Loss of SUMOylation on ATF3 Inhibits Proliferation of Prostate Cancer Cells by Modulating CCND1/2 Activity
Abstract
:1. Introduction
2. Results
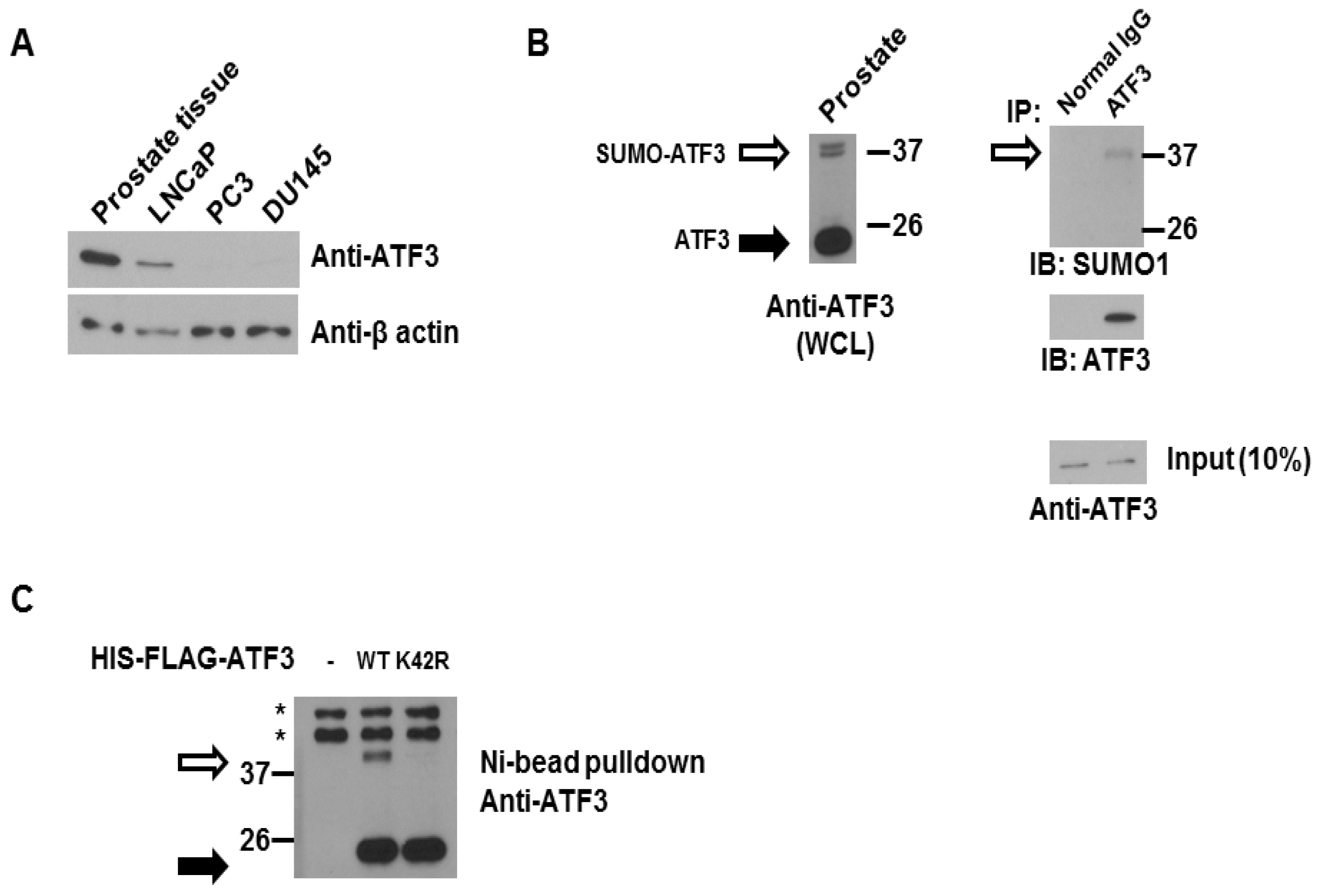
2.1. ATF3 Can Be SUMOylated Endogenously in the Overexpression System
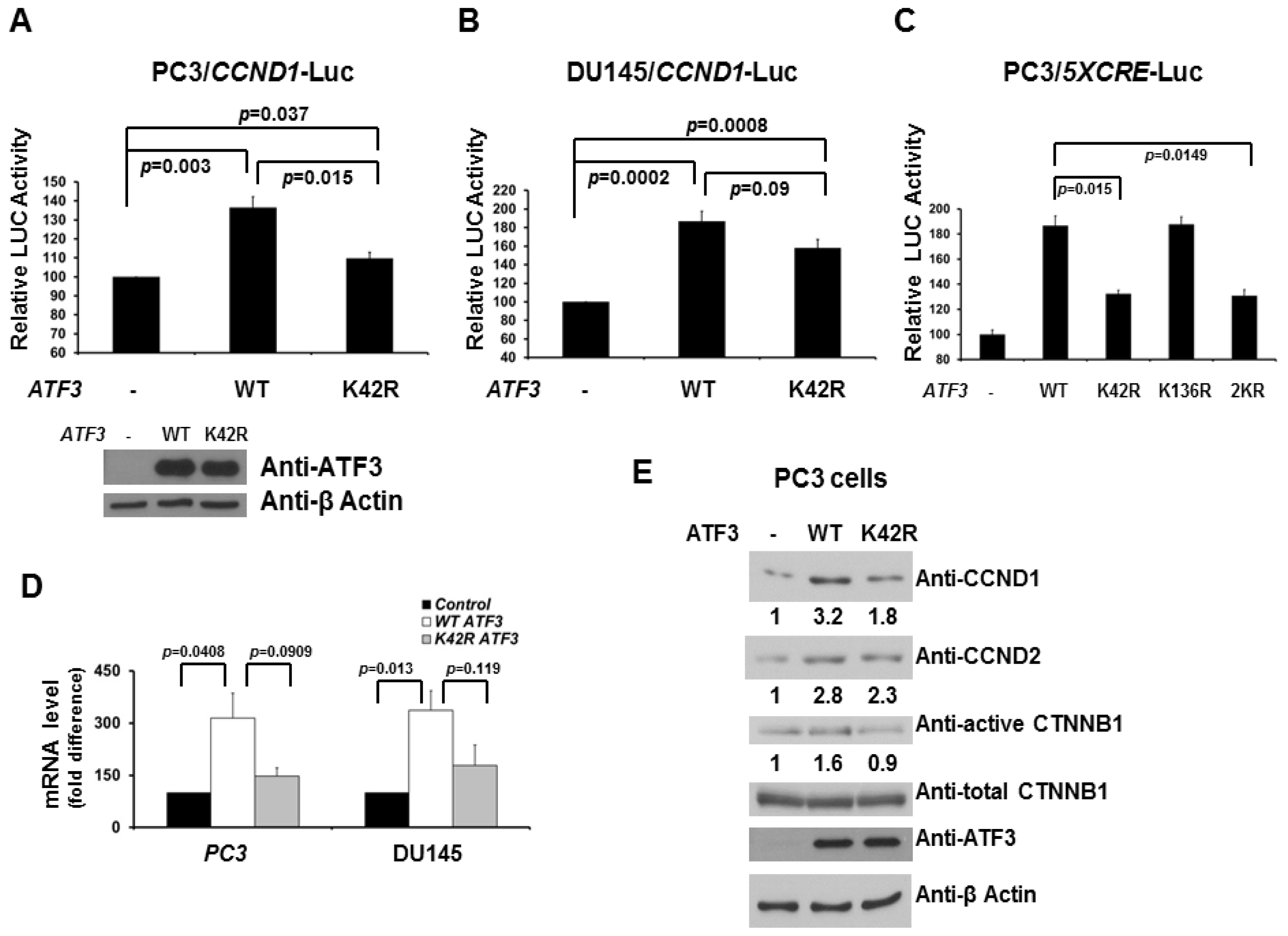
2.2. De-SUMOylation Reduces ATF3-Mediated CCND1 Activation in PC3 and DU145 Cells
2.3. Loss of SUMOylation on ATF3 Reduces Proliferation of Prostate Cancer Cells
3. Experimental Section
3.1. Reagents
3.2. DNA Constructs
3.3. Cell Culture and Transfection
3.4. Tissues
3.5. Immunoprecipitation Assay
3.6. Immunoblotting
3.7. In Vivo SUMOylation Assays
3.8. Colony Formation Assay
3.9. Cell Proliferation Assay
3.10. RT-PCR and Real-Time PCR
3.11. Statistical Analysis
4. Discussion and Conclusions
Acknowledgments
Abbreviations
| ATF3 | cyclic AMP-dependent transcription factor-3/activating transcription factor-3 |
| p53 | cellular tumor antigen p53 |
| TP53 | gene name for p53 protein |
| SUMO | small ubiquitin-like modifier |
| SENP | sentrin-specific protease |
| CCND1 | G1/S-specific cyclin D1 |
| CCND2 | G1/S-specific cyclin D2 |
| CTNNB1 | β-catenin |
Conflict of Interest
References
- Paller, C.J.; Antonarakis, E.S. Management of biochemically recurrent prostate cancer after local therapy: Evolving standards of care and new directions. Clin. Adv. Hematol. Oncol 2013, 11, 14–23. [Google Scholar]
- Klotz, L.; Toren, P. Androgen deprivation therapy in advanced prostate cancer: Is intermittent therapy the new standard of care? Curr. Oncol 2012, 19, S13–S21. [Google Scholar]
- Saylor, P.J.; Smith, M.R. Metabolic complications of androgen deprivation therapy for prostate cancer. J. Urol 2013, 189, S34–S42. [Google Scholar]
- Wolfgang, C.D.; Chen, B.P.; Martindale, J.L.; Holbrook, N.J.; Hai, T. gadd153/Chop10, a potential target gene of the transcriptional repressor ATF3. Mol. Cell. Biol 1997, 17, 6700–6707. [Google Scholar]
- Giraldo, A.; Barrett, O.P.; Tindall, M.J.; Fuller, S.J.; Amirak, E.; Bhattacharya, B.S.; Sugden, P.H.; Clerk, A. Feedback regulation by Atf3 in the endothelin-1-responsive transcriptome of cardiomyocytes: Egr1 is a principal Atf3 target. Biochem. J 2012, 444, 343–355. [Google Scholar]
- Yan, L.; della Coletta, L.; Powell, K.L.; Shen, J.; Thames, H.; Aldaz, C.M.; MacLeod, M.C. Activation of the canonical Wnt/β-catenin pathway in ATF3-induced mammary tumors. PLoS One 2011, 6, e16515. [Google Scholar]
- Yin, X.; Dewille, J.W.; Hai, T. A potential dichotomous role of ATF3, an adaptive-response gene, in cancer development. Oncogene 2008, 27, 2118–2127. [Google Scholar]
- Wang, H.; Mo, P.; Ren, S.; Yan, C. Activating transcription factor 3 activates p53 by preventing E6-associated protein from binding to E6. J. Biol. Chem 2010, 285, 13201–13210. [Google Scholar]
- Yan, C.; Lu, D.; Hai, T.; Boyd, D.D. Activating transcription factor 3, a stress sensor, activates p53 by blocking its ubiquitination. EMBO J 2005, 24, 2425–2435. [Google Scholar]
- Kang, Y.; Chen, C.R.; Massagué, J. A self-enabling TGFbeta response coupled to stress signaling: Smad engages stress response factor ATF3 for Id1 repression in epithelial cells. Mol. Cell 2003, 11, 915–926. [Google Scholar]
- Lu, D.; Wolfgang, C.D.; Hai, T. Activating transcription factor 3, a stress-inducible gene, suppresses Ras-stimulated tumorigenesis. J. Biol. Chem 2006, 281, 10473–10481. [Google Scholar]
- Hai, T.; Wolfgang, C.D.; Marsee, D.K.; Allen, A.E.; Sivaprasad, U. ATF3 and stress responses. Gene Expr 1999, 7, 321–335. [Google Scholar]
- Gilchrist, M.; Thorsson, V.; Li, B.; Rust, A.G.; Korb, M.; Roach, J.C.; Kennedy, K.; Hai, T.; Bolouri, H.; Aderem, A. Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature 2006, 441, 173–178. [Google Scholar]
- Rynes, J.; Donohoe, C.D.; Frommolt, P.; Brodesser, S.; Jindra, M.; Uhlirova, M. Activating transcription factor 3 regulates immune and metabolic homeostasis. Mol. Cell. Biol 2012, 32, 3949–3962. [Google Scholar]
- Zhou, H.; Shen, D.F.; Bian, Z.Y.; Zong, J.; Deng, W.; Zhang, Y.; Guo, Y.Y.; Li, H.; Tang, Q.Z. Activating transcription factor 3 deficiency promotes cardiac hypertrophy, dysfunction, and fibrosis induced by pressure overload. PLoS One 2011, 6, e26744. [Google Scholar]
- Pelzer, A.E.; Bektic, J.; Haag, P.; Berger, A.P.; Pycha, A.; Schäfer, G.; Rogatsch, H.; Horninger, W.; Bartsch, G.; Klocker, H. The expression of transcription factor activating transcription factor 3 in the human prostate and its regulation by androgen in prostate cancer. J. Urol 2006, 175, 1517–1522. [Google Scholar]
- Huang, X.; Li, X.; Guo, B. KLF6 induces apoptosis in prostate cancer cells through up-regulation of ATF3. J. Biol. Chem 2008, 283, 29795–29801. [Google Scholar]
- Taketani, K.; Kawauchi, J.; Tanaka-Okamoto, M.; Ishizaki, H.; Tanaka, Y.; Sakai, T.; Miyoshi, J.; Maehara, Y.; Kitajima, S. Key role of ATF3 in p53-dependent DR5 induction upon DNA damage of human colon cancer cells. Oncogene 2012, 31, 2210–2221. [Google Scholar]
- Wang, H.; Jiang, M.; Cui, H.; Chen, M.; Buttyan, R.; Hayward, S.W.; Hai, T.; Wang, Z.; Yan, C. The stress response mediator ATF3 represses androgen signaling by binding the androgen receptor. Mol. Cell Biol 2012, 32, 3190–3202. [Google Scholar]
- Wang, X.D.; Lapi, E.; Sullivan, A.; Ratnayaka, I.; Goldin, R.; Hay, R.; Lu, X. SUMO-modified nuclear cyclin D1 bypasses Ras-induced senescence. Cell Death Differ 2011, 18, 304–314. [Google Scholar]
- Wang, Z.B.; Ou, X.H.; Tong, J.S.; Li, S.; Wei, L.; Ouyang, Y.C.; Hou, Y.; Schatten, H.; Sun, Q.Y. The SUMO pathway functions in mouse oocyte maturation. Cell. Cycle 2010, 9, 2640–2646. [Google Scholar]
- Pebernard, S.; Schaffer, L.; Campbell, D.; Head, S.R.; Boddy, M.N. Localization of Smc5/6 to centromeres and telomeres requires heterochromatin and SUMO, respectively. EMBO J 2008, 27, 3011–3023. [Google Scholar]
- Dou, H.; Huang, C.; Singh, M.; Carpenter, P.B.; Yeh, E.T. Regulation of DNA repair through deSUMOylation and SUMOylation of replication protein A complex. Mol. Cell 2010, 39, 333–345. [Google Scholar]
- Plant, L.D.; Dementieva, I.S.; Kollewe, A.; Olikara, S.; Marks, J.D.; Goldstein, S.A. One SUMO is sufficient to silence the dimeric potassium channel K2P1. Proc. Natl. Acad. Sci. USA 2010, 107, 10743–10748. [Google Scholar]
- Sydorskyy, Y.; Srikumar, T.; Jeram, S.M.; Wheaton, S.; Vizeacoumar, F.J.; Makhnevych, T.; Chong, Y.T.; Gingras, A.C.; Raught, B. A novel mechanism for SUMO system control: Regulated Ulp1 nucleolar sequestration. Mol. Cell Biol 2010, 30, 4452–4462. [Google Scholar]
- Grünwald, M.; Bono, F. Structure of Importin13-Ubc9 complex: Nuclear import and release of a key regulator of sumoylation. EMBO J 2011, 30, 427–438. [Google Scholar]
- Yan, S.; Sun, X.; Xiang, B.; Cang, H.; Kang, X.; Chen, Y.; Li, H.; Shi, G.; Yeh, E.T.; Wang, B.; et al. Redox regulation of the stability of the SUMO protease SENP3 via interactions with CHIP and Hsp90. EMBO J 2010, 29, 3773–3786. [Google Scholar]
- Yang, W.H.; Heaton, J.H.; Brevig, H.; Mukherjee, S.; Iñiguez-Lluhí, J.A.; Hammer, G.D. SUMOylation inhibits SF-1 activity by reducing CDK7-mediated serine 203 phosphorylation. Mol. Cell Biol 2009, 29, 613–625. [Google Scholar]
- Huang, J.; Yan, J.; Zhang, J.; Zhu, S.; Wang, Y.; Shi, T.; Zhu, C.; Chen, C.; Liu, X.; Cheng, J.; et al. SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane. Nat. Commun 2012, 3, 911. [Google Scholar]
- Guzzo, C.M.; Berndsen, C.E.; Zhu, J.; Gupta, V.; Datta, A.; Greenberg, R.A.; Wolberger, C.; Matunis, M.J. RNF4-dependent hybrid SUMO-ubiquitin chains are signals for RAP80 and thereby mediate the recruitment of BRCA1 to sites of DNA damage. Sci. Signal. 2012, 5, ra88. [Google Scholar]
- Mukherjee, S.; Cruz-Rodríguez, O.; Bolton, E.; Iñiguez-Lluhí, J.A. The in vivo role of androgen receptor SUMOylation as revealed by androgen insensitivity syndrome and prostate cancer mutations targeting the proline/glycine residues of synergy control motifs. J. Biol. Chem 2012, 287, 31195–31206. [Google Scholar]
- Kubota, Y.; O’Grady, P.; Saito, H.; Takekawa, M. Oncogenic Ras abrogates MEK SUMOylation that suppresses the ERK pathway and cell transformation. Nat. Cell. Biol 2011, 13, 282–291. [Google Scholar]
- Haas, A.L. Structural insights into early events in the conjugation of ubiquitin and ubiquitin-like proteins. Mol. Cell 2007, 27, 174–175. [Google Scholar]
- Gareau, J.R.; Lima, C.D. The SUMO pathway: Emerging mechanisms that shape specificity, conjugation and recognition. Nat. Rev. Mol. Cell Biol 2010, 11, 861–871. [Google Scholar]
- Wilkinson, K.A.; Henley, J.M. Analysis of metabotropic glutamate receptor 7 as a potential substrate for SUMOylation. Neurosci. Lett 2011, 491, 181–186. [Google Scholar]
- Martin, S.; Wilkinson, K.A.; Nishimune, A.; Henley, J.M. Emerging extranuclear roles of protein SUMOylation in neuronal function and dysfunction. Nat. Rev. Neurosci 2007, 8, 948–959. [Google Scholar]
- Abed, M.; Barry, K.C.; Kenyagin, D.; Koltun, B.; Phippen, T.M.; Delrow, J.J.; Parkhurst, S.M.; Orian, A. Degringolade, a SUMO-targeted ubiquitin ligase, inhibits Hairy/Groucho-mediated repression. EMBO J 2011, 30, 1289–1301. [Google Scholar]
- Gong, Z.; Brackertz, M.; Renkawitz, R. SUMO modification enhances p66-mediated transcriptional repression of the Mi-2/NuRD complex. Mol. Cell Biol 2006, 26, 4519–4528. [Google Scholar]
- Rytinki, M.M.; Palvimo, J.J. SUMOylation modulates the transcription repressor function of RIP140. J. Biol. Chem 2008, 283, 11586–11595. [Google Scholar]
- Holmstrom, S.R.; Chupreta, S.; So, A.Y.; Iñiguez-Lluhí, J.A. SUMO-mediated inhibition of glucocorticoid receptor synergistic activity depends on stable assembly at the promoter but not on DAXX. Mol. Endocrinol 2008, 22, 2061–2075. [Google Scholar]
- Duverger, O.; Chen, S.X.; Lee, D.; Li, T.; Chock, P.B.; Morasso, M.I. SUMOylation of DLX3 by SUMO1 promotes its transcriptional activity. J. Cell Biochem 2011, 112, 445–452. [Google Scholar]
- Wang, C.M.; Brennan, V.C.; Gutierrez, N.M.; Wang, X.; Wang, L.; Yang, W.H. SUMOylation of ATF3 alters its transcriptional activity on regulation of TP53 gene. J. Cell. Biochem 2013, 114, 589–598. [Google Scholar]
- St Germain, C.; O’Brien, A.; Dimitroulakos, J. Activating Transcription Factor 3 regulates in part the enhanced tumour cell cytotoxicity of the histone deacetylase inhibitor M344 and cisplatin in combination. Cancer Cell Int 2010, 10, 32. [Google Scholar]
- Allan, A.L.; Albanese, C.; Pestell, R.G.; LaMarre, J. Activating transcription factor 3 induces DNA synthesis and expression of cyclin D1 in hepatocytes. J. Biol. Chem 2001, 276, 27272–27280. [Google Scholar]
- James, C.G.; Woods, A.; Underhill, T.M.; Beier, F. The transcription factor ATF3 is upregulated during chondrocyte differentiation and represses cyclin D1 and A gene transcription. BMC Mol. Biol 2006, 7, 30. [Google Scholar]
- Mo, P.; Wang, H.; Lu, H.; Boyd, D.D.; Yan, C. MDM2 mediates ubiquitination and degradation of activating transcription factor 3. J. Biol. Chem 2010, 285, 26908–26915. [Google Scholar]


© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, C.-M.; Yang, W.-H. Loss of SUMOylation on ATF3 Inhibits Proliferation of Prostate Cancer Cells by Modulating CCND1/2 Activity. Int. J. Mol. Sci. 2013, 14, 8367-8380. https://doi.org/10.3390/ijms14048367
Wang C-M, Yang W-H. Loss of SUMOylation on ATF3 Inhibits Proliferation of Prostate Cancer Cells by Modulating CCND1/2 Activity. International Journal of Molecular Sciences. 2013; 14(4):8367-8380. https://doi.org/10.3390/ijms14048367
Chicago/Turabian StyleWang, Chiung-Min, and Wei-Hsiung Yang. 2013. "Loss of SUMOylation on ATF3 Inhibits Proliferation of Prostate Cancer Cells by Modulating CCND1/2 Activity" International Journal of Molecular Sciences 14, no. 4: 8367-8380. https://doi.org/10.3390/ijms14048367




