1. Origin and Geographical Distribution
In the monogeneric genus Moringa of Moringaceae family there are 13 species (namely,
M. arborea, indigenous to Kenya;
M. rivae indigenous to Kenya and Ethiopia;
M. borziana, indigenous to Somalia and Kenia;
M. pygmaea indigenous to Somalia;
M. longituba indigenous to Kenia, Ethiopia and Somalia;
M. stenopetala indigenous to Kenya and Ethiopia;
M ruspoliana indigenous to Ethiopia;
M. ovalifolia indigenous to Namibia and Angola;
M. drouhardii,
M. hildebrandi indigenous to Madagascar;
M. peregrine indigenous o Red sea and Horn of Africa,
M. concanensis,
Moringa oleifera indigenous to sub-Himalayan tracts of Northern India [
1]), among which
Moringa oleifera (
Figure 1) has so far become the most used and studied.
This species is a fast growing soft wood tree that can reach 12 m in height and is indigenous to the Himalayan foothills (northern India Pakistan and Nepal) [
2,
3]. Its multiple uses and potential attracted the attention of farmers and researchers in past historical eras. Ayurvedic traditional medicine says that
Moringa oleifera can prevent 300 diseases and its leaves have been exploited both for preventive and curative purposes [
4]. Moreover, a study in the Virudhunagar district of Tamil Nadu India reports Moringa among the species utilized by traditional Siddha healers [
5]. Ancient Egyptians used
Moringa oleifera oil for its cosmetic value and skin preparation [
6]; even if the species never became popular among Greeks and Romans, they were aware of its medical properties [
7].
Moringa oleifera has been grown and consumed in its original areas until recently (the 1990s) when a few researchers started to study its potential use in clarifying water treatments, while only later were its nutritional and medical properties “discovered” and the species was spread throughout almost all tropical countries. In 2001, the first international conference on
Moringa oleifera was held in Tanzania and since then the number of congresses and studies increased disseminating the information about the incredible properties of
Moringa oleifera. Now this species has been dubbed “miracle tree”, or “natural gift”, or “mother’s best friend”.
Moringa oleifera grows in any tropical and subtropical country with peculiar environmental features, namely, dry to moist tropical or subtropical clime, with annual precipitation of 760 to 2500 mm (it requires less than 800 mm irrigation) and temperature between 18 and 28 °C. It grows in any soil type, but heavy clay and waterlogged, with pH between 4.5 and 8, at an altitude up to 2000 m [
8,
9].
A study on local uses and geographical distribution of
Moringa oleifera [
10] that covers the major agro-ecological region in Nigeria, clearly established that “though considered a not indigenous species,
Moringa oleifera has found wide acceptance among various ethnic Nigeria, who have exploited different uses (e.g., food, medicine, fodder
etc.).
Nowadays,
Moringa oleifera and its derivatives are distributed mainly in Middle East, African and Asian countries [
11] and are still spreading to other areas.
Figure 1.
(a) A tree of Moringa oleifera; (b) Moringa flowers and leaves.
Figure 1.
(a) A tree of Moringa oleifera; (b) Moringa flowers and leaves.
2. Cultivation and Production
Moringa oleifera development is achieved in two main ways: sowing and cutting.
Traditionally in Sudan the seeds are preferred while vegetative propagation is common in India, Indonesia and in some areas of West Africa [
8].
Sowing requires selection of the seeds, when they are easily available and human labor is limited, while the possibility to transplant seedlings allows flexibility in field planting even if it requires extra labor and costs.
Seeds germinate within two weeks, at a maximum 2 cm depth. When sowing is planned in nursery, the seedlings can be transplanted when they reach about 30 cm (3–6 weeks after germination) [
12].
The number of seeds per kilogram ranges from 3000 to 9000, depending on the variety, with a germination rate of 80%–90% for ideal storage conditions (3 °C, 5%–8% moisture). However, the viability decreases if seeds remain at ambient temperature and high relative humidity, their germination rate dropping to 7.5% after three months [
3,
13].
Cutting is preferred when seeds availability is scarce and/or when labor is not a limiting factor. Ramachandran
et al. [
14] reports that plants raised from seeds produce fruits of poorer quality, while Animashaun
et al. [
15] suggest that trees grown from seeds develop longer roots (an advantage for stabilization and access to water) compare to that grown from cuttings that have much shorter roots.
When hard woodcuttings (1–2 m long 4–16 cm diameter [
8,
15]) from adult trees are planted during the rainy season burying one third in the soil, they readily develop roots that in few months reach a considerable size [
16].
Moringa oleifera is an exceptionally fast growing tree, in three months it can be 3 m high and in few years reaches 12 m if it is left to growth naturally. Since the tree vigorously re-sprouts after cutting, pruning or pollarding are usually practiced to enhance lateral branching and give the tree a bush shape in order to facilitate the harvest. Nevertheless, since literature reports about the good practice management of
Moringa oleifera are scant, practical trials are needed [
12]. Leaves and seeds are the parts of the plant of interest. Accordingly, the spatial distribution in planting
Moringa oleifera trees is designed to facilitate the relevant harvest and the management practices.
For production of leaves,
Moringa oleifera plantation can be designed as follows:
- (i)
intensive production with spacing ranging from 10 cm × 10 cm to 20 cm × 20 cm, harvest interval between 35 to 45 days, irrigation and fertilization are needed;
- (ii)
semintensive production with spacing about 50 cm × 100 cm, harvest interval between 50 to 60 days, irrigation and fertilization suggested;
- (iii)
integrate in an agroforestry system with spacing distance of 2–4 m between rows, harvest interval around 60 days, fertilization and irrigation not strictly necessary.
Production decreases from intensive production to less dense spacing (agroforestry system), although a tremendous variability can be observed for a given spatial distribution and the same cultivation management. For example, the yield of an intensive plantation can range from 580 to 40 m/ha/year [
15], being season dependent with the largest yield in wet or cold season. There is a need for further studies to assess optimum spacing and harvest intervals that comply with the different climates and production systems [
17,
18,
19]. Harvest can be mechanical or manual. Shoots are cut at a 0.5–1 m height above the ground; but leaves can be picked directly off the tree; this practice, however, albeit quicker, leads to a less vigorous re-growth.
For the production of seed a low density plantation has a positive effect on yields: typically 2.5 × 2.5 m or 3 × 3 m triangular pattern [
20]. Fruits (trilobite capsule), referred as pods (brown color and dry and split longitudinally), ripen about three months after flowering and must be harvested as soon as possible. Each pod usually contains about 26 1-cm diameter seeds lined by three whitish papery leaflets on the edge. Like for leaves, also the production of seed shows a tremendous variability. A single tree can produce from 15.000 to 25.000 seeds with an average weight of 0.3 gr per seed [
21]; moreover early flowering varieties produce pods in six month, while other varieties require more than one year. After pruning, branches develop new pods within 6 months [
1].
3. Genetic and Breeding
The major
Moringa oleifera producer is India with an annual production of 1.1 to 1.3 million tons of tender fruits from an area of 380 km
2 [
22]. Information about the production in other countries is scarce. The great interest in
Moringa oleifera does not concern its commercial value, being mainly related to its multipurpose uses and its ability to guarantee a reliable yield, while other crops cannot, in countries where people are mostly at risk of suffering from nutritional deficiencies. Indeed its cultivation is localized in developing countries where different parts of the plant are utilized: seeds for oil and water purification; leaves, seeds and fruits for their high nutritional value (nutritional integrator); leaves and seeds for biomass and animal feeding; different parts in traditional medicine. Moreover, Moringa has been planted around the world and is naturalized in many areas (
i.e., almost the entire tropical belt) increasing the variability of the species.
As Moringa is a cross-pollinated tree, high heterogeneity in form and yield is expected. Several works indeed report variability in flowering time [
23] (from annual type to perennial type), tree nature (from deciduous to evergreen), tree shape (from semi spread to upright), resistance to hairy caterpillar [
22,
23,
24], flowering time (
i.e., some tree flowering throughout the year while others flower in two distinct season) [
14].
Although Moringa oleifera shows diversification in many characters and high morphological variability, which may become a resource for its improvement, the major factors that limit productivity are the absence of elite varieties adapted to local conditions and the use of seeds obtained through open pollination from plants in the planted area. Furthermore, despite the various uses of Moringa oleifera and its morphological differentiation, the number of accessions to collections and active germplasm banks are incipient across the world.
Many ecotypes are present in India: Jaffna (soft and taste fruits), Chavakacheri murungai (similar to Jaffna), Chemmurungai (red tipped fruits), Kadumurungai (small and inferior fruits) Palmurungai (bitter taste), Punamurungai (similar to Palmurungai), Kodikalmurungai (short fruit), Palmurungai, Puna Murungai and Kodikkal Murungai and wild Kadumurunga [
14,
25]. Recently two varieties (PKM-1; PKM2) have been developed at Horti Nursery Networks, Tamil Nadu, India, to improve pod production: usually those varieties are grown as annual; after two harvests the tree is dragged out and new seedlings are planted [
26]. At Kerala Agricultural University (India) several varieties have been developed and available.
Outside India there are research centers focused on Moringa oleifera improvement across the world: AVDRC (Taiwan), Rural development initiative (Zambia), Moringa Philippines foundation (Philippines) Moringa community (Zambia).
In spite of the great variability of Moringa oleifera no institutions have a germplasm bank or data base with either cultivated or spontaneous accessions. The divergence between genetic variability inherent to the species and poor variability reflected in germplasm banks should be fixed since it represents an obstacle for the progress of breeding programs.
Moringa cytological studies revealed that
Moringa oleifera has 2c genome size of 1.2 pg [
27] and it is a true diploid with 2
n = 28 [
14]. Since 1999 molecular markers have become standards for the genetic characterization of
Moringa oleifera. Studies started with the use of dominant markers until the development of co-dominant markers (in 2010) that allow distinction between homozygotes and heterozygotes, which provides optimal genetic information profiles.
Interestingly, out of 2857 scientific publications on Moringa oleifera in the primary database (Web of Science), only 12 include genetic characterization based on molecular markers. Furthermore, only 77 fragments of DNA and RNA sequences are available (data from NCBI nucleotide database). This means that the genetic approach and its potential application in breeding programs are just at the beginning step. The common aim of almost all the studies is the genetic diversification among different populations and/or accessions: dominant markers are the most used (66% of all papers).
In spite of the limited range of dominant markers (heterozygote cannot be distinguished from homozygote specimens), the studies among commercial, cultivated or natural accessions have contributed to the understanding of genetic variability of
Moringa oleifera. In this context, Amplified Fragment Length Polymorphism (AFLP) and Random Amplified Polymorphic DNA (RAPD) analyses along with Inter-Simple Sequence Repeat (ISSR) and cytochrome P
450 were used. Muluvi
et al. [
28] used AFLP to investigate seven natural populations from India and introduced populations in Malawi and Kenya. Authors found a significant level of population differentiation and separation of genotypes based on geographical origin. Moreover, high portion of genetic variability was within Indian accessions. In line with these findings, the authors argued that Kenya populations presumably came from India, as suggested by the small number of genetically related accessions. Thank to the same molecular markers [
29] the outcrossing rate in
Moringa oleifera was detected: 26% of selfing in
Moringa trees. This evidence had a strong impact on the breeding program, as inbreed lines and hybridization allowed improvement of the species.
RAPD were used by different authors to investigate cultivated and non-cultivated population of Tanzania [
30], different accessions in Nigeria [
31], accessions present in Embra Cosatal Teblelands Sergipe germplasm bank in Brazil [
32], commercially grown varieties in India [
33], new genotype developed in different countries (Thailand, USA, India and Malaysia, Tanzania, Taiwan) [
34], and further accessions in Nigeria [
35]. All these studies showed the higher level of genetic diversity in natural population with respect to the cultivated ones. Cultivated accessions present in the considered germplasm banks are genetically close and need to be widen to promote increased diversity and used in breeding programs. Many studies disagree with Muluvi’s conclusions that a significant level of population differentiation and separation of genotypes can be based on the geographical origin. Indeed no clusters were found according to geographical origins. This could be due to the planting spread that produced a high rate of gene flow through cross-pollination. Interestingly, Popoola
et al. [
35] investigated morpho-metric characters along with molecular markers and showed a good correlation between 100 seeds weight with pod length, pod weight, number of seeds per lobule and number of seed per pod.
Studies on
Moringa oleifera with co-dominat markers started in 2010 when Wu
et al. [
36] developed microsatellite markers. The first successful estimates of genetic diversity and population structure were obtained with Simple Sequence Repeat (SSR) in 2013 by Shahazad
et al. [
37]. These authors evaluated accessions collected in different locations of Pakistan and accessions from different countries (India, Tanzania, Senegal, Mozambique, Zimbabwe, Florida, Mexico, Haiti, Belize) obtained from Educational Concerns foe Hunger Organization (ECHO). They found high genetic diversity in wild Pakistan accessions, whereas low genetic diversity in ECHO accessions. Moreover, ECHO accessions are more similar to those of a single province of Pakistan. Most likely, British colonialists introduced
Moringa oleifera in early of twentieth century in Africa from India, while in the 1784 an Englishman took
Moringa oleifera over to Jamaica [
37]. The export pathway was restricted to Indian coastal region (where most movement of goods and people took place) and involved a relatively small number of accessions that belonged to a common or few populations. This explains the low genetic variability within ECHO accessions with respect to the Pakistan ones.
Later on, a further investigation on twelve Indian populations, from northern and southern regions of India, was performed through SSR together with morphological markers [
38]. In this study too, individuals from various geographical areas were not significantly different genetically, while a large variability exist in Indian populations. Morphological analysis on fourteen quantitative and eleven qualitative characters showed correlation among some quantitative characters, e.g., between tree tallness with fruit girth, trunk girth with tree branching. More SSR were identified in 2014 thanks to EST examination involving several plant species [
39] and not utilized so far in
Moringa oleifera genetic investigation.
Even if all the reported studies are valuables and have a tremendous importance for conservation, selection and collection of Moringa oleifera seeds, same questions are still to be addresses in order to develop an improved Moringa oleifera cultivation. Considering the cultivation challenges, some research activities should be prioritized: (i) collection and characterization of world accessions both cultivated and natural in order to obtain a true understanding of the genetic diversity and structure of Moringa oleifera; (ii) set a collaborative network among National and International Research Centre, O.N.G, farmers that already work on Moringa oleifera.
This will help scientists and producers to:
have a reliable access to information about genetics and materials to develop better Moringa varieties and technologies for farming practices: phenotypic characterization is a priority to evaluated the accessions;
ensure that Moringa production is improved along with best cultivation practice;
focus research on the association between phenotypic and molecular data within the contest of breeding;
define maps (both association map and physical map) to identify genes that may confer resistance to biotic and abiotic stress and quantitative traits loci (QTL) for a possible introgression of genes into commercial and cultivated accessions.
Today next generation sequencing (NGS) [
40] is an approachable tool to discovery genome-wide genetic markers. This technique could be applied to species with no existing genome data like Moringa. Thank to NGS a saturated genetic map could be obtain within reasonable cost and time, in turn interesting characters could be identified and exploited in breeding programmes.
4. Traditional Uses
All plant parts of
Moringa oleifera are traditionally used for different purposes, but leaves are generally the most used [
10,
41]. In particular, they are used in human and animal nutrition and in the traditional medicine. Leaves are rich in protein, mineral, beta-carotene and antioxidant compounds, which are often lacking among the populations of underdeveloped or developing countries.
Moringa leaves are added to food preparations as integrators of the diet. In traditional medicine, these leaves are used to treat several ailments including malaria, typhoid fever, parasitic diseases, arthritis, swellings, cuts, diseases of the skin, genito-urinary ailments, hypertension and diabetes. They are also used to elicit lactation and boost the immune system (to treat HIV/AIDS related symptoms) [
10,
41,
42,
43,
44,
45], as well as cardiac stimulants and contraceptive remedy. One can directly consume either raw and dried leaves or the extract of an aqueous infusion.
Similarly, the use of seeds concerns both human nutrition and traditional medicine. Barks are boiled in water and soaked in alcohol to obtained drinks and infusions that can be used to treat stomach ailments (ease stomach pain, ulcer and aiding digestion), poor vision, joint pain, diabetes, anemia and hypertension [
10,
43], toothache, hemorrhoids, uterine disorder [
10,
44]. In a well known practice,
Moringa seeds are used to sediment impurities of water [
10].
Roots are soaked in water or alcohol and boiled with other herbs to obtained drinks and infusions as remedies for toothache, as anthelmintic and antiparalytic [
10,
41,
42] drugs and as sex enhancers.
Finally, flowers are used to produce aphrodisiac substances and to treat inflammations, muscle diseases, hysteria, tumors and enlargement of the spleen [
42,
44].
5. Non Food or Medicinal Uses
Beyond the uses of Moringa as a food and for human health, other possible uses exist. It can be used as a natural plant growth enhancer; indeed leaves are rich in zeatin (a plant hormone belong to the cytokinin group). Leaf extracts can stimulate plant growth and increasing crop yield. Researches performed using a spray based on leaf extracts of wheat, maize and rice support the wide range of beneficial effect on crops [
46].
Moringa seed powder can be used for water purification, replacing dangerous and expensive chemicals such as aluminum sulfate [
10].
Interestingly, leaf extracts and also seed extracts show biopesticide activity, effective against larvae and adults of
Trigoderma granarium and can reduce the incidence of fungi on groundnut seeds [
46].
One of the interesting applications of Moringa seeds is their utilization as biomass for biodiesel production.
Due to the increasing energy demand and environmental problems associated with fossil fuels, the improvement of alternative fuels and renewable sources of energy is required. Biodiesel can replace petroleum-derived oil (petrodiesel), without any sulphur or aromatic compound and with lower emission of monoxides, hydrocarbons and particulates. Furthermore, biodiesel can reduce dependence on imported fuels: a crucial problem in developing countries [
47].
Moringa seeds have an oil content of 30%–40%, with a high-quality fatty acid composition
i.e., high oleic acid (>70%) [
48]. In addition they posses significant resistance to oxidative degradation. These proprieties make Moringa oil a good candidate to produce biodiesel after transesterificaton [
48,
49,
50]. Biswas and John [
51], in a study conducted in Australia, report that approximately 3030 kg of oil are required to produce 1000 liters of biodiesel. Furthermore, an equivalent of 3.03 tonnes/ha of oil seeds can be harvested from dry land, and 6.06 tonnes/ha can be harvested from irrigated land. Since biodiesel production with Moringa seed oil is a second generation production (
i.e., not in direct competition with existing farmland and with food crops) and as Moringa can grown on degraded land, studies suggest that Moringa biodiesel is an acceptable substitute to fossil fuels, even when compared against biodiesel derived from vegetable oil of other species.
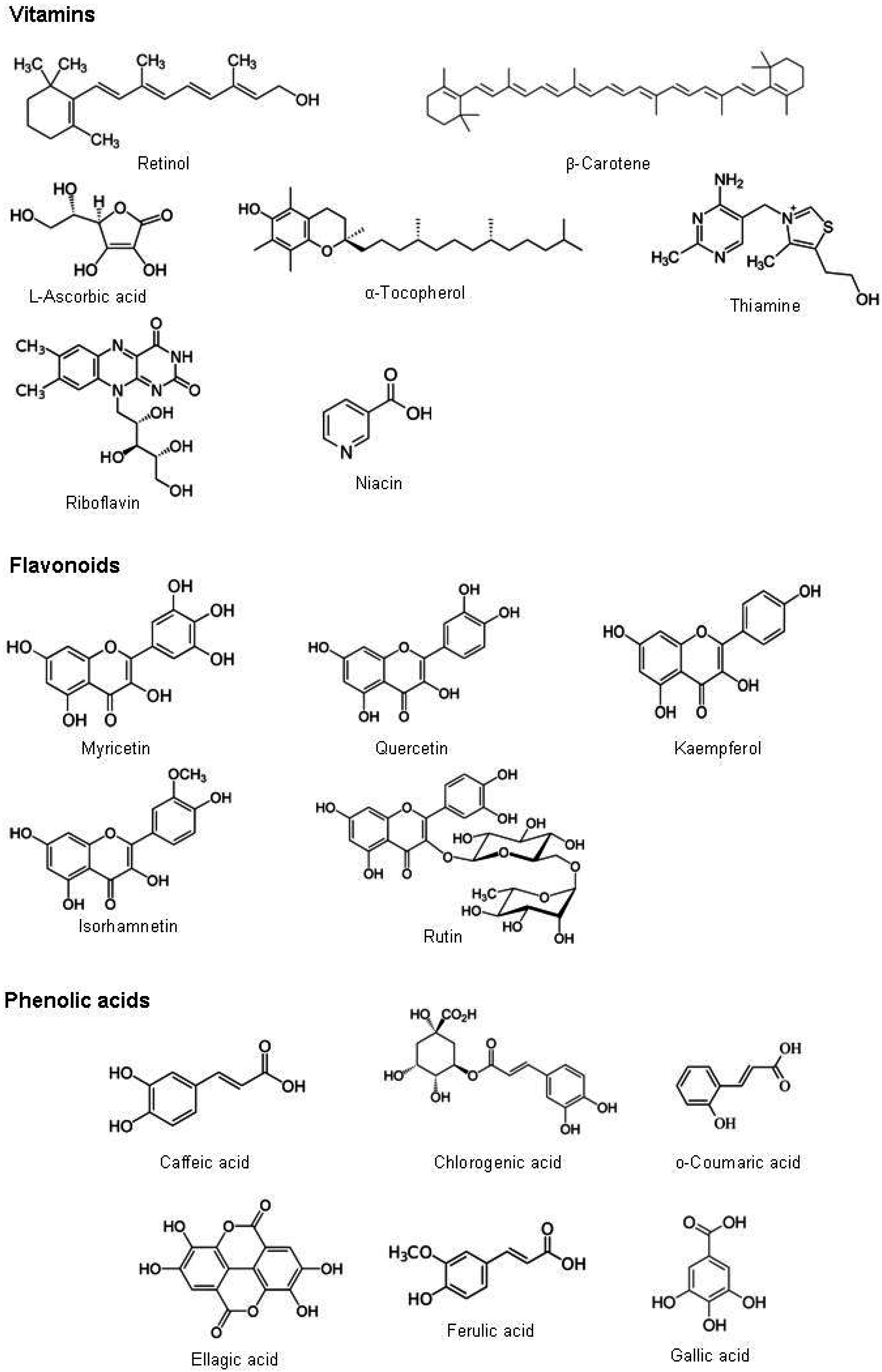
6. Phytochemistry
As Moringa oleifera leaves are most used part of the plant, we review articles concerning phytochemistry and pharmacological properties of leaves.
Several bioactive compounds were recognized in the leaves of
Moringa oleifera. They are grouped as vitamins, carotenoids, polyphenol, phenolic acids, flavonoids, alkaloids, glucosinolates, isothiocyanates, tannins, saponins and oxalates and phytates (
Figure 2). The amounts of different bioactive compounds found in
Moringa oleifera leaves and reported in literature are summarized in
Table 1,
Table 2,
Table 3,
Table 4,
Table 5,
Table 6,
Table 7,
Table 8 and
Table 9.
Figure 2.
Chemical structure of bioactive compounds found in Moringa oleifera leaves.
Figure 2.
Chemical structure of bioactive compounds found in Moringa oleifera leaves.
Table 1.
Vitamins content in Moringa oleifera leaves.
Table 1.
Vitamins content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Vitamins | | | | | | | | |
| Vitamin A | fresh | 11,300 IU | 45,200 IU | | N/A | N/A | India | [14] |
| fresh | 23,000 IU | 92,000 IU a | | N/A | N/A | Brazil | [52] |
| Vitamin B1–Thiamine | fresh | 0.06 mg/100 g | 0.24 mg/100 g | | N/A | N/A | India | [14] |
| fresh | 0.21 mg/100 g | 0.84 mg/100 g | | N/A | N/A | N/A | [53] |
| fresh | 0.6 mg/100 g | 2.58 mg/100 g | | N/A | Microbiological method | India | [54] |
| dried | 2.64 mg/100 g | 2.85 mg/100 g | N/A | N/A | N/A | N/A | [53] |
| Vitamin B2–Riboflavin | fresh | 0.05 mg/100 g | 0.20 mg/100 g | | N/A | N/A | India | [14] |
| fresh | 0.05 mg/100 g | 0.20 mg/100 g | | N/A | N/A | N/A | [53] |
| fresh | 0.17 mg/100 g | 0.726 mg/100 g | | N/A | Microbiological method | India | [54] |
| dried | 20.5 mg/100 g | 22.16 mg/100 g | N/A | N/A | N/A | N/A | [53] |
| Vitamin B3–Niacin | fresh | 0.8 mg/100 g | 3.20 mg/100 g | | N/A | N/A | India | [14] |
| fresh | 0.8 mg/100 g | 3.20 mg/100 g | | N/A | N/A | N/A | [53] |
| fresh | 0.82 mg/100 g | 3.5 mg/100 g | | N/A | Microbiological method | India | [54] |
| dried | 8.2 mg/100 g | 8.86 mg/100 g | N/A | N/A | N/A | N/A | [53] |
| Vitamin C–Ascorbic acid | fresh | 220 mg/100 g | 880 mg/100 g | | N/A | N/A | India | [14] |
| dried | 17.3 mg/100 g | 18.7 mg/100 g | N/A | N/A | N/A | N/A | [53] |
| dried | 92 mg/100 g | 92 mg/100 g | Sun-drying for 4 days | N/A | AOAC 2004 | India | [55] |
| 140 mg/100 g | 140 mg/100 g | Shadow-drying for 6 days |
| 56 mg/100 g | 56 mg/100 g | Oven-drying at 60 °C for 1 h |
| dried | 38.8 mg/100 g b | 38.8 mg/100 g b | Air-drying | Metaphosphoric acid | Indophenol titration | Pakistan | [56] |
| freeze-dried | 271 mg/100 g | 271 mg/100 g | Freeze-drying | Deionized water | Colorimetric method | Florida, USA | [57] |
| freeze-dried | 920 mg/100 g | 920 mg/100 g | Freeze-drying | 6% metaphosphoric acid | Titration against 2,6-dichlorophenolindophenol | Nicaragua | [58] |
| 840 mg/100 g | 840 mg/100 g | India |
| 680 mg/100 g | 680 mg/100 g | Niger |
| Vitamin E–Tocopherol | fresh | 9.0 mg/100 g | 16.21 mg/100 g | | N-hexane + ethyl acetate + BHT | Reverse-phase HPLC | Malaysia | [59] |
| dried | 113 mg/100 g | 122.16 mg/100 g | N/A | N/A | N/A | N/A | [53] |
| dried | 74.45 mg/100 g | 74.45 mg/100 g | Drying at 60 °C for 8 h | Microscale saponification and extraction with n-hexane | HPLC | Mexico | [60] |
| dried | 77.0 mg/100 g | 85.08 mg/100 g | Air-dried under shade | N/A | HPLC Fluorescence | South Africa | [61] |
Table 2.
Carotenoids content in Moringa oleifera leaves.
Table 2.
Carotenoids content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Carotenoids | | | | | | | | |
| β-carotene | fresh | 6.63 mg/100 g | 33.48 mg/100 g | | Acetone–n-hexane | HPLC | Taiwan | [62] |
| fresh | 6.8 mg/100 g | 27.22 mg/100 g | | N/A | N/A | N/A | [53] |
| dried | 36 mg/100 g | 36 mg/100 g | Sun-drying for 4 days | N/A | AOAC 2004 | India | [55] |
| 39.6 mg/100 g | 39.6 mg/100 g | Shadow-drying for 6 days |
| 37.8 mg/100 g | 37.8 mg/100 g | Oven-drying at 60 °C for 1 h |
| dried | 16.3 mg/100 g | 17.62 mg/100 g | N/A | N/A | N/A | N/A | [53] |
| dried | 18.5 mg/100 g | 20.44 mg/100 g | Air-dried under shade | N/A | HPLC | South Africa | [61] |
| freeze-dried | 66 mg/100 g | 66 mg/100 g | Freeze-drying | Acetone | HPLC | Florida, USA | [57] |
| Lutein | fresh | 6.94 mg/100 g | 35.05 mg/100 g | | Acetone–n-hexane | HPLC | Taiwan | [62] |
| freeze-dried | 102 mg/100 g | 102 mg/100 g | Freeze-drying | Acetone | HPLC | Florida, USA | [57] |
Table 3.
Polyphenols content in Moringa oleifera leaves.
Table 3.
Polyphenols content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Polyphenols | | | | | | | | |
| Total phenols | dried | 4581 mgGAE/100 g a | 4581 mgGAE/100 g a | Shade-drying | Water Soxhlet extraction for 18–20 h | Folin-Ciocalteau | India | [63] |
| 3602 mgGAE/100 g b | 3602 mgGAE/100 g b |
| dried | 3290 mgGAE/100 g | 3290 mgGAE/100 g | N/A | 50% MeOH | Folin-Ciocalteau | India | [64] |
| dried | 2090 mgGAE/100 g | 2090 mgGAE/100 g | N/A | 50% MeOH, 100% MeOH and water | Folin-Ciocalteau | India | [65] |
| dried | 10,504 mgGAE/100 g | 10,504 mgGAE/100 g | N/A | Water at 80 °C for 2 h | Folin-Ciocalteau | India | [66] |
| dried | 10,616 mgGAE/100 g c | 10,616 mgGAE/100 g c | Air-drying | 80% MeOH | Folin-Ciocalteau | Pakistan | [56] |
| dried | 10,300 mgGAE/100 g | 10,300 mgGAE/100 g | Air-drying | 100% MeOH | Extraction by shaker | Folin-Ciocalteau | Pakistan | [67] |
| 12,200 mgGAE/100 g | 12,200 mgGAE/100 g | 80% MeOH |
| 9720 mgGAE/100 g | 9720 mgGAE/100 g | 100% EtOH |
| 11,600 mgGAE/100 g | 11,600 mgGAE/100 g | 80% EtOH |
| dried | 9630 mgGAE/100 g | 9630 mgGAE/100 g | Air-drying | 100% MeOH | Extraction by reflux | Folin-Ciocalteau | Pakistan | [67] |
| 10,700 mgGAE/100 g | 10,700 mgGAE/100 g | 80% MeOH |
| 6160 mgGAE/100 g | 6160 mgGAE/100 g | 100% EtOH |
| 8210 mgGAE/100 g | 8210 mgGAE/100 g | 80% EtOH |
| dried | 2070 mg TAE/100 g | 2070 mg TAE/100 g | Air-drying | Acetone/Water (7:3) | Folin-Ciocalteau | India | [68] |
| dried | 1600 mgTEA/100 g d | 1600 mgTEA/100 g d | Air-drying | 80% EtOH | Folin-Ciocalteau | Nicaragua | [69] |
| 3400 mgTEA/100 g e | 3400 mgTEA/100 g e |
| dried | 5350 mgCAE/100 g | 5350 mgCAE/100 g | Oven-drying at 60 °C for 24 h | Maceration with 70% EtOH | Folin-Ciocalteau | Thailand | [70] |
| 2930 mgCAE/100 g | 2930 mgCAE/100 g | Maceration with 50% EtOH |
| 3710 mgCAE/100 g | 3710 mgCAE/100 g | Percolation with 70% EtOH |
| 3280 mgCAE/100 g | 3280 mgCAE/100 g | Percolation with 50% EtOH |
| 4550 mgCAE/100 g | 4550 mgCAE/100 g | Soxhlet extraction with 70% EtOH |
| 4460 mgCAE/100 g | 4460 mgCAE/100 g | Soxhlet extraction with 50% EtOH |
| freeze-dried | 1535.6 mgGAE/100 g | 1535.6 mgGAE/100 g | Freeze-drying | 80% EtOH | Folin-Ciocalteau | Florida, USA | [57] |
Table 4.
Phenolic acids content in Moringa oleifera leaves.
Table 4.
Phenolic acids content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Phenolic acids | | | | | | | | |
| Caffeic acid | dried | ND | ND | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 0.409 mg/g | 0.409 mg/g | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| freeze-dried | 0.536 mg/g | 0.536 mg/g | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
| Chlorogenic acid | dried | 0.018 mg/g | 0.018 mg/g | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 0.489 mg/g | 0.489 mg/g | N/A | Water at 80 °C for 2 h | HPLC and MS/MS | India | [66] |
| o-Coumaric acid | freeze-dried | 6.457 mg/g | 6.457 mg/g | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
| p-Coumaric acid | freeze-dried | ND | ND | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
| Ellagic acid | dried | ND | ND | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| dried | 0.009 mg/g | 0.018 mg/g | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 0.189 mg/g | 0.189 mg/g | N/A | Water at 80 °C for 2 h | HPLC and MS/MS | India | [66] |
| Ferulic acid | dried | 0.078 mg/g | 0.078 mg/g | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 0.078 mg/g | 0.078 mg/g | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| dried | 0.128 mg/g | 0.128 mg/g | N/A | Water at 80°C for 2 h | HPLC and MS/MS | India | [66] |
| Gallic acid | dried | ND | ND | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| dried | 1.034 mg/g | 1.034 mg/g | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 1.034 mg/g | 1.034 mg/g | N/A | Water at 80 °C for 2 h | HPLC and MS/MS | India | [66] |
| Gentistic acid | freeze-dried | ND | ND | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
| Sinapic acid | freeze-dried | ND | ND | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
| Syringic acid | freeze-dried | ND | ND | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
Table 5.
Flavonoids content in Moringa oleifera leaves.
Table 5.
Flavonoids content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Flavonoids | | | | | | | | |
| Total flavonoids | dried | 1.29 mg/g a | 5.059 mg/g a | Vacuum-drying | MeOH + HCl + ascorbic acid | HPLC-DAD | Taiwan | [71] |
| dried | 6.0 mg/g a,b | 6.0 mg/g a,b | Air-drying | 70% MeOH + 0.1% acetic acid | LC/MS | Ghana | [72] |
| 7.03 mg/g a,b | 7.03 mg/g a,b | Senegal |
| 12.16 mg/g a,b | 12.16 mg/g a,b | Zambia |
| dried | 31.28 mgQE/g | 31.28 mgQE/g | N/A | Water at 80 °C for 2 h | HPLC and MS/MS | India | [66] |
| dried | 27.0 mgQE/g c | 27.0 mgQE/g c | Shade-drying | Water Soxhlet extraction for 18–20 h | Colorimetric method | India | [63] |
| 15.0 mgQE/g d | 15.0 mgQE/g d |
| dried | 96.12 mg ECE/100g e | 96.12 mg ECE/100 g e | Air-drying | 80% MeOH | Colorimetric method | Pakistan | [56] |
| dried | 60.6 mgCE/g | 60.6 mgCE/g | Air-drying | 100% MeOH | Extraction by shaker | Spectrophotometric method | Pakistan | [67] |
| 86.6 mgCE/g | 86.6 mgCE/g | 80% MeOH |
| 53.3 mgCE/g | 53.3 mgCE/g | 100% EtOH |
| 62.1 mgCE/g | 62.1 mgCE/g | 80% EtOH |
| dried | 59.0 mgCE/g | 59.0 mgCE/g | Air-drying | 100% MeOH | Extraction by reflux | Spectrophotometric method | Pakistan | [67] |
| 72.9 mgCE/g | 72.9 mgCE/g | 80% MeOH |
| 41.9 mgCE/g | 41.9 mgCE/g | 100% MeOH |
| 53.1 mgCE/g | 53.1 mgCE/g | 80% EtOH |
| dried | 25.1 mgIQE/g | 25.1 mgIQE/g | Oven-drying at 60° C for 24 h | Maceration with 70% EtOH | Colorimetric method | Thailand | [70] |
| 12.3 mgIQE/g | 12.3 mgIQE/g | Maceration with 50% EtOH |
| 18.0 mgIQE/g | 18.0 mgIQE/g | Percolation with 70% EtOH |
| 14.6 mgIQE/g | 14.6 mgIQE/g | Percolation with 50% EtOH |
| 24.5 mgIQE/g | 24.5 mgIQE/g | Soxhlet extraction with 70% EtOH |
| 12.7 mgIQE/g | 12.7 mgIQE/g | Soxhlet extraction with 50% EtOH |
| freeze-dried | 61.62 mgRE/g | 61.62 mgRE/g | Freeze-drying | 80% EtOH | Spectrophotometric method | Florida, USA | [57] |
| freeze-dried | 44.3 mgRE/g | 44.3 mgRE/g | Freeze-drying | 80% MeOH | Spectrophotometric method | Nicaragua India Niger | [58] |
| 21.0 mgRE/g | 21.0 mgRE/g | Nicaragua India Niger |
| 38.1 mgRE/g | 38.1 mgRE/g | Nicaragua India Niger |
| Apigenin | dried | ND | ND | N/A | MeOH + HCl + ascorbic acid | HPLC | Taiwan | [71] |
| Daidzein | dried | ND | ND | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| Epicatechin | freeze-dried | 5.68 mg/g | 5.68 mg/g | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
| Genistein | dried | ND | ND | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| Isorhamnetin | dried | 0.03 mg/g | 0.118 mg/g | Vacuum-drying | MeOH + HCl + ascorbic acid | HPLC | Taiwan | [71] |
| freeze-dried | 0.13 mg/g f | 0.52 mg/g f,g | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 0.18 mg/g h | 0.72 mg/g g,h |
| Kaempferol | dried | 0.04 mg/g | 0.04 mg/g | Air-drying | MeOH + 1% v/v HCl + TBHQ | HPLC | Pakistan | [74] |
| dried | ND | ND | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 2.360 mg/g | 2.360 mg/g | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| dried | 0.198 mg/g | 0.198 mg/g | N/A | Water at 80°C for 2 h | HPLC and MS/MS | India | [66] |
| dried | 0.36 mg/g | 1.412 mg/g | Vacuum-drying | MeOH + HCl + 10 mg ascorbic acid | HPLC | Taiwan | [71] |
| dried | 0.8 mg/g | 0.8 mg/g | Air-drying | 70% MeOH + 0.1% acetic acid | LC/MS | Ghana | [72] |
| 1.23 mg/g | 1.23 mg/g | Senegal |
| 4.59 mg/g | 4.59 mg/g | Zambia |
| freeze-dried | 0.98 mg/g f | 3.92 mg/g f,g | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 0.54 mg/g h | 2.16 mg/g g,h |
| freeze-dried | 2.25 mg/g | 2.25 mg/g | Freeze-drying | 80% MeOH | HPLC-DAD | Nicaragua | [58] |
| 1.75 mg/g | 1.75 mg/g | India |
| 1.05 mg/g | 1.05 mg/g | Niger |
| freeze-dried | 2.9 mg/g d | 2.9 mg/g d | Freeze-drying | 70% MeOH | LC/MS | Malawi | [75] |
| 2.3 mg/g | 2.3 mg/g | Senegal |
| 3.5 mg/g | 3.5 mg/g | Nicaragua |
| 0.3 mg/g c | 0.3 mg/g c | ECHO |
| 0.16 mg/g d | 0.16 mg/g d | ECHO |
| Luteolin | dried | ND | ND | N/A | MeOH + HCl + ascorbic acid | HPLC | Taiwan | [71] |
| Myricetin | dried | 5.804 mg/g | 5.804 mg/g | Air-drying | MeOH + 1% v/v HCl + TBHQ | HPLC | Pakistan | [74] |
| Quercetin | dried | 0.281 mg/g | 0.281 mg/g | Air-drying | MeOH + 1% v/v HCl + TBHQ | HPLC | Pakistan | [74] |
| dried | 0.207 mg/g | 0.207 mg/g | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| dried | 0.207 mg/g | 0.207 mg/g | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| dried | 0.807 mg/g | 0.807 mg/g | N/A | Water at 80 °C for 2 h | HPLC and MS/MS | India | [66] |
| dried | 0.90 mg/g | 3.529 mg/g | Vacuum-drying | MeOH + HCl + 10 mg ascorbic acid | HPLC | Taiwan | [71] |
| dried | 5.2 mg/g | 5.2 mg/g | Air-drying | 70% MeOH + 0.1% acetic acid | LC/MS | Ghana | [72] |
| 5.8 mg/g | 5.8 mg/g | Senegal |
| 7.57 mg/g | 7.57 mg/g | Zambia |
| freeze-dried | 3.21 mg/g f | 12.84 mg/g f,g | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 4.16 mg/g h | 16.64 mg/g g,h |
| freeze-dried | 9.26 mg/g | 9.26 mg/g | Freeze-drying | 80% MeOH | HPLC-DAD | Nicaragua | [58] |
| 6.34 mg/g | 6.34 mg/g | India |
| 7.70 mg/g | 7.70 mg/g | Niger |
| freeze-dried | 5.47 mg/g b | 5.47 mg/g b | Freeze-drying | 70% MeOH | LC/MS | Malawi | [75] |
| 9.1 mg/g | 9.1 mg/g | Senegal |
| 15.2 mg/g | 15.2 mg/g | Nicaragua |
| 0.58 mg/g c | 0.58 mg/g c | ECHO |
| 0.46 mg/g d | 0.46 mg/g d | ECHO |
| Rutin | dried | 0.390 mg/g | 0.390 mg/g | N/A | 50% MeOH, 100% MeOH and water | HPLC | India | [65] |
| dried | ND | ND | N/A | 50% MeOH | HPLC and MS/MS | India | [64] |
| freeze-dried | 1.674 mg/g | 1.674 mg/g | Freeze-drying | 80% EtOH | HPLC | Florida, USA | [57] |
Table 6.
Glucosinolates content in Moringa oleifera leaves.
Table 6.
Glucosinolates content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Glucosinolates | | | | | | | | |
| Benzyl | freeze-dried | ND a ND b | ND a ND b | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| freeze-dried | ND c ND d | ND c ND d | Freeze-drying | 70% MeOH | LC/MS | Many countries | [75] |
| 4-hydroxybenzyl (sinalbin) | freeze-dried | 0.59 mg/g a ND b | 2.36 mg/g a,e ND b,e | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 4-(α-L-rhamnopyranosyloxy)-benzyl | freeze-dried | 5.64 mg/g a | 22.56 mg/g a,e | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 5.46 mg/g b | 21.84 mg/g b,e |
| freeze-dried | 33.9 mg/g c | 33.9 mg/g c | Freeze-drying | 70% MeOH | LC/MS | Many countries | [75] |
| 59.4 mg/g d | 59.4 mg/g d |
| 4-O-(α-L-acetylrhamnopyranosyloxy)-benzyl isomer 1 | freeze-dried | 0.69 mg/g a | 2.76 mg/g a,e | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 0.54 mg/g b | 2.16 mg/g b,e |
| freeze-dried | 2.9 mg/g c | 2.9 mg/g c | Freeze-drying | 70% MeOH | LC/MS | Many countries | [75] |
| 5.0 mg/g d | 5.0 mg/g d |
| 4-O-(α-L-acetylrhamnopyranosyloxy)-benzyl isomer 2 | freeze-dried | 0.45 mg/g a | 1.80 mg/g a,e | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 0.38 mg/g b | 1.52 mg/g b,e |
| freeze-dried | 1.2 mg/g c | 1.2 mg/g c | Freeze-drying | 70% MeOH | LC/MS | Many countries | [75] |
| 1.5 mg/g d | 1.5 mg/g d |
| 4-O-(α-L-acetylrhamnopyranosyloxy)-benzyl isomer 3 | freeze-dried | 5.04 mg/g a | 20.16 mg/g a,e | Freeze-drying | 70% MeOH | HPLC-DAD-electrospray mass spectrometry | Ghana | [73] |
| 3.19 mg/g b | 12.76 mg/g b,e |
| freeze-dried | 17.4 mg/g c | 17.4 mg/g c | Freeze-drying | 70% MeOH | LC/MS | Many countries | [75] |
| 50.2 mg/g d | 50.2 mg/g d |
Table 7.
Tannins content in Moringa oleifera leaves.
Table 7.
Tannins content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Tannins | | | | | | | | |
| Total tannins | dried | 13.2 gTAE/kg | 13.2 gTAE/kg | Air-drying | Acetone/Water (7:3) | Folin-Ciocalteau modified | India | [68] |
| dried | ND a 14.0 gTAE/kg b | ND a 14.0 gTAE/kg b | Air-drying | 80% EtOH | Folin-Ciocalteau modified | Nicaragua | [69] |
| dried | 20.6 g/kg | 20.6 g/kg | Air-drying at 35 °C for 24 h | Double lipid extraction with n-hexane (1:5) | N/A | Brazil | [76] |
| freeze-dried | 12 g/kg | 12 g/kg | Freeze-drying | 80% MeOH | Folin-Ciocalteau modified | Nicaragua | [77] |
| freeze-dried | 5 g/kg | 5 g/kg | Freeze-drying | 80% EtOH | Folin-Ciocalteau modified | Niger | [78] |
| Condensed tannins | dried | 1.05 gLE/kg | 1.05 gLE/kg | Air-drying | Acetone/Water (7:3) | Butanol–HCl–iron method | India | [68] |
| dried | 3.12 g/kg | 3.12 g/kg | Air-dried under shade | N/A | Butanol–HCl–iron method | South Africa | [61] |
Table 8.
Saponins content in Moringa oleifera leaves.
Table 8.
Saponins content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Saponins | | | | | | | | |
| Total saponins | dried | 2.0 gDE/kg a | 2.0 gDE/kg a | Air-drying | 80% EtOH | Spectrophotometric method | Nicaragua | [69] |
| 50.0 gDE/kg b | 50.0 gDE/kg b |
| freeze-dried | 81 gDE/kg | 81 gDE/kg | Freeze-drying | 80% MeOH | Spectrophotometric method | Nicaragua | [77] |
| freeze-dried | 64 gDE/kg | 64 gDE/kg | Freeze-drying | 80% EtOH | Spectrophotometric method | Niger | [78] |
Table 9.
Oxalates and phytates content in Moringa oleifera leaves.
Table 9.
Oxalates and phytates content in Moringa oleifera leaves.
| Bioactive Compound | Leaves | Value Found in Literature | Value Express as Dry Weight | Drying Method | Extractive Method | Analytical Method | Country | Reference |
|---|
| Oxalates and phytates | | | | | | | | |
| Oxalates | dried | 430 mg/100 g | 430 mg/100 g | Sun-drying for 4 days | N/A | AOAC 2004 | India | [55] |
| 500 mg/100 g | 500 mg/100 g | Shadow-drying for 6 days |
| 450 mg/100 g | 450 mg/100 g | Oven-drying at 60 °C for 1 h |
| dried | 1050 mg/100 g | 1050 mg/100 g | Air-drying at 35 °C for 24 h | Double lipid extraction with n-hexane (1:5) | N/A | Brazil | [76] |
| Phytates | dried | 25.0 g/kg a | 25.0 g/kg a | Air-drying | 3.5% HCl for 1 h | Colorimetric method | Nicaragua | [69] |
| 31.0 g/kg b | 31.0 g/kg b |
| freeze-dried | 21.0 g/kg | 21.0 g/kg | Freeze-drying | 3.5% HCl for 1 h | Colorimetric method | Nicaragua | [77] |
| freeze-dried | 23.0 g/kg | 23.0 g/kg | Freeze-drying | 3.5% HCl for 1 h | Colorimetric method | Niger | [78] |
6.1. Vitamins
Fresh leaves of
Moringa oleifera are reported to contain 11,300–23,000 IU of vitamin A [
14,
52]. Vitamin A plays key roles in many physiological processes such as vision, reproduction, embryonic growth and development, immune competence, cell differentiation, cell proliferation and apoptosis, maintenance of epithelial tissue, and brain function. Its deficiency is still prevalent in many developing countries, and considered responsible for child and maternal mortality [
79].
Fresh leaves of
Moringa oleifara are also a good source of carotenoids with pro-vitamin A action. They contain 6.6–6.8 mg/100 g [
53,
62] of β-carotene, greater that carrots, pumpkin and apricots (6.9, 3.6 and 2.2 mg/100 g, respectively) [
80].
β-carotene is more concentrated in the dried leaves, with amounts ranging from 17.6 to 39.6 mg/100 g of dry weight (DW) [
53,
55,
61]. This wide range may be explained by the different environmental conditions existing among different origin countries, genetic of the plant, drying method [
55] and the different extraction and analysis methods employed as well. Freeze-drying seems to be the most conservative dehydration method. In freeze-drying leaves the β-carotene content is approximately 66 mg/100 g [
57].
Moringa oleifera is an interesting source of vitamin C. Fresh leaves contain approximately 200 mg/100 g [
14], greater than orange [
80]. These amounts are of particular interest, as the vitamin C intervenes in the synthesis and metabolism of many compounds, like tyrosine, folic acid and tryptophan, hydroxylation of glycine, proline, lysine carnitine and catecholamine. It facilitates the conversion of cholesterol into bile acids and hence lowers blood cholesterol levels and increases the absorption of iron in the gut by reducing ferric to ferrous state. Finally, it acts as antioxidant, protecting the body from various deleterious effects of free radicals, pollutants and toxins [
81]. However, being vitamin C sensitive to heat and oxygen, it is rapidly oxidized, so much so that its concentration in the
Moringa oleifera dried leaves is lower than in the fresh leaves, dropping to 18.7 to 140 mg/100 g of DW [
53,
55,
56].
Difference in (i) environmental conditions in the various origin countries; (ii) genetic of the plant; (iii) drying method [
55] and (iv) different extraction and analysis methods, may explain the wide range of vitamin C content in
Moringa leaves reported in literature. Freeze-drying seem to better preserve vitamin C from oxidation, so much so that greater amounts of this vitamin were found in leaves undergone to freeze-drying soon after the collection. In these latter, vitamin C concentration ranges between 271 and 920 mg/100 g of DW [
57,
58].
Moringa oleifera fresh leaves are a good source of vitamin E (in particular α-tocopherol) and contain approximately 9.0 mg/100 g [
59] of this compound, similarly to nuts [
80]. Vitamin E acts mainly as liposoluble antioxidants, but it is also involved in the modulation of gene expression, inhibition of cell proliferation, platelet aggregation, monocyte adhesion and regulation of bone mass [
82]. Drying procedure determines a concentration of vitamin E up to values of 74.45–122.16 mg/100 g of DW [
53,
60,
61].
Among vitamins of group B, only thiamine, riboflavin and niacin seem present in
Moringa oleifera leaves. These vitamins mainly act as cofactors of many enzymes involved in the metabolism of nutrients and energy production, and their concentration in fresh leaves ranges between 0.06 and 0.6 mg/100 g, 0.05 and 0.17 mg/100 g and 0.8 and 0.82 mg/100 g for thiamine, riboflavin and niacin, respectively [
14,
53,
60], similarly to fruits and vegetable [
80]. Only one study reported the contribution of vitamin B1, B2 and B3 of dried leaves of
Moringa oleifera [
53]. Their concentrations were 2.85, 22.16 and 8.86 mg/100g of DW, respectively. However, the amount of riboflavin in dried leaves seems very high compared to that of fresh leaves. Further studies are needed to confirm these values. Finally, Girija
et al. showed an appreciable physiological availability of these three vitamins in leaves of
Moringa oleifera (61.6%, 51.5% and 39.9%, respectively) [
54].
We did not find studies about other vitamin of group B or vitamin D and K in Moringa oleifera leave; therefore further studies on this topic are needed.
6.2. Polyphenols
Moringa oleifera dried leaves are a great source of polyphenols. Their concentrations range from 2090 to 12,200 mgGAE/100 g of DW [
63,
64,
65,
66,
67] (or 1600 to 3400 mgTAE/100g of DW) [
68,
69]. These amounts are greater than those found in fruits and vegetable [
83,
84,
85]. The different environmental conditions in the various origin countries, the harvesting season [
56], the genetic of the plant, the drying method, the leaf maturity stage [
63] and the extractive method used [
67] may explain such wide range of reported values. Principal polyphenol compounds in
Moringa oleifera leaves are flavonoids and phenolic acids.
6.3. Flavonoids
Flavonoids are a sub-group of polyphenolic compounds having a benzo-γ-pyrone structure and are ubiquitous in plants, as they are synthesized in response to microbial infections [
86]. Epidemiological studies have consistently shown that high intake of flavonoids has protective effects against many infectious (bacterial and viral diseases) and degenerative diseases such as cardiovascular diseases, cancers, and other age-related diseases [
86,
87].
Moringa oleifera leaves are an interesting source of flavonoids compounds. Total flavonoids concentration in dried leaves ranges from 5.059 to 12.16 mg/g of DW [
71], namely, close to or larger than that in many fruits and vegetable normally consumed [
72,
88]. These values are indeed the overall sum of the amounts of single flavonoids. However, some flavonoids were studied only by some authors and, therefore, these amounts may be inaccurate. The total concentration of flavonoids in freeze-dried leaves ranges from 21.0 to 61.62 mgRE/g of DW [
57,
58]. Myricetin, quercetin and kaempferol are the main flavonoids found in
Moringa oleifera leaves. In dried leaves, myricetin concentration is approximately 5.804 mg/g of DW, while quercetin and kaempferol concentrations range from 0.207 to 7.57 mg/g of DW and not detectable amounts (ND) to 4.59 mg/g of DW, respectively [
64,
65,
66,
71,
72,
74]. Higher amounts were found in freeze-dried leaves. In particular, quercitin and kaempferol concentrations range from 5.47 to 16.64 mg/g and 1.5 to 3.5 mg/g of DW, respectively [
58,
73,
75]. Isorhamnetin concentration in dried leaves is approximately 0.118 mg/g of DW [
71], while, in freeze-dried leaves, its concentration is up to 7 times larger, ranging from 0.52 to 0.72 mg/g of DW [
73]. Other flavonoids, such as luteolin, apigenin, daidzein and genistein, were found in not detectable concentrations in
Moringa oleifera leaves [
65,
71]. However these compounds were investigated only in few studies and, therefore, further investigations are needed. In addition, in this case, the high inter-studies variations for these compounds may be explained taking into account different environmental conditions, harvesting season, genetic of the plant, drying method, leaf maturity stage, extraction method used and, finally, the different sensitivity of the analytical methods.
6.4. Phenolic Acids
Phenolic acids are a sub-group of phenolic compounds derived from hydroxybenzoic acid and hydroxycinnamic acid, naturally present in plants. Thanks to their documented effects on human health, the contribution of food-supplied phenolic acids is a subject of increasing interest. In particular, these compounds are mainly studied for their documented antioxidant, anti-inflammatory, antimutagenic and anticancer properties [
89,
90,
91,
92]. Particularly abundant in fruit and vegetables, phenolic acids were found in great amounts in
Moringa oleifera leaves too. In dried leaves, gallic acid seems to be the most abundant, with a concentration of approximately 1.034 mg/g of DW [
64,
66], although Bajpai
et al. [
65] only found poorly detectable amounts. The concentration of chlorogenic and caffeic acids ranges from 0.018 to 0.489 mg/g of DW and ND to 0.409 mg/g of DW, respectively [
64,
65,
66]. Lower, but appreciable, concentrations were found for ellagic and ferulic acids. Their concentrations range from ND to 0.189 mg/g and 0.078 to 0.128 mg/g of DW, respectively [
64,
65,
66]. Some of these compounds were found more concentrated in freeze-dried leaves. Specifically, Zhang
et al. [
57], in leaves harvested in Florida and subsequently freeze-dried, found approximately 6.457 mg/g of DW of o-coumaric acid and 0.536 mg/g of DW of caffeic acid, while p-coumaric, synaptic, gentistic and syringic acids were found in poorly detectable amounts [
57]. Like for the flavonoids, the different environmental conditions, harvesting season, genetic of the plant, drying method, leaf maturity stage, extraction method used and the different sensitivity of the analytical methods may have contributed to the high inter-study variation in the concentrations of phenolic acids in
Moringa oleifera leaves.
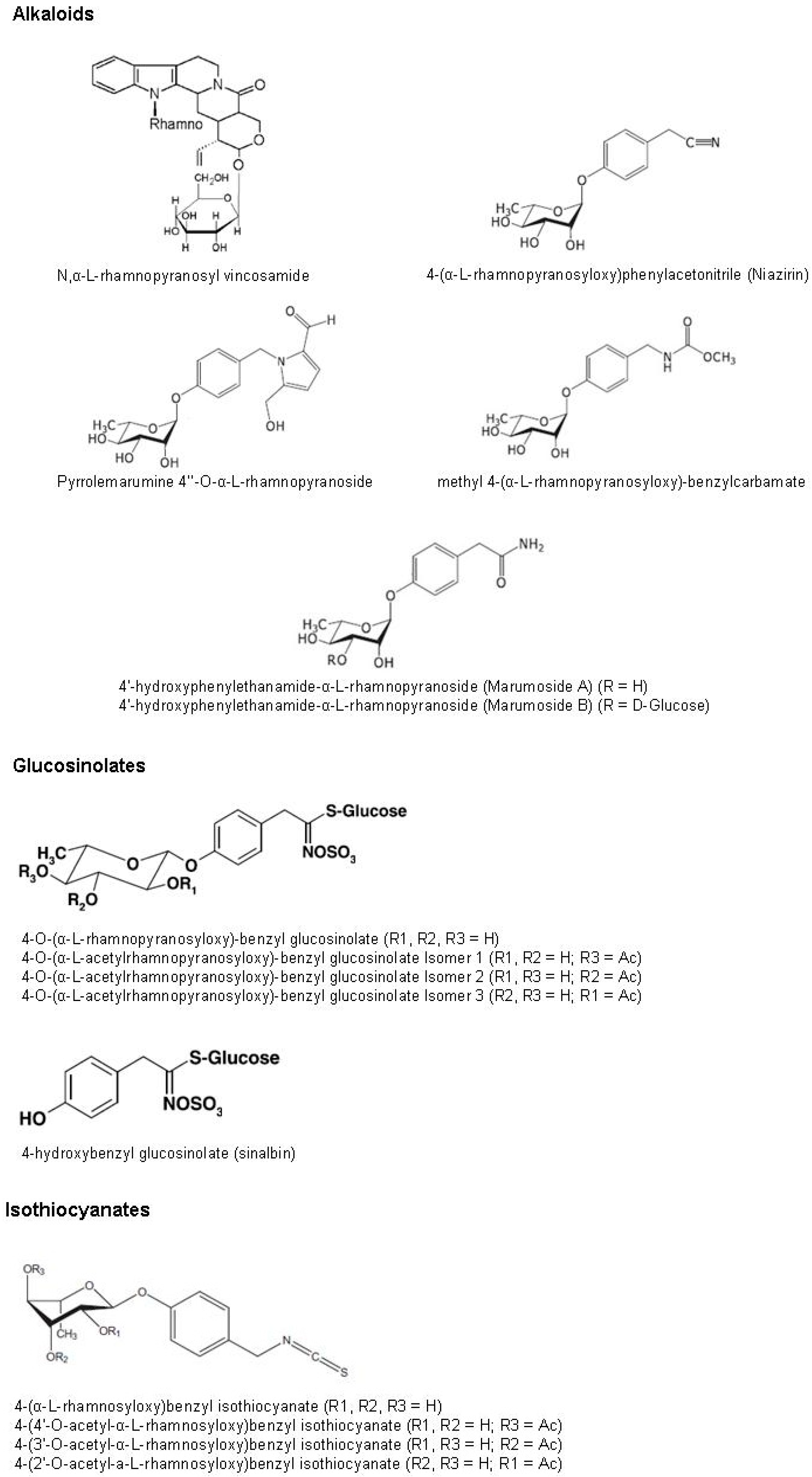
6.5. Alkaloids
Alkaloids are a group of naturally occurring chemical compounds that contain mostly basic nitrogen atoms. This nitrogen may occur in the form of a primary amine (RNH
2), a secondary amine (R
2NH) or a tertiary amine (R
3N). In addition to carbon, hydrogen and nitrogen, most alkaloids contain oxygen [
93]. Alkaloids are of particular interest thanks to their pharmacological properties. The presence of these compounds has been confirmed in
Moringa oleifera leaves [
45,
63]. Several of these compounds, such as
N,α-
l-rhamnopyranosyl vincosamide, 4-(α-
l-rhamnopyranosyloxy) phenylacetonitrile (niazirin), pyrrolemarumine 4′′-
O-α-
l-rhamnopyranoside, 4′-hydroxy phenylethanamide-α-
l-rhamnopyranoside (marumoside A) and its 3-
O-β-
d-glucopyranosyl-derivative (marumoside B) and methyl 4-(α-
l-rhamnopyranosyloxy)-benzylcarbamate, have been isolated in
Moringa oleifera leaves [
94,
95]. However, their amounts in the leaves are still unknown.
6.6. Glucosinolates and Isothiocyanates
Glucosinolates are a group of secondary metabolites in plants. Structurally they are β-
S-glucosides of thio-oxime-
O-sulfates and synthesized from amino acids. Appreciable amounts of these compounds were found in
Moringa oleifera leaves. In particular, around 116 and 63 mg/g of DW in young and older leaves, respectively, are reported [
75,
96]. These amounts are close to, and in some case larger than, those found in many cruciferous vegetables (e.g., broccoli, cabbage, radish), mainly sources of these compounds [
97]. 4-
O-(α-
l-rhamnopyranosyloxy)-benzyl glucosinolate has been identified as the dominant leaf glucosinolate of
Moringa oleifera and is accompanied by lower levels of three isomeric 4-
O-(α-
l-acetylrhamnopyranosyloxy)-benzyl glucosinolates, which reflect the three position of the acetyl group at the rhamnose moiety of the molecule [
75,
96]. The concentrations of these compounds seem affected by the physiological stage of the plant and by the maturity stage of the leaves. The concentration of 4-
O-(α-
l-rhamnopyranosyloxy)-benzyl glucosinolate ranges from 21.84 to 59.4 mg/g of DW, while the concentrations of the three isomer of 4-
O-(α-
l-acetylrhamnopyranosyloxy)-benzyl glucosinolates range from 2.16 to 5.0 mg/g of DW, 1.2 to 1.8 mg/g of DW and 12.76 to 50.2 mg/g of DW for isomer 1, 2 and 3, respectively [
73,
75]. Amaglo
et al. [
73] report the presence of 4-hydroxybenzyl (sinalbin), with a concentration ranging between ND and 2.36 mg/g of DW. Glucosinolates can be hydrolyzed by myrosinase to produce
d-glucose and various other degradation products like isothiocyanates [
98], which are also present in
Moringa oleifera leaves [
99,
100]. Both glucosinolates and isothiocyanates play an important role in health promoting and prevention of disease [
101].
6.7. Tannins
Tannins are water-soluble phenolic compounds that bind to and precipitate alkaloids, gelatin and other proteins. They exhibit various biological properties: anti-cancer, antiatherosclerotic, anti-inflammatory, anti-hepatoxic, antibacterial and anti-HIV replication activity [
102].
Moringa oleifera leaves are an appreciable source of tannins. Their concentrations range between 13.2 and 20.6 gTAE/kg [
68,
69,
76] in dried leaves and between 5.0 and 12.0 gTAE/kg in freeze-dried leaves [
77,
78]. These amounts are greater than concentrations found in nuts [
103], similar to those found in some plants [
104] and berries [
105], but much lower compared to the concenctrations found in other medicinal plants [
106].
6.8. Saponins
Saponins are a group of natural compounds that consist of an isoprenoidal-derived aglycone, designated genin or sapogenin, covalently linked to one or more sugar moieties [
107]. Even though some saponins have hemolytic side effects, they are studied for their anti-cancer properties [
108,
109]. Moringa oleifera leaves are a good source of saponins. Their concentration in dried leaves is approximately 50 gDE/kg of DW [
69], while in freeze-dried leaves it ranges between 64 and 81 gDE/kg of DW [
77,
78]. These amounts are greater than the concentrations found in other plants [
106], but slighty lower than ginseng root [
110], one of the mainly source of these compounds.
6.9. Oxalates and Phytates
Oxalates and phytates are anti-nutritional compounds as they bind minerals inhibiting the intestinal absorption. Moringa oleifera leaves present high contents of these compunds. Oxalates content of dried leaves range from 430 to 1050 mg/100 g of DW [
55,
76], similar to other plants rich in these compounds [
111], while phytates concentration range from 25 to 31 g/kg of DW [
69] in dried leaves and from 21 and 23 g/kg of DW in freeze-dried leaves [
77,
78]. These amounts are greater than those found in legumes and cereals [
112,
113], but lower than brans [
112].
8. Conclusions
Moringa oleifera is one of the most studied and used plants. Its uses stretch from food and medicinal uses to water purification, biopesticide and production of biodiesel.
Moringa shows diversifications in many features and high morphological variability which may become a resource for the conservation and the selection of Moringa oleifera germplasm. However, some questions still have to be addressed, i.e., collection and characterization of world accessions (both cultivated and natural) and setting a collaborative network among all institutions that already work on Moringa oleifera. This will help scientists and producers to have reliable access to information and materials and better develop Moringa. Moreover, researches focused on the association between phenotypical and molecular data and on genetic maps (both association map and physical map) in order to identify genes are needed within the contest of breeding. Next generation sequencing (NGS) could be an approachable tool to discover genome-wide genetic markers and building saturated genetic map within reasonable cost and time.
Nevertheless Moringa oleifera is an interesting plant for its contribution in bioactive compounds. In particular, leaves, the most used part of the plant, are rich in vitamins, carotenoids, polyphenol, phenolic acids, flavonoids, alkaloids, glucosinolates, isothiocyanates, tannins and saponins. In addition, even if leaves present high variation in the amounts of bioactive compounds as a result of the genetic characteristics of the plant, the environmental conditions to which the plant is subjected and the post-harvest treatments as well, they present greater amounts of these compounds than fruits, vegetables and other plants generally used in human nutrition. On the other hand, the high leaves content of oxalates and phytates could limit the intestinal adsorption of minerals. Therefore, this aspect should be taken in to account for future nutritional researches focused on using Moringa as minerals supplementation.
The high contribution in bioactive compounds may explain the pharmacological properties ascribed to Moringa oleifera leaves. Many in vitro and in vivo studies in animals have widely confirmed numerous pharmacological properties. However, few evidences on human beings are available. Therefore, it is too early to recommend Moringa oleifera leaves as medication in the prevention or treatment of diabetes, cardiovascular disease, dyslipidemia, cancer and infective diseases. Further studies aimed to confirm the pharmacological effects of moringa on human beings and, at the same time, ensuring its safety on human health consequently to a chronic or long-term use should be encouraged.