hnRNP A1: The Swiss Army Knife of Gene Expression
Abstract
:1. Introduction
2. The hnRNP A/B Family
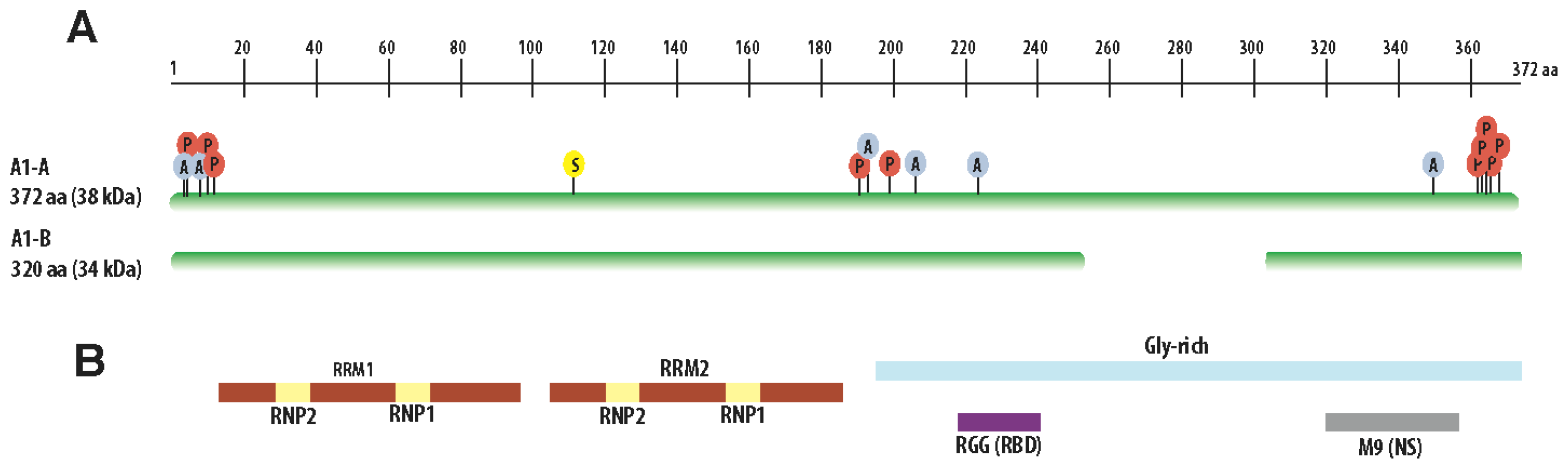
3. Structural Features of hnRNP A1
4. Transcriptional Functions of hnRNP A1
5. The Role of hnRNP A1 in Constitutive and Alternative mRNA Splicing
6. hnRNP A1, Telomeres Maintenance and Telomerase Activity
7. mRNA Nuclear Export and hnRNP A1
8. hnRNP A1 Regulates mRNA Translation and Turnover
9. hnRNP A1 Regulates miRNA Processing
10. hnRNP A1 Role in Human Disease and Therapy
11. Conclusions
Conflicts of Interest
References
- Dreyfuss, G.; Matunis, M.J.; Piñol-Roma, S.; Burd, C.G. hnRNP proteins and the biogenesis of mRNA. Ann. Rev. Biochem 1993, 62, 289–321. [Google Scholar]
- Beyer, A.L.; Christensen, M.E.; Walker, B.W.; LeStourgeon, W.M. Identification and characterisation of the packaging proteins of core 40S hnRNP particles. Cell 1977, 11, 127–138. [Google Scholar]
- Piñol-Roma, S.; Choi, Y.D.; Matunis, M.J.; Dreyfuss, G. Immunopurification of heterogeneous nuclear ribonucleoprotein particles reveals an assortment of RNA binding proteins. Genes Dev 1988, 2, 215–227. [Google Scholar]
- Dreyfuss, G.; Philipson, L.; Mattaj, I.W. Ribonucleoprotein particles in cellular processes. J. Cell Biol 1988, 106, 1419–1425. [Google Scholar]
- Kiledjian, M.; Dreyfuss, G. Primary structure and binding activity of the hnRNP U protein: Binding RNA through RGG box. EMBO J 1992, 11, 2655–2664. [Google Scholar]
- Burd, C.G.; Dreyfuss, G. Conserved structures and diversity of functions of RNA-binding proteins. Science (N. Y.) 1994, 265, 615–621. [Google Scholar]
- Han, S.P.; Tang, Y.H.; Smith, R. Functional diversity of the hnRNPs: Past, present and perspectives. Biochem. J 2010, 430, 379–392. [Google Scholar]
- Dreyfuss, G.; Kim, V.N.; Kataoka, N. Messenger-RNA-binding proteins and the messages they carry. Nat. Rev. Mol. Cell Biol 2002, 3, 195–205. [Google Scholar]
- Singh, R.; Valcarcel, J. Building specificity with nonspecific RNA-binding proteins. Nat. Struct. Mol. Biol 2005, 12, 645–653. [Google Scholar]
- Kamma, H.; Portman, D.S.; Dreyfuss, G. Cell type specific expression of of hnRNP proteins. Exp. Cell Res 1995, 221, 187–196. [Google Scholar]
- Huelga, S.C.; Vu, A.Q.; Arnold, J.D.; Liang, T.Y.; Liu, P.P.; Yan, B.Y.; Donohue, J.P.; Shiue, L.; Hoon, S.; Brenner, S.; et al. Integrative genome-wide analysis reveals cooperative regulation of alternative splicing by hnRNP proteins. Cell Rep 2012, 1, 167–178. [Google Scholar]
- Dangli, A.; Plomaritoglou, A.; Boutou, E.; Vassiliadou, N.; Moutsopoulos, H.M.; Guialis, A. Recognition of subsets of the mammalian A/B-type core heterogeneous nuclear ribonucleoprotein polypeptides by novel autoantibodies. Biochem. J 1996, 320, 761–767. [Google Scholar]
- Kamma, H.; Horiguchi, H.; Wan, L.; Matsui, M.; Fujiwara, M.; Fujimoto, M.; Yazawa, T.; Dreyfuss, G. Molecular characterization of the hnRNP A2/B1 proteins: Tissue-specific expression and novel isoforms. Exp. Cell Res 1999, 246, 399–411. [Google Scholar]
- Huang, M.; Rech, J.E.; Northington, S.J.; Flicker, P.F.; Mayeda, A.; Krainer, A.R.; LeStourgeon, W.M. The C-protein tetramer binds 230 to 240 nucleotides of pre-mRNA and nucleates the assembly of 40S heterogeneous nuclear ribonucleoprotein particles. Mol. Cell. Biol 1994, 14, 518–533. [Google Scholar]
- Han, K.; Yeo, G.; An, P.; Burge, C.B.; Grabowski, P.J. A combinatorial code for splicing silencing: UAGG and GGGG motifs. PLoS Biol 2005, 3, e158. [Google Scholar]
- Hutchison, S.; LeBel, C.; Blanchette, M.; Chabot, B. Distinct sets of adjacent heterogeneous nuclear ribonucleoprotein (hnRNP) A1/A2 binding sites control 5′ splice site selection in the hnRNP A1 mRNA precursor. J. Biol. Chem 2002, 277, 29745–29752. [Google Scholar]
- Chabot, B.; Blanchette, M.; Lapierre, I.; la Branche, H. An intron element modulating 5′ splice site selection in the hnRNP A1 pre-mRNA interacts with hnRNP A1. Mol. Cell. Biol 1997, 17, 1776–1786. [Google Scholar]
- Zahler, A.M.; Damgaard, C.K.; Kjems, J.; Caputi, M. SC35 and heterogeneous nuclear ribonucleoprotein A/B proteins bind to a juxtaposed exonic splicing enhancer/exonic splicing silencer element to regulate HIV-1 tat exon 2 splicing. J. Biol. Chem 2004, 279, 10077–10084. [Google Scholar]
- He, Y.; Smith, R. Nuclear functions of heterogeneous nuclear ribonucleoproteins A/B. Cell. Mol. Life Sci 2009, 66, 1239–1256. [Google Scholar]
- Barraud, P.; Allain, F.H. Solution structure of the two RNA recognition motifs of hnRNP A1 using segmental isotope labeling: How the relative orientation between RRMs influences the nucleic acid binding topology. J. Biomol. NMR 2013, 55, 119–138. [Google Scholar]
- Xu, R.M.; Jokhan, L.; Cheng, X.; Mayeda, A.; Krainer, A.R. Crystal structure of human UP1, the domain of hnRNP A1 that contains two RNA-recognition motifs. Structure 1997, 5, 559–570. [Google Scholar]
- Shamoo, Y.; Krueger, U.; Rice, L.M.; Williams, K.R.; Steitz, T.A. Crystal structure of the two RNA binding domains of human hnRNP A1 at 1.75 A resolution. Nat. Struct. Biol 1997, 4, 215–222. [Google Scholar]
- Vitali, J.; Ding, J.; Jiang, J.; Zhang, Y.; Krainer, A.R.; Xu, R.M. Correlated alternative side chain conformations in the RNA-recognition motif of heterogeneous nuclear ribonucleoprotein A1. Nucleic Acids Res 2002, 30, 1531–1538. [Google Scholar]
- Ding, J.; Hayashi, M.K.; Zhang, Y.; Manche, L.; Krainer, A.R.; Xu, R.M. Crystal structure of the two-RRM domain of hnRNP A1 (UP1) complexed with single-stranded telomeric DNA. Genes Dev 1999, 13, 1102–1115. [Google Scholar]
- Myers, J.C.; Shamoo, Y. Human UP1 as a model for understanding purine recognition in the family of proteins containing the RNA recognition motif (RRM). J. Mol. Biol 2004, 342, 743–756. [Google Scholar]
- Gorlach, M.; Wittekind, M.; Beckman, R.A.; Mueller, L.; Dreyfuss, G. Interaction of the RNA-binding domain of the hnRNP C proteins with RNA. EMBO J 1992, 11, 3289–3295. [Google Scholar]
- Mayeda, A.; Munroe, S.H.; Xu, R.M.; Krainer, A.R. Distinct functions of the closely related tandem RNA-recognition motifs of hnRNP A1. RNA (N. Y.) 1998, 4, 1111–1123. [Google Scholar]
- Fisette, J.F.; Toutant, J.; Dugre-Brisson, S.; Desgroseillers, L.; Chabot, B. hnRNP A1 and hnRNP H can collaborate to modulate 5′ splice site selection. RNA (N. Y.) 2010, 16, 228–238. [Google Scholar]
- Nadler, S.G.; Merrill, B.M.; Roberts, W.J.; Keating, K.M.; Lisbin, M.J.; Barnett, S.F.; Wilson, S.H.; Williams, K.R. Interactions of the A1 heterogeneous nuclear ribonucleoprotein and its proteolytic derivative, UP1, with RNA and DNA: Evidence for multiple RNA binding domains and salt-dependent binding mode transitions. Biochemistry 1991, 30, 2968–2976. [Google Scholar]
- Kumar, A.; Wilson, S.H. Studies of the strand-annealing activity of mammalian hnRNP complex protein A1. Biochemistry 1990, 29, 10717–10722. [Google Scholar]
- Mayeda, A.; Munroe, S.H.; Cáceres, J.F.; Krainer, A.R. Function of conserved domains of hnRNP A1 and other hnRNP A/B proteins. EMBO J 1994, 13, 5483–5495. [Google Scholar]
- Cartegni, L.; Maconi, M.; Morandi, E.; Cobianchi, F.; Riva, S.; Biamonti, G. hnRNP A1 selectively interacts through its gly-rich domain with different RNA-binding proteins. J. Mol. Biol 1996, 259, 337–348. [Google Scholar]
- Eperon, I.C.; Makarova, O.V.; Mayeda, A.; Munroe, S.H.; Caceres, J.F.; Hayward, D.G.; Krainer, A.R. Selection of alternative 5′ splice sites: Role of U1 snRNP and models for the antagonistic effects of SF2/ASF and hnRNP A1. Mol. Cell. Biol 2000, 20, 8303–8318. [Google Scholar]
- Michael, W.M.; Choi, M.; Dreyfuss, G. A nuclear export signal in hnRNP A1: A signal mediated, temperature-dependent nuclear protein export pathway. Cell 1995, 83, 415–422. [Google Scholar]
- Flynn, R.L.; Centore, R.C.; O’Sullivan, R.J.; Rai, R.; Tse, A.; Songyang, Z.; Chang, S.; Karlseder, J.; Zou, L. TERRA and hnRNP A1 orchestrate an RPA-to-POT1 switch on telomeric single-stranded DNA. Nature 2011, 471, 532–536. [Google Scholar]
- Siomi, H.; Dreyfuss, G. A nuclear localization domain in the hnRNP A1 protein. J. Cell Biol 1995, 129, 551–560. [Google Scholar]
- Pinol-Roma, S.; Dreyfuss, G. Shuttling of pre-mRNA binding proteins between nucleus and cytoplasm. Nature 1992, 355, 730–732. [Google Scholar]
- Allemand, E.; Guil, S.; Myers, M.; Moscat, J.; Caceres, J.F.; Krainer, A.R. Regulation of heterogenous nuclear ribonucleoprotein A1 transport by phosphorylation in cells stressed by osmotic shock. Proc. Natl. Acad. Sci. USA 2005, 102, 3605–3610. [Google Scholar]
- Siomi, M.C.; Eder, P.S.; Kataoka, N.; Wan, L.; Liu, Q.; Dreyfuss, G. Transportin-Mediated nuclear import of heterogeneous nuclear RNP proteins. J. Cell Biol 1997, 138, 1181–1192. [Google Scholar]
- Pollard, V.W.; Michael, W.M.; Nakielny, S.; Siomi, M.C.; Wang, F.; Dreyfuss, G. A novel receptor-mediated nuclear protein import pathway. Cell 1996, 86, 985–994. [Google Scholar]
- Rebane, A.; Aab, A.; Steitz, J.A. Transportins 1 and 2 are redundant nuclear import factors for hnRNP A1 and HuR. RNA (N. Y.) 2004, 10, 590–599. [Google Scholar]
- Herrick, G.; Alberts, B. Purification and physical characterization of nucleic acid helix-unwinding proteins from calf thymus. J. Biol. Chem 1976, 251, 2124–2132. [Google Scholar]
- Chen, H.; Hewison, M.; Hu, B.; Adams, J.S. Heterogeneous nuclear ribonucleoprotein (hnRNP) binding to hormone response elements: A cause of vitamin D resistance. Proc. Natl. Acad. Sci. USA 2003, 100, 6109–6114. [Google Scholar]
- Pontius, B.W.; Berg, P. Rapid assembly and disassembly of complementary DNA strands through an equilibrium intermediate state mediated by A1 hnRNP protein. J. Biol. Chem 1992, 267, 13815–13818. [Google Scholar]
- Kim, S.; Merrill, B.M.; Rajpurohit, R.; Kumar, A.; Stone, K.L.; Papov, V.V.; Schneiders, J.M.; Szer, W.; Wilson, S.H.; Paik, W.K.; et al. Identification of N(G)-methylarginine residues in human heterogeneous RNP protein A1: Phe/Gly-Gly-Gly-Arg-Gly-Gly-Gly/Phe is a preferred recognition motif. Biochemistry 1997, 36, 5185–5192. [Google Scholar]
- Burd, C.G.; Dreyfuss, G. RNA binding specificity of hnRNP A1: Significance of hnRNP A1 high-affinity binding sites in pre-mRNA splicing. EMBO J 1994, 13, 1197–1204. [Google Scholar]
- Hamilton, B.J.; Nagy, E.; Malter, J.S.; Arrick, B.A.; Rigby, W.F. Association of heterogeneous nuclear ribonucleoprotein A1 and C proteins with reiterated AUUUA sequences. J. Biol. Chem 1993, 268, 8881–8887. [Google Scholar]
- Zhu, J.; Mayeda, A.; Krainer, A.R. Exon identity established through differential antagonism between exonic splicing silencer-bound hnRNP A1 and enhancer-bound SR proteins. Mol. Cell 2001, 8, 1351–1361. [Google Scholar]
- Caputi, M.; Mayeda, A.; Krainer, A.R.; Zahler, A.M. hnRNP A/B proteins are required for inhibition of HIV-1 pre-mRNA splicing. EMBO J 1999, 18, 4060–4067. [Google Scholar]
- Li, H.P.; Zhang, X.; Duncan, R.; Comai, L.; Lai, M.M. Heterogeneous nuclear ribonucleoprotein A1 binds to the transcription- regulatory region of mouse hepatitis virus RNA. Proc. Natl. Acad. Sci. USA 1997, 94, 9544–9549. [Google Scholar]
- Marchand, V.; Mereau, A.; Jacquenet, S.; Thomas, D.; Mougin, A.; Gattoni, R.; Stevenin, J.; Branlant, C. A Janus splicing regulatory element modulates HIV-1 tat and rev mRNA production by coordination of hnRNP A1 cooperative binding. J. Mol. Biol 2002, 323, 629–652. [Google Scholar]
- Del Gatto, F.; Breathnach, R. Exon and intron sequences, respectively, repress and activate splicing of a fibroblast growth factor receptor 2 alternative exon. Mol. Cell. Biol 1995, 15, 4825–4834. [Google Scholar]
- Rajpurohit, R.; Paik, W.K.; Kim, S. Effect of enzymic methylation of heterogeneous ribonucleoprotein particle A1 on its nucleic-acid binding and controlled proteolysis. Biochem. J 1994, 304, 903–909. [Google Scholar]
- Shen, E.C.; Henry, M.F.; Weiss, V.H.; Valentini, S.R.; Silver, P.A.; Lee, M.S. Arginine methylation facilitates the nuclear export of hnRNP proteins. Genes Dev 1998, 12, 679–691. [Google Scholar]
- Municio, M.M.; Lozano, J.; Sanchez, P.; Moscat, J.; Diaz-Meco, M.T. Identification of heterogeneous ribonucleoprotein A1 as a novel substrate for protein kinase C zeta. J. Biol. Chem 1995, 270, 15884–15891. [Google Scholar]
- Idriss, H.; Kumar, A.; Casas-Finet, J.R.; Guo, H.; Damuni, Z.; Wilson, S.H. Regulation of in vitro nucleic acid strand annealing activity of heterogeneous nuclear ribonucleoprotein protein A1 by reversible phosphorylation. Biochemistry 1994, 33, 11382–11390. [Google Scholar]
- Buxade, M.; Parra, J.L.; Rousseau, S.; Shpiro, N.; Marquez, R.; Morrice, N.; Bain, J.; Espel, E.; Proud, C.G. The Mnks are novel components in the control of TNF alpha biosynthesis and phosphorylate and regulate hnRNP A1. Immunity 2005, 23, 177–189. [Google Scholar]
- Rigbolt, K.T.; Prokhorova, T.A.; Akimov, V.; Henningsen, J.; Johansen, P.T.; Kratchmarova, I.; Kassem, M.; Mann, M.; Olsen, J.V.; Blagoev, B. System-wide temporal characterization of the proteome and phosphoproteome of human embryonic stem cell differentiation. Sci. Signal. 2011, 4. [Google Scholar] [CrossRef]
- Dephoure, N.; Zhou, C.; Villen, J.; Beausoleil, S.A.; Bakalarski, C.E.; Elledge, S.J.; Gygi, S.P. A quantitative atlas of mitotic phosphorylation. Proc. Natl. Acad. Sci. USA 2008, 105, 10762–10767. [Google Scholar]
- Li, T.; Evdokimov, E.; Shen, R.F.; Chao, C.C.; Tekle, E.; Wang, T.; Stadtman, E.R.; Yang, D.C.; Chock, P.B. Sumoylation of heterogeneous nuclear ribonucleoproteins, zinc finger proteins, and nuclear pore complex proteins: A proteomic analysis. Proc. Natl. Acad. Sci. USA 2004, 101, 8551–8556. [Google Scholar]
- Lau, J.S.; Baumeister, P.; Kim, E.; Roy, B.; Hsieh, T.Y.; Lai, M.; Lee, A.S. Heterogeneous nuclear ribonucleoproteins as regulators of gene expression through interactions with the human thymidine kinase promoter. J. Cell. Biochem 2000, 79, 395–406. [Google Scholar]
- Xia, H. Regulation of gamma-fibrinogen chain expression by heterogeneous nuclear ribonucleoprotein A1. J. Biol. Chem 2005, 280, 13171–13178. [Google Scholar]
- Campillos, M.; Lamas, J.R.; Garcia, M.A.; Bullido, M.J.; Valdivieso, F.; Vazquez, J. Specific interaction of heterogeneous nuclear ribonucleoprotein A1 with the -219T allelic form modulates APOE promoter activity. Nucleic Acids Res 2003, 31, 3063–3070. [Google Scholar]
- Das, S.; Ward, S.V.; Markle, D.; Samuel, C.E. DNA damage-binding proteins and heterogeneous nuclear ribonucleoprotein A1 function as constitutive KCS element components of the interferon-inducible RNA-dependent protein kinase promoter. J. Biol. Chem 2004, 279, 7313–7321. [Google Scholar]
- Fukuda, H.; Katahira, M.; Tsuchiya, N.; Enokizono, Y.; Sugimura, T.; Nagao, M.; Nakagama, H. Unfolding of quadruplex structure in the G-rich strand of the minisatellite repeat by the binding protein UP1. Proc. Natl. Acad. Sci. USA 2002, 99, 12685–12690. [Google Scholar]
- Kan, Z.Y.; Lin, Y.; Wang, F.; Zhuang, X.Y.; Zhao, Y.; Pang, D.W.; Hao, Y.H.; Tan, Z. G-quadruplex formation in human telomeric (TTAGGG)4 sequence with complementary strand in close vicinity under molecularly crowded condition. Nucleic Acids Res 2007, 35, 3646–3653. [Google Scholar]
- Paramasivam, M.; Membrino, A.; Cogoi, S.; Fukuda, H.; Nakagama, H.; Xodo, L.E. Protein hnRNP A1 and its derivative Up1 unfold quadruplex DNA in the human KRAS promoter: Implications for transcription. Nucleic Acids Res 2009, 37, 2841–2853. [Google Scholar]
- Takimoto, M.; Tomonaga, T.; Matunis, M.; Avigan, M.; Krutzsch, H.; Dreyfuss, G.; Levens, D. Specific binding of heterogeneous ribonucleoprotein particle protein K to the human c-myc promoter, in vitro. J. Biol. Chem 1993, 268, 18249–18258. [Google Scholar]
- Diribarne, G.; Bensaude, O. 7SK RNA, a non-coding RNA regulating P-TEFb, a general transcription factor. RNA Biol 2009, 6, 122–128. [Google Scholar]
- Barrandon, C.; Bonnet, F.; Nguyen, V.T.; Labas, V.; Bensaude, O. The transcription-dependent dissociation of P-TEFb-HEXIM1–7SK RNA relies upon formation of hnRNP-7SK RNA complexes. Mol. Cell. Biol 2007, 27, 6996–7006. [Google Scholar]
- Van Herreweghe, E.; Egloff, S.; Goiffon, I.; Jady, B.E.; Froment, C.; Monsarrat, B.; Kiss, T. Dynamic remodelling of human 7SK snRNP controls the nuclear level of active P-TEFb. EMBO J 2007, 26, 3570–3580. [Google Scholar]
- Hay, D.C.; Kemp, G.D.; Dargemont, C.; Hay, R.T. Interaction between hnRNP A1 and IkappaBalpha is required for maximal activation of NF-kappaB-dependent transcription. Mol. Cell. Biol 2001, 21, 3482–3490. [Google Scholar]
- Moore, M.J.; Query, C.C.; Sharp, P.A. Splicing of Precursors to mRNA by the Spliceosome. In The RNA World; Gesteland, R.F., Atkins, J.F., Eds.; Cold Spring Harbor Press: New York, NY, USA, 1993; pp. 303–357. [Google Scholar]
- Nilsen, T.W. RNA-RNA interactions in the spliceosome: Unraveling the ties that bind. Cell 1994, 78, 1–4. [Google Scholar]
- Black, D.L. Finding splice sites within a wilderness of RNA. RNA (N. Y.) 1995, 1, 763–771. [Google Scholar]
- Jurica, M.S.; Licklider, L.J.; Gygi, S.R.; Grigorieff, N.; Moore, M.J. Purification and characterization of native spliceosomes suitable for three-dimensional structural analysis. RNA (N. Y.) 2002, 8, 426–439. [Google Scholar]
- Black, D.L. Mechanisms of alternative pre-messenger RNA splicing. Ann. Rev. Biochem 2003, 72, 291–336. [Google Scholar]
- Smith, C.W.J.; Valcarcel, J. Alternative pre-mRNA splicing: The logic of combinatorial control. TIBS 2000, 25, 381–388. [Google Scholar]
- Johnson, J.M.; Castle, J.; Garrett-Engele, P.; Kan, Z.; Loerch, P.M.; Armour, C.D.; Santos, R.; Schadt, E.E.; Stoughton, R.; Shoemaker, D.D. Genome-wide survey of human alternative pre-mRNA splicing with exon junction microarrays. Science (N. Y.) 2003, 302, 2141–2144. [Google Scholar]
- Blencowe, B.J. Alternative splicing: new insights from global analyses. Cell 2006, 126, 37–47. [Google Scholar]
- Wang, E.T.; Sandberg, R.; Luo, S.; Khrebtukova, I.; Zhang, L.; Mayr, C.; Kingsmore, S.F.; Schroth, G.P.; Burge, C.B. Alternative isoform regulation in human tissue transcriptomes. Nature 2008, 456, 470–476. [Google Scholar]
- Krecic, A.M.; Swanson, M.S. hnRNP complexes: Composition, structure, and function. Curr. Opin. Cell Biol 1999, 11, 363–371. [Google Scholar]
- Graveley, B.R. Sorting out the complexity of SR protein functions. RNA (N. Y.) 2000, 6, 1197–1211. [Google Scholar]
- Caceres, J.F.; Stamm, S.; Helfman, D.M.; Krainer, A.R. Regulation of alternative splicing in vivo by overexpression of antagonistic splicing factors. Science (N. Y.) 1994, 265, 1706–1709. [Google Scholar]
- Zhou, Z.; Licklider, L.J.; Gygi, S.P.; Reed, R. Comprehensive proteomic analysis of the human spliceosome. Nature 2002, 419, 182–185. [Google Scholar]
- Tavanez, J.P.; Madl, T.; Kooshapur, H.; Sattler, M.; Valcarcel, J. hnRNP A1 proofreads 3′ splice site recognition by U2AF. Mol. Cell 2012, 45, 314–329. [Google Scholar]
- Mayeda, A.; Krainer, A.R. Regulation of alternative pre-mRNA splicing by hnRNP A1 and splicing factor SF2. Cell 1992, 68, 365–375. [Google Scholar]
- Venables, J.P.; Bourgeois, C.F.; Dalgliesh, C.; Kister, L.; Stevenin, J.; Elliott, D.J. Up-Regulation of the ubiquitous alternative splicing factor Tra2beta causes inclusion of a germ cell-specific exon. Hum. Mol. Genet 2005, 14, 2289–2303. [Google Scholar]
- Dirksen, W.P.; Li, X.; Mayeda, A.; Krainer, A.R.; Rottman, F.M. Mapping the SF2/ASF binding sites in the bovine growth hormone exonic splicing enhancer. J. Biol. Chem 2000, 275, 29170–29177. [Google Scholar]
- Sun, Q.; Hampson, R.K.; Rottman, F.M. In vitro analysis of bovine growth hormone pre-messenger RNA alternative splicing-involvement of exon sequences and trans-acting factor(s). J. Biol. Chem 1993, 268, 15659–15666. [Google Scholar]
- Rooke, N.; Markovtsov, V.; Cagavi, E.; Black, D.L. Roles for SR proteins and hnRNP A1 in the regulation of c-src exon N1. Mol. Cell. Biol 2003, 23, 1874–1884. [Google Scholar]
- Kashima, T.; Manley, J.L. A negative element in SMN2 exon 7 inhibits splicing in spinal muscular atrophy. Nat. Genet 2003, 34, 460–463. [Google Scholar]
- Cartegni, L.; Krainer, A.R. Disruption of an SF2/ASF-dependent exonic splicing enhancer in causes spinal muscular atrophy in the absence of SMN1. Nat. Genet 2002, 30, 377–384. [Google Scholar]
- Okunola, H.L.; Krainer, A.R. Cooperative-Binding and splicing-repressive properties of hnRNP A1. Mol. Cell. Biol 2009, 29, 5620–5631. [Google Scholar]
- Guo, R.; Li, Y.; Ning, J.; Sun, D.; Lin, L.; Liu, X. HnRNP A1/A2 and SF2/ASF regulate alternative splicing of interferon regulatory factor-3 and affect immunomodulatory functions in human non-small cell lung cancer cells. PLoS One 2013, 8, e62729. [Google Scholar]
- Tange, T.O.; Damgaard, C.K.; Guth, S.; Valcarcel, J.; Kjems, J. The hnRNP A1 protein regulates HIV-1 tat splicing via a novel intron silencer element. EMBO J 2001, 20, 5748–5758. [Google Scholar]
- Blanchette, M.; Chabot, B. Modulation of exon skipping by high-affinity hnRNP A1-binding sites and by intron elements that repress splice site utilization. EMBO J 1999, 18, 1939–1952. [Google Scholar]
- Nasim, F.U.; Hutchison, S.; Cordeau, M.; Chabot, B. High-affinity hnRNP A1 binding sites and duplex-forming inverted repeats have similar effects on 5′ splice site selection in support of a common looping out and repression mechanism. RNA (N. Y.) 2002, 8, 1078–1089. [Google Scholar]
- Bruun, G.H.; Doktor, T.K.; Andresen, B.S. A synonymous polymorphic variation in ACADM exon 11 affects splicing efficiency and may affect fatty acid oxidation. Mol. Genet. Metab 2013, 110, 122–128. [Google Scholar]
- Zearfoss, N.R.; Johnson, E.S.; Ryder, S.P. hnRNP A1 and secondary structure coordinate alternative splicing of Mag. RNA (N. Y.) 2013, 19, 948–957. [Google Scholar]
- Oh, H.; Lee, E.; Jang, H.N.; Lee, J.; Moon, H.; Sheng, Z.; Jun, Y.; Loh, T.J.; Cho, S.; Zhou, J.; et al. hnRNP A1 contacts exon 5 to promote exon 6 inclusion of apoptotic Fas gene. Apoptosis 2013, 18, 825–835. [Google Scholar]
- Pelisch, F.; Khauv, D.; Risso, G.; Stallings-Mann, M.; Blaustein, M.; Quadrana, L.; Radisky, D.C.; Srebrow, A. Involvement of hnRNP A1 in the matrix metalloprotease-3-dependent regulation of Rac1 pre-mRNA splicing. J. Cell. Biochem 2013, 113, 2319–2329. [Google Scholar]
- Talukdar, I.; Sen, S.; Urbano, R.; Thompson, J.; Yates, J.R., III; Webster, N.J. hnRNP A1 and hnRNP F modulate the alternative splicing of exon 11 of the insulin receptor gene. PLoS One 2013, 6, e27869. [Google Scholar]
- Goina, E.; Skoko, N.; Pagani, F. Binding of DAZAP1 and hnRNP A1/A2 to an exonic splicing silencer in a natural BRCA1 exon 18 mutant. Mol. Cell. Biol 2008, 28, 3850–3860. [Google Scholar]
- Raponi, M.; Kralovicova, J.; Copson, E.; Divina, P.; Eccles, D.; Johnson, P.; Baralle, D.; Vorechovsky, I. Prediction of single-nucleotide substitutions that result in exon skipping: Identification of a splicing silencer in BRCA1 exon 6. Hum. Mutat 2011, 32, 436–444. [Google Scholar]
- Dirksen, W.P.; Sun, Q.; Rottman, F.M. Multiple splicing signals control alternative intron retention of bovine growth hormone pre-mRNA. J. Biol. Chem 1995, 270, 5346–5352. [Google Scholar]
- Doktor, T.K.; Schroeder, L.D.; Vested, A.; Palmfeldt, J.; Andersen, H.S.; Gregersen, N.; Andresen, B.S. SMN2 exon 7 splicing is inhibited by binding of hnRNP A1 to a common ESS motif that spans the 3′ splice site. Hum. Mutat 2011, 32, 220–230. [Google Scholar]
- Kashima, T.R.N.; David, C.J.; Manley, J.L. hnRNP A1 functions with specificity in repression of SMN2 exon 7 splicing. Hum. Mol. Genet 2007, 16, 3149–3159. [Google Scholar]
- Singh, N.N.; Androphy, E.J.; Singh, R.N. An extended inhibitory context causes skipping of exon 7 of SMN2 in spinal muscular atrophy. Biochem. Biophys. Res. Commun 2004, 315, 381–388. [Google Scholar]
- Kashima, T.; Rao, N.; Manley, J.L. An intronic element contributes to splicing repression in spinal muscular atrophy. Proc. Natl. Acad. Sci. USA 2007, 104, 3426–3431. [Google Scholar]
- DelGatto-Konczak, F.; Olive, M.; Gesnel, M.C.; Breathnach, R. hnRNP A1 recruited to an exon in vivo can function as an exon splicing silencer. Mol. Cell. Biol 1999, 19, 251–260. [Google Scholar]
- Gesnel, M.C.; del Gatto-Konczak, F.; Breathnach, R. Combined use of MS2 and PP7 coat fusions shows that TIA-1 dominates hnRNP A1 for K-SAM exon splicing control. J. Biomed. Biotechnol 2009, 2009, 104853. [Google Scholar]
- Donev, R.; Newall, A.; Thome, J.; Sheer, D. A role for SC35 and hnRNP A1 in the determination of amyloid precursor protein isoforms. Mol. Psychiatr 2007, 12, 681–690. [Google Scholar]
- Disset, A.; Bourgeois, C.F.; Benmalek, N.; Claustres, M.; Stevenin, J.; Tuffery-Giraud, S. An exon skipping-associated nonsense mutation in the dystrophin gene uncovers a complex interplay between multiple antagonistic splicing elements. Hum. Mol. Genet 2006, 15, 999–1013. [Google Scholar]
- Expert-Bezancon, A.; Sureau, A.; Durosay, P.; Salesse, R.; Groeneveld, H.; Lecaer, J.P.; Marie, J. hnRNP A1 and the SR proteins ASF/SF2 and SC35 have antagonistic functions in splicing of beta-tropomyosin exon 6B. J. Biol. Chem 2004, 279, 38249–38259. [Google Scholar]
- Princler, G.L.; Julias, J.G.; Hughes, S.H.; Derse, D. Roles of viral and cellular proteins in the expression of alternatively spliced HTLV-1 pX mRNAs. Virology 2003, 317, 136–145. [Google Scholar]
- Guil, S.; Gattoni, R.; Carrascal, M.; Abian, J.; Stevenin, J.; Bach-Elias, M. Roles of hnRNP A1, SR proteins, and p68 helicase in c-H-ras alternative splicing regulation. Mol. Cell. Biol 2003, 23, 2927–2941. [Google Scholar]
- Damgaard, C.K.; Tange, T.O.; Kjems, J. hnRNP A1 controls HIV-1 mRNA splicing through cooperative binding to intron and exon splicing silencers in the context of a conserved secondary structure. RNA (N. Y.) 2002, 8, 1401–1415. [Google Scholar]
- Dery, K.J.; Gaur, S.; Gencheva, M.; Yen, Y.; Shively, J.E.; Gaur, R.K. Mechanistic control of carcinoembryonic antigen-related cell adhesion molecule-1 (CEACAM1) splice isoforms by the heterogeneous nuclear ribonuclear proteins hnRNP L, hnRNP A1, and hnRNP M. J. Biol. Chem 2011, 286, 16039–16051. [Google Scholar]
- David, C.J.; Chen, M.; Assanah, M.; Canoll, P.; Manley, J.L. HnRNP proteins controlled by c-Myc deregulate pyruvate kinase mRNA splicing in cancer. Nature 2010, 463, 364–368. [Google Scholar]
- Bilodeau, P.S.; Domsic, J.K.; Mayeda, A.; Krainer, A.R.; Stoltzfus, C.M. RNA splicing at human immunodeficiency virus type 1 3′ splice site A2 is regulated by binding of hnRNP A/B proteins to an exonic splicing silencer element. J. Virol 2001, 75, 8487–8497. [Google Scholar]
- Rosenberger, S.; De-Castro Arce, J.; Langbein, L.; Steenbergen, R.D.; Rosl, F. Alternative splicing of human papillomavirus type-16 E6/E6* early mRNA is coupled to EGF signaling via Erk1/2 activation. Proc. Natl. Acad. Sci. USA 2010, 107, 7006–7011. [Google Scholar]
- Pastor, T.; Pagani, F. Interaction of hnRNP A1/A2 and DAZAP1 with an Alu-derived intronic splicing enhancer regulates ATM aberrant splicing. PLoS One 2011, 6, e23349. [Google Scholar]
- Blackburn, E.H. Switching and signaling at the telomere. Cell 2001, 106, 661–673. [Google Scholar]
- Palm, W.; de Lange, T. How shelterin protects mammalian telomeres. Ann. Rev. Genet 2008, 42, 301–334. [Google Scholar]
- Parkinson, G.N.; Lee, M.P.; Neidle, S. Crystal structure of parallel quadruplexes from human telomeric DNA. Nature 2002, 417, 876–880. [Google Scholar]
- Meeker, A.K.; de Marzo, A.M. Recent advances in telomere biology: Implications for human cancer. Curr. Opin. Oncol 2004, 16, 32–38. [Google Scholar]
- Zhang, Q.S.; Manche, L.; Xu, R.M.; Krainer, A.R. hnRNP A1 associates with telomere ends and stimulates telomerase activity. RNA (N. Y.) 2006, 12, 1116–1128. [Google Scholar]
- LaBranche, H.; Dupuis, S.; Ben-David, Y.; Bani, M.R.; Wellinger, R.J.; Chabot, B. Telomere elongation by hnRNP A1 and a derivative that interacts with telomeric repeats and telomerase. Nat. Genet 1998, 19, 199–202. [Google Scholar]
- Fiset, S.; Chabot, B. hnRNP A1 may interact simultaneously with telomeric DNA and the human telomerase RNA in vitro. Nucleic Acids Res 2001, 29, 2268–2275. [Google Scholar]
- Lopez de Silanes, I.; Stagnod’ Alcontres, M.; Blasco, M.A. TERRA transcripts are bound by a complex array of RNA-binding proteins. Nat. Commun 2010, 1, 33. [Google Scholar]
- Le, P.N.; Maranon, D.G.; Altina, N.H.; Battaglia, C.L.; Bailey, S.M. TERRA, hnRNP A1, and DNA-PKcs Interactions at Human Telomeres. Front. Oncol 2013, 3, 91. [Google Scholar]
- Dallaire, F.; Dupuis, S.; Fiset, S.; Chabot, B. Heterogeneous nuclear ribonucleoprotein A1 and UP1 protect mammalian telomeric repeats and modulate telomere replication in vitro. J. Biol. Chem 2000, 275, 14509–14516. [Google Scholar]
- Mili, S.; Shu, H.J.; Zhao, Y.; Pinol-Roma, S. Distinct RNP complexes of shuttling hnRNP proteins with pre-mRNA and mRNA: Candidate intermediates in formation and export of mRNA. Mol. Cell. Biol 2001, 21, 7307–7319. [Google Scholar]
- Nakielny, S.; Dreyfuss, G. Transport of proteins and RNAs in and out of the nucleus. Cell 1999, 99, 677–690. [Google Scholar]
- Izaurralde, E.; Jarmolowski, A.; Beisel, C.; Mattaj, I.W.; Dreyfuss, G.; Fischer, U. A role for the M9 transport signal of hnRNP A1 in mRNA nuclear export. J. Cell Biol 1997, 137, 27–35. [Google Scholar]
- Gallouzi, I.E.; Steitz, J.A. Delineation of mRNA export pathways by the use of cell-permeable peptides. Science (N. Y.) 2001, 294, 1895–1901. [Google Scholar]
- Visa, N.; Alzhanova-Ericsson, A.T.; Sun, X.; Kiseleva, E.; Bjorkroth, B.; Wurtz, T.; Daneholt, B. A pre-mRNA-binding protein accompanies the RNA from the gene through the nuclear pores and into polysomes. Cell 1996, 84, 253–264. [Google Scholar]
- Siebrasse, J.P.; Kaminski, T.; Kubitscheck, U. Nuclear export of single native mRNA molecules observed by light sheet fluorescence microscopy. Proc. Natl. Acad. Sci. USA 2012, 109, 9426–9431. [Google Scholar]
- Vautier, D.; Chesne, P.; Cunha, C.; Calado, A.; Renard, J.P.; Carmo-Fonseca, M. Transcription-Dependent nucleocytoplasmic distribution of hnRNP A1 protein in early mouse embryos. J. Cell Sci 2001, 114, 1521–1531. [Google Scholar]
- Kataoka, N.; Yong, J.; Kim, V.N.; Velazquez, F.; Perkinson, R.A.; Wang, F.; Dreyfuss, G. Pre-mRNA splicing imprints mRNA in the nucleus with a novel RNA-binding protein that persists in the cytoplasm. Mol. Cell 2000, 6, 673–682. [Google Scholar]
- Barreau, C.; Paillard, L.; Osborne, H.B. AU-Rich elements and associated factors: Are there unifying principles? Nucleic Acids Res 2005, 33, 7138–7150. [Google Scholar]
- Henics, T.; Sanfridson, A.; Hamilton, B.J.; Nagy, E.; Rigby, W.F. Enhanced stability of interleukin-2 mRNA in MLA 144 cells. Possible role of cytoplasmic AU-rich sequence-binding proteins. J. Biol. Chem 1994, 269, 5377–5383. [Google Scholar]
- Hamilton, B.J.; Burns, C.M.; Nichols, R.C.; Rigby, W.F. Modulation of AUUUA response element binding by heterogeneous nuclear ribonucleoprotein A1 in human T lymphocytes. The roles of cytoplasmic location, transcription, and phosphorylation. J. Biol. Chem 1997, 272, 28732–28741. [Google Scholar]
- Svitkin, Y.V.; Ovchinnikov, L.P.; Dreyfuss, G.; Sonenberg, N. General RNA binding proteins render translation cap dependent. EMBO J 1996, 15, 7147–7155. [Google Scholar]
- Martinez-Salas, E.; Pineiro, D.; Fernandez, N. Alternative mechanisms to initiate translation in eukaryotic mRNAs. Comp. Funct. Gen 2012, 2012, 391546. [Google Scholar]
- Bonnal, S.; Pileur, F.; Orsini, C.; Parker, F.; Pujol, F.; Prats, A.C.; Vagner, S. Heterogeneous nuclear ribonucleoprotein A1 is a novel internal ribosome entry site trans-acting factor that modulates alternative initiation of translation of the fibroblast growth factor 2 mRNA. J. Biol. Chem 2005, 280, 4144–4153. [Google Scholar]
- Cammas, A.; Pileur, F.; Bonnal, S.; Lewis, S.M.; Leveque, N.; Holcik, M.; Vagner, S. Cytoplasmic relocalization of heterogeneous nuclear ribonucleoprotein A1 controls translation initiation of specific mRNAs. Mol. Biol. Cell 2007, 18, 5048–5059. [Google Scholar]
- Damiano, F.; Rochira, A.; Tocci, R.; Alemanno, S.; Gnoni, A.; Siculella, L. hnRNP A1 mediates the activation of the IRES-dependent SREBP-1a mRNA translation in response to endoplasmic reticulum stress. Biochem. J 2013, 449, 543–553. [Google Scholar]
- Jo, O.D.; Martin, J.; Bernath, A.; Masri, J.; Lichtenstein, A.; Gera, J. Heterogeneous nuclear ribonucleoprotein A1 regulates cyclin D1 and c-myc internal ribosome entry site function through Akt signaling. J. Biol. Chem 2008, 283, 23274–23287. [Google Scholar]
- Martin, J.; Masri, J.; Cloninger, C.; Holmes, B.; Artinian, N.; Funk, A.; Ruegg, T.; Anderson, L.; Bashir, T.; Bernath, A.; et al. Phosphomimetic substitution of heterogeneous nuclear ribonucleoprotein A1 at serine 199 abolishes AKT-dependent internal ribosome entry site-transacting factor (ITAF) function via effects on strand annealing and results in mammalian target of rapamycin complex 1 (mTORC1) inhibitor sensitivity. J. Biol. Chem 2011, 286, 16402–16413. [Google Scholar]
- Lewis, S.M.; Veyrier, A.; Hosszu Ungureanu, N.; Bonnal, S.; Vagner, S.; Holcik, M. Subcellular relocalization of a trans-acting factor regulates XIAP IRES-dependent translation. Mol. Biol. Cell 2007, 18, 1302–1311. [Google Scholar]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar]
- Kim, V.N.; Han, J.; Siomi, M.C. Biogenesis of small RNAs in animals. Nat. Rev. Mol. Cell Biol 2009, 10, 126–139. [Google Scholar]
- Guil, S.; Caceres, J.F. The multifunctional RNA-binding protein hnRNP A1 is required for processing of miR-18a. Nat. Struct. Mol. Biol 2007, 14, 591–596. [Google Scholar]
- Michlewski, G.; Guil, S.; Semple, C.A.; Caceres, J.F. Posttranscriptional regulation of miRNAs harboring conserved terminal loops. Mol. Cell 2008, 32, 383–393. [Google Scholar]
- Roush, S.; Slack, F.J. The let-7 family of microRNAs. Trends Cell Biol 2008, 18, 505–516. [Google Scholar]
- Michlewski, G.; Caceres, J.F. Antagonistic role of hnRNP A1 and KSRP in the regulation of let-7a biogenesis. Nat. Struct. Mol. Biol 2010, 17, 1011–1018. [Google Scholar]
- Gherzi, R.; Chen, C.Y.; Trabucchi, M.; Ramos, A.; Briata, P. The role of KSRP in mRNA decay and microRNA precursor maturation. Wiley Interdiscip. Rev 2010, 1, 230–239. [Google Scholar]
- Pino, I.; Pio, R.; Toledo, G.; Zabalegui, N.; Vicent, S.; Rey, N.; Lozano, M.D.; Torre, W.; Garcia-Foncillas, J.; Montuenga, L.M. Altered patterns of expression of members of the heterogeneous nuclear ribonucleoprotein (hnRNP) family in lung cancer. Lung Cancer (Amsterdam, The Netherlands) 2003, 41, 131–143. [Google Scholar]
- Ushigome, M.; Ubagai, T.; Fukuda, H.; Tsuchiya, N.; Sugimura, T.; Takatsuka, J.; Nakagama, H. Up-regulation of hnRNP A1 gene in sporadic human colorectal cancers. Int. J. Oncol 2005, 26, 635–640. [Google Scholar]
- Carpenter, B.; MacKay, C.; Alnabulsi, A.; MacKay, M.; Telfer, C.; Melvin, W.T.; Murray, G.I. The roles of heterogeneous nuclear ribonucleoproteins in tumour development and progression. Biochim. Biophys. Acta 2006, 1765, 85–100. [Google Scholar]
- Boukakis, G.; Patrinou-Georgoula, M.; Lekarakou, M.; Valavanis, C.; Guialis, A. Deregulated expression of hnRNP A/B proteins in human non-small cell lung cancer: Parallel assessment of protein and mRNA levels in paired tumour/non-tumour tissues. BMC Cancer 2010, 10, 434. [Google Scholar]
- Venables, J.P. Unbalanced alternative splicing and its significance in cancer. Bioessays 2006, 28, 378–386. [Google Scholar]
- Venables, J.P.; Koh, C.S.; Froehlich, U.; Lapointe, E.; Couture, S.; Inkel, L.; Bramard, A.; Paquet, E.R.; Watier, V.; Durand, M.; et al. Multiple and specific mRNA processing targets for the major human hnRNP proteins. Mol. Cell. Biol 2008, 28, 6033–6043. [Google Scholar]
- Zhou, Z.J.; Dai, Z.; Zhou, S.L.; Fu, X.T.; Zhao, Y.M.; Shi, Y.H.; Zhou, J.; Fan, J. Overexpression of HnRNP A1 promotes tumor invasion through regulating CD44v6 and indicates poor prognosis for hepatocellular carcinoma. Int. J. Cancer 2012, 132, 1080–1089. [Google Scholar]
- Patry, C.; Bouchard, L.; Labrecque, P.; Gendron, D.; Lemieux, B.; Toutant, J.; Lapointe, E.; Wellinger, R.; Chabot, B. Small interfering RNA-mediated reduction in heterogeneous nuclear ribonucleoparticule A1/A2 proteins induces apoptosis in human cancer cells but not in normal mortal cell lines. Cancer Res 2003, 63, 7679–7688. [Google Scholar]
- Jiang, Z.H.; Zhang, W.J.; Rao, Y.; Wu, J.Y. Regulation of Ich-1 pre-mRNA alternative splicing and apoptosis by mammalian splicing factors. Proc. Natl. Acad. Sci. USA 1998, 95, 9155–9160. [Google Scholar]
- Bekenstein, U.; Soreq, H. Heterogeneous nuclear ribonucleoprotein A1 in health and neurodegenerative disease: From structural insights to post-transcriptional regulatory roles. Mol. Cell. Neurosci. 2012. [Google Scholar] [CrossRef]
- Berson, A.; Barbash, S.; Shaltiel, G.; Goll, Y.; Hanin, G.; Greenberg, D.S.; Ketzef, M.; Becker, A.J.; Friedman, A.; Soreq, H. Cholinergic-associated loss of hnRNP-A/B in Alzheimer’s disease impairs cortical splicing and cognitive function in mice. EMBO Mol. Med 2012, 4, 730–742. [Google Scholar]
- Kim, H.J.; Kim, N.C.; Wang, Y.D.; Scarborough, E.A.; Moore, J.; Diaz, Z.; MacLea, K.S.; Freibaum, B.; Li, S.; Molliex, A.; et al. Mutations in prion-like domains in hnRNPA2B1 and hnRNP A1 cause multisystem proteinopathy and ALS. Nature 2013, 495, 467–473. [Google Scholar]
- Shih, S.R.; Stollar, V.; Li, M.L. Host factors in enterovirus 71 replication. J. Virol 2011, 85, 9658–9666. [Google Scholar]
- Lin, J.Y.; Shih, S.R.; Pan, M.; Li, C.; Lue, C.F.; Stollar, V.; Li, M.L. hnRNP A1 interacts with the 5′ untranslated regions of enterovirus 71 and Sindbis virus RNA and is required for viral replication. J. Virol 2009, 83, 6106–6114. [Google Scholar]
- Kim, C.S.; Seol, S.K.; Song, O.K.; Park, J.H.; Jang, S.K. An RNA-binding protein, hnRNP A1, and a scaffold protein, septin 6, facilitate hepatitis C virus replication. J. Virol 2007, 81, 3852–3865. [Google Scholar]
- Zhao, X.; Schwartz, S. Inhibition of HPV-16 L1 expression from L1 cDNAs correlates with the presence of hnRNP A1 binding sites in the L1 coding region. Virus Genes 2008, 36, 45–53. [Google Scholar]
- Zhao, X.; Fay, J.; Lambkin, H.; Schwartz, S. Identification of a 17-nucleotide splicing enhancer in HPV-16 L1 that counteracts the effect of multiple hnRNP A1-binding splicing silencers. Virology 2007, 369, 351–363. [Google Scholar]
- Karn, J.; Stoltzfus, C.M. Transcriptional and posttranscriptional regulation of HIV-1 gene expression. Cold Spring Harbor Perspect. Med 2012, 2, a006916. [Google Scholar]
- Jablonski, J.A.; Caputi, M. Role of cellular RNA processing factors in human immunodeficiency virus type 1 mRNA metabolism, replication, and infectivity. J. Virol 2009, 83, 981–992. [Google Scholar]
- Monette, A.; Ajamian, L.; Lopez-Lastra, M.; Mouland, A.J. Human immunodeficiency virus type 1 (HIV-1) induces the cytoplasmic retention of heterogeneous nuclear ribonucleoprotein A1 by disrupting nuclear import: Implications for HIV-1 gene expression. J. Biol. Chem 2009, 284, 31350–31362. [Google Scholar]
- Najera, I.; Krieg, M.; Karn, J. Synergistic stimulation of HIV-1 Rev-dependent export of unspliced mRNA to the cytoplasm by hnRNP A1. J. Mol. Biol 1999, 285, 1951–1964. [Google Scholar]
- Lorson, C.L.; Rindt, H.; Shababi, M. Spinal muscular atrophy: Mechanisms and therapeutic strategies. Hum. Mol. Genet 2010, 19, R111–R118. [Google Scholar]
- Mitrpant, C.; Porensky, P.; Zhou, H.; Price, L.; Muntoni, F.; Fletcher, S.; Wilton, S.D.; Burghes, A.H. Improved antisense oligonucleotide design to suppress aberrant SMN2 gene transcript processing: Towards a treatment for spinal muscular atrophy. PLoS One 2013, 8, e62114. [Google Scholar]
- Rigo, F.; Hua, Y.; Krainer, A.R.; Bennett, C.F. Antisense-Based therapy for the treatment of spinal muscular atrophy. J. Cell Biol 2012, 199, 21–25. [Google Scholar]
- Hua, Y.; Sahashi, K.; Hung, G.; Rigo, F.; Passini, M.A.; Bennett, C.F.; Krainer, A.R. Antisense correction of SMN2 splicing in the CNS rescues necrosis in a type III SMA mouse model. Genes Dev 2010, 24, 1634–1644. [Google Scholar]
- Hua, Y.; Vickers, T.A.; Okunola, H.L.; Bennett, C.F.; Krainer, A.R. Antisense masking of an hnRNP A1/A2 intronic splicing silencer corrects SMN2 splicing in transgenic mice. Am. J. Hum. Genet 2008, 82, 834–848. [Google Scholar]
- Li, S.; Wang, W.; Ding, H.; Xu, H.; Zhao, Q.; Li, J.; Li, H.; Xia, W.; Su, X.; Chen, Y.; et al. Aptamer BC15 against heterogeneous nuclear ribonucleoprotein A1 has potential value in diagnosis and therapy of hepatocarcinoma. Nucleic Acid Ther 2012, 22, 391–398. [Google Scholar]

| Gene | Organism | Spicing event | Reference |
|---|---|---|---|
| Medium-chain acyl-CoA dehydrogenase (MCAD) | Human | Exon 11 skipping | [99] |
| Myelin-associated glycoprotein (MAG) | Human | Exon 12 skipping | [100] |
| Interferon regulatory factor-3 (IRF-3) | Human | Exons 2 and 3 skipping | [95] |
| TNF Receptor Superfamily Member 6 (Fas) | Human | Exon 6 skipping | [101] |
| Ras-related C3 botulinum toxin substrate 1 (Rac1) | Human | Exon 3b skipping | [102] |
| Insulin receptor gene (INSR) | Human | Exon 11 skipping | [103] |
| Breast cancer 1 (BRCA1) | Human | Exon 18 skipping | [104] |
| Breast cancer 1 (BRCA1) | Human | Exon 6 skipping | [105] |
| Homeodomain interacting protein kinase 3 (HIPK3) | Human | Testis-specific Exon skipping | [88] |
| Bovine growth hormone (BGH) | Bovine | Exon 5 skipping | [89,90,106] |
| Survival of Motor Neuron 2, (SMN2) | Human | Exon 7 skipping | [92,107–110] |
| Fibroblast growth factor receptor 2 (FGFR2) | Human | K-SAM exon skipping | [111,112] |
| Amyloid precursor protein (APP) | Human | Exon 7 and 8 skipping | [113] |
| Dystrophin | Human | Exon 31 skipping | [114] |
| β-tropomyosin | Chicken | Exon 6B skipping | [115] |
| pX region | Human T-cell leukemia virus type 1 (HTLV-1) | Exon skipping | [116] |
| V-Ha-ras Harvey rat sarcoma viral oncogene homolog (C–H-ras) | Human | Exon IDX skipping | [117] |
| Proto-oncogene tyrosine-protein kinase Src (c-SRC) | Human | Exon N1 skipping | [91] |
| Trans-activator of transcription (Tat) | Human immunodeficiency virus type 1 (HIV-1) | Exon 3 3′ ss repression | [48,51,94,96,118] |
| Trans-activator of transcription (Tat) | Human immunodeficiency virus type 1 (HIV-1) | Exon 2 3′ ss repression | [18,49] |
| Carcinoembryonic antigen-related cell adhesion molecule-1 (CEACAM1) | Human | Exon 7 skipping | [119] |
| heterogeneous ribonucleoprotein A1 (hnRNP A1) | Human | Exon 7B skipping | [16,97,98] |
| Pyruvate kinase (PKM) | Human | Exon 9 skipping | [120] |
| Viral protein R (VPR) | Human immunodeficiency virus type 1 (HIV-1) | Repression 3′ splice site A2 | [121] |
| E6/E7 | Human papillomavirus type-16 (HPV-16) | E6 exon skipping | [122] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jean-Philippe, J.; Paz, S.; Caputi, M. hnRNP A1: The Swiss Army Knife of Gene Expression. Int. J. Mol. Sci. 2013, 14, 18999-19024. https://doi.org/10.3390/ijms140918999
Jean-Philippe J, Paz S, Caputi M. hnRNP A1: The Swiss Army Knife of Gene Expression. International Journal of Molecular Sciences. 2013; 14(9):18999-19024. https://doi.org/10.3390/ijms140918999
Chicago/Turabian StyleJean-Philippe, Jacques, Sean Paz, and Massimo Caputi. 2013. "hnRNP A1: The Swiss Army Knife of Gene Expression" International Journal of Molecular Sciences 14, no. 9: 18999-19024. https://doi.org/10.3390/ijms140918999
APA StyleJean-Philippe, J., Paz, S., & Caputi, M. (2013). hnRNP A1: The Swiss Army Knife of Gene Expression. International Journal of Molecular Sciences, 14(9), 18999-19024. https://doi.org/10.3390/ijms140918999




