Arsenic Trioxide Inhibits Cell Growth and Induces Apoptosis through Inactivation of Notch Signaling Pathway in Breast Cancer
Abstract
:1. Introduction
2. Results
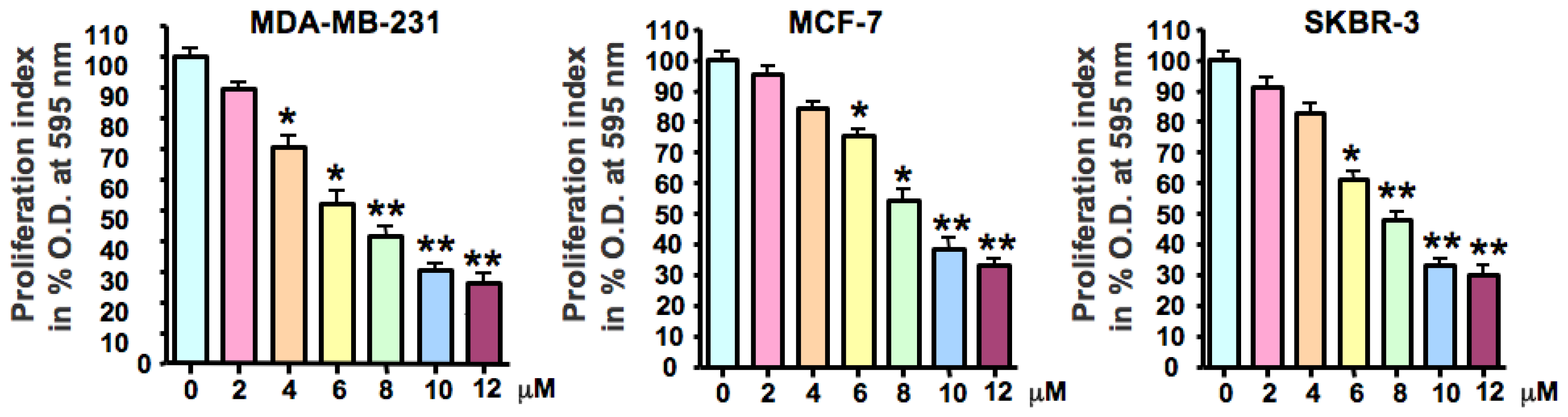
2.1. As2O3 Inhibited Breast Cancer Cell Growth
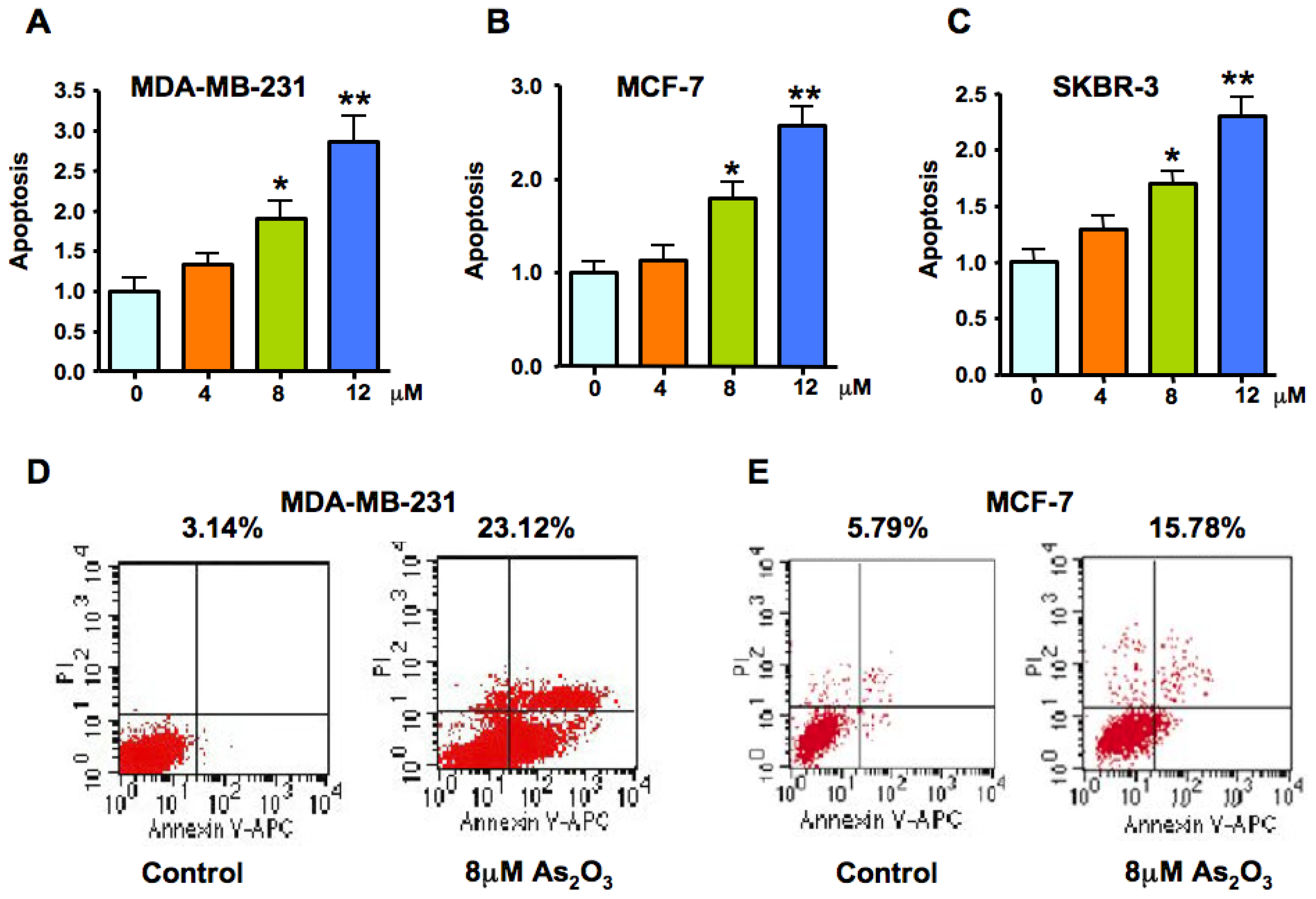
2.2. As2O3 Induced Apoptosis in Breast Cancer Cell Lines
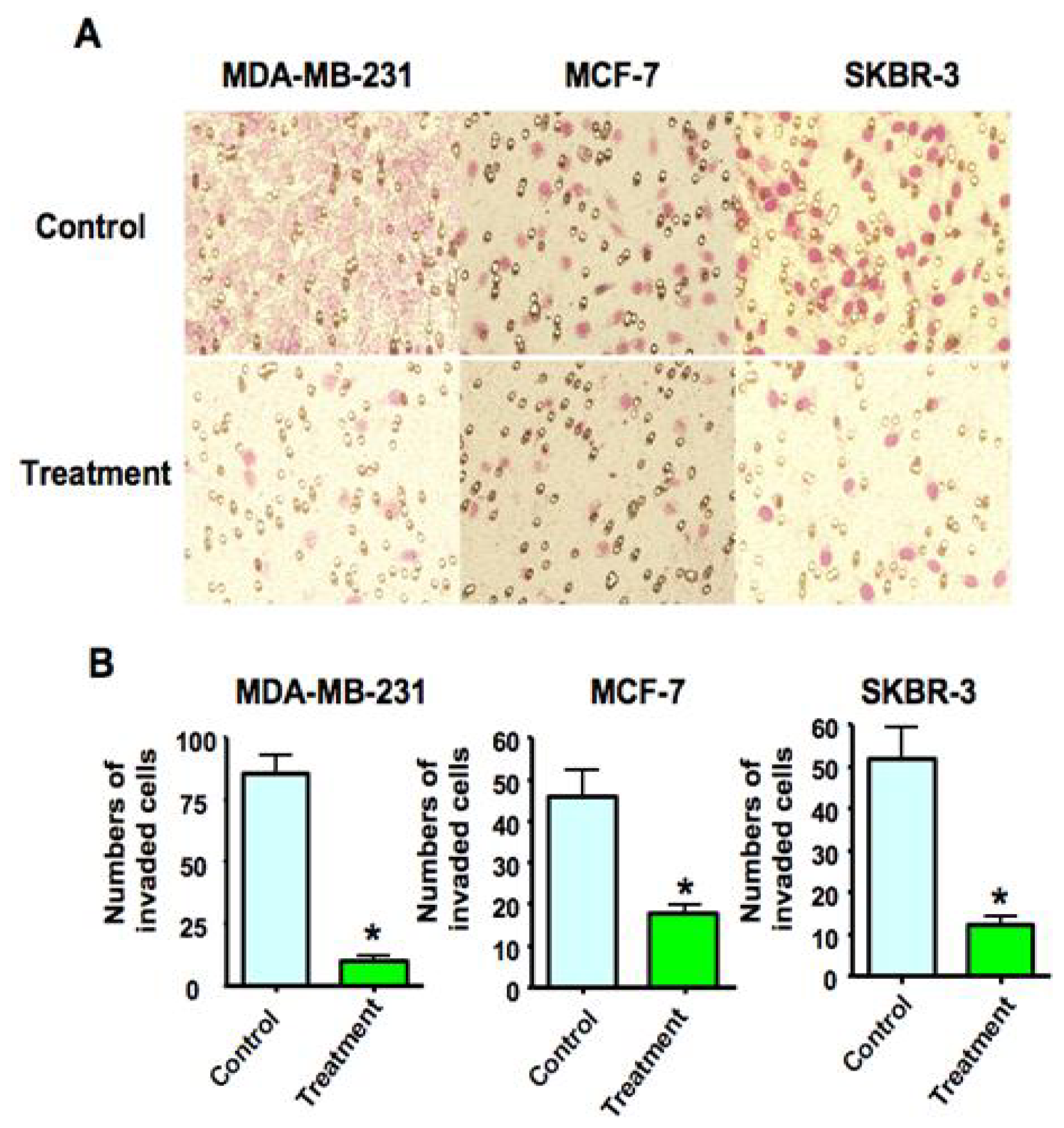
2.3. As2O3 Suppressed Breast Cancer Cell Invasion
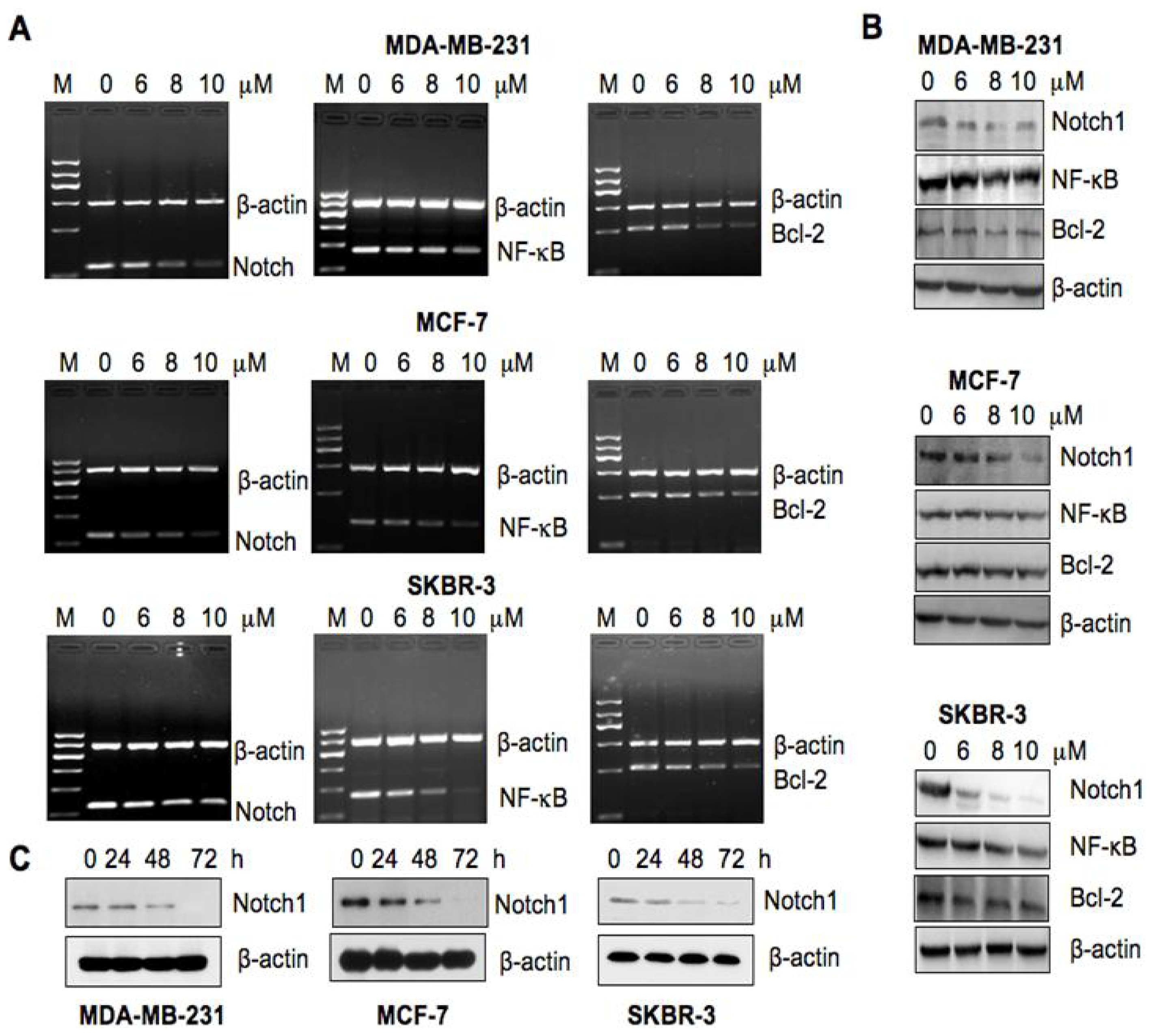
2.4. As2O3 Inhibited the Notch-1 Expression in Breast Cancer Cells
2.5. As2O3 Inhibited the Expression of Notch-1 Downstream Genes
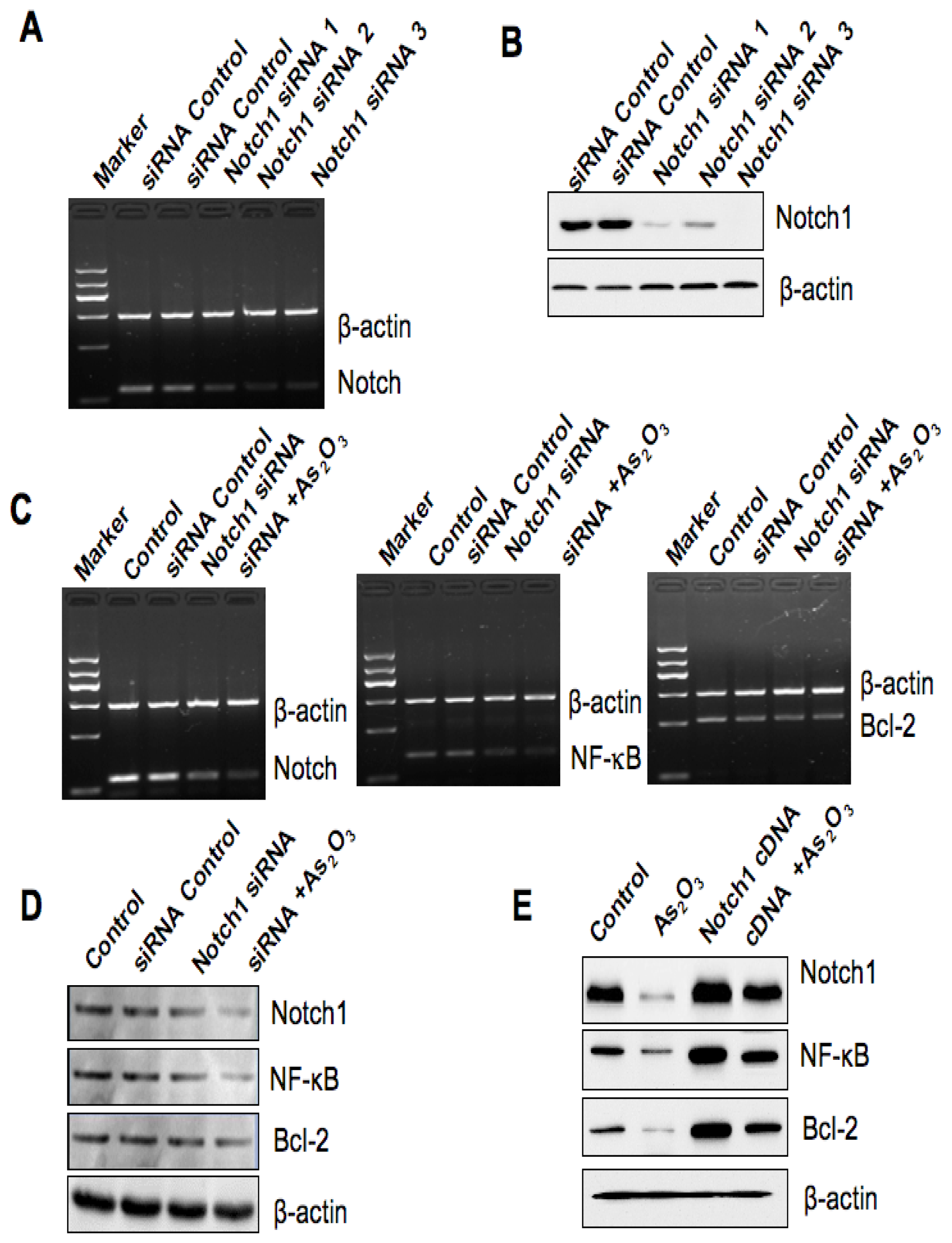
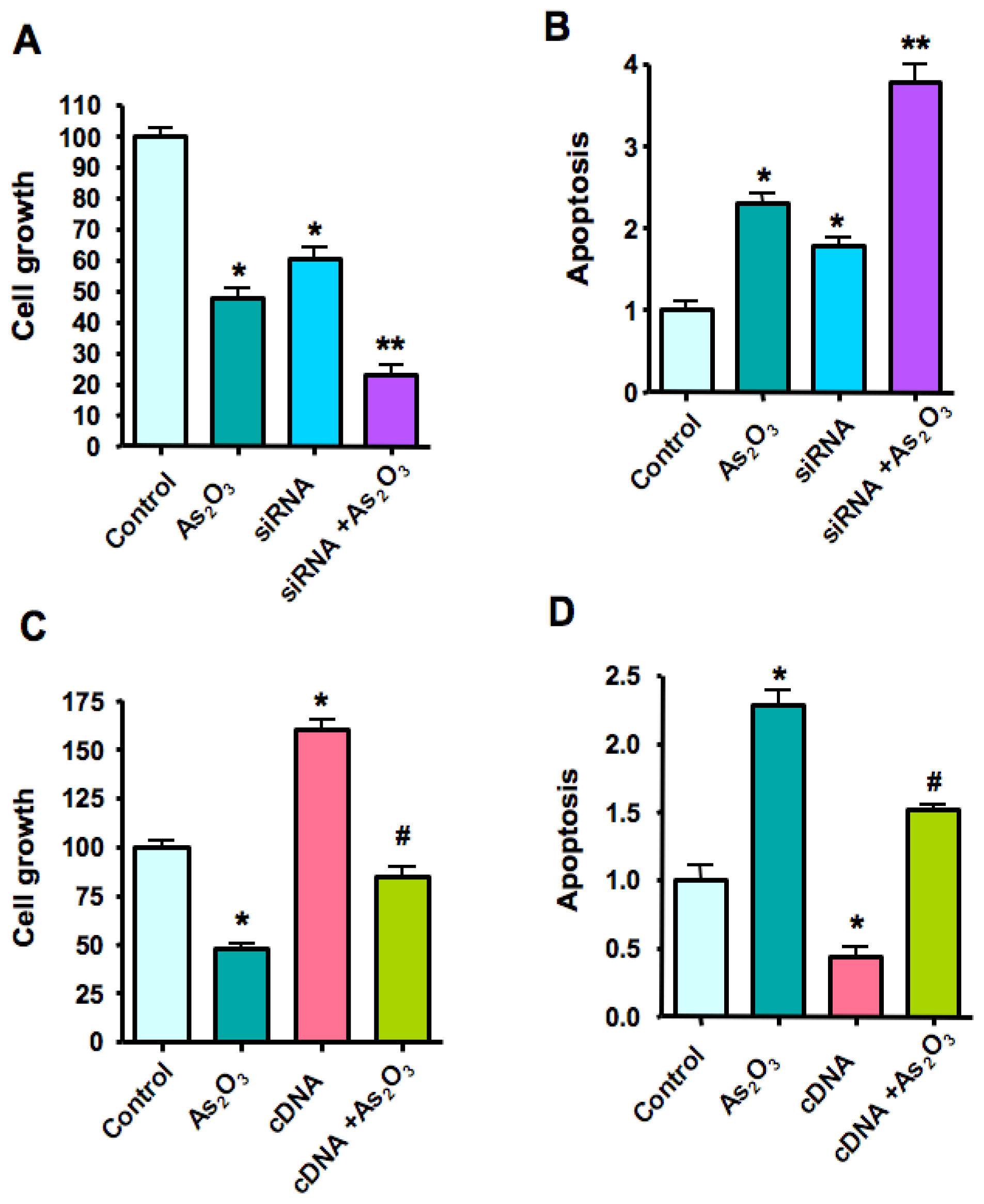
2.6. Down-Regulation of Notch-1 Expression by SiRNA and the Effect of As2O3 Treatment
2.7. Over-Expression of Notch-1 by cDNA Transfection Reduced As2O3-Induced Cell Growth Inhibition and Apoptosis
3. Discussion
4. Experiment Section
4.1. Cell Lines and Experimental Reagents
4.2. Cell Growth Inhibition Studies by MTT Assay
4.3. Histone-DNA Enzyme-Linked Immunosorbent Assay (ELISA) for Detecting Apoptosis
4.4. Annexin V-FITC Method for Apoptosis Analysis
4.5. Cell Invasion Assay
4.6. Reverse Transcription-PCR (RT-PCR) Analysis for Gene Expression Studies
4.7. Western Blot Analysis
4.8. Plasmids and Transfections
4.9. Densitometric and Statistical Analysis
5. Conclusions
Acknowledgement
- Conflict of InterestThe authors declare that they have no conflict of interest.
References and Notes
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics. CA Cancer J. Clin 2012, 62, 10–29. [Google Scholar]
- Chlebowski, R.T.; Anderson, G.L. Changing concepts: Menopausal hormone therapy and breast cancer. J. Natl. Cancer Inst 2012, 104, 517–527. [Google Scholar]
- Reedijk, M. Notch signaling and breast cancer. Adv. Exp. Med. Biol 2012, 727, 241–257. [Google Scholar]
- Guo, S.; Liu, M.; Gonzalez-Perez, R.R. Role of Notch and its oncogenic signaling crosstalk in breast cancer. Biochim. Biophys. Acta 2011, 1815, 197–213. [Google Scholar]
- Harrison, H.; Farnie, G.; Brennan, K.R.; Clarke, R.B. Breast cancer stem cells: something out of notching? Cancer Res. 2010, 70, 8973–8976. [Google Scholar]
- Rizzo, P.; Osipo, C.; Pannuti, A.; Golde, T.; Osborne, B.; Miele, L. Targeting Notch signaling cross-talk with estrogen receptor and ErbB-2 in breast cancer. Adv. Enzym. Regul 2009, 49, 134–141. [Google Scholar]
- Ranganathan, P.; Weaver, K.L.; Capobianco, A.J. Notch signalling in solid tumours: A little bit of everything but not all the time. Nat. Rev. Cancer 2011, 11, 338–351. [Google Scholar]
- Han, J.; Hendzel, M.J.; Allalunis-Turner, J. Notch signaling as a therapeutic target for breast cancer treatment? Breast Cancer Res 2011, 13, 210. [Google Scholar]
- Dickson, B.C.; Mulligan, A.M.; Zhang, H.; Lockwood, G.; O’Malley, F.P.; Egan, S.E.; Reedijk, M. High-level JAG1 mRNA and protein predict poor outcome in breast cancer. Mod. Pathol 2007, 20, 685–693. [Google Scholar]
- Reedijk, M.; Odorcic, S.; Chang, L.; Zhang, H.; Miller, N.; McCready, D.R.; Lockwood, G.; Egan, S.E. High-level coexpression of JAG1 and NOTCH1 is observed in human breast cancer and is associated with poor overall survival. Cancer Res 2005, 65, 8530–8537. [Google Scholar]
- Reedijk, M.; Pinnaduwage, D.; Dickson, B.C.; Mulligan, A.M.; Zhang, H.; Bull, S.B.; O’Malley, F.P.; Egan, S.E.; Andrulis, I.L. JAG1 expression is associated with a basal phenotype and recurrence in lymph node-negative breast cancer. Breast Cancer Res. Treat 2008, 111, 439–448. [Google Scholar]
- Speiser, J.; Foreman, K.; Drinka, E.; Godellas, C.; Perez, C.; Salhadar, A.; Ersahin, C.; Rajan, P. Notch-1 and notch-4 biomarker expression in triple-negative breast cancer. Int. J. Surg. Pathol 2012, 20, 137–143. [Google Scholar]
- Yao, K.; Rizzo, P.; Rajan, P.; Albain, K.; Rychlik, K.; Shah, S.; Miele, L. Notch-1 and notch-4 receptors as prognostic markers in breast cancer. Int. J. Surg. Pathol 2011, 19, 607–613. [Google Scholar]
- Wang, Z.; Li, Y.; Ahmad, A.; Azmi, A.S.; Banerjee, S.; Kong, D.; Sarkar, F.H. Targeting Notch signaling pathway to overcome drug resistance for cancer therapy. Biochim. Biophys. Acta 2010, 1806, 258–267. [Google Scholar]
- Baj, G.; Arnulfo, A.; Deaglio, S.; Mallone, R.; Vigone, A.; de Cesaris, M.G.; Surico, N.; Ferrero, E. Arsenic trioxide and breast cancer: Analysis of the apoptotic, differentiative and immunomodulatory effects. Breast Cancer Res. Treat 2002, 73, 61–73. [Google Scholar]
- Chow, S.K.; Chan, J.Y.; Fung, K.P. Suppression of cell proliferation and regulation of estrogen receptor α signaling pathway by arsenic trioxide on human breast cancer MCF-7 cells. J. Endocrinol 2004, 182, 325–337. [Google Scholar]
- Chow, S.K.; Chan, J.Y.; Fung, K.P. Inhibition of cell proliferation and the action mechanisms of arsenic trioxide (As2O3) on human breast cancer cells. J. Cell Biochem 2004, 93, 173–187. [Google Scholar]
- Lengfelder, E.; Hofmann, W.K.; Nowak, D. Impact of arsenic trioxide in the treatment of acute promyelocytic leukemia. Leukemia 2012, 26, 433–442. [Google Scholar]
- Emadi, A.; Gore, S.D. Arsenic trioxide—An old drug rediscovered. Blood Rev 2010, 24, 191–199. [Google Scholar]
- Chen, G.C.; Guan, L.S.; Hu, W.L.; Wang, Z.Y. Functional repression of estrogen receptor a by arsenic trioxide in human breast cancer cells. Anticancer Res 2002, 22, 633–638. [Google Scholar]
- Wang, X.; Gao, P.; Long, M.; Lin, F.; Wei, J.X.; Ren, J.H.; Yan, L.; He, T.; Han, Y.; Zhang, H.Z. Essential role of cell cycle regulatory genes p21 and p27 expression in inhibition of breast cancer cells by arsenic trioxide. Med. Oncol 2011, 28, 1225–1254. [Google Scholar]
- Osipo, C.; Golde, T.E.; Osborne, B.A.; Miele, L.A. Off the beaten pathway: The complex cross talk between Notch and NF-κB. Lab. Invest 2008, 88, 11–17. [Google Scholar]
- Ferreira, A.C.; Suriano, G.; Mendes, N.; Gomes, B.; Wen, X.; Carneiro, F.; Seruca, R.; Machado, J.C. E-cadherin impairment increases cell survival through Notch-dependent upregulation of Bcl-2. Hum. Mol. Genet 2012, 21, 334–343. [Google Scholar]
- Clementz, A.G.; Rogowski, A.; Pandya, K.; Miele, L.; Osipo, C. NOTCH-1 and NOTCH-4 are novel gene targets of PEA3 in breast cancer: Novel therapeutic implications. Breast Cancer Res 2011, 13, R63. [Google Scholar]
- Osipo, C.; Patel, P.; Rizzo, P.; Clementz, A.G.; Hao, L.; Golde, T.E.; Miele, L. ErbB-2 inhibition activates Notch-1 and sensitizes breast cancer cells to a γ-secretase inhibitor. Oncogene 2008, 27, 5019–5032. [Google Scholar]
- Zang, S.; Chen, F.; Dai, J.; Guo, D.; Tse, W.; Qu, X.; Ma, D.; Ji, C. RNAi-mediated knockdown of Notch-1 leads to cell growth inhibition and enhanced chemosensitivity in human breast cancer. Oncol. Rep 2010, 23, 893–899. [Google Scholar]
- Zhang, J.; Wang, B. Arsenic trioxide (As2O3) inhibits peritoneal invasion of ovarian carcinoma cells in vitro and in vivo. Gynecol. Oncol 2006, 103, 199–206. [Google Scholar]
- Rizzo, P.; Miao, H.; D’Souza, G.; Osipo, C.; Song, L.L.; Yun, J.; Zhao, H.; Mascarenhas, J.; Wyatt, D.; Antico, G.; et al. Cross-talk between notch and the estrogen receptor in breast cancer suggests novel therapeutic approaches. Cancer Res 2008, 68, 5226–5235. [Google Scholar]
- Korkaya, H.; Wicha, M.S. HER-2, notch, and breast cancer stem cells: targeting an axis of evil. Clin. Cancer Res 2009, 15, 1845–1847. [Google Scholar]
- Malhotra, G.K.; Zhao, X.; Band, H.; Band, V. Shared signaling pathways in normal and breast cancer stem cells. J. Carcinog 2011, 10, 38. [Google Scholar]
- Kondratyev, M.; Kreso, A.; Hallett, R.M.; Girgis-Gabardo, A.; Barcelon, M.E.; Ilieva, D.; Ware, C.; Majumder, P.K.; Hassell, J.A. Gamma-secretase inhibitors target tumor-initiating cells in a mouse model of ERBB2 breast cancer. Oncogene 2012, 31, 93–103. [Google Scholar]
- McGowan, P.M.; Simedrea, C.; Ribot, E.J.; Foster, P.J.; Palmieri, D.; Steeg, P.S.; Allan, A.L.; Chambers, A.F. Notch1 inhibition alters the CD44hi/CD24lo population and reduces the formation of brain metastases from breast cancer. Mol. Cancer Res 2011, 9, 834–844. [Google Scholar]
- Grudzien, P.; Lo, S.; Albain, K.S.; Robinson, P.; Rajan, P.; Strack, P.R.; Golde, T.E.; Miele, L.; Foreman, K.E. Inhibition of Notch signaling reduces the stem-like population of breast cancer cells and prevents mammosphere formation. Anticancer Res 2010, 30, 3853–3867. [Google Scholar]
- Efferson, C.L.; Winkelmann, C.T.; Ware, C.; Sullivan, T.; Giampaoli, S.; Tammam, J.; Patel, S.; Mestiti, G.; Reilly, J.F.; Gibson, R.E.; et al. Downregulation of Notch pathway by a γ-secretase inhibitor attenuates AKT/mammalian target of rapamycin signaling and glucose uptake in an ERBB2 transgenic breast cancer model. Cancer Res 2010, 70, 2476–2484. [Google Scholar]
- Imbimbo, B.P. Therapeutic potential of γ-secretase inhibitors and modulators. Curr. Top Med. Chem 2008, 8, 54–61. [Google Scholar]
- De Thé, H.; Chen, Z. Acute promyelocytic leukaemia: novel insights into the mechanisms of cure. Nat. Rev. Cancer 2010, 10, 775–783. [Google Scholar]
- Diepart, C.; Karroum, O.; Magat, J.; Feron, O.; Verrax, J.; Calderon, P.B.; Grégoire, V.; Leveque, P.; Stockis, J.; Dauguet, N.; et al. Arsenic trioxide treatment decreases the oxygen consumption rate of tumor cells and radiosensitizes solid tumors. Cancer Res 2012, 72, 482–490. [Google Scholar]
- Pettersson, H.M.; Pietras, A.; Munksgaard Persson, M.; Persson, M.; Karlsson, J.; Johansson, L.; Shoshan, M.C.; Pahlman, S. Arsenic trioxide is highly cytotoxic to small cell lung carcinoma cells. Mol. Cancer Ther 2009, 8, 160–170. [Google Scholar]
- Chiu, H.W.; Chen, Y.A.; Ho, S.Y.; Wang, Y.J. Arsenic trioxide enhances the radiation sensitivity of androgen-dependent and -independent human prostate cancer cells. PLoS One 2012, 7, e31579. [Google Scholar]
- Uslu, R.; Sanli, U.A.; Sezgin, C.; Karabulut, B.; Terzioglu, E.; Omay, S.B.; Goker, E. Arsenic trioxide-mediated cytotoxicity and apoptosis in prostate and ovarian carcinoma cell lines. Clin. Cancer Res 2000, 6, 4957–4964. [Google Scholar]
- Liu, Y.; Zhang, W.; Zhang, X.; Qi, Y.; Huang, D.; Zhang, Y. Arsenic trioxide inhibits invasion/migration in SGC-7901 cells by activating the reactive oxygen species-dependent cyclooxygenase-2/matrix metalloproteinase-2 pathway. Exp. Biol. Med. (Maywood) 2011, 236, 592–597. [Google Scholar]
- Tingting, R.; Wei, G.; Changliang, P.; Xinchang, L.; Yi, Y. Arsenic trioxide inhibits osteosarcoma cell invasiveness via MAPK signaling pathway. Cancer Biol. Ther 2010, 10, 251–257. [Google Scholar]
- Park, M.J.; Lee, J.Y.; Kwak, H.J.; Park, C.M.; Lee, H.C.; Woo, S.H.; Jin, H.O.; Han, C.J.; An, S.; Lee, S.H.; et al. Arsenic trioxide (As2O3) inhibits invasion of HT1080 human fibrosarcoma cells: role of nuclear factor-kappaB and reactive oxygen species. J. Cell Biochem 2005, 95, 955–969. [Google Scholar]
- Zhen, Y.; Zhao, S.; Li, Q.; Li, Y.; Kawamoto, K. Arsenic trioxide-mediated Notch pathway inhibition depletes the cancer stem-like cell population in gliomas. Cancer Lett 2010, 292, 64–72. [Google Scholar]
- Wang, Z.; Kong, D.; Banerjee, S.; Li, Y.; Adsay, N.V.; Abbruzzese, J.; Sarkar, F.H. Down-regulation of platelet-derived growth factor-D inhibits cell growth and angiogenesis through inactivation of Notch-1 and nuclear factor-kappaB signaling. Cancer Res 2007, 67, 11377–11385. [Google Scholar]
- Wang, Z.; Zhang, Y.; Li, Y.; Banerjee, S.; Liao, J.; Sarkar, F.H. Down-regulation of Notch-1 contributes to cell growth inhibition and apoptosis in pancreatic cancer cells. Mol. Cancer Ther. 2006, 5, 483–493. [Google Scholar]
- Wang, Z.; Azmi, A.S.; Ahmad, A.; Benerjee, S.; Wang, S.; Sarkar, F.H.; Mohammad, R.M. TW-37, a small-molecule inhibitor of Bcl-2, inhibits cell growth and induces apoptosis in pancreatic cancer: Involvement of Notch-1 signaling pathway. Cancer Res 2009, 69, 2757–2765. [Google Scholar]
- Weijzen, S.; Rizzo, P.; Braid, M.; Vaishnav, R.; Jonkheer, S.M.; Zlobin, A.; Osborne, B.A.; Gottipati, S.; Aster, J.C.; Hann, W.C.; et al. Activation of Notch-1 signaling maintains the neoplastic phenotype in human Ras-transformed cells. Nat. Med 2002, 8, 979–986. [Google Scholar]
- Wang, Z.; Banerjee, S.; Li, Y.; Rahman, K.M.; Zhang, Y.; Sarkar, F.H. Down-regulation of notch-1 inhibits invasion by inactivation of nuclear factor-kappaB, vascular endothelial growth factor, and matrix metalloproteinase-9 in pancreatic cancer cells. Cancer Res. 2006, 66, 2778–2784. [Google Scholar]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xia, J.; Li, Y.; Yang, Q.; Mei, C.; Chen, Z.; Bao, B.; Ahmad, A.; Miele, L.; Sarkar, F.H.; Wang, Z. Arsenic Trioxide Inhibits Cell Growth and Induces Apoptosis through Inactivation of Notch Signaling Pathway in Breast Cancer. Int. J. Mol. Sci. 2012, 13, 9627-9641. https://doi.org/10.3390/ijms13089627
Xia J, Li Y, Yang Q, Mei C, Chen Z, Bao B, Ahmad A, Miele L, Sarkar FH, Wang Z. Arsenic Trioxide Inhibits Cell Growth and Induces Apoptosis through Inactivation of Notch Signaling Pathway in Breast Cancer. International Journal of Molecular Sciences. 2012; 13(8):9627-9641. https://doi.org/10.3390/ijms13089627
Chicago/Turabian StyleXia, Jun, Youjian Li, Qingling Yang, Chuanzhong Mei, Zhiwen Chen, Bin Bao, Aamir Ahmad, Lucio Miele, Fazlul H Sarkar, and Zhiwei Wang. 2012. "Arsenic Trioxide Inhibits Cell Growth and Induces Apoptosis through Inactivation of Notch Signaling Pathway in Breast Cancer" International Journal of Molecular Sciences 13, no. 8: 9627-9641. https://doi.org/10.3390/ijms13089627
APA StyleXia, J., Li, Y., Yang, Q., Mei, C., Chen, Z., Bao, B., Ahmad, A., Miele, L., Sarkar, F. H., & Wang, Z. (2012). Arsenic Trioxide Inhibits Cell Growth and Induces Apoptosis through Inactivation of Notch Signaling Pathway in Breast Cancer. International Journal of Molecular Sciences, 13(8), 9627-9641. https://doi.org/10.3390/ijms13089627



