Effect of Calcium Sprays on Mechanical Strength and Cell Wall Fractions of Herbaceous Peony (Paeonia Lactiflora Pall.) Inflorescence Stems
Abstract
:1. Introduction
2. Results and Discussion
2.1. Breaking Force
2.2. Calcium Concentrations
2.3. Cell Wall Fractions
2.3.1. Pectin in Different Forms
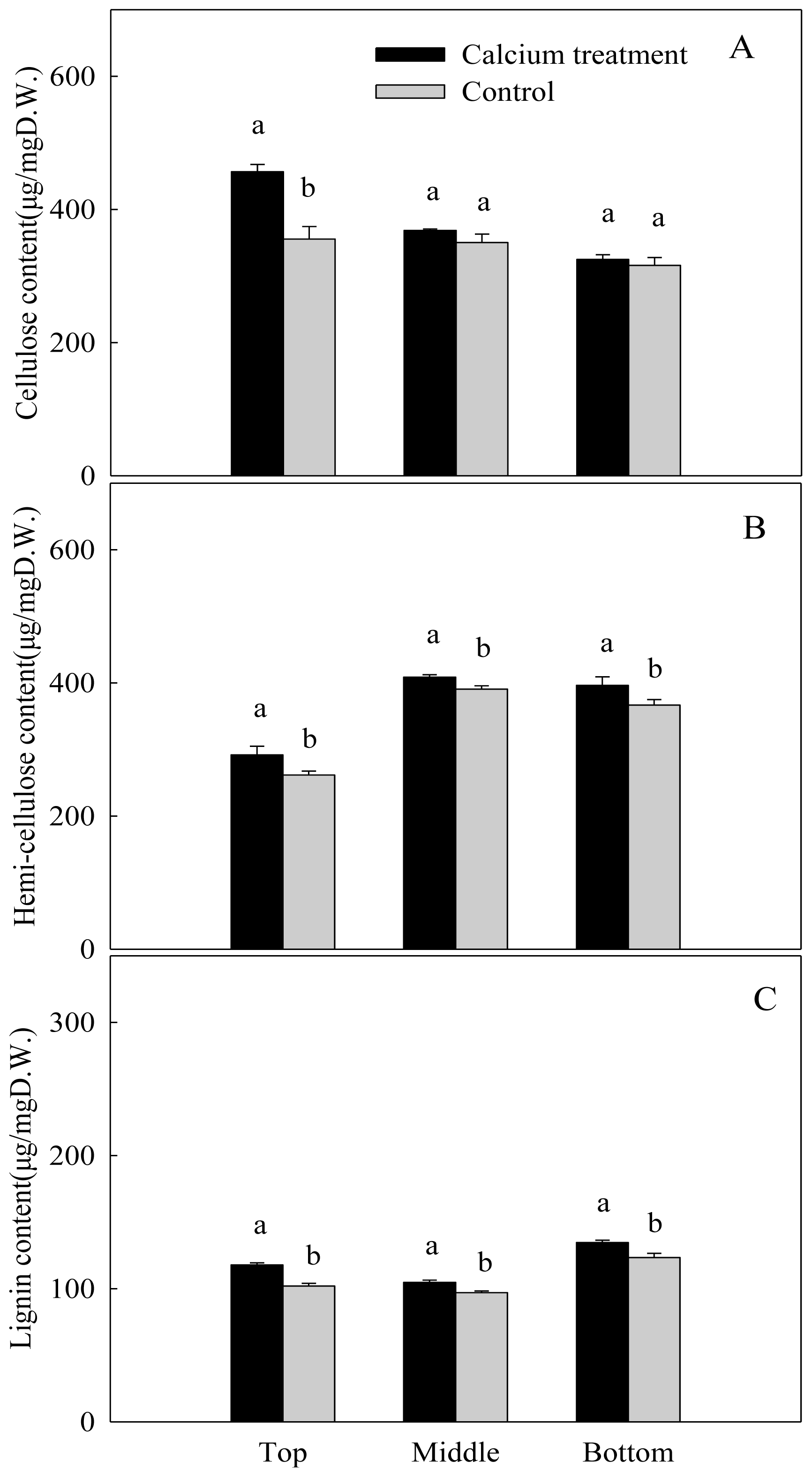
2.3.2. Cellulose, Hemi-Cellulose and Lignin
3. Experimental Section
3.1. Plant Material
3.2. Calcium Treatments
3.3. Breaking Strength Measurement
3.4. Calcium Concentration Determination
3.5. Cell Wall Materials (CWM) Preparation
3.6. Uronic Acid, Hemi-Cellulose, Cellulose and Lignin Contents Determination
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Guo, X.F.; Fu, X.L.; Zang, D.K.; Ma, Y. Effect of auxin treatments, cuttings’ collection date and initial characteristics on Paeonia “Yang Fei Chu Yu” cutting propagation. Sci. Hortic 2009, 119, 177–181. [Google Scholar]
- Hall, A.J.; Catley, J.L.; Walton, E.F. The effect of forcing temperature on peony shoot and flower develepoment. Sci. Hortic 2007, 113, 188–195. [Google Scholar]
- Guo, X.F.; Zang, D.K.; Yuan, T.; Liu, X.K.; Wang, L.Y. Discussion about the origion of cultivated herbaceous peonies native to China (in Chinese). J. Shandong Agric. Univ. (Nat. Sci.) 2008, 39, 388–392. [Google Scholar]
- Kamenetsky, R.; Barzilay, A.; Erez, A.; Halevy, A.H. Temperature requirements for floral development of herbaceous peony cv. “Sarah Bernhardt”. Sci. Hortic 2003, 97, 309–320. [Google Scholar]
- Li, X.; Yang, Y.; Yao, J.; Chen, G.; Li, X.; Zhang, Q.; Wu, C. FLEXIBLE CULM 1 encoding a cinnamyl-alcohol dehydrogenase controls culm mechanical strength in rice. Plant Mol. Biol 2009, 69, 685–697. [Google Scholar]
- Fulton, T.A.; Hall, A.J.; Catley, J.L. Chilling requirements of Paeoniacultivars. Sci. Hortic 2001, 89, 237–248. [Google Scholar]
- Halevy, A.H.; Levi, M.; Cohen, M.; Naor, V. Evaluation of methods forflowering advancement of herbaceous peonies. HortScience 2002, 37, 885–889. [Google Scholar]
- Cheng, F.Y.; Gao, S.P.; Yu, X.N. Stage identification and morphological types of bud maturity or flowering in Paeonia lactiflora cutivars (in Chinese). Acta Hortic. Sin 2009, 36, 611–613. [Google Scholar]
- Cosgrove, D.J. Growth of the plant cell wall. Nat. Rev. Mol. Cell Biol 2005, 6, 850–861. [Google Scholar]
- Hirano, K.; Kotake, T.; Kamihara, K.; Tsuna, K.; Aohara, T.; Kaneko, Y.; Takatsuji, H.; Tsumuraya, Y.; Kawasaki, S. Rice BRITTLE CULM 3 (BC3) encodes a classical dynamin OsDRP2B essential for proper secondary cell wall synthesis. Planta 2010, 232, 95–108. [Google Scholar]
- Li, Y.; Qian, Q.; Zhou, Y.; Yan, M.; Sun, L.; Zhang, M.; Fu, Z.; Wang, Y.; Han, B.; Pang, X.; et al. BRITTLE CULM1, which encodes a COBRA-like protein, affects the mechanical properties of rice plants. Plant Cell 2003, 15, 2020–2031. [Google Scholar]
- Zhong, R.Q.; Burk, D.H.; Morrison, W.H.; Ye, Z.H. A kinesin-like protein is essential for oriented deposition of cellulose microfibrils and cell wall strength. Plant Cell 2002, 14, 3101–3117. [Google Scholar]
- Hepler, P.K. Calcium: A central regulator of plant growth and development. Plant Cell 2005, 17, 2142–2155. [Google Scholar]
- Hepler, P.K.; Winship, L.J. Calcium at the cell wall-cytoplast interface. J. Integr. Plant Biol 2010, 52, 147–160. [Google Scholar]
- Gerasopoulos, D.; Chehli, B. Effects of pre-and postharvest calcium applications on the vase life of cut gerberas. J. Hortic. Sci. Biotechnol 1999, 74, 78–81. [Google Scholar]
- Chen, D.S.; Li, N.H.; Wang, J.M.; Ding, Y.X.; Wang, X.J. Effect of calcium chloride on preservation of cut flowers of Gerbera hybrid (in Chinese). Acta Botanica Yunnanica 2004, 26, 345–348. [Google Scholar]
- Yu, X.N.; Lu, G.P.; Cheng, F.Y.; Zheng, L.W. Effect of calcium on the stem quality of cut herbaceous peony. J. Hunan Agric. Univ. (Nat. Sci.) 2010, 36, 531–535. [Google Scholar]
- Zhang, B.; Zhou, Y. Rice brittleness mutants: A way to open the ‘black box’ of monocot cell wall biosynthesis. J. Integr. Plant Biol 2011, 53, 136–142. [Google Scholar]
- Walton, E.F.; Boldingh, H.L.; McLarenc, G.F.; Williamsa, M.H.; Jackman, R. The dynamics of starch and sugar utilisation in cut peony (Paeonia lactiflora Pall.) stems during storage and vase life. Postharvest Biol. Technol 2010, 58, 142–146. [Google Scholar]
- Burk, D.H.; Liu, B.; Zhong, R.; Morrison, W.H.; Ye, Z.H. A katanin-like protein regulates normal cell wall biosynthesis and cell elongation. Plant Cell 2001, 13, 807–827. [Google Scholar]
- Rose, J.K.; Hadfield, K.A.; Labavitch, J.M.; Bennett, A.B. Temporal sequence of cell wall disassembly in rapidly ripening melon fruit. Plant Physiol 1998, 117, 345–361. [Google Scholar]
- Manganaris, G.A.; Vasilakakis, M.; Mignani, I.; Diamantidis, G.; Tzavella-Klonari, K. The effect of preharvest calcium sprays on quality attributes, physicochemical aspects of cell wall components and susceptibility to brown rot of peach fruits (Prunus persica L. cv. Andross). Sci. Hortic 2005, 107, 43–50. [Google Scholar]
- Selvendran, R.R.; O’Neill, M.A. Isolation and analysis of cell wall from plant material. Meth. Biochem. Anal 1987, 32, 25–153. [Google Scholar]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem 1973, 54, 484–489. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem 1956, 28, 350–356. [Google Scholar]
- Updegraff, D.M. Semimicro determination of cellulose inbiological materials. Anal. Biochem 1969, 32, 420–424. [Google Scholar]
- Müse, G.; Schindler, T.; Bergfeld, R.; Ruel, K.; Jacquet, G.; Lapierre, C.; Speth, V.; Schopfer, P. Structure and distribution of lignin in primary and secondary cell walls of maize coleoptiles analyzed by chemical and immunological probes. Planta 1997, 201, 146–159. [Google Scholar]
- Hui, G.J.; Zheng, G.S.; Zhang, W.; Guo, S.X. Physiological and biochemical characteristics of cut peony flowers with preharvest spraying calcium (in Chinese). Acta Bot. Boreal. -Occident. Sin 2009, 29, 1246–1251. [Google Scholar]
- Capdevillea, G.D.; Maffiab, L.A.; Fingerc, F.L.; Batistab, U.G. Pre-harvest calcium sulfate applications affect vase life and severity of gray mold in cut roses. Sci. Hortic 2005, 103, 329–338. [Google Scholar]
- Huang, X.M.; Wang, H.C.; Gao, F.F.; Huang, H.B. A comparative study of the pericarp of litchi cultivars susceptible and resistant to fruit-cracking. J. Hortic. Sci. Biotechnol 1999, 74, 351–354. [Google Scholar]
- Lara, I.; Garcia, P.; Vendrell, M. Modifications in cell wall composition after cold storage of calcium-treated strawberry (Fragaria × ananassa Duch.) fruit. Postharvest Biol. Technol 2004, 34, 331–339. [Google Scholar]
- White, P.J.; Broadley, M.R. Calcium in plants. Ann. Bot 2003, 92, 487–511. [Google Scholar]
- Fang, Y.; Al-Assaf, S.; Phillips, G.O.; Nishinari, K.; Funami, T.; Williams, P.A. Binding behavior of calcium to polyuronates: Comparison of pectin with alginate. Carbohydr. Polym 2008, 72, 334–341. [Google Scholar]
- Fraeye, I.; Doungla, E.; Duvetter, T.; Moldenaers, P.; van Loey, A.; Hendrickx, M. Influence of intrinsic and extrinsic factors on rheology of pectin-calcium gels. Food Hydrocol 2009, 23, 2069–2077. [Google Scholar]
- Cao, Y.; Cao, Y.; Li, Z.; Ren, J.; Leng, P. Effect of pre-harvest calcium sprays on fruit quality and softening during storage of melting flesh peach. J. China Agric. Univ 2008, 13, 31–36. [Google Scholar]
- Manganaris, G.A.; Vasilakakis, M.; Diamantidis, G.; Ilaria, M. Effect of calcium additives on physicochemical aspects of cell wall pectin and sensory attributes of canned peach (Prunus persica (L) Batsch cv Andross). J. Sci. Food Agric 2005, 85, 1773–1778. [Google Scholar]
- Zhong, R.Q.; Peña, M.J.; Zhou, G.K.; Nairn, C.J.; Wood-Jones, A.; Richardson, E.A.; Morrison, W.H.; Darvill, A.G.; York, W.S.; Ye, Z.H. Arabidopsis Fragile Fiber 8, which encodes a putative glucuronyltransferase, is essential for normal secondary wall synthesis. Plant Cell 2005, 17, 3390–3408. [Google Scholar]
- Whang, J.; Zhu, J.; Lin, Q.; Li, X.; Teng, N.; Li, Z.; Li, B.; Zhang, A.; Lin, J. Influence of Stem structural features and cell wall components on the bending strength of wheat (Triticum aestivum L.) stems. Chin. Sci. Bull 2006, 51, 679–685. [Google Scholar]
- Liao, L.X.; Peng, Y.H.; Ye, Q.S. Neck-bending phenomena in cut gerbera flower (in Chinese). Acta Hortic. Sin 2003, 30, 110–112. [Google Scholar]

| Spray schedules | Breaking Force (N) | ||
|---|---|---|---|
| Top | Middle | Bottom | |
| Control (water) | 21.7 ± 0.8 e | 53.6 ± 3.1 c | 106.7 ± 4.5 c |
| Treatment 1 (4% calcium once) | 28.4 ± 1.4 bc | 60.3 ± 1.9 b | 113.9 ± 3.0 b |
| Treatment 2 (4% calcium twice) | 29.6 ± 1.1 b | 62.2 ± 1..5 b | 115.3 ± 3.9 b |
| Treatment 3 (4% calcium three times) | 32.9 ± 1.1 a | 67.3 ± 2.6 a | 125.5 ± 4.9 a |
| Treatment 4 (2% calcium three times) | 25.6 ± 1.6 d | 63.0 ± 1.8 b | 113.6 ± 2.8 b |
| Treatment 5 (6% calcium three times) | 27.0 ± 2.1 cd | 61.6 ± 2.8 b | 113.5 ± 2.8 b |
| Treatments | Calcium concentration | ||
|---|---|---|---|
| Top | Middle | Bottom | |
| Control | 14.40 ± 0.21 b | 13.10 ± 0.27 a | 11.81 ± 0.81 a |
| Calcium sprays | 16.03 ± 1.25 a | 14.04 ± 1.23 a | 12.50 ± 0.15 a |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, C.; Tao, J.; Zhao, D.; You, C.; Ge, J. Effect of Calcium Sprays on Mechanical Strength and Cell Wall Fractions of Herbaceous Peony (Paeonia Lactiflora Pall.) Inflorescence Stems. Int. J. Mol. Sci. 2012, 13, 4704-4713. https://doi.org/10.3390/ijms13044704
Li C, Tao J, Zhao D, You C, Ge J. Effect of Calcium Sprays on Mechanical Strength and Cell Wall Fractions of Herbaceous Peony (Paeonia Lactiflora Pall.) Inflorescence Stems. International Journal of Molecular Sciences. 2012; 13(4):4704-4713. https://doi.org/10.3390/ijms13044704
Chicago/Turabian StyleLi, Chengzhong, Jun Tao, Daqiu Zhao, Chao You, and Jintao Ge. 2012. "Effect of Calcium Sprays on Mechanical Strength and Cell Wall Fractions of Herbaceous Peony (Paeonia Lactiflora Pall.) Inflorescence Stems" International Journal of Molecular Sciences 13, no. 4: 4704-4713. https://doi.org/10.3390/ijms13044704
APA StyleLi, C., Tao, J., Zhao, D., You, C., & Ge, J. (2012). Effect of Calcium Sprays on Mechanical Strength and Cell Wall Fractions of Herbaceous Peony (Paeonia Lactiflora Pall.) Inflorescence Stems. International Journal of Molecular Sciences, 13(4), 4704-4713. https://doi.org/10.3390/ijms13044704




