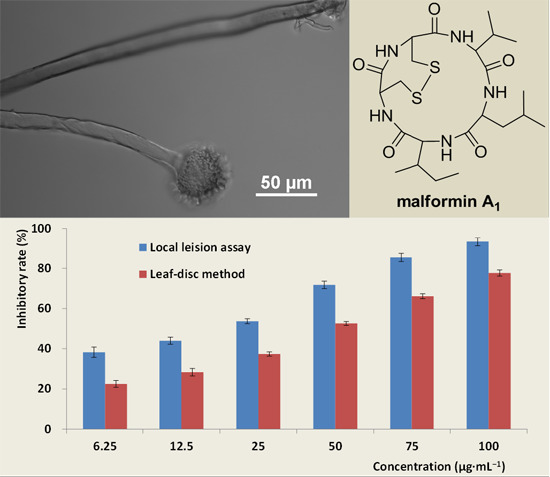
Anti-TMV Activity of Malformin A1, a Cyclic Penta-Peptide Produced by an Endophytic Fungus Aspergillus tubingensis FJBJ11
Abstract
:1. Introduction
2. Results
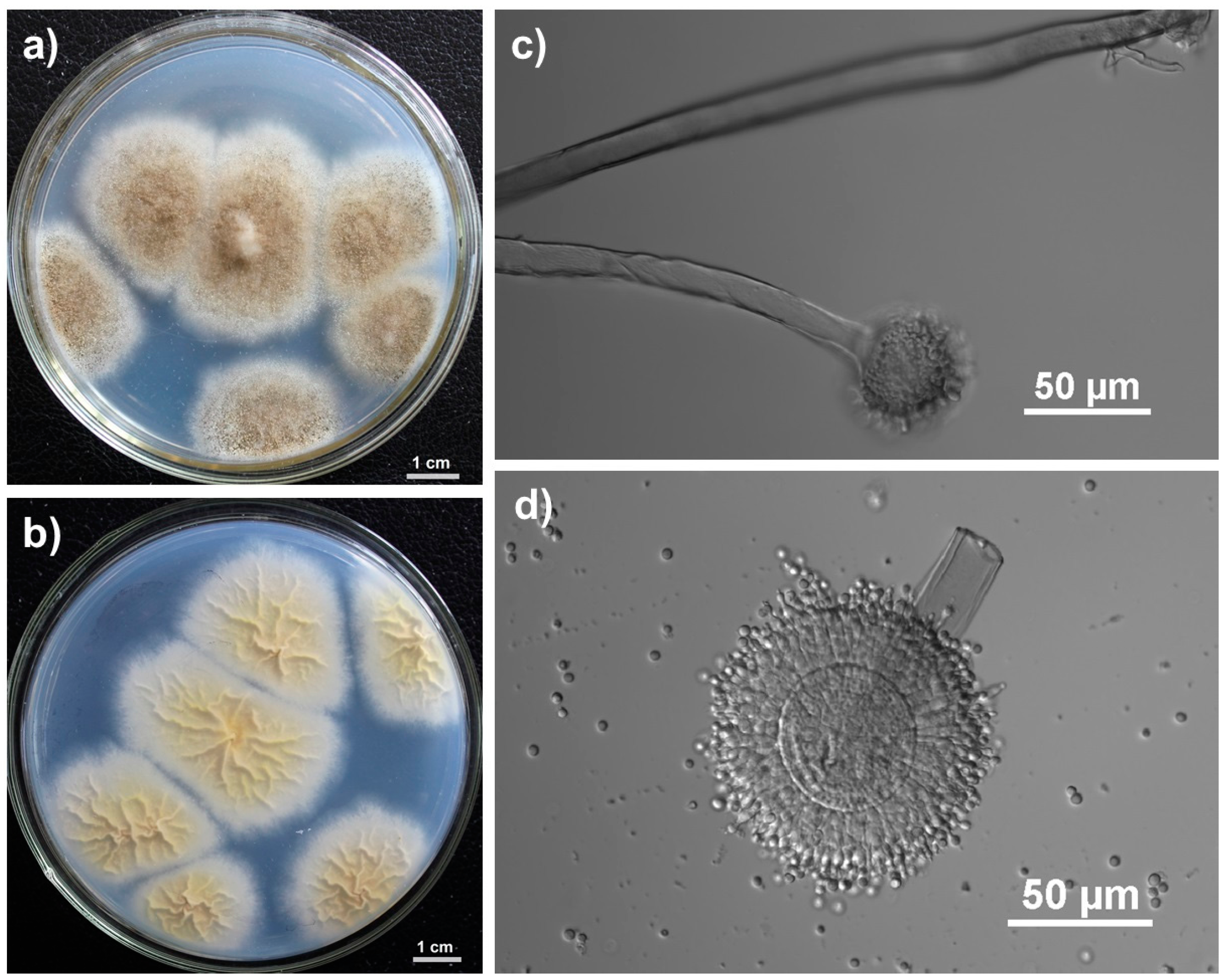
2.1. Identification of the Endophytic Fungus

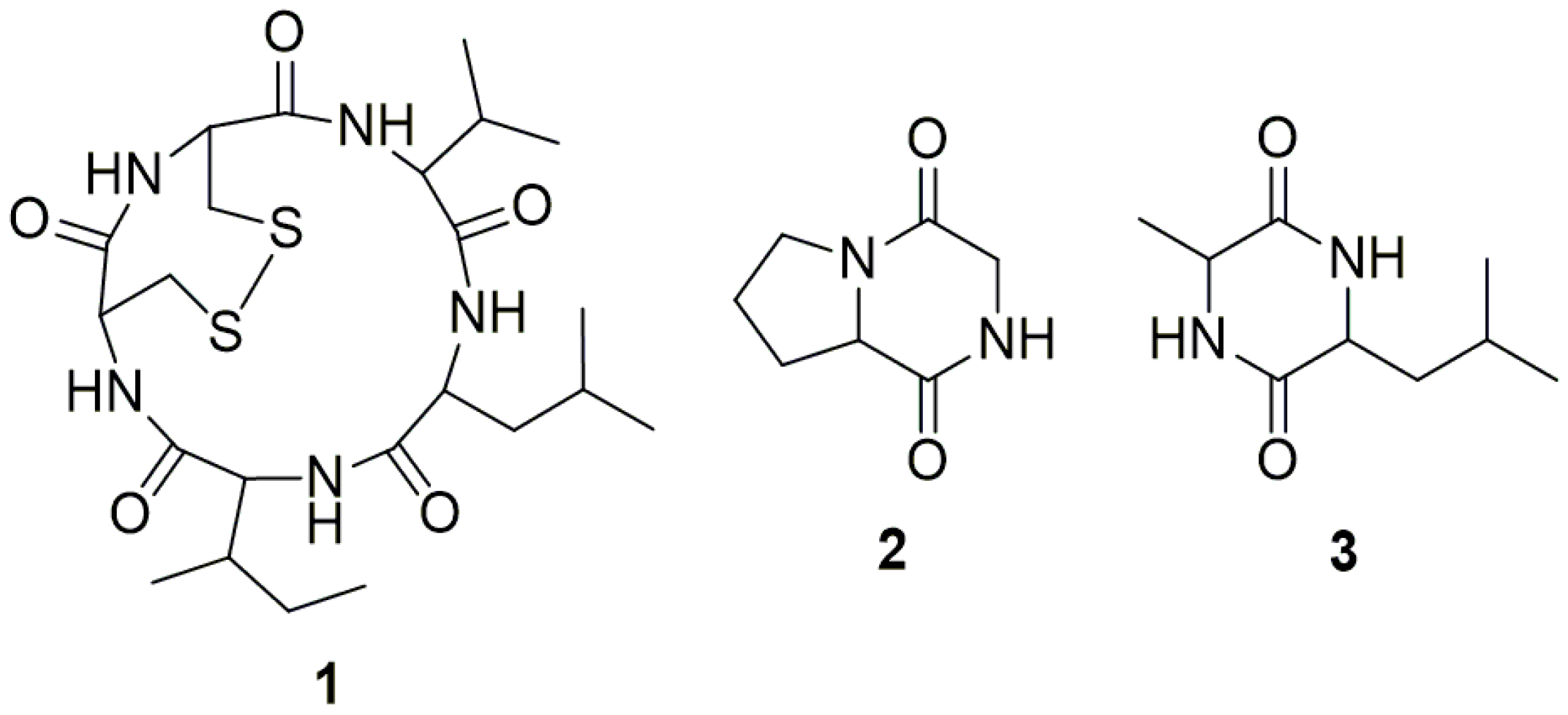
2.3. Structure Determination
| Amino Acid | Position | δH (mult., J in Hz) | δC (mult.) |
|---|---|---|---|
| Leu1 | C=O | – | 172.8 (s) |
| NH | 7.38 (d, 9.2) | – | |
| α | 4.47 (dt, 9.2, 2.8) | 50.4 (d) | |
| β | 1.37 (m) | 40.9 (d) | |
| γ | 1.56 (m) | 24.5 (d) | |
| δ | 0.89 (d, 6.6) | 22.7 (q) | |
| 0.85 (d, 6.6) | 21.8 (q) | ||
| Ile2 | C=O | – | 173.0 (s) |
| NH | 8.59 (d, 6.6) | – | |
| α | 3.87 (dd, 10.2, 6.6) | 58.0 (d) | |
| β | 1.69 (m) | 34.0 (d) | |
| γ | 1.50 (m); 1.13 (m) | 24.8 (t) | |
| 0.77 (d, 6.9) | 14.9 (q) | ||
| δ | 0.81 (t, 10.9) | 10.0 (q) | |
| Cys3 | C=O | – | 174.0 (s) |
| NH | 8.84 (d, 3.7) | – | |
| α | 3.98 (dd, 6.3, 3.3) | 52.4 (d) | |
| β | 3.51 (dd, 11.9, 3.2); 3.14 (m) | 46.2 (d) | |
| Cys4 | C=O | – | 169.8 (s) |
| NH | 7.11 (d, 11.0) | – | |
| α | 4.71 (dt, 11.0, 4.4) | 52.9 (d) | |
| β | 3.24 (m); 3.19 (m) | 45.2 (t) | |
| Val5 | C=O | – | 170.6 (s) |
| NH | 7.94 (d, 8.5) | – | |
| α | 3.92 (m) | 58.8 (d) | |
| β | 2.05 (m) | 26.9 (d) | |
| γ | 0.82 (d, 6.8) | 19.7 (q) | |
| 0.82 (d, 6.8) | 18.7 (q) |
3. Discussion
4. Experimental Section
4.1. General
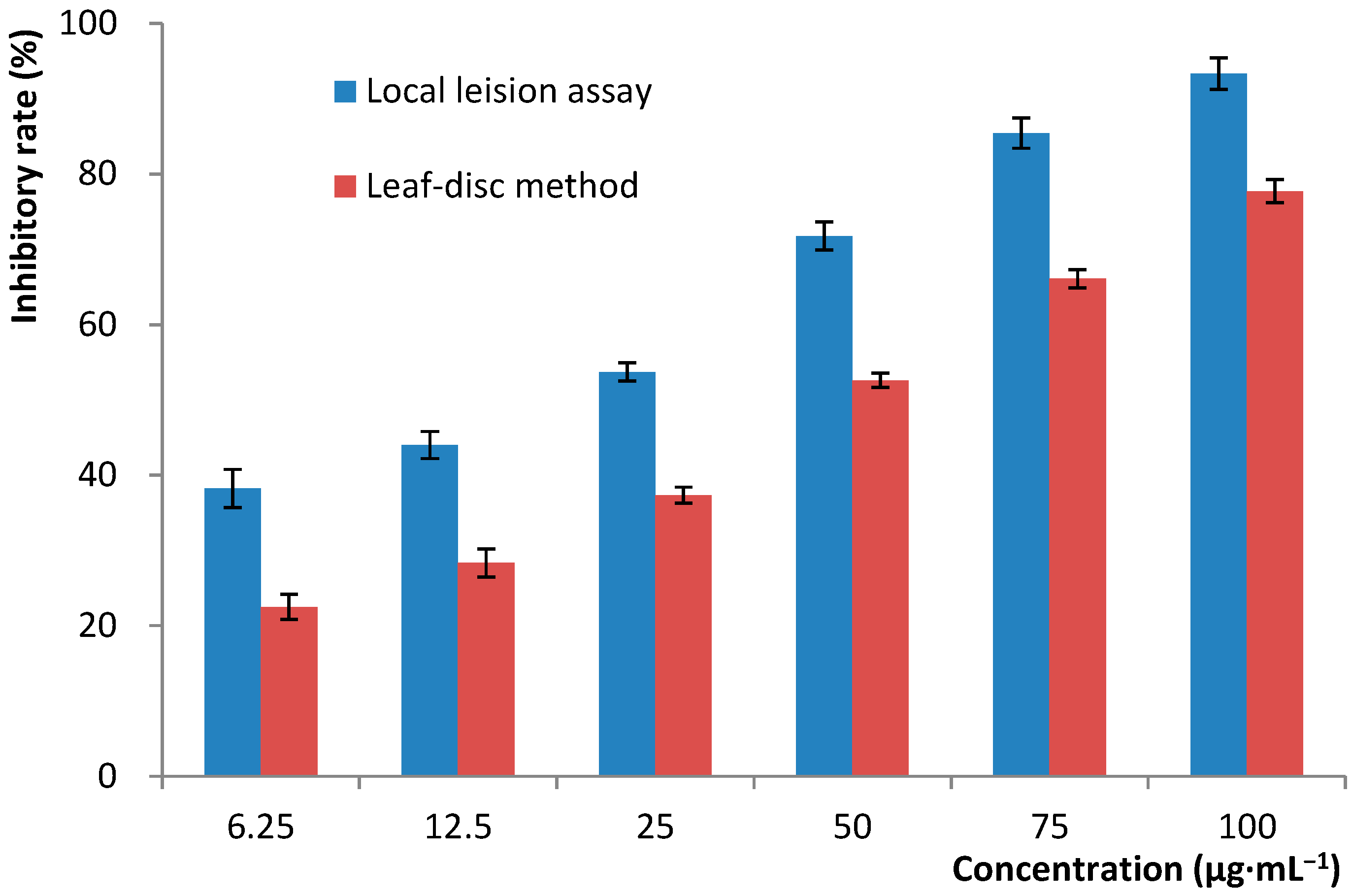
4.2. Bioassay of TMV-Inhibitory Activity
4.2.1. Local-Lesion Assay
4.2.2. Leaf-Disc Method
4.2.3. Indirect ELISA Procedure
4.2.4. Statistical Analysis
4.3. Identification of Fungi
4.4. Endophytic Fungus and Fermentation
4.5. Extraction and Isolation
4.5.1. Malformin A1 (1)
4.5.2. Cyclo (Gly-l-Pro) (2)
4.5.3. Cyclo (Ala-Leu) (3)
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Craeger, A.N.; Scholthof, K.B.; Citovsky, V.; Scholthof, H.B. Tobacco mosaic virus: Pioneering research for a century. Plant Cell 1999, 11, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Ritzenthaler, C. Resistance to plant viruses: Old issue, new answer. Curr. Opin. Biotechnol. 2005, 16, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.H.; Chen, J.; Di, Y.T.; Fang, X.; Dong, J.H.; Peng, S.; Wang, Y.H.; He, H.P.; Zhang, Z.K.; Hao, X.J. Anti-Tobacco mosaic virus (TMV) quassinoids from Brucea javanica (L.) Merr. J. Agric. Food Chem. 2010, 58, 1572–1577. [Google Scholar] [CrossRef]
- Li, Y.M.; Wang, L.H.; Li, S.L.; Chen, X.Y.; Shen, Y.M.; Zhang, Z.K.; He, H.P.; Xu, W.B.; Shu, Y.L.; Liang, G.D.; et al. Seco-pregnane steroids target the subgenomic RNA of alphavirus-like RNA viruses. Proc. Natl. Acad. Sci. USA 2007, 104, 8083–8088. [Google Scholar]
- Li, Y.M.; Zhang, Z.K.; Jia, Y.T.; Shen, Y.M.; He, H.P.; Fang, R.X.; Chen, X.Y.; Hao, X.J. 3-Acetony-3-hydroxyoxindole: A new inducer of systemic acquired resistance in plants. Plant Biotechnol. J. 2008, 6, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.H.; Zhang, Z.K.; Yang, F.M.; Sun, Q.Y.; He, H.P.; Di, Y.T.; Mu, S.Z.; Lu, Y.; Chang, Y.; Zheng, Q.T.; et al. Benzylphenethylamine alkaloids from Hosta plantaginea with inhibitory activity against tobacco mosaic virus and acetylcholinesterase. J. Nat. Prod. 2007, 70, 1458–1461. [Google Scholar]
- Tan, R.X.; Zou, W.X. Endophytes: A rich source of functional metabolites. Nat. Prod. Rep. 2001, 18, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.; Daisy, B.; Castillo, U.; Harper, J. Natural products from endophytic microorganisms. J. Nat. Prod. 2004, 67, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Gunatilaka, A.A.L. Natural products from plant-associated microorganisms: Distribution, structural diversity, bioactivity and implications of their occurrence. J. Nat. Prod. 2006, 69, 509–526. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, F.; Kim, K.W.; Uzawa, J.; Yoshida, S.; Takahashi, N.; Curtis, R.W. Structure of malformin A2, reinvestigation of phytotoxic metabolites produced by Aspergillus niger. Tetrahedron Lett. 1990, 31, 4337–4340. [Google Scholar] [CrossRef]
- Kojima, Y.; Sunazuka, T.; Nagai, K.; Hirose, T.; Namatame, M.; Ishiyama, A.; Otoguro, K.; Ōmura, S. Solid-phase synthesis and biological activity of malformin C and its derivatives. J. Antibiot. 2009, 62, 681–686. [Google Scholar] [CrossRef] [PubMed]
- John, W.W.; Curtis, R.W. Stimulation of plant growth by malformin A. Experientia 1974, 30, 1392–1393. [Google Scholar] [CrossRef] [PubMed]
- Curtis, R.W.; John, W.W. Effect of malformin on phytochromeand ethrel-mediated responses. Plant Cell Physiol. 1975, 16, 719–728. [Google Scholar]
- Suda, S.; Curtis, R.W. Antibiotic properties of malformin. Appl. Microbiol. 1966, 14, 475–476. [Google Scholar] [PubMed]
- Franck, B. Mycotoxins from mold fungi-weapons of uninvited fellow-boarders of man and animal: Structures, biological activity, biosynthesis, and precautions. Angew. Chem. Int. Ed. 1984, 23, 493–505. [Google Scholar] [CrossRef]
- Dawes, J. Malformin A prevents IL-1 induced endothelial changes by inhibition of protein synthesis. Thromb. Haemost. 1994, 72, 482–483. [Google Scholar] [PubMed]
- Koizumi, Y.; Fukudome, H.; Hasumi, K. Fibrinolytic activation promoted by the cyclopentapeptide malformin: Involvement of cytoskeletal reorganization. Biol. Pharm. Bull. 2011, 34, 1426–1431. [Google Scholar] [CrossRef] [PubMed]
- Zhan, J.X.; Gunaherath, G.M.K.B.; Wijeratne, E.M.K.; Gunatilaka, A.A.L. Asperpyrone D and other metabolites of the plant-associated fungal strain Aspergillus tubingensis. Phytochemistry 2007, 68, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Li, X.B.; Xie, F.; Liu, S.S.; Li, Y.; Zhou, J.C.; Liu, Y.Q.; Yuan, H.Q.; Lou, H.X. Naphtho-γ-pyrones from endophyte Aspergillus niger occurring in the liverwort Heteroscyphus tener (Steph.) Schiffn. Chem. Biodivers. 2013, 10, 1193–1201. [Google Scholar] [CrossRef]
- Kim, S.Y.; Cho, A.; Kim, K.W.; Oh, S.E. Dose-dependent effects of malformin A1 on IAA-induced ethylene production in mung bean (Vigna radiate L.) hypocotyl segments. J. Plant Biol. 2004, 47, 254–261. [Google Scholar] [CrossRef]
- Van Loon, L.C.; Geraats, B.P.J.; Linthorst, H.J.M. Ethylene as a modulator of disease resistance in plants. Trends Plant Sci. 2006, 11, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Faoro, F.; Gozzo, F. Is modulating virus virulence by induced systemic resistance realistic? Plant Sci. 2015, 234, 1–13. [Google Scholar] [CrossRef]
- Padmanabhan, M.S.; Goregaoker, S.P.; Golem, S.; Shiferaw, H.; Culver, J.N. Interaction of the Tobacco mosaic virus replicase protein with the Aux/IAA protein PAP1/IAA26 is associated with disease development. J. Virol. 2005, 79, 2549–2558. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, M.S.; Shiferaw, H.; Culver, J.N. The Tobacco mosaic virus replicase protein disrupts the localization and function of interaction Aux/IAA proteins. Mol. Plant-Microbe Interact. 2006, 19, 846–873. [Google Scholar] [CrossRef]
- Padmanabhan, M.S.; Kramer, S.R.; Wang, X.; Culver, J.N. Tobacco mosaic virus replicase-Auxin/Indole acetic acid protein interactions: Reprogramming the auxin response pathway to enhance virus infection. J. Virol. 2008, 82, 2477–2485. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.J.; Ouyang, M.A.; Tan, Q.W.; Zhang, Z.K.; Wu, Z.J.; Lin, Q.Y. Constituents from the seeds of Brucea javanica with inhibitory activity of Tobacco mosaic virus. J. Asian Nat. Prod. Res. 2009, 11, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Ouyang, M.A.; Tan, Q.W. New asperxanthone and asperbiphenyl from the marine fungus Aspergillus sp. Pest Manag. Sci. 2009, 65, 60–65. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press Inc: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar]
- Nylander, J.A.A. MrModeltest v2.3. Program Distributed by the Author; Evolutionary Biology Centre, Uppsala University: Uppsala, Sweden, 2008. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, Q.-W.; Gao, F.-L.; Wang, F.-R.; Chen, Q.-J. Anti-TMV Activity of Malformin A1, a Cyclic Penta-Peptide Produced by an Endophytic Fungus Aspergillus tubingensis FJBJ11. Int. J. Mol. Sci. 2015, 16, 5750-5761. https://doi.org/10.3390/ijms16035750
Tan Q-W, Gao F-L, Wang F-R, Chen Q-J. Anti-TMV Activity of Malformin A1, a Cyclic Penta-Peptide Produced by an Endophytic Fungus Aspergillus tubingensis FJBJ11. International Journal of Molecular Sciences. 2015; 16(3):5750-5761. https://doi.org/10.3390/ijms16035750
Chicago/Turabian StyleTan, Qing-Wei, Fang-Luan Gao, Fu-Rong Wang, and Qi-Jian Chen. 2015. "Anti-TMV Activity of Malformin A1, a Cyclic Penta-Peptide Produced by an Endophytic Fungus Aspergillus tubingensis FJBJ11" International Journal of Molecular Sciences 16, no. 3: 5750-5761. https://doi.org/10.3390/ijms16035750
APA StyleTan, Q.-W., Gao, F.-L., Wang, F.-R., & Chen, Q.-J. (2015). Anti-TMV Activity of Malformin A1, a Cyclic Penta-Peptide Produced by an Endophytic Fungus Aspergillus tubingensis FJBJ11. International Journal of Molecular Sciences, 16(3), 5750-5761. https://doi.org/10.3390/ijms16035750