MAPKs and Signal Transduction in the Control of Gastrointestinal Epithelial Cell Proliferation and Differentiation
Abstract
:1. Introduction
2. MAPK Signaling Pathway
2.1. ERK Pathway
2.2. JNK Pathway
2.3. p38 Pathway
2.4. ERK5 Pathway
3. Crosstalk with Other Signaling Pathways
4. MAPK and Epithelial Cell Proliferation and Differentiation
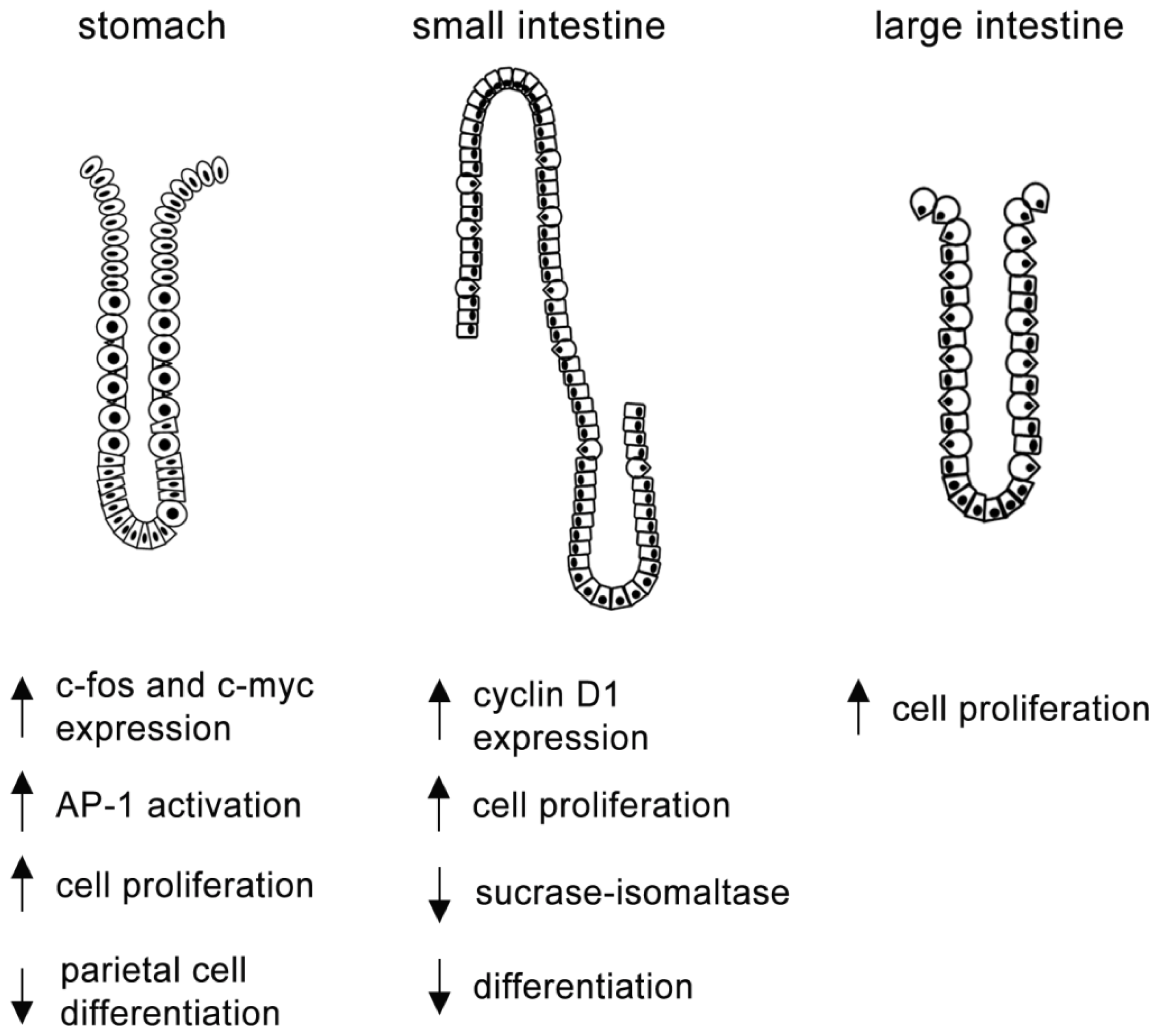
4.1. MAPKs and Gastric Cells
4.2. MAPKs and Intestinal Cells
5. Nutritional Influence on Cell Signaling
6. Conclusion
Conflict of Interest
References
- Avruch, J. Map kinase pathways: The first twenty years. Biochim. Biophys. Acta 2007, 1773, 1150–1160. [Google Scholar]
- Wagner, E.F.; Nebreda, A.R. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat. Rev. Cancer 2009, 9, 537–549. [Google Scholar]
- Asrih, M.; Mach, F.; Nencioni, A.; Dallegri, F.; Quercioli, A.; Montecucco, F. Role of mitogen-activated protein kinase pathways in multifactorial adverse cardiac remodeling associated with metabolic syndrome. Mediators Inflamm 2013, 2013, e367245. [Google Scholar]
- Schlessinger, J. Common and distinct elements in cellular signaling via EGF and FGF receptors. Science 2004, 306, 1506–1507. [Google Scholar]
- Kyriakis, J.M.; Avruch, J. Mammalian MAPK signal transduction pathways activated by stress and inflammation: A 10-year update. Physiol. Rev 2012, 92, 689–737. [Google Scholar]
- Payne, D.M.; Rossomando, A.J.; Martino, P.; Erickson, A.K.; Her, J.H.; Shabanowitz, J.; Hunt, D.F.; Weber, M.J.; Sturgill, T.W. Identification of the regulatory phosphorylation sites in pp42/mitogen-activated protein kinase (MAP kinase). EMBO J 1991, 10, 885–892. [Google Scholar]
- Biondi, R.M.; Nebreda, A.R. Signalling specificity of Ser/Thr protein kinases through docking-site-mediated interactions. Biochem. J 2003, 372, 1–13. [Google Scholar]
- Tanoue, T.; Adachi, M.; Moriguchi, T.; Nishida, E. A conserved docking motif in MAP kinases common to substrates, activators and regulators. Nat. Cell Biol 2000, 2, 110–116. [Google Scholar]
- Pan, C.Q.; Sudol, M.; Sheetz, M.; Low, B.C. Modularity and functional plasticity of scaffold proteins as p(l)acemakers in cell signaling. Cell. Signal 2012, 24, 2143–2165. [Google Scholar]
- Brown, M.D.; Sacks, D.B. Protein scaffolds in map kinase signalling. Cell. Signal 2009, 21, 462–469. [Google Scholar]
- Owens, D.M.; Keyse, S.M. Differential regulation of map kinase signalling by dual-specificity protein phosphatases. Oncogene 2007, 26, 3203–3213. [Google Scholar]
- Andreadi, C.; Noble, C.; Patel, B.; Jin, H.; Aguilar Hernandez, M.M.; Balmanno, K.; Cook, S.J.; Pritchard, C. Regulation of MEK/ERK pathway output by subcellular localization of B-Raf. Biochem. Soc. Trans 2012, 40, 67–72. [Google Scholar]
- Marshall, C.J. Specificity of receptor tyrosine kinase signaling: Transient versus sustained extracellular signal-regulated kinase activation. Cell 1995, 80, 179–185. [Google Scholar]
- Yang, S.H.; Sharrocks, A.D.; Whitmarsh, A.J. Map kinase signalling cascades and transcriptional regulation. Gene 2013, 513, 1–13. [Google Scholar]
- Xiao, Z.Q.; Li, J.; Majumdar, A.P. Regulation of TGF-alpha-induced activation of AP-1 in the aging gastric mucosa. Am. J. Physiol. Gastrointest. Liver Physiol 2003, 285, G396–G403. [Google Scholar]
- Lv, Z.M.; Wang, Q.; Wan, Q.; Lin, J.G.; Hu, M.S.; Liu, Y.X.; Wang, R. The role of the p38 MAPK signaling pathway in high glucose-induced epithelial-mesenchymal transition of cultured human renal tubular epithelial cells. PLoS One 2011, 6, e22806. [Google Scholar]
- Yoon, S.; Seger, R. The extracellular signal-regulated kinase: Multiple substrates regulate diverse cellular functions. Growth Factors 2006, 24, 21–44. [Google Scholar]
- Ramos, J.W. The regulation of extracellular signal-regulated kinase (ERK) in mammalian cells. Int. J. Biochem. Cell Biol 2008, 40, 2707–2719. [Google Scholar]
- Seki, E.; Brenner, D.A.; Karin, M. A liver full of JNK: Signaling in regulation of cell function and disease pathogenesis, and clinical approaches. Gastroenterology 2012, 143, 307–320. [Google Scholar]
- Cuenda, A.; Rousseau, S. p38 MAP-kinases pathway regulation, function and role in human diseases. Biochim. Biophys. Acta 2007, 1773, 1358–1375. [Google Scholar]
- Cuadrado, A.; Nebreda, A.R. Mechanisms and functions of p38 MAPK signalling. Biochem. J 2010, 429, 403–417. [Google Scholar]
- Lee, J.D.; Ulevitch, R.J.; Han, J. Primary structure of BMK1: A new mammalian map kinase. Biochem. Biophys. Res. Commun 1995, 213, 715–724. [Google Scholar]
- Zhou, G.; Bao, Z.Q.; Dixon, J.E. Components of a new human protein kinase signal transduction pathway. J. Biol. Chem 1995, 270, 12665–12669. [Google Scholar]
- Nithianandarajah-Jones, G.N.; Wilm, B.; Goldring, C.E.; Müller, J.; Cross, M.J. ERK5: Structure, regulation and function. Cell. Signal 2012, 24, 2187–2196. [Google Scholar]
- Kato, Y.; Kravchenko, V.V.; Tapping, R.I.; Han, J.; Ulevitch, R.J.; Lee, J.D. BMK1/ERK5 regulates serum-induced early gene expression through transcription factor MEF2C. EMBO J 1997, 16, 7054–7066. [Google Scholar]
- Junttila, M.R.; Li, S.P.; Westermarck, J. Phosphatase-mediated crosstalk between MAPK signaling pathways in the regulation of cell survival. FASEB J 2008, 22, 954–965. [Google Scholar]
- Fey, D.; Croucher, D.R.; Kolch, W.; Kholodenko, B.N. Crosstalk and signaling switches in mitogen-activated protein kinase cascades. Front. Physiol. 2012, 3. [Google Scholar] [CrossRef]
- Liu, Q.; Hofmann, P.A. Protein phosphatase 2A-mediated cross-talk between p38 MAPK and ERK in apoptosis of cardiac myocytes. Am. J. Physiol. Heart Circ. Physiol 2004, 286, H2204–H2212. [Google Scholar]
- Grethe, S.; Coltella, N.; di Renzo, M.F.; Pörn-Ares, M.I. p38 MAPK downregulates phosphorylation of Bad in doxorubicin-induced endothelial apoptosis. Biochem. Biophys. Res. Commun 2006, 347, 781–790. [Google Scholar]
- Lee, J.; Hong, F.; Kwon, S.; Kim, S.S.; Kim, D.O.; Kang, H.S.; Lee, S.J.; Ha, J. Activation of p38 MAPK induces cell cycle arrest via inhibition of Raf/ERK pathway during muscle differentiation. Biochem. Biophys. Res. Commun 2002, 298, 765–771. [Google Scholar]
- Li, S.P.; Junttila, M.R.; Han, J.; Kähäri, V.M.; Westermarck, J. p38 mitogen-activated protein kinase pathway suppresses cell survival by inducing dephosphorylation of mitogen-activated protein/extracellular signal-regulated kinase kinase1,2. Cancer Res 2003, 63, 3473–3477. [Google Scholar]
- Monick, M.M.; Powers, L.S.; Gross, T.J.; Flaherty, D.M.; Barrett, C.W.; Hunninghake, G.W. Active ERK contributes to protein translation by preventing JNK-dependent inhibition of protein phosphatase 1. J. Immunol 2006, 177, 1636–1645. [Google Scholar]
- Zhang, Q.G.; Wang, R.M.; Han, D.; Yang, L.C.; Li, J.; Brann, D.W. Preconditioning neuroprotection in global cerebral ischemia involves NMDA receptor-mediated ERK-JNK3 crosstalk. Neurosci. Res 2009, 63, 205–212. [Google Scholar]
- Peng, T.; Zhang, T.; Lu, X.; Feng, Q. JNK1/c-fos inhibits cardiomyocyte TNF-α expression via a negative crosstalk with ERK and p38 MAPK in endotoxaemia. Cardiovasc. Res 2009, 81, 733–741. [Google Scholar]
- Aksamitiene, E.; Kiyatkin, A.; Kholodenko, B.N. Cross-talk between mitogenic Ras/MAPK and survival PI3K/Akt pathways: A fine balance. Biochem. Soc. Trans 2012, 40, 139–146. [Google Scholar]
- Barthwal, M.K.; Sathyanarayana, P.; Kundu, C.N.; Rana, B.; Pradeep, A.; Sharma, C.; Woodgett, J.R.; Rana, A. Negative regulation of mixed lineage kinase 3 by protein kinase B/Akt leads to cell survival. J. Biol. Chem 2003, 278, 3897–3902. [Google Scholar]
- Kim, A.H.; Khursigara, G.; Sun, X.; Franke, T.F.; Chao, M.V. Akt phosphorylates and negatively regulates apoptosis signal-regulating kinase 1. Mol. Cell Biol 2001, 21, 893–901. [Google Scholar]
- Vazquez de la Torre, A.; Junyent, F.; Folch, J.; Pelegri, C.; Vilaplana, J.; Auladell, C.; Beas-Zarate, C.; Pallas, M.; Verdaguer, E.; Camins, A. PI3 K/Akt inhibition induces apoptosis through p38 activation in neurons. Pharmacol. Res 2013, 70, 116–125. [Google Scholar]
- Hartsough, M.T.; Mulder, K.M. Transforming growth factor beta activation of p44MAPK in proliferating cultures of epithelial cells. J. Biol. Chem 1995, 270, 7117–7124. [Google Scholar]
- Hough, C.; Radu, M.; Doré, J.J. TGF-beta induced Erk phosphorylation of smad linker region regulates smad signaling. PLoS One 2012, 7, e42513. [Google Scholar]
- Tian, Y.C.; Chen, Y.C.; Chang, C.T.; Hung, C.C.; Wu, M.S.; Phillips, A.; Yang, C.W. Epidermal growth factor and transforming growth factor-β1 enhance HK-2 cell migration through a synergistic increase of matrix metalloproteinase and sustained activation of ERK signaling pathway. Exp. Cell Res 2007, 313, 2367–2377. [Google Scholar]
- Browne, J.A.; Pearson, A.L.; Zahr, R.A.; Niculescu-Duvaz, I.; Baines, D.L.; Dockrell, M.E. TGF-β activates ERK5 in human renal epithelial cells. Biochem. Biophys. Res. Commun 2008, 373, 440–444. [Google Scholar]
- Marchetti, A.; Colletti, M.; Cozzolino, A.M.; Steindler, C.; Lunadei, M.; Mancone, C.; Tripodi, M. ERK5/MAPK is activated by TGFbeta in hepatocytes and required for the GSK-3beta-mediated Snail protein stabilization. Cell. Signal 2008, 20, 2113–2118. [Google Scholar]
- Joko, T.; Shiraishi, A.; Akune, Y.; Tokumaru, S.; Kobayashi, T.; Miyata, K.; Ohashi, Y. Involvement of P38MAPK in human corneal endothelial cell migration induced by TGF-β(2). Exp. Eye Res 2012, 108C, 23–32. [Google Scholar]
- Kretzschmar, M.; Liu, F.; Hata, A.; Doody, J.; Massague, J. The TGF-beta family mediator Smad1 is phosphorylated directly and activated functionally by the BMP receptor kinase. Genes Dev 1997, 11, 984–995. [Google Scholar]
- Kretzschmar, M.; Doody, J.; Timokhina, I.; Massague, J. A mechanism of repression of TGFβ/Smad signaling by oncogenic Ras. Genes Dev 1999, 13, 804–816. [Google Scholar]
- Kretzschmar, M.; Doody, J.; Massague, J. Opposing BMP and EGF signalling pathways converge on the TGF-β family mediator Smad1. Nature 1997, 389, 618–622. [Google Scholar]
- Červenka, I.; Wolf, J.; Mašek, J.; Krejci, P.; Wilcox, W.R.; Kozubík, A.; Schulte, G.; Gutkind, J.S.; Bryja, V. Mitogen-activated protein kinases promote WNT/β-catenin signaling via phosphorylation of LRP6. Mol. Cell Biol 2011, 31, 179–189. [Google Scholar]
- Krejci, P.; Aklian, A.; Kaucka, M.; Sevcikova, E.; Prochazkova, J.; Masek, J.K.; Mikolka, P.; Pospisilova, T.; Spoustova, T.; Weis, M.; et al. Receptor tyrosine kinases activate canonical WNT/β-catenin signaling via MAP kinase/LRP6 pathway and direct β-catenin phosphorylation. PLoS One 2012, 7, e35826. [Google Scholar]
- Biechele, T.L.; Kulikauskas, R.M.; Toroni, R.A.; Lucero, O.M.; Swift, R.D.; James, R.G.; Robin, N.C.; Dawson, D.W.; Moon, R.T.; Chien, A.J. Wnt/β-catenin signaling and AXIN1 regulate apoptosis triggered by inhibition of the mutant kinase BRAFV600E in human melanoma. Sci. Signal. 2012, 5. [Google Scholar] [CrossRef]
- Bikkavilli, R.K.; Feigin, M.E.; Malbon, C.C. p38 mitogen-activated protein kinase regulates canonical Wnt-beta-catenin signaling by inactivation of GSK3β. J. Cell Sci 2008, 121, 3598–3607. [Google Scholar]
- Bikkavilli, R.K.; Feigin, M.E.; Malbon, C.C. Gαo mediates WNT-JNK signaling through dishevelled 1 and 3, RhoA family members, and MEKK 1 and 4 in mammalian cells. J. Cell Sci 2008, 121, 234–245. [Google Scholar]
- Singh, A.M.; Reynolds, D.; Cliff, T.; Ohtsuka, S.; Mattheyses, A.L.; Sun, Y.; Menendez, L.; Kulik, M.; Dalton, S. Signaling network crosstalk in human pluripotent cells: A Smad2/3-regulated switch that controls the balance between self-renewal and differentiation. Cell Stem Cell 2012, 10, 312–326. [Google Scholar]
- Rivard, N.; Boucher, M.J.; Asselin, C.; L’Allemain, G. MAP kinase cascade is required for p27 downregulation and S phase entry in fibroblasts and epithelial cells. Am. J. Physiol 1999, 277, C652–C664. [Google Scholar]
- Coleman, M.L.; Marshall, C.J.; Olson, M.F. RAS and RHO GTPases in G1-phase cell-cycle regulation. Nat. Rev. Mol. Cell Biol 2004, 5, 355–366. [Google Scholar]
- Chambard, J.C.; Lefloch, R.; Pouysségur, J.; Lenormand, P. ERK implication in cell cycle regulation. Biochim. Biophys. Acta 2007, 1773, 1299–1310. [Google Scholar]
- Huang, K.T.; Pavlides, S.C.; Lecanda, J.; Blank, S.V.; Mittal, K.R.; Gold, L.I. Estrogen and progesterone regulate p27kip1 levels via the ubiquitin-proteasome system: Pathogenic and therapeutic implications for endometrial cancer. PLoS One 2012, 7, e46072. [Google Scholar]
- Ullrich, A.; Schlessinger, J. Signal transduction by receptors with tyrosine kinase activity. Cell 1990, 61, 203–212. [Google Scholar]
- Kholodenko, B.N. Map kinase cascade signaling and endocytic trafficking: A marriage of convenience? Trends Cell Biol 2002, 12, 173–177. [Google Scholar]
- Ceresa, B.P. Spatial regulation of epidermal growth factor receptor signaling by endocytosis. Int. J. Mol. Sci 2012, 14, 72–87. [Google Scholar]
- Mills, J.C.; Shivdasani, R.A. Gastric epithelial stem cells. Gastroenterology 2011, 140, 412–424. [Google Scholar]
- Karam, S.M.; Leblond, C.P. Dynamics of epithelial cells in the corpus of the mouse stomach. I. Identification of proliferative cell types and pinpointing of the stem cell. Anat. Rec 1993, 236, 259–279. [Google Scholar]
- Pan, Q.; Nicholson, A.M.; Barr, H.; Harrison, L.A.; Wilson, G.D.; Burkert, J.; Jeffery, R.; Alison, M.R.; Looijenga, L.; Lin, W.R.; et al. Identification of lineage-uncommitted, long-lived, label-retaining cells in healthy human esophagus and stomach, and in metaplastic esophagus. Gastroenterology 2013, 144, 761–770. [Google Scholar]
- Bredemeyer, A.J.; Geahlen, J.H.; Weis, V.G.; Huh, W.J.; Zinselmeyer, B.H.; Srivatsan, S.; Miller, M.J.; Shaw, A.S.; Mills, J.C. The gastric epithelial progenitor cell niche and differentiation of the zymogenic (chief) cell lineage. Dev. Biol 2009, 325, 211–224. [Google Scholar]
- Barker, N.; Huch, M.; Kujala, P.; van de Wetering, M.; Snippert, H.J.; van Es, J.H.; Sato, T.; Stange, D.E.; Begthel, H.; van den Born, M.; et al. Lgr5+ve stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell 2010, 6, 25–36. [Google Scholar]
- Alvares, E.P. Extensive networks of tmpase positive basal lysosomes are present in fetal rat gastric epithelium before overt differentiation. J. Submicrosc. Cytol. Pathol 1994, 26, 515–523. [Google Scholar]
- Kim, B.M.; Buchner, G.; Miletich, I.; Sharpe, P.T.; Shivdasani, R.A. The stomach mesenchymal transcription factor Barx1 specifies gastric epithelial identity through inhibition of transient Wnt signaling. Dev. Cell 2005, 8, 611–622. [Google Scholar]
- Kiefer, S.M.; Robbins, L.; Rauchman, M. Conditional expression of Wnt9b in Six2-positive cells disrupts stomach and kidney function. PLoS One 2012, 7, e43098. [Google Scholar]
- Beauchamp, R.D.; Barnard, J.A.; McCutchen, C.M.; Cherner, J.A.; Coffey, R.J. Localization of transforming growth factor alpha and its receptor in gastric mucosal cells. Implications for a regulatory role in acid secretion and mucosal renewal. J. Clin. Invest 1989, 84, 1017–1023. [Google Scholar]
- Slomiany, B.L.; Liu, J.; Yao, P.; Wu-Wang, C.Y.; Keogh, J.P.; Wang, S.L.; Slomiany, A. Characterization of the epidermal growth factor receptor in the gastric mucosa. Digestion 1990, 47, 181–190. [Google Scholar]
- Osaki, L.H.; Curi, M.A.; Alvares, E.P.; Gama, P. Early weaning accelerates the differentiation of mucous neck cells in rat gastric mucosa: Possible role of TGFalpha/EGFR. Differentiation 2010, 79, 48–56. [Google Scholar]
- Osaki, L.H.; Figueiredo, P.M.; Alvares, E.P.; Gama, P. EGFR is involved in control of gastric cell proliferation through activation of MAPK and Src signalling pathways in early-weaned rats. Cell Prolif 2011, 44, 174–182. [Google Scholar]
- Alison, M.R.; Sarraf, C.E. The role of growth factors in gastrointestinal cell proliferation. Cell Biol. Int 1994, 18, 1–10. [Google Scholar]
- Hormi, K.; Lehy, T. Developmental expression of transforming growth factor-α and epidermal growth factor receptor proteins in the human pancreas and digestive tract. Cell Tissue Res 1994, 278, 439–450. [Google Scholar]
- Nakajima, N.; Kuwayama, H. Stimulatory effect of transforming growth factor-alpha on gastric epithelial cell migration through proliferation. J. Clin. Gastroenterol 1995, 21, S45–S49. [Google Scholar]
- Turner, J.R.; Liu, L.; Fligiel, S.E.; Jaszewski, R.; Majumdar, A.P. Aging alters gastric mucosal responses to epidermal growth factor and transforming growth factor-α. Am. J. Physiol. Gastrointest. Liver Physiol 2000, 278, G805–G810. [Google Scholar]
- Xiao, Z.Q.; Majumdar, A.P. Increased in vitro activation of EGFR by membrane-bound TGF-alpha from gastric and colonic mucosa of aged rats. Am. J. Physiol. Gastrointest. Liver Physiol 2001, 281, G111–G116. [Google Scholar]
- Pai, R.; Ohta, M.; Itani, R.M.; Sarfeh, I.J.; Tarnawski, A.S. Induction of mitogen-activated protein kinase signal transduction pathway during gastric ulcer healing in rats. Gastroenterology 1998, 114, 706–713. [Google Scholar]
- Coffey, R.J.; Washington, M.K.; Corless, C.L.; Heinrich, M.C. Ménétrier disease and gastrointestinal stromal tumors: Hyperproliferative disorders of the stomach. J. Clin. Invest 2007, 117, 70–80. [Google Scholar]
- Tétreault, M.P.; Chailler, P.; Beaulieu, J.F.; Rivard, N.; Ménard, D. Epidermal growth factor receptor-dependent PI3K-activation promotes restitution of wounded human gastric epithelial monolayers. J. Cell. Physiol 2008, 214, 545–557. [Google Scholar]
- Albeck, J.G.; Mills, G.B.; Brugge, J.S. Frequency-modulated pulses of ERK activity transmit quantitative proliferation signals. Mol. Cell 2013, 49, 249–261. [Google Scholar]
- Shin, S.H.; Park, S.Y.; Kang, G.H. Down-regulation of dual-specificity phosphatase 5 in gastric cancer by promoter CpG island hypermethylation and its potential role in carcinogenesis. Am. J. Pathol . 2013, 182, 1275–1285. [Google Scholar]
- Tureaud, J.; Sarkar, F.H.; Fligiel, S.E.; Kulkarni, S.; Jaszewski, R.; Reddy, K.; Yu, Y.; Majumdar, A.P. Increased expression of EGFR in gastric mucosa of aged rats. Am. J. Physiol 1997, 273, G389–G398. [Google Scholar]
- Nakamizo, H.; Suzuki, H.; Miura, S.; Mogami, S.; Kishikawa, H.; Yoshida, H.; Matsui, H.; Hibi, T. Transmural pressure loading enhances gastric mucosal cell proliferation. Dig. Dis. Sci 2012, 57, 2545–2554. [Google Scholar]
- Schneider, R.; Bornstein, S.R.; Chrousos, G.P.; Boxberger, S.; Ehninger, G.; Breidert, M. Leptin mediates a proliferative response in human gastric mucosa cells with functional receptor. Horm. Metab. Res 2001, 33, 1–6. [Google Scholar]
- Sasaki, E.; Tominaga, K.; Watanabe, T.; Fujiwara, Y.; Oshitani, N.; Matsumoto, T.; Higuchi, K.; Tarnawski, A.S.; Arakawa, T. COX-2 is essential for EGF induction of cell proliferation in gastric RGM1 cells. Dig. Dis. Sci 2003, 48, 2257–2262. [Google Scholar]
- Osawa, H.; Ohnishi, H.; Takano, K.; Noguti, T.; Mashima, H.; Hoshino, H.; Kita, H.; Sato, K.; Matsui, H.; Sugano, K. Sonic hedgehog stimulates the proliferation of rat gastric mucosal cells through ERK activation by elevating intracellular calcium concentration. Biochem. Biophys. Res. Commun 2006, 344, 680–687. [Google Scholar]
- Ohsawa, R.; Miyazaki, H.; Niisato, N.; Shiozaki, A.; Iwasaki, Y.; Otsuji, E.; Marunaka, Y. Intracellular chloride regulates cell proliferation through the activation of stress-activated protein kinases in MKN28 human gastric cancer cells. J. Cell. Physiol 2010, 223, 764–770. [Google Scholar]
- Ruggiero, P. Helicobacter pylori infection: What’s new. Curr. Opin. Infect. Dis 2012, 25, 337–344. [Google Scholar]
- Ding, S.Z.; Smith, M.F.; Goldberg, J.B. Helicobacter pylori and mitogen-activated protein kinases regulate the cell cycle, proliferation and apoptosis in gastric epithelial cells. J. Gastroenterol. Hepatol 2008, 23, e67–e78. [Google Scholar]
- Burdick, J.S.; Chung, E.; Tanner, G.; Sun, M.; Paciga, J.E.; Cheng, J.Q.; Washington, K.; Goldenring, J.R.; Coffey, R.J. Treatment of ménétrier’s disease with a monoclonal antibody against the epidermal growth factor receptor. N. Engl. J. Med 2000, 343, 1697–1701. [Google Scholar]
- Fiske, W.H.; Threadgill, D.; Coffey, R.J. Erbbs in the gastrointestinal tract: Recent progress and new perspectives. Exp. Cell Res 2009, 315, 583–601. [Google Scholar]
- Van den Brink, G.R.; Hardwick, J.C.; Tytgat, G.N.; Brink, M.A.; Ten Kate, F.J.; van Deventer, S.J.; Peppelenbosch, M.P. Sonic hedgehog regulates gastric gland morphogenesis in man and mouse. Gastroenterology 2001, 121, 317–328. [Google Scholar]
- Itoh, K.; Kataoka, H.; Sasaki, M.; Tanida, S.; Oshima, T.; Ogasawara, N.; Ohara, H.; Nakao, H.; Joh, T. Bone morphogenetic protein 2 induced differentiation toward superficial epithelial cells in the gastric mucosa. J. Gastroenterol 2006, 41, 1064–1075. [Google Scholar]
- Nyeng, P.; Norgaard, G.A.; Kobberup, S.; Jensen, J. FGF10 signaling controls stomach morphogenesis. Dev. Biol 2007, 303, 295–310. [Google Scholar]
- Keeley, T.M.; Samuelson, L.C. Cytodifferentiation of the postnatal mouse stomach in normal and Huntingtin-interacting protein 1-related-deficient mice. Am. J. Physiol. Gastrointest. Liver Physiol 2010, 299, G1241–G1251. [Google Scholar]
- Bockman, D.E.; Sharp, R.; Merlino, G. Regulation of terminal differentiation of zymogenic cells by transforming growth factor α in transgenic mice. Gastroenterology 1995, 108, 447–454. [Google Scholar]
- Fiorucci, S.; Lanfrancone, L.; Santucci, L.; Calabro, A.; Orsini, B.; Federici, B.; Morelli, A. Epidermal growth factor modulates pepsinogen secretion in guinea pig gastric chief cells. Gastroenterology 1996, 111, 945–958. [Google Scholar]
- Stepan, V.; Pausawasdi, N.; Ramamoorthy, S.; Todisco, A. The Akt and MAPK signal-transduction pathways regulate growth factor actions in isolated gastric parietal cells. Gastroenterology 2004, 127, 1150–1161. [Google Scholar]
- Chew, C.S.; Parente, J.A.; Zhou, C.; Baranco, E.; Chen, X. Lasp-1 is a regulated phosphoprotein within the cAMP signaling pathway in the gastric parietal cell. Am. J. Physiol 1998, 275, C56–C67. [Google Scholar]
- Chew, C.S.; Parente, J.A.; Chen, X.; Chaponnier, C.; Cameron, R.S. The LIM and SH3 domain-containing protein, lasp-1, may link the cAMP signaling pathway with dynamic membrane restructuring activities in ion transporting epithelia. J. Cell. Sci 2000, 113, 2035–2045. [Google Scholar]
- Kopic, S.; Murek, M.; Geibel, J.P. Revisiting the parietal cell. Am. J. Physiol. Cell Physiol 2010, 298, C1–C10. [Google Scholar]
- Todisco, A.; Pausawasdi, N.; Ramamoorthy, S.; del Valle, J.; van Dyke, R.W.; Askari, F.K. Functional role of protein kinase B/Akt in gastric acid secretion. J. Biol. Chem 2001, 276, 46436–46444. [Google Scholar]
- Barker, N.; van Es, J.H.; Kuipers, J.; Kujala, P.; van den Born, M.; Cozijnsen, M.; Haegebarth, A.; Korving, J.; Begthel, H.; Peters, P.J.; et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 2007, 449, 1003–1007. [Google Scholar]
- Barker, N.; van de Wetering, M.; Clevers, H. The intestinal stem cell. Genes Dev 2008, 22, 1856–1864. [Google Scholar]
- Sato, T.; van Es, J.H.; Snippert, H.J.; Stange, D.E.; Vries, R.G.; van den Born, M.; Barker, N.; Shroyer, N.F.; van de Wetering, M.; Clevers, H. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 2011, 469, 415–418. [Google Scholar]
- Van der Flier, L.G.; van Gijn, M.E.; Hatzis, P.; Kujala, P.; Haegebarth, A.; Stange, D.E.; Begthel, H.; van den Born, M.; Guryev, V.; Oving, I.; et al. Transcription factor achaete scute-like 2 controls intestinal stem cell fate. Cell 2009, 136, 903–912. [Google Scholar]
- Simons, B.D.; Clevers, H. Stem cell self-renewal in intestinal crypt. Exp. Cell Res 2011, 317, 2719–2724. [Google Scholar]
- Ireland, H.; Kemp, R.; Houghton, C.; Howard, L.; Clarke, A.R.; Sansom, O.J.; Winton, D.J. Inducible Cre-mediated control of gene expression in the murine gastrointestinal tract: Effect of loss of β-catenin. Gastroenterology 2004, 126, 1236–1246. [Google Scholar]
- Fevr, T.; Robine, S.; Louvard, D.; Huelsken, J. Wnt/β-catenin is essential for intestinal homeostasis and maintenance of intestinal stem cells. Mol. Cell Biol 2007, 27, 7551–7559. [Google Scholar]
- Schepers, A.; Clevers, H. Wnt signaling, stem cells, and cancer of the gastrointestinal tract. Cold Spring Harb. Perspect. Biol 2012, 4, a007989. [Google Scholar]
- Guardavaccaro, D.; Clevers, H. Wnt/β-catenin and MAPK signaling: Allies and enemies in different battlefields. Sci. Signal. 2012, 5, pe15. [Google Scholar]
- Schwitalla, S.; Fingerle, A.A.; Cammareri, P.; Nebelsiek, T.; Göktuna, S.I.; Ziegler, P.K.; Canli, O.; Heijmans, J.; Huels, D.J.; Moreaux, G.; et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell 2013, 152, 25–38. [Google Scholar]
- Aliaga, J.C.; Deschênes, C.; Beaulieu, J.F.; Calvo, E.L.; Rivard, N. Requirement of the map kinase cascade for cell cycle progression and differentiation of human intestinal cells. Am. J. Physiol 1999, 277, G631–G641. [Google Scholar]
- Singh, R.; Subramanian, S.; Rhodes, J.M.; Campbell, B.J. Peanut lectin stimulates proliferation of colon cancer cells by interaction with glycosylated CD44v6 isoforms and consequential activation of c-Met and MAPK: Functional implications for disease-associated glycosylation changes. Glycobiology 2006, 16, 594–601. [Google Scholar]
- Chiu, T.; Wu, S.S.; Santiskulvong, C.; Tangkijvanich, P.; Yee, H.F.; Rozengurt, E. Vasopressin-mediated mitogenic signaling in intestinal epithelial cells. Am. J. Physiol. Cell Physiol 2002, 282, C434–C450. [Google Scholar]
- Jasleen, J.; Shimoda, N.; Shen, E.R.; Tavakkolizadeh, A.; Whang, E.E.; Jacobs, D.O.; Zinner, M.J.; Ashley, S.W. Signaling mechanisms of glucagon-like peptide 2-induced intestinal epithelial cell proliferation. J. Surg. Res 2000, 90, 13–18. [Google Scholar]
- Jiang, R.; Lonnerdal, B. Apo- and holo-lactoferrin stimulate proliferation of mouse crypt cells but through different cellular signaling pathways. Int. J. Biochem. Cell Biol 2012, 44, 91–100. [Google Scholar]
- Wakeman, D.; Schneider, J.E.; Liu, J.; Wandu, W.S.; Erwin, C.R.; Guo, J.; Stappenbeck, T.S.; Warner, B.W. Deletion of p38-alpha mitogen-activated protein kinase within the intestinal epithelium promotes colon tumorigenesis. Surgery 2012, 152, 286–293. [Google Scholar]
- Suzuki, A.; Sekiya, S.; Gunshima, E.; Fujii, S.; Taniguchi, H. EGF signaling activates proliferation and blocks apoptosis of mouse and human intestinal stem/progenitor cells in long-term monolayer cell culture. Lab. Invest 2010, 90, 1425–1436. [Google Scholar]
- Malecka-Panas, E.; Relan, N.K.; Majumdar, A.P. Increased activation of EGF-receptor tyrosine kinase by EGF and TGF-alpha in the colonic mucosa of aged rats. J. Gerontol. A Biol. Sci. Med. Sci 1996, 51, B60–B65. [Google Scholar]
- Xiao, Z.Q.; Moragoda, L.; Jaszewski, R.; Hatfield, J.A.; Fligiel, S.E.; Majumdar, A.P. Aging is associated with increased proliferation and decreased apoptosis in the colonic mucosa. Mech. Ageing Dev 2001, 122, 1849–1864. [Google Scholar]
- Majumdar, A.P.; Du, J. Phosphatidylinositol 3-kinase/Akt signaling stimulates colonic mucosal cell survival during aging. Am. J. Physiol. Gastrointest. Liver Physiol 2006, 290, G49–G55. [Google Scholar]
- Houde, M.; Laprise, P.; Jean, D.; Blais, M.; Asselin, C.; Rivard, N. Intestinal epithelial cell differentiation involves activation of p38 mitogen-activated protein kinase that regulates the homeobox transcription factor CDx2. J. Biol. Chem 2001, 276, 21885–21894. [Google Scholar]
- Lorentz, O.; Duluc, I.; Arcangelis, A.D.; Simon-Assmann, P.; Kedinger, M.; Freund, J.N. Key role of the CDx2 homeobox gene in extracellular matrix-mediated intestinal cell differentiation. J. Cell Biol 1997, 139, 1553–1565. [Google Scholar]
- Lemieux, E.; Boucher, M.J.; Mongrain, S.; Boudreau, F.; Asselin, C.; Rivard, N. Constitutive activation of the MEK/ERK pathway inhibits intestinal epithelial cell differentiation. Am. J. Physiol. Gastrointest. Liver Physiol 2011, 301, G719–G730. [Google Scholar]
- Koldovský, O. Search for role of milk-borne biologically active peptides for the suckling. J. Nutr 1989, 119, 1543–1551. [Google Scholar]
- Gama, P.; Alvares, E.P. Early weaning and prolonged nursing induce changes in cell proliferation in the gastric epithelium of developing rats. J. Nutr 2000, 130, 2594–2598. [Google Scholar]
- de Andrade Sa, E.R.; Bitencourt, B.; Alvares, E.P.; Gama, P. In vivo effects of TGFbeta1 on the growth of gastric epithelium in suckling rats. Regul. Pept 2008, 146, 293–302. [Google Scholar]
- Ogias, D.; de Andrade Sa, E.R.; Alvares, E.P.; Gama, P. Opposite effects of fasting on TGF-beta3 and Tbetari distribution in the gastric mucosa of suckling and early weanling rats. Nutrition 2010, 26, 224–229. [Google Scholar]
- Dvorak, B. Milk epidermal growth factor and gut protection. J. Pediatr 2010, 156, S31–S35. [Google Scholar]
- Moran, J.R.; Courtney, M.E.; Orth, D.N.; Vaughan, R.; Coy, S.; Mount, C.D.; Sherrell, B.J.; Greene, H.L. Epidermal growth factor in human milk: Daily production and diurnal variation during early lactation in mothers delivering at term and at premature gestation. J. Pediatr 1983, 103, 402–405. [Google Scholar]
- Letterio, J.J.; Geiser, A.G.; Kulkarni, A.B.; Roche, N.S.; Sporn, M.B.; Roberts, A.B. Maternal rescue of transforming growth factor-beta 1 null mice. Science 1994, 264, 1936–1938. [Google Scholar]
- Penttila, I.A.; van Spriel, A.B.; Zhang, M.F.; Xian, C.J.; Steeb, C.B.; Cummins, A.G.; Zola, H.; Read, L.C. Transforming growth factor-beta levels in maternal milk and expression in postnatal rat duodenum and ileum. Pediatr. Res 1998, 44, 524–531. [Google Scholar]
- Lin, C.H.; Lyons, H.; Seelbach, M.S.; Tolia, V.; Vijesurier, R. Induction of gastric ornithine decarboxylase in early weaning rats. Digestion 2001, 63, 214–219. [Google Scholar]
- Morikawa, Y.; Ueyama, E.; Senba, E. Fasting-induced activation of mitogen-activated protein kinases (ERK/p38) in the mouse hypothalamus. J. Neuroendocrinol 2004, 16, 105–112. [Google Scholar]
- Ueyama, E.; Morikawa, Y.; Yasuda, T.; Senba, E. Attenuation of fasting-induced phosphorylation of mitogen-activated protein kinases (ERK/p38) in the mouse hypothalamus in response to refeeding. Neurosci. Lett 2004, 371, 40–44. [Google Scholar]
- Alvares, E.P.; Gama, P. Fasting enhances cell proliferation of gastric epithelium during the suckling period in rats. Braz. J. Med. Biol. Res 1993, 26, 869–873. [Google Scholar]
- Gama, P.; Alvares, E.P. Corticosterone treatment inhibits cell proliferation in the gastric epithelium of suckling rats. J. Gastroenterol 1998, 33, 32–38. [Google Scholar]
- Ogias, D.; de Andrade Sa, E.R.; Kasai, A.; Moisan, M.P.; Alvares, E.P.; Gama, P. Fasting differentially regulates plasma corticosterone-binding globulin, glucocorticoid receptor, and cell cycle in the gastric mucosa of pups and adult rats. Am. J. Physiol. Gastrointest. Liver Physiol 2010, 298, G117–G125. [Google Scholar]
- Angevine, K.R.; Wuescher, L.M.; Andrews, K.; Alexander, L.A.; McInerney, M.F.; Kieffer, T.J.; Mensah-Osman, E.J. Menin and GIP are inversely regulated by food intake and diet via PI3/AKT signaling in the proximal duodenum. Nutr. Diabetes 2012, 2, e55. [Google Scholar]
- Sabio, G.; Davis, R.J. cJun NH2-terminal kinase 1 (JNK1): Roles in metabolic regulation of insulin resistance. Trends Biochem. Sci 2010, 35, 490–496. [Google Scholar]
- Schultze, S.M.; Hemmings, B.A.; Niessen, M.; Tschopp, O. PI3K/AKT, MAPK and AMPK signalling: Protein kinases in glucose homeostasis. Expert Rev. Mol. Med 2012, 14, e1. [Google Scholar]
- Nautiyal, J.; Banerjee, S.; Kanwar, S.S.; Yu, Y.; Patel, B.B.; Sarkar, F.H.; Majumdar, A.P. Curcumin enhances dasatinib-induced inhibition of growth and transformation of colon cancer cells. Int. J. Cancer 2011, 128, 951–961. [Google Scholar]
- Fenton, J.I.; McCaskey, S.J. Curcumin and docosahexaenoic acid block insulin-induced colon carcinoma cell proliferation. Prostaglandins Leukot. Essent. Fatty Acids 2013, 88, 219–226. [Google Scholar]
- Bao, M.J.; Shen, J.; Jia, Y.L.; Li, F.F.; Ma, W.J.; Shen, H.J.; Shen, L.L.; Lin, X.X.; Zhang, L.H.; Dong, X.W.; et al. Apple polyphenol protects against cigarette smoke-induced acute lung injury. Nutrition 2013, 29, 235–243. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Osaki, L.H.; Gama, P. MAPKs and Signal Transduction in the Control of Gastrointestinal Epithelial Cell Proliferation and Differentiation. Int. J. Mol. Sci. 2013, 14, 10143-10161. https://doi.org/10.3390/ijms140510143
Osaki LH, Gama P. MAPKs and Signal Transduction in the Control of Gastrointestinal Epithelial Cell Proliferation and Differentiation. International Journal of Molecular Sciences. 2013; 14(5):10143-10161. https://doi.org/10.3390/ijms140510143
Chicago/Turabian StyleOsaki, Luciana H., and Patrícia Gama. 2013. "MAPKs and Signal Transduction in the Control of Gastrointestinal Epithelial Cell Proliferation and Differentiation" International Journal of Molecular Sciences 14, no. 5: 10143-10161. https://doi.org/10.3390/ijms140510143
APA StyleOsaki, L. H., & Gama, P. (2013). MAPKs and Signal Transduction in the Control of Gastrointestinal Epithelial Cell Proliferation and Differentiation. International Journal of Molecular Sciences, 14(5), 10143-10161. https://doi.org/10.3390/ijms140510143



