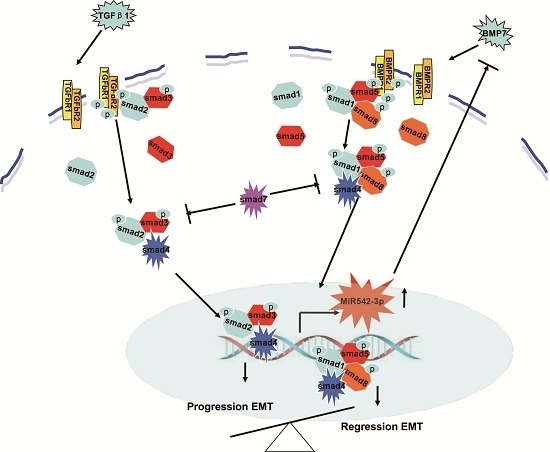
MiR542-3p Regulates the Epithelial-Mesenchymal Transition by Directly Targeting BMP7 in NRK52e
Abstract
:1. Introduction
2. Results
2.1. MiR542-3p Expression Was Up-Regulated in Fibrotic Kidney
2.2. Identification of TGFβ1 as a Positive Regulator of MiR542-3p Expression
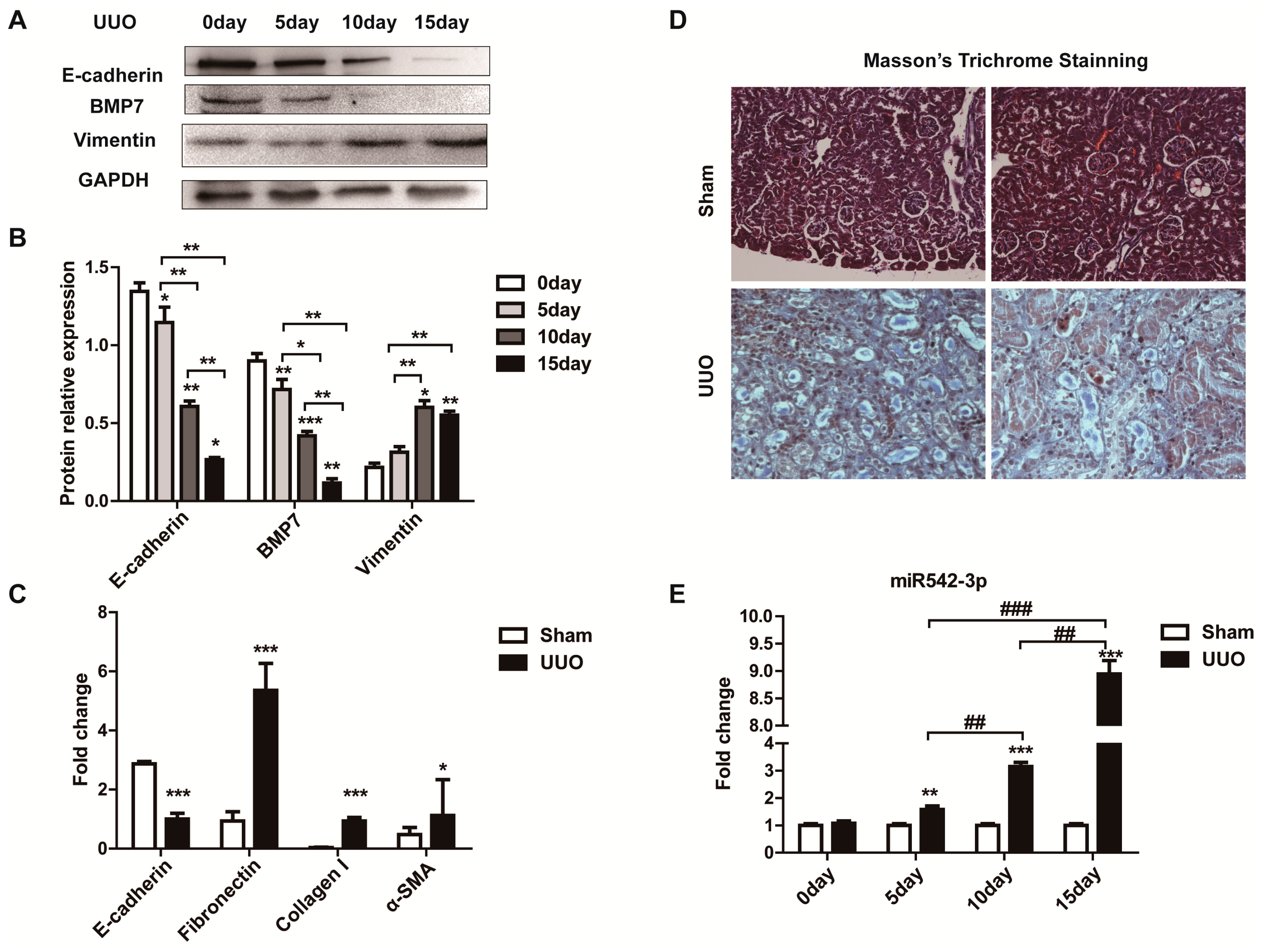
2.3. MiR542-3p Induce Epithelial-to-Mesenchymal Transition (EMT)
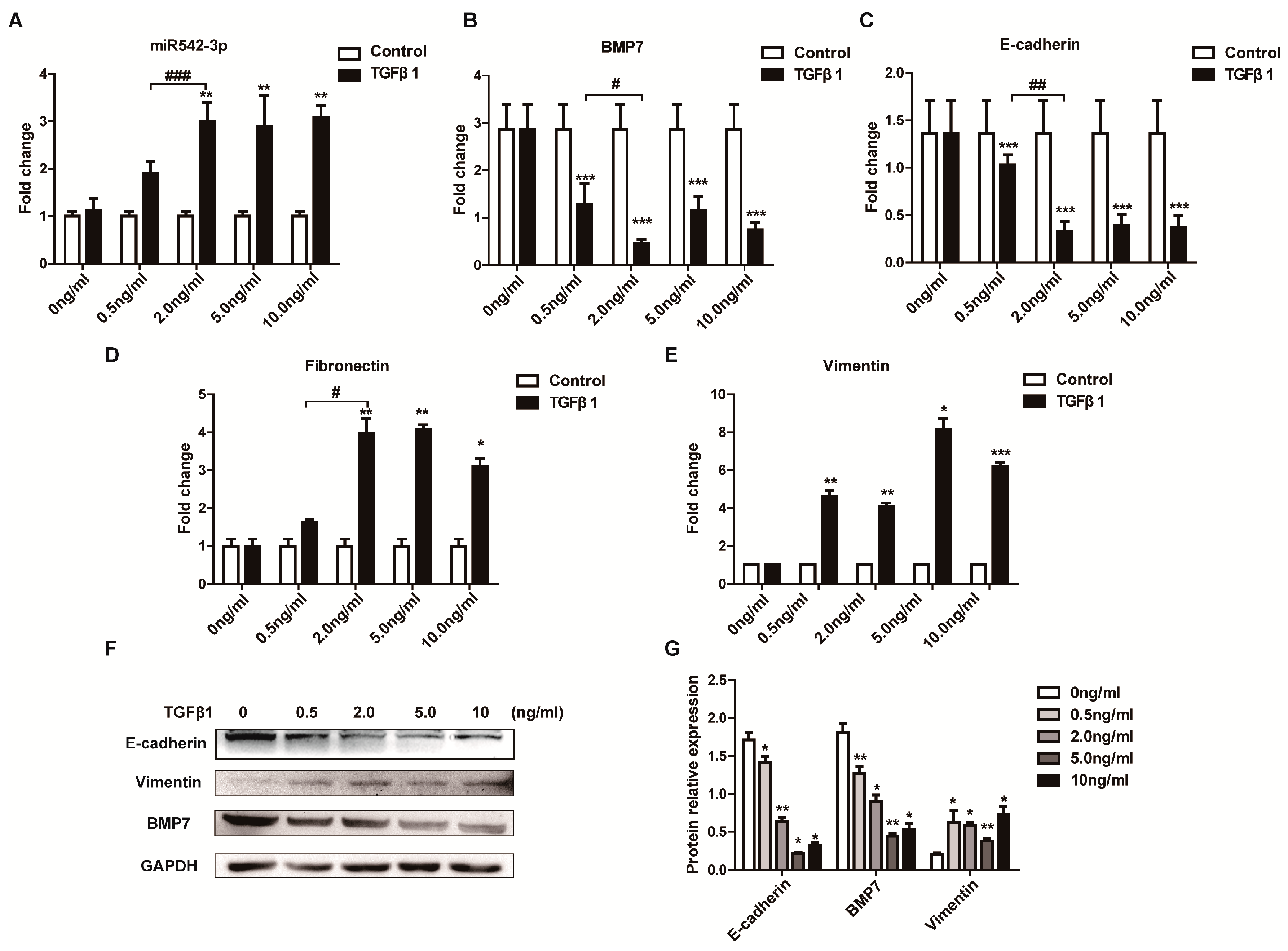
2.4. BMP7 Is a Direct Target of MiR542-3p
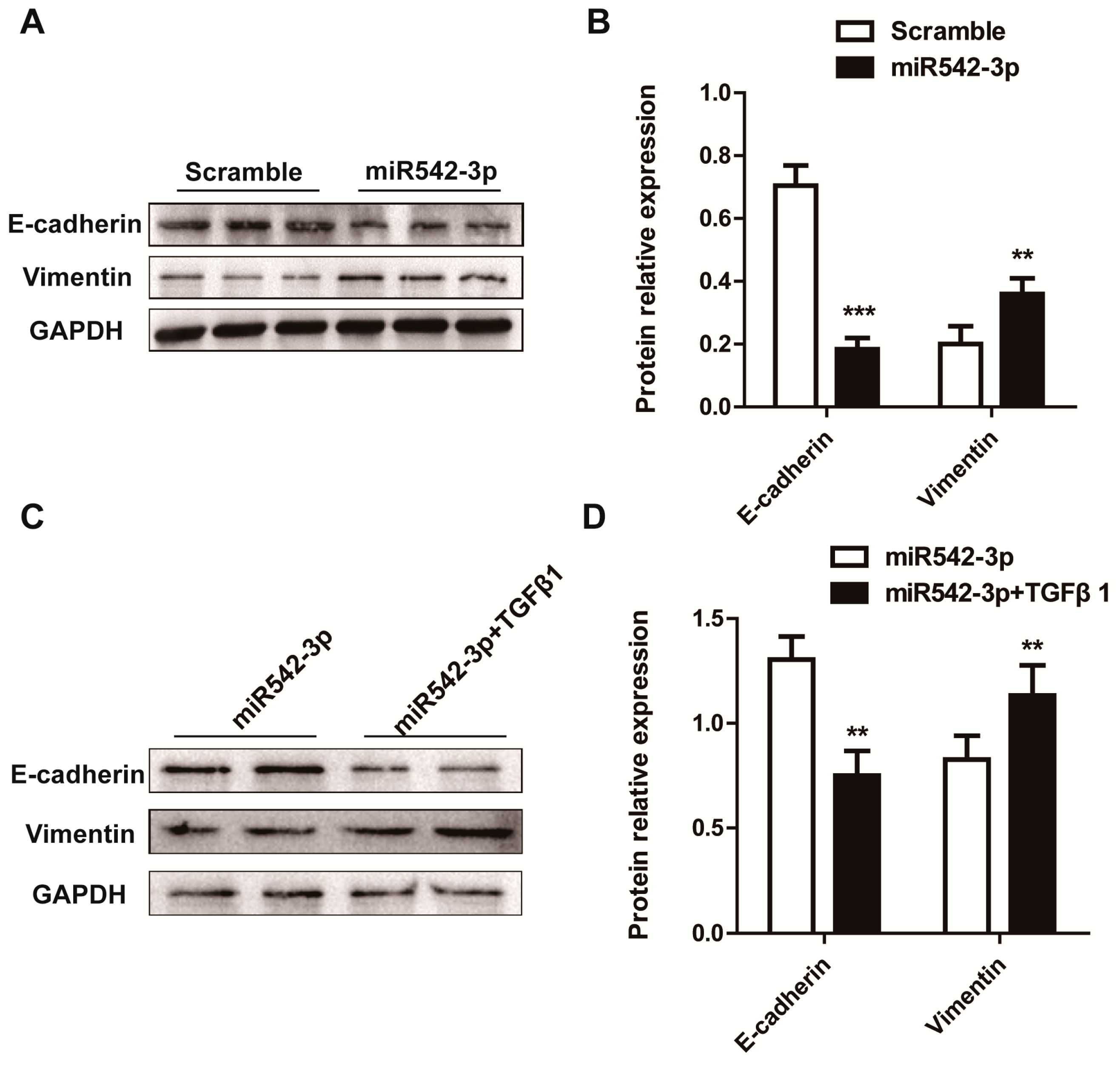
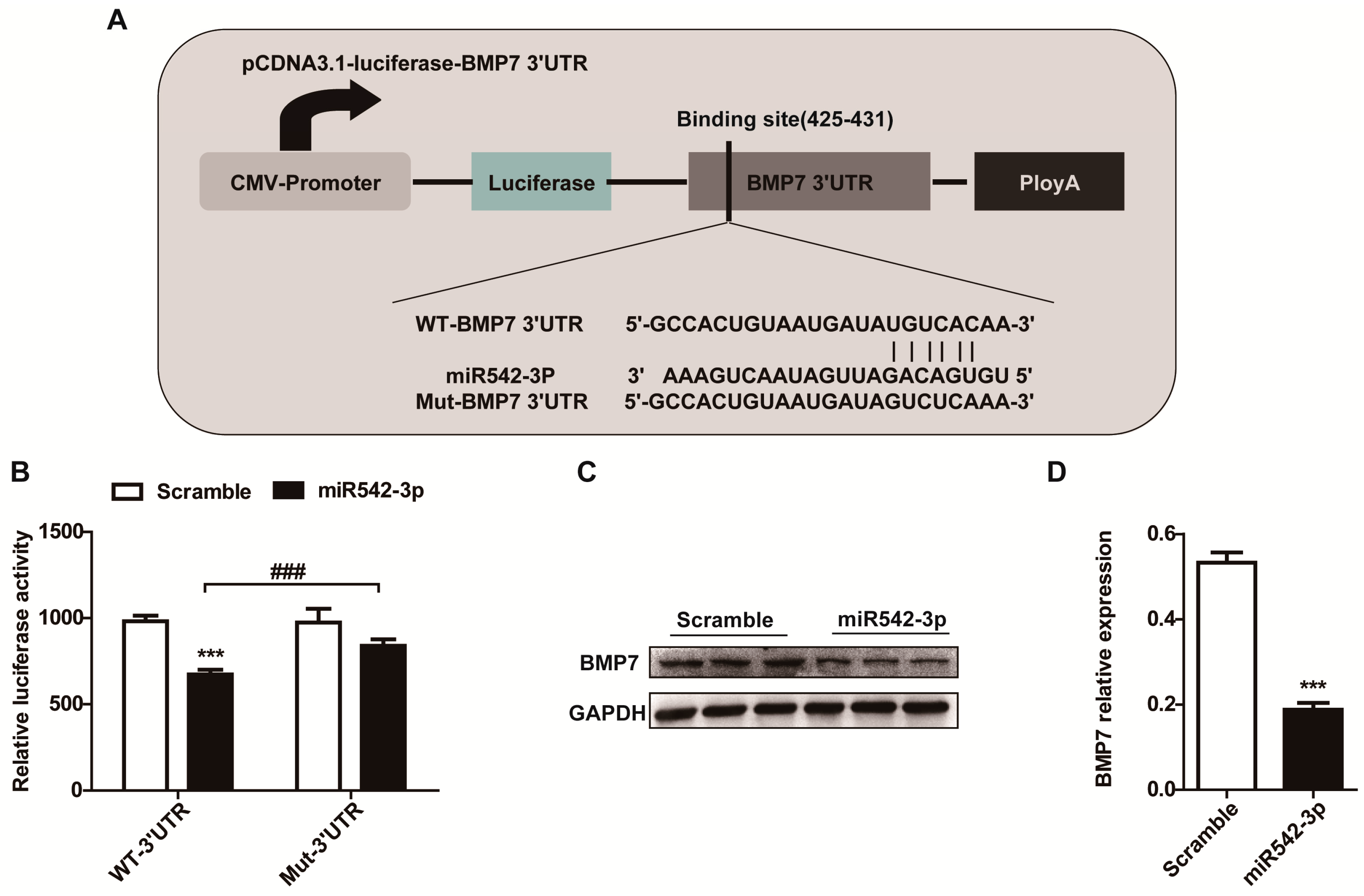
2.5. Overexpression of BMP7 Attenuates the MiR542-3p-Induced EMT
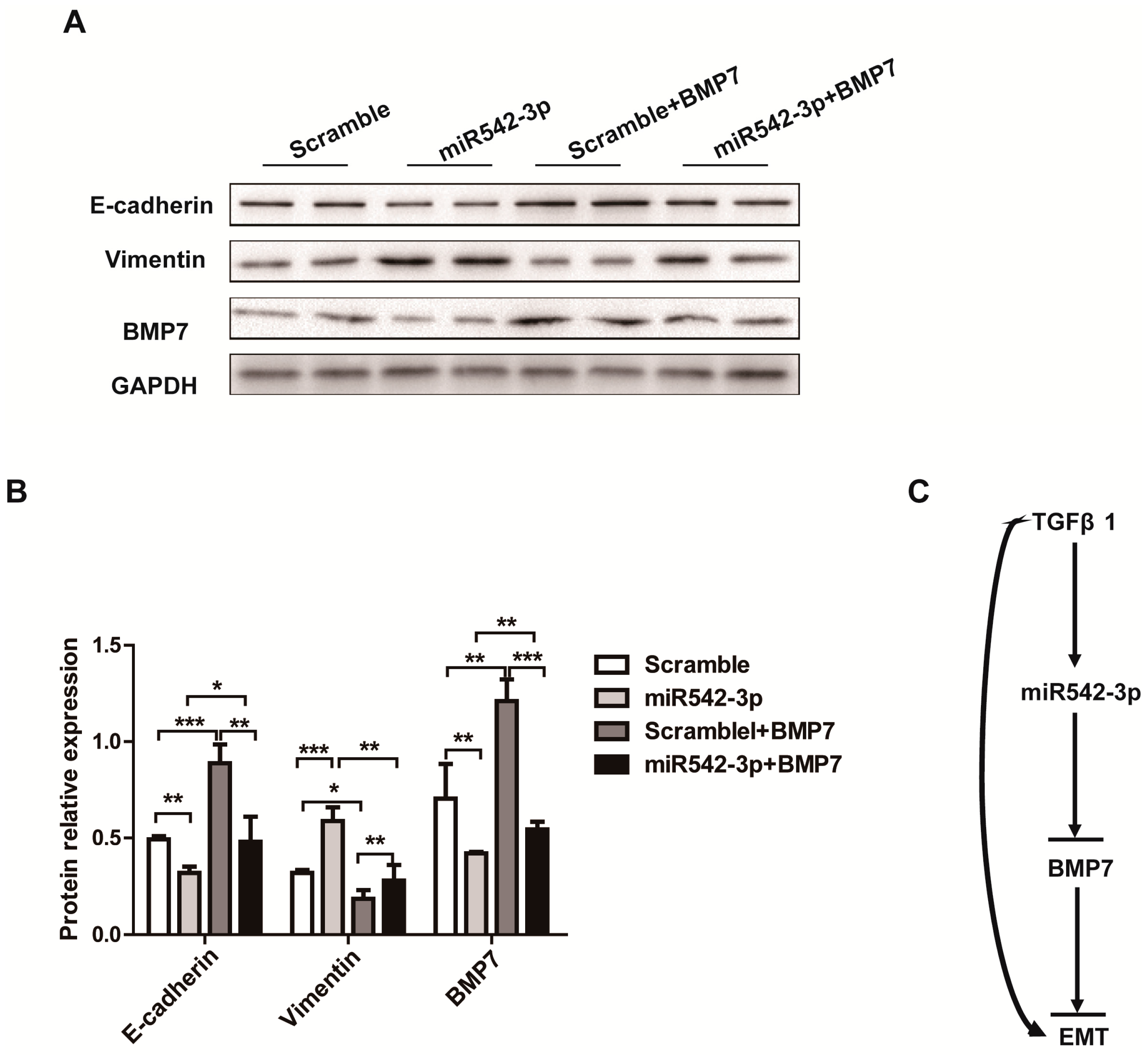
3. Discussion
4. Experimental Section
4.1. Obstructive Kidney Disease Model
4.2. Cell Culture and Transfection
4.3. RNA Isolation and Quantitative Real-Time PCR Analysis
| Gene Name | Sense | Antisense |
|---|---|---|
| BMP7 | CAGCCACCAGCAACCACT | GTCCATGCCGTCCAATCA |
| Ecadherin | GTCAACACCTACAACGCTGC | ACGTGCTTGGGTTGAAGACA |
| Vimentin | TGACCGCTTCGCCAACTA | CGCAACTCCCTCATCTCCT |
| Collagen I | AACTTTGCTTCCCAGATFTCCTATG | GCTTCCCCATCATCTCCATTCTTGC |
| a-SMA | GGGGTGATGGTGGGAATG | GGGGTGATGGTGGGAATG |
| Fibnectin | TGTGACCCAGACTTACGG | TGTAGGTGAACGGGAGAA |
| U6 | CTCGCTTCGGCAGCACA | AACGCTTCACGAATTTGCGT |
| 18sRNA | GTAACCCGTTGAACCCCATT | CCATCCAATCGGTAGTAGCG |
4.4. Western Bolt Analysis
4.5. Dual Luciferase Reporter Assay
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bohle, A.; Christ, H.; Grund, K.E.; Mackensen, S. The role of the interstitium of the renal cortex in renal disease. Contrib. Nephrol. 1979, 16, 109–114. [Google Scholar] [PubMed]
- Borges, F.T.; Melo, S.A.; Ozdemir, B.C.; Kato, N.; Revuelta, I.; Miller, C.A.; Gattone, V.H., 2nd; LeBleu, V.S.; Kalluri, R. TGF-β1-containing exosomes from injured epithelial cells activate fibroblasts to initiate tissue regenerative responses and fibrosis. J. Am. Soc. Nephrol. 2013, 24, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Kim, S.I.; Choi, M.E. Therapeutic targets for treating fibrotic kidney diseases. Transl. Res. J. Lab. Clin. Med. 2015, 165, 512–530. [Google Scholar] [CrossRef] [PubMed]
- Han, H.J.; Lee, Y.J.; Park, S.H.; Lee, J.H.; Taub, M. High glucose-induced oxidative stress inhibits Na+/glucose cotransporter activity in renal proximal tubule cells. Am. J. Physiol. Ren. Physiol. 2005, 288, F988–F996. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, A.; Nagase, S.; Ueda, A.; Oteki, T.; Takada, K.; Obara, M.; Inoue, M.; Yoh, K.; Hirayama, K.; Koyama, A. In vivo imaging of oxidative stress in ischemia-reperfusion renal injury using electron paramagnetic resonance. Am. J. Physiol. Ren. Physiol. 2005, 288, F597–F603. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Iturbe, B.; Vaziri, N.D.; Herrera-Acosta, J.; Johnson, R.J. Oxidative stress, renal infiltration of immune cells, and salt-sensitive hypertension: All for one and one for all. Am. J. Physiol. Ren. Physiol. 2004, 286, F606–F616. [Google Scholar] [CrossRef] [PubMed]
- Genovese, F.; Manresa, A.A.; Leeming, D.J.; Karsdal, M.A.; Boor, P. The extracellular matrix in the kidney: A source of novel non-invasive biomarkers of kidney fibrosis? Fibrogenes. Tissue Repair 2014, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Fu, W.; Xu, J.; Shao, L.; Wang, Y. Effect of BMP7 on podocyte transdifferentiation and Smad7 expression induced by hyperglycemia. Clin. Nephrol. 2015, 84, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Zhou, X.; Zhou, X.; Pi, C.; Xu, R.; Wan, M.; Yang, J.; Zhou, Y.; Liu, C.; Sun, J.; et al. BMP7 and EREG Contribute to the Inductive Potential of Dental Mesenchyme. Sci. Rep. 2015, 5, 9903. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; He, S.; Zeng, N.; Ma, J.; Zhang, B.; Shi, B.; Jia, Z. BMP7 Gene involved in nonsyndromic orofacial clefts in Western han Chinese. Med. Oral Patol. Oral Cir. Bucal 2015, 20, e298–e304. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Shao, Q.; Ji, D.; Li, F.; Chen, G. Mesenchymal stem cells mitigate cirrhosis through BMP7. Cell. Physiol. Biochem. 2015, 35, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Pauk, M.; Grgurevic, L.; Brkljacic, J.; Kufner, V.; Bordukalo-Niksic, T.; Grabusic, K.; Razdorov, G.; Rogic, D.; Zuvic, M.; Oppermann, H.; et al. Exogenous BMP7 corrects plasma iron overload and bone loss in Bmp6−/− mice. Int. Orthop. 2015, 39, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Yasmin, N.; Bauer, T.; Modak, M.; Wagner, K.; Schuster, C.; Koffel, R.; Seyerl, M.; Stockl, J.; Elbe-Burger, A.; Graf, D.; et al. Identification of bone morphogenetic protein 7 (BMP7) as an instructive factor for human epidermal Langerhans cell differentiation. J. Exp. Med. 2013, 210, 2597–2610. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomita, M.; Asada, M.; Asada, N.; Nakamura, J.; Oguchi, A.; Higashi, A.Y.; Endo, S.; Robertson, E.; Kimura, T.; Kita, T.; et al. BMP7 maintains undifferentiated kidney progenitor population and determines nephron numbers at birth. PLoS ONE 2013, 8, e73554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, I.; Agarwal, P.; Thangjam, G.S.; Radhesh, R.; Rao, S.G.; Kondaiah, P. Role of TGF-β and BMP7 in the pathogenesis of oral submucous fibrosis. Growth Factors 2011, 29, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Dean, C.; Ito, M.; Makarenkova, H.P.; Faber, S.C.; Lang, R.A. BMP7 regulates branching morphogenesis of the lacrimal gland by promoting mesenchymal proliferation and condensation. Development 2004, 131, 4155–4165. [Google Scholar] [CrossRef] [PubMed]
- Zeisberg, M.; Kalluri, R. Reversal of experimental renal fibrosis by BMP7 provides insights into novel therapeutic strategies for chronic kidney disease. Pediatr. Nephrol. 2008, 23, 1395–1938. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.R.; Dressler, G.R. BMP7 signaling in renal development and disease. Trends Mol. Med. 2005, 11, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Zhao, J.; Zhang, N.; Xu, X.; Li, R.; Yi, Y.; Fang, L.; Zhang, L.; Li, M.; Wu, J.; et al. MicroRNA-542-3p Suppresses Tumor Cell Invasion via Targeting AKT Pathway in Human Astrocytoma. J. Biol. Chem. 2015, 290, 24678–24688. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Si, Y.; Yang, Z.; Wang, Q.; Yuan, J.; Zhang, X. MicroRNA-542-3p suppresses cell growth of gastric cancer cells via targeting oncogene astrocyte-elevated gene-1. Med. Oncol. 2015, 32. [Google Scholar] [CrossRef] [PubMed]
- Althoff, K.; Lindner, S.; Odersky, A.; Mestdagh, P.; Beckers, A.; Karczewski, S.; Molenaar, J.J.; Bohrer, A.; Knauer, S.; Speleman, F.; et al. MiR-542-3p exerts tumor suppressive functions in neuroblastoma by downregulating Survivin. Int. J. Cancer 2015, 136, 1308–1320. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, J.W.; Castella, M.; Huntsman, D.G.; Taniguchi, T. p53 is positively regulated by miR-542-3p. Cancer Res. 2014, 74, 3218–3227. [Google Scholar] [CrossRef] [PubMed]
- Kureel, J.; Dixit, M.; Tyagi, A.M.; Mansoori, M.N.; Srivastava, K.; Raghuvanshi, A.; Maurya, R.; Trivedi, R.; Goel, A.; Singh, D. miR-542-3p suppresses osteoblast cell proliferation and differentiation, targets BMP-7 signaling and inhibits bone formation. Cell Death Dis. 2014, 5, e1050. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Qi, F.; Jia, L.; Wang, S.; Song, N.; Guo, L.; Fu, Y.; Luo, Y. MicroRNA-542-3p inhibits tumour angiogenesis by targeting angiopoietin-2. J. Pathol. 2014, 232, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Bray, I.; Tivnan, A.; Bryan, K.; Foley, N.H.; Watters, K.M.; Tracey, L.; Davidoff, A.M.; Stallings, R.L. MicroRNA-542-5p as a novel tumor suppressor in neuroblastoma. Cancer Lett. 2011, 303, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Choi, Y.C.; Lee, S.; Jeong, Y.; Yoon, J.; Baek, K. Induction of growth arrest by miR-542-3p that targets survivin. FEBS Lett. 2010, 584, 4048–4052. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Gan, H.; Zhang, H.; Tang, W.; Sun, Y.; Tang, X.; Kong, D.; Zhou, J.; Wang, Y.; Zhu, Y. MicroRNA21 inhibits SMAD7 expression through a target sequence in the 3′ untranslated region and inhibits proliferation of renal tubular epithelial cells. Mol. Med. Rep. 2014, 10, 707–712. [Google Scholar] [PubMed]
- Wang, J.Y.; Gao, Y.B.; Zhang, N.; Zou, D.W.; Wang, P.; Zhu, Z.Y.; Li, J.Y.; Zhou, S.N.; Wang, S.C.; Wang, Y.Y.; et al. MiR-21 overexpression enhances TGF-beta1-induced epithelial-to-mesenchymal transition by target smad7 and aggravates renal damage in diabetic nephropathy. Mol. Cell. Endocrinol. 2014, 392, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.B.; Jiang, Z.P. Role of miR-21 and its signaling pathways in renal diseases. J. Recept. Signal Transduct. 2014, 34, 335–337. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Huang, C.; Li, J. MiR-21 is a critical therapeutic target for renal fibrosis. Cell Biochem. Biophys. 2014, 68, 635–636. [Google Scholar] [CrossRef] [PubMed]
- Chung, A.C.; Huang, X.R.; Meng, X.; Lan, H.Y. MiR-192 mediates TGF-beta/Smad3-driven renal fibrosis. J. Am. Soc. Nephrol. 2010, 21, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Herman-Edelstein, M.; Koh, P.; Burns, W.; Jandeleit-Dahm, K.; Watson, A.; Saleem, M.; Goodall, G.J.; Twigg, S.M.; Cooper, M.E.; et al. E-cadherin expression is regulated by miR-192/215 by a mechanism that is independent of the profibrotic effects of transforming growth factor-beta. Diabetes 2010, 59, 1794–1802. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Qi, F.; Jia, L.; Wang, S.; Wang, C.; Song, N.; Fu, Y.; Li, L.; Luo, Y. Tumor cell-secreted angiogenin induces angiogenic activity of endothelial cells by suppressing miR-542-3p. Cancer Lett. 2015, 368, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Qian, D.H.; Gao, P.; Feng, H.; Qin, Z.X.; Li, J.B.; Huang, L. Down-regulation of mir-542-3p promotes neointimal formation in the aging rat. Vasc. Pharmacol. 2015, 72, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Oneyama, C.; Morii, E.; Okuzaki, D.; Takahashi, Y.; Ikeda, J.; Wakabayashi, N.; Akamatsu, H.; Tsujimoto, M.; Nishida, T.; Aozasa, K.; et al. MicroRNA-mediated upregulation of integrin-linked kinase promotes Src-induced tumor progression. Oncogene 2012, 31, 1623–1635. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Bo, Y.; Shen, W.; Tan, J.; Jia, Z.; Xu, C.; Li, F. Leonurine ameliorates kidney fibrosis via suppressing TGF-β and NF-κB signaling pathway in UUO mice. Int. Immunopharmacol. 2015, 25, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Choi, M.E. Regulation of autophagy by TGF-β: Emerging role in kidney fibrosis. Semin. Nephrol. 2014, 34, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Yanagita, M. Inhibitors/antagonists of TGF-β system in kidney fibrosis. Nephrol. Dial. Transplant. 2012, 27, 3686–3691. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.E.; Ding, Y.; Kim, S.I. TGF-β signaling via TAK1 pathway: Role in kidney fibrosis. Semin. Nephrol. 2012, 32, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Treiber, T.; Meister, G. SMADs stimulate miRNA processing. Mol. Cell 2010, 39, 315–316. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Chung, A.C.; Chen, H.Y.; Meng, X.M.; Lan, H.Y. Smad3-mediated upregulation of miR-21 promotes renal fibrosis. J. Am. Soc. Nephrol. 2011, 22, 1668–1681. [Google Scholar] [CrossRef] [PubMed]
- Dodd, S. The pathogenesis of tubulointerstitial disease and mechanisms of fibrosis. Curr. Top. Pathol. 1995, 88, 51–67. [Google Scholar] [PubMed]
- Iwano, M.; Neilson, E.G. Mechanisms of tubulointerstitial fibrosis. Curr. Opin. Nephrol. Hypertens. 2004, 13, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Muller, G.A.; Zeisberg, M.; Strutz, F. The importance of tubulointerstitial damage in progressive renal disease. Nephrol. Dial. Transplant. 2000, 15 (Suppl. 6), 76–77. [Google Scholar] [CrossRef] [PubMed]
- Zeisberg, M.; Neilson, E.G. Mechanisms of tubulointerstitial fibrosis. J. Am. Soc. Nephrol. 2010, 21, 1819–1834. [Google Scholar] [CrossRef] [PubMed]
- Tandon, A.; Sharma, A.; Rodier, J.T.; Klibanov, A.M.; Rieger, F.G.; Mohan, R.R. BMP7 gene transfer via gold nanoparticles into stroma inhibits corneal fibrosis in vivo. PLoS ONE 2013, 8, e66434. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Hirschberg, R. BMP7 antagonizes TGF-β-dependent fibrogenesis in mesangial cells. Am. J. Physiol. Ren. Physiol. 2003, 284, F1006–F1013. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Zhou, Y.; Yuan, Y.; Nie, F.; Peng, R.; Li, Q.; Lyu, Z.; Mao, Z.; Huang, L.; Zhou, L.; et al. MiR542-3p Regulates the Epithelial-Mesenchymal Transition by Directly Targeting BMP7 in NRK52e. Int. J. Mol. Sci. 2015, 16, 27945-27955. https://doi.org/10.3390/ijms161126075
Liu Z, Zhou Y, Yuan Y, Nie F, Peng R, Li Q, Lyu Z, Mao Z, Huang L, Zhou L, et al. MiR542-3p Regulates the Epithelial-Mesenchymal Transition by Directly Targeting BMP7 in NRK52e. International Journal of Molecular Sciences. 2015; 16(11):27945-27955. https://doi.org/10.3390/ijms161126075
Chicago/Turabian StyleLiu, Zhicheng, Yuru Zhou, Yue Yuan, Fang Nie, Rui Peng, Qianyin Li, Zhongshi Lyu, Zhaomin Mao, Liyuan Huang, Li Zhou, and et al. 2015. "MiR542-3p Regulates the Epithelial-Mesenchymal Transition by Directly Targeting BMP7 in NRK52e" International Journal of Molecular Sciences 16, no. 11: 27945-27955. https://doi.org/10.3390/ijms161126075
APA StyleLiu, Z., Zhou, Y., Yuan, Y., Nie, F., Peng, R., Li, Q., Lyu, Z., Mao, Z., Huang, L., Zhou, L., Li, Y., Hao, J., Ni, D., Jin, Q., Long, Y., Ju, P., Yu, W., Liu, J., Hu, Y., & Zhou, Q. (2015). MiR542-3p Regulates the Epithelial-Mesenchymal Transition by Directly Targeting BMP7 in NRK52e. International Journal of Molecular Sciences, 16(11), 27945-27955. https://doi.org/10.3390/ijms161126075