Prediction of Partition Coefficients of Organic Compounds between SPME/PDMS and Aqueous Solution
Abstract
:1. Introduction
2. Results and Discussion
2.1 Correlation Analysis
2.2 Empirical Models
3. Method
3.1. PDMS-Water Partition Coefficients
3.2. Physicochemical Descriptors
4. Conclusions
Conflicts of Interest
References
- Pawliszyn, J. Applications of Solid Phase Microextraction; Royal Society of Chemistry: Cornwall, UK, 1999. [Google Scholar]
- Chao, K.P.; Wang, V.S.; Yang, H.W.; Wang, C.I. Estimation of effective diffusion coefficients for benzene and toluene in PDMS for direct solid phase microextraction. Polym. Test 2011, 30, 501–508. [Google Scholar]
- Mahugo Santana, C.; Sosa Ferrera, Z.; Esther Torres Padrón, M.; Juan Santana Rodríguez, J. Methodologies for the extraction of phenolic compounds from environmental samples: New approaches. Molecules 2009, 14, 298–320. [Google Scholar]
- Benhabib, K.; Mimanne, G. Optimized parameters of SMPE analysis for atrazine and its application to measure speciation. Appl. Clay Sci 2014, 87, 260–264. [Google Scholar]
- Benhabib, K.; ter Laak, T.L.; van Leeuwen, H.P. Steady-state diffusion regime in solid-phase microextraction kinetics. Anal. Chim. Acta 2008, 609, 113–119. [Google Scholar]
- Lord, H.; Pawliszyn, J. Evolution of solid-phase microextraction technology. J. Chromatogr. A 2000, 885, 153–193. [Google Scholar]
- LaGrega, M.D.; Buckingham, P.L.; Evans, J.C. Hazardous Waste Management; McGraw-Hill Inc.: New York, NY, USA, 1994. [Google Scholar]
- Stenzel, A.; Endo, S.; Goss, K.W. Measurements and predictions of hexadecane/air partition coefficients for 387 environmentally relevant compounds. J. Chromatogr. A 2012, 1220, 132–142. [Google Scholar]
- Zhao, H.; Zhang, Q.; Chen, J.; Xue, Z.; Liang, X. Prediction of octanol-air partition coefficients of semivolatile organic compounds based on molecular connectivity index. Chemosphere 2005, 59, 1421–1426. [Google Scholar]
- Hong, H.; Wang, L.; Han, S. Prediction adsorption coefficients (Koc) for aromatic compounds by HPLC retention factors (K). Chemosphere 1996, 32, 343–351. [Google Scholar]
- Nirmalakhandan, N.N.; Speece, R.E. Prediction of aqueous solubility of organic chemicals based on molecular structure. Environ. Sci. Technol 1988, 22, 328–338. [Google Scholar]
- Nirmalakhandan, N.N.; Speece, R.E. QSAR model for predicting Henry’s constant. Environ. Sci. Technol 1988, 22, 1349–1357. [Google Scholar]
- Pawliszyn, J. Solid Phase Microextraction—Theory and Practice; Wiley-VCH: New York, NY, USA, 1997. [Google Scholar]
- Xia, X.R.; Baynes, N.A.; Monteiro-Riviere, N.A.; Riviere, J.E. A system coefficient approach for quantitative assessment of the solvent effects on membrane absorption from chemical mixtures. SAR QSAR Environ. Res 2007, 18, 579–593. [Google Scholar]
- Silbey, R.J.; Alberty, R.A.; Bawendi, M.G. Physical Chemistry, 4th ed.; Wiley: New York, NY, USA, 2005. [Google Scholar]
- DiFilippo, E.L.; Eganhouse, R.P. Assessment of PDMS-water partition coefficients: implications for passive environmental sampling of hydrophobic organic compounds. Environ. Sci. Technol 2010, 44, 6917–6925. [Google Scholar]
- Avila, M.A.S.; Breiter, R. Estimating the DMS-coated SPME-fibre/water- and fibre/gas-partition coefficients of chlorinated ethenes by headspace-SPME. Chromatographia 2007, 66, 369–376. [Google Scholar]
- Kier, L.B.; Hall, L.H. Molecular Connectivity in Structure—Activity Analysis; Wiley: Chichester, UK, 1986. [Google Scholar]
- Endo, S.; Droge, S.T.J.; Goss, K.U. Polyparameter linear free energy models for polyacrylate fiber-water partition coefficients to evaluate the efficiency of solid-phase microextraction. Anal. Chem 2011, 83, 1394–1400. [Google Scholar]
- Sprunger, L.; Proctor, A.; Acree, W.E., Jr.; Abrahamb, M.H. Characterization of the sorption of gaseous and organic solutes onto polydimethyl siloxane solid-phase microextraction surfaces using the Abraham model. J. Chromatogr. A 2007, 1175, 162–173. [Google Scholar]
- Goss, K.U. Predicting equilibrium sorption of neutral organic chemicals into various polymeric sorbents with COSMO-RS. Anal. Chem 2011, 83, 5304–5308. [Google Scholar]
- Horvath, A.L. Halogenated Hydrocarbons; Dekker: New York, NY, USA, 1982. [Google Scholar]
- Randic, M. On characterization of molecular branching. J. Am. Chem. Soc 1975, 97, 6609–6615. [Google Scholar]
- Hansch, C.; Vittoria, A.; Silipo, C.; Jow, P.Y.C. Partition coefficients and the structure-activity relationship of the anesthetic gases. J. Med. Chem 1975, 18, 546–548. [Google Scholar]

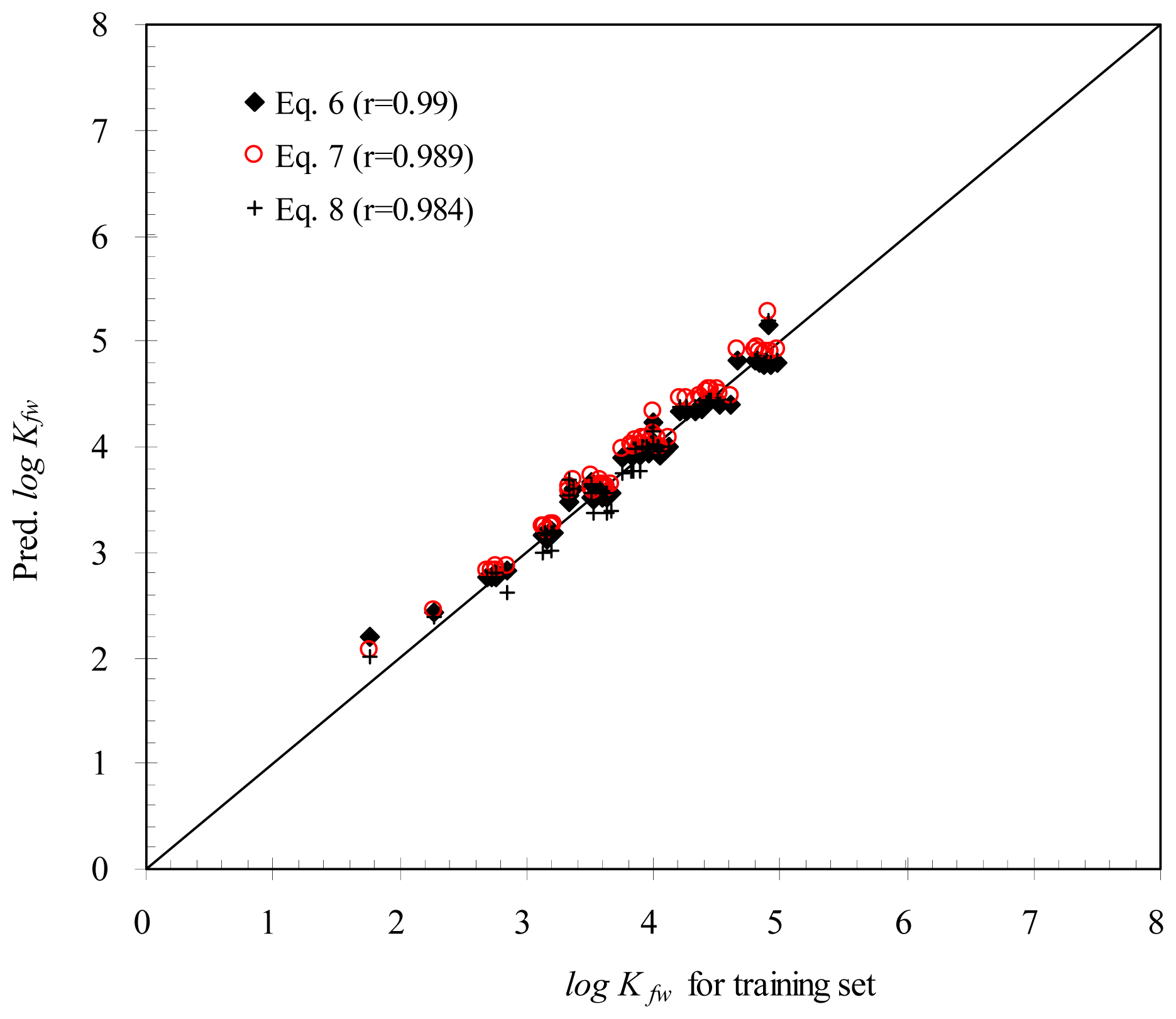
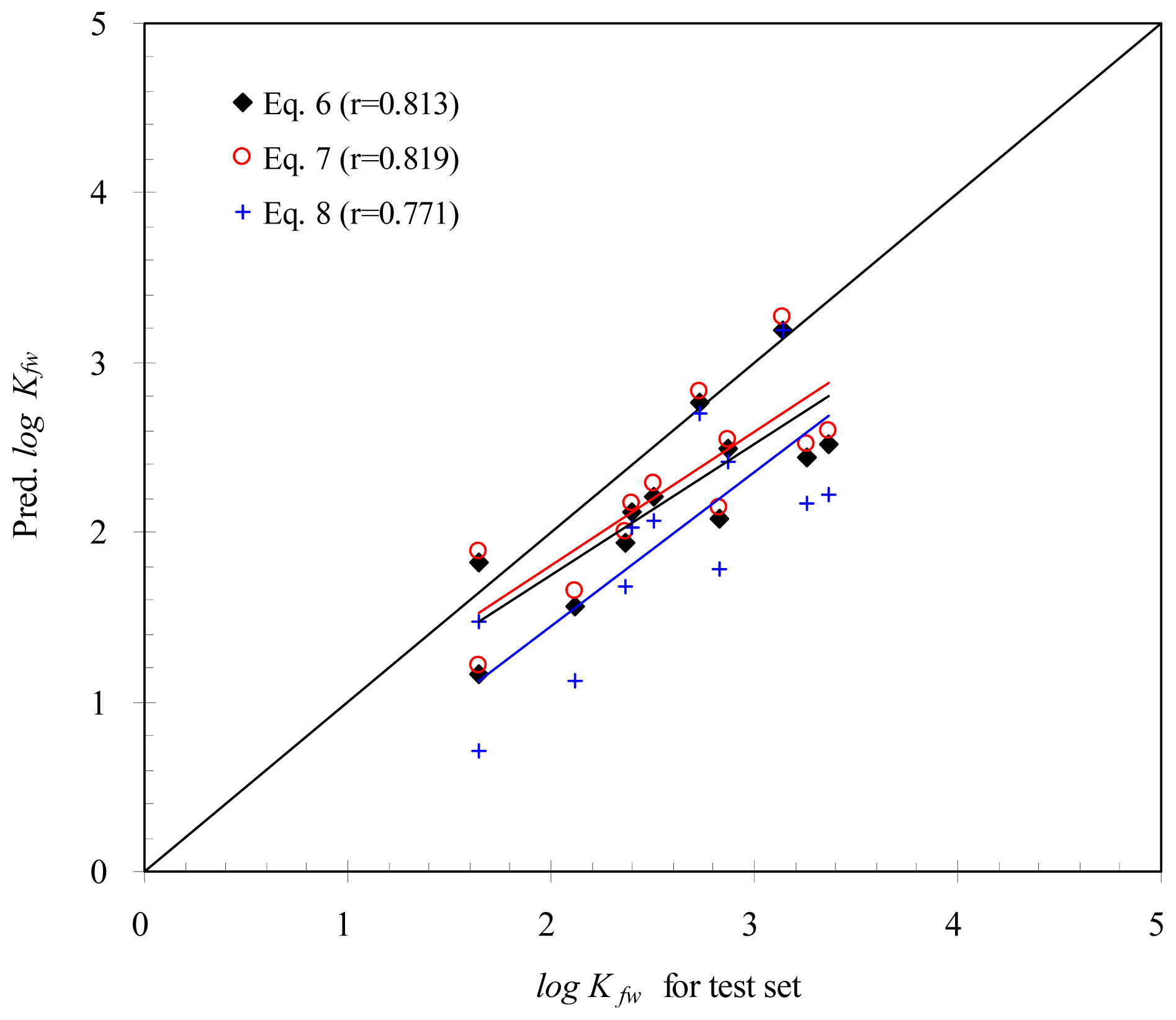
| Descriptor | Correlation |
|---|---|
| Φ | log Kfw = 1.933 + 1.026 Φ (r = 0.977, p < 0.05) |
| log Kow | log Kfw = 0.319 + 0.881 log Kow (r = 0.774, p < 0.05) |
| 1χ | log Kfw = 0.750 + 0.862 1χ (r = 0.743, p < 0.05) |
| I | log Kfw = 4.144 − 0.846 I (r = 0.583, p < 0.05) |
| WS | log Kfw = 3.936 − 0.002 WS (r = 0.559, p < 0.05) |
| MW | log Kfw = 1.859 + 0.017 MW (r = 0.474, p < 0.05) |
| Compound | Kfw | 1χ | Φ | I | MW † | WS ‡ | log Kow |
|---|---|---|---|---|---|---|---|
| Benzene | 58 | 2 | 0.165 | 1 | 78 | 1790 | 2.13 |
| Toluene | 189 | 2.411 | 0.502 | 1 | 92 | 526 | 2.73 |
| o-Xylene | 485 | 2.827 | 0.839 | 1 | 106 | 178 | 3.12 |
| m-Xylene | 533 | 2.821 | 0.839 | 1 | 106 | 161 | 3.2 |
| p-Xylene | 564 | 2.821 | 0.839 | 1 | 106 | 162 | 3.15 |
| Ethylbenzene | 566 | 2.971 | 0.839 | 1 | 106 | 169 | 3.15 |
| Cyclopentane | 712 | 2.5 | 0.733 | 0 | 70 | 156 | 3.01 |
| Methylcyclopentane | 1356 | 2.894 | 1.07 | 0 | 84 | 42 | 3.37 |
| Isopropylbenzene | 1412 | 3.354 | 1.176 | 1 | 120 | 61.3 | 3.66 |
| 1,3,5-Trimethylbenzene | 1451 | 3.232 | 1.176 | 1 | 120 | 48.2 | 3.42 |
| 1-Methyl-4-Ethylbenzene | 1581 | 3.382 | 1.176 | 1 | 120 | 94.9 | 3.63 |
| Cyclohexane | 1592 | 3 | 1.07 | 0 | 84 | 55 | 3.44 |
| n-Propylbenzene | 1664 | 3.471 | 1.176 | 1 | 120 | 52.2 | 3.69 |
| 1,2,4-Trimethylbenzene | 2183 | 3.238 | 1.76 | 1 | 120 | 57 | 3.63 |
| tert-Butylbenzene | 2185 | 3.661 | 1.513 | 1 | 134 | 29.5 | 4.11 |
| 2,3-Dimethylbutane | 2359 | 2.643 | 1.782 | 0 | 86 | 22.5 | 3.42 |
| 2-Methylpentane | 3224 | 2.77 | 1.782 | 0 | 86 | 14 | 3.21 |
| 3-Methylpentane | 3270 | 2.808 | 1.782 | 0 | 86 | 17.9 | 3.6 |
| 1-Methyl-3-Isopropylbenzene | 3284 | 3.765 | 1.513 | 1 | 134 | 42.5 | 4.5 |
| trans-1,2-Dimethylcyclopentane | 3372 | 3.207 | 1.407 | 0 | 98 | 33.9 | 3.52 |
| 1-Methyl-3-n-Propylbenzene | 3772 | 3.882 | 1.513 | 1 | 134 | 9.09 | 4.67 |
| n-Butylbenzene | 3872 | 3.971 | 1.513 | 1 | 134 | 11.8 | 4.38 |
| 1,2-Dimethyl-4-Ethylbenzene | 3984 | 3.8 | 1.513 | 1 | 134 | 12.7 | 4.5 |
| sec-Butylbenzene | 4011 | 3.892 | 1.513 | 1 | 134 | 17.6 | 4.57 |
| Isobutylbenzene | 4197 | 3.827 | 1.513 | 1 | 134 | 10.1 | 4.68 |
| cis-1,3-Dimethylcyclopentane | 4289 | 3.288 | 1.407 | 0 | 98 | 33.9 | 3.52 |
| 1,3-Dimethyl-2-Ethylbenzene | 4345 | 3.805 | 1.513 | 1 | 134 | 19.6 | 4.28 |
| Methylcyclohexane | 4657 | 3.394 | 1.407 | 0 | 98 | 14 | 3.61 |
| cis-trans-cis-1,2,4-Trimethylcyclopentane | 5621 | 3.698 | 1.744 | 0 | 112 | 14.8 | 3.94 |
| trans-1,2-Dimethylcyclohexane | 6638 | 3.805 | 1.744 | 0 | 112 | 5.2 | 4.01 |
| 1-Ethyl-1-methylcyclopentane | 6831 | 3.768 | 1.744 | 0 | 122 | 11.6 | 4.05 |
| 2,2-Dimethylpentane | 7349 | 3.061 | 2.119 | 0 | 100 | 4.4 | 3.67 |
| cis-1,2-Dimethylcyclohexane | 7826 | 3.805 | 1.744 | 0 | 112 | 6 | 4.01 |
| n-Pentylbenzene | 8195 | 4.471 | 1.85 | 1 | 148 | 3.37 | 4.9 |
| 2,4-Dimethylpentane | 8989 | 3.126 | 2.119 | 0 | 100 | 5.5 | 3.63 |
| 2-Methylbutylbenzene | 9099 | 4.365 | 1.85 | 1 | 148 | 12.7 | 4.43 |
| 2,2,3-Trimethylbutane | 9802 | 3.944 | 2.119 | 0 | 100 | 28.9 | 3.59 |
| 2-Methylhexane | 10,202 | 3.27 | 2.119 | 0 | 100 | 2.54 | 3.71 |
| 3,3-Dimethylpentane | 10,963 | 3.121 | 2.119 | 0 | 100 | 5.92 | 3.67 |
| 3-Methylhexane | 11,146 | 2.9 | 2.119 | 0 | 100 | 4.95 | 3.71 |
| 2,3-Dimethylpentane | 13,074 | 3.181 | 2.119 | 0 | 100 | 5.25 | 3.63 |
| 1,2,4-Triethylbenzene | 16,253 | 4.92 | 2.187 | 1 | 162 | 2.9 | 5.11 |
| 1,3,5-Triethylbenzene | 18,517 | 4.914 | 2.187 | 1 | 162 | 0.014 | 5.11 |
| 2,2,3-Trimethylpentane | 21,205 | 3.481 | 2.456 | 0 | 114 | 2.4 | 4.09 |
| 2,5-Dimethylhexane | 23,519 | 3.626 | 2.456 | 0 | 114 | 9.2 | 4.12 |
| 2,2-Dimethylhexane | 24,504 | 3.561 | 2.456 | 0 | 114 | 0.2 | 4.16 |
| 2-Methylheptane | 25,806 | 3.77 | 2.456 | 0 | 114 | 7.97 | 4.2 |
| 4-Methylheptane | 27,274 | 3.808 | 2.456 | 0 | 114 | 7.97 | 4.2 |
| 3-Ethylhexane | 28,370 | 3.846 | 2.456 | 0 | 114 | 7.97 | 4.2 |
| 3-Methylheptane | 31,856 | 3.808 | 2.456 | 0 | 114 | 0.792 | 4.2 |
| 2,3-Dimethylhexane | 33,749 | 3.681 | 2.456 | 0 | 114 | 9.2 | 4.12 |
| 2,4-Dimethylhexane | 41,133 | 3.664 | 2.456 | 0 | 114 | 9.87 | 4.12 |
| 2-Methyloctane | 45,267 | 4.27 | 2.793 | 0 | 128 | 2.87 | 4.69 |
| 3,3-Diethylpentane | 63,718 | 4.243 | 2.793 | 0 | 128 | 2.9 | 4.65 |
| 3-Methyloctane | 66,682 | 4.308 | 2.793 | 0 | 128 | 2.87 | 4.69 |
| 2,3-Dimethylheptane | 68,675 | 4.181 | 2.793 | 0 | 128 | 3.37 | 4.61 |
| 3,3-Dimethylheptane | 76,013 | 4.121 | 2.793 | 0 | 128 | 2.9 | 4.65 |
| 3,5-Dimethylheptane | 78,829 | 4.202 | 2.793 | 0 | 128 | 3.11 | 4.61 |
| 3,3-Dimethyloctane | 82,430 | 4.621 | 3.13 | 0 | 142 | 2.9 | 5.14 |
| 2,5-Dimethylheptane | 84,142 | 4.164 | 2.793 | 0 | 128 | 3.11 | 4.61 |
| 3,4-Dimethylheptane | 93,292 | 4.219 | 2.793 | 0 | 128 | 3.11 | 4.61 |
| Compound | Kfw | 1χ | Φ | I | WS ‡ |
|---|---|---|---|---|---|
| Benzyl alcohol | 0.447 | 2.580 | −0.323 | 1 | 4.29 × 104 |
| 4-Fluorophenol | 0.525 | 2.234 | −1.110 | 1 | 1.25 × 104 |
| Phenol | 0.661 | 2.134 | −0.540 | 1 | 8.28 × 104 |
| m-Cresol | 0.933 | 2.545 | −0.323 | 1 | 2.27 × 104 |
| Phenethyl alcohol | 1.318 | 3.081 | 0.014 | 1 | 2 × 104 |
| 3-Methylbenzyl alcohol | 1.479 | 2.991 | 0.014 | 1 | 5 × 104 |
| 3-Chlorophenol | 2.042 | 2.647 | −0.727 | 1 | 2.6 × 104 |
| 3,5-Dimethylphenol | 2.630 | 2.956 | 0.014 | 1 | 4.88 × 103 |
| 3-Bromophenol | 2.884 | 3.026 | −0.762 | 1 | 2.4 × 104 |
| 4-Ethylphenol | 3.981 | 3.106 | 0.014 | 1 | 4.9 × 103 |
| 4-Chloroaniline | 6.918 | 2.676 | −0.022 | 1 | 3.9 × 103 |
| Phenyl acetate | 7.244 | 3.023 | −1.430 | 1 | 4.64 × 103 |
| Benzonitrile | 10.96 | 2.384 | 0.753 | 1 | 2 × 103 |
| Acetophenone | 10.96 | 2.865 | −0.605 | 1 | 6.13 × 103 |
| 4-Chloroacetophenone | 43.7 | 3.342 | −0.672 | 1 | 111 |
| Methyl benzoate | 44.7 | 2.977 | −1.430 | 1 | 2.1 × 103 |
| Ethylbenzoate | 131.8 | 3.565 | −1.093 | 1 | 720 |
| 4-Chloroanisole | 234.4 | 3.036 | −0.390 | 1 | 237 |
| Chlorobenzene | 251.2 | 2.477 | 0.098 | 1 | 498 |
| Bromobenzene | 323.6 | 2.891 | 0.063 | 1 | 410 |
| Iodobenzene | 537.1 | 3.161 | 0.692 | 1 | 340 |
| Naphthalene | 676.1 | 3.405 | −0.344 | 1 | 31 |
| 4-Chlorotoluene | 741.3 | 3.095 | 0.435 | 1 | 106 |
| Propylbenzene | 1380 | 3.471 | 1.176 | 1 | 52.2 |
| 1-Methylnaphthalene | 1819 | 3.821 | −0.007 | 1 | 25.8 |
| Biphenyl | 2344 | 4.071 | −0.007 | 1 | 6.9 |
| Atom/bond | Contribution factor | Atom/bond | Contribution factor |
|---|---|---|---|
| Carbon | 0.577 | Iodine | 0.407 |
| Hydrogen * | −0.120 | Fluorine | −0.570 |
| Oxygen | −0.825 | Cycle | −0.952 |
| Hydroxyl | −3.701 | Double bond | −0.859 |
| Chlorine | −0.187 | Triple bond | −0.109 |
| Bromine | −0.222 | - | - |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chao, K.-P.; Lu, Y.-T.; Yang, H.-W. Prediction of Partition Coefficients of Organic Compounds between SPME/PDMS and Aqueous Solution. Int. J. Mol. Sci. 2014, 15, 2585-2595. https://doi.org/10.3390/ijms15022585
Chao K-P, Lu Y-T, Yang H-W. Prediction of Partition Coefficients of Organic Compounds between SPME/PDMS and Aqueous Solution. International Journal of Molecular Sciences. 2014; 15(2):2585-2595. https://doi.org/10.3390/ijms15022585
Chicago/Turabian StyleChao, Keh-Ping, Yu-Ting Lu, and Hsiu-Wen Yang. 2014. "Prediction of Partition Coefficients of Organic Compounds between SPME/PDMS and Aqueous Solution" International Journal of Molecular Sciences 15, no. 2: 2585-2595. https://doi.org/10.3390/ijms15022585
APA StyleChao, K.-P., Lu, Y.-T., & Yang, H.-W. (2014). Prediction of Partition Coefficients of Organic Compounds between SPME/PDMS and Aqueous Solution. International Journal of Molecular Sciences, 15(2), 2585-2595. https://doi.org/10.3390/ijms15022585




