The Safety Evaluation of Salvianolic Acid B and Ginsenoside Rg1 Combination on Mice
Abstract
:1. Introduction
2. Results
2.1. Purity of SalB and Rg1

2.2. LD50 from the Up and Down Procedure
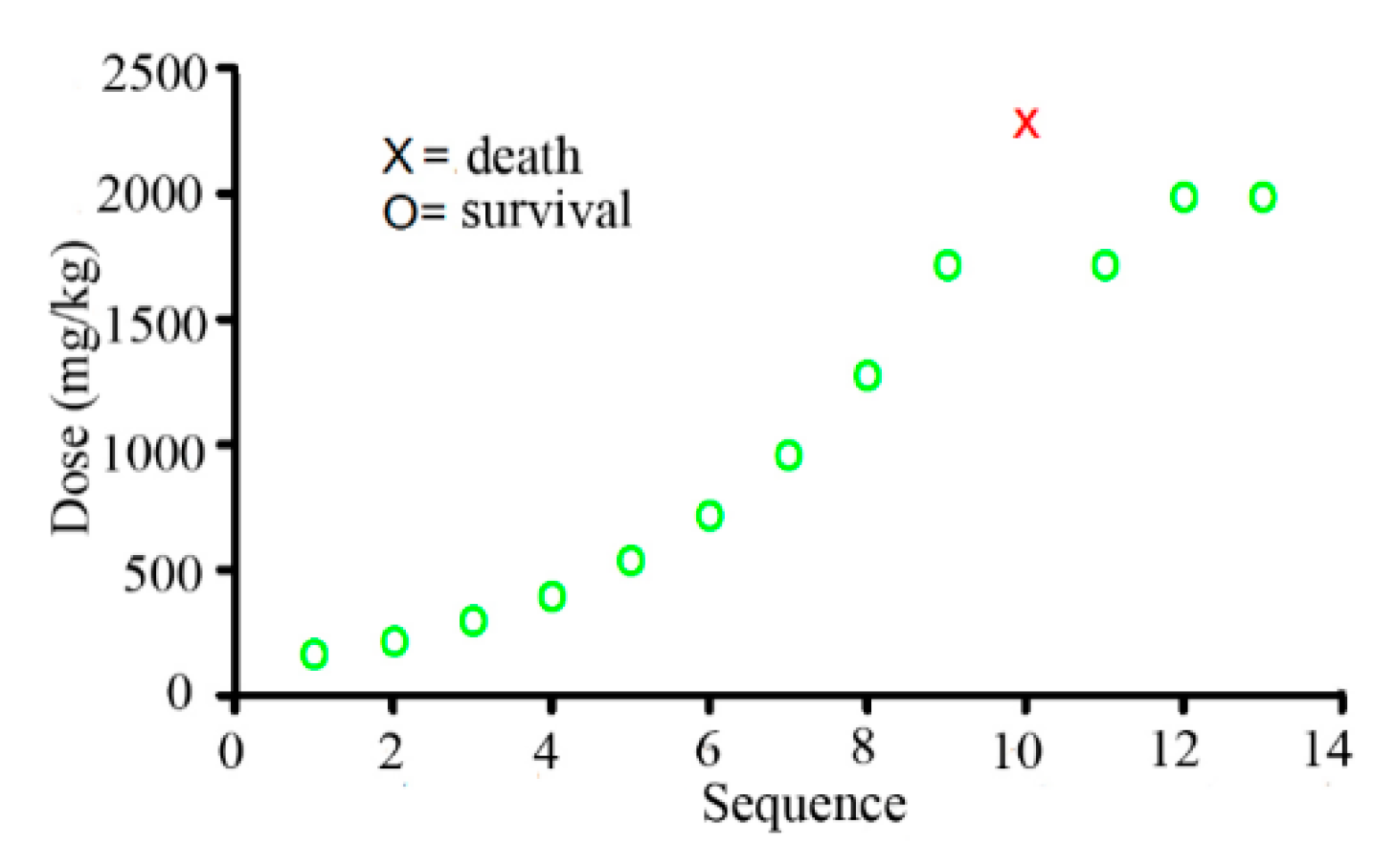
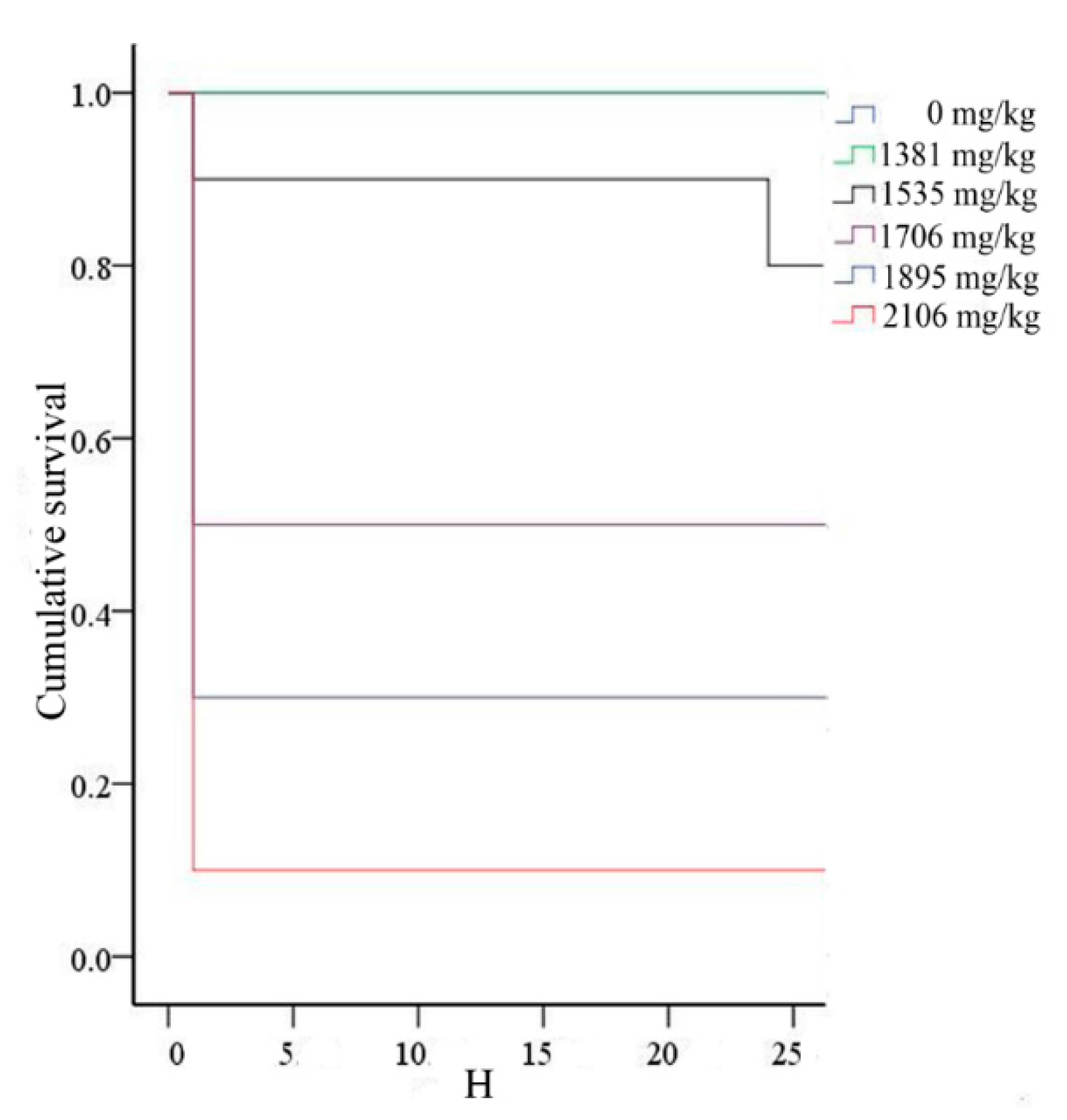
2.3. LD50 from the Bliss Method with a Single Dose
| Day 0 | Day 7 | Day 14 | |||||
|---|---|---|---|---|---|---|---|
| Dose (mg/kg) | Body Weight (g) | Animal Number | Body Weight (g) | Animal Number | Body Weight (g) | Animal Number | |
| Males | 0 | 38.1 ± 2.1 | 5 | 42.2 ± 3.3 | 5 | 40.6 ± 3.2 | 5 |
| 1381 | 37.5 ± 1.9 | 5 | 40.6 ± 1.8 | 5 | 40.4 ± 1.9 | 5 | |
| 1535 | 38.5 ± 2.5 | 5 | 42.2 ± 3.0 | 5 | 42.0 ± 2.9 | 5 | |
| 1706 | 38.0 ± 2.5 | 3 | 40.0 ± 3.3 | 3 | 39.5 ± 2.9 | 3 | |
| 1895 | 37.6 ± 1.2 | 1 | 38.7 | 1 | 35.7 | 1 | |
| 2106 | 38.3 ± 2.1 | 0 | – | – | – | – | |
| Females | 0 | 32.1 ± 1.2 | 5 | 33.8 ± 1.7 | 5 | 35.6 ± 1.7 | 5 |
| 1381 | 30.5 ± 1.3 | 5 | 31.5 ± 1.9 | 5 | 33.8 ± 2.1 | 5 | |
| 1535 | 31.7 ± 2.0 | 4 | 33.7 ± 1.9 | 3 | 33.4 ± 1.7 | 3 | |
| 1706 | 30.5 ± 1.6 | 2 | 35.2 ± 0.1 | 2 | 32.4 ± 0.2 | 2 | |
| 1895 | 29.9 ± 2.4 | 2 | 31.5 ± 2.5 | 2 | 32.1 ± 2.1 | 2 | |
| 2106 | 30.3 ± 1.6 | 1 | 30.9 | 1 | 31.3 | 1 | |
2.4. Effects of SalB-Rg1 on the Organ Index with a Single Dose in Acute Toxicity
| Organ Index (g/kg) | Dose Group (mg/kg) | |||||
|---|---|---|---|---|---|---|
| 0 | 1381 | 1535 | 1706 | 1895 | 2106 | |
| Males | ||||||
| Brain | 8.4 ± 1.5 | 9.2 ± 0.8 | 8.9 ± 0.8 | 8.5 ± 0.3 | 10.6 | – |
| Heart | 5.1 ± 0.2 | 5.4 ± 0.6 | 5.6 ± 0.7 | 4.7 ± 0.7 | 4.5 | – |
| Kidney | 15.4 ± 1.3 | 15.2 ± 1.3 | 15.2 ± 0.8 | 16.7 ± 2.2 | 17.9 | – |
| Liver | 39.9 ± 3.7 | 45.1 ± 5.4 | 43.8 ± 1.9 | 40.1 ± 0.8 | 44.7 | – |
| Lung | 7.1 ± 0.9 | 7.1 ± 1.0 | 6.8 ± 1.5 | 6.6 ± 0.7 | 7.27 | – |
| Spleen | 2.4 ± 0.4 | 3.7 ± 1.9 | 2.5 ± 0.6 | 3.4 ± 1.5 | 4.5 | – |
| Females | ||||||
| Brain | 9.9 ± 1.0 | 10.8 ± 1.4 | 11.7 ± 2.6 | 13.3 ± 1.1 | 11.1 ± 2.0 | 11 |
| Heart | 4.8 ± 0.4 | 5.7 ± 0.4 | 5.8 ± 0.5 | 5.2 ± 0.4 | 3.5 ± 0.4 | 6.6 |
| Kidney | 12.6 ± 1.5 | 13.0 ± 1.4 | 13.5 ± 2.0 | 13.3 ± 0.4 | 13.2 ± 0.2 | 13.4 |
| Liver | 43.7 ± 4.7 | 46.0 ± 3.9 | 45.0 ± 3.0 | 38.5 ± 0.17 | 50.8 ± 0.8 | 43.5 |
| Lung | 7.22 ± 1.3 | 6.9 ± 0.7 | 6.9 ± 1.2 | 6.9 ± 0.2 | 7.02 ± 0.5 | 6.4 |
| Spleen | 3.5 ± 0.3 | 4.1 ± 0.5 | 4.8 ± 0.6 ** | 2.6 ± 0.38 | 5.8 ± 0.4 ** | 3.7 |
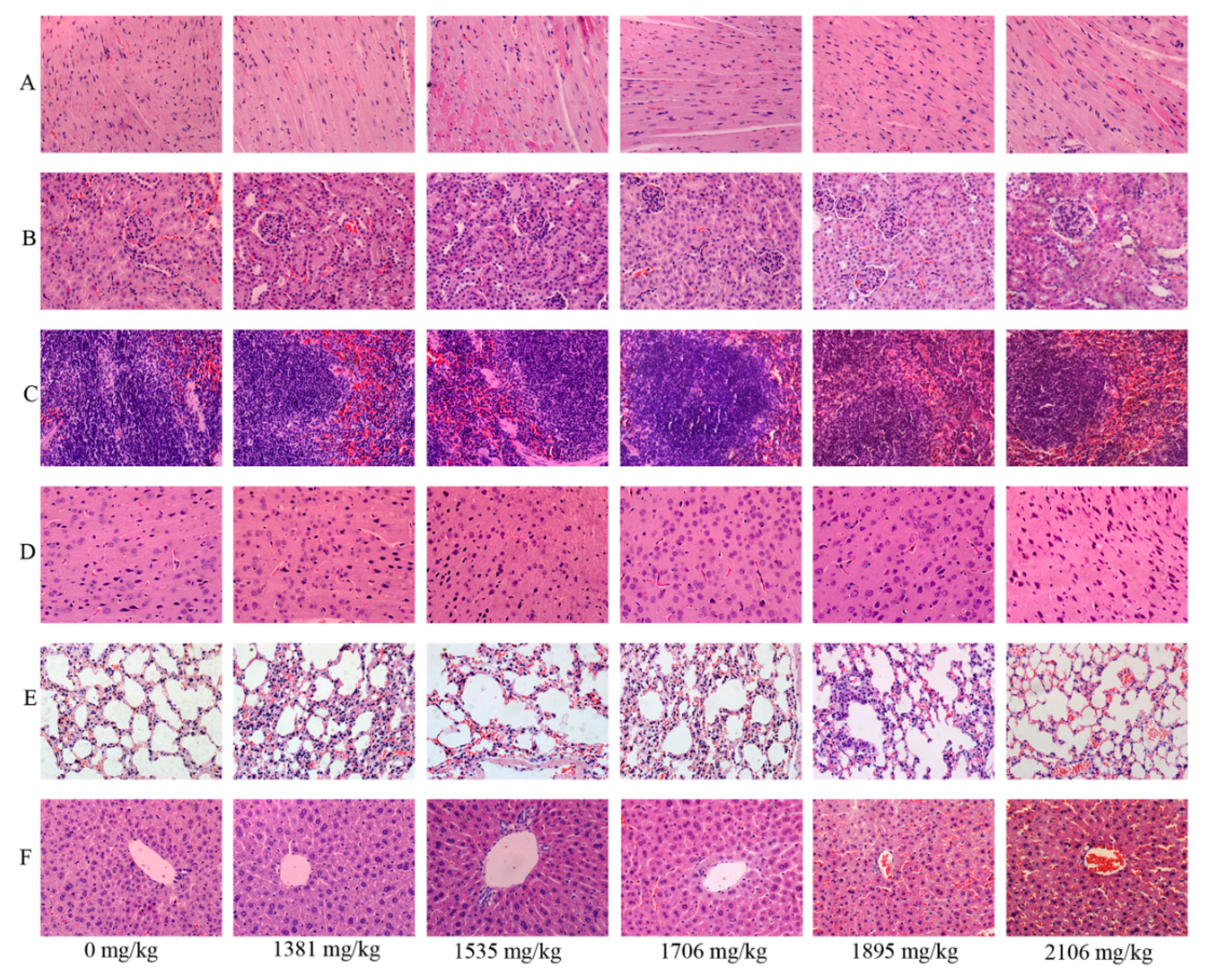
2.5. Effects of SalB-Rg1 on the Organ Structure with a Single Dose in Acute Toxicity
2.6. Effects of SalB-Rg1 on Biochemical Parameters with a Single Dose in Acute Toxicity
| Dose Group (mg/kg) | ||||||
|---|---|---|---|---|---|---|
| 0 | 1381 | 1535 | 1706 | 1895 | 2106 | |
| Males | ||||||
| Cr (μmol/L) | 201.2 ± 88.2 | 159.5 ± 38.8 | 193.6 ± 12.4 | 162.3 ± 87.2 | – | – |
| TP (mg/mL) | 48.7 ± 2.4 | 41.2 ± 7.0 | 38.5 ± 7.5 | 45.2 ± 10.8 | – | – |
| Alb (mg/mL) | 19.2 ± 2.2 | 19.7 ± 4.6 | 17.3 ± 3.7 | 18.6 ± 3.3 | – | – |
| Females | ||||||
| Cr (μmol/L) | 125.3 ± 81.3 | 153.8 ± 58.7 | 100.6 ± 71.7 | 323.3 ± 194.6 | 162.3 ± 33.6 | 176.5 |
| TP (mg/mL) | 44.0 ± 3.7 | 41.0 ± 2.2 | 47.5 ± 6.5 | 47.9 ± 3.0 | 41.7 ± 6.2 | 23.2 |
| Alb (mg/mL) | 20.2 ± 2.9 | 18.0 ± 3.8 | 16.8 ± 1.0 | 19.7 ± 0.2 | 17.9 ± 3.2 | 47.8 |
2.7. Effects of SalB-Rg1 on Body Weight and Survival with a Seven-Day Repeated Dose for Toxicity Evaluation
| Day 0 | Day 7 | Day 14 | |||||
|---|---|---|---|---|---|---|---|
| Dose (mg/kg) | Body Weight (g) | Animal Number | Body Weight (g) | Animal Number | Body Weight (g) | Animal Number | |
| Males | 0 | 23.5 ± 1.1 | 5 | 27.4 ± 1.5 | 5 | 30.8 ± 2.2 | 5 |
| 15 | 23.5 ± 1.1 | 5 | 27.4 ± 1.5 | 5 | 30.8 ± 2.2 | 5 | |
| 30 | 22.8 ± 0.8 | 5 | 26.4 ± 1.1 | 5 | 30.0 ± 1.5 | 5 | |
| 60 | 23.1 ± 0.9 | 5 | 27.0 ± 1.3 | 5 | 30.0 ± 1.3 | 5 | |
| Females | 0 | 22.2 ± 1.2 | 5 | 24.7 ± 1.3 | 5 | 26.6 ± 1.1 | 5 |
| 15 | 22.9 ± 0.7 | 5 | 26.9 ± 1.0 | 5 | 26.8 ± 1.2 | 5 | |
| 30 | 23.0 ± 0.7 | 5 | 26.6 ± 2.2 | 5 | 28.5 ± 1.2 | 5 | |
| 60 | 22.2 ± 1.5 | 5 | 25.7 ± 1.9 | 5 | 26.7 ± 1.7 | 5 | |
2.8. Effects of SalB-Rg1 on the Organ Index with a Seven-Day Repeated Dose for Toxicity Evaluation
| Organ Index (g/kg) | Dose Group (mg/kg) | |||
|---|---|---|---|---|
| 0 | 15 | 30 | 60 | |
| Males | ||||
| Brain | 11.5 ± 1.2 | 11.6 ± 1.1 | 11.8 ± 1.3 | 11.7 ± 1.1 |
| Heart | 5.7 ± 0.5 | 5.2 ± 0.3 | 5.8 ± 0.5 | 5.5 ± 0.5 |
| Kidney | 14.0 ± 0.7 | 13.6 ± 0.6 | 13.0 ± 1.3 | 13.6 ± 0.9 |
| Lung | 7.3 ± 0.4 | 6.6 ± 0.4 | 7.0 ± 0.5 | 6.8 ± 0.6 |
| Spleen | 8.3 ± 3.4 | 6.4 ± 0.4 | 6.8 ± 0.7 | 6.1 ± 0.4 |
| Females | ||||
| Brain | 13.4 ± 1.7 | 12.6 ± 1.0 | 12.1 ± 1.3 | 13.6 ± 0.5 |
| Heart | 5.1 ± 0.5 | 4.9 ± 0.2 | 4.9 ± 0.5 | 4.8 ± 0.5 |
| Kidney | 13.3 ± 0.6 | 13.5 ± 1.1 | 13.4 ± 0.9 | 12.4 ± 0.9 |
| Lung | 8.2 ± 1.8 | 7.5 ± 0.7 | 6.8 ± 1.1 | 7.4 ± 1.2 |
| Spleen | 7.3 ± 0.9 | 6.5 ± 1.0 | 5.9 ± 1.2 | 6.3 ± 1.2 |
2.9. Effects of SalB-Rg1 on the Organ Structure with a Seven-Day Repeated Dose for Toxicity Evaluation
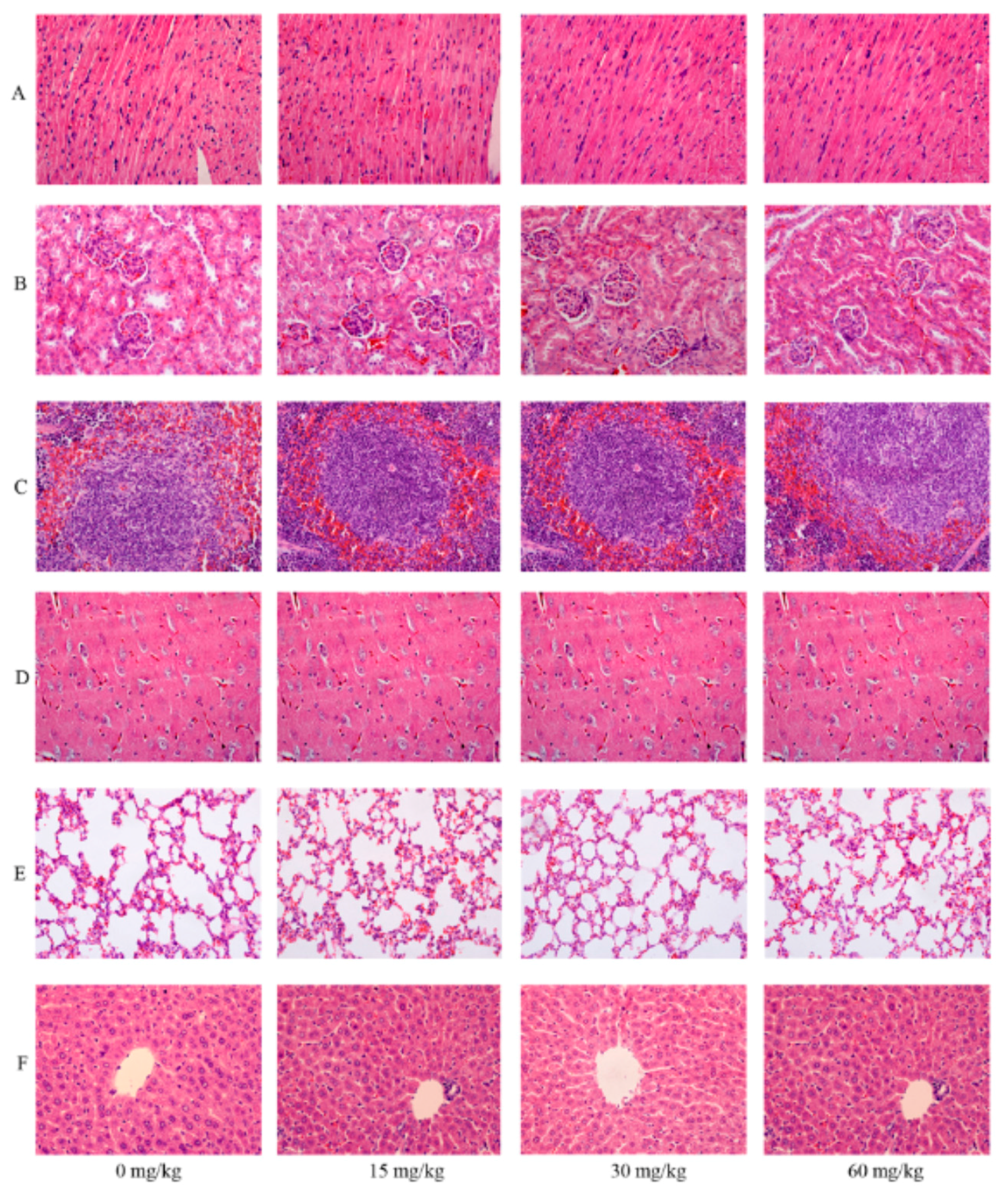
2.10. Effects of SalB-Rg1 on Biochemical Parameters of the Seven-Day Repeated Dose for Toxicity Evaluation
| Dose Group (mg/kg) | ||||
|---|---|---|---|---|
| 0 | 15 | 30 | 60 | |
| Males | ||||
| Cr (μmol/L) | 76.3 ± 49.8 | 38.7 ± 22.0 | 67.1 ± 11.3 | 58.9 ± 12.3 |
| TP (mg/mL) | 52.3 ± 5.4 | 56.1 ± 5.4 | 52.4 ± 4.9 | 52.7 ± 5.6 |
| Alb (mg/mL) | 28.0 ± 3.2 | 29.6 ± 1.7 | 28.5 ± 4.9 | 28.6 ± 3.1 |
| Females | ||||
| Cr (μmol/L) | 81.5 ± 14.7 | 81.5 ± 10.6 | 70.2 ± 17.4 | 44.5 ± 20.6 * |
| TP (mg/mL) | 54.3 ± 3.9 | 56.4 ± 4.9 | 54.1 ± 6.9 | 53.1 ± 4.6 |
| Alb (mg/mL) | 28.7 ± 3.3 | 28.7 ± 1.8 | 29.2 ± 3.5 | 33.3 ± 3.9 |
3. Discussion
4. Experimental Section
4.1. Reagents
4.2. Animals
4.3. Purity Assay for SalB and Rg1
4.4. Up and Down Procedure
4.5. Bliss Method
4.6. Seven-Day Repeat Dose
4.7. Collection of Blood and Organ Samples
4.8. Histopathological Detection
4.9. Measurement of Biochemical Parameters on Serum
4.10. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chun-Sheng, L.; Hsiao-Meng, Y.; Yun-Hsiang, H.; Chun, P.; Chi-Fen, S. Radix salviae miltiorrhizae and Rhizoma ligustici wallichii in coronary heart disease. Chin. Med. J. 1978, 4, 43–46. [Google Scholar] [PubMed]
- Shang, Q.; Xu, H.; Liu, Z.; Chen, K.; Liu, J. Oral Panax notoginseng preparation for coronary heart disease: A systematic review of randomized controlled trials. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Yang, M.; Xu, F.; Zhang, Q.; Zhao, Q.; Yu, H.; Li, D.; Zhang, G.; Lu, A.; Cho, K.; et al. Combined salvianolic acid B and ginsenoside Rg1 exerts cardioprotection against ischemia/reperfusion injury in rats. PLoS ONE 2015, 10, e0135435. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P. Mechanisms of drug toxicity and relevance to pharmaceutical development. Drug Metab. Pharmacokinet. 2011, 26, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.Y.; Wei, J.C.; Lee, M.Y.; Chen, C.M.; Ueng, K.C. A randomized, double-blind, placebo-controlled study to evaluate the efficacy and tolerability of Fufang Danshen (Salvia miltiorrhiza) as add-on antihypertensive therapy in Taiwanese patients with uncontrolled hypertension. Phytother. Res. 2012, 26, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.M.; Zhang, C.G.; Lin, H.; Lu, W.L.; Cheng, H.P.; Wang, J. Determination of effective components in different positions of Panax notoginseng by HPLC. Zhong Yao Cai 2009, 32, 1810–1813. [Google Scholar] [PubMed]
- Li, L.; Sheng, Y.; Zhang, J.; Wang, C.; Guo, D. HPLC determination of four active saponins from Panax notoginseng in rat serum and its application to pharmacokinetic studies. Biomed. Chromatogr. 2004, 18, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Huang, Z.G.; Cao, H.; Wang, Y.T.; Hui, P.; Hoo, C.; Li, S.P. Screening of anti-platelet aggregation agents from Panax notoginseng using human platelet extraction and HPLC-DAD-ESI-MS/MS. J. Sep. Sci. 2008, 31, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.J.; Li, S.L.; Li, P. Simultaneous determination of seven active components of Fufang Danshen tablet by high performance liquid chromatography. Biomed. Chromatogr. 2007, 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.D.; Zhang, T.T.; Teng, F.K.; Li, D.F.; Xu, F.; Cho, K.; Xu, J.; Yin, J.; Zhang, L.; Liu, Q.; et al. Ginsenoside Rg1 and Rb1, in combination with salvianolic acid B, play different roles in myocardial infarction in rats. J. Chin. Med. Assoc. 2015, 78, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.L.; Wang, X.J.; Sun, Y.N.; Li, C.R.; Xing, Y.L.; Zhao, H.B.; Duan, M.; Zhou, Z.; Wang, S.Q. Salvianolic acid B, an antioxidant from Salvia miltiorrhiza, prevents 6-hydroxydopamine induced apoptosis in SH-SY5Y cells. Int. J. Biochem. Cell Biol. 2008, 40, 409–422. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.X.; Hu, L.M.; Gao, X.M.; Guo, H.; Fan, G.W. Anti-inflammatory activity of salvianolic acid B in microglia contributes to its neuroprotective effect. Neurochem. Res. 2010, 35, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Mu, X.; Zeng, J.; Xu, C.; Liu, J.; Zhang, M.; Li, C.; Chen, J.; Li, T.; Wang, Y. Ginsenoside Rg1 prevents cognitive impairment and hippocampus senescence in a rat model of d-galactose-induced aging. PLoS ONE 2014, 9, e101291. [Google Scholar] [CrossRef] [PubMed]
- Kojouharov, B.M.; Brackett, C.M.; Veith, J.M.; Johnson, C.P.; Gitlin, II; Gitlin, II; Toshkov, I.A.; Gleiberman, A.S.; Gudkov, A.V.; Burdelya, L.G. Toll-like receptor-5 agonist Entolimod broadens the therapeutic window of 5-fluorouracil by reducing its toxicity to normal tissues in mice. Oncotarget 2014, 5, 802–814. [Google Scholar] [CrossRef] [PubMed]
- Querfeld, U.; Mak, R.H. Vitamin D deficiency and toxicity in chronic kidney disease: In search of the therapeutic window. Pediatr. Nephrol. 2010, 25, 2413–2430. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Luo, X.; Ju, G.; Yu, X.; Hao, X.; Huang, Q.; Xiao, J.; Cui, L.; Kai, G. Increased accumulation of the cardio-cerebrovascular disease treatment drug tanshinone in Salvia miltiorrhiza hairy roots by the enzymes 3-hydroxy-3-methylglutaryl CoA reductase and 1-deoxy-d-xylulose 5-phosphate reductoisomerase. Funct. Integr. Genom. 2014, 14, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Huang, F.; Zhang, S.; Leung, S.W. Is danshen (Salvia miltiorrhiza) dripping pill more effective than isosorbide dinitrate in treating angina pectoris? A systematic review of randomized controlled trials. Int. J. Cardiol. 2012, 157, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zuo, Z.; Chow, M.S. Danshen: An overview of its chemistry, pharmacology, pharmacokinetics, and clinical use. J. Clin. Pharmacol. 2005, 45, 1345–1359. [Google Scholar] [CrossRef] [PubMed]
- Flood, M.T.; Kondo, M. Safety evaluation of lipase produced from Candida rugosa: Summary of toxicological data. Regul. Toxicol. Pharmacol. 2001, 33, 157–164. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Q.; Yang, M.; Deng, Y.; Yu, H.; Wang, L.; Teng, F.; Cho, K.; Ma, H.; Wu, P.; Li, X.; et al. The Safety Evaluation of Salvianolic Acid B and Ginsenoside Rg1 Combination on Mice. Int. J. Mol. Sci. 2015, 16, 29345-29356. https://doi.org/10.3390/ijms161226176
Zhao Q, Yang M, Deng Y, Yu H, Wang L, Teng F, Cho K, Ma H, Wu P, Li X, et al. The Safety Evaluation of Salvianolic Acid B and Ginsenoside Rg1 Combination on Mice. International Journal of Molecular Sciences. 2015; 16(12):29345-29356. https://doi.org/10.3390/ijms161226176
Chicago/Turabian StyleZhao, Qun, Min Yang, Yanping Deng, Haitao Yu, Linlin Wang, Fukang Teng, Kenka Cho, Hongmei Ma, Peng Wu, Xue Li, and et al. 2015. "The Safety Evaluation of Salvianolic Acid B and Ginsenoside Rg1 Combination on Mice" International Journal of Molecular Sciences 16, no. 12: 29345-29356. https://doi.org/10.3390/ijms161226176
APA StyleZhao, Q., Yang, M., Deng, Y., Yu, H., Wang, L., Teng, F., Cho, K., Ma, H., Wu, P., Li, X., Wu, W., Liu, X., Xu, F., Jiang, B., & Guo, D.-A. (2015). The Safety Evaluation of Salvianolic Acid B and Ginsenoside Rg1 Combination on Mice. International Journal of Molecular Sciences, 16(12), 29345-29356. https://doi.org/10.3390/ijms161226176






