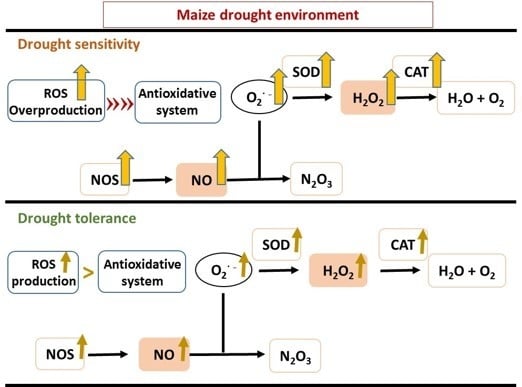
Stress Sensitivity Is Associated with Differential Accumulation of Reactive Oxygen and Nitrogen Species in Maize Genotypes with Contrasting Levels of Drought Tolerance
Abstract
:1. Introduction
2. Results
2.1. Seedling Morphological Responses to Drought in Selected Maize Genotypes
| Genotypes | Pedigree | Origin | Tolerance to Drought | Reference |
|---|---|---|---|---|
| B73 | Recurrent selection population (C5) of Iowa Stiff Stalk Synthetic | Iowa, USA | Susceptible | [40,41,42] |
| Lo1016 | P3369A × Lo876o2 | Italy | Susceptible | [37,43,44] |
| A638 | (V3 × Wf9) × Wf9 | Minnesota, USA | Moderate | [45] |
| Lo964 | P3183 | Italy | Tolerant | [37,43,44] |
| Va35 | (C103 × T8) × T8 | Virginia, USA | Tolerant | [46,47] |
| Grace-E5 | – | CIMMYT, Mexico | Tolerant | [48] |
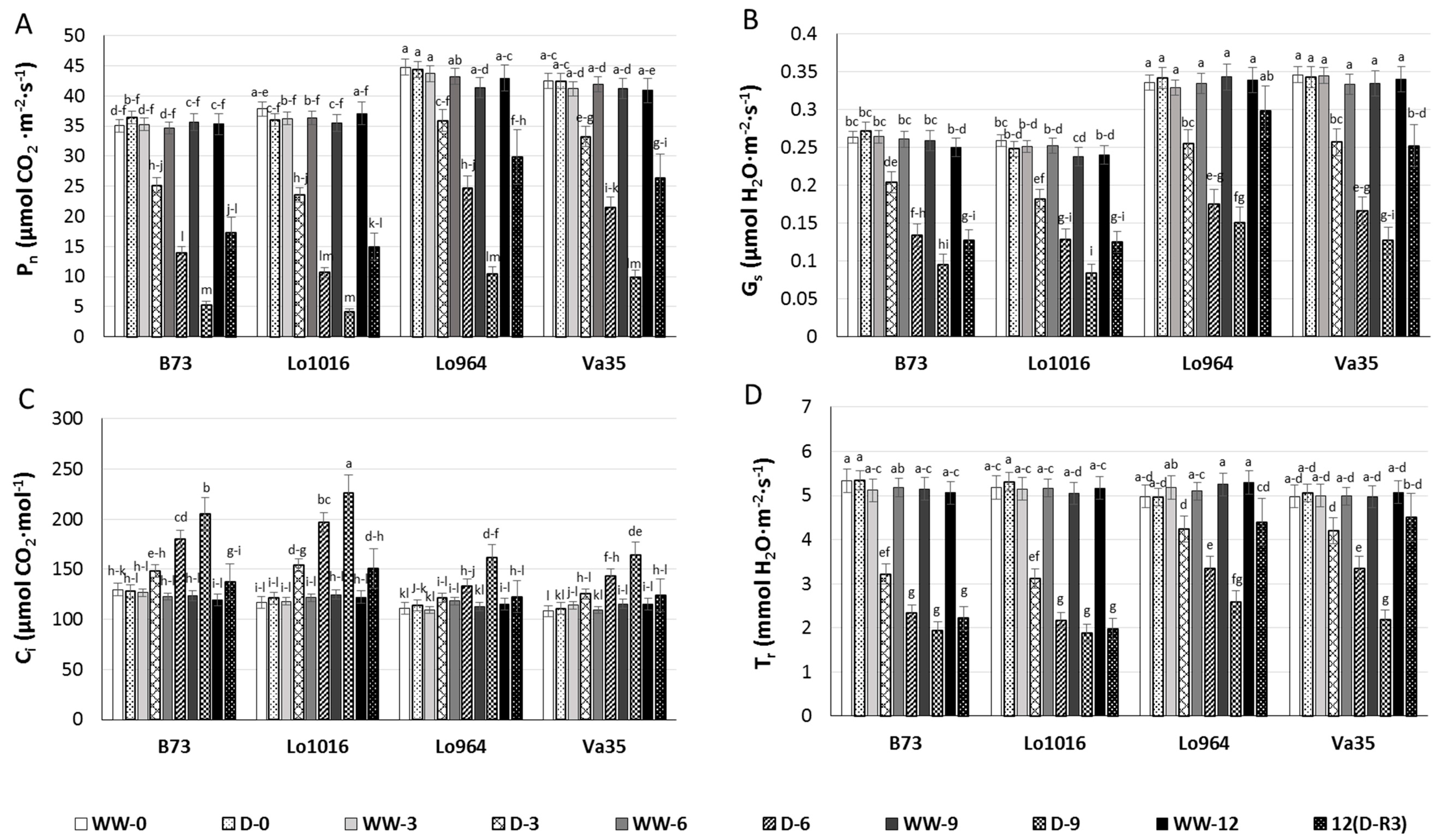
2.2. Changes of Chlorophyll Content and Photosynthesis Parameters in Response to Progressive Drought Stress and Recovery
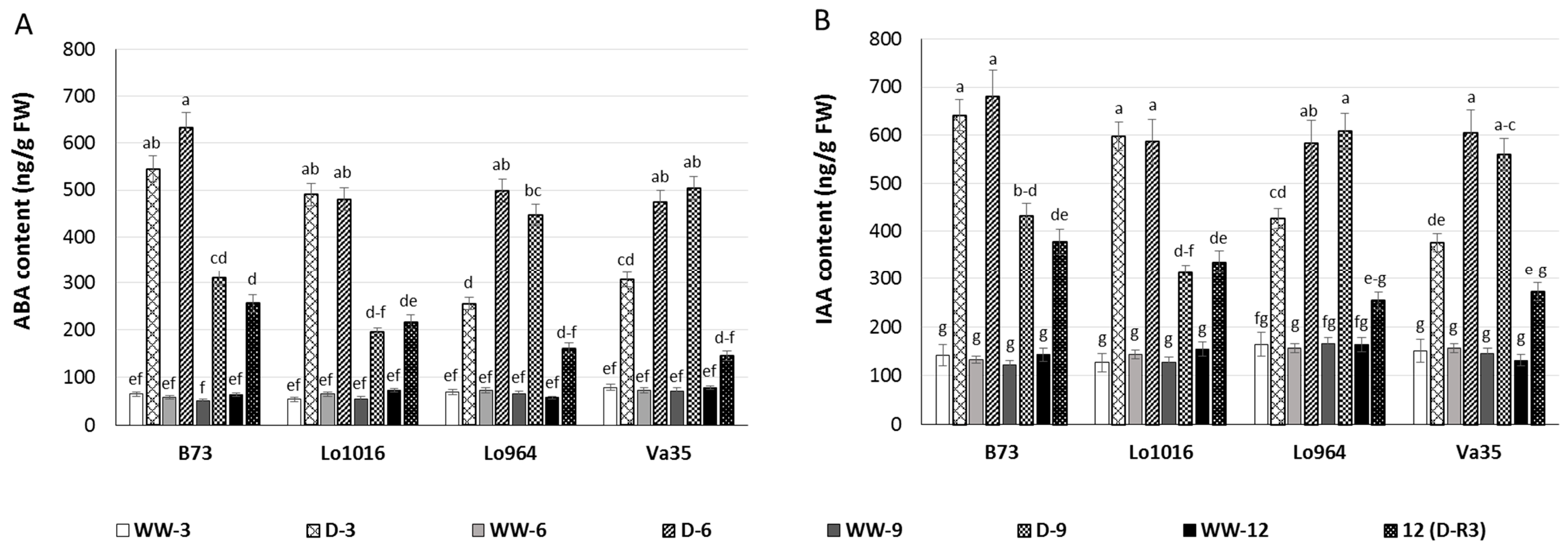
2.3. Drought Responses of Abscisic Acid (ABA) and Indole-3-Acetic Acid (IAA) Contents
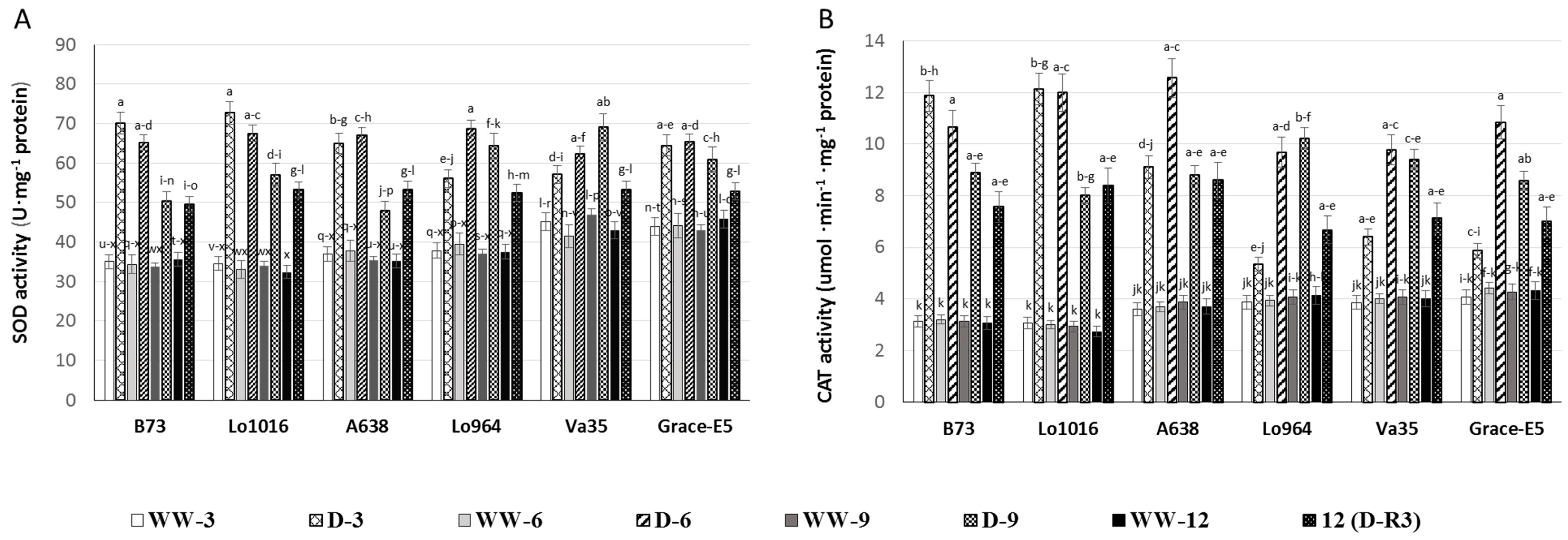
2.4. Effect of Drought Stress on ROS Metabolism
2.5. NO Production and RNS-Related Enzyme Activities during Drought Stress

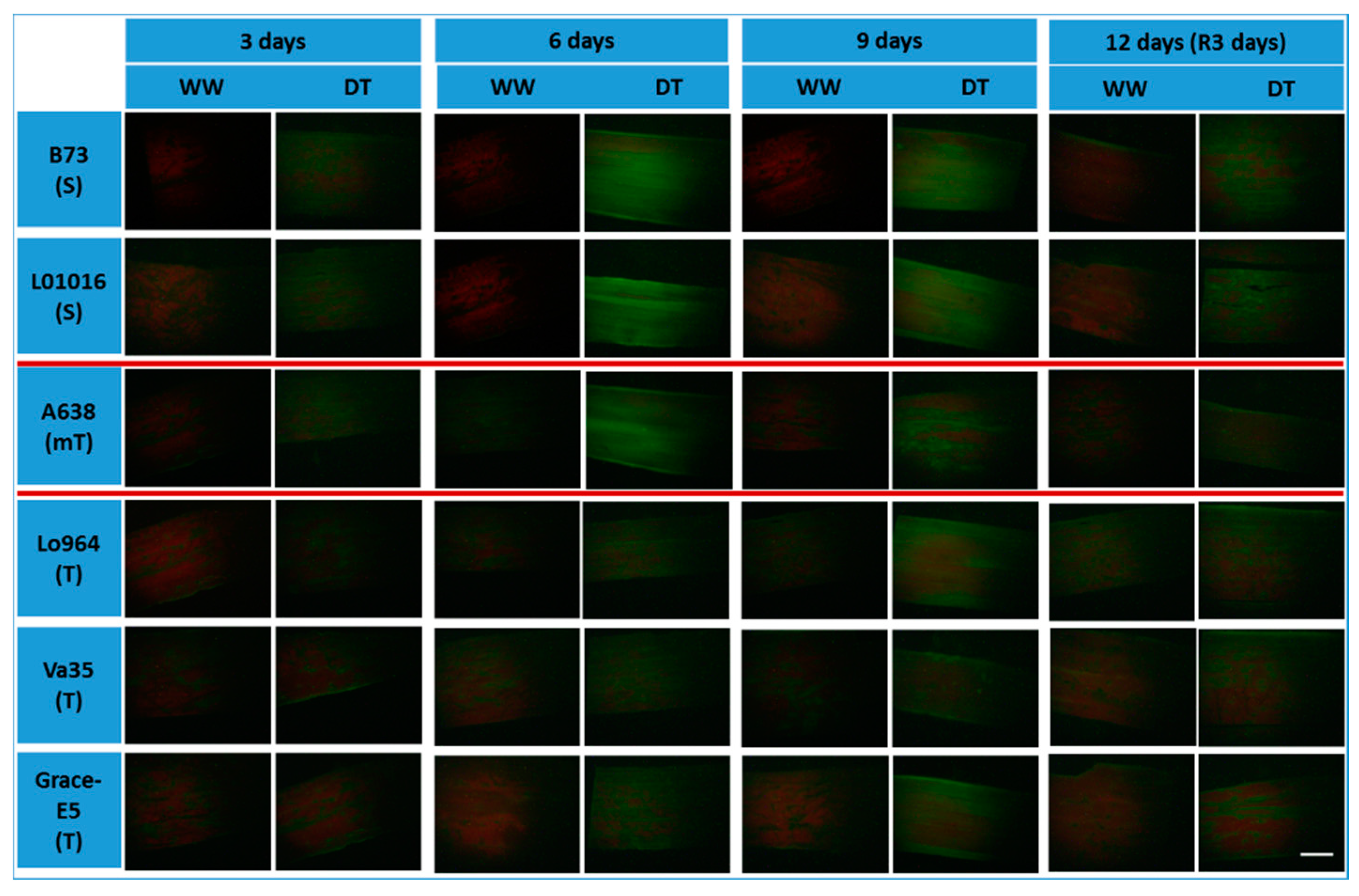
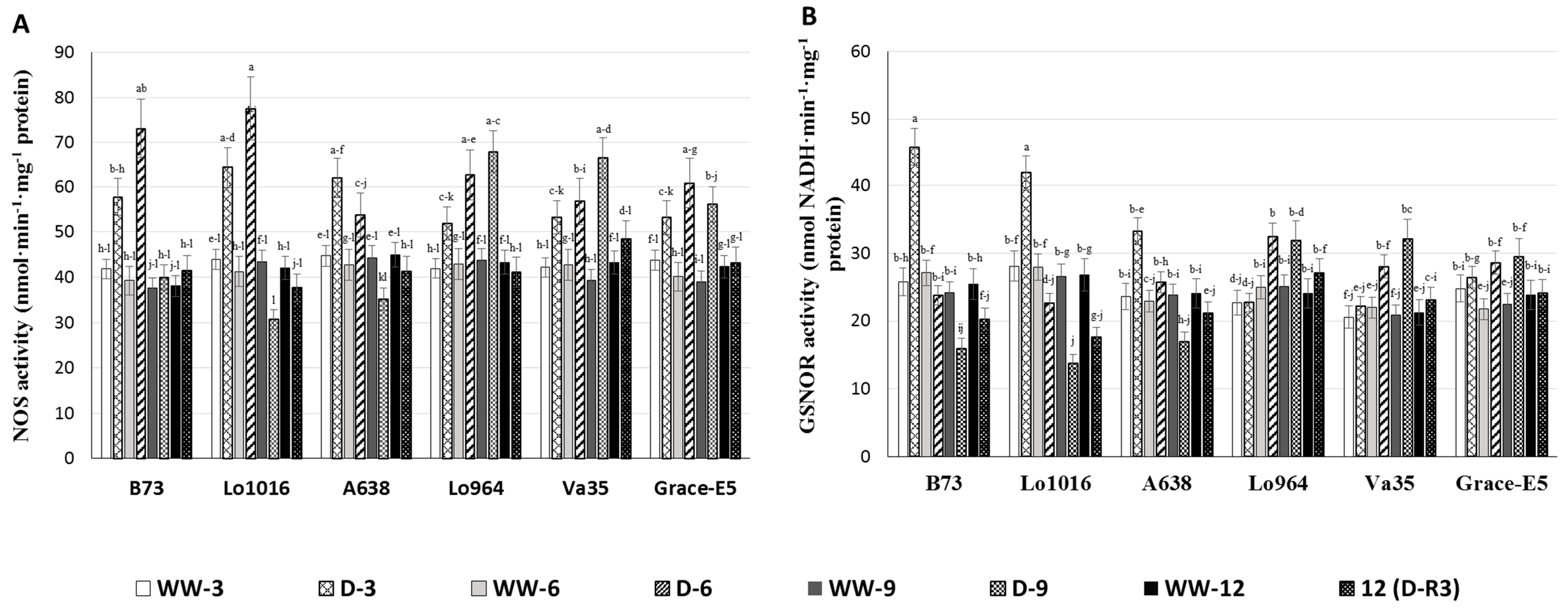
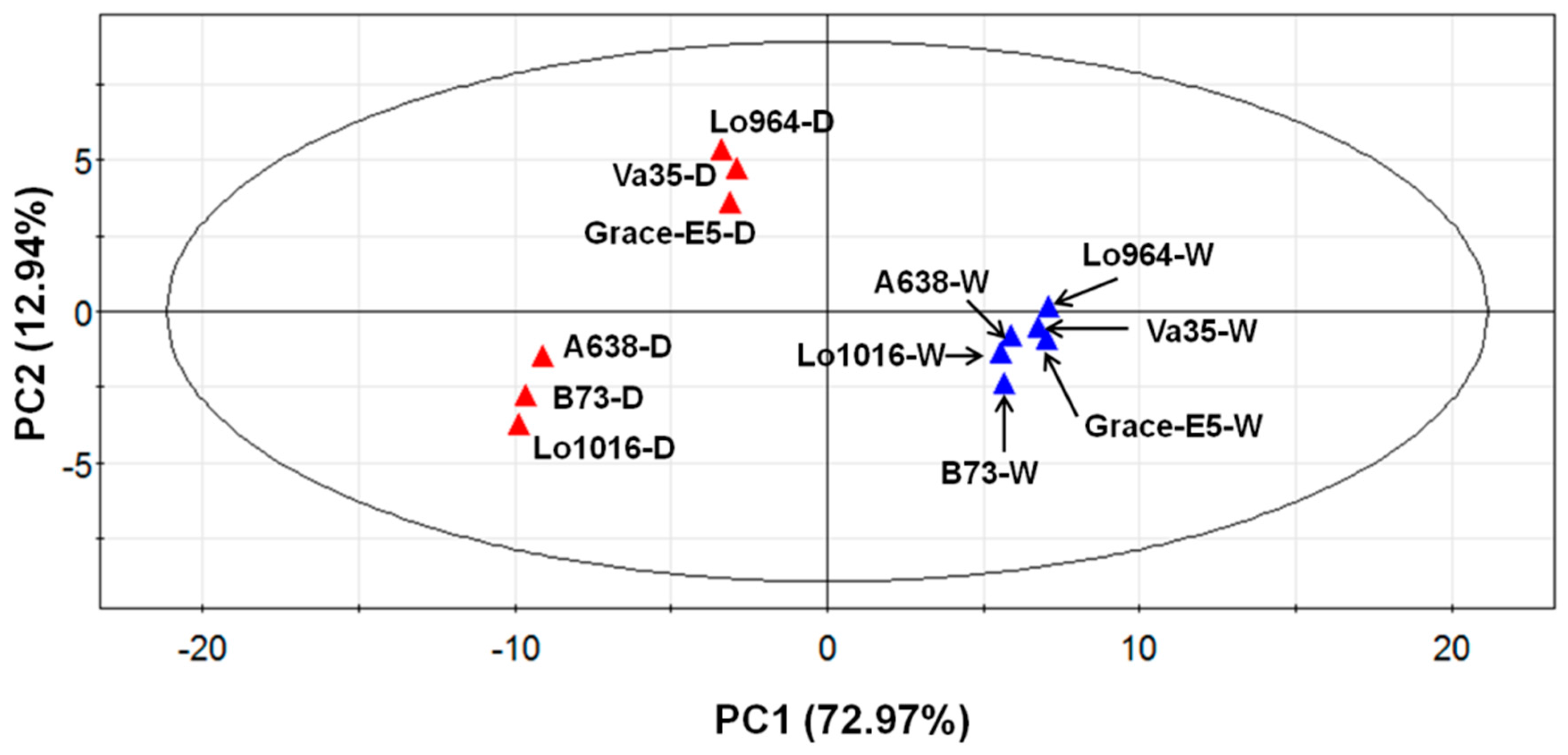
2.6. Physiological and Biochemical Patterns Allow Clear Separation of Drought Tolerance Characterization between Different Maize Lines
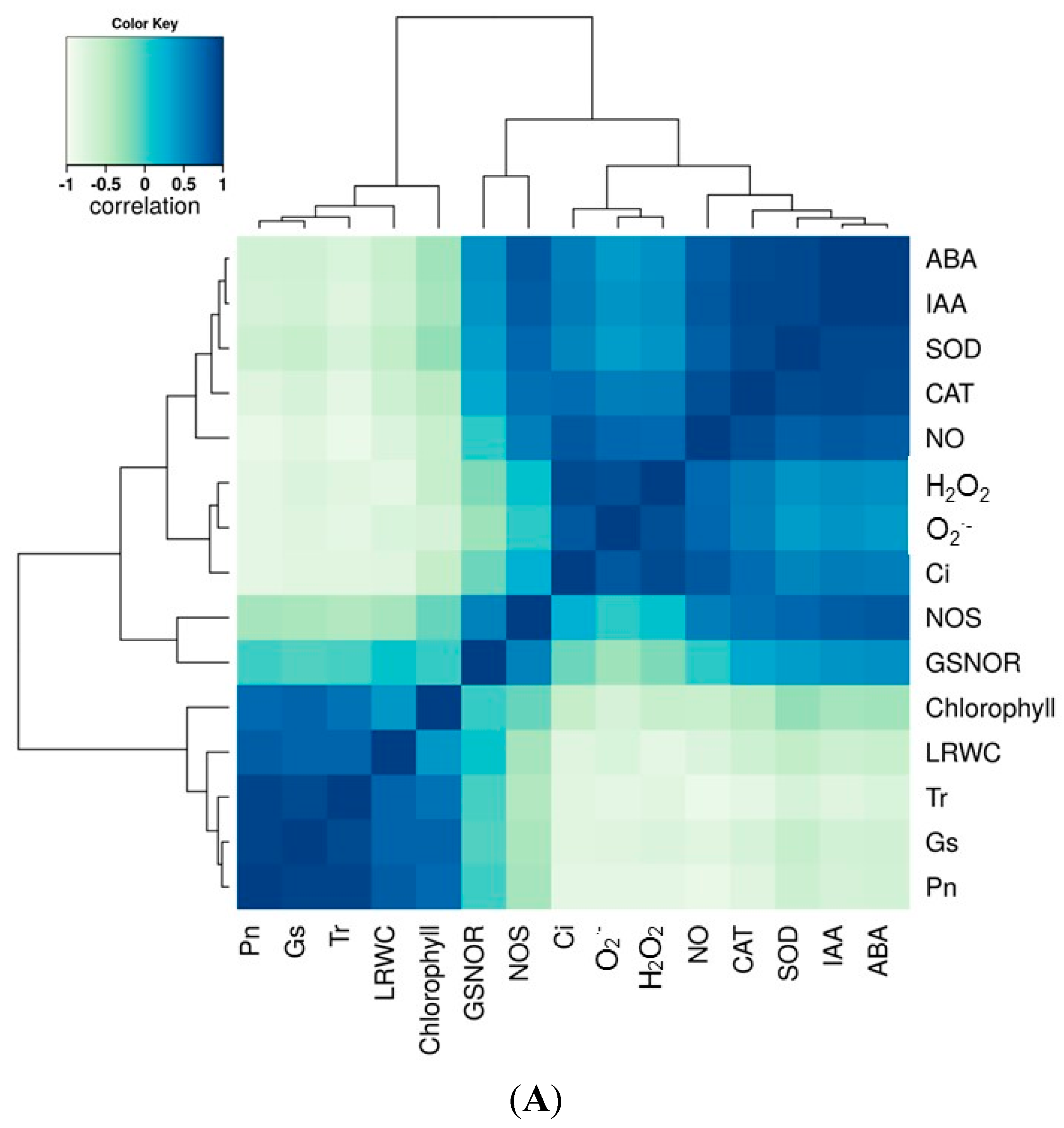
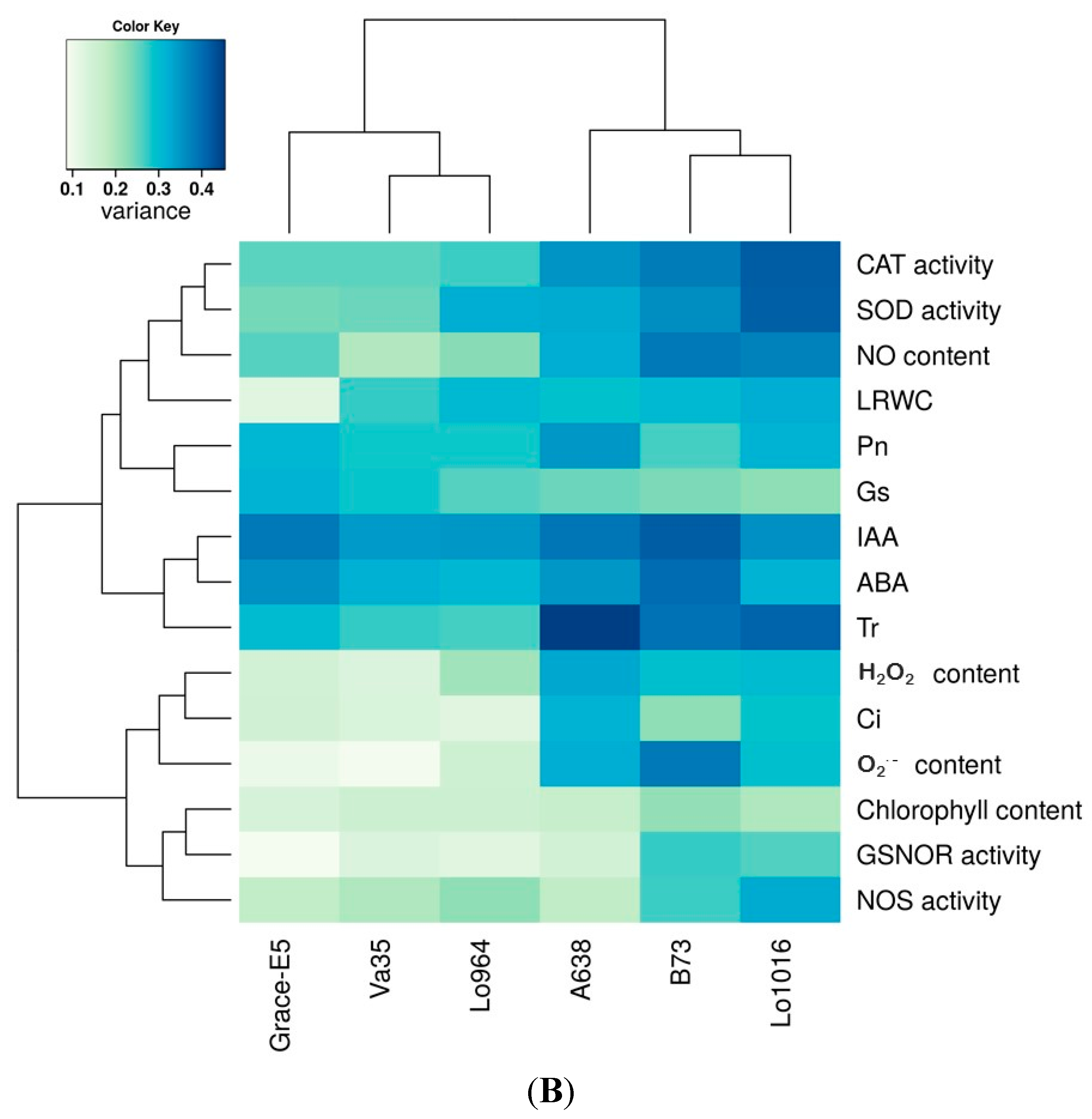
2.7. Correlation and Variance Analysis of All Tested Traits in All Lines
3. Discussion
3.1. Morphological and Physiological Responses to Drought Stress
3.2. Drought Stress Induces an ROS Burst and Disturbs the Redox Homeostasis
3.3. Drought Stress Affects NO Homeostasis
3.4. Drought Stress Responses Involve Interconnections between ROS and RNS
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Measurement of Plant Growth Rate and Leaf Relative Water Content
4.3. Photosynthesis Parameters Measurements and Determination of Chlorophyll
4.4. ABA and IAA Content Measurement in Leaves
4.5. Superoxide Radical Staining and Quantification
4.6. Hydrogen Peroxide Staining and Quantification
4.7. Extraction and Activity Determination of SOD and CAT
4.8. Detection and Quantification of NO
4.9. Determination of the NOS and GSNOR Activities
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guo, B.Z.; Chen, Z.Y.; Lee, R.D.; Scully, B.T. Drought stress and preharvest aflatoxin contamination in agricultural commodity: Genetics, genomics and proteomics. J. Integr. Plant Biol. 2008, 50, 1281–1291. [Google Scholar] [CrossRef] [PubMed]
- Scully, B.T.; Krakowsky, M.D.; Ni, X.Z.; Wilson, J.P.; Lee, R.D.; Guo, B.Z. Preharvest aflatoxincontamination of corn and other grain crops grown on the U.S. Southeastern Coastal Plain. Toxin Rev. 2009, 28, 169–179. [Google Scholar] [CrossRef]
- Fountain, J.C.; Scully, B.T.; Ni, X.; Kemerait, R.C.; Lee, R.D.; Chen, Z.Y.; Guo, B. Environmental influences on maize-Aspergillus flavus interactions and aflatoxin production. Front. Microbiol. 2014, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Fink, G. Encyclopedia of Stress; Academic Press: San Diego, CA, USA, 2000; Volume 3, p. 1341. [Google Scholar]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Luo, M.; Liu, J.; Lee, R.D.; Scully, B.T.; Guo, B.Z. Monitoring the expression of maize genes in developing kernels under drought stress using oligo-microarray. J. Integr. Plant Biol. 2010, 52, 1059–1074. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, S.; Roy Choudhury, S.; Pandey, S. Comparative quantitative proteomics analysis of the ABA response of roots of drought-sensitive and drought-tolerant wheat varieties identifies proteomic signatures of drought adaptability. J. Proteome Res. 2014, 13, 1688–1701. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M. Inducing drought tolerance in plants: Recent advances. Biotechnol. Adv. 2010, 28, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Miao, Y.; Song, C. Behind the scenes: The roles of reactive oxygen species in guard cells. New Phytol. 2014, 201, 1121–1140. [Google Scholar] [CrossRef]
- Dat, J.F.; Vandenabeele, E.; Vranova, M.; Mantagu, V.; Inze, D.; Breusegem, F.V. Dual action of the active oxygen species during plant stress responses. Cell. Mol. Life Sci. 2000, 57, 779–795. [Google Scholar] [CrossRef] [PubMed]
- Dalton, T.D.; Shertzer, H.G.; Puga, A. Regulation of gene expression by reactive oxygen. Annu. Rev. Pharmacol. Toxicol. 1999, 39, 67–101. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; van Breusegem, F. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Kerchev, P.; Mühlenbock, P.; Denecker, J.; Morreel, K.; Hoeberichts, F.A.; van Der Kelen, K.; Vandorpe, M.; Nguyen, L.; Audenaert, D.; van Breusegem, F. Activation of auxin signalling counteracts photorespiratory H2O2-dependent cell death. Plant Cell Environ. 2015, 38, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.Y.; Liu, J.Y. Comparative proteomics analysis reveals an intimate protein network provoked by hydrogen peroxide stress in rice seedling leaves. Mol. Cell. Proteom. 2008, 7, 1469–1488. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lin, A.; Loake, G.J.; Chu, C. H2O2-induced leaf cell death and the crosstalk of reactive nitric/oxygen species. J. Integr. Plant Biol. 2013, 55, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Hossain, M.A.; Silva, J.A.T.; Fujita, F. Plant response and tolerance to abiotic oxidative stress: Antioxidant defense is a key factor. In Crop Stress and Its Management: Perspectives and Strategies; Venkateswarlu, B., Shanker, A.K., Shanker, C., Maheswari, M., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 261–315. [Google Scholar]
- Signorelli, S.; Corpas, F.J.; Borsani, O.; Barroso, J.B.; Monza, J. Water stress induces a differential and spatially distributed nitro-oxidative stress response in roots and leaves of Lotus japonicus. Plant Sci. 2013, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Jiang, T.; Fountain, J.C.; Scully, B.T.; Lee, R.D.; Kemerait, R.C.; Chen, S.; Guo, B. Protein profiles reveal diverse responsive signaling pathways in kernels of two maize inbred lines with contrasting drought sensitivity. Int. J. Mol. Sci. 2014, 15, 18892–18918. [Google Scholar] [CrossRef]
- Valderrama, R.; Corpas, F.J.; Carreras, A.; Gómez-Rodríguez, M.V.; Chaki, M.; Pedrajas, J.R.; Fernández-Ocaña, A.; del Río, L.A.; Barroso, J.B. The dehydrogenase-mediated recycling of NADPH is a key antioxidant system against salt-induced oxidative stress in olive plants. Plant Cell Environ. 2006, 29, 1449–1459. [Google Scholar] [CrossRef] [PubMed]
- Hill, B.G.; Dranka, B.P.; Bailey, S.M.; Lancaster, J.R.; Darley-Usmar, V.M. What part of NO don’t you understand? Some answers to the cardinal questions in nitric oxide biology. J. Biol. Chem. 2010, 285, 19699–19704. [Google Scholar] [CrossRef] [PubMed]
- Groß, F.; Durner, J.; Gaupels, F. Nitric oxide, antioxidants and pro-oxidants in plant defense responses. Front. Plant Sci. 2013, 4, 419. [Google Scholar] [CrossRef] [PubMed]
- Airaki, M.; Leterrier, M.; Mateos, R.M.; Valderrama, R.; Chaki, M.; Barroso, J.B.; del Río, L.A.; Palma, J.M.; Corpas, F.J. Metabolism of reactive oxygen species and reactive nitrogen species in pepper (Capsicum annuum L.) plants under low temperature stress. Plant Cell Environ. 2012, 35, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Yang, L.; Yang, Y.; Ahmad, P.; Yang, Y.; Hu, X. Deciphering the protective role of nitric oxide against salt stress at the physiological and proteomic levels in maize. J. Proteome Res. 2011, 10, 4349–4364. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.M.; Tian, D.G.; Todd, C.D.; Luo, Y.M.; Hu, X.Y. Comparative proteome analyses reveal that nitric oxide is an important signal molecule in the response of rice to aluminum toxicity. J. Proteome Res. 2013, 12, 1316–1330. [Google Scholar] [CrossRef] [PubMed]
- Díaz, M.; Achkor, H.; Titarenko, E.; Martínez, M.C. The gene encoding glutathione-dependent formaldehyde dehydrogenase/GSNO reductase is responsive to wounding, jasmonic acid and salicylic acid. FEBS Lett. 2003, 543, 136–139. [Google Scholar] [CrossRef]
- Corpas, F.J.; Chaki, M.; Fernández-Ocaña, A.; Valderrama, R.; Palma, J.M.; Carreras, A.; Airaki, M.; Begara-Morales, J.C.; del Río, L.A.; Barroso, J.B. Metabolism of reactive nitrogen species in pea plants under abiotic stress conditions. Plant Cell Physiol. 2008, 49, 1711–1722. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Liu, J. Nitric oxide is involved in dehydration/drought tolerance in Poncirus trifoliata seedlings through regulation of antioxidant systems and stomatal response. Plant Cell Rep. 2012, 31, 145–154. [Google Scholar] [PubMed]
- Shan, C.; Zhou, Y.; Liu, M. Nitric oxide participates in the regulation of the ascorbate-glutathione cycle by exogenous jasmonic acid in the leaves of wheat seedlings under drought stress. Protoplasma 2015, 252, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, X.; Zhang, C.; Fu, G.; Chen, T.; Tao, L. Effects of nitric oxide on drought stress induced physiological characteristics in leaves of nipponbare (Oryza sativa L.). Chin. J. Rice Sci. 2015, 29, 65–72. [Google Scholar]
- Wang, P.; Du, Y.; Hou, Y.J.; Zhao, Y.; Hsu, C.C.; Yuan, F.; Zhu, X.; Tao, W.A.; Song, C.P.; Zhu, J.K. Nitric oxide negatively regulates abscisic acid signaling in guard cells by S-nitrosylation of OST1. Proc. Natl. Acad. Sci. USA 2015, 112, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.B.; Huang, G.B.; Yu, J.H.; Zhang, M.L. Nitric oxide and hydrogen peroxide alleviate drought stress in marigold explants and promote its adventitious root development. Plant Physiol. Biochem. 2012, 58, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Jiang, M.; Zhang, J.; Ding, H.; Xu, S.; Hu, X.; Tan, M. Nitric oxide induced by hydrogen peroxide mediates abscisic acid-induced activation of the mitogen-activated protein kinase cascade involved in antioxidant defense in maize leaves. New Phytol. 2007, 175, 36–50. [Google Scholar] [CrossRef] [PubMed]
- Hayano-Kanashiro, C.; Calderón-Vázquez, C.; Ibarra-Laclette, E.; Herrera-Estrella, L.; Simpson, J. Analysis of gene expression and physiological responses in three Mexican maize landraces under drought stress and recovery irrigation. PLoS ONE 2009. [Google Scholar] [CrossRef] [PubMed]
- Fountain, J.C.; Chen, Z.Y.; Scully, B.T.; Kemerait, R.C.; Lee, R.D.; Guo, B.Z. Pathogenesis-related gene expressions in different maize genotypes under drought stressed conditions. Afr. J. Plant Sci. 2010, 4, 433–440. [Google Scholar]
- Jiang, T.B.; Zhou, B.R.; Luo, M.; Abbas, H.K.; Kemerait, R.; Lee, R.D.; Scully, B.T.; Guo, B.Z. Expression analysis of stress-related genes in kernels of different maize (Zea mays L.) inbred lines with different resistance to aflatoxin contamination. Toxins 2011, 3, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.B.; Fountain, J.; Davis, G.; Kemerait, R.; Scully, B.T.; Lee, R.D.; Guo, B.Z. Root morphology and gene expression analysis in response to drought stress in maize (Zea mays). Plant Mol. Biol. Rep. 2012, 30, 360–369. [Google Scholar] [CrossRef]
- Krishnan, A.; Batlang, U.; Myers, E.; Grene, R.; Pereira, A. Effects of drought on gene expression in maize reproductive and leaf meristem tissue revealed by RNA-Seq. Plant Physiol. 2012, 160, 846–867. [Google Scholar]
- Humbert, S.; Subedi, S.; Cohn, J.; Zeng, B.; Bi, Y.M.; Chen, X.; Zhu, T.; McNicholas, P.D.; Rothstein, S.J. Genome-wide expression profiling of maize in response to individual and combined water and nitrogen stresses. BMC Genom. 2013. [Google Scholar] [CrossRef] [PubMed]
- Russell, W.A. Registration of B70 and B73 parental lines of maize. Crop Sci. 1972, 12, 721. [Google Scholar] [CrossRef]
- Chen, J.; Xu, W.; Velten, J.; Xin, Z.; Stout, J. Characterization of maize inbred lines for drought and heat tolerance. J. Soil Water Conserv. 2012, 67, 354–364. [Google Scholar] [CrossRef]
- Xu, J.; Yuan, Y.; Xu, Y.; Zhang, G.; Guo, X.; Wu, F.; Wang, Q.; Rong, T.; Pan, G.; Cao, M.; et al. Identification of candidate genes for drought tolerance by whole-genome resequencing in maize. BMC Plant Biol. 2014, 14, 83. [Google Scholar] [CrossRef] [PubMed]
- Sanguineti, M.C.; Giuliani, M.M.; Govi, G.; Tuberosa, R.; Landi, P. Root and shoot traits of maize inbred lines grown in the field and in hydroponic culture and their relationship with root lodging. Maydica 1998, 43, 211–216. [Google Scholar]
- Losa, A.; Hartings, H.; Verderio, A.; Motto, M. Assessment of genetic diversity and relationships among maize inbred lines developed in Italy. Maydica 2010, 56, 95–104. [Google Scholar]
- Aslam, M.; Khan, I.A.; Saleem, M.; Ali, Z. Assessment of water stress tolerance in different maize accessions at germination and early growth stage. Pakistan J. Bot. 2006, 38, 1571–1579. [Google Scholar]
- Marković, K.; Anđelković, V.; Šukalović, H.T.V.; Vuletić, M. The influence of osmotic stress on the superoxide dismutase and peroxidase isozymes in roots of two maize genotypes. In Genetics, Plant Breeding and Seed Production. Proceedings of 51th Croatian and 11th International Symposium on Agriculture, Opatija, Croatia, 15–18 February 2015; Agriculturae Conspectus Scientificus: Zagreb, Croatia, 2010; pp. 461–465. [Google Scholar]
- Vuletić, M.; Hadži-TaškovićŠukalović, V.; Marković, K.; DragišićMaksimović, J. Antioxidative system in maize roots as affected by osmotic stress and different nitrogen sources. Biol. Plant. 2010, 54, 530–534. [Google Scholar] [CrossRef]
- Ramugondo, R.; McDonald, A. New maize offers better livelihoods for poor farmers. Available online: http://repository.cimmyt.org/xmLui/bitstream/handle/10883/3679/74981.pdf (accessed on 13 March 2014).
- Harris, M.J.; Outlaw, W.H.; Mertens, R.; Weiler, E.W. Water-stress-induced changes in the abscisic acid content of guard cells and other cells of Vicia faba L. leaves as determined by enzyme-amplified immunoassay. Proc. Natl. Acad. Sci. USA 1988, 85, 2584–2588. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y. Auxin biosynthesis and its role in plant development. Annu. Rev. Plant Biol. 2010, 61, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.Q.; Okamoto, M.; Crawford, N.M. Identification of a plant nitric oxide synthase gene involved in hormonal signaling. Science 2003, 302, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Gas, E.; Flores-Pérez, U.; Sauret-Güeto, S.; Rodríguez-Concepción, M. Hunting for plant nitric oxide synthase provides new evidence of a central role for plastids in nitric oxide metabolism. Plant Cell 2009, 21, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, K.; Tsumoto, H.; Wei, W.; Tang, C.H.; Komatsubara, A.T.; Kawafune, H.; Shimizu, K.; Liu, L.; Tsujimoto, G. Proteomic analysis of the role of S-nitrosoglutathione reductase in lipopolysaccharide-challenged mice. Proteomics 2012, 12, 2024–2035. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhou, G.; Wang, Y.; Han, G.; Li, Y. Changes in chlorophyll fluorescence in maize plants with imposed rapid dehydration at different leaf ages. J. Plant Growth Regul. 2008, 27, 83–92. [Google Scholar] [CrossRef]
- De Souza, T.C.; Magalhaes, P.C.; de Castro, E.M.; de Albuquerque, P.E.P.; Marabesi, M.A. The influence of ABA on water relation, photosynthesis parameters, and chlorophyll fluorescence under drought conditions in two maize hybrids with contrasting drought resistance. Acta Physiol. Plant. 2013, 35, 515–527. [Google Scholar] [CrossRef]
- Benešová, M.; Holá, D.; Fischer, L.; Jedelský, P.L.; Hnilička, F.; Wilhelmová, N.; Rothová, O.; Kočová, M.; Procházková, D.; Honnerová, J.; et al. The physiology and proteomics of drought tolerance in maize: Early stomatal closure as a cause of lower tolerance to short-term dehydration? PLoS ONE 2012, 7, e38017. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.S.; Araus, J.L.; van Heerden, P.D.; Foyer, C.H. Enhancing drought tolerance in C4 crops. J. Exp. Bot. 2011, 62, 3135–3153. [Google Scholar] [CrossRef] [PubMed]
- De Micco, V.; Aronne, G. Morpho-anatomical traits for plant adaptation to drought. In Plant Responses to Drought Stress: From Morphological to Molecular Features; Ricardo, A., Ed.; Springer: New York, NY, USA, 2012; pp. 37–61. [Google Scholar]
- Wilkinson, S.; Davies, W.J. Drought, ozone, ABA and ethylene: New insights from cell to plant to community. Plant Cell Environ. 2010, 33, 510–525. [Google Scholar] [CrossRef] [PubMed]
- Harb, A.; Krishnan, A.; Ambavaram, M.M.; Pereira, A. Molecular and physiological analysis of drought stress in Arabidopsis reveals early responses leading to acclimation in plant growth. Plant Physiol. 2010, 154, 1254–1271. [Google Scholar] [CrossRef] [PubMed]
- Aimar, D.; Calafat, M.; Andrade, A.M.; Carassay, L.; Abdala, G.; Molas, M.L. Drought tolerance and stress hormones: From model organisms to forage crops. In Plants and Environment; Vasanthaiah, H.K.N., Kambiranda, D., Eds.; InTech: Rijeka, Croatia, 2011; pp. 137–164. [Google Scholar]
- Bai, L.; Wang, P.; Song, C. Reactive Oxygen Species (ROS) and ABA Signalling. In Abscisic Acid: Metabolism, Transport and Signaling; Zhang, D.P., Ed.; Springer: Dordrecht, The Netherlands, 2014; pp. 191–223. [Google Scholar]
- Kwak, J.M.; Mori, I.C.; Pei, Z.M.; Leonhardt, N.; Torres, M.A.; Dangl, J.L.; Bloom, R.E.; Bodde, S.; Jones, J.D.G.; Schroeder, J.I. NADPH oxidase AtrbohD and AtrbohF genes function in ROS-dependent ABA signaling in Arabidopsis. EMBO J. 2003, 22, 2623–2633. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, Y.; Wen, F.; Yao, D.; Wang, L.; Guo, J.; Ni, L.; Zhang, A.; Tan, M.; Jiang, M. A novel rice C2H2-type zinc finger protein, ZFP36, is a key player involved in abscisic acid-induced antioxidant defence and oxidative stress tolerance in rice. J. Exp. Bot. 2014, 65, 5795–5809. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Schumaker, K.S.; Zhu, J.K. Cell signaling during cold, drought, and salt stress. Plant Cell. 2002, 14, S165–S183. [Google Scholar] [PubMed]
- Cruz de Carvalho, M.H. Drought stress and reactive oxygen species: Production, scavenging and signaling. Plant Signal. Behav. 2008, 3, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Filippou, P.; Antoniou, C.; Fotopoulos, V. Effect of drought and rewatering on the cellular status and antioxidant response of Medicago truncatula plants. Plant Signal. Behav. 2011, 6, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Sofo, A.; Scopa, A.; Nuzzaci, M.; Vitti, A. Ascorbate peroxidase and catalase activities and their genetic regulation in plants subjected to drought and salinity stresses. Int. J. Mol. Sci. 2015, 16, 13561–13578. [Google Scholar] [CrossRef] [PubMed]
- Laloi, C.; Przybyla, D.; Apel, K. A genetic approach towrds elucidating the biological activity of different reactive oxygen species in Arabidopsis thalliana. J. Exp. Bot. 2006, 57, 1719–1724. [Google Scholar] [CrossRef] [PubMed]
- Marok, M.A.; Tarrago, L.; Ksas, B.; Henri, P.; Abrous-Belbachir, O.; Havaux, M.; Rey, P. A drought-sensitive barley variety displays oxidative stress and strongly increased contents in low-molecular weight antioxidant compounds during water deficit compared to a tolerant variety. J. Plant Physiol. 2013, 170, 633–645. [Google Scholar] [CrossRef] [PubMed]
- Loggini, B.; Scartazza, A.; Brugnoli, E.; Navari-Izzo, F. Antioxidative defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought. Plant Physiol. 1999, 119, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Hao, G.P.; Xing, Y.; Zhang, J.H. Role of nitric oxide dependence on nitric oxide synthase-like activity in the water stress signaling of maize seedling. J. Integr. Plant Biol. 2008, 50, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Chaki, M.; Valderrama, R.; Fernández-Ocaña, A.M.; Carreras, A.; Gómez-Rodríguez, M.V.; Pedrajas, J.R.; Begara-Morales, J.C.; Sánchez-Calvo, B.; Luque, F.; Leterrier, M.; et al. Mechanical wounding induces a nitrosative stress by down-regulation of GSNO reductase and an increase in S-nitrosothiols in sunflower (Helianthus annuus) seedlings. J. Exp. Bot. 2011, 62, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Benhar, M.; Forrester, M.T.; StamLer, J.S. Protein denitrosylation: Enzymatic mechanisms and cellular functions. Nat. Rev. Mol. Cell Biol. 2009, 10, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.I.; Hussain, A.; Yun, B.W.; Spoel, S.H.; Loake, G.J. GSNOR-mediated de-nitrosylation in the plant defence response. Plant Sci. 2011, 181, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Frungillo, L.; Skelly, M.J.; Loake, G.J.; Spoel, S.H.; Salgado, I. S-nitrosothiols regulate nitric oxide production and storage in plants through the nitrogen assimilation pathway. Nat. Commun. 2014, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Delledonne, M.; Zeier, J.; Marocco, A.; Lamb, C. Signal interactions between nitric oxide and reactive oxygen intermediates in the plant hypersensitive disease resistance response. Proc. Natl. Acad. Sci. USA 2001, 98, 13454–13459. [Google Scholar] [CrossRef] [PubMed]
- Lindermayr, C.; Durner, J. Interplay of reactive oxygen species and nitric oxide: Nitric oxide coordinates reactive oxygen species homeostasis. Plant Physiol. 2015, 167, 1209–1210. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ries, A.; Wu, K.; Yang, A.; Crawford, N.M. The Arabidopsis prohibitin gene PHB3 functions in nitric oxide-mediated responses and in hydrogen peroxide-induced nitric oxide accumulation. Plant Cell 2010, 22, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Yun, B.W.; Feechan, A.; Yin, M.; Saidi, N.B.; Le Bihan, T.; Yu, M.; Moore, J.W.; Kang, J.G.; Kwon, E.; Spoel, S.H.; et al. S-Nitrosylation of NADPH oxidase regulates cell death in plant immunity. Nature 2011, 478, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Fountain, J.C.; Khera, P.; Yang, L.; Nayak, S.N.; Scully, B.T.; Lee, R.D.; Chen, Z.Y.; Kemerait, R.C.; Varshney, R.K.; Guo, B. Resistance to Aspergillus flavus in maize and peanut: Molecular biology, breeding, environmental stress, and future perspectives. Crop J. 2015, 3, 229–237. [Google Scholar] [CrossRef]
- Baldwin, T.T.; Glenn, A.E. Evaluation of nitric oxide detoxifying flavohaemoglobin in the Fusarium verticillioides–maize interaction. Phytopathology 2010, 100, S10–S11. [Google Scholar]
- Baldwin, T.T. Maize Seedling Blight Caused by Fusarium verticillioides Involves Fumonisin B1 Mobility and Modulation of Nitric Oxide by the Denitrification Pathway. Ph.D. Thesis, University of Georgia, Athens, GA, USA, 2013. [Google Scholar]
- Ghanem, M.E.; Marrou, H.; Sinclair, T.R. Physiological phenotyping of plants for crop improvement. Trends Plant Sci. 2015, 20, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Cellier, F.; Conéjéro, G.; Breitler, J.C.; Casse, F. Molecular and physiological responses to water deficit in drought-tolerant and drought-sensitive lines of sunflower. Accumulation of dehydrin transcripts correlates with tolerance. Plant Physiol. 1998, 116, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Jabs, T.; Dietrich, R.A.; Dangl, J.L. Initiation of runaway cell death in an Arabidopsis mutant by extracellular superoxide. Science 1996, 273, 1853–1856. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Kumar, M.M.; Rao, M.A. In vitro and in vivo effects of phenolic antioxidants against cisplatin-induced nephrotoxicity. J. Biochem. 1999, 125, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Rook, G.A.; Steele, J.; Umar, S.; Dockrell, H.M. A simple method for the solubilization of reduced NBT, and its use as a colorimetric assay for activation of human macrophages by γ-interferon. J. Immunol. Methods 1985, 82, 161–167. [Google Scholar] [CrossRef]
- Mookerjee, A.; Basu, J.M.; Majumder, S.; Chatterjee, S.; Panda, G.S.; Dutta, P.; Pal, S; Mukherjee, P.; Efferth, T.; Roy, S. A novel copper complex induces ROS generation in doxorubicin resistant Ehrlich ascites carcinoma cells and increases activity of antioxidant enzymes in vital organs in vivo. BMC Cancer 2006, 6, 267. [Google Scholar] [CrossRef] [PubMed]
- Thordal-Christensen, H.; Zhang, Z.; Wei, Y.; Collinge, D.B. Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J. 1997, 11, 1187–1194. [Google Scholar] [CrossRef]
- Bindschedler, L.V.; Dewdney, J.; Blee, K.A.; Stone, J.M.; Asai, T.; Plotnikov, J.; Denoux, C.; Hayes, T.; Gerrish, C.; Davies, D.R.; et al. Peroxidase-dependent apoplastic oxidative burst in Arabidopsis required for pathogen resistance. Plant J. 2006, 47, 851–863. [Google Scholar] [CrossRef] [PubMed]
- Daudi, A.; Cheng, Z.; O’Brien, J.A.; Mammarella, N.; Khan, S.; Ausubel, F.M.; Bolwell, G.P. The apoplastic oxidative burst peroxidase in Arabidopsis is a major component of pattern-triggered immunity. Plant Cell 2012, 24, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Kotchoni, S.O.; Kuhns, C.; Ditzer, A.; Kirch, H.H.; Bartels, D. Overexpression of different aldehyde dehydrogenase genes in Arabidopsis thaliana confers tolerance to abiotic stress and protects plants against lipid peroxidation and oxidative stress. Plant Cell Environ. 2006, 29, 1033–1048. [Google Scholar] [CrossRef] [PubMed]
- Ramel, F.; Sulmon, C.; Bogard, M.; Couée, I.; Gouesbet, G. Differential patterns of reactive oxygen species and antioxidative mechanisms during atrazine injury and sucrose-induced tolerance in Arabidopsis thaliana plantlets. BMC Plant Biol. 2009, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Meth. Enzymol. 1984, 105, 121–126. [Google Scholar] [PubMed]
- Mur, L.A.; Mandon, J.; Cristescu, S.M.; Harren, F.J.; Prats, E. Methods of nitric oxide detection in plants: A commentary. Plant Sci. 2011, 181, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Zielonka, J.; Zielonka, M.; Sikora, A.; Adamus, J.; Joseph, J.; Hardy, M.; Ouari, O.; Dranka, B.P.; Kalyanaraman, B. Global profiling of reactive oxygen and nitrogen species in biological systems: High-throughput real-time analyses. J. Biol. Chem. 2012, 287, 2984–2995. [Google Scholar] [CrossRef] [PubMed]
- Stacklies, W.; Redestig, H.; Scholz, M.; Walther, D.; Selbig, J. pcaMethods—A bioconductor package providing PCA methods for incomplete data. Bioinformatics 2007, 23, 1164–1167. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 2001, 29, 1165–1188. [Google Scholar]
- Sharov, A.A.; Dudekula, D.B.; Ko, M.S.H. A web-based tool for principal component and significance analysis of microarray data. Bioinformatics 2005, 21, 2548–2549. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Cheng, T.; Wang, P.; Liu, W.; Xiao, J.; Yang, Y.; Shi, J. Salinity-induced changes in protein expression in the halophytic plant Nitraria sphaerocarpa. J. Proteom. 2012, 75, 5226–5243. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, L.; Fountain, J.C.; Wang, H.; Ni, X.; Ji, P.; Lee, R.D.; Kemerait, R.C.; Scully, B.T.; Guo, B. Stress Sensitivity Is Associated with Differential Accumulation of Reactive Oxygen and Nitrogen Species in Maize Genotypes with Contrasting Levels of Drought Tolerance. Int. J. Mol. Sci. 2015, 16, 24791-24819. https://doi.org/10.3390/ijms161024791
Yang L, Fountain JC, Wang H, Ni X, Ji P, Lee RD, Kemerait RC, Scully BT, Guo B. Stress Sensitivity Is Associated with Differential Accumulation of Reactive Oxygen and Nitrogen Species in Maize Genotypes with Contrasting Levels of Drought Tolerance. International Journal of Molecular Sciences. 2015; 16(10):24791-24819. https://doi.org/10.3390/ijms161024791
Chicago/Turabian StyleYang, Liming, Jake C. Fountain, Hui Wang, Xinzhi Ni, Pingsheng Ji, Robert D. Lee, Robert C. Kemerait, Brian T. Scully, and Baozhu Guo. 2015. "Stress Sensitivity Is Associated with Differential Accumulation of Reactive Oxygen and Nitrogen Species in Maize Genotypes with Contrasting Levels of Drought Tolerance" International Journal of Molecular Sciences 16, no. 10: 24791-24819. https://doi.org/10.3390/ijms161024791
APA StyleYang, L., Fountain, J. C., Wang, H., Ni, X., Ji, P., Lee, R. D., Kemerait, R. C., Scully, B. T., & Guo, B. (2015). Stress Sensitivity Is Associated with Differential Accumulation of Reactive Oxygen and Nitrogen Species in Maize Genotypes with Contrasting Levels of Drought Tolerance. International Journal of Molecular Sciences, 16(10), 24791-24819. https://doi.org/10.3390/ijms161024791