Cloning of the Lycopene β-cyclase Gene in Nicotiana tabacum and Its Overexpression Confers Salt and Drought Tolerance
Abstract
:1. Introduction
2. Results
2.1. RNA Sequencing Analysis of Genes in the Carotenoid Biosynthetic Pathway and the Characterization of Ntβ-LCY Genes
| Gene | Transcript ID | Gene ID | Sample_FGS | Sample_FS | Sample_TS | Sample_LLMS |
|---|---|---|---|---|---|---|
| PSY1 | mRNA_108630_cds | Ntab0523090 | 50.00 | 20.26 | 26.26 | 26.84 |
| PSY2 | mRNA_24759_cds | Ntab0141080 | 5.72 | 6.43 | 5.15 | 4.42 |
| PSY3 | mRNA_28820_cds | Ntab0582610 | 90.19 | 31.8 | 35.02 | 48.05 |
| PSY4 | mRNA_3350_cds | Ntab0470140 | 46.29 | 35.54 | 24.17 | 26.29 |
| PDS1 | mRNA_13725_cds | Ntab0746310 | 79.50 | 42.74 | 56.13 | 65.04 |
| PDS2 | mRNA_73042_cds | Ntab0595110 | 103.66 | 63.22 | 80.42 | 64.72 |
| ZDS | mRNA_101234_cds | Ntab0653840 | 82.89 | 62.56 | 70.39 | 62.62 |
| CRTISO1 | mRNA_114973_cds | Ntab0634540 | 44.61 | 26.42 | 33.46 | 23.74 |
| CRTISO2 | mRNA_122944_cds | Ntab0027300 | 24.79 | 14.27 | 19.2 | 22.76 |
| CRTISO3 | mRNA_78351_cds | Ntab0736080 | 14.34 | 15.26 | 15.03 | 18.19 |
| β-LCY1 | mRNA_46713_cds | Ntab0268950 | 54.02 | 37.97 | 37.51 | 55.58 |
| β-LCY2 | mRNA_18729_cds | Ntab0383390 | 43.56 | 34.1 | 29.14 | 44.63 |
| ε-LCY1 | mRNA_60735_cds | Ntab0006110 | 48.65 | 22.31 | 29.74 | 32.46 |
| ε-LCY2 | mRNA_99724_cds | Ntab0455950 | 26.79 | 14.13 | 11.97 | 13.38 |
| β-OHase1 | mRNA_106915_cds | Ntab0677920 | 55.63 | 26.45 | 25.08 | 30.60 |
| β-OHase2 | mRNA_120276_cds | Ntab0861090 | 4.22 | 1.65 | 2.48 | 0.16 |
| β-OHase3 | mRNA_121754_cds | Ntab0486180 | 77.56 | 70.69 | 73.00 | 96.59 |
| ε-OHase1 | mRNA_131608_cds | Ntab0299130 | 28.18 | 10.21 | 23.92 | 20.13 |
| ε-OHase2 | mRNA_140553_cds | Ntab0895820 | 27.02 | 15.45 | 19.14 | 26.27 |
| VDE1 | mRNA_114230_cds | Ntab0858420 | 53.14 | 39.84 | 69.32 | 21.80 |
| VDE2 | mRNA_119637_cds | Ntab0230700 | 107.99 | 84.86 | 95.45 | 41.86 |
| VDE3 | mRNA_130498_cds | Ntab0721110 | 8.82 | 5.59 | 10.51 | 5.70 |
| VDE4 | mRNA_86361_cds | Ntab0189070 | 8.52 | 4.45 | 5.66 | 5.53 |
| VDE5 | mRNA_95599_cds | Ntab0607170 | 1.03 | 0.00 | 0.46 | 0.84 |
| ZE1 | mRNA_119539_cds | Ntab0136170 | 316.73 | 279.76 | 267.74 | 297.37 |
| ZE2 | mRNA_42563_cds | Ntab0384590 | 381.6 | 339.56 | 352.04 | 353.57 |

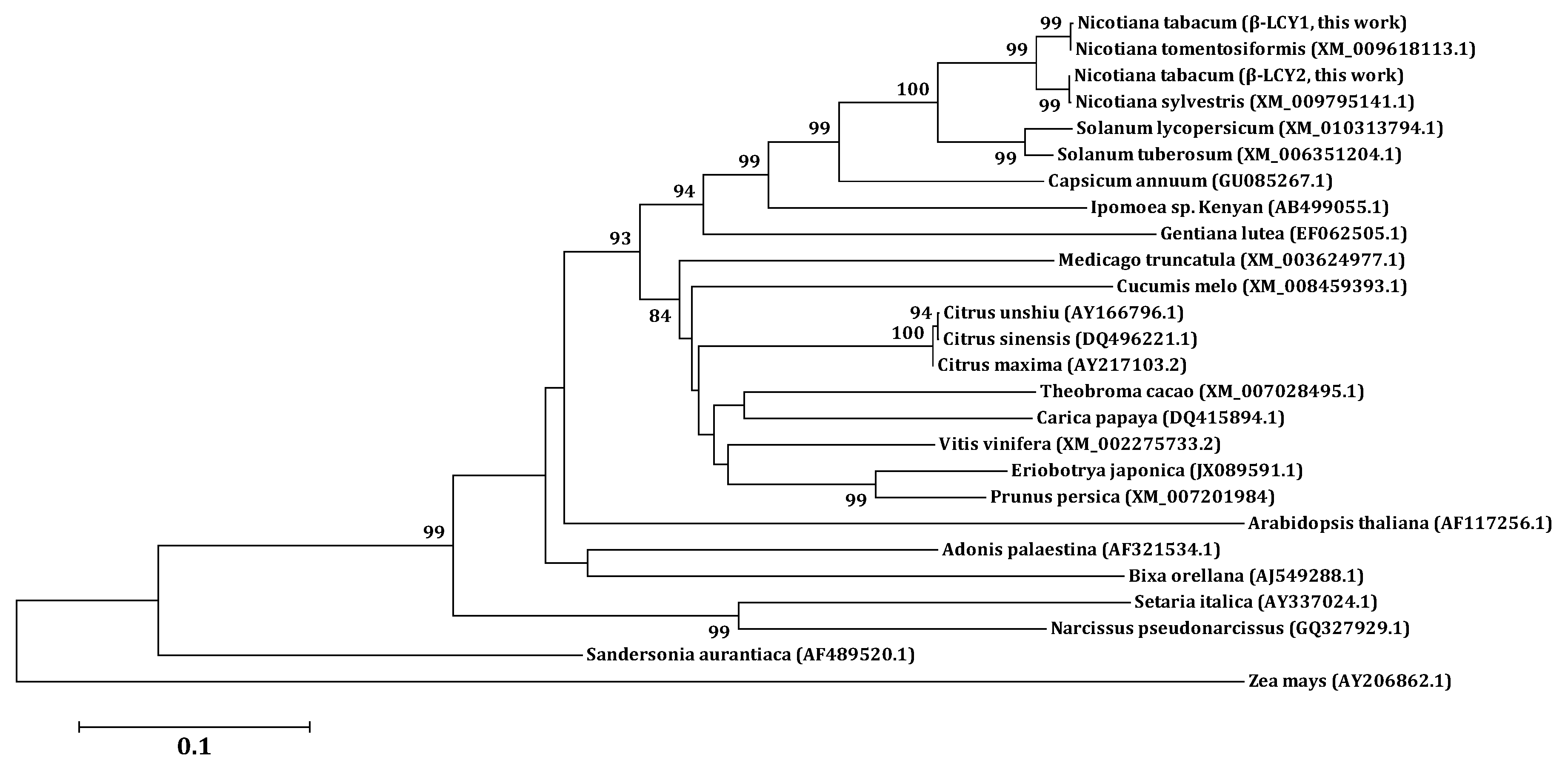
| Amino Acid | Ntomβ-LCY | Nsyβ-LCY | Stβ-LCY | Slβ-LCY | Caβ-LCY | Atβ-LCY | |
|---|---|---|---|---|---|---|---|
| Identity (%) | Ntβ-LCY1 | 99.8 | 96.8 | 91.4 | 90.6 | 89.2 | 77.0 |
| Ntβ-LCY2 | 97.0 | 100 | 91.0 | 90.4 | 89.2 | 77.0 |
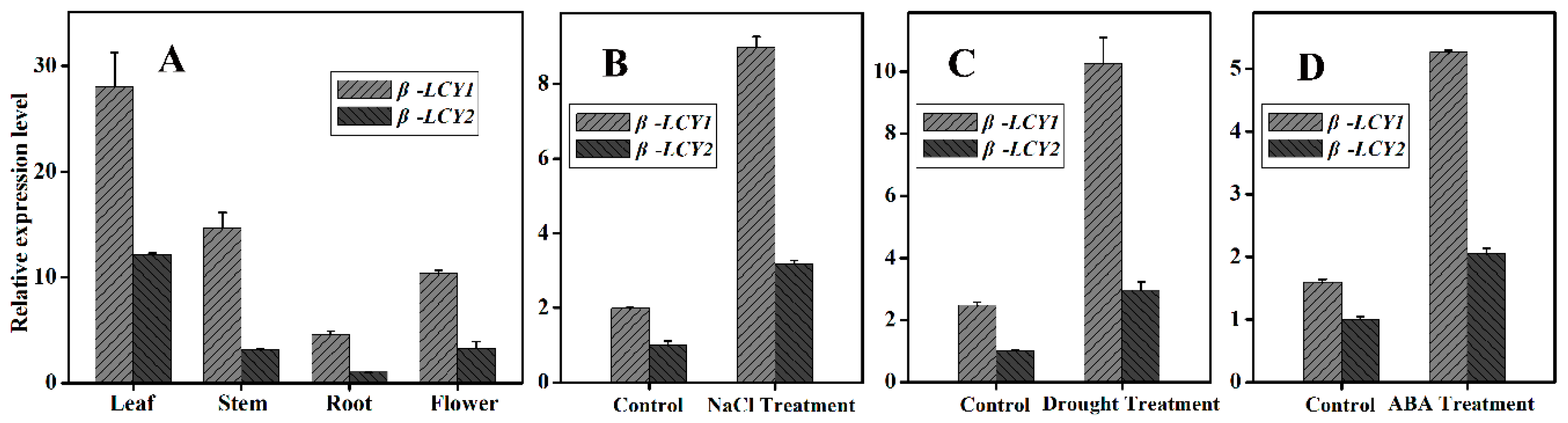
2.2. Examination of Ntβ-LCY Transcript Levels in Tobacco Organs and in Response to Stress Treatment
2.3. Characterization of Ntβ-LCY1 OE and RNAi Transgenic Tobacco Plants
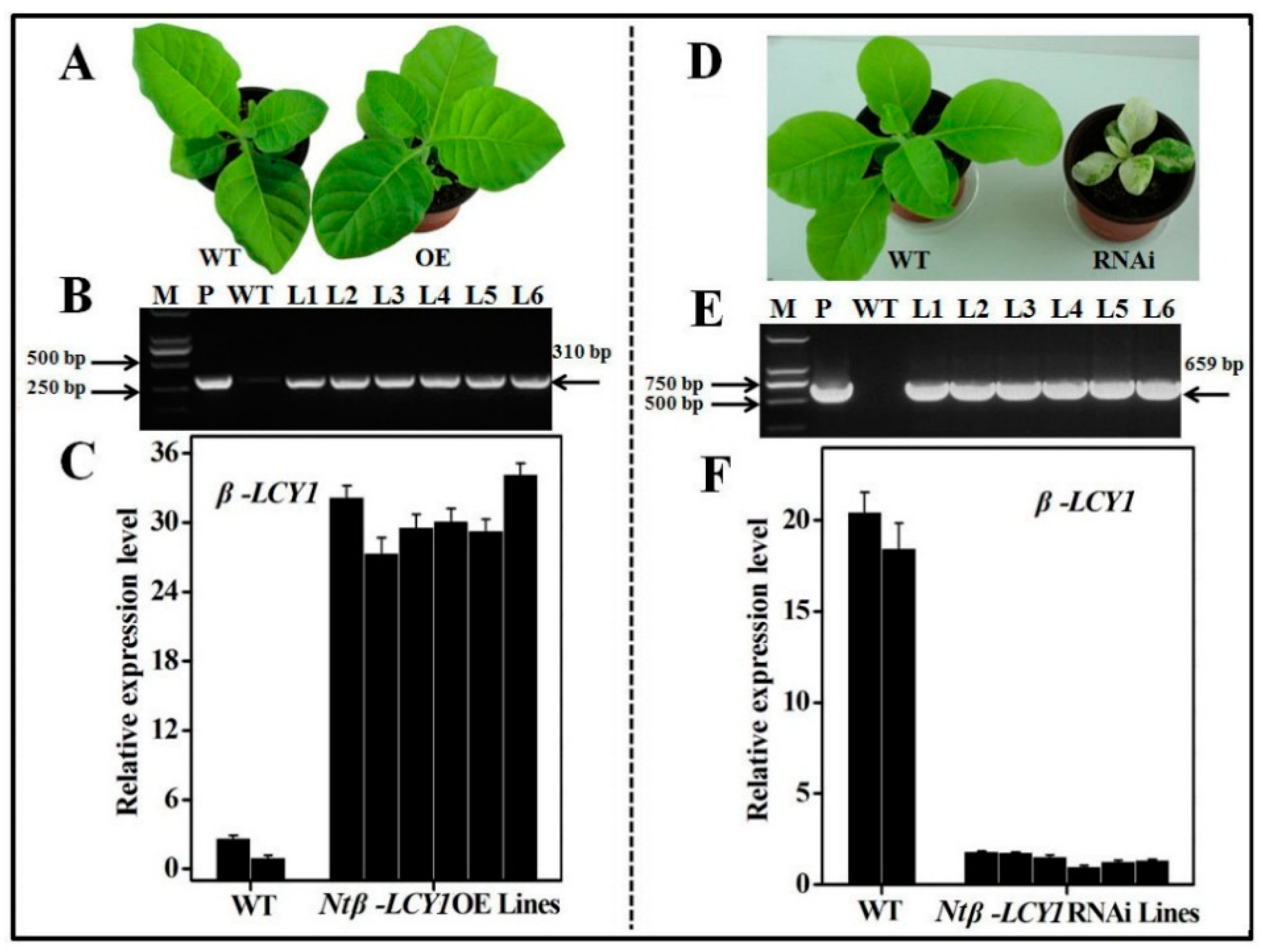
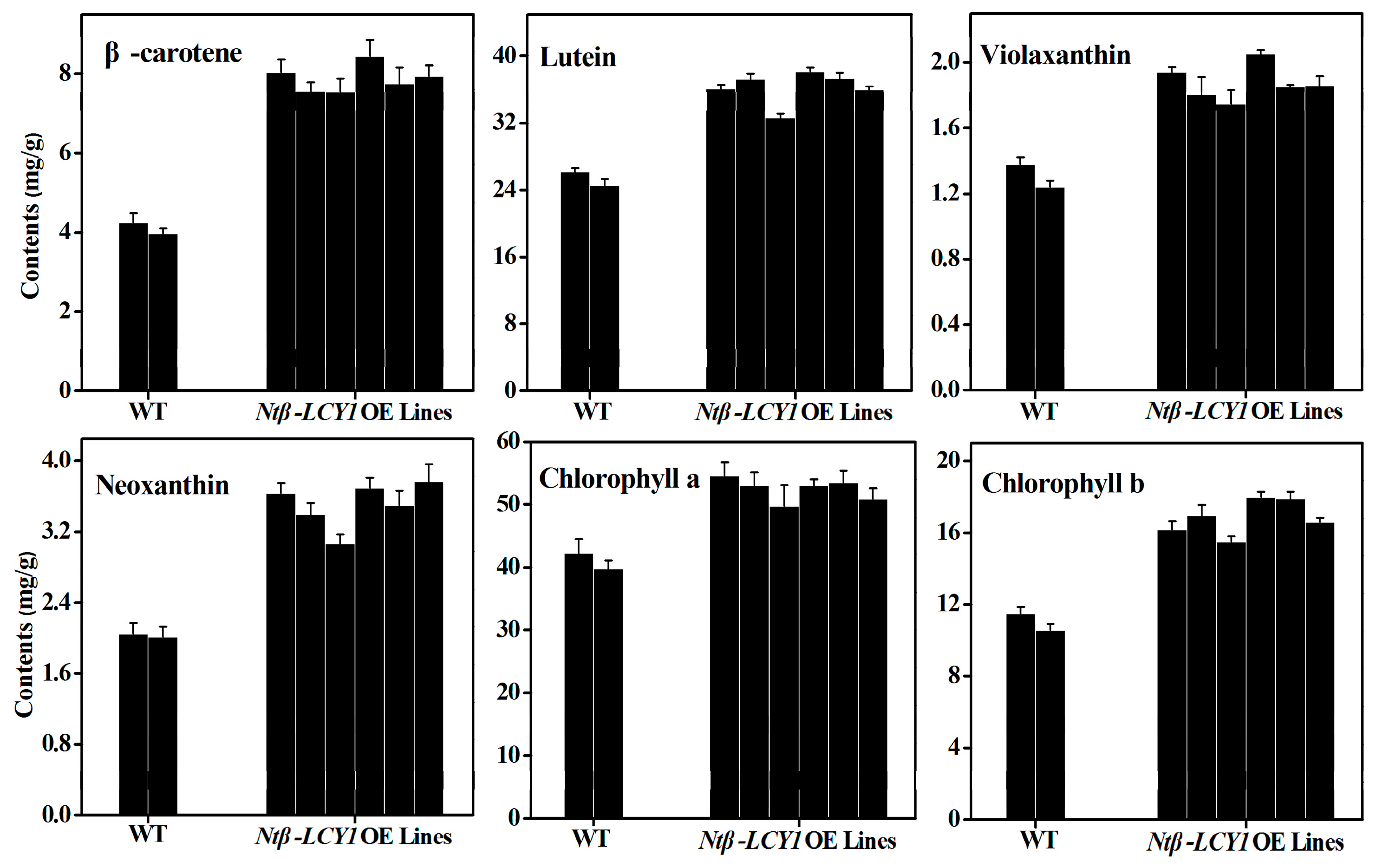

2.4. Functional Analysis of Ntβ-LCY1 under Salt Stress
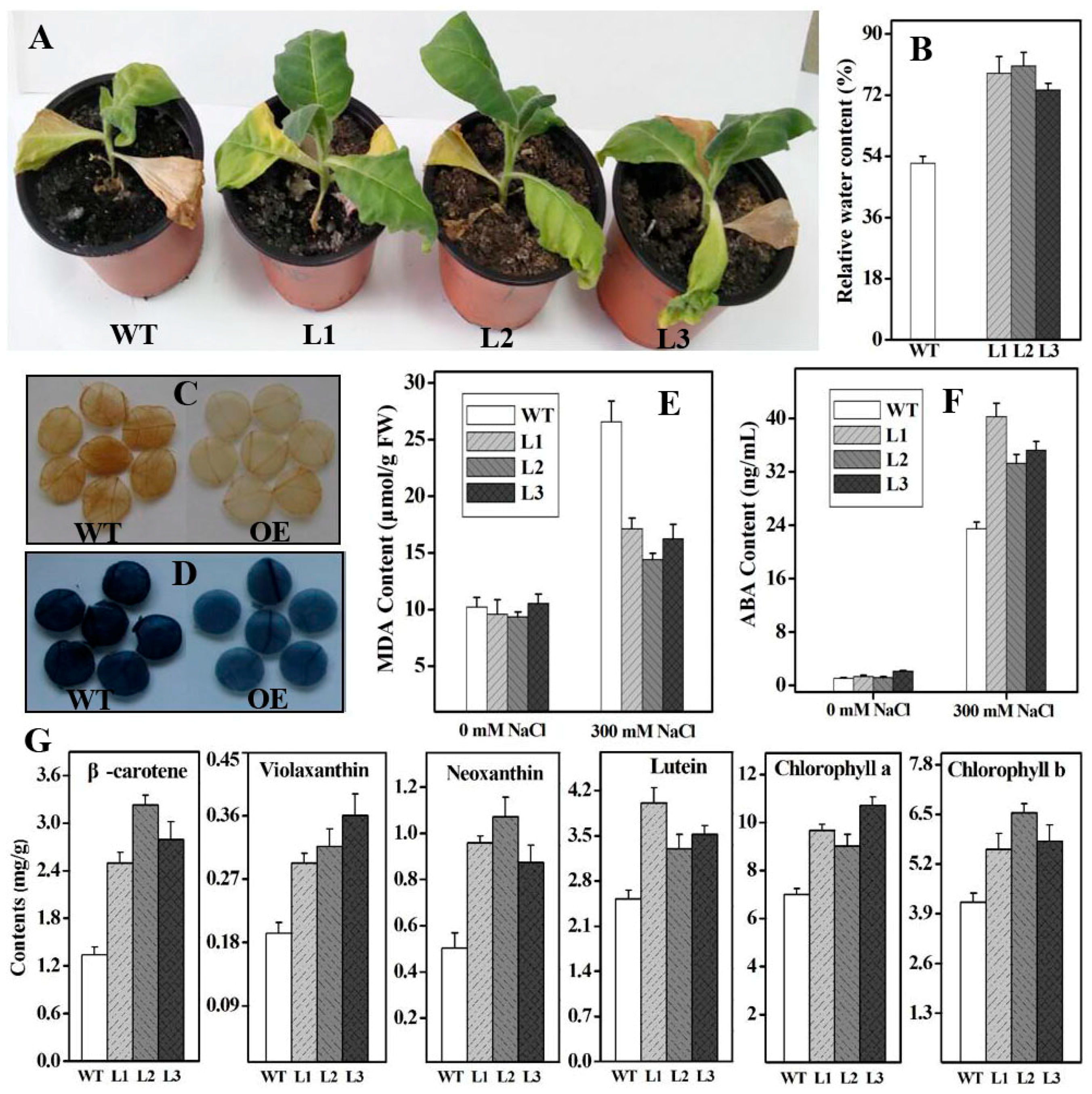
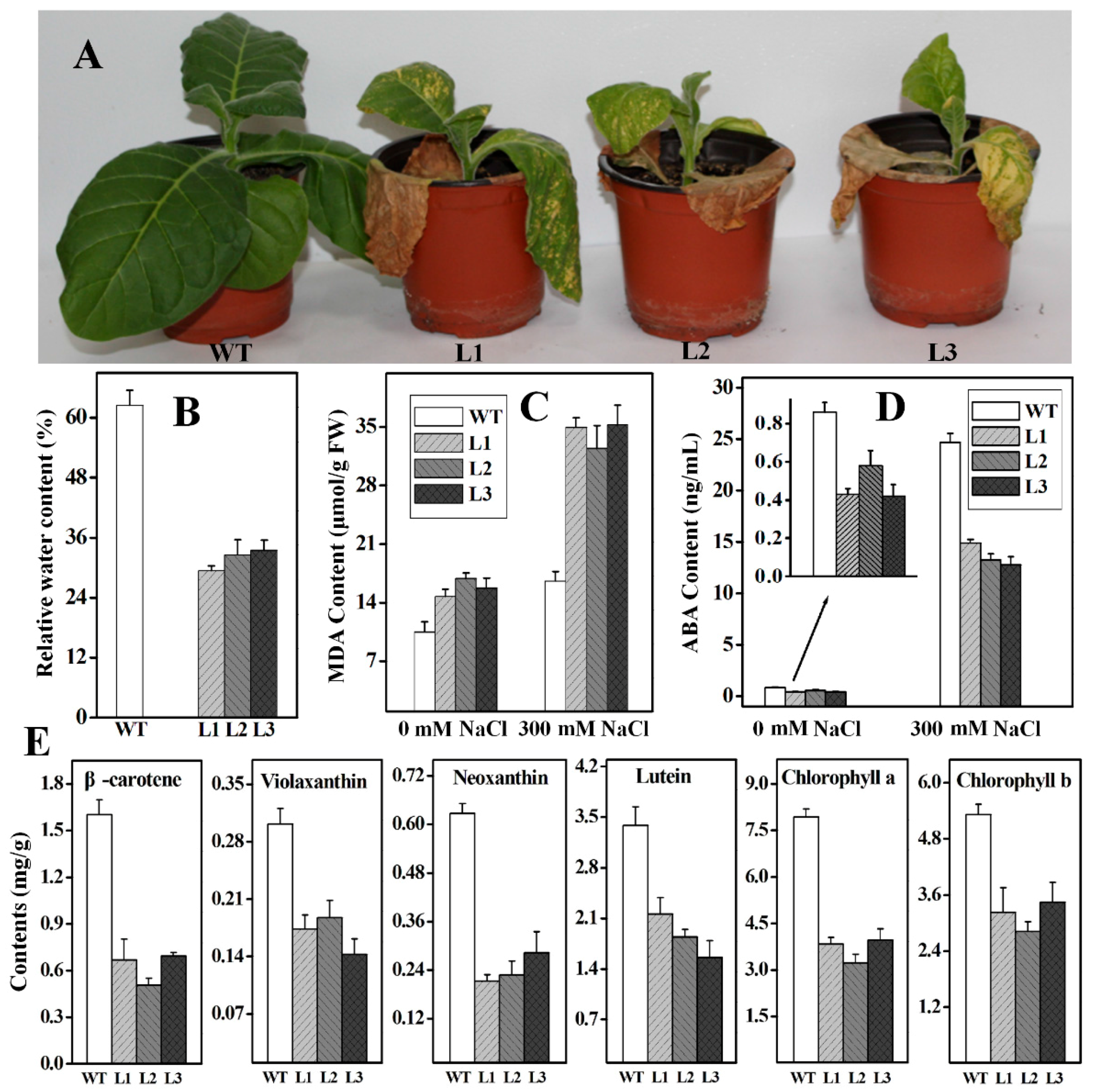
2.5. Functional Analysis of Ntβ-LCY1 under Drought Stress
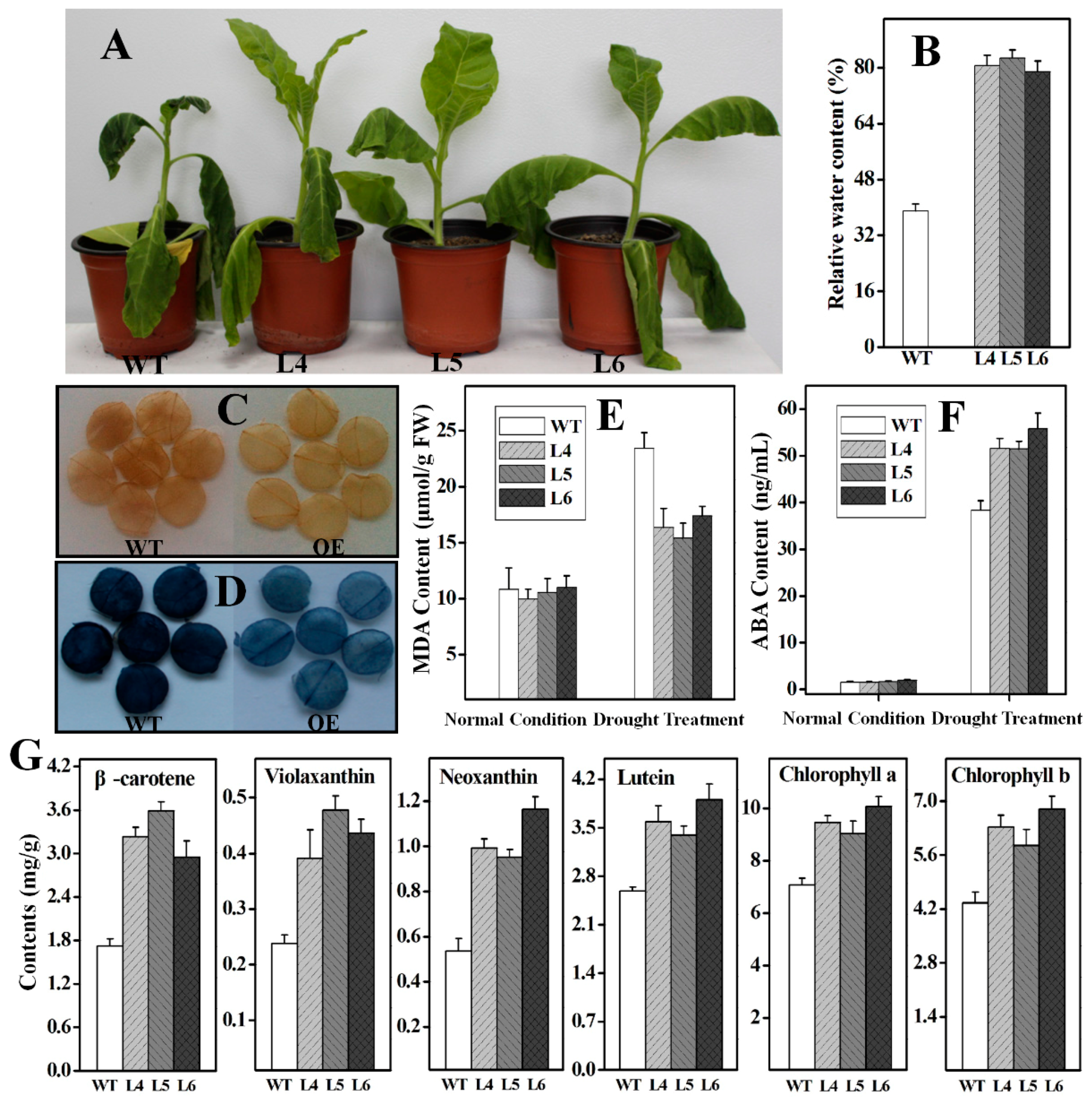
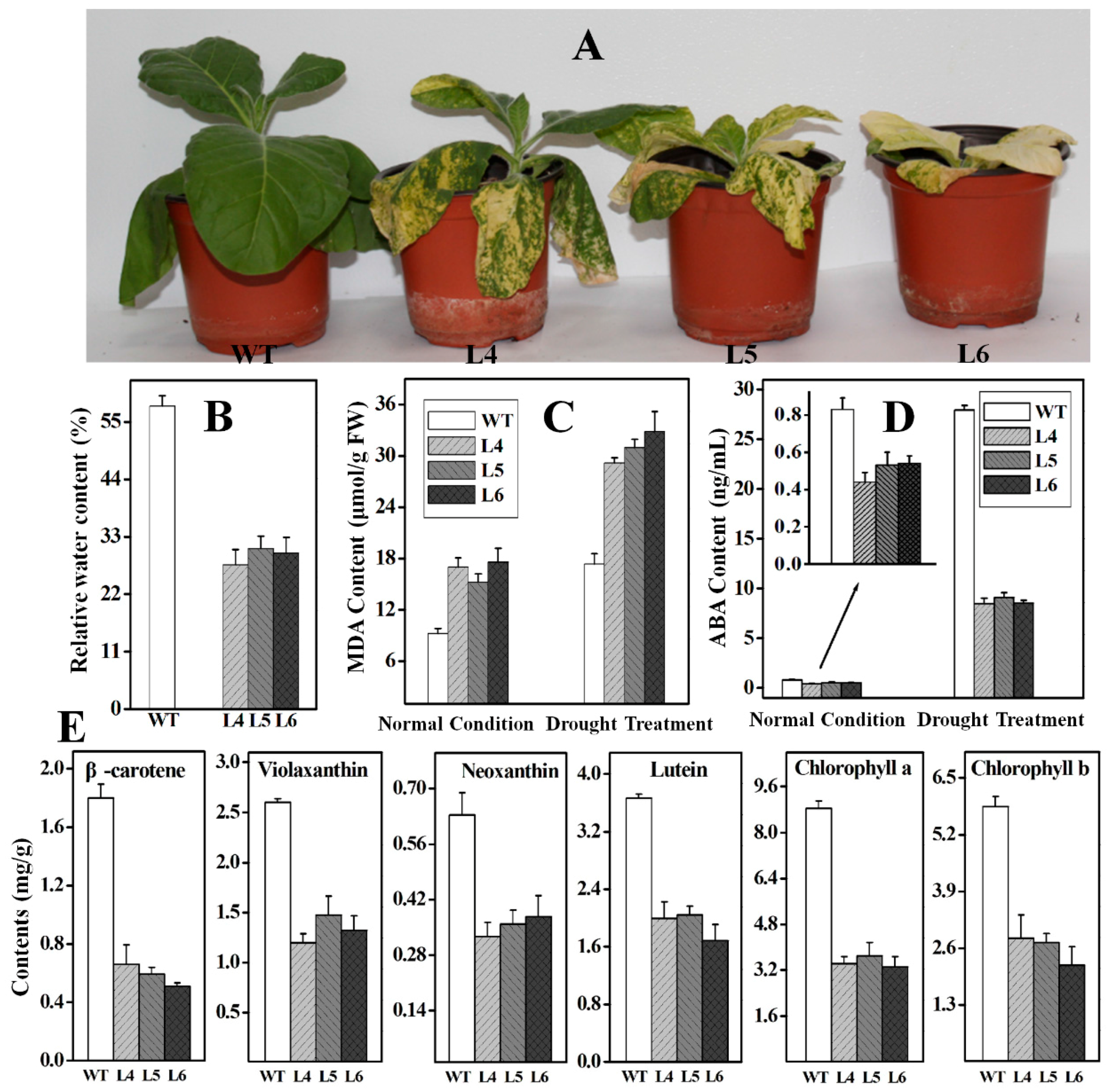
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. RNA Isolation and cDNA Preparation
4.3. cDNA Library Construction and Sequencing
4.4. Cloning of Ntβ-LCY1 and Vector Construction
4.5. Plant Transformation and Confirmation
4.6. Gene Expression Analysis
4.7. Carotenoid and Chlorophyll Extraction and Quantification
4.8. Salt and Drought Stress Treatments
4.9. Relative Water Content (RWC)
4.10. Detection of H2O2 and O2−
4.11. Determination of MDA Content
4.12. ABA Extraction and Quantification
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mahajan, S.; Tuteja, N. Cold, salinity and drought stresses: An overview. Arch. Biochem. Biophys. 2005, 444, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Bromham, L.; Saslis-Lagoudakis, C.H.; Bennett, T.H.; Flowers, T.J. Soil alkalinity and salt tolerance: adapting to multiple stresses. Biol. Lett. 2013, 9, 1744. [Google Scholar] [CrossRef] [PubMed]
- Evers, D.; Lefevre, I.; Legay, S.; Lamoureux, D.; Hausman, J.F.; Rosales, R.O.G.; Marca, L.R.T.; Hoffmann, L.; Bonierbale, M.; Schafleitner, R. Identification of drought-responsive compounds in potato through a combined transcriptomic and targeted metabolite approach. J. Exp. Bot. 2010, 61, 2327–2343. [Google Scholar] [CrossRef] [PubMed]
- Natwar, S.; Avinash, M.; Bhavanath, J. Over-expression of the Peroxisomal Ascorbate Peroxidase (SbpAPX) gene cloned from Halophyte Salicornia brachiata confers salt and drought stress tolerance in transgenic Tobacco. Mar. Biotechnol. 2014, 16, 321–332. [Google Scholar]
- Wu, G.; Wang, G.; Ji, J.; Gao, H.; Guan, W.; Wu, J.; Guan, C.; Wang, Y. Cloning of a cytosolic ascorbate peroxidase gene from Lycium chinense Mill. and enhanced salt tolerance by overexpressing in tobacco. Gene 2014, 543, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Y.; Yang, H.L.; Li, X.S.; Li, H.Y.; Wang, Y.C. Overexpression of Tamarix albiflonum TaMnSOD increases drought tolerance in transgenic cotton. Mol. Breed. 2014, 34, 1–11. [Google Scholar] [CrossRef]
- Tang, L.L.; Cai, H.; Zhai, H.; Luo, X.; Wang, Z.Y.; Cui, L.; Bai, X. Overexpression of Glycine soja WRKY20 enhances both drought and salt tolerance in transgenic alfalfa (Medicago sativa L.). Plant Cell Tissue Organ Cult. 2014, 118, 77–86. [Google Scholar] [CrossRef]
- Xu, Q.; He, Q.; Li, S.; Tian, Z. Molecular characterization of StNAC2 in potato and its overexpression confers drought and salt tolerance. Acta Physiol. Plant 2014, 36, 1841–1851. [Google Scholar] [CrossRef]
- Min, D.H.; Zhao, Y.; Huo, D.Y.; Li, L.C.; Chen, M.; Xu, Z.S.; Ma, Y.Z. Isolation and identification of a wheat gene encoding a zinc finger protein (TaZnFP) responsive to abiotic stresses. Acta Physiol. Plant 2013, 35, 1597–1604. [Google Scholar] [CrossRef]
- Sun, P.P.; Zhu, X.F.; Huang, X.S.; Liu, J.H. Overexpression of a stress-responsive MYB transcription factor of Poncirus trifoliata confers enhanced dehydration tolerance and increases polyamine biosynthesis. Plant Physiol. Biochem. 2014, 78, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, R.; Khurram, S.; Rashid, A.; Moddassir, A.; Imran, H.; Shahid, M.; Gerald, A.B.; Nasir, A.S. Cloning and characterization of Na+/H+ antiporter (LfNHX1) gene from a halophyte grass Leptochloa fusca for drought and salt tolerance. Mol. Biol. Rep. 2014, 41, 1669–1682. [Google Scholar]
- Yarra, R.; He, S.J.; Abbagani, S.; Ma, B.; Bulle, M.; Zhang, W.K. Overexpression of a wheat Na+/H+ antiporter gene (TaNHX2) enhances tolerance to salt stress in transgenic tomato plants (Solanum lycopersicum L.). Plant Cell Tissue Organ Cult. 2012, 111, 49–57. [Google Scholar] [CrossRef]
- Bramley, P.M. The regulation and genetic manipulation of carotenoid biosynthesis in tomato fruit. Pure Appl. Chem. 1997, 69, 2159–2162. [Google Scholar] [CrossRef]
- Dall’Osto, L.; Fiore, A.; Cazzaniga, S.; Giuliano, G.; Bassi, R. Different roles of α- and β-branch xanthophylls in photosystem assembly and photoprotection. J. Biol. Chem. 2007, 282, 35056–35068. [Google Scholar] [CrossRef] [PubMed]
- Andrade-Souza, V.; Costa, M.G.C.; Chen, C.X.; Gmitter, F.G., Jr.; Costa, M.A. Physical location of the carotenoid biosynthesis genes Psy and β-Lcy in Capsicum annuum (Solanaceae) using heterologous probes from Citrus sinensis (Rutaceae). Genet. Mol. Res. 2011, 10, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Nambara, E.; Marion-Poll, A. Abscisic acid biosynthesis and catabolism. Annu. Rev. Plant Biol. 2005, 56, 165–185. [Google Scholar] [CrossRef] [PubMed]
- Taylor, I.B.; Sonneveld, T.; Bugg, T.D.H.; Thompson, A.J. Regulation and manipulation of the biosynthesis of abscisic acid, including the supply of xanthophyll precursors. J. Plant Growth Regul. 2005, 24, 253–273. [Google Scholar] [CrossRef]
- Miller, G.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis and signaling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Götz, T.; Sandmann, G.; Römer, S. Expression of bacterial carotene hydroxylase gene (crtZ) enhances UV tolerance in tobacco. Plant Mol. Biol. 2002, 50, 127–140. [Google Scholar] [CrossRef]
- Davison, P.A.; Hunter, C.N.; Horton, P. Overexpression of β-carotene hydroxylase enhances stress tolerance in Arabidopsis. Nature 2002, 418, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Li, Y.; Zhou, S. Overexpression of phytoene synthase gene from Salicornia europaea alters response to reactive oxygen species under salt stress in transgenic Arabidopsis. Biotechnol. Lett. 2008, 30, 1501–1507. [Google Scholar] [CrossRef] [PubMed]
- Howitt, C.A.; Cavanagh, C.R.; Bowerman, A.F.; Cazzonelli, C.; Rampling, L.; Mimica, J.L.; Pogson, B.J. Alternative splicing, activation of cryptic exons and amino acid substitutions in carotenoid biosynthetic genes are associated with lutein accumulation in wheat endosperm. Funct. Integr. Genom. 2009, 9, 363–376. [Google Scholar] [CrossRef] [PubMed]
- Harjes, C.E.; Rocheford, T.R.; Bai, L.; Brutnell, T.P.; Kandianis, C.B.; Sowinski, S.G.; Stapleton, A.E.; Vallabhaneni, R.; Williams, M.; Wurtzel, E.T.; et al. Natural genetic variation in lycopene epsilon cyclase tapped for maize biofortification. Science 2008, 319, 330–333. [Google Scholar] [CrossRef] [PubMed]
- Apel, W.; Bock, R. Enhancement of carotenoid biosynthesis in transplastomic tomatoes by induced lycopene-to-provitamin A conversion. Plant Physiol. 2009, 151, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Diretto, G.; Welsch, R.; Tavazza, R.; Mourgues, F.; Pizzichini, D.; Beyer, P.; Giuliano, G. Silencing of beta-carotene hydroxylase increases total carotenoid and beta-carotene levels in potato tubers. BMC Plant Biol. 2007, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.; Kim, S.; Leskovar, D.; King, S. Development of a codominant CAPS marker for allelic selection between canary yellow and red watermelon based on SNP in lycopene β-cyclase (LCYB) gene. Mol. Breed. 2007, 20, 63–72. [Google Scholar] [CrossRef]
- Cazzonelli, C.I.; Pogson, B.J. Source to sink: regulation of carotenoid biosynthesis in plants. Trends Plant Sci. 2010, 15, 266–274. [Google Scholar] [CrossRef] [PubMed]
- D’Ambrosio, C.; Giorio, G.; Marino, I.; Merendino, A.; Petrozza, A.; Salfi, L.; Stigliani, A.; Cellini, F. Virtually complete conversion of lycopene into β-carotene in fruits of tomato plants transformed with the tomato lycopene β-cyclase (tlcy-β) cDNA. Plant Sci. 2004, 166, 207–214. [Google Scholar] [CrossRef]
- Cunningham, F.X.; Pogson, B.; Sun, Z.; McDonald, K.A.; DellaPenna, D.; Gantt, E. Functional analysis of the beta and epsilon lycopene cyclase enzymes of Arabidopsis reveals a mechanism for control of cyclic carotenoid formation. Plant Cell 1996, 8, 1613–1626. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Musetti, V.; Kim, J.; Magallanes-Lundback, M.; DellaPenna, D. The Arabidopsis LUT1 locus encodes a member of the cytochrome P450 family that is required for carotenoid epsilon-ring hydroxylation activity. Proc. Natl. Acad. Sci. USA 2004, 101, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Zhou, W.; Zhang, J.; Xu, Q.; Deng, X. Effect of the Citrus Lycopene β-Cyclase Transgene on Carotenoid Metabolism in Transgenic Tomato Fruits. PLoS ONE 2012, 7, e32221. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Y.; Han, H.P.; Jiang, P.; Nie, L.L.; Bao, H.X.; Fan, P.X.; Lv, S.; Feng, J.; Li, Y. Transformation of β-lycopene cyclase genes from Salicornia europaea and Arabidopsis conferred salt tolerance in Arabidopsis and tobacco. Plant Cell Physiol. 2011, 52, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Robles, P.; Micol, J.L.; Quesada, V. Arabidopsis MDA1, a nuclearencoded protein, functions in chloroplast development and abiotic stress responses. PLoS ONE 2012, 7, e42924. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.L.; Ma, J.T.; Zhao, Y.M.; Wei, Y.H.; Tang, Y.X.; Wu, Y.M. Improvement of drought and salt tolerance in Arabidopsis and Lotus corniculatus by overexpression of a novel DREB transcription factor from Populus euphratica. Gene 2012, 506, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Danquah, A.; de Zelicourt, A.; Colcombet, J.; Hirt, H. The role of ABA and MAPK signaling pathways in plant abiotic stress responses. Biotechnol. Adv. 2014, 32, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.X.; Chiou, C.Y.; Shen, C.H.; Chen, P.J.; Liu, Y.C.; Jian, C.D.; Shen, X.L.; Shen, F.Q.; Yeh, K.W. RNA interference-based gene silencing of phytoene synthase impairs growth, carotenoids, and plastid phenotype in Oncidium hybrid orchid. SpringerPlus 2014, 3, 478. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Jeong, J.C.; Park, S.; Bae, J.Y.; Ahn, M.J.; Lee, H.S.; Kwak, S.S. Down-regulation of sweetpotato lycopene β-cyclase gene enhances tolerance to abiotic stress in transgenic calli. Mol. Biol. Rep. 2014, 41, 8137–8148. [Google Scholar] [CrossRef] [PubMed]
- Pogson, B.J.; Rissler, H.M. Genetic manipulation of carotenoid biosynthesis and photoprotection. Philos. Trans. R. Soc. Lond. B 2000, 355, 1395–1403. [Google Scholar] [CrossRef] [PubMed]
- De Azevedo Neto, A.D.; Prisco, J.T.; Enéas-Filho, J.; Braga de Abreu, C.E.; Gomes-Filho, E. Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ. Exp. Bot. 2006, 56, 87–94. [Google Scholar] [CrossRef]
- Bode, S.; Quentmeier, C.C.; Liao, P.N.; Barrosc, T.; Walla, P.J. Xanthophyll-cycle dependence of the energy transfer between carotenoid dark states and chlorophylls in NPQ mutants of living plants and in LHC II. Chem. Phys. Lett. 2008, 450, 379–385. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W. Antioxidants in photosynthesis and human nutrition. Science 2002, 298, 2149–2153. [Google Scholar] [CrossRef] [PubMed]
- Parvaiz, A.; Satyawati, S. Salt stress and phyto-biochemical responses of plants-a review. Plant Soil Environ. 2008, 54, 89–99. [Google Scholar]
- Jithesh, M.N.; Prashanth, S.R.; Sivaprakash, K.R.; Parida, A.K. Antioxidative response mechanisms in halophytes: Their role in stress defense. J. Genet. 2006, 85, 237–254. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Ahn, Y.O.; Ahn, M.J.; Lee, H.S.; Kwak, S.S. Down-regulation of β-carotene hydroxylase increases β-carotene and total carotenoids enhancing salt stress tolerance in transgenic cultured cells of sweet potato. Phytochemistry 2012, 74, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.M.; Wang, R.; Luo, Z.P.; Jin, L.F.; Liu, P.P.; Chen, Q.S.; Li, Z.F.; Li, F.; Wei, C.Y.; Wu, M.Z.; et al. Molecular cloning and functional characterization of the lycopene ε-Cyclase gene via virus-induced gene silencing and its expression pattern in Nicotiana tabacum. Int. J. Mol. Sci. 2014, 15, 14766–14785. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.M.; Liu, P.P.; Xia, Y.Z.; Wei, P.; Li, W.Z.; Zhang, W.; Chen, X.; Cao, P.J.; Xu, Y.L.; Jin, L.F.; et al. Downregulation of the lycopene ε-cyclase gene confers tolerance to salt and drought stress in Nicotiana tabacum. Acta Physiol. Plant 2015, 37, 210. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, Y.H.; Ahn, Y.O.; Ahn, M.J.; Jeong, J.C.; Lee, H.S.; Kwak, S.S. Down-regulation of the lycopene-ε-cyclase gene increases carotenoid synthesis via the β-branch-specific pathway and enhances salt-stress tolerance in sweetpotato transgenic calli. Physiol. Plant 2012, 147, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Gene expression and signal transduction in water-stress response. Plant Physiol. 1997, 115, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Pang, T.; Ye, C.Y.; Xia, X.L.; Yin, W.L. De novo sequencing and transcriptome analysis of the desert shrub, Ammopiptanthus mongolicus, during cold acclimation using Illumina/Solexa. BMC Genom. 2013, 14, 488. [Google Scholar] [CrossRef] [PubMed]
- Horsch, R.B.; Fry, J.E.; Hoffmann, N.L.; Eicholtz, D.; Rogers, S.G.; Fraley, R.T. A simple method for transferring genes into plants. Science 1985, 227, 1229–1231. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Boyer, J.S. Measurement of the water status of plants. Annu. Rev. Plant Physiol. 1969, 9, 351–363. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, L.; Meng, H.; Wen, H.T.; Fan, Y.L.; Zhao, J. Maize ABP9 enhances tolerance to multiple stresses in transgenic Arabidopsis by modulating ABA signaling and cellular levels of reactive oxygen species. Plant Mol. Biol. 2011, 75, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sun, P.P.; Chen, C.L.; Wang, Y.; Fu, X.Z.; Liu, J.H. An arginine decarboxylase gene PtADC from Poncirus trifoliata confers abiotic stress tolerance and promotes primary root growth in Arabidopsis. J. Exp. Bot. 2011, 62, 2899–2914. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.B.; Li, X.H.; Xiao, J.H.; Wang, S.P. A convenient method for simultaneous quantification of multiple phytohormones and metabolites: Application in study of rice-bacterium interaction. Plant Methods 2012, 8, 2. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, Y.; Guo, J.; Zhang, W.; Jin, L.; Liu, P.; Chen, X.; Li, F.; Wei, P.; Li, Z.; Li, W.; et al. Cloning of the Lycopene β-cyclase Gene in Nicotiana tabacum and Its Overexpression Confers Salt and Drought Tolerance. Int. J. Mol. Sci. 2015, 16, 30438-30457. https://doi.org/10.3390/ijms161226243
Shi Y, Guo J, Zhang W, Jin L, Liu P, Chen X, Li F, Wei P, Li Z, Li W, et al. Cloning of the Lycopene β-cyclase Gene in Nicotiana tabacum and Its Overexpression Confers Salt and Drought Tolerance. International Journal of Molecular Sciences. 2015; 16(12):30438-30457. https://doi.org/10.3390/ijms161226243
Chicago/Turabian StyleShi, Yanmei, Jinggong Guo, Wei Zhang, Lifeng Jin, Pingping Liu, Xia Chen, Feng Li, Pan Wei, Zefeng Li, Wenzheng Li, and et al. 2015. "Cloning of the Lycopene β-cyclase Gene in Nicotiana tabacum and Its Overexpression Confers Salt and Drought Tolerance" International Journal of Molecular Sciences 16, no. 12: 30438-30457. https://doi.org/10.3390/ijms161226243
APA StyleShi, Y., Guo, J., Zhang, W., Jin, L., Liu, P., Chen, X., Li, F., Wei, P., Li, Z., Li, W., Wei, C., Zheng, Q., Chen, Q., Zhang, J., Lin, F., Qu, L., Snyder, J. H., & Wang, R. (2015). Cloning of the Lycopene β-cyclase Gene in Nicotiana tabacum and Its Overexpression Confers Salt and Drought Tolerance. International Journal of Molecular Sciences, 16(12), 30438-30457. https://doi.org/10.3390/ijms161226243







