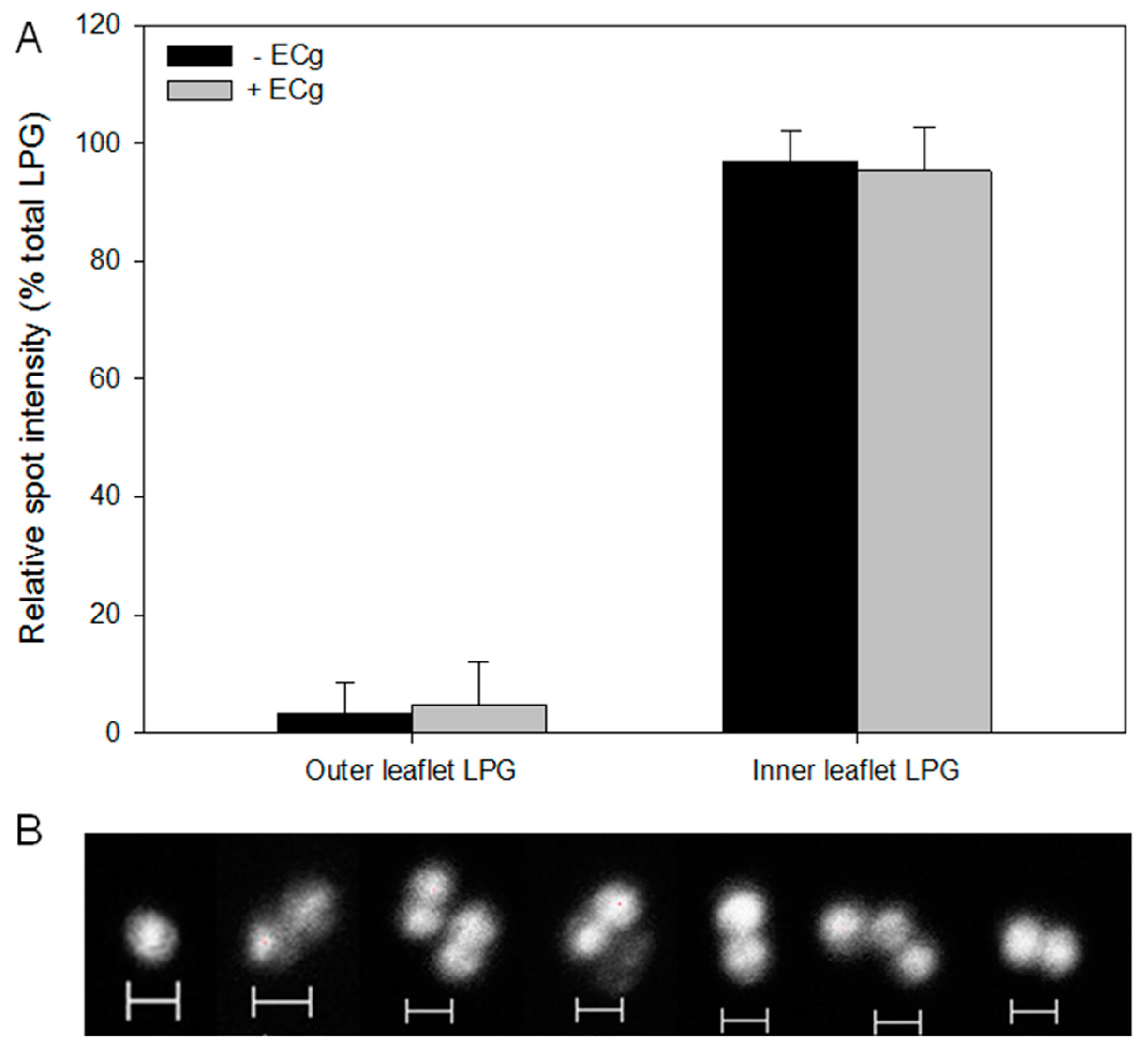
2.3. ECg Does Not Alter the Mode of Peptidoglycan Biosynthesis or Septum Formation in EMRSA-16
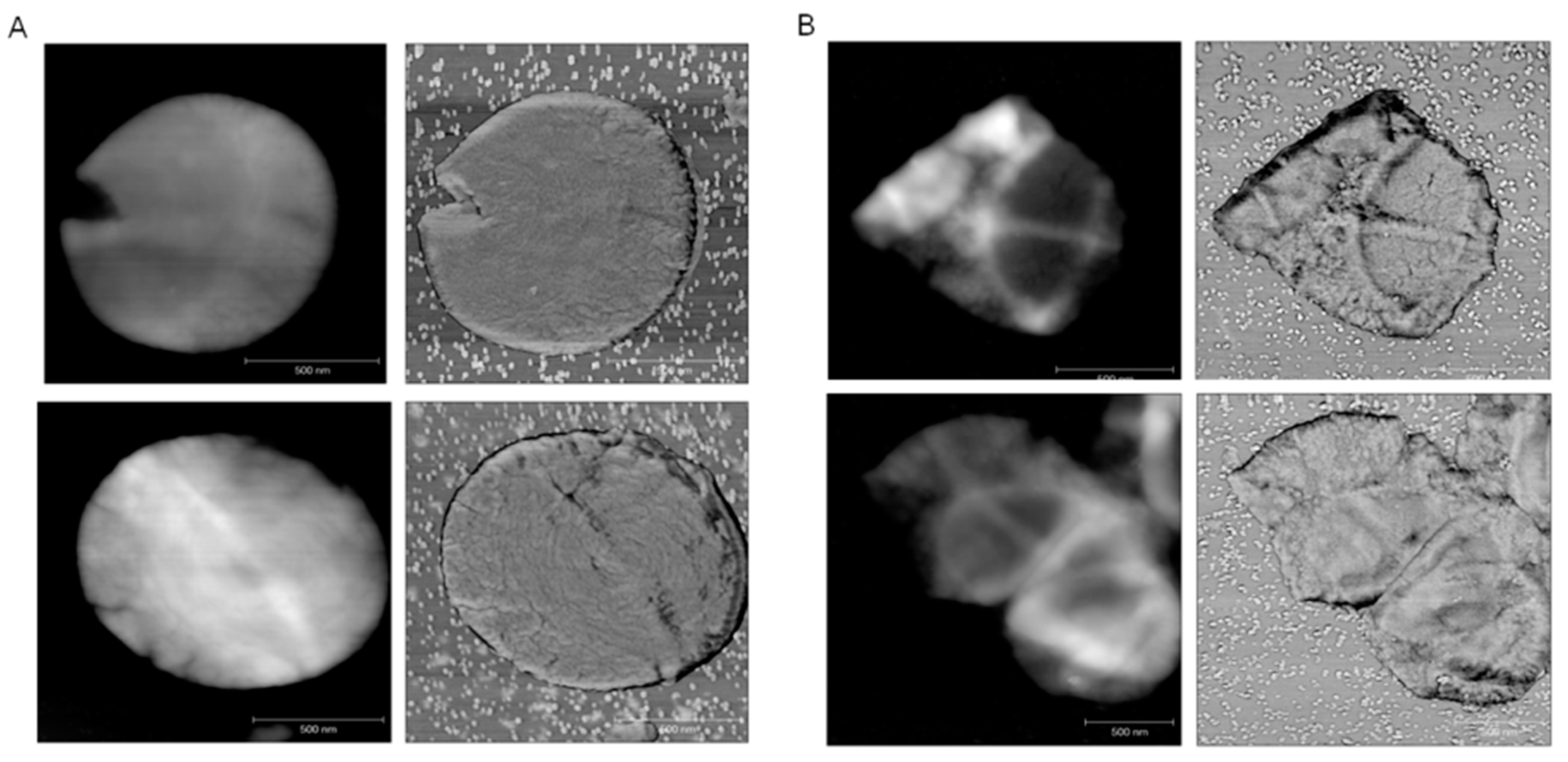
3. Discussion
4. Experimental Section
4.1. Bacteria
4.2. Bilayer Distribution of LPG
4.3. Bilayer Distribution of PG
4.4. Bilayer Distribution of CL
4.5. Flow Cytometry
4.6. Confocal Laser Scanning Microscopy (CLSM)
4.7. Atomic Force Microscopy (AFM)
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lingwood, D.; Simons, K. Lipid rafts as a membrane-organizing principle. Science 2010, 327, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Cross, T.A.; Murray, D.T.; Watts, A. Helical membrane protein conformations and their environment. Eur. J. Biophys. 2013, 42, 731–755. [Google Scholar] [CrossRef] [PubMed]
- Michel, V.; Bakovic, M. Lipid rafts in health and disease. Biol. Cell 2007, 99, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: Where they are and how they behave. Nat. Rev. Mol. Cell Biol 2008, 9, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Fadeel, B.; Xue, D. The ins and outs of phospholipid asymmetry in the plasma membrane: roles in health and disease. Crit. Rev. Biochem. Mol. Biol. 2009, 44, 264–277. [Google Scholar] [CrossRef] [PubMed]
- Venegas, V.; Zhou, Z. Two alternative mechanisms that regulate the presentation of apoptotic cell engulfment signal in Caenorhabditis elegans. Mol. Biol. Cell 2007, 18, 3180–3192. [Google Scholar] [CrossRef] [PubMed]
- Daleke, D.L. Regulation of phospholipid asymmetry in the erythrocyte membrane. Curr. Opin. Hematol. 2008, 15, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Schoenwaelder, S.M.; Yuan, Y.; Josefsson, E.C.; White, M.J.; Yao, Y.; Mason, K.D.; O’Reilly, L.A.; Henley, K.J.; Ono, A.; Hsiao, S.; et al. Two distinct pathways regulate platelet phosphatidylserine exposure and procoagulant function. Blood 2009, 114, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Lhermusier, T.; Chap, H.; Payrastre, B. Platelet membrane phospholipid asymmetry: From the characterization of a scramblase activity to the identification of an essential protein mutated in Scott syndrome. J. Thromb. Haemost. 2011, 9, 1883–1891. [Google Scholar] [CrossRef] [PubMed]
- Mileykovskaya, E.; Dowhan, W. Visualization of phospholipid domains in Escherichia coli by using the cardiolipin-specific fluorescent dye 10-N-nonyl acridine orange. J. Bacteriol. 2000, 182, 1172–1175. [Google Scholar] [CrossRef] [PubMed]
- Kawai, F.; Shoda, M.; Harashima, R.; Sadaie, Y.; Hara, H.; Matsumoto, K. Cardiolipin domains in Bacillus subtilis marburg membranes. J. Bacteriol. 2004, 186, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Barák, I.; Muchová, K.; Wilkinson, A.J.; O'Toole, P.J.; Pavlendová, N. Lipid spirals in Bacillus subtilis and their role in cell division. Mol. Microbiol. 2008, 68, 1315–1327. [Google Scholar] [CrossRef] [PubMed]
- López, D.; Kolter, R. Functional microdomains in bacterial membranes. Genes Dev. 2010, 24, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, M.; Dowhan, W. Lipid-dependent generation of dual topology for a membrane protein. J. Biol. Chem. 2012, 287, 37939–37948. [Google Scholar] [CrossRef] [PubMed]
- Malinverni, J.C.; Silhavy, T.J. An ABC transport system that maintains lipid asymmetry in the Gram-negative outer membrane. Proc. Natl. Acad. Sci. USA 2009, 106, 8009–8014. [Google Scholar] [CrossRef] [PubMed]
- Norris, V.; Misevic, G.; Delosme, J.M.; Oshima, A. Hypothesis: A phospholipid translocase couples lateral and transverse bilayer asymmetries in dividing bacteria. J. Mol. Biol. 2002, 318, 455–462. [Google Scholar] [CrossRef]
- Kol, M.A.; de Kroon, A.I.P.M.; Killian, J.A.; de Kruijff, B. Transbilayer movement of phospholipids in biogenic membranes. Biochemistry 2004, 43, 2673–2681. [Google Scholar] [CrossRef] [PubMed]
- Benfield, T.; Espersen, F.; Frimodt-Møller, N.; Jensen, A.G.; Larsen, A.R.; Pallesen, L.V.; Skov, R.; Westh, H.; Skinhøj, P. Increasing incidence but decreasing in-hospital mortality of adult Staphylococcus aureus bacteraemia between 1981 and 2000. Clin. Microbiol. Infect. 2007, 13, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Thwaites, G.E.; Edgeworth, J.D.; Gkrania-Klotsas, E.; Kirby, A.; Tilley, R.; Török, M.E.; Walker, S.; Wertheim, H.F.; Wilson, P.; Llewelyn, M.J. Clinical management of Staphylococcus aureus bacteraemia. Lancet Infect. Dis. 2011, 11, 208–222. [Google Scholar] [CrossRef]
- Strommenger, B.; Bartels, M.D.; Kurt, K.; Layer, F.; Rohde, S.M.; Boye, K.; Westh, H.; Witte, W.; de Lencastre, H.; Nübel, U. Evolution of methicillin-resistant Staphylococcus aureus towards increasing resistance. J. Antimicrob. Chemother. 2014, 69, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Short, S.A.; White, D.C. Metabolism of phosphatidylglycerol, lysylphosphatidylglycerol, and cardiolipin of Staphylococcus aureus. J. Bacteriol. 1971, 108, 219–226. [Google Scholar] [PubMed]
- Mukhopadhyay, K.; Whitmire, W.; Xiong, Y.Q.; Molden, J.; Jones, T.; Peschel, A.; Staubitz, P.; Adler-Moore, J.; McNamara, P.J.; Proctor, R.A.; et al. In vitro susceptibility of Staphylococcus aureus to thrombin-induced platelet microbicidal protein-1 (tPMP-1) is influenced by cell membrane phospholipid composition and asymmetry. Microbiology 2007, 153, 1187–1197. [Google Scholar] [CrossRef] [PubMed]
- Bernal, P.; Lemaire, S.; Pinho, M.G.; Mobashery, S.; Hinds, J.; Taylor, P.W. Insertion of epicatechin gallate into the cytoplasmic membrane of methicillin-resistant Staphylococcus aureus disrupts penicillin-binding protein (PBP) 2a-mediated β-lactam resistance by delocalizing PBP2. J. Biol. Chem. 2010, 285, 24055–24065. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.; Yeaman, M.R.; Sakoulas, G.; Yang, S.J.; Proctor, R.A.; Sahl, H.G.; Schrenzel, J.; Xiong, Y.Q.; Bayer, A.S. Failures in clinical treatment of Staphylococcus aureus infection with daptomycin are associated with alterations in surface charge, membrane phospholipid asymmetry, and drug binding. Antimicrob. Agents Chemother. 2008, 52, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Fuda, C.C.S.; Fisher, J.F.; Mobashery, S. β-lactam resistance in Staphylococcus aureus: The adaptive resistance of a plastic genome. Cell Mol. Life Sci. 2005, 62, 2617–2633. [Google Scholar] [CrossRef] [PubMed]
- Paterson, G.K.; Harrison, E.M.; Holmes, M.A. The emergence of mecC methicillin-resistant Staphylococcus aureus. Trends Microbiol. 2014, 22, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Egan, A.J.F.; Vollmer, W. The physiology of bacterial cell division. Ann. N. Y. Acad. Sci. 2013, 1277, 8–28. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.W.; Hamilton-Miller, J.M.T.; Stapleton, P.D. Antimicrobial properties of green tea catechins. Food Sci. Technol. Bull. 2005, 2, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Paulin, S.; Jamshad, M.; Dafforn, T.R.; Garcia-Lara, J.; Foster, S.J.; Galley, N.F.; Roper, D.I.; Rosado, H.; Taylor, P.W. Surfactant-free purification of membrane protein complexes from bacteria: Application to the staphylococcal penicillin-binding protein complex PBP2/PBP2a. Nanotechnology 2014, 25, 285101. [Google Scholar] [CrossRef] [PubMed]
- Palacios, L.; Rosado, H.; Micol, V.; Rosato, A.E.; Bernal, P.; Arroyo, R.; Grounds, H.; Anderson, J.C.; Stabler, R.A.; Taylor, P.W. Staphylococcal phenotypes induced by naturally occurring and synthetic membrane-interactive polyphenolic β-lactam resistance modifiers. PLoS ONE 2014, 9, e93830. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.D.; Ratcliffe, E.C.; Wheeler, R.; Golestanian, R.; Hobbs, J.K.; Foster, S.J. Peptidoglycan architecture can specify division planes in Staphylococcus aureus. Nat. Commun. 2010, 1, 26. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, P.D.; Shah, S.; Anderson, J.C.; Hara, Y.; Hamilton-Miller, J.M.T.; Taylor, P.W. Modulation of β-lactam resistance in Staphylococcus aureus by catechins and gallates. Int. J. Antimicrob. Agents 2004, 23, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.F.; Edwards, D.I.; Hawkey, P.M.; Morrison, D.; Ridgway, G.L.; Towner, K.J.; Wren, M.W. Guidelines for the laboratory diagnosis and susceptibility testing of methicillin-resistant Staphylococcus aureus (MRSA). J. Antimicrob. Chemother. 2005, 56, 1000–1018. [Google Scholar] [CrossRef] [PubMed]
- Bernal, P.; Zloh, M.; Taylor, P.W. Disruption of d-alanyl esterification of Staphylococcus aureus cell wall teichoic acid by the β-lactam resistance modifier (−)-epicatechin gallate. J. Antimicrob. Chemother. 2009, 63, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, P.D.; Shah, S.; Ehlert, K.; Hara, Y.; Taylor, P.W. The β-lactam-resistance modifier (−)-epicatechin gallate alters the architecture of the cell wall of Staphylococcus aureus. Microbiology 2007, 153, 2093–2103. [Google Scholar] [CrossRef] [PubMed]
- Pomorski, T.; Muller, P.; Zimmermann, B.; Burger, K.; Devaux, P.F.; Herrmann, A. Transbilayer movement of fluorescent and spin-labeled phospholipids in the plasma membrane of human fibroblasts: A quantitative approach. J. Cell Sci. 1996, 109, 687–698. [Google Scholar] [PubMed]
- Devaux, P.F.; Fellmann, P.; Hervé, P. Investigation on lipid asymmetry using lipid probes: Comparison between spin-labeled lipids and fluorescent lipids. Chem. Phys. Lipids 2002, 116, 115–134. [Google Scholar] [CrossRef]
- Angeletti, C.; Nichols, J.W. Dithionite quenching rate measurement of the inside-outside membrane bilayer distribution of 7-nitrobenz-2-oxa-1,3-diazol-4-yl-labeled phospholipids. Biochemistry 1998, 37, 15114–15119. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, D.W.; Comfurius, P.; van Gool, R.G.; Bevers, E.M.; Zwaal, R.F. Multidrug resistance protein 1 regulates lipid asymmetry in erythrocyte membranes. Biochem. J. 2000, 350, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Pomorski, T.; Hrafnsdóttir, S.; Devaux, P.F.; van Meer, G. Lipid distribution and transport across cellular membranes. Semin. Cell Dev. Biol. 2001, 12, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Bayer, A.S.; McNamara, P.; Yeaman, M.R.; Lucindo, N.; Jones, T.; Cheung, A.L.; Sahl, H.G.; Proctor, R.A. Transposon disruption of the complex I NADH oxidoreductase gene (snoD) in Staphylococcus aureus is associated with reduced susceptibility to the microbicidal activity of thrombin-induced platelet microbicidal protein 1. J. Bacteriol. 2006, 188, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Stapleton, P.D.; Taylor, P.W. The polyphenol (–)-epicatechin gallate disrupts the secretion of virulence-related proteins by Staphylococcus aureus. Lett. Appl. Microbiol. 2008, 46, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Pinho, M.G.; Errington, J. Dispersed mode of Staphylococcus aureus cell wall synthesis in the absence of the division machinery. Mol. Microbiol. 2003, 50, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, K.; Schroit, A.J. Aminophospholipid asymmetry: A matter of life and death. Annu. Rev. Physiol. 2003, 65, 701–734. [Google Scholar] [CrossRef] [PubMed]
- Fuda, C.; Hesek, D.; Lee, M.; Morio, K.; Nowak, T.; Mobashery, S. Activation for catalysis of penicillin-binding protein 2a from methicillin-resistant Staphylococcus aureus by bacterial cell wall. J. Am. Chem. Soc. 2005, 127, 2056–2057. [Google Scholar] [CrossRef] [PubMed]
- Pinho, M.G.; Errington, J. Recruitment of penicillin-binding protein PBP2 to the division site of Staphylococcus aureus is dependent on its transpeptidation substrates. Mol. Microbiol. 2005, 55, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, K.; Gupta, C.M. Transbilayer phosphatidylethanolamine movements in yeast plasma membranes. Evidence for protein-mediated, energy-dependent mechanism. Eur. J. Biochem. 1996, 240, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Petit, J.M.; Maftah, A.; Ratinaud, M.H.; Julien, R. 10 N-nonyl acridine orange interacts with cardiolipin and allows the quantification of this phospholipid in isolated mitochondria. Eur. J. Biochem. 1992, 209, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Gallet, P.F.; Petit, J.M.; Maftah, A.; Zachowski, A.; Julien, R. Asymmetrical distribution of cardiolipin in yeast inner mitochondrial membrane triggered by carbon catabolite repression. Biochem. J. 1997, 324, 627–634. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosado, H.; Turner, R.D.; Foster, S.J.; Taylor, P.W. Impact of the β-Lactam Resistance Modifier (−)-Epicatechin Gallate on the Non-Random Distribution of Phospholipids across the Cytoplasmic Membrane of Staphylococcus aureus. Int. J. Mol. Sci. 2015, 16, 16710-16727. https://doi.org/10.3390/ijms160816710
Rosado H, Turner RD, Foster SJ, Taylor PW. Impact of the β-Lactam Resistance Modifier (−)-Epicatechin Gallate on the Non-Random Distribution of Phospholipids across the Cytoplasmic Membrane of Staphylococcus aureus. International Journal of Molecular Sciences. 2015; 16(8):16710-16727. https://doi.org/10.3390/ijms160816710
Chicago/Turabian StyleRosado, Helena, Robert D. Turner, Simon J. Foster, and Peter W. Taylor. 2015. "Impact of the β-Lactam Resistance Modifier (−)-Epicatechin Gallate on the Non-Random Distribution of Phospholipids across the Cytoplasmic Membrane of Staphylococcus aureus" International Journal of Molecular Sciences 16, no. 8: 16710-16727. https://doi.org/10.3390/ijms160816710