Increased Chondrocyte Apoptosis Is Associated with Progression of Osteoarthritis in Spontaneous Guinea Pig Models of the Disease
Abstract
:1. Introduction
2. Results
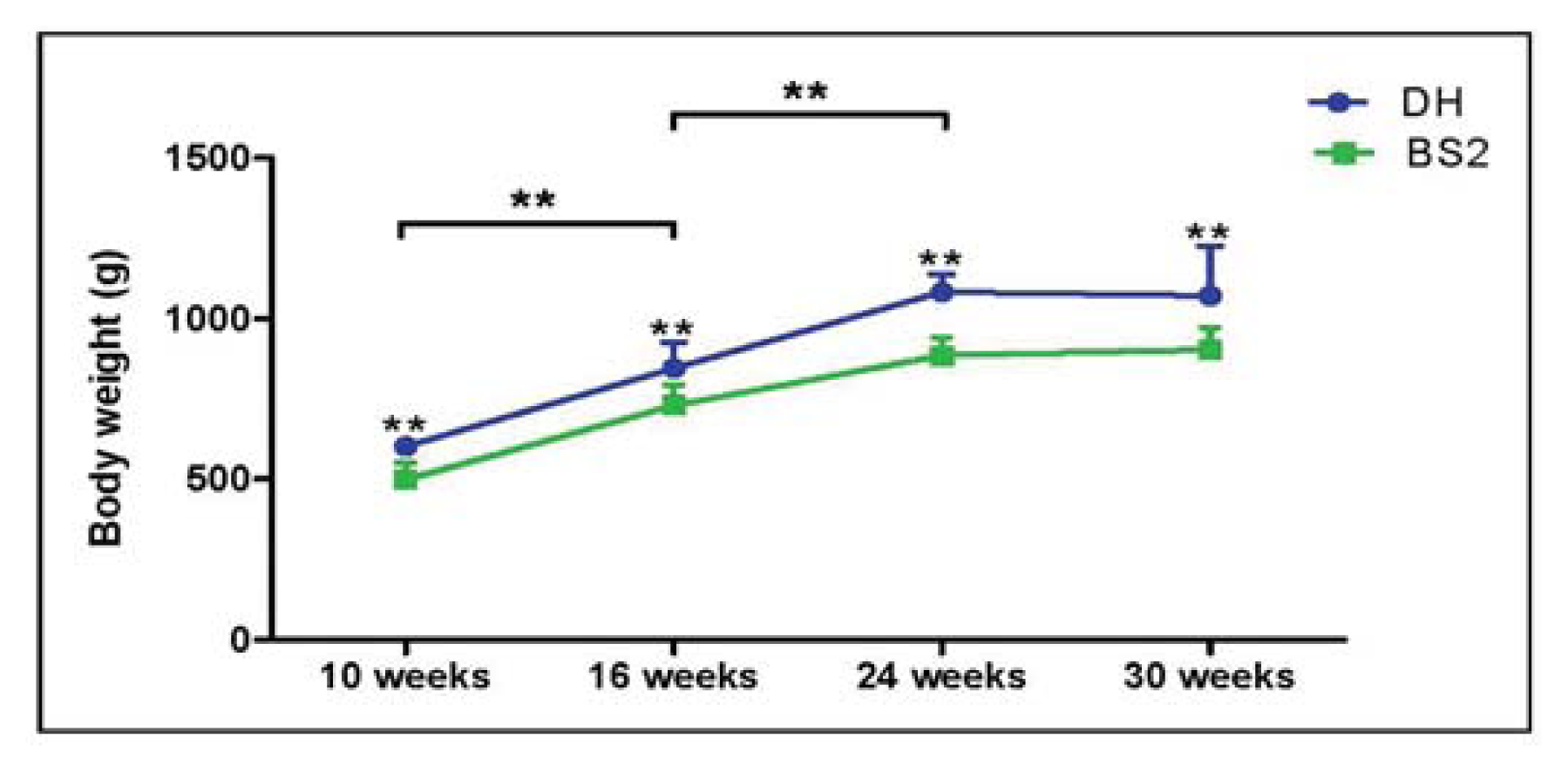
2.1. Body Weight
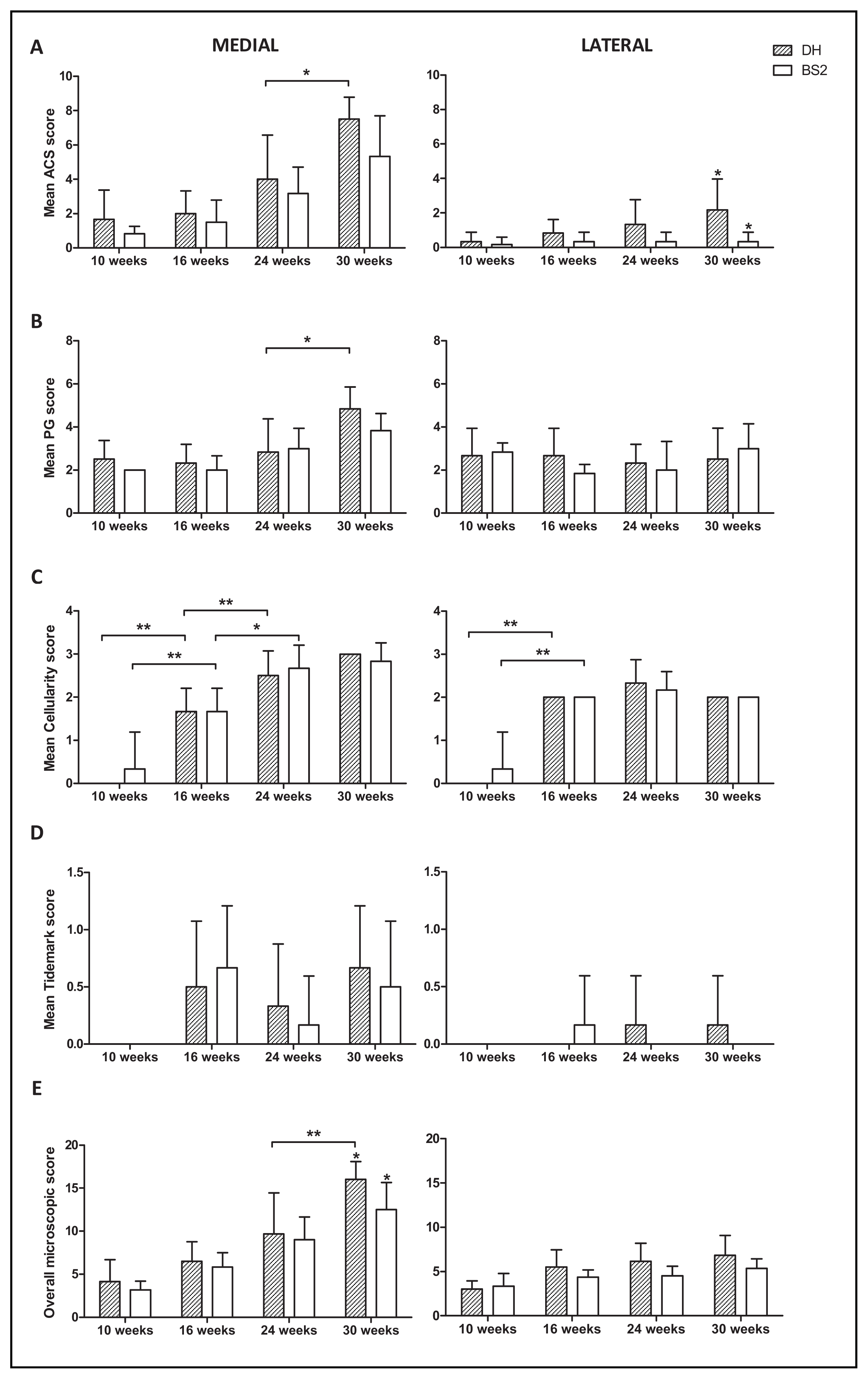
2.2. Histopathological Changes in AC
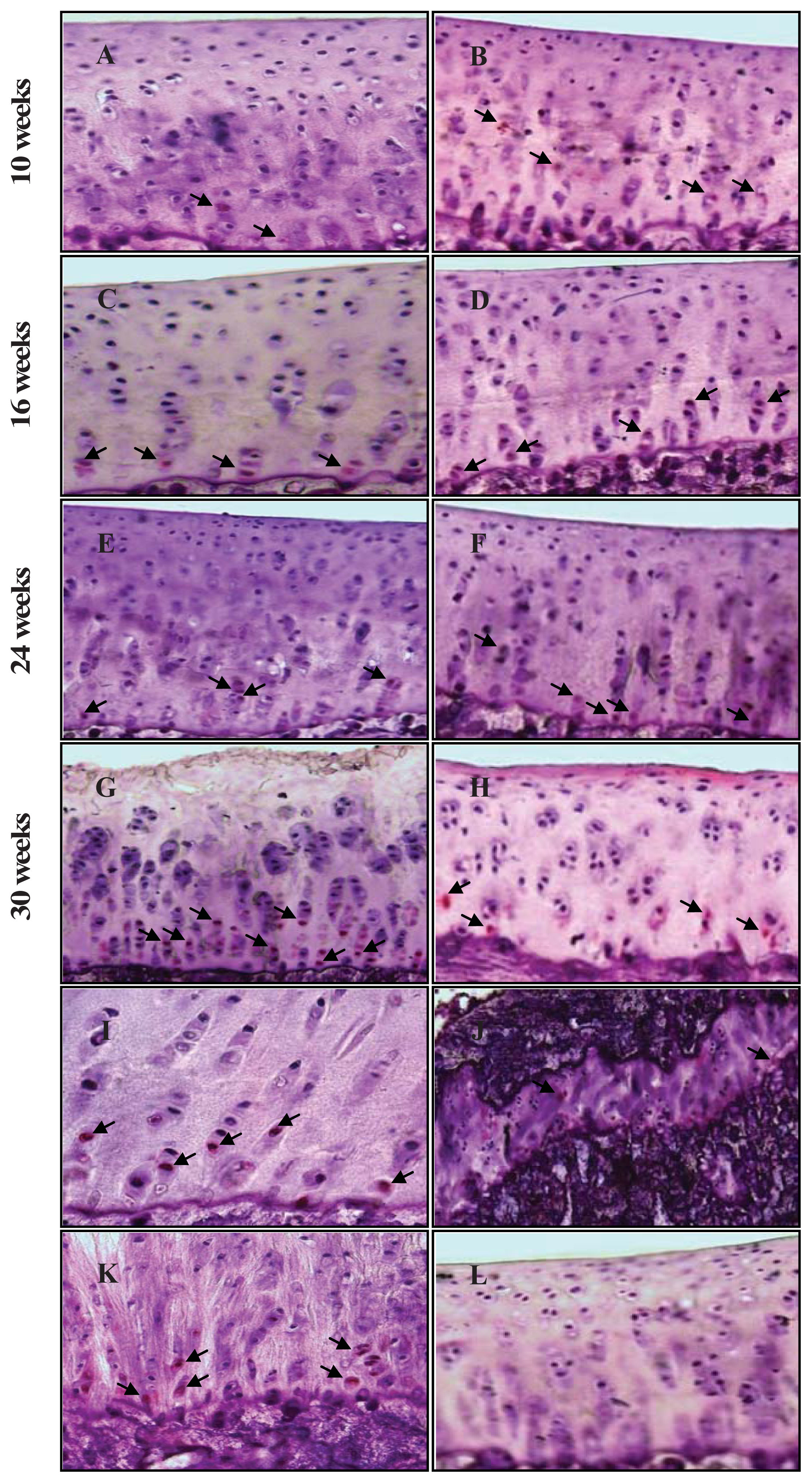
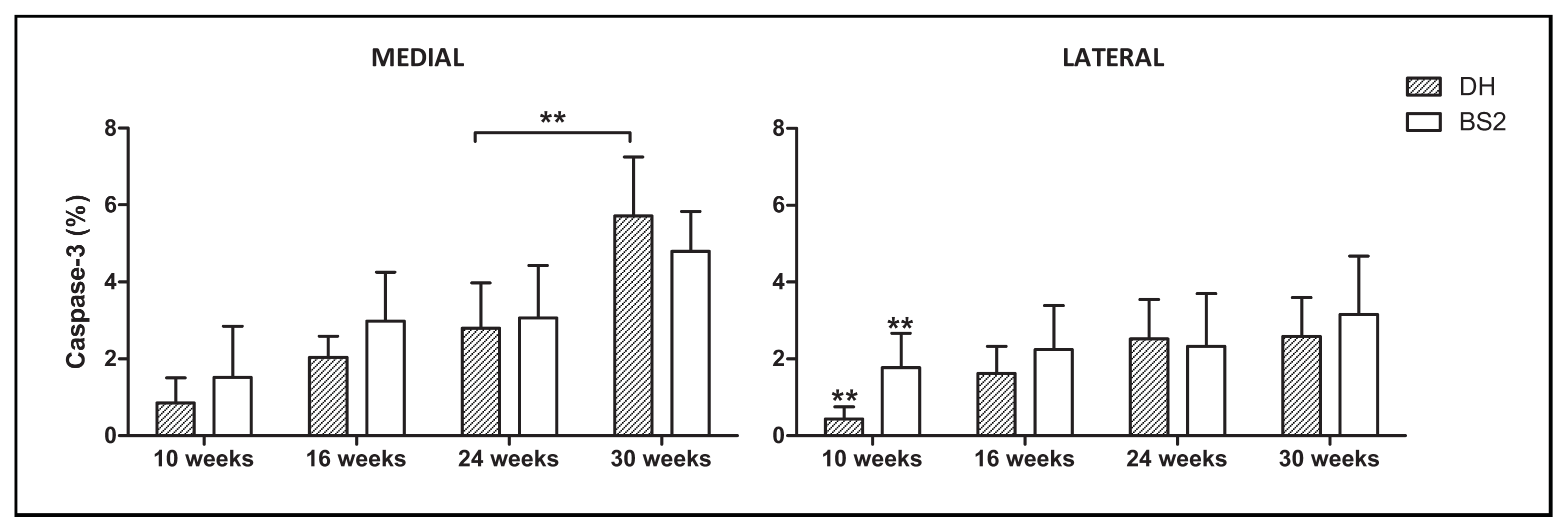
2.3. Chondrocyte Apoptosis
2.4. Correlation between Chondrocyte Apoptosis and AC Degradation
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Histology and Microscopic Scoring
4.3. Caspase-3 Staining
4.4. Terminal Deoxyribonucleotidyl Transferase (TdT)-Mediated dUTP Nick-End Labelling (TUNEL) Staining
4.5. Statistics
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Mistry, D.; Oue, Y.; Chambers, M.G.; Kayser, M.V.; Mason, R.M. Chondrocyte death during murine osteoarthritis. Osteoarthr. Cartil 2004, 12, 131–141. [Google Scholar]
- Kim, H.A.; Lee, Y.J.; Seong, S.C.; Choe, K.W.; Song, Y.W. Apoptotic chondrocyte death in human osteoarthritis. J. Rheumatol 2000, 27, 455–462. [Google Scholar]
- Thomas, C.M.; Fuller, C.J.; Whittles, C.E.; Sharif, M. Chondrocyte death by apoptosis is associated with cartilage matrix degradation. Osteoarthr. Cartil 2007, 15, 27–34. [Google Scholar]
- Zemmyo, M.; Meharra, E.J.; Kühn, K.; Creighton-Achermann, L.; Lotz, M. Accelerated, aging-dependent development of osteoarthritis in α1 integrin-deficient mice. Arthritis Rheum 2003, 48, 2873–2880. [Google Scholar]
- Jimenez, P.A.; Glasson, S.S.; Trubetskoy, O.V.; Haimes, H.B. Spontaneous osteoarthritis in Dunkin Hartley guinea pigs: Histologic, radiologic, and biochemical changes. Lab. Anim. Sci 1997, 47, 598–601. [Google Scholar]
- Mobasheri, A. Role of chondrocyte death and hypocellularity in ageing human articular cartilage and the pathogenesis of osteoarthritis. Med. Hypotheses 2002, 58, 193–197. [Google Scholar]
- Todd Allen, R.; Robertson, C.M.; Harwood, F.L.; Sasho, T.; Williams, S.K.; Pomerleau, A.C.; Amiel, D. Characterization of mature vs aged rabbit articular cartilage: Analysis of cell density, apoptosis-related gene expression and mechanisms controlling chondrocyte apoptosis. Osteoarthr. Cartil 2004, 12, 917–923. [Google Scholar]
- Hashimoto, S.; Ochs, R.L.; Rosen, F.; Quach, J.; McCabe, G.; Solan, J.; Seegmiller, J.E.; Terkeltaub, R.; Lotz, M. Chondrocyte-derived apoptotic bodies and calcification of articular cartilage. Proc. Natl. Acad. Sci. USA 1998, 95, 3094–3099. [Google Scholar]
- Adams, C.S.; Horton, W.E., Jr. Chondrocyte apoptosis increases with age in the articular cartilage of adult animals. Anat. Rec. 1998, 250, 418–425. [Google Scholar]
- D’Lima, D.D.; Hashimoto, S.; Chen, P.C.; Colwell, C.W., Jr; Lotz, M.K. Human chondrocyte apoptosis in response to mechanical injury. Osteoarthr. Cartil. 2001, 9, 712–719. [Google Scholar]
- Loening, A.M.; James, I.E.; Levenston, M.E.; Badger, A.M.; Frank, E.H.; Kurz, B. Injurious mechanical compression of bovine articular cartilage induces chondrocyte apoptosis. Arch. Biochem. Biophys 2000, 381, 205–212. [Google Scholar]
- Del Carlo, M.; Loeser, R. Cell death in osteoarthritis. Curr. Rheumatol. Rep 2008, 10, 37–42. [Google Scholar]
- Hashimoto, S.; Ochs, R.L.; Komiya, S.; Lotz, M. Linkage of chondrocyte apoptosis and cartilage degradation in human osteoarthritis. Arthritis Rheum 1998, 41, 1632–1638. [Google Scholar]
- Wei, L.; Sun, X-J.; Wang, Z.; Chen, Q. CD95-induced osteoarthritic chondrocyte apoptosis and necrosis: Dependency on p38 mitogen-activated protein kinase. Arthritis Res. Ther. 2006, 8, R37. [Google Scholar]
- Roach, H.I.; Aigner, T.; Kouri, J.B. Chondroptosis: A variant of apoptotic cell death in chondrocytes? Apoptosis 2004, 9, 265–277. [Google Scholar]
- Perez, H.E.; Luna, M.J.; Rojas, M.L.; Kouri, J.B. Chondroptosis: An immunohistochemical study of apoptosis and Golgi complex in chondrocytes from human osteoarthritic cartilage. Apoptosis 2005, 10, 1105–1110. [Google Scholar]
- Blanco, F.J.; Guitian, R.; Vazquez-Martul, E.; de Toro, F.J.; Galdo, F. Osteoarthritis chondrocytes die by apoptosis. A possible pathway for osteoarthritis pathology. Arthritis Rheum 1998, 41, 284–289. [Google Scholar]
- Sharif, M.; Whitehouse, A.; Sharman, P.; Perry, M.; Adams, M. Increased apoptosis in human osteoarthritic cartilage corresponds to reduced cell density and expression of caspase-3. Arthritis Rheum 2004, 50, 507–515. [Google Scholar]
- Matsuo, M.; Nishida, K.; Yoshida, A.; Murakami, T.; Inoue, H. Expression of caspase-3 and -9 relevant to cartilage destruction and chondrocyte apoptosis in human osteoarthritic cartilage. Acta Med. Okayama 2001, 55, 333–340. [Google Scholar]
- Héraud, F.; Héraud, A.; Harmand, M.-F. Apoptosis in normal and osteoarthritic human articular cartilage. Ann. Rheum. Dis 2000, 59, 959–965. [Google Scholar]
- Thomas, C.M.; Murray, R.; Sharif, M. Chondrocyte apoptosis determined by caspase-3 expression varies with fibronectin distribution in equine articular cartilage. Int. J. Rheum. Dis 2011, 14, 290–297. [Google Scholar]
- Almonte-Becerril, M.; Navarro-Garcia, F.; Gonzalez-Robles, A.; Vega-Lopez, M.; Lavalle, C.; Kouri, J. Cell death of chondrocytes is a combination between apoptosis and autophagy during the pathogenesis of Osteoarthritis within an experimental model. Apoptosis 2010, 15, 631–638. [Google Scholar]
- Taniguchi, S.; Ryu, J.; Seki, M.; Sumino, T.; Tokuhashi, Y.; Esumi, M. Long-term oral administration of glucosamine or chondroitin sulfate reduces destruction of cartilage and up-regulation of MMP-3 mRNA in a model of spontaneous osteoarthritis in Hartley guinea pigs. J. Orthop. Res 2012, 30, 673–678. [Google Scholar]
- Lo, M.Y.; Kim, H.T. Chondrocyte apoptosis induced by collagen degradation: Inhibition by caspase inhibitors and IGF-1. J. Orthop. Res 2004, 22, 140–144. [Google Scholar]
- Turunen, M.J.; Töyräs, J.; Lammi, M.J.; Jurvelin, J.S.; Korhonen, R.K. Hyperosmolaric contrast agents in cartilage tomography may expose cartilage to overload-induced cell death. J. Biomech 2012, 45, 497–503. [Google Scholar]
- Clements, K.M.; Bee, Z.C.; Crossingham, G.V.; Adams, M.A.; Sharif, M. How severe must repetitive loading be to kill chondrocytes in articular cartilage? Osteoarthr. Cartil 2001, 9, 499–507. [Google Scholar]
- Pascual Garrido, C.; Hakimiyan, A.A.; Rappoport, L.; Oegema, T.R.; Wimmer, M.A.; Chubinskaya, S. Anti-apoptotic treatments prevent cartilage degradation after acute trauma to human ankle cartilage. Osteoarthr. Cartil 2009, 17, 1244–1251. [Google Scholar]
- D’Lima, D.; Hermida, J.; Hashimoto, S.; Colwell, C.; Lotz, M. Caspase inhibitors reduce severity of cartilage lesions in experimental osteoarthritis. Arthritis Rheum 2006, 54, 1814–1821. [Google Scholar]
- Nakagawa, S.; Arai, Y.; Mazda, O.; Kishida, T.; Takahashi, K.A.; Sakao, K.; Saito, M.; Honjo, K.; Imanishi, J.; Kubo, T. N-acetylcysteine prevents nitric oxide-induced chondrocyte apoptosis and cartilage degeneration in an experimental model of osteoarthritis. J. Orthop. Res 2010, 28, 156–163. [Google Scholar]
- Hashimoto, S.; Takahashi, K.; Amiel, D.; Coutts, R.D.; Lotz, M. Chondrocyte apoptosis and nitric oxide production during experimentally induced osteoarthritis. Arthritis Rheum 1998, 41, 1266–1274. [Google Scholar]
- Yatsugi, N.; Tsukazaki, T.; Osaki, M.; Koji, T.; Yamashita, S.; Shindo, H. Apoptosis of articular chondrocytes in rheumatoid arthritis and osteoarthritis: Correlation of apoptosis with degree of cartilage destruction and expression of apoptosis-related proteins of p53 and c-myc. J. Orthop. Sci 2000, 5, 150–156. [Google Scholar]
- Zamli, Z.; Sharif, M. Chondrocyte apoptosis: A cause or consequence of osteoarthritis? Int. J. Rheum. Dis 2011, 14, 159–166. [Google Scholar]
- Bendele, A.M.; Hulman, J.F. Spontaneous cartilage degeneration in guinea pigs. Arthritis Rheum 1988, 31, 561–565. [Google Scholar]
- Huebner, J.L.; Hanes, M.A.; Beekman, B.; TeKoppele, J.M.; Kraus, V.B. A comparative analysis of bone and cartilage metabolism in two strains of guinea-pig with varying degrees of naturally occurring osteoarthritis. Osteoarthr. Cartil 2002, 10, 758–767. [Google Scholar]
- Muraoka, T.; Hagino, H.; Okano, T.; Enokida, M.; Teshima, R. Role of subchondral bone in osteoarthritis development: A comparative study of two strains of guinea pigs with and without spontaneously occurring osteoarthritis. Arthritis Rheum 2007, 56, 3366–3374. [Google Scholar]
- Anderson-MacKenzie, J.M.; Quasnichka, H.L.; Starr, R.L.; Lewis, E.J.; Billingham, M.E.; Bailey, A.J. Fundamental subchondral bone changes in spontaneous knee osteoarthritis. Int. J. Biochem. Cell Biol 2005, 37, 224–236. [Google Scholar]
- Quasnichka, H.L.; Anderson-MacKenzie, J.M.; Tarlton, J.F.; Sims, T.J.; Billingham, M.E.J.; Bailey, A.J. Cruciate ligament laxity and femoral intercondylar notch narrowing in early-stage knee osteoarthritis. Arthritis Rheum 2005, 52, 3100–3109. [Google Scholar]
- Knott, L.; Avery, N.C.; Hollander, A.P.; Tarlton, J.F. Regulation of osteoarthritis by omega-3 (n-3) polyunsaturated fatty acids in a naturally occurring model of disease. Osteoarthr. Cartil 2011, 19, 1150–1157. [Google Scholar]
- Thomsen, J.; Straarup, T.; Danielsen, C.; Oxlund, H.; Brüel, A. Relationship between articular cartilage damage and subchondral bone properties and meniscal ossification in the Dunkin Hartley guinea pig model of osteoarthritis. Scand. J. Rheumatol 2011, 40, 391–399. [Google Scholar]
- Huebner, J.L.; Williams, J.M.; Deberg, M.; Henrotin, Y.; Kraus, V.B. Collagen fibril disruption occurs early in primary guinea pig knee osteoarthritis. Osteoarthr. Cartil 2010, 18, 397–405. [Google Scholar]
- De Bri, E.; Reinholt, F.P.; Svensson, O. Primary osteoarthrosis in guinea pigs: A stereological study. J. Orthop. Res 1995, 13, 769–776. [Google Scholar]
- Osborne, D.; Woodhouse, S.; Meacock, R. Early changes in the sulfation of chondroitin in guinea-pig articular cartilage, a possible predictor of osteoarthritis. Osteoarthr. Cartil 1994, 2, 215–223. [Google Scholar]
- Johnson, K.; Svensson, C.I.; Etten, D.V.; Ghosh, S.S.; Murphy, A.N.; Powell, H.C.; Terkeltaub, R. Mediation of spontaneous knee osteoarthritis by progressive chondrocyte ATP depletion in Hartley guinea pigs. Arthritis Rheum 2004, 50, 1216–1225. [Google Scholar]
- Tessier, J.J.; Bowyer, J.; Brownrigg, N.J.; Peers, I.S.; Westwood, F.R.; Waterton, J.C.; Maciewicz, R.A. Characterisation of the guinea pig model of osteoarthritis by in vivo three-dimensional magnetic resonance imaging. Osteoarthr. Cartil 2003, 11, 845–853. [Google Scholar]
- Noyes, F.R.; Schipplein, O.D.; Andriacchi, T.P.; Saddemi, S.R.; Weise, M. The anterior cruciate ligament-deficient knee with varus alignment. Am. J. Sports Med 1992, 20, 707–716. [Google Scholar]
- Hughston, J.C.; Andrews, J.R.; Cross, M.J.; Moschi, A. Classification of knee ligament instabilities. Part I. The medial compartment and cruciate ligaments. J. Bone Jt. Surg. Am 1976, 58, 159–172. [Google Scholar]
- Pelletier, J.-P.; Jovanovic, D.V.; Lascau-Coman, V.; Fernandes, J.C.; Manning, P.T.; Connor, J.R.; Currie, M.G.; Di Battista, J.A.; Martel-Pelletier, J. Selective inhibition of inducible nitric oxide synthase reduces progression of experimental osteoarthritis in vivo: Possible link with the reduction in chondrocyte apoptosis and caspase 3 level. Arthritis Rheum 2000, 43, 1290–1299. [Google Scholar]
- Chen, C.-T.; Bhargava, M.; Lin, P.M.; Torzilli, P.A. Time, stress, and location dependent chondrocyte death and collagen damage in cyclically loaded articular cartilage. J. Orthop. Res 2003, 21, 888–898. [Google Scholar]
- Chen, C.-T.; Burton-Wurster, N.; Borden, C.; Hueffer, K.; Bloom, S.E.; Lust, G. Chondrocyte necrosis and apoptosis in impact damaged articular cartilage. J. Orthop. Res 2001, 19, 703–711. [Google Scholar]
- Ewers, B.J.; Dvoracek-Driksna, D.; Orth, M.W.; Haut, R.C. The extent of matrix damage and chondrocyte death in mechanically traumatized articular cartilage explants depends on rate of loading. J. Orthop. Res 2001, 19, 779–784. [Google Scholar]
- Quinn, T.M.; Allen, R.G.; Schalet, B.J.; Perumbuli, P.; Hunziker, E.B. Matrix and cell injury due to sub-impact loading of adult bovine articular cartilage explants: Effects of strain rate and peak stress. J. Orthop. Res 2001, 19, 242–249. [Google Scholar]
- Ding, M.; Danielsen, C.; Hvid, I. Age-related three-dimensional microarchitectural adaptations of subchondral bone tissues in guinea pig primary osteoarthrosis. Calcif. Tissue Int 2006, 78, 113–122. [Google Scholar]
- Wong, A.H.C.; Gottesman, I.I.; Petronis, A. Phenotypic differences in genetically identical organisms: The epigenetic perspective. Hum. Mol. Genet 2005, 14, R11–R18. [Google Scholar]
- Bendele, A.M.; Hulman, J.F. Effects of body weight restriction on the development and progression of spontaneous osteoarthritis in guinea pigs. Arthritis Rheum 1991, 34, 1180–1184. [Google Scholar]
- Kawamoto, T. Use of a new adhesive film for the preparation of multi-purpose fresh-frozen sections from hard tissues, whole-animals, insects and plants. Arch. Histol. Cytol 2003, 66, 123–143. [Google Scholar]
- Kraus, V.B.; Huebner, J.L.; DeGroot, J.; Bendele, A. The OARSI histopathology initiative-recommendations for histological assessments of osteoarthritis in the guinea pig. Osteoarthr. Cartil 2010, 18, S35–S52. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zamli, Z.; Adams, M.A.; Tarlton, J.F.; Sharif, M. Increased Chondrocyte Apoptosis Is Associated with Progression of Osteoarthritis in Spontaneous Guinea Pig Models of the Disease. Int. J. Mol. Sci. 2013, 14, 17729-17743. https://doi.org/10.3390/ijms140917729
Zamli Z, Adams MA, Tarlton JF, Sharif M. Increased Chondrocyte Apoptosis Is Associated with Progression of Osteoarthritis in Spontaneous Guinea Pig Models of the Disease. International Journal of Molecular Sciences. 2013; 14(9):17729-17743. https://doi.org/10.3390/ijms140917729
Chicago/Turabian StyleZamli, Zaitunnatakhin, Michael A. Adams, John F. Tarlton, and Mohammed Sharif. 2013. "Increased Chondrocyte Apoptosis Is Associated with Progression of Osteoarthritis in Spontaneous Guinea Pig Models of the Disease" International Journal of Molecular Sciences 14, no. 9: 17729-17743. https://doi.org/10.3390/ijms140917729
APA StyleZamli, Z., Adams, M. A., Tarlton, J. F., & Sharif, M. (2013). Increased Chondrocyte Apoptosis Is Associated with Progression of Osteoarthritis in Spontaneous Guinea Pig Models of the Disease. International Journal of Molecular Sciences, 14(9), 17729-17743. https://doi.org/10.3390/ijms140917729



