Lipoprotein Lipase, Tissue Expression and Effects on Genes Related to Fatty Acid Synthesis in Goat Mammary Epithelial Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cloning and Sequence Analysis of Lipoprotein Lipase (LPL)
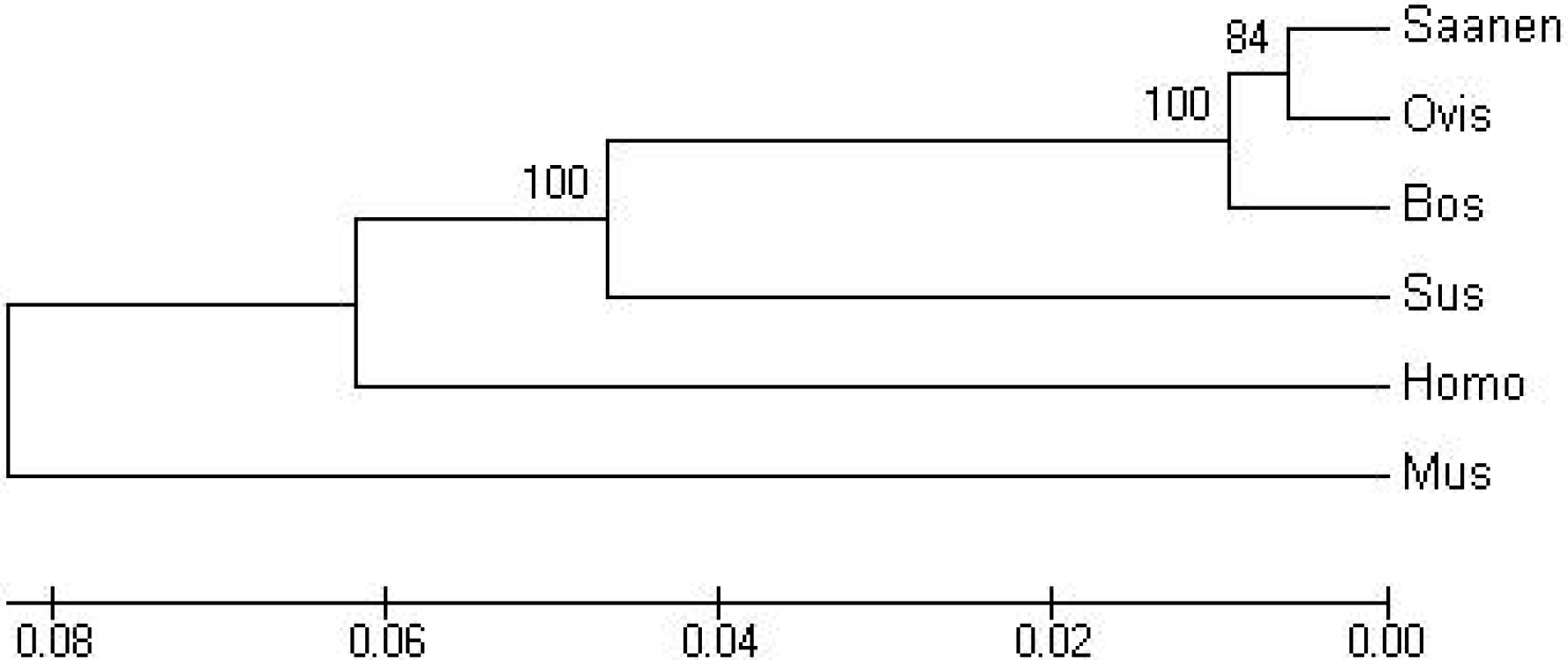
2.2. Phylogenetic Analysis of LPL Protein
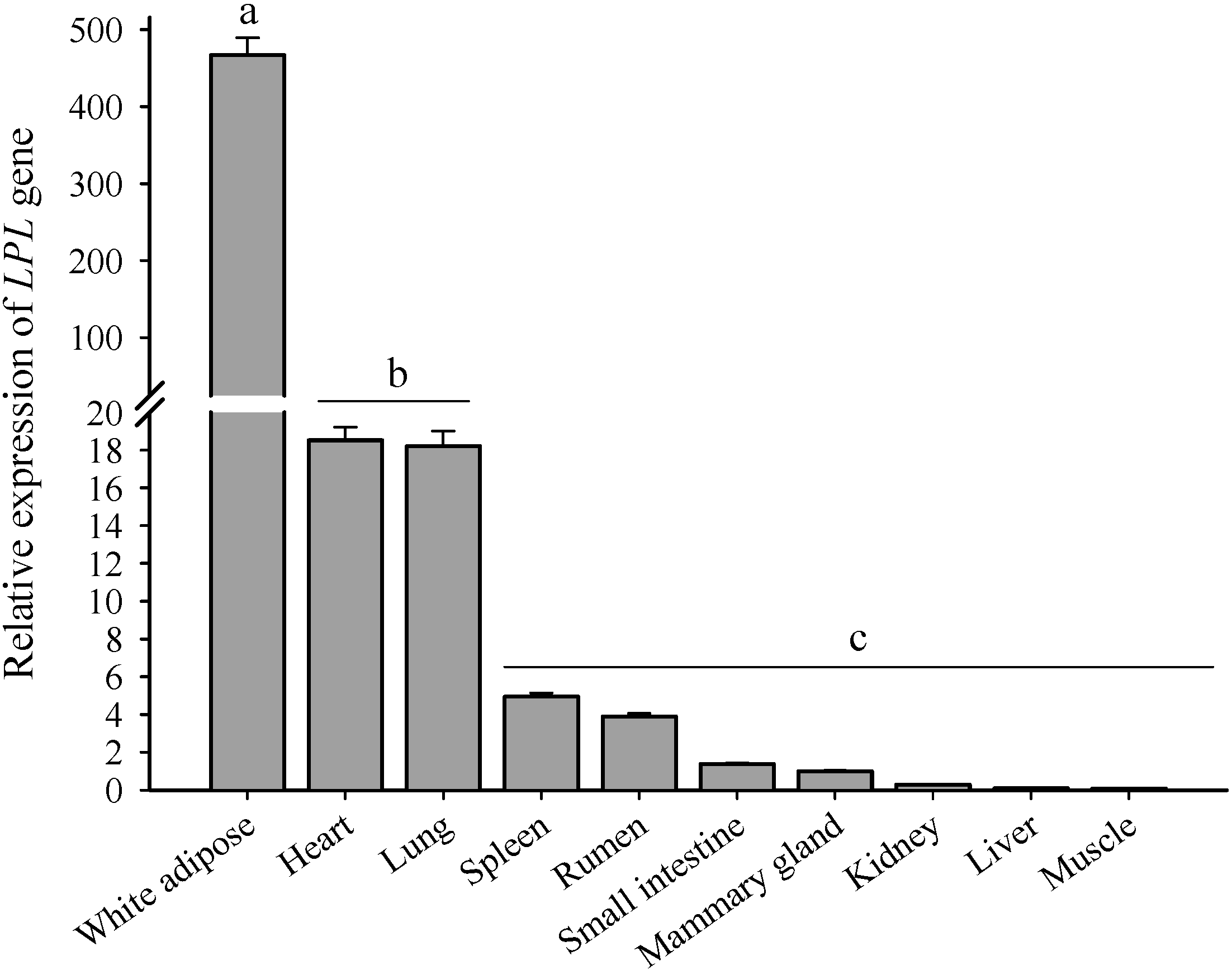
2.3. The Expression Profiles of LPL in 10 Tissues and Different Stages of Lactation

2.4. Effects of LPL Knockdown and Orlistat on mRNA Expression of Genes Related to Milk Fat Synthesis in Goat Mammary Epithelial Cells (GMEC)

3. Experimental Section
3.1. Animals and Samples Collection
3.2. Cloning, Sequencing Analysis and Tissue Expression of LPL
3.2.1. Total RNA Extraction, cDNA Synthesis and Quantification
3.2.2. Full-Length Cloning of LPL cDNA
3.2.3. Bioinformatics Analysis of LPL
3.3. Expression and Statistical Analysis
3.4. Effects of Orlistat (Tetrahydrolipstain, THL) and LPL Knockdown on mRNA Expression of LPL and Genes Related to Milk Fat Synthesis in GMEC
3.4.1. Adenovirus Establishment
3.4.2. Cell Culture and Treatments
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Abbreviation
| ACACA | acetyl-coenzyme A carboxylase alpha |
| ANOVA | analysis of variance |
| bp | base pair(s) |
| BLAST | basic local alignment search tool |
| bMEC | bovine mammary epithelial cells |
| cDNA | DNA complimentary to RNA |
| CDS | coding sequence |
| CTR | control |
| DMEM | Dulbecco’s modified Eagle medium |
| EC | enzyme class |
| EGF | epidermal growth factor |
| FASN | fatty acid synthase |
| FBS | fetal bovine serum |
| FFA | free fatty acids |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| GMEC | goat mammary epithelial cells |
| FFAR3 | free fatty acid receptor 3 |
| LIPE | lipase, hormone-sensitive |
| kDa | kilo Dalton(s) |
| LCFA | long chain fatty acid |
| LPL | Lipoprotein lipase |
| MG | mammary gland |
| MOI | multiplicity of infection |
| MW | molecular weight |
| NC | negative control |
| NCBI | national center for biotechnology information |
| NEFA | non-esterified fatty acids |
| NJ | neighbor-joining |
| nt | nucleotide |
| ORF | open reading frame |
| PBS | phosphate buffered saline |
| pI | isoelectric point |
| PPARG | peroxisome proliferator-activated receptor-γ |
| RACE | rapid amplification of cDNA ends |
| RPS9 | ribosomal protein S9 |
| RT-PCR | reverse transcription polymerase chain reaction |
| SCFAs | short-chain fatty acids |
| SD | standard deviation |
| shRNA | short hairpin RNA |
| SREBF1 | sterol regulatory element binding factor1 |
| TAG | triacylgleryerol |
| TG | triglyceride |
| UTR | un-translated region(s) |
| UXT | Ubiquitously expressed transcript protein |
| VLDL | very-low density lipoproteins |
Conflicts of Interest
References
- Stins, M.F.; Maxfield, F.R.; Goldberg, I.J. Polarized binding of lipoprotein lipase to endothelial cells. Implications for its physiological actions. Arterioscler. Thromb. 1992, 12, 1437–1446. [Google Scholar] [CrossRef] [PubMed]
- Bernard, L.; Leroux, C.; Chilliard, Y. Expression and nutritional regulation of lipogenic genes in the ruminant lactating mammary gland. Adv. Exp. Med. Biol. 2008, 606, 67–108. [Google Scholar] [PubMed]
- Fielding, B.A.; Frayn, K.N. Lipoprotein lipase and the disposition of dietary fatty acids. Br. J. Nutr. 1998, 80, 495–502. [Google Scholar] [PubMed]
- Mead, J.R.; Irvine, S.A.; Ramji, D.P. Lipoprotein lipase: Structure, function, regulation, and role in disease. J. Mol. Med. 2002, 80, 753–769. [Google Scholar] [CrossRef] [PubMed]
- Jensen, D.R.; Gavigan, S.; Sawicki, V.; Witsell, D.L.; Eckel, R.H.; Neville, M.C. Regulation of lipoprotein lipase activity and mrna in the mammary gland of the lactating mouse. Biochem. J. 1994, 298, 321–327. [Google Scholar] [PubMed]
- Finucane, K.A.; McFadden, T.B.; Bond, J.P.; Kennelly, J.J.; Zhao, F.Q. Onset of lactation in the bovine mammary gland: Gene expression profiling indicates a strong inhibition of gene expression in cell proliferation. Funct. Integr. Genomics 2008, 8, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, M.; Leroux, C.; Faulconnier, Y.; Hocquette, J.F.; Bocquier, F.; Martin, P.; Chilliard, Y. Lipoprotein lipase activity and mrna are up-regulated by refeeding in adipose tissue and cardiac muscle of sheep. J. Nutr. 2000, 130, 749–756. [Google Scholar] [PubMed]
- Eckel, R.H.; Yost, T.J. Weight reduction increases adipose tissue lipoprotein lipase responsiveness in obese women. J. Clin. Investig. 1987, 80, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Chilliard, Y.; Ferlay, A.; Rouel, J.; Lamberet, G. A review of nutritional and physiological factors affecting goat milk lipid synthesis and lipolysis. J. Dairy Sci. 2003, 86, 1751–1770. [Google Scholar] [CrossRef] [PubMed]
- Kern, P.A.; Mandic, A.; Eckel, R.H. Regulation of lipoprotein lipase by glucose in primary cultures of isolated human adipocytes. Relevance to hypertriglyceridemia of diabetes. Diabetes 1987, 36, 1238–1245. [Google Scholar] [CrossRef] [PubMed]
- Jensen, D.R.; Bessesen, D.H.; Etienne, J.; Eckel, R.H.; Neville, M.C. Distribution and source of lipoprotein lipase in mouse mammary gland. J. Lipid Res. 1991, 32, 733–742. [Google Scholar]
- Moioli, B.; D’Andrea, M.; Pilla, F. Candidate genes affecting sheep and goat milk quality. Small Rumin. Res. 2007, 68, 179–192. [Google Scholar]
- Merkel, M.; Eckel, R.H.; Goldberg, I.J. Lipoprotein lipase: Genetics, lipid uptake, and regulation. J. Lipid Res. 2002, 43, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Lookene, A.; Skottova, N.; Olivecrona, G. Interactions of lipoprotein lipase with the active-site inhibitor tetrahydrolipstatin (Orlistat). Eur. J. Biochem. 1994, 222, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Burn, P. Lipid metabolic enzymes: Emerging drug targets for the treatment of obesity. Nat. Rev. Drug Discov. 2004, 3, 695–710. [Google Scholar] [CrossRef] [PubMed]
- Zhi, J.G.; Mulligan, T.E.; Hauptman, J.B. Long-term systemic exposure of Orlistat, a lipase inhibitor, and its metabolites in obese patients. J. Clin. Pharmacol. 1999, 39, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Zhang, Y.; Yin, Y.; Xu, F.; Gao, B.; Shi, Q.; Zhu, C.; Li, W.; Li, B. Cloning of Xuhuai goat lipoprotein lipase gene and the preparation of transgenic sheep. Mol. Biol. Rep. 2012, 39, 8439–8446. [Google Scholar] [CrossRef] [PubMed]
- Bionaz, M.; Loor, J.J. Gene networks driving bovine milk fat synthesis during the lactation cycle. BMC Genomics 2008, 9, 366. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Li, W.H.; Gouy, M.; Sharp, P.M.; O’HUigin, C.; Yang, Y.W. Molecular phylogeny of rodentia, lagomorpha, primates, artiodactyla, and carnivora and molecular clocks. Proc. Natl. Acad. Sci. USA 1990, 87, 6703–6707. [Google Scholar] [CrossRef] [PubMed]
- Camps, L.; Reina, M.; Llobera, M.; Bengtsson-Olivecrona, G.; Olivecrona, T.; Vilaro, S. Lipoprotein lipase in lungs, spleen, and liver: Synthesis and distribution. J. Lipid Res. 1991, 32, 1877–1888. [Google Scholar] [PubMed]
- Hocquette, J.F.; Graulet, B.; Olivecrona, T. Lipoprotein lipase activity and mrna levels in bovine tissues. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1998, 121, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Hamosh, M.; Yeager, M., Jr.; Shechter, Y.; Hamosh, P. Lipoprotein lipase in rat lung. Effect of dexamethasone. Biochim. Biophys. Acta 1976, 431, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Ranganathan, G.; Ong, J.M.; Yukht, A.; Saghizadeh, M.; Simsolo, R.B.; Pauer, A.; Kern, P.A. Tissue-specific expression of human lipoprotein lipase. Effect of the 3'-untranslated region on translation. J. Biol. Chem. 1995, 270, 7149–7155. [Google Scholar] [CrossRef] [PubMed]
- Faucon, F.; Rebours, E.; Bevilacqua, C.; Helbling, J.C.; Aubert, J.; Makhzami, S.; Dhorne-Pollet, S.; Robin, S.; Martin, P. Terminal differentiation of goat mammary tissue during pregnancy requires the expression of genes involved in immune functions. Physiol. Genomics 2009, 40, 61–82. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.C.; McManaman, J.L.; Phang, T.; Russell, T.; Kominsky, D.J.; Serkova, N.J.; Stein, T.; Anderson, S.M.; Neville, M.C. Metabolic regulation in the lactating mammary gland: A lipid synthesizing machine. Physiol. Genomics 2007, 28, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Cundiff, J.K.; Maria, S.D.; McMahon, R.J.; Woo, J.G.; Davidson, B.S.; Morrow, A.L. Time-resolved quantitative analysis of human milk proteome reveals developing milk and mammary-gland functions during the first year of lactation. FASEB J. 2013, 27, 628-1. [Google Scholar]
- Bell, A.W. Regulation of organic nutrient metabolism during transition form late pregnancy to early lactation. J. Anim. Sci. 1995, 73, 2804–2819. [Google Scholar] [PubMed]
- Thompson, G.E. Prolactin and the onset of mammary extraction of plasma triacylglycerols during lactogenesis in the goat. Comp. Biochem. Physiol. Comp. Physiol. 1992, 102, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Bauman, D.E.; Currie, W.B. Partitioning of nutrients during pregnancy and lactation: A review of mechanisms involving homeostasis and homeorhesis. J. Dairy Sci. 1980, 63, 1514–1529. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.O.; Grunnet, I.; Knudsen, J. Triacylglycerol synthesis in goat mammary gland. The effect of ATP, Mg2+ and glycerol 3-phosphate on the esterification of fatty acids synthesized de novo. Biochem. J. 1984, 220, 513–519. [Google Scholar] [PubMed]
- Hansen, H.O.; Grunnet, I.; Knudsen, J. Triacylglycerol synthesis in goat mammary gland. Factors influencing the esterification of fatty acids synthesized de novo. Biochem. J. 1984, 220, 521–527. [Google Scholar] [PubMed]
- Hansen, H.O.; Knudsen, J. Effect of exogenous long-chain fatty acids on individual fatty acid synthesis by dispersed ruminant mammary gland cells. J. Dairy Sci. 1987, 70, 1350–1354. [Google Scholar] [CrossRef] [PubMed]
- Yonezawa, T.; Haga, S.; Kobayashi, Y.; Katoh, K.; Obara, Y. Short-chain fatty acid signaling pathways in bovine mammary epithelial cells. Regul. Pept. 2009, 153, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Schoonjans, K.; Gelman, L.; Haby, C.; Briggs, M.; Auwerx, J. Induction of LPL gene expression by sterols is mediated by a sterol regulatory element and is independent of the presence of multiple E boxes. J. Mol. Biol. 2000, 304, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Schoonjans, K.; Peinado-Onsurbe, J.; Lefebvre, A.M.; Heyman, R.A.; Briggs, M.; Deeb, S.; Staels, B.; Auwerx, J. Pparalpha and ppargamma activators direct a distinct tissue-specific transcriptional response via a PPRE in the lipoprotein lipase gene. EMBO J. 1996, 15, 5336–5348. [Google Scholar] [PubMed]
- Neville, M.C.; Waxman, L.J.; Jensen, D.; Eckel, R.H. Lipoprotein lipase in human milk: Compartmentalization and effect of fasting, insulin, and glucose. J. Lipid Res. 1991, 32, 251–257. [Google Scholar] [PubMed]
- Shi, H.; Luo, J.; Zhu, J.; Li, J.; Sun, Y.; Lin, X.; Zhang, L.; Yao, D.; Shi, H. PPAR γ regulates genes involved in triacylglycerol synthesis and secretion in mammary gland epithelial cells of dairy goats. PPAR Res. 2013, 2013, 310948. [Google Scholar] [CrossRef] [PubMed]
- Ruby, M.A.; Goldenson, B.; Orasanu, G.; Johnston, T.P.; Plutzky, J.; Krauss, R.M. VLDL hydrolysis by LPL activates PPAR-α through generation of unbound fatty acids. J. Lipid Res. 2010, 51, 2275–2281. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.D.; Thuillier, P.; Baillie, R.A.; Sha, X.M. Peroxisome proliferator-activated receptors: A family of lipid-activated transcription factors. Am. J. Clin. Nutr. 1999, 70, 566–571. [Google Scholar] [PubMed]
- Langin, D.; Holm, C.; Lafontan, M. Adipocyte hormone-sensitive lipase: A major regulator of lipid metabolism. Proc. Nutr. Soc. 1996, 55, 93–109. [Google Scholar] [CrossRef]
- Frayn, K.N.; Coppack, S.W.; Fielding, B.A.; Humphreys, S.M. Coordinated regulation of hormone-sensitive lipase and lipoprotein lipase in human adipose tissue in vivo: Implications for the control of fat storage and fat mobilization. Adv. Enzym. Regul. 1995, 35, 163–178. [Google Scholar] [CrossRef]
- Martin-Hidalgo, A.; Huerta, L.; Alvarez, N.; Alegria, G.; del Val Toledo, M.; Herrera, E. Expression, activity, and localization of hormone-sensitive lipase in rat mammary gland during pregnancy and lactation. J. Lipid Res. 2005, 46, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Boizard, M.; le Liepvre, X.; Lemarchand, P.; Foufelle, F.; Ferre, P.; Dugail, I. Obesity-related overexpression of fatty-acid synthase gene in adipose tissue involves sterol regulatory element-binding protein transcription factors. J. Biol. Chem. 1998, 273, 29164–29171. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.M.; Bennett, M.K.; Sanchez, H.B.; Rosenfeld, J.M.; Osborne, T.F. Sterol regulation of acetyl coenzyme a carboxylase: A mechanism for coordinate control of cellular lipid. Proc. Natl. Acad. Sci. USA 1996, 93, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.B.; Spiegelman, B.M. ADD1/SREBP1 promotes adipocyte differentiation and gene expression linked to fatty acid metabolism. Genes Dev. 1996, 10, 1096–1107. [Google Scholar] [CrossRef]
- Ma, L.; Corl, B.A. Transcriptional regulation of lipid synthesis in bovine mammary epithelial cells by sterol regulatory element binding protein-1. J. Dairy Sci. 2012, 95, 3743–3755. [Google Scholar] [CrossRef] [PubMed]
- Oppi-Williams, C.; Suagee, J.K.; Corl, B.A. Regulation of lipid synthesis by liver X receptor α and sterol regulatory element-binding protein 1 in mammary epithelial cells. J. Dairy Sci. 2013, 96, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Kadegowda, A.K.; Bionaz, M.; Piperova, L.S.; Erdman, R.A.; Loor, J.J. Peroxisome proliferator-activated receptor-γ activation and long-chain fatty acids alter lipogenic gene networks in bovine mammary epithelial cells to various extents. J. Dairy Sci. 2009, 92, 4276–4289. [Google Scholar] [CrossRef] [PubMed]
- Farr, V.C.; Stelwagen, K.; Cate, L.R.; Molenaar, A.J.; McFadden, T.B.; Davis, S.R. An improved method for the routine biopsy of bovine mammary tissue. J. Dairy Sci. 1996, 79, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Ramunno, L.; Cosenza, G.; Rando, A.; Pauciullo, A.; Illario, R.; Gallo, D.; di Berardino, D.; Masina, P. Comparative analysis of gene sequence of goat CSN1S1 F and N alleles and characterization of CSN1S1 transcript variants in mammary gland. Gene 2005, 345, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Bionaz, M.; Loor, J.J. Identification of reference genes for quantitative real-time PCR in the bovine mammary gland during the lactation cycle. Physiol. Genomics 2007, 29, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Jager, L.; Hausl, M.A.; Rauschhuber, C.; Wolf, N.M.; Kay, M.A.; Ehrhardt, A. A rapid protocol for construction and production of high-capacity adenoviral vectors. Nat. Protoc. 2009, 4, 547–564. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Luo, J.; Wang, W.; Zhao, W.; Lin, X. Characterization and culture of isolated primary dairy goat mammary gland epithelial cells. Sheng Wu Gong Cheng Xue Bao 2010, 26, 1123–1127. (In Chinese) [Google Scholar] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, W.-S.; Hu, S.-L.; Yu, K.; Wang, H.; Wang, W.; Loor, J.; Luo, J. Lipoprotein Lipase, Tissue Expression and Effects on Genes Related to Fatty Acid Synthesis in Goat Mammary Epithelial Cells. Int. J. Mol. Sci. 2014, 15, 22757-22771. https://doi.org/10.3390/ijms151222757
Zhao W-S, Hu S-L, Yu K, Wang H, Wang W, Loor J, Luo J. Lipoprotein Lipase, Tissue Expression and Effects on Genes Related to Fatty Acid Synthesis in Goat Mammary Epithelial Cells. International Journal of Molecular Sciences. 2014; 15(12):22757-22771. https://doi.org/10.3390/ijms151222757
Chicago/Turabian StyleZhao, Wang-Sheng, Shi-Liang Hu, Kang Yu, Hui Wang, Wei Wang, Juan Loor, and Jun Luo. 2014. "Lipoprotein Lipase, Tissue Expression and Effects on Genes Related to Fatty Acid Synthesis in Goat Mammary Epithelial Cells" International Journal of Molecular Sciences 15, no. 12: 22757-22771. https://doi.org/10.3390/ijms151222757
APA StyleZhao, W.-S., Hu, S.-L., Yu, K., Wang, H., Wang, W., Loor, J., & Luo, J. (2014). Lipoprotein Lipase, Tissue Expression and Effects on Genes Related to Fatty Acid Synthesis in Goat Mammary Epithelial Cells. International Journal of Molecular Sciences, 15(12), 22757-22771. https://doi.org/10.3390/ijms151222757




