Beneficial Effects of the RESMENA Dietary Pattern on Oxidative Stress in Patients Suffering from Metabolic Syndrome with Hyperglycemia Are Associated to Dietary TAC and Fruit Consumption
Abstract
:1. Introduction
2. Results and Discussion
2.1. Anthropometrical, Body Composition and Blood Pressure Parameters
2.2. General Biochemical Parameters
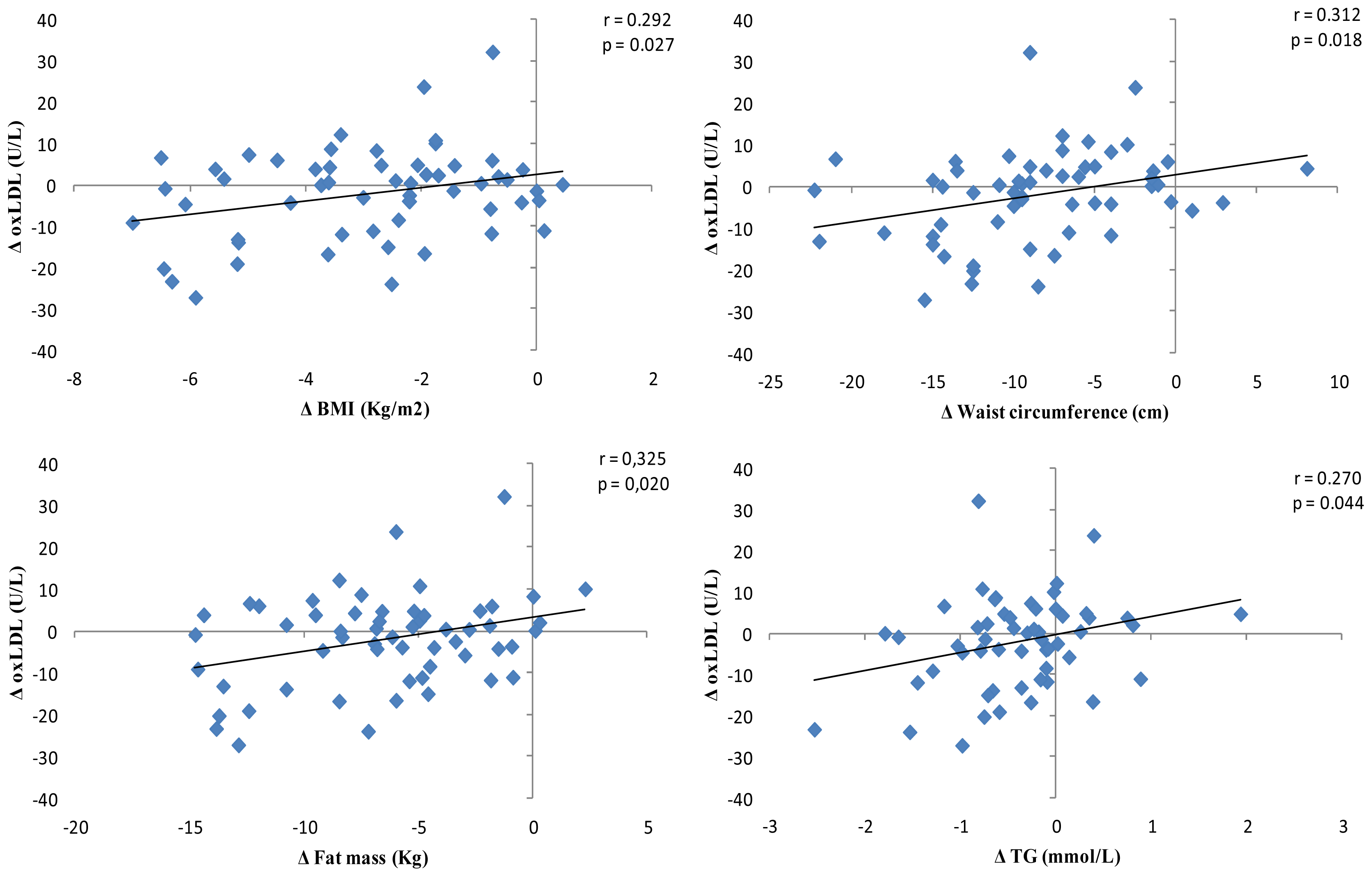
2.3. Oxidative Stress Biomarkers
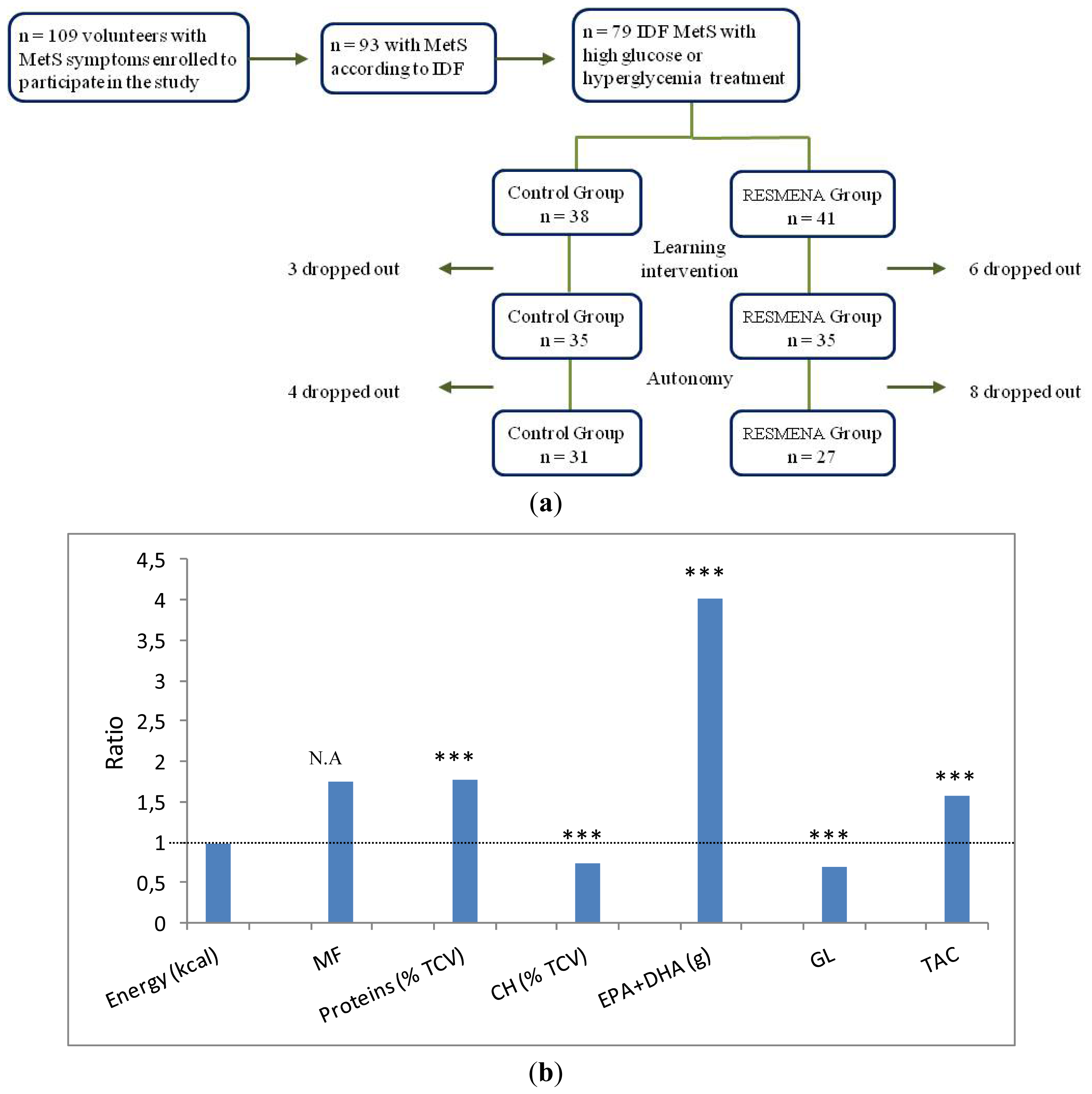
2.4. Dietary Records
3. Experimental Section
3.1. Subjects
3.2. Study Protocol
3.3. Diets
3.4. Clinical and Biochemical Assessments
3.5. Statistical Analyses
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Zimmet, P.; Shaw, J.; Aschner, P.; Balkau, B.; Bennett, P.; Boyko, E.; Brunzell, J.; Chan, J.; DeFronzo, R.; Després, J.; et al. A new international diabetes federation worldwide definition of the metabolic syndrome: The rationale and the results. Rev. Esp. Cardiol 2005, 58, 1371–1376. [Google Scholar]
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med 2011, 9, 48. [Google Scholar]
- Crepaldi, G. Origin and Development of the Metabolic Syndrome. In The Metabolic Syndrome at the Beginning of the XXIst Century: A Genetic and Molecular Approach; Ríos, M.S., Caro, J.F., Carraro, R., Fuentes, J.A.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 5–13. [Google Scholar]
- Perez-Matute, P.; Zulet, M.A.; Martinez, J.A. Reactive species and diabetes: Counteracting oxidative stress to improve health. Curr. Opin. Pharmacol 2009, 9, 771–779. [Google Scholar]
- Huri, Z.H.; Wee, H.F. Drug related problems in type 2 diabetes patients with hypertension: A cross-sectional retrospective study. BMC Endocr. Disord 2013, 13, 2. [Google Scholar]
- Mansego, M.L.; Redon, J.; Martinez-Hervas, S.; Real, J.T.; Martinez, F.; Blesa, S.; Gonzalez-Albert, V.; Saez, G.T.; Carmena, R.; Chaves, F.J. Different impacts of cardiovascular risk factors on oxidative stress. Int. J. Mol. Sci 2011, 12, 6146–6163. [Google Scholar]
- Mitra, A.; Dewanjee, D.; Dey, B. Mechanistic studies of lifestyle interventions in type 2 diabetes. World J. Diabetes 2012, 3, 201–207. [Google Scholar]
- Santos, A.C.; Ebrahim, S.; Barros, H. Alcohol intake, smoking, sleeping hours, physical activity and the metabolic syndrome. Prev. Med 2007, 44, 328–334. [Google Scholar]
- Hermsdorff, H.H.; Barbosa, K.B.; Volp, A.C.; Puchau, B.; Bressan, J.; Zulet, M.Á.; Martínez, J.A. Vitamin C and fibre consumption from fruits and vegetables improves oxidative stress markers in healthy young adults. Br. J. Nutr. 2012, 107, 1119–1127. [Google Scholar]
- Hermsdorff, H.H.; Zulet, M.A.; Puchau, B.; Martínez, J.A. Central adiposity rather than total adiposity measurements are specifically involved in the inflammatory status from healthy young adults. Inflammation 2011, 34, 161–170. [Google Scholar]
- Isomaa, B.; Almgren, P.; Tuomi, T.; Forsén, B.; Lahti, K.; Nissén, M.; Taskinen, M.R.; Groop, L. Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 2001, 24, 683–689. [Google Scholar]
- Nikolopoulou, A.; Kadoglou, N.P. Obesity and metabolic syndrome as related to cardiovascular disease. Expert Rev. Cardiovasc. Ther 2012, 10, 933–939. [Google Scholar]
- Lopez-Huertas, E. The effect of EPA and DHA on metabolic syndrome patients: A systematic review of randomised controlled trials. Br. J. Nutr 2012, 107, S185–S194. [Google Scholar]
- Bahadoran, Z.; Golzarand, M.; Mirmiran, P.; Shiva, N.; Azizi, F. Dietary total antioxidant capacity and the occurrence of metabolic syndrome and its components after a 3-year follow-up in adults: Tehran Lipid and Glucose Study. Nutr. Metab 2012, 9, 70. [Google Scholar]
- Puchau, B.; Zulet, M.A.; de Echávarri, A.G.; Hermsdorff, H.H.; Martínez, J.A. Dietary total antioxidant capacity is negatively associated with some metabolic syndrome features in healthy young adults. Nutrition 2010, 26, 534–541. [Google Scholar]
- Mirza, N.M.; Palmer, M.G.; Sinclair, K.B.; McCarter, R.; He, J.; Ebbeling, C.B.; Ludwig, D.S.; Yanovski, J.A. Effects of a low glycemic load or a low-fat dietary intervention on body weight in obese Hispanic American children and adolescents: A randomized controlled trial. Am. J. Clin. Nutr 2013, 97, 276–285. [Google Scholar]
- Jääskeläinen, A.; Schwab, U.; Kolehmainen, M.; Pirkola, J.; Järvelin, M.R.; Laitinen, J. Associations of meal frequency and breakfast with obesity and metabolic syndrome traits in adolescents of Northern Finland Birth Cohort 1986. Nutr. Metab. Cardiovasc. Dis. 2012. [Google Scholar] [CrossRef]
- Barbosa, K.B.; Bressan, J.; Zulet, M.A.; Martínez Hernández, J.A. Influence of dietary intake on plasma biomarkers of oxidative stress in humans. An. Sist. Sanit. Navar 2008, 31, 259–280. [Google Scholar]
- Zulet, M.A.; Bondia-Pons, I.; Abete, I.; de la Iglesia, R.; López-Legarrea, P.; Forga, L.; Navas-Carretero, S.; Martínez, J.A. The reduction of the metabolyc syndrome in Navarra-Spain (RESMENA-S) study: A multidisciplinary strategy based on chrononutrition and nutritional education, together with dietetic and psychological control. Nutr. Hosp 2011, 26, 16–26. [Google Scholar]
- ClinicalTrials.gov. U.S. National Institutes of Health. http://clinicaltrials.gov/show/NCT01087086 (accessed on 21 November 2011).
- Radzeviciene, L.; Ostrauskas, R. Body mass index, waist circumference, waist-hip ratio, waist-height ratio and risk for type 2 diabetes in women: A case-control study. Public Health 2013. [Google Scholar] [CrossRef]
- Santos, F.L.; Esteves, S.S.; da Costa Pereira, A.; Yancy, W.S., Jr; Nunes, J.P. Systematic review and meta-analysis of clinical trials of the effects of low carbohydrate diets on cardiovascular risk factors. Obes. Rev. 2012, 13, 1048–1066. [Google Scholar]
- Carey, V.J.; Bishop, L.; Laranjo, N.; Harshfield, B.J.; Kwiat, C.; Sacks, F.M. Contribution of high plasma triglycerides and low high-density lipoprotein cholesterol to residual risk of coronary heart disease after establishment of low-density lipoprotein cholesterol control. Am. J. Cardiol 2010, 106, 757–763. [Google Scholar]
- Clifton, P.M.; Keogh, J. Metabolic effects of high-protein diets. Curr. Atheroscler. Rep 2007, 9, 472–478. [Google Scholar]
- McQueen, M.J.; Hawken, S.; Wang, X.; Ounpuu, S.; Sniderman, A.; Probstfield, J.; Steyn, K.; Sanderson, J.E.; Hasani, M.; Volkova, E.; et al. Lipids, lipoproteins, and apolipoproteins as risk markers of myocardial infarction in 52 countries (the INTERHEART study): A case-control study. Lancet 2008, 372, 224–233. [Google Scholar]
- Tani, S.; Saito, Y.; Anazawa, T.; Kawamata, H.; Furuya, S.; Takahashi, H.; Iida, K.; Matsumoto, M.; Washio, T.; Kumabe, N.; et al. Low-density lipoprotein cholesterol/apolipoprotein B ratio may be a useful index that differs in statin-treated patients with and without coronary artery disease: A case control study. Int. Heart J 2011, 52, 343–347. [Google Scholar]
- Sanchez-Muniz, F.J.; Bastida, S.; Viejo, J.M.; Terpstra, A.H. Small supplements of n-3 fatty acids change serum low density lipoprotein composition by decreasing phospholid and apolipoprotein B concentrations in young adult women. Eur. J. Nutr 1999, 38, 20–27. [Google Scholar]
- Miles, R.R.; Perry, W.; Haas, J.V.; Mosior, M.K.; N’cho, M.; Wang, J.W.; Yu, P.; Calley, J.; Yue, Y.; Carter, Q.; et al. Genome-wide screen for modulation of hepatic Apolipoprotein A-I (ApoA-I) secretion. J. Biol. Chem. 2013. [Google Scholar] [CrossRef]
- Johnson, R.J.; Lanaspa, M.A.; Gaucher, E.A. Uric acid: A danger signal from the RNA world that may have a role in the epidemic of obesity, metabolic syndrome, and cardiorenal disease: Evolutionary considerations. Semin. Nephrol 2011, 31, 394–399. [Google Scholar]
- Pasalic, D.; Marinkovic, N.; Feher-Turkovic, L. Uric acid as one of the important factors in multifactorial disorders—Facts and controversies. Biochem. Med 2012, 22, 63–75. [Google Scholar]
- Rider, O.J.; Holloway, C.J.; Emmanuel, Y.; Bloch, E.; Clarke, K.; Neubauer, S. Increasing plasma free fatty acids in healthy subjects induces aortic distensibility changes seen in obesity. Circ. Cardiovasc. Imaging 2012, 5, 367–375. [Google Scholar]
- Yahalom, G.; Kivity, S.; Segev, S.; Sidi, Y.; Kurnik, D. Estimated glomerular filtration rate in a population with normal to mildly reduced renal function as predictor of cardiovascular disease. Eur. J. Prev. Cardiol. 2013. [Google Scholar] [CrossRef]
- Wu, H.L.; Sung, J.M.; Kao, M.D.; Wang, M.C.; Tseng, C.C.; Chen, S.T. Nonprotein calorie supplement improves adherence to low-protein diet and exerts beneficial responses on renal function in chronic kidney disease. J. Ren. Nutr. 2012. [Google Scholar] [CrossRef]
- Brinkworth, G.D.; Buckley, J.D.; Noakes, M.; Clifton, P.M. Renal function following long-term weight loss in individuals with abdominal obesity on a very-low-carbohydrate diet vs high-carbohydrate diet. J. Am. Diet. Assoc 2010, 110, 633–638. [Google Scholar]
- Beasley, J.M.; Aragaki, A.K.; LaCroix, A.Z.; Neuhouser, M.L.; Tinker, L.F.; Cauley, J.A.; Ensrud, K.E.; Jackson, R.D.; Prentice, R.L. Higher biomarker-calibrated protein intake is not associated with impaired renal function in postmenopausal women. J. Nutr 2011, 141, 1502–1507. [Google Scholar]
- Larson-Meyer, D.M.; Newcomer, B.R.; Heilbronn, L.K.; Volaufova, J.; Smith, S.R.; Alfonso, A.J.; Lefevre, M.; Rood, J.C.; Williamson, D.A.; Ravussin, E.; et al. Effect of 6-month calorie restriction and exercise on serum and liver lipids and markers of liver function. Obesity 2008, 16, 1355–1362. [Google Scholar]
- Straznicky, N.E.; Lambert, E.A.; Grima, M.T.; Eikelis, N.; Nestel, P.J.; Dawood, T.; Schlaich, M.P.; Masuo, K.; Chopra, R.; Sari, C.I.; et al. The effects of dietary weight loss with or without exercise training on liver enzymes in obese metabolic syndrome subjects. Diabetes Obes. Metab 2012, 14, 139–148. [Google Scholar]
- Rodríguez-Hernández, H.; Cervantes-Huerta, M.; Rodríguez-Moran, M.; Guerrero-Romero, F. Decrease of aminotransferase levels in obese women is related to body weight reduction, irrespective of type of diet. Ann. Hepatol 2011, 10, 486–492. [Google Scholar]
- Jialal, I.; Devaraj, S.; Adams-Huet, B.; Chen, X.; Kaur, H. Increased cellular and circulating biomarkers of oxidative stress in nascent metabolic syndrome. J. Clin. Endocrinol. Metab 2012, 97, E1844–E1850. [Google Scholar]
- De la Iglesia, R.; Milagro, F.I.; Campión, J.; Boqué, N.; Martínez, J.A. Healthy properties of proanthocyanidins. Biofactors 2010, 36, 159–168. [Google Scholar]
- Kaneto, H.; Matsuoka, T.A. Involvement of oxidative stress in suppression of insulin biosynthesis under diabetic conditions. Int. J. Mol. Sci 2012, 13, 13680–13690. [Google Scholar]
- Fernández-Sánchez, A.; Madrigal-Santillán, E.; Bautista, M.; Esquivel-Soto, J.; Morales-González, Á.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sánchez-Rivera, G.; Valadez-Vega, C.; Morales-González, J.A. Inflammation, oxidative stress, and obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar]
- Kaefer, M.; de Carvalho, J.A.; Piva, S.J.; da Silva, D.B.; Becker, A.M.; Sangoi, M.B.; Almeida, T.C.; Hermes, C.L.; Coelho, A.C.; Tonello, R.; et al. Plasma malondialdehyde levels and risk factors for the development of chronic complications in type 2 diabetic patients on insulin therapy. Clin. Lab 2012, 58, 973–978. [Google Scholar]
- Crujeiras, A.B.; Parra, D.; Goyenechea, E.; Abete, I.; González-Muniesa, P.; Martínez, J.A. Energy restriction in obese subjects impact differently two mitochondrial function markers. J. Physiol. Biochem 2008, 64, 211–219. [Google Scholar]
- Nicholls, S.J.; Hazen, S.L. Myeloperoxidase and cardiovascular disease. Arterioscler Thromb. Vasc. Biol 2005, 25, 1102–1111. [Google Scholar]
- Rector, R.S.; Warner, S.O.; Liu, Y.; Hinton, P.S.; Sun, G.Y.; Cox, R.H.; Stump, C.S.; Laughlin, M.H.; Dellsperger, K.C.; Thomas, T.R. Exercise and diet induced weight loss improves measures of oxidative stress and insulin sensitivity in adults with characteristics of the metabolic syndrome. Am. J. Physiol. Endocrinol. Metab 2007, 293, E500–E506. [Google Scholar]
- Nus, M.; Frances, F.; Librelotto, J.; Canales, A.; Corella, D.; Sánchez-Montero, J.M.; Sánchez-Muniz, F.J. Arylesterase activity and antioxidant status depend on PON1-Q192R and PON1-L55M polymorphisms in subjects with increased risk of cardiovascular disease consuming walnut-enriched meat. J. Nutr 2007, 137, 1783–1788. [Google Scholar]
- Nus, M.; Sanchez-Muniz, F.J.; Sanchez-Montero, J.M. A new method for the determination of arylesterase activity in human serum using simulated body fluid. Atherosclerosis 2006, 188, 155–159. [Google Scholar]
- Sanchez-Muniz, F.J. Dietary fibre and cardiovascular health. Nutr. Hosp 2012, 27, 31–45. [Google Scholar]
- Lixandru, D.; Mohora, M.; Coman, A.; Stoian, I.; van Gils, C.; Aerts, P.; Manuel-Y-Keenoy, B. Diet and paraoxonase 1 enzymatic activity in diabetic foot patients from Romania and Belgium: Favorable association of high flavonoid dietary intake with arylesterase activity. Ann. Nutr. Metab 2010, 56, 294–301. [Google Scholar]
- Rock, W.; Rosenblat, M.; Miller-Lotan, R.; Levy, A.P.; Elias, M.; Aviram, M. Consumption of wonderful variety pomegranate juice and extract by diabetic patients increases paraoxonase 1 association with high-density lipoprotein and stimulates its catalytic activities. J. Agric. Food Chem 2008, 56, 8704–8713. [Google Scholar]
- Ghorbanihaghjo, A.; Kolahi, S.; Seifirad, S.; Rashtchizadeh, N.; Argani, H.; Hajialilo, M.; Khabazi, A.; Alizadeh, S.; Bahreini, E. Effect of fish oil supplements on serum paraoxonase activity in female patients with rheumatoid arthritis: A double-blind randomized controlled trial. Arch. Iran Med 2012, 15, 549–552. [Google Scholar]
- Hayden, M.R.; Tyagi, S.C. Homocysteine and reactive oxygen species in metabolic syndrome, type 2 diabetes mellitus, and atheroscleropathy: The pleiotropic effects of folate supplementation. Nutr. J 2004, 10, 4. [Google Scholar]
- Colas, R.; Pruneta-Deloche, V.; Guichardant, M.; Luquain-Costaz, C.; Cugnet-Anceau, C.; Moret, M.; Vidal, H.; Moulin, P.; Lagarde, M.; Calzada, C. Increased lipid peroxidation in LDL from type-2 diabetic patients. Lipids 2010, 45, 723–731. [Google Scholar]
- Linna, M.S.; Borg, P.; Kukkonen-Harjula, K.; Fogelholm, M.; Nenonen, A.; Ahotupa, M.; Vasankari, T.J. Successful weight maintenance preserves lower levels of oxidized LDL achieved by weight reduction in obese men. Int. J. Obes 2007, 31, 245–253. [Google Scholar]
- Alberti, K.G.; Zimmet, P.; Shaw, J. The metabolic syndrome—A new worldwide definition. Lancet 2005, 366, 1059–1062. [Google Scholar]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gøtzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials. Int. J. Surg 2012, 10, 28–55. [Google Scholar]
- Navas-Carretero, S.; Pérez-Granados, A.M.; Schoppen, S.; Sarria, B.; Carbajal, A.; Vaquero, M.P. Iron status biomarkers in iron deficient women consuming oily fish versus red meat diet. J. Physiol. Biochem 2009, 65, 165–174. [Google Scholar]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C., Jr; et al. Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 2005, 112, 2735–2752. [Google Scholar]
- Abete, I.; Parra, D.; de Morentin, B.M.; Alfredo Martinez, J. Effects of two energy-restricted diets differing in the carbohydrate/protein ratio on weight loss and oxidative changes of obese men. Int. J. Food Sci. Nutr 2009, 60, 1–13. [Google Scholar]
- INGENIERIA, A DIAL Programme. Evaluation and design diets programme. Available online: http://www.alceingenieria.net/nutricion.htm (accessed on 1 March 2012).
- Carlsen, M.H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutr. J 2010, 9, 3. [Google Scholar]
- The University of Sydney. Available online: http://www.glycemicindex.com (accessed on 5 July 2012).
- Pérez, S.; Martínez, P.M.; de Morentín, B.; Rodríguez, M.; Martínez, J. Evaluación de la variabilidad intraindividual de la medida de composición corporal mediante bioimpedancia en voluntarias sanas y su relación con el índice de masa corporal y el pliegue tricipital. Enferm. Clín 2005, 15, 307–314. [Google Scholar]
- Whitworth, J.A.; Chalmers, J. World health organisation-international society of hypertension (WHO/ISH) hypertension guidelines. Clin. Exp. Hypertens 2004, 26, 747–752. [Google Scholar]
- Aller, E.E.J.G.; Abete, I.; Astrup, A.; Martinez, J.A.; van Baak, M.A. Starches, sugars and obesity. Nutrients 2011, 3, 341–369. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem 1972, 18, 499–502. [Google Scholar]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar]

| Control | RESMENA | P† Difference | |||
|---|---|---|---|---|---|
| Day 0 | Day 180 | Day 0 | Day 180 | ||
| Weight (kg) | 103.1 ± 2.9 | 95.35 ± 2.9 *** | 106.0 ± 3.2 | 96.7 ± 3.0 *** | 0.281 |
| BMI (kg/m2) | 36.4 ± 0.7 | 33.7 ± 0.8 *** | 37.41 ± 0.8 | 34.12 ± 0.8 *** | 0.206 |
| Waist circumference (cm) | 114.6 ± 2.0 | 107.4 ± 2.0 *** | 117.2 ± 2.1 | 107.1 ± 2.0 *** | 0.060 |
| WHR | 1.00 ± 0.02 | 0.97 ± 0.02 *** | 0.99 ± 0.02 | 0.95 ± 0.02 *** | 0.098 |
| Total fat Mass (kg) | 42.3 ± 1.5 | 36.4 ± 1.6 *** | 45.4 ± 1.9 | 37.9 ± 1.8 *** | 0.139 |
| Android Fat Mass (kg) | 4.7 ± 0.2 | 4.3 ± 0.3 | 5.3 ± 0.2 | 4.0 ± 0.2 *** | 0.044 |
| Lean mass (kg) | 58.0 ± 2.2 | 55.6 ± 2.1 *** | 57.1 ± 2.1 | 55.5 ± 2.0 ** | 0.197 |
| Fat-free mass (kg) | 60.9 ± 2.3 | 58.6 ± 2.2 *** | 60.0 ± 2.1 | 58.4 ± 2.1 ** | 0.220 |
| SBP (mmHg) | 152.9 ± 3.3 | 138.7 ± 2.2 ** | 154.2 ± 4.4 | 137.1 ± 3.1 ** | 0.637 |
| DBP (mmHg) | 86.3 ± 1.6 | 79.2 ± 1.8 ** | 85.8 ± 1.8 | 79.5 ± 2.0 * | 0.766 |
| Heart rate (bpm) | 75 ± 3 | 72 ± 3 | 82.3 ± 2.6 | 72.1 ± 2.5 *** | 0.587 |
| Activity level1 | 1.59 ± 0.04 | 1.54 ± 0.04 | 1.54 ± 0.03 | 1.55 ± 0.03 | 0.191 |
| Control | RESMENA | P† Difference | |||
|---|---|---|---|---|---|
| Day 0 | Day 180 | Day 0 | Day 180 | ||
| Total Cholesterol (mmol/L) | 5.56 ± 0.19 | 5.66 ± 0.19 | 5.44 ± 0.21 | 5.44 ± 0.20 | 0.397 |
| HDL-c (mmol/L) | 1.14 ± 0.05 | 1.28 ± 0.06 *** | 1.11 ± 0.04 | 1.15 ± 0.04 | 0.057 |
| LDL-c (mmol/L) | 3.47 ± 0.18 | 4.38 ± 0.17 *** | 3.34 ± 0.17 | 4.29 ± 0.19 *** | 0.884 |
| LDL-c/ApoB | 1.43 ± 0.04 | 1.91 ± 0.04 *** | 1.50 ± 0.11 | 1.92 ± 0.03 ** | 0.593 |
| TG (mmol/L) | 2.06 ± 0.21 | 1.67 ± 0.21 * | 2.17 ± 0.21 | 1.72 ± 0.20 ** | 0.574 |
| Apo A-I (mg/dL) | 134.3 ± 4.3 | 139.2 ± 4.1 | 126.3 ± 3.5 | 131.2 ± 4.3 | 0.978 |
| Apo B (mg/dL) | 93.4 ± 3.7 | 88.7 ± 3.4 | 90.3 ± 4.6 | 86.9 ± 4.1 | 0.737 |
| FFA (mmol/L) | 0.55 ± 0.04 | 0.48 ± 0.04 | 0.60 ± 0.18 | 0.50 ± 0.23 * | 0.349 |
| Glucose (mmol/L) | 7.14 ± 0.36 | 6.68 ± 0.28 | 7.59 ± 0.43 | 6.49 ± 0.35 ** | 0.118 |
| Insulin (μU/mL) | 15.22 ± 1.56 | 10.01 ± 1.54 *** | 15.36 ± 1.53 | 9.41 ± 1.21 *** | 0.685 |
| HOMA-IR | 4.92 ± 0.55 | 3.25 ± 0.61 ** | 5.24 ± 0.56 | 2.80 ± 0.37 *** | 0.475 |
| Uric Acid (mg/dL) | 6.08 ± 0.21 | 6.29 ± 0.22 | 6.19 ± 0.28 | 6.23 ± 0.22 | 0.310 |
| Total Proteins (mg/dL) | 73.01 ± 0.94 | 76.30 ± 1.19 *** | 71.48 ± 0.79 | 73.51 ± 0.97 * | 0.186 |
| eGFRs (mL/min/1.73 m2) | 83.97 ± 2.92 | 79.85 ± 2.60 | 79.07 ± 2.72 | 81.46 ± 3.08 | 0.080 |
| ALT (U/L) | 41.59 ± 4.29 | 27.16 ± 1.56 ** | 28.90 ± 2.13 | 22.54 ± 1.60 ** | 0.172 |
| AST (U/L) | 27.73 ± 2.26 | 22.86 ± 1.15* | 22.68 ± 1.08 | 20.38 ± 1.00 | 0.685 |
| Control | RESMENA | P† Difference | |||
|---|---|---|---|---|---|
| Day 0 | Day 180 | Day 0 | Day 180 | ||
| MDA (μM) | 0.86 ± 0.07 | 0.75 ± 0.07 * | 0.83 ± 0.07 | 0.76 ± 0.05 | 0.449 |
| MPO (μg/L) | 71.69 ± 7.36 | 65.39 ± 7.65 | 69.53 ± 8.39 | 66.48 ± 7.42 | 0.723 |
| ARE (U/L) | 458 ± 44 | 442 ± 43 | 370 ± 31 | 361 ± 28 | 0.778 |
| ARE:HDL-c (U/mmol) | 413.6 ± 0.1 | 366.8 ± 0.1 * | 343.8 ± 0.1 | 327.1 ± 0.1 | 0.227 |
| ARE:Apo A-I (U/mg) | 0.347 ± 0.030 | 0.319 ± 0.027 * | 0.295 ± 0.024 | 0.281 ± 0.022 | 0.424 |
| oxLDL (U/L) | 35.36 ± 1.80 | 36.39 ± 2.60 | 46.53 ± 4.46 | 41.03 ± 3.22 * | 0.025 |
| oxLDL:LDL-c (U/mmol) | 10.34 ± 0.52 | 8.25 ± 0.62 ** | 14.88 ± 1.80 | 9.52 ± 0.58 ** | 0.046 |
| oxLDL:HDL-c (U/mmol) | 30.89 ± 1.52 | 28.46 ± 1.76 | 42.78 ± 4.19 | 4.19 ± 2.64 * | 0.186 |
| oxLDL:Apo B (U/mg) | 0.038 ± 0.002 | 0.043 ± 0.004 | 0.051 ± 0.004 | 0.048 ± 0.003 | 0.040 |
| Control | RESMENA | p | |
|---|---|---|---|
| Energy (kcal/day) | 1513 ± 54 | 1569 ± 77 | 0.542 |
| Meal Frequency (meals/day) | 4.3 ± 0.2 | 5.8 ± 0.2 | <0.001 |
| Proteins (% TCV/day) | 16.9 ± 0.4 | 20.4 ± 0.9 | 0.001 |
| Lipids (% TCV/day) | 40.8 ± 1.5 | 37.7 ± 1.0 | 0.108 |
| CHO (% TCV/day) | 37.1 ± 1.5 | 36.9 ± 1.1 | 0.940 |
| Fiber (% TCHO/day) | 11.4 ± 0.8 | 12.0 ± 0.6 | 0.573 |
| GL (U/day) | 73.4 ± 5.9 | 70.0 ± 5.5 | 0.682 |
| EPA+DHA (g/day) | 0.30 ± 0.08 | 0.39 ± 0.17 | 0.617 |
| TAC (mmol/day) | 6.1 ± 0.6 | 8.5 ± 0.9 | 0.031 |
| Fruits (kcal/day) | 117 ± 21 | 185 ± 27 | 0.049 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De la Iglesia, R.; Lopez-Legarrea, P.; Celada, P.; Sánchez-Muniz, F.J.; Martinez, J.A.; Zulet, M.A. Beneficial Effects of the RESMENA Dietary Pattern on Oxidative Stress in Patients Suffering from Metabolic Syndrome with Hyperglycemia Are Associated to Dietary TAC and Fruit Consumption. Int. J. Mol. Sci. 2013, 14, 6903-6919. https://doi.org/10.3390/ijms14046903
De la Iglesia R, Lopez-Legarrea P, Celada P, Sánchez-Muniz FJ, Martinez JA, Zulet MA. Beneficial Effects of the RESMENA Dietary Pattern on Oxidative Stress in Patients Suffering from Metabolic Syndrome with Hyperglycemia Are Associated to Dietary TAC and Fruit Consumption. International Journal of Molecular Sciences. 2013; 14(4):6903-6919. https://doi.org/10.3390/ijms14046903
Chicago/Turabian StyleDe la Iglesia, Rocio, Patricia Lopez-Legarrea, Paloma Celada, Francisco J. Sánchez-Muniz, J. Alfredo Martinez, and M. Angeles Zulet. 2013. "Beneficial Effects of the RESMENA Dietary Pattern on Oxidative Stress in Patients Suffering from Metabolic Syndrome with Hyperglycemia Are Associated to Dietary TAC and Fruit Consumption" International Journal of Molecular Sciences 14, no. 4: 6903-6919. https://doi.org/10.3390/ijms14046903
APA StyleDe la Iglesia, R., Lopez-Legarrea, P., Celada, P., Sánchez-Muniz, F. J., Martinez, J. A., & Zulet, M. A. (2013). Beneficial Effects of the RESMENA Dietary Pattern on Oxidative Stress in Patients Suffering from Metabolic Syndrome with Hyperglycemia Are Associated to Dietary TAC and Fruit Consumption. International Journal of Molecular Sciences, 14(4), 6903-6919. https://doi.org/10.3390/ijms14046903





