Overnutrition Determines LPS Regulation of Mycotoxin Induced Neurotoxicity in Neurodegenerative Diseases
Abstract
:1. Introduction
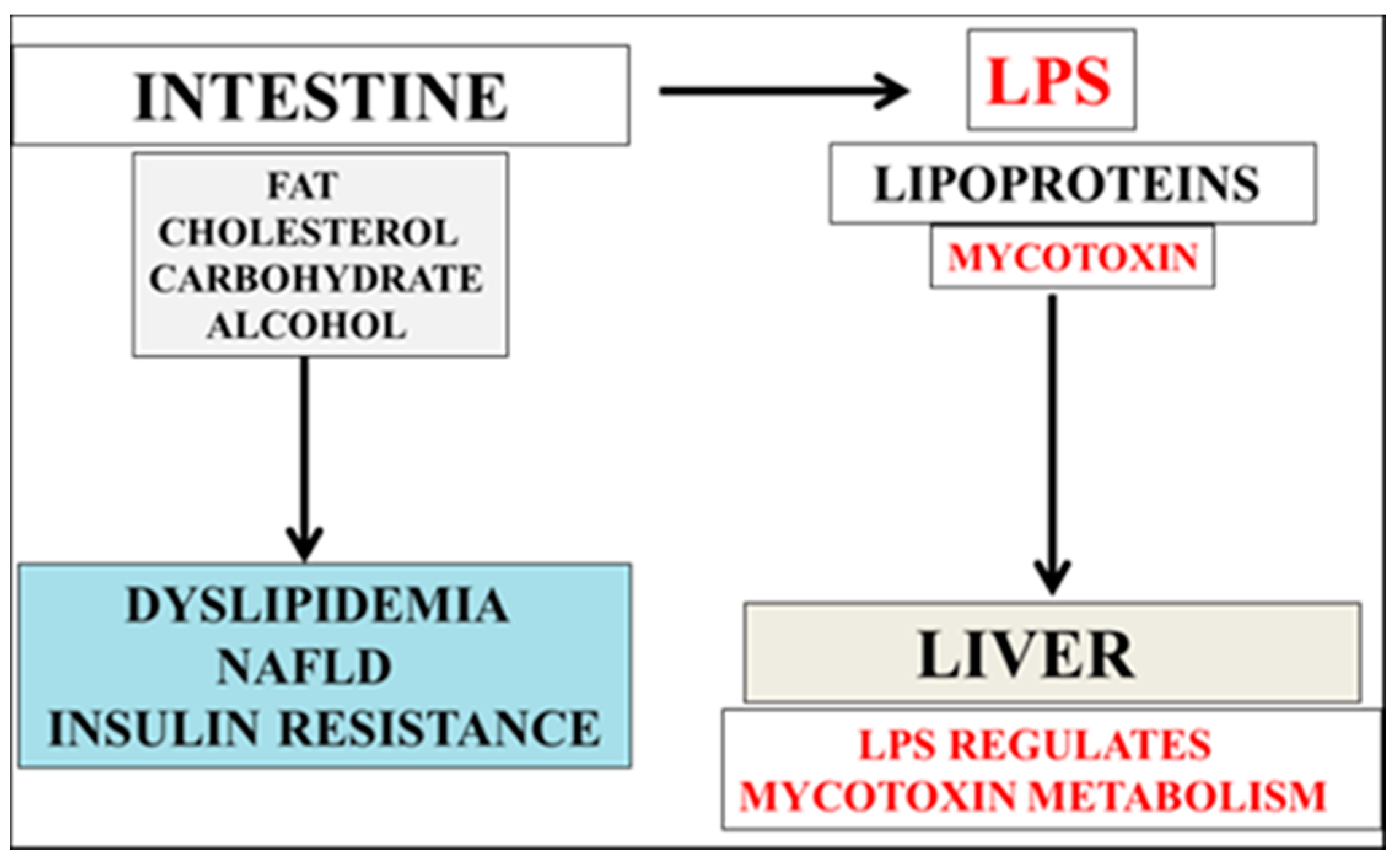
2. Mycotoxin and LPS Regulate Cholesterol and Aβ Metabolism
3. LPS/Mycotoxin Interactions Interfere with Apolipoprotein E/Aβ Peptide Interactions and Determine Neuron Survival
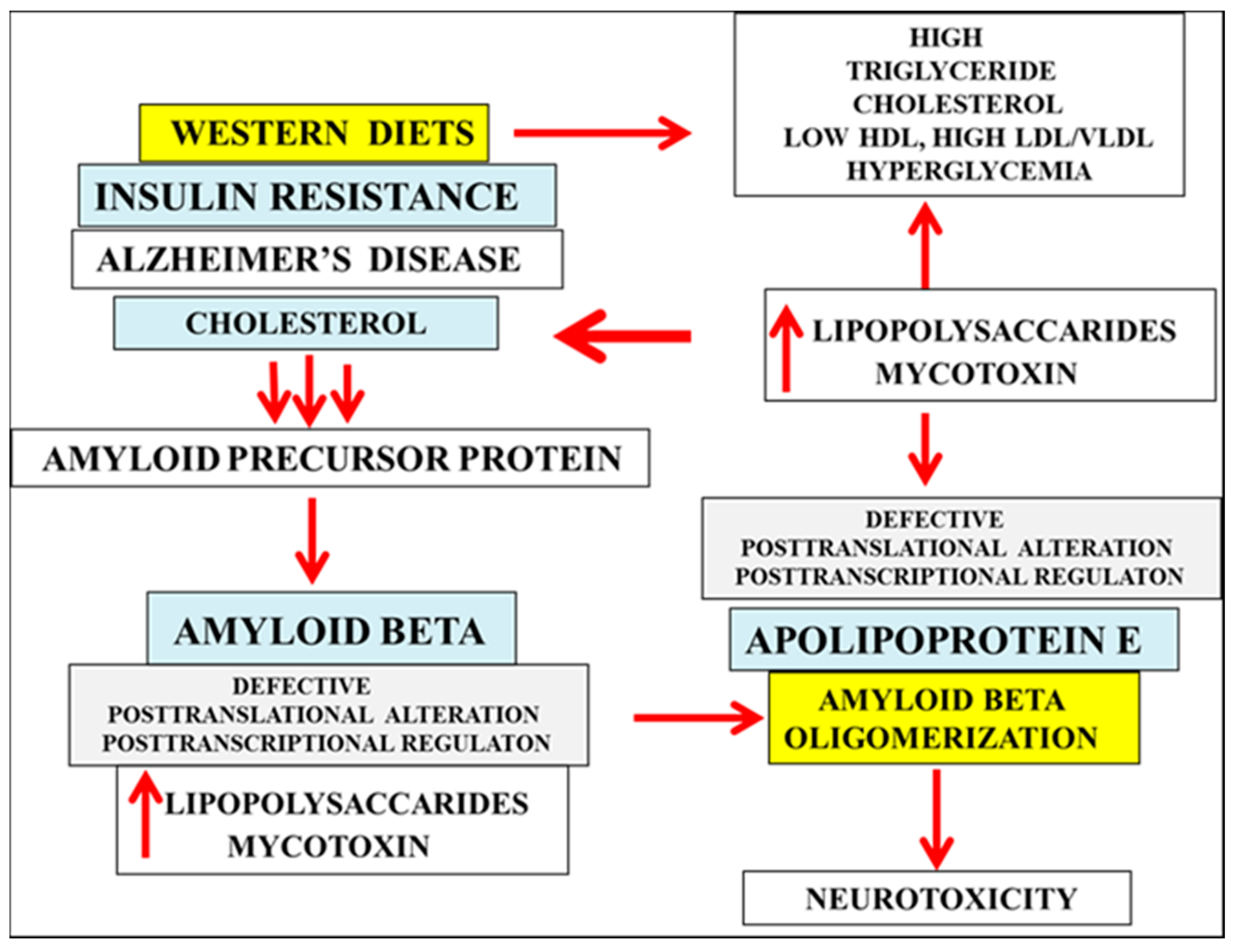
4. Patulin and LPS Effect Electrostatic Aβ Oligomer Formation via Post Translational or Post Transcriptional Modifications
5. Nutritional Diets Reduce Neurotoxins and Allow Effective Drug Treatment Programs
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Martín, M.G.; Pfrieger, F.; Dotti, C.G. Cholesterol in brain disease: Sometimes determinant and frequently implicated. EMBO Rep. 2014, 15, 1036–1052. [Google Scholar] [CrossRef] [PubMed]
- Vance, J.E. Dysregulation of cholesterol balance in the brain: Contribution to neurodegenerative diseases. Dis. Models Mech. 2012, 5, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Benarroch, E.E. Brain cholesterol metabolism and neurologic disease. Neurology 2008, 71, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J.; Fernando, W. High fibre diets and Alzheimer’s disease. Food Nutr. Sci. 2014, 5, 410–424. [Google Scholar] [CrossRef]
- Jonkers, I.J.; Smelt, A.H.; Hattori, H.; Scheek, L.M.; van Gent, T.; de Man, F.H.; van der Laarse, A.; van Tol, A. Decreased PLTP mass but elevated PLTP activity linked to insulin resistance in HTG: Effects of bezafibrate therapy. J. Lipid Res. 2003, 44, 1462–1469. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.C.; Shiu, S.W.; Wong, Y.; Wong, W.K.; Tam, S. Plasma apolipoprotein E concentration is an important determinant of phospholipid transfer protein activity in type 2 diabetes mellitus. Diabetes Metab. Res. Rev. 2006, 22, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J.; Creegan, R. Links between insulin resistance, lipoprotein metabolism and amyloidosis in Alzheimer’s disease. Health 2014, 6, 1549–1579. [Google Scholar] [CrossRef]
- Dombrink-Kurtzman, M.A. Chapter 2—Economic Aspects of Mycotoxins in Fruits and Vegetables. In Mycotoxins in Fruits and Vegetables; Academic Press: San Diego, CA, USA, 2008; pp. 27–44. [Google Scholar]
- Hymery, N.; Vasseur, V.; Coton, M.; Mounier, J.; Jany, J.L.; Barbier, G.; Coton, E. Filamentous fungi and mycotoxins in cheese: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 437–456. [Google Scholar] [CrossRef]
- Makun, H.S. (Ed.) Mycotoxin and Food Safety in Developing Countries. 2013. Available online: http://www.intechopen.com (accessed on 9 September 2015).
- Shephard, G.S. Impact of mycotoxins on human health in developing countries. Food Addit. Contam. 2008, 25, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.D. Fungi and mycotoxins in grain: Implications for stored product research. J. Stored Prod. Res. 1995, 31, 1–16. [Google Scholar] [CrossRef]
- Levi, C. Mycotoxins in coffee. J. Assoc. Off. Anal. Chem. 1980, 63, 1282–1285. [Google Scholar] [PubMed]
- Wild, C.P.; Gong, Y.Y. Mycotoxins and human disease: A largely ignored global health issue. Carcinogenesis 2010, 31, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Afsah-Hejri, L.; Jinap, S.; Hajeb, P.; Radu, S. A review on mycotoxins in food and feed: Malaysia case study. Shakibazadeh 2013, 12, 629–651. [Google Scholar]
- Culliao, A.G.; Barcelo, J.M. Fungal and mycotoxin contamination of coffee beans in Benguet province, Philippines. Food Addit. Contam. 2015, 32, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Peraica, M.; Radić, B.; Lucić, A.; Pavlović, M. Toxic effects of mycotoxins in humans. Bull. World Health Organ 1999, 77, 754–766. [Google Scholar] [PubMed]
- Modified Nanoparticles for Lipophilic Toxin Sequestration Published on SBIR.gov Modified Nanoparticles for Lipophilic Toxin Sequestration Principal Investigator: Weston Daniel Research Scientist, 2013. Northwestern University. Available online: https://www.sbir.gov (accessed on 7 May 2015).
- Anyanwu, E.; Ehiri, J.; Kanu, I. High cholesterol levels and chronic exposure to toxigenic molds in damp buildings: A high risk for cardiovascular diseases and stroke. Internet J. Toxicol. 2006, 3, 1–9. [Google Scholar]
- Costantini, A.V.; Am Muehlbach, M.D. (Eds.) Mycotoxin-induced atherosclerosis. The Fungalbionic Book Series. Available online: www.fungalbionicbookseries.com/blue (accessed on 4 September 2015).
- Martins, I.J.; Mortimer, B.-C.; Redgrave, T.G. Effect of the ACAT inhibitor CL 277,082 on apolipoprotein B48 transport in mesenteric lymph and on the plasma clearance of chylomicrons and remnants. Arteriosclerosis. Thromb. Vasc. Biol. 1997, 17, 211–216. [Google Scholar] [CrossRef]
- Martins, I.J.; Sainsbury, A.J.; Mamo, J.C.L.; Redgrave, T.G. Lipid and apolipoprotein B48 transport in mesenteric lymph and the effect of hyperphagia on chylomicron clearance in insulin-deficient rats. Diabetologia 1994, 37, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Mason, T.M. The role of factors that regulate the synthesis and secretion of very-low-density lipoprotein by hepatocytes. Crit. Rev. Clin. Lab. Sci. 1998, 35, 461–487. [Google Scholar] [CrossRef] [PubMed]
- Apolipoproteins: the Apoproteins in Lipoproteins 2008. Available online: https://biochemistryquestions.wordpress.com (accessed on 17 November 2015).
- Shaw, I.; Vanoort, R. Food Chemical Safety; Watson, D.H., Ed.; CRC Press: Leiden, The Netherlands; Woodhead Publishing Limited: Cambridge, UK, 2001; Chapter 10 Mycotoxins; Volume 1 Contaminants, pp. 239–245. [Google Scholar]
- Zain, Z.E. Impact of mycotoxins on humans and animals. J. Saudi Chem. Soc. 2011, 15, 129–144. [Google Scholar] [CrossRef]
- Plumlee, K.H.; Galey, F.D. Neurotoxic mycotoxins: A review of fungal toxins that cause neurological disease in large animals. J. Vet. Intern. Med. 1994, 8, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F.; Mezes, M.; Vladimir, I.; Fisinin, V.I.; Fotina, T.I. Effects of mycotoxins on animal health: From oxidative stress to gene expression. Available online: www.kgzs-ms.si/slike/ZED08/06Surai.pdf (accessed on 27 August 2015).
- Ikegwuonu, F.I. The neurotoxicity of aflatoxin B1 in the rat. Toxicology 1983, 28, 247–259. [Google Scholar] [CrossRef]
- Roze, L.V.; Hong, S.-Y.; Linz, J.E. Aflatoxin biosynthesis: Current frontiers. Annu. Rev. Food Sci. Technol. 2013, 4, 293–311. [Google Scholar] [CrossRef] [PubMed]
- Doi, K.; Uetsuka, K. Mechanisms of mycotoxin-induced neurotoxicity through oxidative stress-associated pathways. Int. J. Mol. Sci. 2011, 12, 5213–5237. [Google Scholar] [CrossRef] [PubMed]
- Gentry, P.A.; Ross, M.L.; Bondy, G.S. Inhibitory effect of trichothecene mycotoxins on bovine platelets stimulated by platelet activating factor. Can. J. Vet. Res. 1987, 51, 490–494. [Google Scholar] [PubMed]
- Grandoni, K.; Gentry, P.; Holub, B.; Yagen, B. Comparative effects of trichothecene mycotoxins on bovine platelet function: Acetyl T-2 toxin, a more potent inhibitor than T-2 toxin. Mycotoxin Res. 1990, 6, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, P.; Upadhyay, A.; Agnihotri, A.; Karmakar, S.; Ghoyary, D.; Veer, V. Hematoxicity of fusirium mycotoxin in experimental Sprague-Dawley rats. Toxicol. Int. 2013, 20, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Ostry, V.; Malir, F.; Ruprich, J. Producers and important dietary sources of ochratoxin A and citrinin. Toxins 2013, 5, 1574–1586. [Google Scholar] [CrossRef] [PubMed]
- Paradells, S.; Rocamonde, B.; Llinares, C.; Herranz-Pérez, V.; Jimenez, M.; Garcia-Verdugo, J.M.; Zipancic, I.; Soria, J.M.; Garcia-Esparza, M.A. Neurotoxic effects of ochratoxin-A on the subventricular zone of adult mouse brain. J. Appl. Toxicol. 2015, 35, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Sava, V.; Reunova, O.; Velasquez, A.; Sanchez-Ramos, J. Can low level exposure to ochratoxin-A cause Parkinsonism? J. Neurol. Sci. 2006, 249, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Dias, I.H.; Polidori, M.C.; Griffiths, H.R. Hypercholesterolaemia-induced oxidative stress at the blood-brain barrier. Biochem. Soc. Trans. 2014, 42, 1001–1005. [Google Scholar] [CrossRef] [PubMed]
- Kalayci, R.; Kaya, M.; Uzun, H.; Bilgic, B.; Ahishali, B.; Arican, N.; Elmas, I.; Küçük, M. Influence of hypercholesterolemia and hypertension on the integrity of the blood-brain barrier in rats. Int. J. Neurosci. 2009, 119, 1881–904. [Google Scholar] [CrossRef] [PubMed]
- Tesoriere, L.; Attanzio, A.; Allegra, M.; Cilla, A.; Gentile, C.; Livrea, M.A. Oxysterol mixture in hypercholesterolemia-relevant proportion causes oxidative stress-dependent eryptosis. Cell. Physiol. Biochem. 2014, 34, 1075–1089. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Kim, B.-H.; Seo, H.S.; Lee, Y.J.; Kim, H.H.; Son, H.-H.; Choi, M.H. Cholesterol-induced non-alcoholic fatty liver disease and atherosclerosis aggravated by systemic inflammation. PLoS ONE 2014, 9, e9784. [Google Scholar] [CrossRef] [PubMed]
- Björkhem, I.; Heverin, M.; Leoni, V.; Meaney, S.; Diczfalusy, U. Oxysterols and Alzheimer’s disease. Acta Neurol. Scand. Suppl. 2006, 185, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Poli, G.; Biasi, F.; Leonarduzzi, G. Oxysterols in the pathogenesis of major chronic diseases. Redox Biol. 2013, 1, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Eckel, R.H. What are lipoproteins doing in the brain? Trends Endocrinol. Metab. 2014, 25, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Wolozin, B.; Brown, J., 3rd; Theisler, C.; Silberman, S. The cellular biochemistry of cholesterol and statins: Insights into the pathophysiology and therapy of Alzheimer’s disease. CNS Drug Rev. 2004, 10, 127–146. [Google Scholar] [CrossRef] [PubMed]
- Papassotiropoulos, A.; Lütjohann, D.; Bagli, M.; Locatelli, S.; Jessen, F.; Buschfort, R.; Ptok, U.; Björkhem, I.; von Bergmann, K.; Heun, R. 24S-hydroxycholesterol in cerebrospinal fluid is elevated in early stages of dementia. J. Psychiatr. Res. 2002, 36, 27–32. [Google Scholar] [CrossRef]
- Tucsek, Z.; Toth, P.; Sosnowska, D.; Gautam, T.; Mitschelen, M.; Koller, A.; Szalai, G.; Sonntag, W.E.; Ungvari, Z.; Csiszar, A. Obesity in aging exacerbates blood-brain barrier disruption, neuroinflammation, and oxidative stress in the mouse hippocampus: Effects on expression of genes involved in beta-amyloid generation and Alzheimer’s disease. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 1212–1226. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Guo, M.; Su, J.; Lu, B.; Ma, D.; Zhang, R.; Yang, L.; Wang, Q.; Ma, Y.; Fan, Y. Simvastatin blocks blood-brain barrier disruptions induced by elevated cholesterol both in vivo and in vitro. Int. J. Alzheimer’s Dis. 2012. [Google Scholar] [CrossRef] [PubMed]
- Goldwaser, E.L.; Acharya, N.K.; Nagele, R.G. Cerebrovascular and blood-brain barrier compromise: A mechanistic link between vascular disease and Alzheimer’s disease subtypes of neurocognitive disorders. J. Parkinsons Dis. Alzheimer’s Dis. 2015, 2, 10. [Google Scholar]
- Acharya, N.K.; Levin, E.C.; Clifford, P.M.; Han, M.; Tourtellotte, R.; Chamberlain, D.; Pollaro, M.; Coretti, N.J.; Kosciuk, M.C.; Nagele, E.P.; et al. Diabetes and hypercholesterolemia increase blood-brain barrier permeability and brain amyloid deposition: Beneficial effects of the LpPLA2 inhibitor darapladib. J. Alzheimer’s Dis. 2013, 35, 179–198. [Google Scholar]
- Campbell, A.W.; Thrasher, J.D.; Gray, M.R.; Vojdani, A. Mold and mycotoxins: Effects on the neurological and immune systems in humans. Adv. Appl. Microbiol. 2004, 55, 375–406. [Google Scholar] [PubMed]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin deoxynivalenol. Toxins 2013, 5, 784–820. [Google Scholar] [CrossRef] [PubMed]
- Weidner, M.; Hüwel, S.; Ebert, F.; Schwerdtle, T.; Galla, H.-J.; Humpf, H.-U. Influence of T-2 and HT-2 toxin on the blood-brain barrier in vitro: New experimental hints for neurotoxic effects. PLoS ONE 2013, 8, e60484. [Google Scholar] [CrossRef] [PubMed]
- Jangula, A.; Murphy, E.J. Lipopolysaccharide-induced blood brain barrier permeability is enhanced by alpha-synuclein expression. Neurosci. Lett. 2013, 551, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Xaio, H.; Banks, W.A.; Nieholf, M.L.; Morley, J.E. Effect of LPS on the permeability of the blood-brain barrier to insulin. Brain Res. 2001, 896, 36–42. [Google Scholar] [CrossRef]
- Qin, L.; Wu, X.; Block, M.L.; Liu, Y.; Breese, G.R.; Hong, J.S.; Knapp, D.J.; Crews, F.T. Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 2007, 55, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Lee, Y.K.; Yuk, D.Y.; Choi, D.Y.; Ban, S.B.; Oh, K.W.; Hong, J.T. Neuro-inflammation induced by lipopolysaccharide causes cognitive impairment through enhancement of β-amyloid generation. J. Neuroinflamm. 2008, 5. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Liu, T.; Rose, J.L.; Stevens, R.L.; Hoyt, D.G. Sensitivity of mice to lipopolysaccharide is increased by a high saturated fat and cholesterol diet. J. Inflamm. 2007, 4. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-A.; Gu, W.; Lee, I.-A.; Joh, E.-H.; Kim, D.-H. High fat diet-induced gut microbiota exacerbates inflammation and obesity in mice via the TLR4 signaling pathway. PLoS ONE 2012, 7, e47713. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y. The Effect of High-fat diet-induced pathophysiological changes in the gut on obesity: What should be the ideal treatment? Clin. Transl. Gastroenterol. 2013, 4, e39. [Google Scholar] [CrossRef] [PubMed]
- Amar, J.; Burcelin, R.; Ruidavets, J.B.; Cani, P.D.; Fauvel, J.; Alessi, M.C.; Chamontin, B.; Ferriéres, J. Energy intake is associated with endotoxemia in apparently healthy men. Am. J. Clin. Nutr. 2008, 87, 1219–1223. [Google Scholar] [PubMed]
- Fenton, M.J.; Golenbock, D.T. LPS-binding proteins and receptors. J. Leukoc. Biol. 1998, 64, 25–32. [Google Scholar] [PubMed]
- Ciesielski, F.; Davis, B.; Rittig, M.; Bonev, B.B.; O’Shea, P. Receptor-independent interaction of bacterial lipopolysaccharide with lipid and lymphocyte membranes; the role of cholesterol. PLoS ONE 2012, 7, e38677. [Google Scholar] [CrossRef] [PubMed]
- Feingold, K.R.; Staprans, I.; Memon, R.A.; Moser, A.H.; Shigenaga, J.K.; Doerrler, W. Endotoxin rapidly induces changes in lipid metabolism that produce hypertriglyceridemia: Low doses stimulate hepatic triglyceride production while high doses inhibit clearance. J. Lipid Res. 1992, 33, 1765–1776. [Google Scholar] [PubMed]
- Miele, L.; Marrone, G.; Lauritano, C.; Cefalo, C.; Gasbarrini, A.; Day, C. Gut-liver axis and microbiota in NAFLD: Insight pathophysiology for novel therapeutic target. Curr. Pharm. Des. 2013, 19, 5314–5324. [Google Scholar] [CrossRef] [PubMed]
- Le Roy, T.; Llopis, M.; Lepage, P.; Bruneau, A.; Rabot, S.; Bevilacqua, C. Intestinal microbiota determines development of non-alcoholic fatty liver disease in mice. Gut 2013, 62, 1787–1794. [Google Scholar]
- Alisi, A.; Ceccarelli, S.; Panera, N.; Nobili, V. Causative role of gut microbiota in non-alcoholic fatty liver disease pathogenesis. Front. Cell. Infect. Microbiol. 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- Duseja, A.; Chawla, Y.K. Obesity and NAFLD: The role of bacteria and microbiota. Clin. Liver Dis. 2014, 18, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. LPS Regulates apolipoprotein E and Aβ Interactions with effects on acute phase proteins and amyloidosis. Adv. Aging Res. 2015, 4, 69–77. [Google Scholar] [CrossRef]
- Martins, I.J. Unhealthy diets determine benign or toxic amyloid beta states and promote brain amyloid beta aggregation. Austin J. Clin. Neurol. 2015, 2, 1060–1066. [Google Scholar]
- Martins, I.J. Diabetes and cholesterol dyshomeostasis involve abnormal α-synuclein and amyloid beta transport in neurodegenerative diseases. Austin Alzheimer’s J. Parkinsons Dis. 2015, 2, 1020–1028. [Google Scholar]
- Deane, R.; Bell, R.D.; Sagare, A.; Zlokovic, B.V. Clearance of amyloid-beta peptide across the blood-brain barrier: Implication for therapies in Alzheimer’s disease. CNS Neurol. Disord. Drug Targets 2009, 8, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Castellano, J.M.; Deane, R.; Gottesdiener, A.J.; Verghese, P.B.; Stewart, F.R.; West, T.; Paoletti, A.C.; Kasper, T.R.; DeMattos, R.B.; Zlokovic, B.V.; et al. Low-density lipoprotein receptor overexpression enhances the rate of brain-to-blood Aβ clearance in a mouse model of β-amyloidosis. Proc. Natl. Acad. Sci. USA 2012, 109, 15502–15507. [Google Scholar] [CrossRef] [PubMed]
- Basak, J.M.; Verghese, P.B.; Yoon, H.; Kim, J.; Holtzman, D.M. Low-density lipoprotein receptor represents an apolipoprotein E-independent pathway of Aβ uptake and degradation by astrocytes. J. Biol. Chem. 2012, 287, 13959–13971. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.T.; Goeger, D.E.; Hinton, D.M. Microbial Toxins in Foods and Feeds; Chapter Mycotoxin-Induced Alterations in Ion Transport across Cell Membranes; Pohland, A.E., Ed.; Plenum Press: New York, NY, USA, 1990. [Google Scholar]
- Kaplan, R.; Gan, X.; Menke, J.G.; Wright, S.D.; Cai, T.Q. Bacterial lipopolysaccharide induces expression of ABCA1 but not ABCG1 via an LXR-independent pathway. J. Lipid Res. 2002, 43, 952–959. [Google Scholar] [PubMed]
- Martin, S.; Parton, R.G. Caveolin, cholesterol, and lipid bodies. Semin. Cell Dev. Biol. 2005, 16, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Frank, P.G.; Cheung, M.W.; Pavlides, S.; Llaverias, G.; Park, D.S.; Lisanti, M.P. Caveolin-1 and regulation of cellular cholesterol homeostasis. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H677–H686. [Google Scholar] [CrossRef] [PubMed]
- Fantini, J.; Barrantes, F.J. How cholesterol interacts with membrane proteins: An exploration of cholesterol-binding sites including CRAC, CARC, and tilted domains. Front. Physiol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Hulce, J.J.; Cognetta, A.B.; Niphakis, M.J.; Tully, S.E.; Cravatt, B.F. Extracting intracellular diffusive states and transition rates from single-molecule tracking data Proteome-wide mapping of cholesterol-interacting proteins in mammalian cells. Nat. Methods 2013, 10, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.M. Review cholesterol and the interaction of proteins with membrane domains. Prog. Lipid Res. 2006, 45, 279–294. [Google Scholar] [CrossRef] [PubMed]
- Gimpl, G. Cholesterol-protein interaction: Methods and cholesterol reporter molecules. Subcell. Biochem. 2010, 51, 1–45. [Google Scholar] [PubMed]
- Parton, R.G.; Simons, K. The multiple faces of caveolae. Nat. Rev. Mol. Cell Biol. 2007, 8, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Parton, R.G. Caveolae Meet Endosomes: A Stable Relationship? Dev. Cell 2004, 7, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Mundy, D.I.; Li, W.P.; Luby-Phelps, K.; Anderson, R.G. Caveolin targeting to late endosome/lysosomal membranes is induced by perturbations of lysosomal pH and cholesterol content. Mol. Biol. Cell 2012, 23, 864–880. [Google Scholar] [CrossRef] [PubMed]
- Frank, P.G.; Galbiati, F.; Volonte, D.; Razani, B.; Cohen, D.E.; Marcel, Y.L.; Lisanti, M.P. Influence of caveolin-1 on cellular cholesterol efflux mediated by high-density lipoproteins. Am. J. Physiol. Cell Physiol. 2001, 280, C1204–C1214. [Google Scholar] [PubMed]
- Matveev, S.; Uittenbogaard, A.; van der Westhuyzen, D.; Smart, E.J. Caveolin-1 negatively regulates SR-BI mediated selective uptake of high-density lipoprotein-derived cholesteryl ester. Eur. J. Biochem. 2001, 268, 5609–5616. [Google Scholar] [CrossRef] [PubMed]
- Hailstones, D.; Sleer, L.S.; Parton, R.G.; Stanley, K.K. Regulation of caveolin and caveolae by cholesterol in MDCK cells. J. Lipid Res. 1998, 39, 369–379. [Google Scholar] [PubMed]
- Lei, M.G.; Tan, X.; Qureshi, N.; Morrison, D.C. Regulation of cellular caveolin-1 protein expression in murine macrophages by microbial products. Infect. Immun. 2005, 73, 8136–8143. [Google Scholar] [CrossRef] [PubMed]
- Medina, F.A.; de Almeida, C.J.; Dew, E.; Li, J.-W.; Bonuccelli, G.; Williams, T.M.; Cohen, A.W.; Pestell, R.G.; Frank, P.J.; Tanowitz, H.B.; et al. Caveolin-1-deficient mice show defects in innate immunity and inflammatory immune response during salmonella enterica serovar typhimurium infection. Infect. Immun. 2006, 74, 6665–6674. [Google Scholar] [CrossRef] [PubMed]
- Henning, M.F.; Garda, H.A.; Bakas, L. Biophysical characterization of interaction between apolipoprotein A-I and bacterial lipopolysaccharide. Cell. Biochem. Biophys. 2006, 44, 490–496. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, X.; Wu, G.; Shen, L.; Chen, B. Effect of lipid-bound apoA-I cysteine mutants on lipopolysaccharide-induced endotoxemia in mice. J. Lipid Res. 2008, 49, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Gupta, H.; Dai, L.; Datta, G.; Garber, D.W.; Grenett, H.; Li, Y.; Mishra, V.; Palgunachari, M.N.; Handattu, S.; Gianturco, S.H.; et al. Inhibition of lipopolysaccharide-induced inflammatory responses by an apolipoprotein AI mimetic peptide. Circ. Res. 2005, 97, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Silver, D.L.; Thiele, C.; Tall, A.R. ATP-binding cassette transporter A1 (ABCA1) functions as a cholesterol efflux regulatory protein. J. Biol. Chem. 2001, 276, 23742–23747. [Google Scholar] [CrossRef] [PubMed]
- Burns, M.P.; Vardanian, L.; Pajoohesh-Ganji, A.; Wang, L.; Cooper, M.; Harris, D.C.; Duff, K.; Rebeck, G.W. The effects of ABCA1 on cholesterol efflux and Aβ levels in vitro and in vivo. J. Neurochem. 2006, 98, 792–800. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, D.; Trischuk, T.C.; Chan, T.; Drover, V.A.B.; Ho, S.; Chimini, G.; Agellon, L.B.; Agnihotri, R.; Francis, A.; Lehner, R. ABCA1-dependent lipid efflux to apolipoprotein A-I mediates HDL particle formation and decreases VLDL secretion from murine hepatocytes. J. Lipid Res. 2004, 45, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, D.A.; Castegna, A.; Lauderback, C.M.; Drake, J. Evidence that amyloid beta-peptide-induced lipid peroxidation and its sequelae in Alzheimer’s disease brain contribute to neuronal death. Neurobiol. Aging 2002, 23, 655–664. [Google Scholar] [CrossRef]
- Oates, J.; Watts, A. Uncovering the intimate relationship between lipids, cholesterol and GPCR activation. Curr. Opin. Struct. Biol. 2011, 21, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Paila, Y.D.; Chattopadhyay, A. Membrane cholesterol in the function and organization of G-protein coupled receptors. Subcell. Biochem. 2010, 51, 439–466. [Google Scholar] [PubMed]
- Sengupta, D.; Chattopadhyay, A. Molecular dynamics simulations of GPCR-cholesterol interaction: An emerging paradigm. Biochim. Biophys. Acta 2015, 1848, 1775–1782. [Google Scholar] [CrossRef] [PubMed]
- Thathiah, A.; de Strooper, B. G protein—Coupled receptors, cholinergic dysfunction, and Aβ toxicity in Alzheimer’s disease. Sci. Signal. 2009, 2. [Google Scholar] [CrossRef] [PubMed]
- Thathiah, A.; de Strooper, B. The role of G protein-coupled receptors in the pathology of Alzheimer’s disease. Nat. Rev. Neurosci. 2011, 12, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Chini, B.; Parenti, M. G-protein coupled receptors in lipid rafts and caveolae: How, when and why do they go there? J. Mol. Endocrinol. 2004, 32, 325–338. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Ma, C.; Hsu, W.C.; Lo, H.F.; Yang, V.C. Molecular interaction between caveolin-1 and ABCA1 on high-density lipoprotein-mediated cholesterol efflux in aortic endothelial cells. Cardiovasc. Res. 2007, 75, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.W.; Combs, T.P.; Scherer, P.E.; Lisanti, M.P. Role of caveolin and caveolae in insulin signaling and diabetes. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E1151–E1160. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Toya, Y.; Schwencke, C.; Lisanti, M.P.; Myers, M.G.; Ishikawa, Y. Caveolin is an activator of insulin receptor signaling. J. Biol. Chem. 1998, 273, 26962–26968. [Google Scholar] [CrossRef] [PubMed]
- Nystrom, F.H.; Chen, H.; Cong, L.N.; Li, Y.; Quon, M.J. Caveolin-1 interacts with the insulin receptor and can differentially modulate insulin signaling in transfected Cos-7 cells and rat adipose cells. Mol. Endocrinol. 1999, 13, 2013–2024. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.W.; Razani, B.; Wang, X.B.; Combs, T.P.; Williams, T.M.; Scherer, P.E.; Lisanti, M.P. Caveolin-1-deficient mice show insulin resistance and defective insulin receptor protein expression in adipose tissue. Am. J. Physiol. Cell Physiol. 2003, 285, C222–C235. [Google Scholar] [CrossRef] [PubMed]
- Head, B.P.; Peart, J.N.; Panneerselvam, M.; Yokoyama, T.; Pearn, M.L.; Niesman, I.R.; Bonds, J.A.; Schilling, J.M.; Miyanohara, A.; Headrick, J.; et al. Loss of caveolin-1 accelerates neurodegeneration and aging. PLoS ONE 2010, 5, e15697. [Google Scholar] [CrossRef] [PubMed]
- Van Helmond, Z.K.; Miners, J.S.; Bednall, E.; Chalmers, K.A.; Zhang, Y.; Wilcock, G.K.; Love, S.; Khoe, P.G. Caveolin-1 and -2 and their relationship to cerebral amyloid angiopathy in Alzheimer’s disease. Neuropathol. Appl. Neurobiol. 2007, 33, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Gaudreault, S.B.; Dea, D.; Poirier, J. Increased caveolin-1 expression in Alzheimer’s disease brain. Neurobiol. Aging 2004, 25, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J.; Gupta, V.; Wilson, A.C.; Fuller, S.J.; Martins, R.N. Interactions between apo E and amyloid beta and their relationship to nutriproteomics and neurodegeneration. Curr. Proteom. 2014, 11, 173–183. [Google Scholar]
- Haziot, A.; Lin, X.Y.; Zhang, F.; Goert, S.M. Cutting edge: The induction of acute phase proteins by lipopolysaccharide uses a novel pathway that is CD14-independent. J. Immunol. 1998, 160, 2570–2572. [Google Scholar] [PubMed]
- Liu, M.; Bing, G. Lipopolysaccharide animal models for Parkinson’s disease. Parkinson’s Dis. 2011, 2011, 327089. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, P.; Herrmann, M.; Klafki, H.W.; Smirnov, A.; Lewczuk, P.; Kornhuber, J. Phagocytosis and LPS alter the maturation state of β-amyloid precursor protein and induce different Aβ peptide release signatures in human mononuclear phagocytes. J. Neuroinflamm. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- Bate, C.; Veerhuis, R.; Eikelenboom, P.; Williams, A. Microglia kill amyloid-β1-42 damaged neurons by a CD14-dependent process. Neuroreport 2004, 15, 1427–1430. [Google Scholar] [CrossRef] [PubMed]
- Erickson, M.A.; Hartvigson, P.E.; Morofuji, Y.; Owen, J.B.; Butterfield, D.A.; Banks, W.A. Lipopolysaccharide impairs amyloid β efflux from brain: Altered vascular sequestration, cerebrospinal fluid reabsorption, peripheral clearance and transporter function at the blood-brain barrier. J. Neuroinflamm. 2012, 9. [Google Scholar] [CrossRef]
- Murray, C.L.; Skelly, D.T.; Cunningham, C. Exacerbation of CNS inflammation and neurodegeneration by systemic LPS treatment is independent of circulating IL-1β and IL-6. J. Neuroinflamm. 2011, 8. [Google Scholar] [CrossRef] [PubMed]
- Waché, Y.J.; Hbabi-Haddioui, L.; Guzylack-Piriou, L.; Belkhelfa, H.; Roques, C.; Oswald, I.P. The mycotoxin deoxynivalenol inhibits the cell surface expression of activation markers in human macrophages. Toxicology 2009, 262, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Garwood, C.J.; Pooler, A.M.; Atherton, J.; Hanger, D.P.; Noble, W. Astrocytes are important mediators of Aβ-inducedneurotoxicity and tau phosphorylation in primary culture. Cell Death Dis. 2011, 202, e167. [Google Scholar] [CrossRef] [PubMed]
- Nagele, R.G.; D’Andrea, M.R.; Lee, H.; Venkataraman, V.; Wang, H.Y. Astrocytes accumulate Aβ 42 and give rise to astrocytic amyloid plaques in Alzheimer disease brains. Brain Res. 2003, 971, 197–209. [Google Scholar] [CrossRef]
- Wyss-Coray, T.; Loike, J.D.; Brionne, T.C.; Lu, E.; Anankov, R.; Yan, F.; Silverstein, S.C.; Husemann, J. Adult mouse astrocytes degrade amyloid-β in vitro and in situ. Nat. Med. 2003, 9, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Canepa, E.; Borghi, R.; Viña, J.; Traverso, N.; Gambini, J.; Domenicotti, C.; Marinari, U.M.; Poli, G.; Pronzato, M.A.; Ricciarelli, R. Cholesterol and amyloid-β: Evidence for a cross-talk between astrocytes and neuronal cells. J. Alzheimer’s Dis. 2011, 25, 645–653. [Google Scholar] [CrossRef]
- Danik, M.; Champagne, D.; Petit-Turcotte, C.; Beffert, U.; Poirier, J. Brain lipoprotein metabolism and its relation to neurodegenerative disease. Crit. Rev. Neurobiol. 1999, 13, 357–407. [Google Scholar] [PubMed]
- Jänis, M.T.; Metso, J.; Lankinen, H.; Strandin, T.; Olkkonen, V.M.; Rye, K.A.; Jauhiainen, M.; Ehnholm, C. Apolipoprotein E activates the low-activity form of human phospholipid transfer protein. Biochem. Biophys. Res. Commun. 2005, 331, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Oram, J.F.; Wolfbauer, G.; Tang, C.; Davidson, W.; Albers, J.J. An amphipathic helical region of the N-terminal barrel of phospholipid transfer protein is critical for ABCA1-dependent cholesterol efflux. J. Biol. Chem. 2008, 283, 11541–11549. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.F.L.; Martins, I.J.; Martins, R.N. Lipid metabolism and lipidomics: An emerging frontier in biology. J. Genet. Genom. 2014, 41, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Desrumaux, C.; Pisoni, A.; Meunier, J.; Deckert, V.; Athias, A.; Perrier, V.; Villard, V.; Lagrost, L.; Verdier, J.M.; Maurice, T. Increased amyloid-β peptide-induced memory deficits in phospholipid transfer protein (PLTP) gene knockout mice. Neuropsychopharmacology 2013, 38, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Lei, H.; Fan, Z.; Zheng, W.; Zheng, S. Difference in LDL receptor feedback regulation in macrophages and vascular smooth muscle cells: Foam cell transformation under inflammatory stress. Inflammation 2014, 37, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.; Urbanc, B.; Cruz, L.; Bitan, G.; Teplow, D.B.; Stanley, H.E. Role of electrostatic interactions in amyloid β-protein (Aβ) oligomer formation: A discrete molecular dynamics study. Biophys. J. 2007, 92, 4064–4077. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.; Mezes, M.; Fotina, T.; Denev, S.D. Modern Dietary Fat Intakes in Disease Promotion; De Meester, F., Zibadi, S., Watson, R.R., Eds.; Medical: New York, NY, USA, 2010; Chapter Mycotoxin in Human Diet: A Hidden Danger; pp. 275–375. [Google Scholar]
- Burghardt, R.C.; Barhoumi, R.; Lewis, E.H.; Bailey, R.H.; Pyle, K.A.; Clement, B.A.; Phillips, T.D. Patulin-induced cellular toxicity: A vital fluorescence study. Toxicol. Appl. Pharmacol. 1992, 112, 235–244. [Google Scholar] [CrossRef]
- Riley, R.T.; Showker, J.L. The mechanism of patulin’s cytotoxicity and the antioxidant activity of indole tetramic acids. Toxicol. Appl. Pharmacol. 1991, 109, 108–126. [Google Scholar] [CrossRef]
- Horváth, E.; Papp, G.; Belágyi, J.; Gazdag, Z.; Vágvölgyi, C.; Pesti, M. In vivo direct patulin-induced fluidization of the plasma membrane of fission yeast Schizosaccharomyces pombe. Food Chem. Toxicol. 2010, 48, 1898–1904. [Google Scholar] [CrossRef] [PubMed]
- Riley, R.T.; Hinton, D.M.; Showker, J.L.; Rigsby, W.; Norred, W.P. Chronology of patulin-induced alterations in membrane function of cultured renal cells, LLC-PK. Toxicol. Appl. Pharmacol. 1990, 102, 128–141. [Google Scholar] [CrossRef]
- Ziprin, R.L.; Holt, P.S.; Mortensen, R.F. T-2 toxin effects on the serum amyloid P-component (SAP) response of Listeria monocytogenes- and Salmonella typhimurium-infected mice. Toxicol. Lett. 1987, 39, 177–184. [Google Scholar] [CrossRef]
- Di Giuseppe, R.; Bertuzzi, T.; Rossi, F.; Rastelli, S.; Mulazzi, A.; Capraro, J.; de Curtis, A.; Iacoviello, L.; Pietri, A. Plasma ochratoxin A levels, food consumption, and risk biomarkers of a representative sample of men and women from the Molise region in Italy. Eur. J. Nutr. 2012, 51, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Bi, B.T.; Lin, H.B.; Cheng, Y.F.; Zhou, H.; Lin, T.; Zhang, M.Z.; Li, T.J.; Xu, J.P. Promotion of β-amyloid production by C-reactive protein and its implications in the early pathogenesis of Alzheimer’s disease. Neurochem. Int. 2012, 60, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, V.K.; Sakthisekaran, D. Effect of patulin on albumin fraction of plasma proteins studied in rats. Biochem. Int. 1991, 25, 461–475. [Google Scholar] [PubMed]
- Salsbury, E.R.; Poole, L.B.; Fetrow, J.S. Electrostatics of cysteine residues in proteins: Parameterization and validation of a simple model. Proteins 2012, 80, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Van Geest, M.; Lolkema, J.S. Membrane topology and insertion of membrane proteins: Search for topogenic signals. Microbiol. Mol. Biol. Rev. 2000, 64, 13–33. [Google Scholar] [CrossRef] [PubMed]
- Hozoji, M.; Kimura, Y.; Kioka, N.; Ueda, K. Formation of two intramolecular disulfide bonds is necessary for apoA-I-dependent cholesterol efflux mediated by ABCA1. J. Biol. Chem. 2009, 284, 11293–11300. [Google Scholar] [CrossRef] [PubMed]
- Nagao, K.; Tomioka, M.; Ueda, K. Function and regulation of ABCA1—Membrane meso-domain organization and reorganization. FEBS J. 2011, 278, 3190–3203. [Google Scholar] [CrossRef] [PubMed]
- Qu, S.J.; Fan, H.Z.; Kilinc, C.; Pownall, H.J. Role of cysteine residues in human plasma phospholipid transfer protein. J. Protein Chem. 1999, 18, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Dietzen, D.J.; Hastings, W.R.; Lublin, D.M. Caveolin is palmitoylated on multiple cysteine residues. Palmitoylation is not necessary for localization of caveolin to caveolae. J. Biol. Chem. 1995, 270, 6838–6842. [Google Scholar] [PubMed]
- Rubenstein, L.A.; Lanzarab, R.G. Activation of G protein-coupled receptors entails cysteine modulation of agonist binding. J. Mol. Struct.: THEOCHEM 1998, 430, 57–71. [Google Scholar] [CrossRef]
- Olivella, M.; Caltabiano, G.; Cordomí, A. The role of Cysteine 6.47 in class A GPCRs. BMC Struct. Biol. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, K.; Sasaki, J.; Takada, Y.; Matsunaga, A.; Fukui, J.; Albers, J.J.; Arakawa, K. A cysteine-containing truncated apo A-I variant associated with HDL deficiency. Arterioscler. Thromb. Vasc. Biol. 1996, 16, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Oda, M.N.; Bielicki, J.K.; Berger, T.; Forte, T.M. Cysteine substitutions in apolipoprotein A-I primary structure modulate paraoxonase activity. Biochemistry 2001, 40, 1710–1718. [Google Scholar] [CrossRef] [PubMed]
- Daly, N.L.; Scanlon, M.J.; Djordjevic, J.T.; Kroon, P.A.; Smith, R. Three-dimensional structure of a cysteine-rich repeat from the low-density lipoprotein receptor. Proc. Natl. Acad. Sci. USA 1995, 92, 6334–6338. [Google Scholar] [CrossRef] [PubMed]
- Lillis, A.P.; van Duyn, L.B.; Murphy-Ullrich, J.-E.; Strickland, D.K. The low density lipoprotein receptor-related protein 1: Unique tissue-specific functions revealed by selective gene knockout studies. Physiol. Rev. 2008, 88, 887–918. [Google Scholar] [CrossRef] [PubMed]
- Ivaturi, S.; Wooten, C.J.; Nguyen, M.D.; Ness, G.C.; Lopez, D. Distribution of the LDL receptor within clathrin-coated pits and caveolae in rat and human liver. Biochem. Biophys. Res. Commun. 2014, 445, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Alves, I.; Oliveira, N.G.; Laires, A.; Rodrigues, A.S.; Rueff, J. Induction of micronuclei and chromosomal aberrations by the mycotoxin patulin in mammalian cells: Role of ascorbic acid as a modulator of patulin clastogenicity. Mutagenesis 2000, 15, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Song, E.; Su, C.; Fu, J.; Xia, X.; Yang, S.; Xiao, C.; Lu, B.; Chen, H.; Sun, Z.; Wu, S.; et al. Selenium supplementation shows protective effects against patulin-induced brain damage in mice via increases in GSH-related enzyme activity and expression. Life Sci. 2014, 109, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Fliege, R.I.; Metzler, M. The mycotoxin patulin induces intra- and inter-molecular protein crosslinks in vitro involving cysteine, lysine, and histidine side chains, and alpha-amino groups. Chem. Biol. Interact. 1999, 123, 85–103. [Google Scholar] [CrossRef]
- Takai, E.; Uda, K.; Yoshida, T.; Zako, T.; Maeda, M.; Shiraki, K. Cysteine inhibits the fibrillisation and cytotoxicity of amyloid-β 40 and 42: Implications for the contribution of the thiophilic interaction. Phys. Chem. Chem. Phys. 2014, 16, 3566–3572. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.-M.; Du, Q.-S.; Meng, J.-Z.; Pang, Z.-W.; Huang, R.-B. The multiple roles of histidine in protein interactions. Chem. Cent. J. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.V.; Gendrault, J.L.; Wolff, C.M. Poly-l-lysine dissolves fibrillar aggregation of the Alzheimer β-amyloid peptide in vitro. Biochem. Biophys. Res. Commun. 2002, 291, 764–768. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Lopes, D.H.; Bitan, G. A key role for lysine residues in amyloid β-protein folding, assembly, and toxicity. ACS Chem. Neurosci. 2012, 3, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.G.; Ciccotosto, G.D.; Tew, D.J.; Perez, K.; Curtain, C.C.; Boas, J.F.; Masters, C.L.; Cappai, R.; Barnham, K.J. Histidine 14 modulates membrane binding and neurotoxicity of the Alzheimer’s disease amyloid-beta peptide. J. Alzheimer’s Dis. 2010, 19, 1387–1400. [Google Scholar]
- Herskovits, A.Z.; Guarente, L. SIRT1 in Neurodevelopment and Brain Senescence. Neuron 2014, 8, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.L.; Chua, C.E.L. SIRT1 and neuronal diseases. Mol. Asp. Med. 2008, 29, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Guarente, L. Sirtuins in Aging and Disease. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.K.; Connolly, T.M. Nuclear receptors as drug targets in obesity, dyslipidemia and atherosclerosis. Curr. Opin. Investig. Drugs 2008, 9, 247–255. [Google Scholar] [PubMed]
- Harrison, C. Neurodegenerative disorders: A neuroprotective role for Sirtuin 1. Nat. Rev. Drug Discov. 2012, 11. [Google Scholar] [CrossRef]
- Kawada, T.; Goto, T.; Hirai, S.; Kang, M.S.; Uemura, T.; Yu, R.; Takahashi, N. Dietary regulation of nuclear receptors in obesity-related metabolic syndrome. Asia Pac. J. Clin. Nutr. 2008, 17, 126–130. [Google Scholar] [PubMed]
- Martins, I.J. Nutritional Diets Accelerate Amyloid Beta Metabolism and Prevent the Induction of Chronic Diseases and Alzheimer’s Disease, 1st ed.; Photon ebooks: Quebec, Canada, 2015; pp. 1–48. [Google Scholar]
- Zee, R.S.; Pimentel, D.R.; Hou, X.L.; Zang, M.; Yao, C.X.; O’Connor, P.B.; Bachschmid, M.M.; Cohen, R.A. Sirtuin-1 zinc thiolate center is a direct molecular target of oxidants. In Proceedings of the Society for Free Radical Biology and Medicine’s 15th Annual Meeting, Indianapolis, IN, USA, 19–23 November 2008; Published in Free Radical Biology and Medicine. Volume 45. Supplement S76.
- Mori, H.; Kawai, K.; Ohbayashi, F.; Kuniyasu, T.; Yamazaki, M.; Hamasaki, T.; Williams, G.M. Genotoxicity of a variety of mycotoxins in the hepatocyte primary culture/DNA repair test using rat and mouse hepatocytes. Cancer Res. 1984, 44, 2918–2923. [Google Scholar] [PubMed]
- Martins, I.J.; Lim, W.L.F.; Wilson, A.; Laws, S.; Martins, R.N. The acceleration of aging and Alzheimer’s disease through the biological mechanisms behind obesity and type II diabetes. Health 2013, 5, 913–920. [Google Scholar] [CrossRef]
- Dawson, V.L.; Dawson, T.M. Nitric oxide neurotoxicity. J. Chem. Neuroanat. 1996, 10, 179–190. [Google Scholar] [CrossRef]
- Calabrese, V.; Mancuso, C.; Calvani, M.; Rizzarelli, E.; Butterfield, D.A.; Stella, A.M. Nitric oxide in the central nervous system: Neuroprotection versus neurotoxicity. Nat. Rev. Neurosci. 2007, 8, 766–775. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Chen, S.H.; Kadiiska, M.B.; Hong, J.S.; Zielonka, J.; Kalyanaraman, B.; Mason, R.P. Inducible nitric oxide synthase is key to peroxynitrite-mediated, LPS-induced protein radical formation in murine microglial BV2 cells. Free Radic. Biol. Med. 2014, 73, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Yoo, B.K.; Choi, J.W.; Shin, C.-Y.; Jeon, S.J.; Park, S.J.; Cheong, J.H.; Han, S.Y.; Ryu, J.R.; Song, M.R.; Ko, K.H.; et al. Activation of p38 MAPK induced peroxynitrite generation in LPS plus IFN-γ-stimulated rat primary astrocytes via activation of iNOS and NADPH oxidase. Neurochem. Int. 2008, 52, 1188–1197. [Google Scholar] [CrossRef] [PubMed]
- Shao, D.; Fry, J.L.; Han, J.; Hou, X.; Pimentel, D.R.; Matsui, R.; Cohen, R.A.; Bachschmid, M.M. A redox-resistant sirtuin-1 mutant protects against hepatic metabolic and oxidant stress. J. Biol. Chem. 2014, 289, 7293–7306. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. Nutritional and genotoxic stress contributes to diabetes and neurodegenerative diseases such as Parkinson’s and Alzheimer’s diseases. In Frontiers in Clinical Drug Research—CNS and Neurological Disorders; Atta-ur-Rahman, Ed.; Bentham Science Publishers: Cambridge, UK, 2015; Volume 35, pp. 158–192. [Google Scholar]
- Martins, I.J. Unhealthy Nutrigenomic Diets Accelerate NAFLD and Adiposity in Global communities. J. Mol. Genet. Med. 2015, 9, 1–11. [Google Scholar]
- Bist, A.; Fielding, C.J.; Fielding, P.E. p53 regulates caveolin gene transcription, cell cholesterol, and growth by a novel mechanism. Biochemistry 2000, 39, 1966–1972. [Google Scholar] [CrossRef] [PubMed]
- Galbiati, F.; Volonte, D.; Liu, J.; Capozza, F.; Frank, P.G.; Zhu, L.; Pestell, R.G.; Lisanti, M.P. Caveolin-1 expression negatively regulates cell cycle progression by inducing G0/G1 arrest via a p53/p21WAF1/Cip1-dependent mechanism. Mol. Biol. Cell 2001, 12, 2229–2244. [Google Scholar] [CrossRef] [PubMed]
- Volonte, D.; Zou, H.; Bartholomew, J.N.; Liu, Z.; Morel, P.A.; Galbiati, F. Oxidative stress-induced inhibition of Sirt1 by caveolin-1 promotes p53-dependent premature senescence and stimulates the secretion of interleukin 6 (IL-6). J. Biol. Chem. 2015, 290, 4202–4214. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. Nutrition increases Survival and Reverses NAFLD and Alzheimers disease; Ebook Project; Jones, E., Ed.; GmbH & Co. KG.: Saarbrucken, Germany, 2015. [Google Scholar]
- Kitagawa, T.; Yokoyama, Y.; Kokuryo, T.; Nagino, M. Protective effects of l-cysteine against LPS-induced liver injuries. FASEB J. 2015, 29. [Google Scholar]
- Deng, J.; Huang, Q.; Wang, F.; Liu, Y.; Wang, Z.; Wang, Z.; Zhang, Q.; Lei, B.; Cheng, Y. The role of caveolin-1 in blood-brain barrier disruption induced by focused ultrasound combined with microbubbles. J. Mol. Neurosci. 2012, 46, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.L.; Song, J.N.; Zhang, M. Role of caveolin-1 in the biology of the blood-brain barrier. Rev. Neurosci. 2014, 25, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Madeira, A.; Yang, J.; Zhang, X.; Vikeved, E.; Nilsson, A.; Andrén, P.E.; Svenningsson, P. Caveolin-1 interacts with α-synuclein and mediates toxic actions of cellular α-synuclein overexpression. Neurochem. Int. 2011, 59, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Takenouchi, T.; Rockenstein, E.; Masliah, E. α-Synuclein up-regulates expression of caveolin-1 and down-regulates extracellular signal-regulated kinase activity in B103 neuroblastoma cells: Role in the pathogenesis of Parkinson’s disease. J. Neurochem. 2003, 85, 1468–1479. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J.; Vilchèze, C.; Mortimer, B.C.; Bittman, R.; Redgrave, T.G. Sterol side chain length and structure affect the clearance of chylomicron-like lipid emulsions in rats and mice. J. Lipid Res. 1998, 39, 302–312. [Google Scholar] [PubMed]
- Pfister, J.A.; Ma, C.; Morrison, B.E.; D’Mello, S.R. Opposing effects of sirtuins on neuronal survival: SIRT1-mediated neuroprotection is independent of its deacetylase activity. PLoS ONE 2008, 3, e4090. [Google Scholar] [CrossRef] [PubMed]
- Akama, K.T.; Albanese, C.; Pestell, R.G.; van Eldik, L.J. Amyloid β-peptide stimulates nitric oxide production in astrocytes through an NFκB-dependent mechanism. Proc. Natl. Acad. Sci. USA 1998, 95, 5795–5800. [Google Scholar] [CrossRef] [PubMed]
- Keil, U.; Bonert, A.; Marques, C.A.; Strosznajder, J.B.; Müller-Spahn, F.; Müller, W.E.; Eckert, A. Elevated nitric oxide production mediates β-amyloid-induced mitochondria failure. Pol. J. Pharmacol. 2004, 56, 631–634. [Google Scholar] [PubMed]
- Xiang, W.; Menges, S.; Schlachetzki, J.C.; Meixner, H.; Hoffmann., A.C.; Schlötzer-Schrehardt, U.; Becker, C.M.; Winkler, J.; Klucken, J. Posttranslational modification and mutation of histidine 50 trigger α synuclein aggregation and toxicity. Mol. Neurodegener. 2015, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Freed, C.R. Tyrosine-to-cysteine modification of human alpha-synuclein enhances protein aggregation and cellular toxicity. J. Biol. Chem. 2004, 279, 10128–10135. [Google Scholar] [CrossRef] [PubMed]
- Williamson, M.P.; Suzuki, Y.; Bourne, N.T.; Asakura, T. Binding of amyloid β-peptide to ganglioside micelles is dependent on histidine-13. Biochem. J. 2006, 397, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Martins, I.J. The global obesity epidemic is related to stroke, dementia and Alzheimer’s disease. JSM Alzheimer’s Dis. Relat. Dement. 2014, 1, 1010–1018. [Google Scholar]
- Martins, I.J. Induction of NAFLD with increased risk of obesity and chronic diseases in developed countries. Open J. Endocr. Metab. Dis. 2014, 4, 90–110. [Google Scholar] [CrossRef]
- Martins, I.J. Diabetes and organ dysfunction in the developing and developed. World Glob. J. Med. Res. F Dis. 2015, 15, 1–8. [Google Scholar]
- Leff, J.W.; Fierer, N. Bacterial communities associated with the surfaces of fresh fruits and vegetables. PLoS ONE 2013, 8, e59310. [Google Scholar] [CrossRef] [PubMed]
- Howel, K. Bacterial contamination of bread. Am. J. Public Health 1912, 2, 321–324. [Google Scholar] [CrossRef]
- Miura, S.; Hasumi, K.; Endo, A. Inhibition of protein prenylation by patulin. FEBS Lett. 1993, 318, 88–90. [Google Scholar] [CrossRef]
- Mohamed, A.; Saavedra, L.; di Pardo, A.; Sipione, S.; Posse de Chaves, E. β-Amyloid inhibits protein prenylation and induces cholesterol sequestration by impairing SREBP-2 cleavage. J. Neurosci. 2012, 32, 6490–6500. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, S.M.; Wilkinson, B.L.; Golde, T.E.; Landreth, G. Statins reduce amyloid-β production through inhibition of protein isoprenylation. J. Biol. Chem. 2007, 282, 26832–26844. [Google Scholar] [CrossRef] [PubMed]
- PATULIN: Food Safety Watch 2013. Available online: http://www.foodsafetywatch.org/factsheets/patulin/ (accessed on 16 September 2015).
- Prabhu, P. Analysis of the mycotoxin patulin in apple juice using the flexar FX-15 UHPLC-UV. Available online: http://www.perkinelmer.com/APP_FlexarAppleJuice.pdf (accessed on 16 September 2015).
- Ramsay Tainsh, A. Alcohol and mycotoxins. Alcohol Alcohol. 1988, 23, 180–181. [Google Scholar] [PubMed]
- Martins, I.J. Appetite Dysregulation and Obesity in Western Countries; Ebook Project; Jones, E., Ed.; GmbH & Co. KG.: Saarbrucken, Germany, 2013. [Google Scholar]
- Swick, R.A. Hepatic metabolism and bioactivation of mycotoxins and plant toxins. J. Anim. Sci. 1984, 58, 1017–1028. [Google Scholar] [PubMed]
- Martins, I.J. Increased risk for obesity and diabetes with neurodegeneration in developing countries. J. Mol. Genet. Med. 2013, S1, 1–8. [Google Scholar]
- Burgess, J.W.; Neville, T.A.; Rouillard, P.; Harder, Z.; Beanlands, D.S.; Sparks, D.L. Phosphatidylinositol increases HDL-C levels in humans. J. Lipid Res. 2005, 46, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Devaraja, S.; Girish, K.S.; Santhosh, M.S.; Hemshekhar, M.; Nayaka, S.C.; Kemparaju, K. Fusaric acid, a mycotoxin, and its influence on blood coagulation and platelet function. Blood Coagul. Fibrinolysis. 2013, 24, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Prociuk, M.A.; Edel, A.L.; Richard, M.N.; Gavel, N.T.; Ander, B.P.; Dupasquier, C.M.; Pierce, G.N. Cholesterol-induced stimulation of platelet aggregation is prevented by a hempseed-enriched diet. Can. J. Physiol. Pharmacol. 2008, 86, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Min, S.H.; Abrams, C.S. Regulation of platelet plug formation by phosphoinositide metabolism. Arch. Biochem. Biophys. 1984, 228, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Graff, G.; Nahas, N.; Nikolopoulou, M.; Natarajan, V.; Schmid, H.H. Possible regulation of phospholipase C activity in human platelets by phosphatidylinositol 4′,5′-bisphosphate. Food Chem. Toxicol. 2013, 57, 11–20. [Google Scholar] [CrossRef]
- Zundel, W.; Swiersz, L.M.; Giaccia, A. Caveolin 1-mediated regulation of receptor tyrosine kinase-associated phosphatidylinositol 3-kinase activity by ceramide. Mol. Cell. Biol. 2000, 20, 1507–1514. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, I.J. Overnutrition Determines LPS Regulation of Mycotoxin Induced Neurotoxicity in Neurodegenerative Diseases. Int. J. Mol. Sci. 2015, 16, 29554-29573. https://doi.org/10.3390/ijms161226190
Martins IJ. Overnutrition Determines LPS Regulation of Mycotoxin Induced Neurotoxicity in Neurodegenerative Diseases. International Journal of Molecular Sciences. 2015; 16(12):29554-29573. https://doi.org/10.3390/ijms161226190
Chicago/Turabian StyleMartins, Ian James. 2015. "Overnutrition Determines LPS Regulation of Mycotoxin Induced Neurotoxicity in Neurodegenerative Diseases" International Journal of Molecular Sciences 16, no. 12: 29554-29573. https://doi.org/10.3390/ijms161226190
APA StyleMartins, I. J. (2015). Overnutrition Determines LPS Regulation of Mycotoxin Induced Neurotoxicity in Neurodegenerative Diseases. International Journal of Molecular Sciences, 16(12), 29554-29573. https://doi.org/10.3390/ijms161226190




