Chelation of Cu(II), Zn(II), and Fe(II) by Tannin Constituents of Selected Edible Nuts
Abstract
:1. Introduction
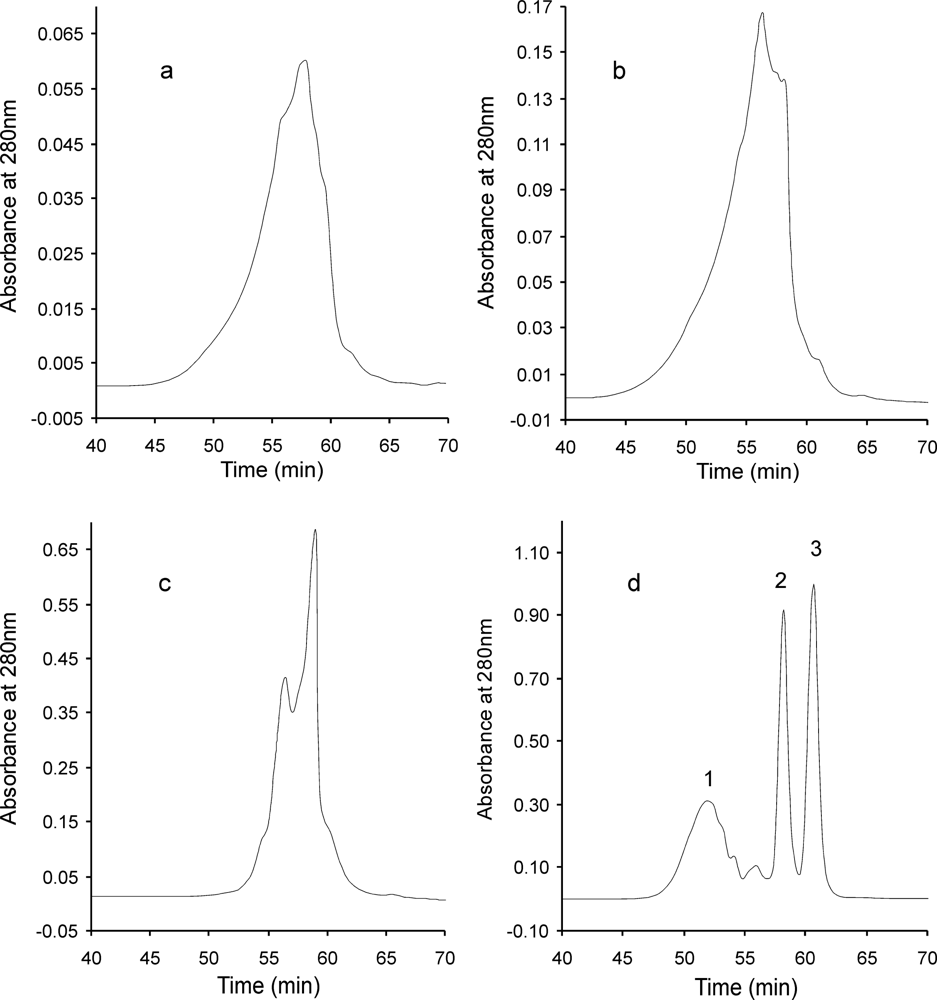
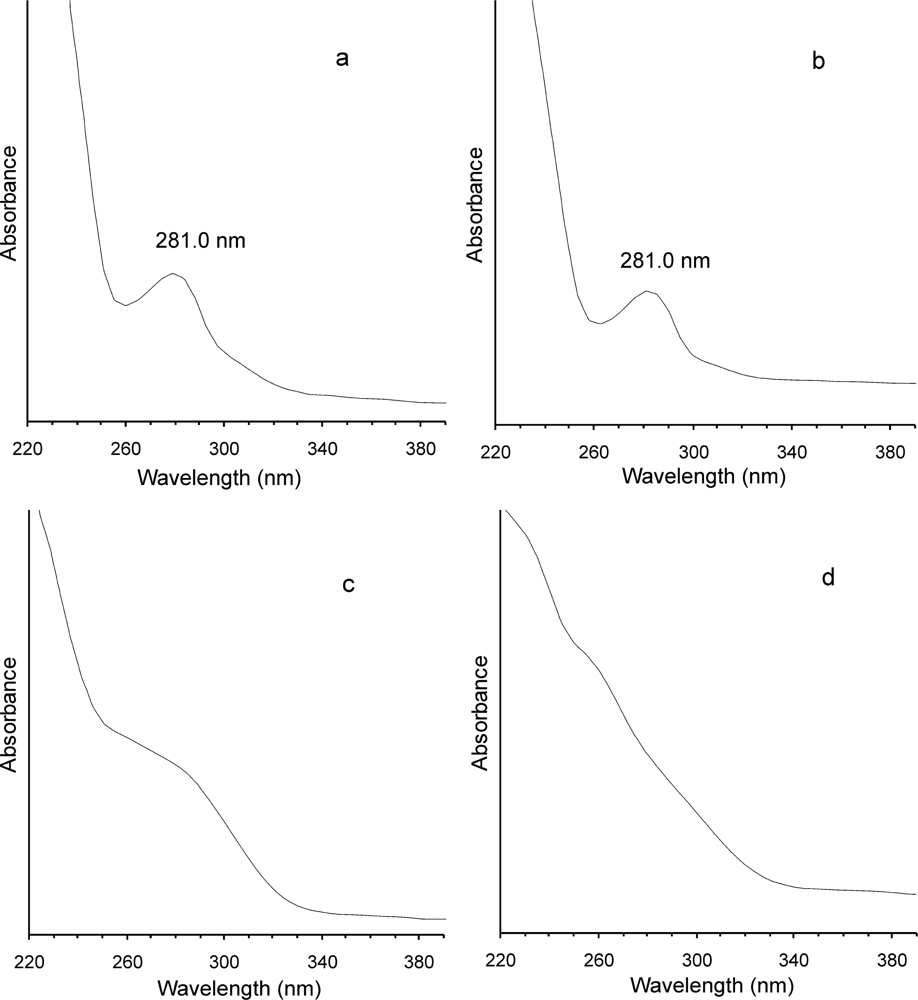
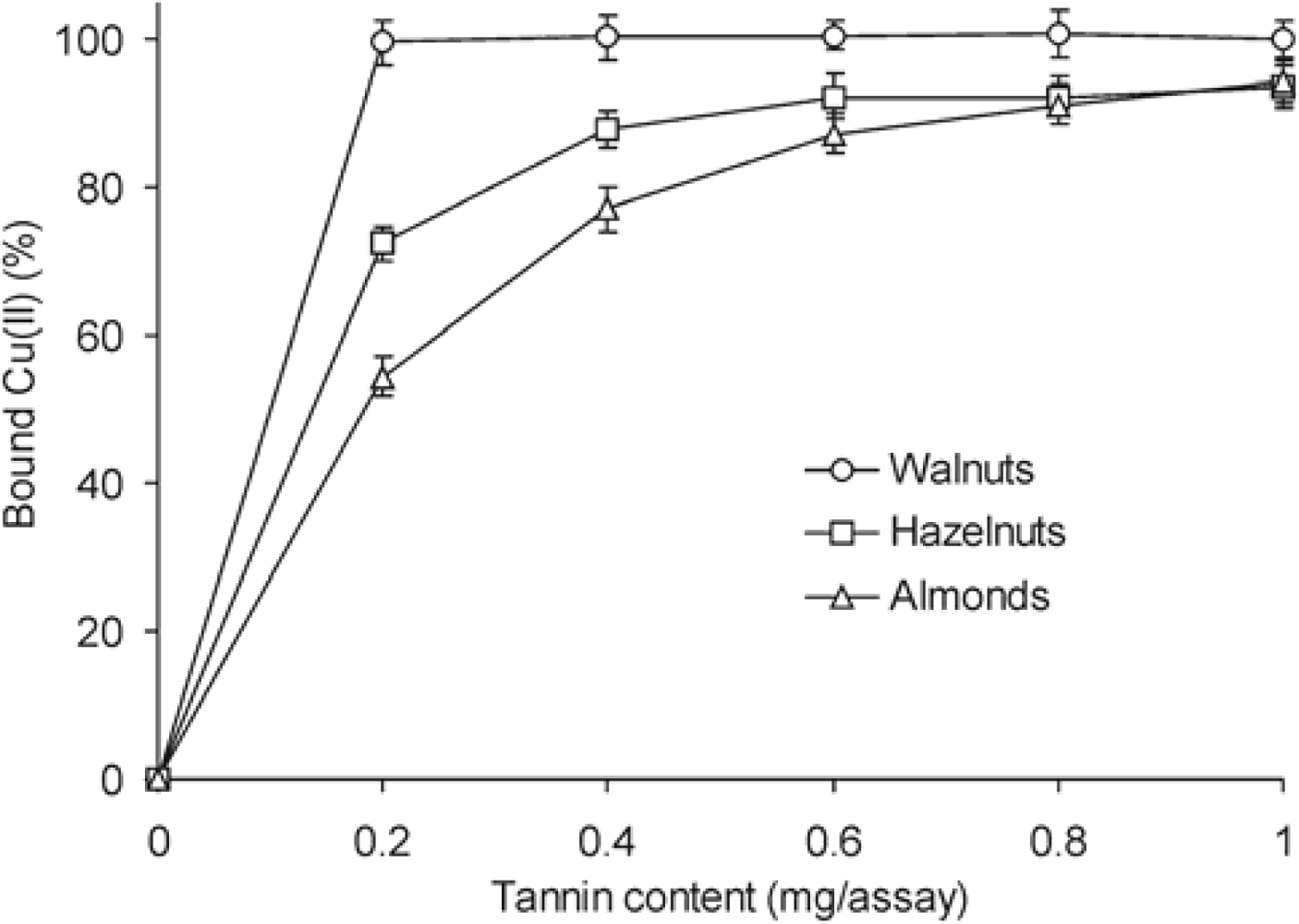
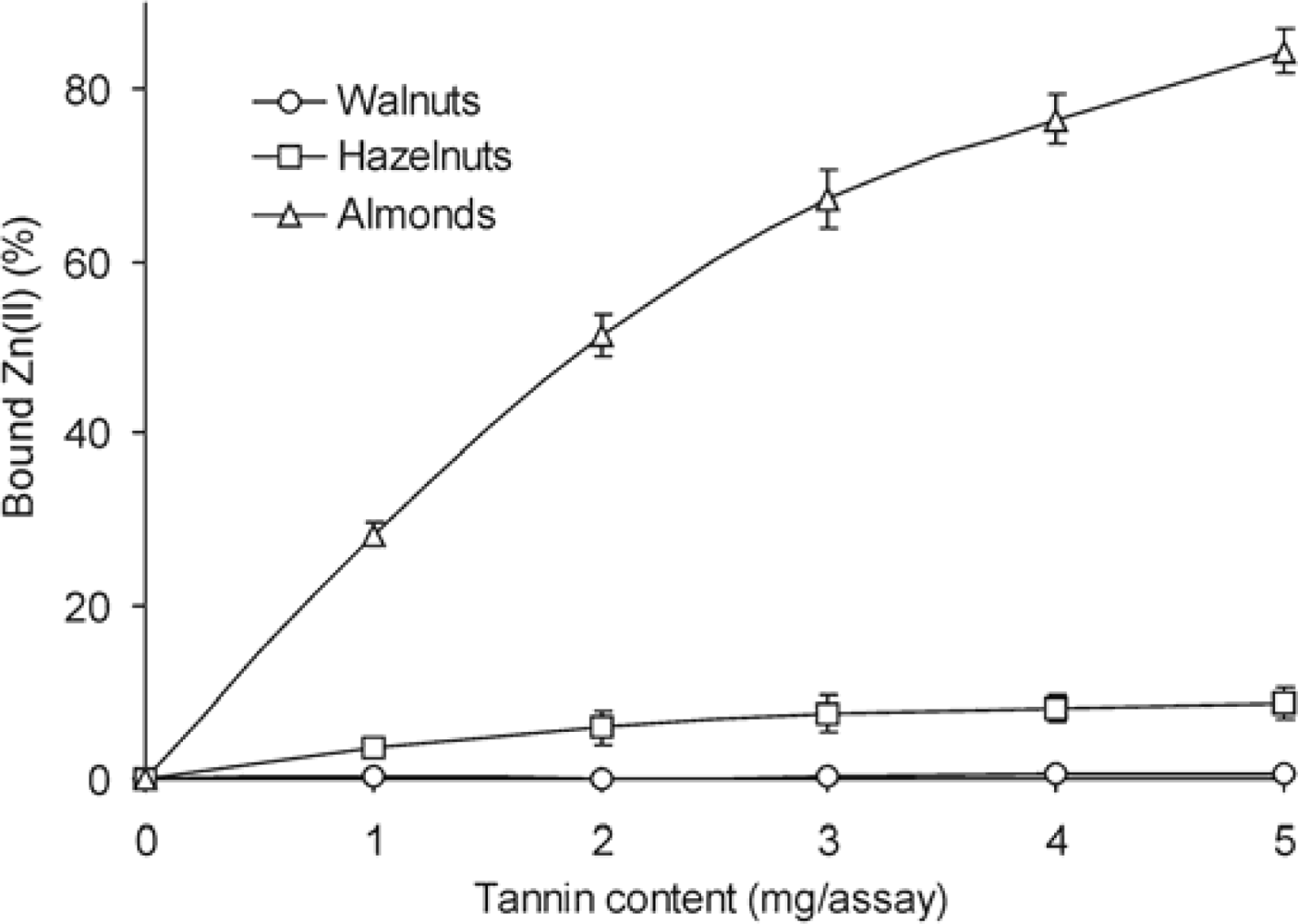
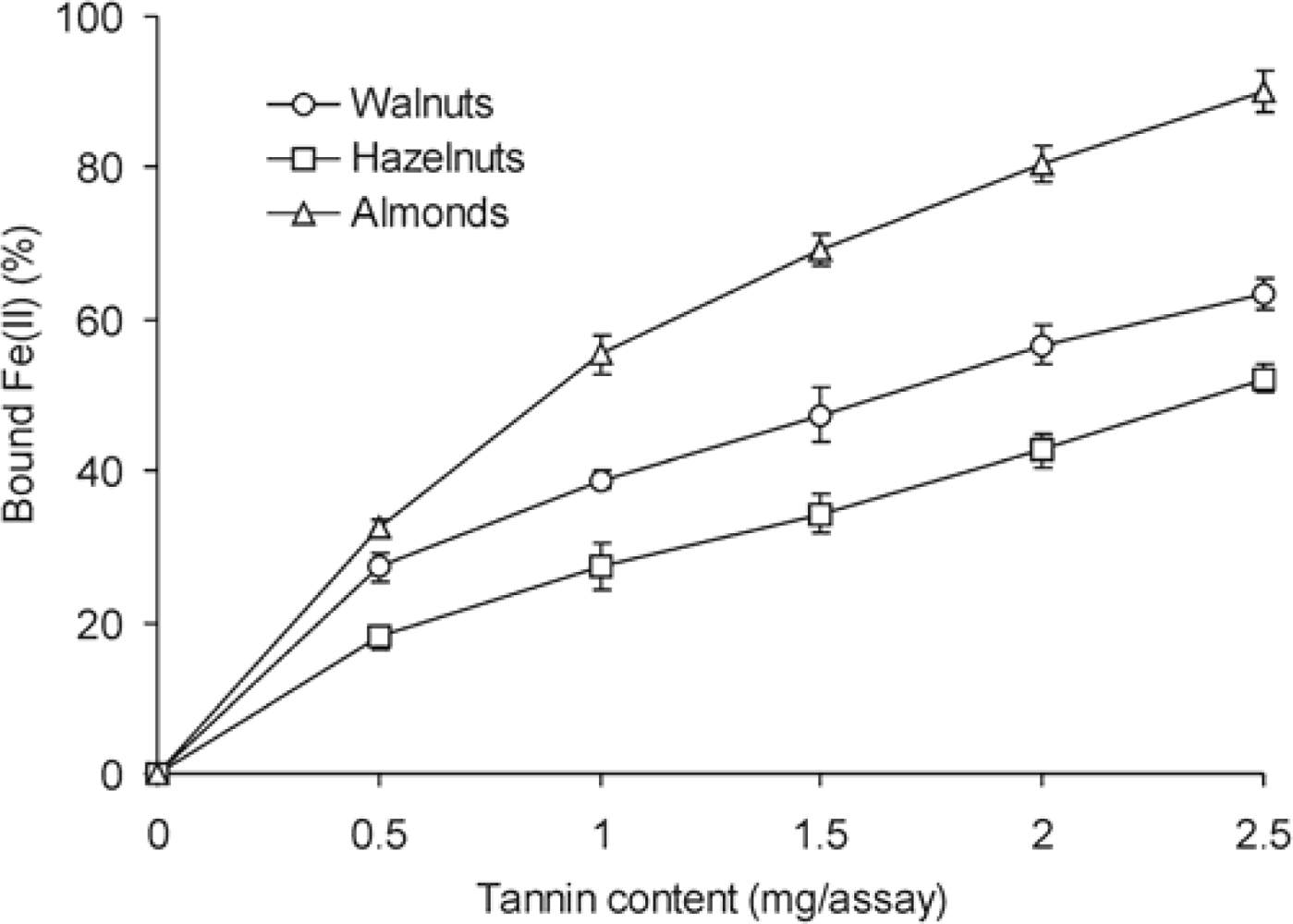
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Chemicals and Reagents
3.3. Isolation of Tannin Fractions
3.4. Content of Condensed Tannins
3.5. Protein Precipitation Capacity
3.6. SE-HPLC Method
3.7. Chelation Capacity
4. Conclusions
Acknowledgments
References and Notes
- Halvorsen, BL; Holte, K; Myhrstad, MCW; Barikmo, I; Hvattum, E; Remberg, SF; Wold, AB; Haffner, K; Baugerod, H; Andersen, LF; Moskaug, JO; Jacobs, DR; Blopmhoff, R. A systematic screening of total antioxidants in dietary plants. J. Nutr 2002, 132, 461–471. [Google Scholar]
- Blomhoff, R; Carlsen, MH; Andersen, LP; Jacobs, DR, Jr. Health benefits of nuts: potential role of antioxidants. Brit J Nutr 2006, 96, S52–S60. [Google Scholar]
- Yurttas, HC; Schafer, HW; Warthesen, JJ. Antioxidant activity of nontocopherol hazelnut (Corylus spp.) phenolics. J Food Sci 2000, 65, 276–280. [Google Scholar]
- Wijeratne, SSK; Abou-Zaid, MM; Shahidi, F. Antioxidant polyphenols in almond and its coproducts. J. Agric. Food Chem 2006, 54, 312–318. [Google Scholar]
- Alasalvar, C; Karamać, M; Amarowicz, R; Shahidi, F. Antioxidant and antiradical activities in extracts of hazelnut kernel (Corylus avellana L.) and hazelnut green leafy cover. J Agric Food Chem 2006, 54, 4826–4832. [Google Scholar]
- Li, L; Tsao, R; Yang, R; Liu, C; Zhu, H; Young, JC. Polyphenolic profiles and antioxidant activities of heartnut (Juglans ailanthifolia var. cordiformis) and persian walnut (Juglans regia L.). J. Agric. Food Chem 2006, 54, 8033–8040. [Google Scholar]
- Garrido, I; Monagas, M; Gomez-Cordoves, C; Bartolome, B. Polyphenols and antioxidant properties of almond skins: Influence of industrial processing. J Food Sci 2008, 73, 106–115. [Google Scholar]
- Gu, L; Kelm, MA; Hammerstone, JF; Beecher, G; Holden, J; Haytowitz, D; Gebhardt, S; Prior, RL. Concentrations of proanthocyanidins in common foods and estimations of normal consumption. J. Nutr 2004, 134, 613–617. [Google Scholar]
- Amarowicz, R; Troszyńska, A; Shahidi, F. Antioxidant activity of almond seed extract and its fractions. J. Food Lipids 2005, 12, 344–358. [Google Scholar]
- Alasalvar, C; Karamać, M; Kosińska, A; Rybarczyk, A; Shahidi, F; Amarowicz, R. Antioxidant activity of hazelnut skin phenolics. J. Agric. Food Chem 2009, 57, 4645–4650. [Google Scholar]
- Prodanov, M; Garrido, I; Vacas, V; Lebron-Aguilar, R; Duenas, M; Gomez-Cordoves, C; Bartolome, B. Ultrafiltration as alternative purification procedure for the characterization of low and high molecular-mass phenolics from almond skins. Anal. Chim. Acta 2008, 609, 241–251. [Google Scholar]
- Arranz, S; Perez-Jimenez, J; Saura-Calixto, F. Antioxidant capacity of walnut (Juglans regia L.): Contribution of oil and defatted matter. Eur. Food Res. Technol 2008, 227, 425–431. [Google Scholar]
- Fukuda, T; Ito, H; Yoshida, T. Antioxidative polyphenols from walnuts (Juglans regia L.). Phytochem 2003, 63, 795–801. [Google Scholar]
- Ito, H; Okuda, T; Fukuda, T; Hatano, T; Yoshida, T. Two novel dicarboxylic acid derivatives and a new dimeric hydrolyzable tannin from walnuts. J. Agric. Food Chem 2007, 55, 672–679. [Google Scholar]
- Hagerman, AE; Riedl, KM; Jones, GA; Sovik, KN; Ritchard, NT; Hartzfeld, PW; Riechel, TL. High molecular weight plant polyphenolics (tannins) as biological antioxidants. J. Agric. Food Chem 1998, 46, 1887–1892. [Google Scholar]
- Yokazawa, T; Chen, CP; Dong, E; Tanaka, T; Nonaka, GI; Nishioka, I. Study on the inhibitory effect of tannins and flavonoids against the 1,1-diphenyl-2-picrylhydrazyl radical. Biochem. Pharmacol 1998, 56, 213–222. [Google Scholar]
- deGaulejac, NS; Provost, C; Vivas, N. Comparative study of polyphenol scavenging activities assessed by different methods. J. Agric. Food Chem 1999, 47, 425–431. [Google Scholar]
- Karamać, M. In vitro study on efficacy of tannin fractions of edible nuts as antioxidants. Eur. J. Lipid Sci. Technol 2009, 111, 1063–1071. [Google Scholar]
- Borg, DC. Oxygen free radicals and tissue injury. In Oxygen Free Radicals in Tissue Damage, 1st ed; Tarr, M, Samson, F, Eds.; Birkhauser: Boston, MA, USA, 1993; pp. 12–53. [Google Scholar]
- Lopes, GKB; Schulman, HM; Hermes-Lima, M. Polyphenol tannic acid inhibits hydroxyl radical formation from Fenton reaction by complexing ferrous ions. Biochim. Biophys. Acta 1999, 1472, 142–152. [Google Scholar]
- Andrade, RG, Jr; Dalvi, LT; Silva, JMC, Jr; Lopes, GKB; Alonso, A; Hermes-Lima, M. The antioxidant effect of tannic acid on the in vitro copper-mediated formation of free radicals. Arch. Biochem. Biophys 2005, 437, 1–9. [Google Scholar]
- Mila, I; Scalbert, A; Expert, D. Iron withholding by plant polyphenols and resistance to pathogens and rots. Phytochem 1996, 42, 1551–1555. [Google Scholar]
- South, PK; Miller, DD. Iron binding by tannic acid: Effects of selected ligands. Food Chem 1998, 63, 167–172. [Google Scholar]
- Gu, L; Kelm, MA; Hammerstone, JF; Beecher, G; Holden, J; Haytowitz, D; Prior, RL. Screening of foods containing proanthocyanidins and their structural characterization using LC-MS/MS and thiolytic degradation. J. Agric. Food Chem 2003, 51, 7513–7521. [Google Scholar]
- Hagerman, AE. Chemistry of tannin-protein complexation. In Chemistry and Significance of Condensed Tannins, 1st ed; Hemingway, RW, Karchesy, JJ, Eds.; Plenum Press: New York, NY, USA, 1989; pp. 323–333. [Google Scholar]
- Sarni-Manchado, P; Cheynier, V; Moutounet, M. Interactions of grape seed tannins with salivary proteins. J. Agric. Food Chem 1999, 47, 42–47. [Google Scholar]
- Kawamoto, H; Nakatsubo, F; Murakami, K. Quantitative determination of tannin and protein precipitates by high-performance liquid chromatography. Phytochemistry 1995, 40, 1503–1505. [Google Scholar]
- Karamać, M. Fe(II), Cu(II) and Zn(II) chelating activity of buckwheat and buckwheat groats tannin fractions. Pol. J. Food Nutr. Sci 2007, 57, 357–362. [Google Scholar]
- Wong, SP; Leong, LP; Koh, JHW. Antioxidant activities of aqueous extracts of selected plants. Food Chem 2006, 99, 775–783. [Google Scholar]
- Asakura, T; Nakamura, Y; Inoue, N; Murata, M; Homma, S. Characterization of zinc chelating compounds in instant coffee. Agric. Biol. Chem 1990, 54, 855–862. [Google Scholar]
- Karamać, M; Pegg, RB. Limitations of the tetramethylmurexide assay for investigating the Fe(II) chelation activity of phenolic compounds. J. Agric. Food Chem 2009, 57, 6425–6431. [Google Scholar]
- Mira, L; Fernandez, MT; Santos, M; Rocha, R; Florencio, MH; Jennings, KR. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res 2002, 36, 1199–1208. [Google Scholar]
- Kumamoto, M; Sonda, T; Nagayama, K; Tabata, M. Effects of pH and metal ions on antioxidative activities of catechins. Biosc. Biotechnol. Biochem 2001, 65, 126–132. [Google Scholar]
- McDonald, M; Mila, I; Scalbert, A. Precipitation of metal ions by plant polyphenols: Optimal conditions and origin of precipitation. J. Agric. Food Chem 1996, 44, 599–606. [Google Scholar]
- Brown, JE; Khodr, H; Hider, RC; Rice-Evans, CA. Structural dependence of flavonoid interactions with Cu2+ ions: Implications for their antioxidant properties. Biochem. J 1998, 330, 1173–1178. [Google Scholar]
- Brown, JE; Kelly, MF. Inhibition of lipid peroxidation by anthocyanins, anthocyanidins and their phenolic degradation products. Eur. J. Lipid Sci. Technol 2007, 109, 66–71. [Google Scholar]
- Zhou, K; Yin, JJ; Yu, LL. ESR determination of the reactions between selected phenolic acids and free radicals or transition metals. Food Chem 2006, 95, 446–457. [Google Scholar]
- Andjelkovic, M; van Camp, J; de Meulenaer, B; Depaemelaere, G; Socaciu, C; Verloo, M; Verhe, R. Iron–chelation properties of phenolic acids bearing catechol and galloyl groups. Food Chem 2006, 98, 23–31. [Google Scholar]
- Miller, NJ; Castelluccio, C; Tijburg, L; RiceEvans, C. The antioxidant properties of theaflavins and their gallate esters–radical scavengers or metal chelators? FEBS Lett 1996, 392, 40–44. [Google Scholar]
- Hider, RC; Liu, ZD; Khodr, HH. Metal chelation of polyphenols. Methods Enzymol 2001, 335, 190–203. [Google Scholar]
- Amarowicz, R; Piskuła, M; Honke, J; Rudnicka, B; Troszyńska, A; Kozłowska, H. Extraction of phenolic compounds from lentil seeds (Lens culinaris) with various solvents. Pol. J. Food Nutr. Sci 1995, 45, 53–62. [Google Scholar]
- Strumeyer, DH; Malin, MJ. Condensed tannins in grain sorghum: Isolation, fractionation, and characterisation. J. Agric. Food Chem 1975, 23, 909–914. [Google Scholar]
- Price, ML; van Scoyoc, S; Butler, LG. A critical evaluation of the vanillic reaction as an assay for tannin in sorghum grain. J. Agric. Food Chem 1978, 26, 1214–1218. [Google Scholar]
- Hagerman, AE; Butler, LG. Protein precipitation method for the quantitative determination of tannins. J. Agric. Food Chem 1978, 26, 809–812. [Google Scholar]
- Boyer, RF; McCleary, CJ. Superoxide ion as a primary reductant in ascorbate-mediated ferritin iron release. Free Radic. Biol. Med 1987, 3, 389–395. [Google Scholar]
| Nuts tannin fraction | Condensed tannins (mg CEa/g) | Protein precipitation capacity (mg TAEb/g) |
|---|---|---|
| Walnuts | 147 ± 4 | 873 ± 12 |
| Hazelnuts | 1261 ± 12 | 940 ± 24 |
| Almonds | 776 ± 8 | 69 ± 9 |
| Nuts tannin fraction | μmol metal ions chelated by 0.5 mg of tannin fraction | ||
|---|---|---|---|
| Fe(II)a | Cu(II)b | Zn(II)b | |
| Walnuts | 2.7 ± 0.08 | 25.0 ± 0.90 | 0.1 ± 0.01 |
| Hazelnuts | 1.8 ± 0.11 | 22.5 ± 1.03 | 1.5 ± 0.08 |
| Almonds | 3.2 ± 0.14 | 20.5 ± 0.89 | 11.4 ± 0.57 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Karamać, M. Chelation of Cu(II), Zn(II), and Fe(II) by Tannin Constituents of Selected Edible Nuts. Int. J. Mol. Sci. 2009, 10, 5485-5497. https://doi.org/10.3390/ijms10125485
Karamać M. Chelation of Cu(II), Zn(II), and Fe(II) by Tannin Constituents of Selected Edible Nuts. International Journal of Molecular Sciences. 2009; 10(12):5485-5497. https://doi.org/10.3390/ijms10125485
Chicago/Turabian StyleKaramać, Magdalena. 2009. "Chelation of Cu(II), Zn(II), and Fe(II) by Tannin Constituents of Selected Edible Nuts" International Journal of Molecular Sciences 10, no. 12: 5485-5497. https://doi.org/10.3390/ijms10125485
APA StyleKaramać, M. (2009). Chelation of Cu(II), Zn(II), and Fe(II) by Tannin Constituents of Selected Edible Nuts. International Journal of Molecular Sciences, 10(12), 5485-5497. https://doi.org/10.3390/ijms10125485





