Rapid Characterization of Fatty Acids in Oleaginous Microalgae by Near-Infrared Spectroscopy
Abstract
:1. Introduction
2. Results
2.1. Algal Samples and Near-Infrared (NIR) Spectra
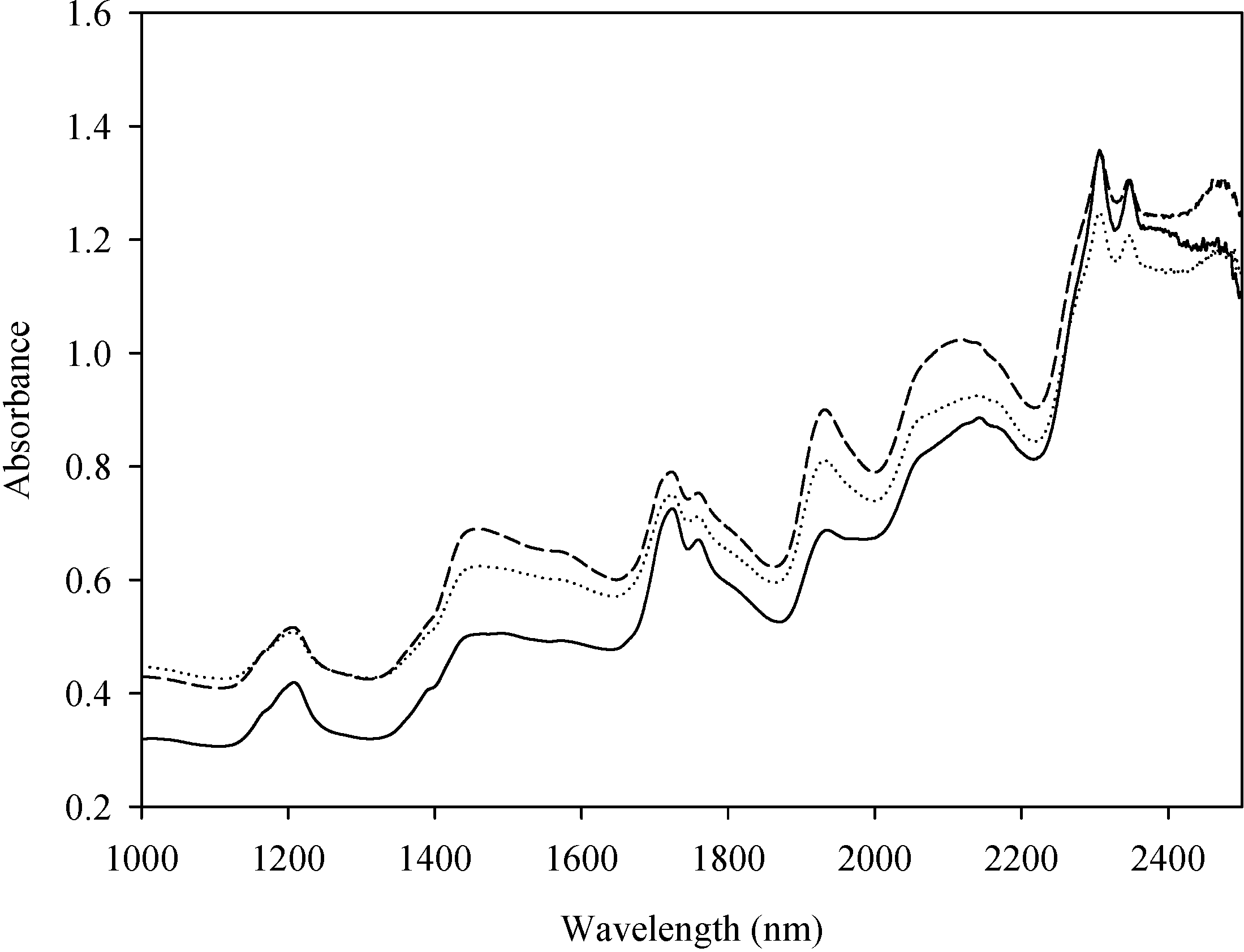
2.2. NIRS Models Based on C. vulgaris Data
| Fatty Acid | Calibration Set (45 Samples) | Validation Set (15 Samples) | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | Max. | Min. | S.D. | Mean | Max. | Min. | S.D. | |
| TFA | 303.42 | 468.56 | 176.64 | 78.12 | 314.27 | 446.49 | 233.76 | 63.65 |
| C16:0 | 59.29 | 114.55 | 29.00 | 23.41 | 62.33 | 105.28 | 40.78 | 19.61 |
| C18:0 | 21.06 | 42.23 | 5.99 | 10.17 | 23.34 | 40.99 | 12.55 | 9.28 |
| C18:1 | 67.81 | 112.97 | 29.90 | 21.39 | 71.68 | 104.22 | 47.59 | 17.54 |
| C18:2 | 78.52 | 86.93 | 65.43 | 5.67 | 78.49 | 86.62 | 72.17 | 4.16 |
| C18:3 | 63.62 | 108.11 | 33.20 | 19.40 | 66.24 | 100.15 | 47.63 | 15.15 |
| Model | RMSEC (mg/g Cell) | R2 b | RMSECV (mg/g Cell) | SEP (mg/g Cell) | r2 c | RPD |
|---|---|---|---|---|---|---|
| Models developed with WR I (CV-NIRS-WR I) | ||||||
| TFA | 5.81 | 0.997 | 7.15 | 7.23 | 0.994 | 10.83 |
| C16:0 | 2.44 | 0.994 | 3.31 | 3.35 | 0.985 | 7.02 |
| C18:0 | 2.53 | 0.969 | 2.68 | 2.94 | 0.951 | 3.76 |
| C18:1 | 2.56 | 0.993 | 3.38 | 2.47 | 0.989 | 6.27 |
| C18:2 | 2.65 | 0.888 | 3.16 | 3.95 | 0.604 | 1.78 |
| C18:3 | 2.56 | 0.992 | 3.13 | 3.44 | 0.976 | 6.15 |
| Models developed with WR II (CV-NIRS-WR II) | ||||||
| TFA | 4.41 | 0.998 | 5.28 | 6.47 | 0.997 | 14.68 |
| C16:0 | 1.68 | 0.997 | 2.19 | 1.95 | 0.995 | 10.59 |
| C18:0 | 1.54 | 0.989 | 1.62 | 1.61 | 0.984 | 6.22 |
| C18:1 | 2.85 | 0.991 | 2.90 | 2.36 | 0.991 | 7.31 |
| C18:2 | 1.96 | 0.940 | 2.18 | 3.98 | 0.678 | 2.58 |
| C18:3 | 1.59 | 0.997 | 2.01 | 2.68 | 0.984 | 9.50 |
2.3. NIRS Models Suitable for Three Species of Chlorella Simultaneously
| Fatty Acid | Calibration Set (119 Samples) | Validation Set (40 Samples) | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | Max. | Min. | S.D. | Mean | Max. | Min. | S.D. | |
| TFA | 289.35 | 468.56 | 95.08 | 93.92 | 297.14 | 463.58 | 93.00 | 90.86 |
| C16:0 | 46.57 | 114.55 | 17.13 | 18.71 | 49.48 | 111.97 | 16.64 | 21.62 |
| C18:0 | 17.96 | 41.39 | 1.03 | 8.45 | 19.46 | 42.23 | 0.93 | 9.74 |
| C18:1 | 100.96 | 198.52 | 22.13 | 50.87 | 98.59 | 201.93 | 26.10 | 47.86 |
| C18:2 | 63.00 | 120.39 | 24.94 | 21.28 | 65.33 | 118.96 | 24.37 | 21.09 |
| C18:3 | 39.05 | 108.11 | 5.70 | 23.14 | 43.50 | 100.15 | 5.99 | 25.50 |
| Model | RMSEC (mg/g Cell) | R2 a | RMSECV (mg/g Cell) | SEP (mg/g Cell) | r2 b | RPD |
|---|---|---|---|---|---|---|
| TFA | 14.68 | 0.988 | 18.81 | 24.16 | 0.964 | 4.98 |
| C16:0 | 4.41 | 0.968 | 5.21 | 5.77 | 0.964 | 3.58 |
| C18:0 | 2.36 | 0.961 | 2.56 | 2.87 | 0.957 | 3.29 |
| C18:1 | 8.37 | 0.986 | 10.55 | 11.56 | 0.970 | 4.81 |
| C18:2 | 6.78 | 0.949 | 7.25 | 7.67 | 0.932 | 2.92 |
| C18:3 | 4.27 | 0.981 | 4.80 | 5.49 | 0.977 | 4.80 |
3. Discussion
4. Experimental Section
4.1. Chlorella Species and Culture Conditions
4.2. Fatty Acid Analysis
4.3. NIR Spectra Collection
4.4. Regression Model Development
4.5. Calibration Performance
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y. Biodiesel from microalgae beats bioethanol. Trends Biotechnol. 2008, 26, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y.; Yan, J.Y. Energy from algae: Current status and future trends algal biofuels—A status report. Appl. Energy 2011, 88, 3277–3279. [Google Scholar] [CrossRef]
- Huang, G.H.; Chen, F.; Wei, D.; Zhang, X.W.; Chen, G. Biodiesel production by microalgal biotechnology. Appl. Energy 2010, 87, 38–46. [Google Scholar] [CrossRef]
- Liu, J.; Huang, J.C.; Sun, Z.; Zhong, Y.J.; Jiang, Y.; Chen, F. Differential lipid and fatty acid profiles of photoautotrophic and heterotrophic Chlorella zofingiensis: Assessment of algal oils for biodiesel production. Bioresour. Technol. 2011, 102, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Peng, X.; Luo, M.; Zhao, C.J.; Gu, C.B.; Zu, Y.G.; Fu, Y.J. Biodiesel production from Camptotheca acuminata seed oil catalyzed by novel Bronsted-Lewis acidic ionic liquid. Appl. Energy 2014, 115, 438–444. [Google Scholar] [CrossRef]
- Liu, J.; Huang, J.C.; Fan, K.W.; Jiang, Y.; Zhong, Y.J.; Sun, Z.; Chen, F. Production potential of Chlorella zofingienesis as a feedstock for biodiesel. Bioresour. Technol. 2010, 101, 8658–8663. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Huang, J.C.; Jiang, Y.; Chen, F. Molasses-based growth and production of oil and astaxanthin by Chlorella zofingiensis. Bioresour. Technol. 2012, 107, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zhou, Z.G.; Gerken, H.; Chen, F.; Liu, J. Screening and characterization of oleaginous Chlorella strains and exploration of photoautotrophic Chlorella protothecoides for oil production. Bioresour. Technol. 2014, 184, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, S.K.; Malik, A.; Vijay, V.K. Comparative evaluation of biomass production and bioenergy generation potential of Chlorella spp. through anaerobic digestion. Appl. Energy 2014, 114, 790–797. [Google Scholar] [CrossRef]
- Ashraful, A.M.; Masjuki, H.H.; Kalam, M.A.; Rahman, S.M.A.; Habibullah, M.; Syazwan, M. Study of the effect of storage time on the oxidation and thermal stability of various biodiesels and their blends. Energy Fuels 2014, 28, 1081–1089. [Google Scholar] [CrossRef]
- Knothe, G. “Designer” biodiesel: Optimizing fatty ester composition to improve fuel properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Park, J.Y.; Kim, D.K.; Lee, J.P.; Park, S.C.; Kim, Y.J.; Lee, J.S. Blending effects of biodiesels on oxidation stability and low temperature flow properties. Bioresour. Technol. 2008, 99, 1196–1203. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Verduzco, L.F.; Rodríguez-Rodríguez, J.E.; Jaramillo-Jacob, A.D. Predicting cetane number, kinematic viscosity, density and higher heating value of biodiesel from its fatty acid methyl ester composition. Fuel 2012, 91, 102–111. [Google Scholar] [CrossRef]
- Tong, D.M.; Hu, C.W.; Jiang, K.H.; Li, Y.S. Cetane number prediction of biodiesel from the composition of the fatty acid methyl esters. J. Am. Oil Chem. Soc. 2011, 88, 415–423. [Google Scholar] [CrossRef]
- Nascimento, I.A.; Marques, S.S.I.; Cabanelas, I.T.D.; Pereira, S.A.; Druzian, J.I.; de Souza, C.O.; Vich, D.V.; de Carvalho, G.C.; Nascimento, M.A. Screening microalgae strains for biodiesel production: Lipid productivity and estimation of fuel quality based on fatty acids profiles as selective criteria. Bioenergy Res. 2013, 6, 1–13. [Google Scholar] [CrossRef]
- Wei, L.; Huang, X.; Huang, Z.; Zhou, Z. Orthogonal test design for optimization of lipid accumulation and lipid property in Nannochloropsis oculata for biodiesel production. Bioresour. Technol. 2013, 147, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Andruleviciute, V.; Makareviciene, V.; Skorupskaite, V.; Gumbyte, M. Biomass and oil content of Chlorella sp., Haematococcus sp., Nannochloris sp and Scenedesmus sp under mixotrophic growth conditions in the presence of technical glycerol. J. Appl. Phycol. 2014, 26, 83–90. [Google Scholar] [CrossRef]
- Huang, X.X.; Wei, L.K.; Huang, Z.Z.; Yan, J.Q. Effect of high ferric ion concentrations on total lipids and lipid characteristics of Tetraselmis subcordiformis, Nannochloropsis oculata and Pavlova viridis. J. Appl. Phycol. 2014, 26, 105–114. [Google Scholar] [CrossRef]
- Holman, R.T.; Edmondson, P.R. Near-infrared spectra of fatty acids and some related substances. Anal. Chem. 1956, 28, 1533–1538. [Google Scholar] [CrossRef]
- Perez-Vich, B.; Velasco, L.; Fernandez-Martinez, J.M. Determination of seed oil content and fatty acid composition in sunflower through the analysis of intact seeds, husked seeds, meal and oil by near-infrared reflectance spectroscopy. J. Am. Chem. Soc. 1998, 75, 547–555. [Google Scholar]
- Coppa, M.; Ferlay, A.; Leroux, C.; Jestin, M.; Chilliard, Y.; Martin, B.; Andueza, D. Prediction of milk fatty acid composition by near infrared reflectance spectroscopy. Int. Dairy J. 2010, 20, 182–189. [Google Scholar] [CrossRef]
- Fernández-Cuesta, Á.; Fernández-Martinez, J.M.; Company, R.S.I.; Velasco, L. Near-infrared spectroscopy for analysis of oil content and fatty acid profile in almond flour. Eur. J. Lipid Sci. Technol. 2013, 115, 211–216. [Google Scholar] [CrossRef]
- Guy, F.; Prache, S.; Thomas, A.; Bauchart, D.; Andueza, D. Prediction of lamb meat fatty acid composition using near-infrared reflectance spectroscopy (NIRS). Food Chem. 2011, 127, 1280–1286. [Google Scholar] [CrossRef] [PubMed]
- Riovanto, R.; de Marchi, M.; Cassandro, M.; Penasa, M. Use of near infrared transmittance spectroscopy to predict fatty acid composition of chicken meat. Food Chem. 2012, 134, 2459–2464. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Rojas, E.; Garrido-Varo, A.; de Pedro-Sanz, E.; Guerrero-Ginel, J.E.; Pérez-Marín, D. Prediction of fatty acids content in pig adipose tissue by near infrared spectroscopy: At-line versus in-situ analysis. Meat Sci. 2013, 95, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.R.; Frampton, D.M.F.; Dunstan, G.A.; Blackburn, S.I. Assessing near-infrared reflectance spectroscopy for the rapid detection of lipid and biomass in microalgae cultures. J. Appl. Phycol. 2014, 26, 191–198. [Google Scholar] [CrossRef]
- Challagulla, V.; Walsh, K.B.; Subedi, P. Biomass and total lipid content assessment of microalgal cultures using near and short wave infrared spectroscopy. Bioenergy Res. 2014, 7, 306–318. [Google Scholar] [CrossRef]
- Laurens, L.M.L.; Wolfrum, E.J. High-throughput quantitative biochemical characterization of algal biomass by NIR spectroscopy; multiple linear regression and multivariate linear regression analysis. J. Agric. Food Chem. 2013, 61, 12307–12314. [Google Scholar] [CrossRef] [PubMed]
- Mayers, J.J.; Flynn, K.J.; Shields, R.J. Rapid determination of bulk microalgal biochemical composition by Fourier-Transform Infrared spectroscopy. Bioresour. Technol. 2013, 148, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Mulbry, W.; Reeves, J.; Liu, Y.; Ruan, Z.H.; Liao, W. Near- and mid-infrared spectroscopic determination of algal composition. J. Appl. Phycol. 2012, 24, 1261–1267. [Google Scholar] [CrossRef]
- Wagner, H.; Liu, Z.X.; Langner, U.; Stehfest, K.; Wilhelm, C. The use of FTIR spectroscopy to assess quantitative changes in the biochemical composition of microalgae. J. Biophotonics 2010, 3, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Gardner, R.; Peters, P.; Peyton, B.; Cooksey, K.E. Medium pH and nitrate concentration effects on accumulation of triacylglycerol in two members of the chlorophyta. J. Appl. Phycol. 2011, 23, 1005–1016. [Google Scholar] [CrossRef]
- Praveenkumar, R.; Shameera, K.; Mahalakshmi, G.; Akbarsha, M.A.; Thajuddin, N. Influence of nutrient deprivations on lipid accumulation in a dominant indigenous microalga Chlorella sp., BUM11008: Evaluation for biodiesel production. Biomass Bioenergy 2012, 37, 60–66. [Google Scholar] [CrossRef]
- Plans, M.; Simo, J.; Casanas, F.; Sabate, J. Near-infrared spectroscopy analysis of seed coats of common beans (Phaseolus vulgaris L.): A potential tool for breeding and quality evaluation. J. Agric. Food Chem. 2012, 60, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Prieto, N.; Dugan, M.E.R.; López-Campos, Ó; Aalhus, J.L.; Uttaro, B. At line prediction of PUFA and biohydrogenation intermediates in perirenal and subcutaneous fat from cattle fed sunflower or flaxseed by near infrared spectroscopy. Meat Sci. 2013, 94, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Workman, J. Handbook of Organic Compounds. Academic Press: Boston, MA, USA, 2001; pp. 174–176. [Google Scholar]
- Kim, Y.; Kays, S.E. Near-infrared (NIR) prediction of trans-fatty acids in ground cereal foods. J. Agric. Food Chem. 2009, 57, 8187–8193. [Google Scholar] [CrossRef] [PubMed]
- Pinzi, S.; Rounce, P.; Herreros, J.M.; Tsolakis, A.; Dorado, M.P. The effect of biodiesel fatty acid composition on combustion and diesel engine exhaust emissions. Fuel 2013, 104, 170–182. [Google Scholar] [CrossRef]
- Laurens, L.M.L.; Wolfrum, E.J. Feasibility of spectroscopic characterization of algal lipids: Chemometric correlation of NIR and FTIR spectra with exogenous lipids in algal biomass. Bioenergy Res. 2011, 4, 22–35. [Google Scholar] [CrossRef]
- Workman, J.J.; Weyer, L. Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscopy, 2nd ed.CRC Press: Boca Taton, FL, USA, 2012; pp. 55–80. [Google Scholar]
- Bao, Y.D.; Kong, W.W.; Liu, F.; Qiu, Z.J.; He, Y. Detection of glutamic acid in oilseed rape leaves using near infrared spectroscopy and the least squares-support vector machine. Int. J. Mol. Sci. 2012, 13, 14106–14114. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Huang, Y.D. Near infrared spectroscopy for on-line monitoring of alkali-free cloth/phenolic resin prepreg during manufacture. Int. J. Mol. Sci. 2007, 8, 541–552. [Google Scholar] [CrossRef]
- Ordog, V.; Stirk, W.A.; Balint, P.; Lovasz, C.; Pulz, O.; van Staden, J. Lipid productivity and fatty acid composition in Chlorella and Scenepdesmus strains grown in nitrogen-stressed conditions. J. Appl. Phycol. 2013, 25, 233–243. [Google Scholar] [CrossRef]
- Davies, A.M.C.; Grant, A. Near-infrared analysis of food. Int. J. Food Sci. Technol. 1987, 22, 191–207. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, B.; Liu, J.; Chen, T.; Yang, B.; Jiang, Y.; Wei, D.; Chen, F. Rapid Characterization of Fatty Acids in Oleaginous Microalgae by Near-Infrared Spectroscopy. Int. J. Mol. Sci. 2015, 16, 7045-7056. https://doi.org/10.3390/ijms16047045
Liu B, Liu J, Chen T, Yang B, Jiang Y, Wei D, Chen F. Rapid Characterization of Fatty Acids in Oleaginous Microalgae by Near-Infrared Spectroscopy. International Journal of Molecular Sciences. 2015; 16(4):7045-7056. https://doi.org/10.3390/ijms16047045
Chicago/Turabian StyleLiu, Bin, Jin Liu, Tianpeng Chen, Bo Yang, Yue Jiang, Dong Wei, and Feng Chen. 2015. "Rapid Characterization of Fatty Acids in Oleaginous Microalgae by Near-Infrared Spectroscopy" International Journal of Molecular Sciences 16, no. 4: 7045-7056. https://doi.org/10.3390/ijms16047045
APA StyleLiu, B., Liu, J., Chen, T., Yang, B., Jiang, Y., Wei, D., & Chen, F. (2015). Rapid Characterization of Fatty Acids in Oleaginous Microalgae by Near-Infrared Spectroscopy. International Journal of Molecular Sciences, 16(4), 7045-7056. https://doi.org/10.3390/ijms16047045







