Comparison of Selective Laser Melted Titanium and Magnesium Implants Coated with PCL
Abstract
:1. Introduction
2. Results
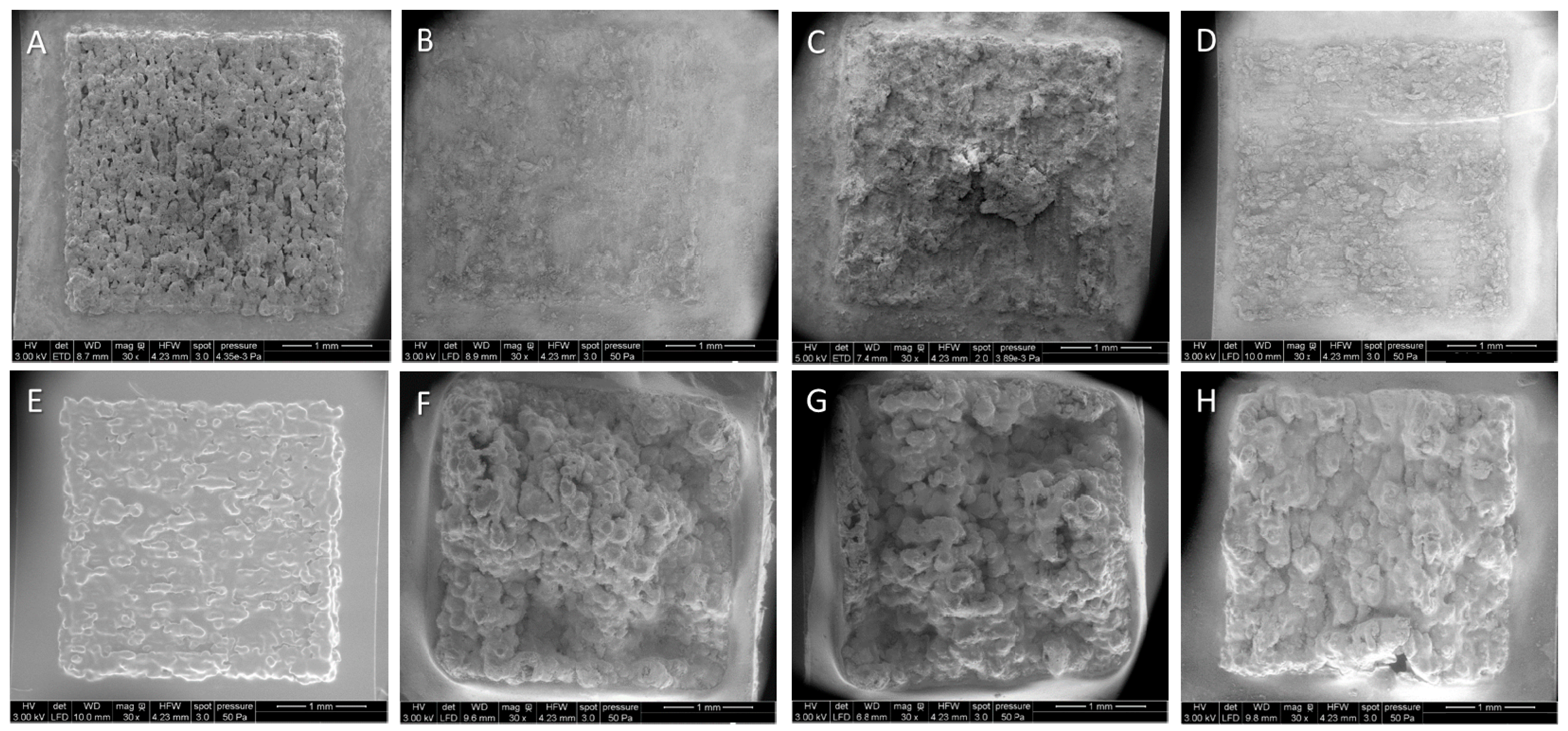
2.1. Manufacturing

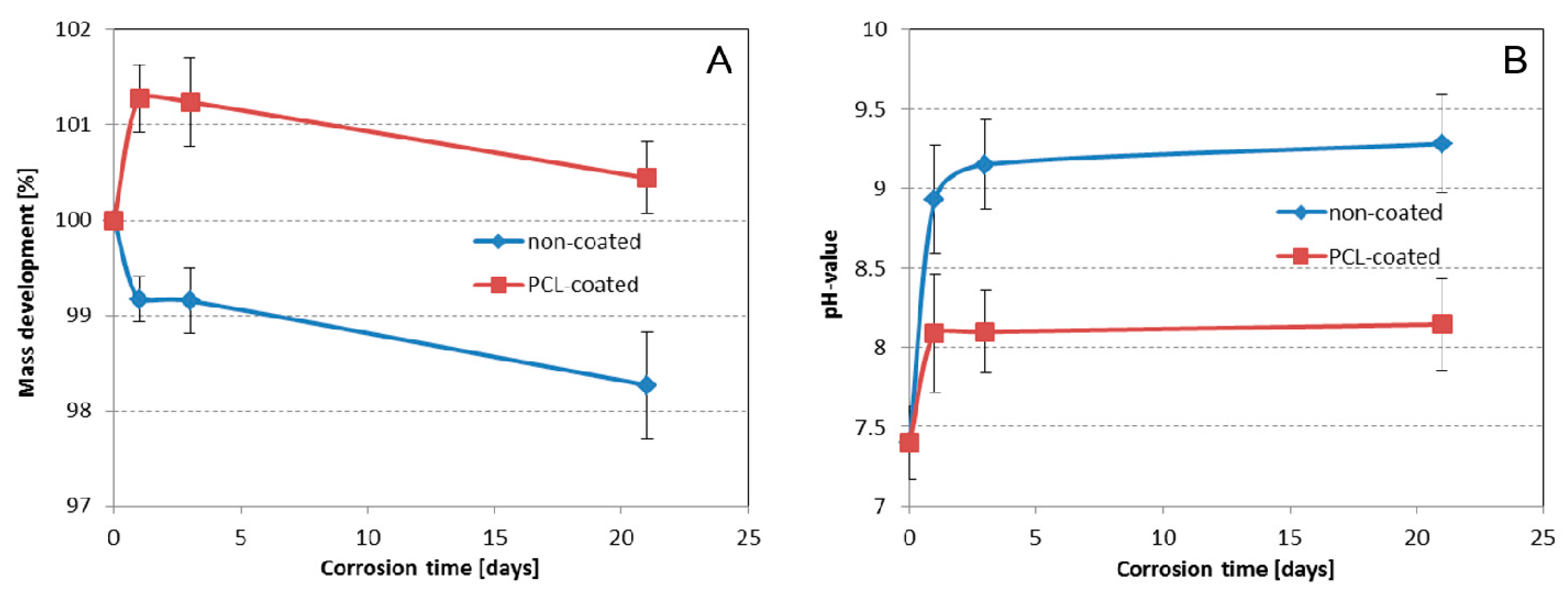
2.2. In Vitro Corrosion Study
| Corrosion Time (Days) | At% Mg | At% O | At% C |
|---|---|---|---|
| 0 | 0.06 | 34.08 | 65.86 |
| 1 | 6.19 | 54.96 | 38.85 |
| 3 | 6.33 | 52.75 | 40.92 |
| 21 | 4.82 | 54.99 | 40.19 |
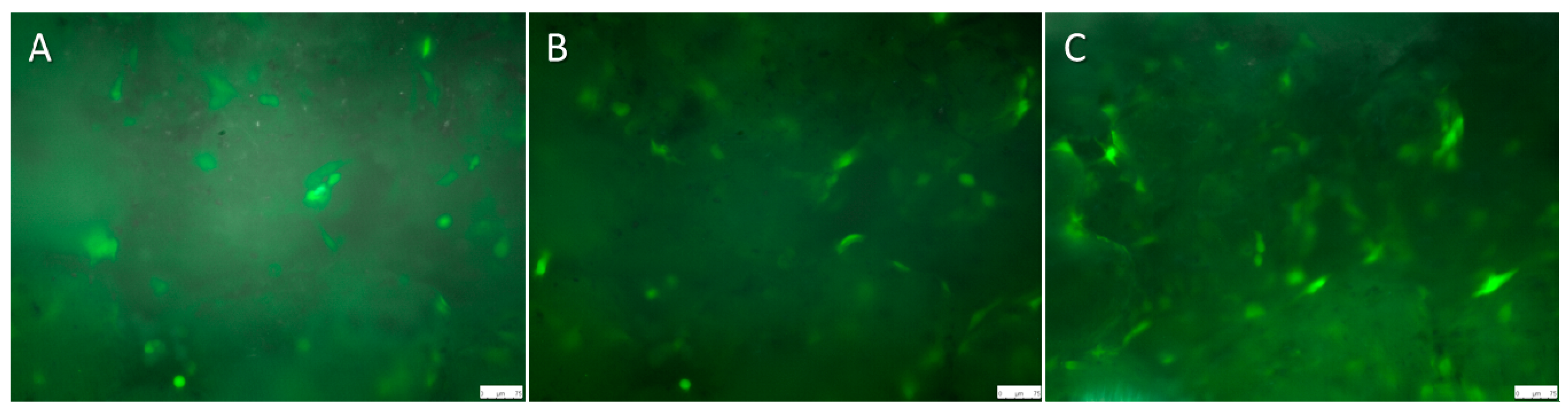
2.3. Live Cell Imaging (LCI) of Magnesium Hybrid Construct Compared with the Titanium Implant Coated with Polycaprolactone (PCL) Seeded with Green Fluorescent Protein (GFP)-Osteoblast
2.4. PCL-Thickness Measurement of Coated Magnesium Implant

| Position | PCL External Coating (µm) | PCL Internal Coating (µm) |
|---|---|---|
| Top | 10.8 ± 1.2 | 1.2 ± 0.6 |
| Center | 3.7 ± 3.5 | 1.3 ± 0.3 |
| Bottom | 1 ± 0.1 | 1.4 ± 0.3 |
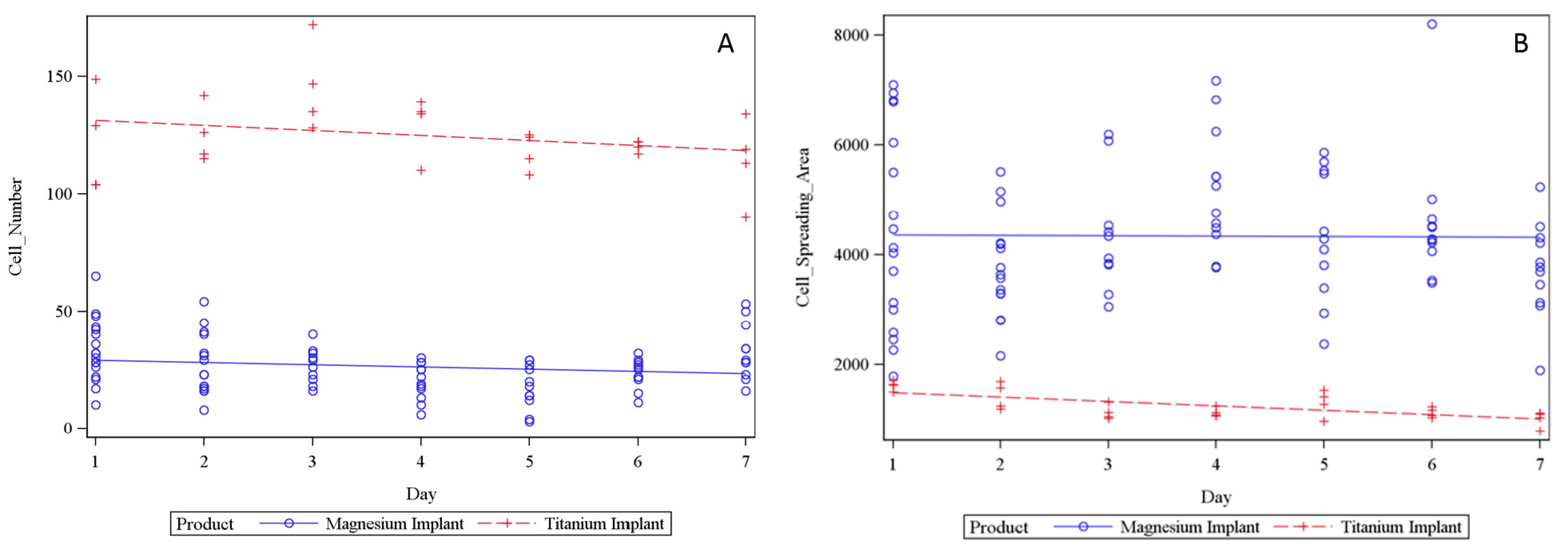
2.5. LCI of Osteoblasts Seeded on Magnesium PCL and Titanium PCL Implants
3. Discussion
4. Experimental Section
4.1. Selective Laser Melting (SLM) of Titanium and Magnesium Implants
4.2. In Vitro Corrosion Study
4.2.1. Spray-Coating Process for Application of Polymeric Coatings to Flat Magnesium Structures for the in Vitro Corrosion
4.2.2. Gravimetry
4.2.3. PH Value Measurement
4.2.4. Scanning Electron Microscopy and EDX Measurements
4.3. Murine GFP-Osteoblast and Murine Osteoblast Isolation
4.4. PCL Coating of Magnesium PCL and Titanium PCL Implant
4.5. PCL-Thickness Measurement of Coated Magnesium Implant
4.6. Live Cell Imaging (LCI)
4.6.1. LCI of Titanium Implant Seeded with GFP-Osteoblast
4.6.2. LCI of Magnesium Hybrid Construct and Magnesium PCL Implant Seeded with GFP-Osteoblast
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gellrich, N.C.; Held, U.; Schoen, R.; Pailing, T.; Schramm, A.; Bormann, K.H. Alveolar zygomatic buttress: A new donor site for limited preimplant augmentation procedures. J. Oral Maxillofac. Surg. 2007, 65, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Silber, J.S.; Anderson, D.G.; Daffner, S.D.; Brislin, B.T.; Leland, J.M.; Hilibrand, A.S.; Vaccaro, A.R.; Albert, T.J. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine 2003, 28, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Sasso, R.C.; LeHuec, J.C.; Shaffrey, C. Iliac crest bone graft donor site pain after anterior lumbar interbody fusion: A prospective patient satisfaction outcome assessment. J. Spinal Disord. Tech. 2005, 18, S77–S81. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.E.; Matta, R.; Mendes, V.C.; Perri de Carvalho, P.S. Development, characterization and clinical use of a biodegradable composite scaffold for bone engineering in oro-maxillo-facial surgery. Organogenesis 2010, 6, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wang, J.; Tang, L.; Ao, H.; Tan, H.; Tang, T.; Liu, C. Mesoporous bioactive glass doped-poly (3-hydroxybutyrate-co-3-hydroxyhexanoate) composite scaffolds with 3-dimensionally hierarchical pore networks for bone regeneration. Colloids Surf. B Biointerfaces 2014, 116, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Willbold, E.; Gu, X.; Albert, D.; Kalla, K.; Bobe, K.; Brauneis, M.; Janning, C.; Nellesen, J.; Czayka, W.; Tillmann, W.; et al. Effect of the addition of low rare earth elements (lanthanum, neodymium, cerium) on the biodegradation and biocompatibility of magnesium. Acta Biomater. 2015, 11, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Waizy, H.; Seitz, J.-M.; Reifenrath, J.; Weizbauer, A.; Bach, F.-W.; Meyer-Lindenberg, A.; Denkena, B.; Windhagen, H. Biodegradable magnesium implants for orthopedic applications. J. Mater. Sci. 2013, 48, 39–50. [Google Scholar] [CrossRef]
- Shadanbaz, S.; Walker, J.; Woodfield, T.B.; Staiger, M.P.; Dias, G.J. Monetite and brushite coated magnesium: In vivo and in vitro models for degradation analysis. J. Mater. Sci. Mater. Med. 2014, 25, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Song, Y.; Zhang, S.; Li, J.; Zhao, C.; Zhang, X. Interaction between a high purity magnesium surface and PCL and PLA coatings during dynamic degradation. Biomed. Mater. 2011, 6, 025005. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yamamoto, A. Characteristics and cytocompatibility of biodegradable polymer film on magnesium by spin coating. Colloids Surf. B Biointerfaces 2012, 93, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Ring, A.; Langer, S.; Homann, H.H.; Kuhnen, C.; Schmitz, I.; Steinau, H.U.; Drucke, D. Analysis of neovascularization of PEGT/PBT-copolymer dermis substitutes in balb/c-mice. Burns 2006, 32, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Artel, A.; Mehdizadeh, H.; Chiu, Y.C.; Brey, E.M.; Cinar, A. An agent-based model for the investigation of neovascularization within porous scaffolds. Tissue Eng. Part A 2011, 17, 2133–2141. [Google Scholar] [CrossRef] [PubMed]
- Rakhmatia, Y.D.; Ayukawa, Y.; Furuhashi, A.; Koyano, K. Current barrier membranes: Titanium mesh and other membranes for guided bone regeneration in dental applications. J. Prosthodont. Res. 2013, 57, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shen, Y.; Wang, Z.; Yang, J.; Liu, N.; Huang, W. Development of highly porous titanium scaffolds by selective laser melting. Mater. Lett. 2010, 64, 674–676. [Google Scholar] [CrossRef]
- Jauer, L.; Meiners, W.; Poprawe, R. Selective laser melting of biodegradable metals. Eur. Cells Mater. 2013, 26, 21. [Google Scholar]
- Matena, J.; Petersen, S.; Gieseke, M.; Kampmann, A.; Teske, M.; Beyerbach, M.; Escobar, H.M.; Haferkamp, H.; Gellrich, N.C.; Nolte, I. SLM produced porous titanium implant improvements for enhanced vascularization and osteoblast seeding. Int. J. Mol. Sci. 2015, 16, 7478–7492. [Google Scholar] [CrossRef] [PubMed]
- Kruth, J.P.; Froyen, L.; van Vaerenbergh, J.; Mercelis, P.; Rombouts, M.; Lauwers, B. Selective laser melting of iron-based powder. J. Mater. Process. Technol. 2004, 149, 616–622. [Google Scholar] [CrossRef]
- Murray, J.L.; Wriedt, H.A. The O-Ti (oxygen-titanium) system. J. Phase Equilib. 1987, 8, 148–165. [Google Scholar] [CrossRef]
- Louvis, E.; Fox, P.; Sutcliffe, C.J. Selective laser melting of aluminium components. J. Mater. Process. Technol. 2011, 211, 275–284. [Google Scholar] [CrossRef]
- Vandenbroucke, B.; Kruth, J.P. Selective laser melting of biocompatible metals for rapid manufacturing of medical parts. Rapid Prototyp. J. 2007, 13, 196–203. [Google Scholar] [CrossRef]
- Wriedt, H.A. The Mg-O (magnesium-oxygen) system. Bull. Alloy Phase Diagr. 1987, 8, 227–233. [Google Scholar] [CrossRef]
- Niemeyer, M. Strahl-Stoff-Wechselwirkung und Resultierende Verbindungseigenschaften Beim Laserstrahlschweißen Von Magnesiumlegierungen; Verein Deutscher Ingenieure: Düsseldorf, Germany, 1999. [Google Scholar]
- Zhang, Y.; Zhang, G.; Wei, M. Controlling the biodegradation rate of magnesium using biomimetic apatite coating. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 89, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.M.; Adewunmi, A.; Schek, R.M.; Flanagan, C.L.; Krebsbach, P.H.; Feinberg, S.E.; Hollister, S.J.; Das, S. Bone tissue engineering using polycaprolactone scaffolds fabricated via selective laser sintering. Biomaterials 2005, 26, 4817–4827. [Google Scholar] [CrossRef] [PubMed]
- Wulf, K.; Teske, M.; Lobler, M.; Luderer, F.; Schmitz, K.P.; Sternberg, K. Surface functionalization of poly(epsilon-caprolactone) improves its biocompatibility as scaffold material for bioartificial vessel prostheses. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 98, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Giannoudis, P.V.; Einhorn, T.A.; Marsh, D. Fracture healing: The diamond concept. Injury 2007, 38, S3–S6. [Google Scholar] [CrossRef]
- Hagihara, K.; Fujii, K.; Matsugaki, A.; Nakano, T. Possibility of Mg- and Ca-based intermetallic compounds as new biodegradable implant materials. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 4101–4111. [Google Scholar] [CrossRef] [PubMed]
- Willbold, E.; Kalla, K.; Bartsch, I.; Bobe, K.; Brauneis, M.; Remennik, S.; Shechtman, D.; Nellesen, J.; Tillmann, W.; Vogt, C.; et al. Biocompatibility of rapidly solidified magnesium alloy rs66 as a temporary biodegradable metal. Acta Biomater. 2013, 9, 8509–8517. [Google Scholar] [CrossRef] [PubMed]
- Pichler, K.; Kraus, T.; Martinelli, E.; Sadoghi, P.; Musumeci, G.; Uggowitzer, P.J.; Weinberg, A.M. Cellular reactions to biodegradable magnesium alloys on human growth plate chondrocytes and osteoblasts. Int. Orthop. 2014, 38, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, S. Biodegradable Mg and Mg alloys: Corrosion and biocompatibility. Mater. Sci. Eng. B 2011, 176, 1600–1608. [Google Scholar] [CrossRef]
- Wang, J.; Qin, L.; Wang, K.; Wang, J.; Yue, Y.; Li, Y.; Tang, J.; Li, W. Cytotoxicity studies of AZ31D alloy and the effects of carbon dioxide on its biodegradation behavior in vitro. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 4416–4426. [Google Scholar] [CrossRef] [PubMed]
- Seuss, F.; Seuss, S.; Turhan, M.C.; Fabry, B.; Virtanen, S. Corrosion of mg alloy AZ91D in the presence of living cells. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 99, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Johnson, I.; Perchy, D.; Liu, H. In vitro evaluation of the surface effects on magnesium-yttrium alloy degradation and mesenchymal stem cell adhesion. J. Biomed. Mater. Res. Part A 2012, 100, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Ulrich, H.; Rudert, M.; Willbold, E. Biodegradable magnesium scaffolds: Part 1: Appropriate inflammatory response. J. Biomed. Mater. Res. A 2007, 81, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Ulrich, H.; Palm, C.; Willbold, E. Biodegradable magnesium scaffolds: Part II: Peri-implant bone remodeling. J. Biomed. Mater. Res. A 2007, 81, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Mueller, W.D.; Lucia Nascimento, M.; Lorenzo de Mele, M.F. Critical discussion of the results from different corrosion studies of Mg and Mg alloys for biomaterial applications. Acta Biomater. 2010, 6, 1749–1755. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.H.M.; Luthringer, B.J.C.; Feyerabend, F.; Willumeit, R. Mg and Mg alloys: How comparable are in vitro and in vivo corrosion rates? A review. Acta Biomater. 2015, 13, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Scheideler, L.; Fuger, C.; Schille, C.; Rupp, F.; Wendel, H.P.; Hort, N.; Reichel, H.P.; Geis-Gerstorfer, J. Comparison of different in vitro tests for biocompatibility screening of Mg alloys. Acta Biomater. 2013, 9, 8740–8745. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.D.; Qian, H.Y.; Neff, L.; Satomura, K.; Horowitz, M.C. Thy-1 antigen expression by cells in the osteoblast lineage. J. Bone Miner. Res. 1999, 14, 362–375. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matena, J.; Petersen, S.; Gieseke, M.; Teske, M.; Beyerbach, M.; Kampmann, A.; Escobar, H.M.; Gellrich, N.-C.; Haferkamp, H.; Nolte, I. Comparison of Selective Laser Melted Titanium and Magnesium Implants Coated with PCL. Int. J. Mol. Sci. 2015, 16, 13287-13301. https://doi.org/10.3390/ijms160613287
Matena J, Petersen S, Gieseke M, Teske M, Beyerbach M, Kampmann A, Escobar HM, Gellrich N-C, Haferkamp H, Nolte I. Comparison of Selective Laser Melted Titanium and Magnesium Implants Coated with PCL. International Journal of Molecular Sciences. 2015; 16(6):13287-13301. https://doi.org/10.3390/ijms160613287
Chicago/Turabian StyleMatena, Julia, Svea Petersen, Matthias Gieseke, Michael Teske, Martin Beyerbach, Andreas Kampmann, Hugo Murua Escobar, Nils-Claudius Gellrich, Heinz Haferkamp, and Ingo Nolte. 2015. "Comparison of Selective Laser Melted Titanium and Magnesium Implants Coated with PCL" International Journal of Molecular Sciences 16, no. 6: 13287-13301. https://doi.org/10.3390/ijms160613287
APA StyleMatena, J., Petersen, S., Gieseke, M., Teske, M., Beyerbach, M., Kampmann, A., Escobar, H. M., Gellrich, N.-C., Haferkamp, H., & Nolte, I. (2015). Comparison of Selective Laser Melted Titanium and Magnesium Implants Coated with PCL. International Journal of Molecular Sciences, 16(6), 13287-13301. https://doi.org/10.3390/ijms160613287





