A Study of the Crystallization, Melting, and Foaming Behaviors of Polylactic Acid in Compressed CO2
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isothermal Crystallization of PLA
2.2. Cold-crystallization and Melting Behavior of PLA Treated by CO2
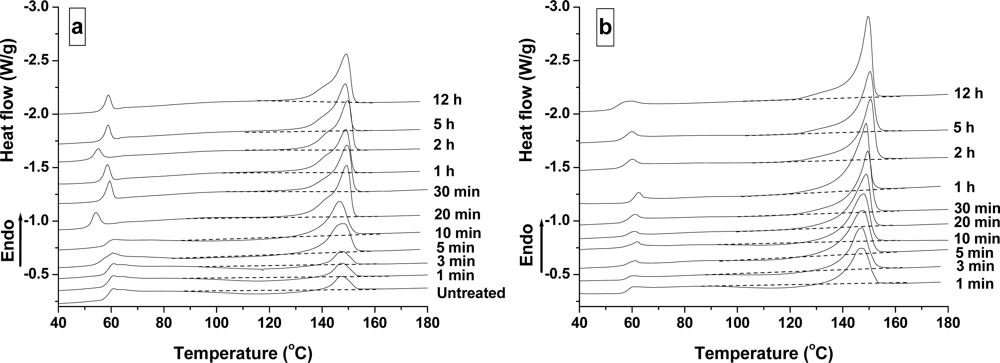
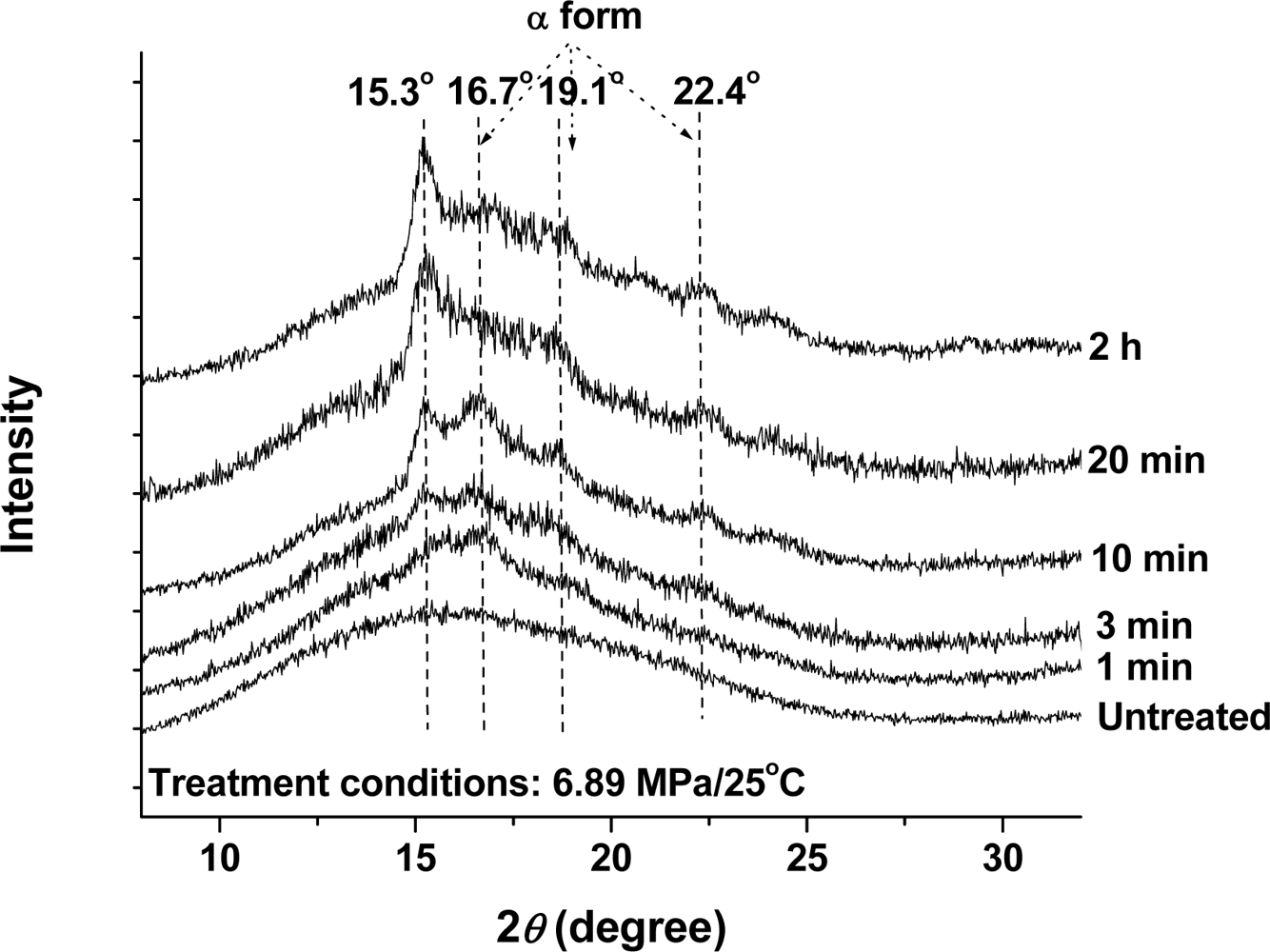
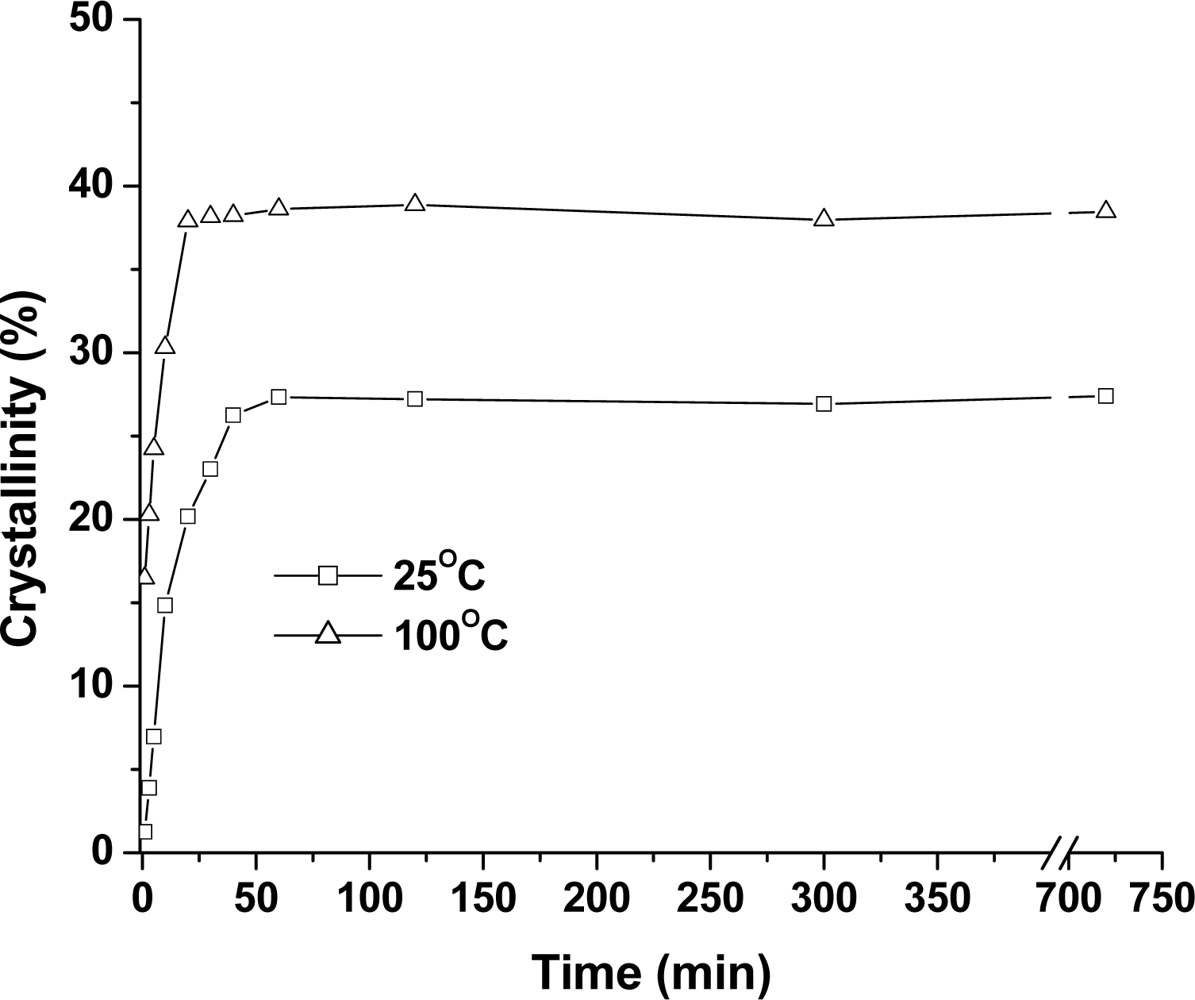
2.2.1. Effect of Time
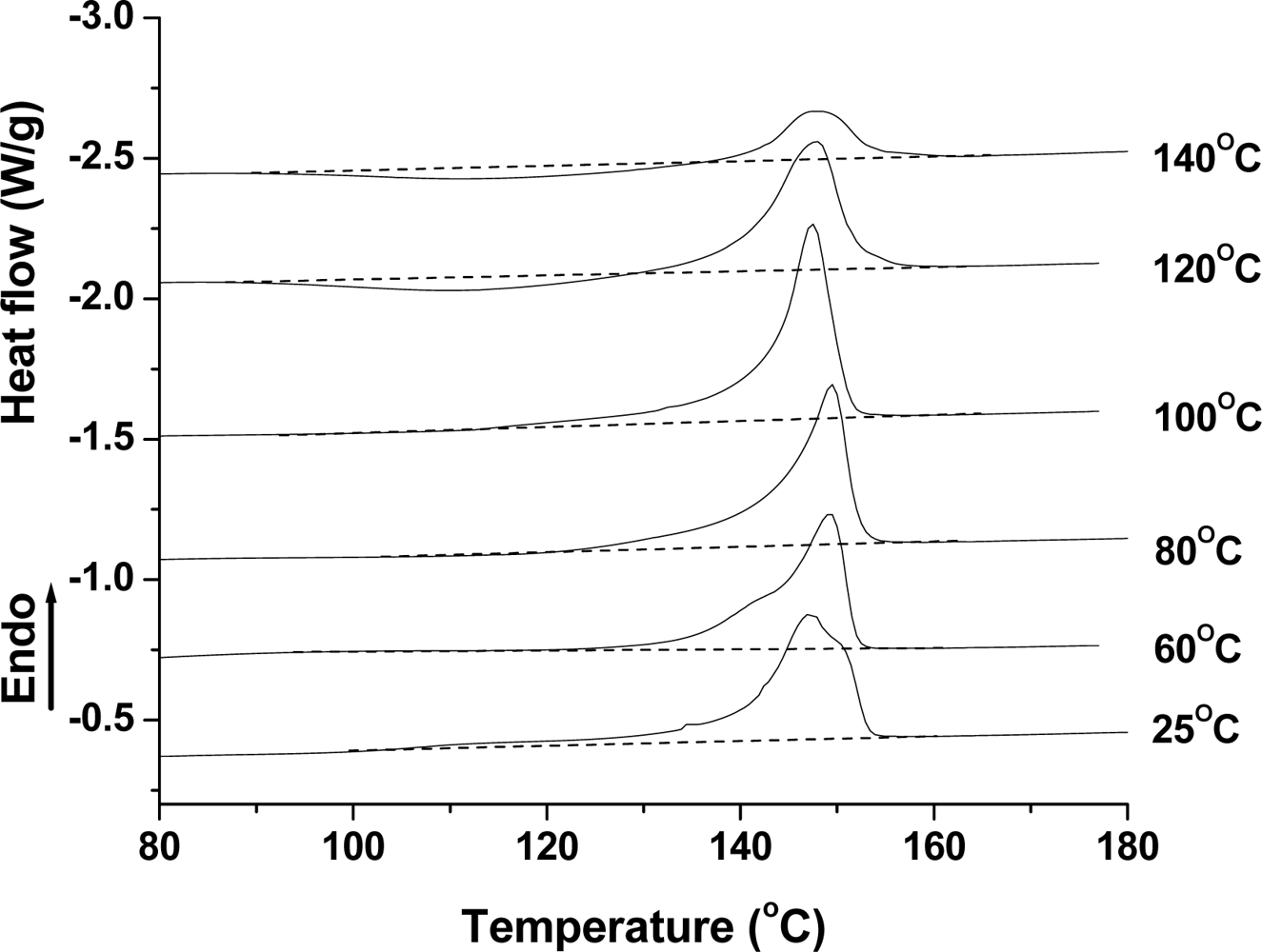
2.2.2. Effect of Temperature
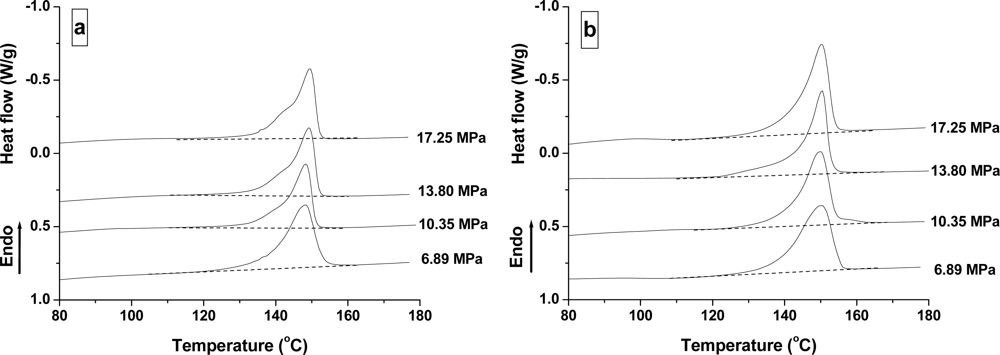
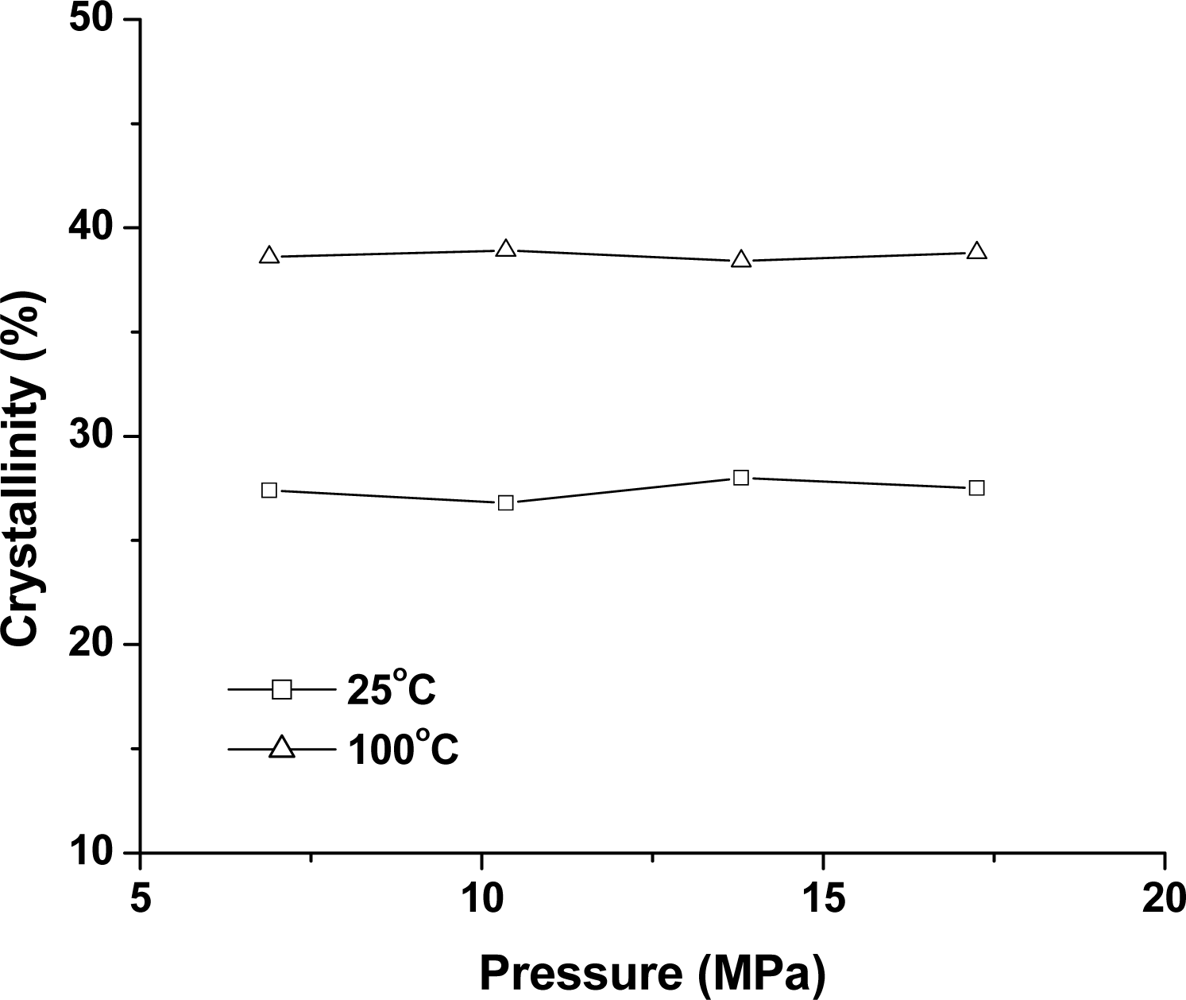
2.2.3. Effect of Pressure
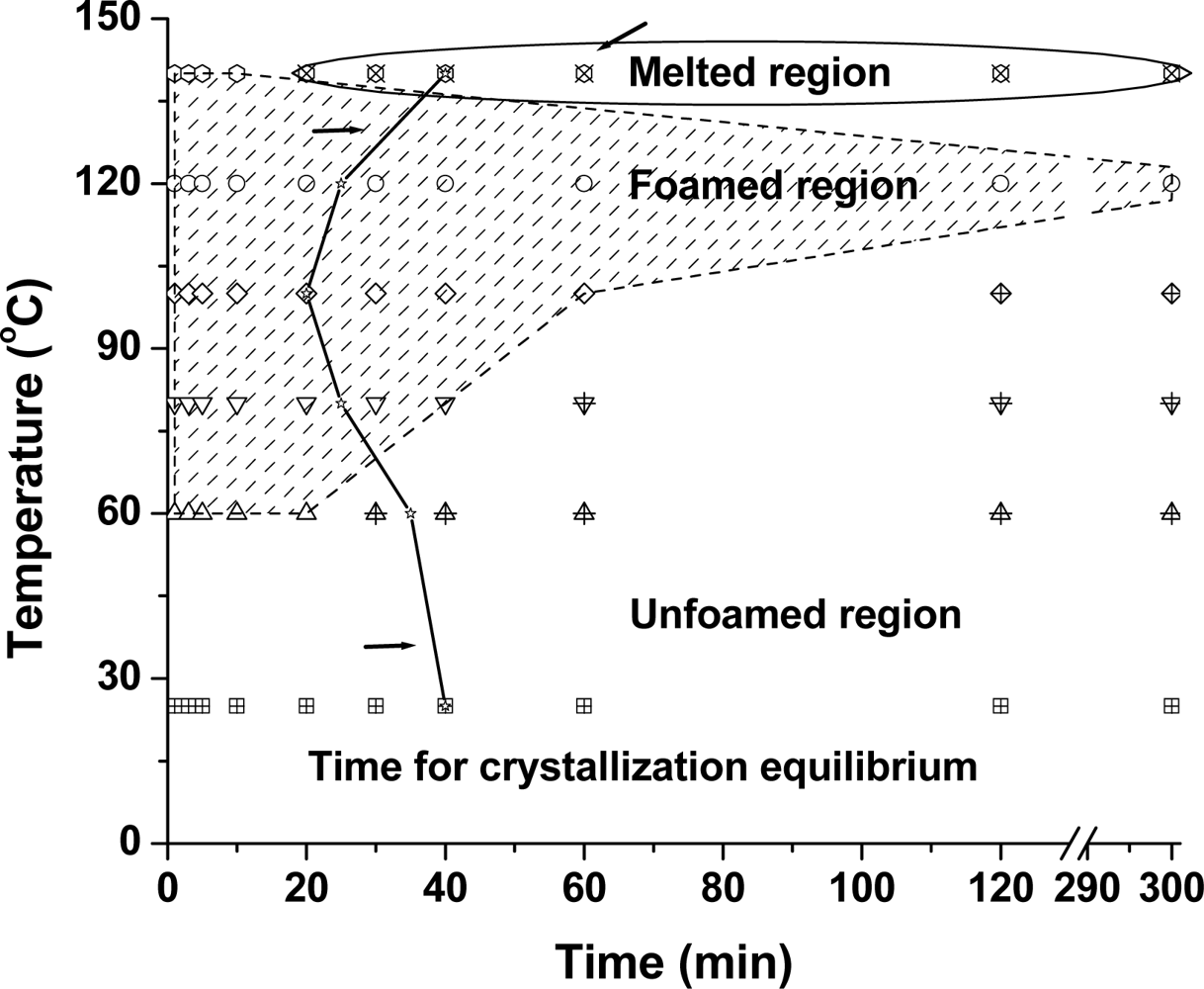
2.3. The Induced Crystallization of PLA during the Foaming Process
3. Experimental Section
3.1. Materials and Samples Preparation
3.2. High Pressure CO2 Treatment
3.3. Analysis
4. Conclusions
Acknowledgments
References and Notes
- Mandelkern, L. Crystallization of Polymers; McGraw-Hill: New York, NY, USA, 1964. [Google Scholar]
- Lunt, J. Large-scale production, properties and commercial applications of polylactic acid polymers. Polym. Degard. Stab 1998, 59, 145–152. [Google Scholar]
- Gupta, B; Revagade, N; Hilborn, J. Poly(lactic acid) fiber: An overview. Prog. Polym. Sci 2007, 32, 455–482. [Google Scholar]
- Södegård, A; Stolt, M. Properties of lactic acid based polymers and their correlation with composition. Prog. Polym. Sci 2002, 27, 1123–1163. [Google Scholar]
- Auras, R; Harte, B; Selke, S. An overview of polylactides as packaging materials. Macromol. Biosci 2004, 4, 835–864. [Google Scholar]
- Lim, LT; Auras, R; Rubino, M. Processing technologies for poly(lactic acid). Prog. Polym. Sci 2008, 33, 820–852. [Google Scholar]
- Yasuniwa, M; Tsubakihara, S; Iura, K; Ono, Y; Dan, Y; Takahashi, K. Crystallization behavior of poly(l-lactic acid). Polymer 2006, 47, 7554–7563. [Google Scholar]
- Yasuniwa, M; Iura, K; Dan, Y. Melting behavior of poly(l-lactic acid): Effects of crystallization temperature and time. Polymer 2007, 48, 5398–5407. [Google Scholar]
- Tsuji, H; Takai, H; Saha, SK. Isothermal and non-isothermal crystallization behavior of poly(l-lactic acid): Effect of stereocomplex as nucleating agent. Polymer 2006, 47, 3826–3837. [Google Scholar]
- Yu, L; Dean, K; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci 2006, 31, 576–602. [Google Scholar]
- Pillin, I; Momtrelay, N; Grohens, Y. Thermo-mechanical characterization of plasticized PLA: Is the miscibility the only significant factor. Polymer 2006, 47, 4676–4682. [Google Scholar]
- Li, H; Huneault, MA. Crystallization of PLA/thermoplastic starch blends. Int. Polym. Process 2008, 5, 412–418. [Google Scholar]
- Yeh, JT; Wu, CJ; Tsou, CH; Chai, WL; Chow, JD; Huang, CY; Chen, KN; Wu, CS. Study on the crystallization, miscibility, morphology, properties of poly(lactic acid)/poly(ɛ-caprolactone) blends. Polym. Plast. Tech. Eng 2009, 48, 571–578. [Google Scholar]
- Li, Y; Wang, Y; Liu, Li; Han, L; Xiang, F; Zhou, Z. Crystallization improvement of poly(l-lactide) induced by functionalized multiwalled carbon nanotubes. J. Polym. Sci. Part: B Polym. Phys 2009, 47, 326–339. [Google Scholar]
- Day, M; Nawaby, AV; Liao, X. A DSC study of the crystallization behaviour of polylactic acid and its nanocomposites. J. Therm. Anal. Calorim 2006, 86, 623–629. [Google Scholar]
- Li, H; Huneault, MA. Effect of nucleation and plasticization on the crystallization of poly(lactic acid). Polymer 2007, 48, 6855–6866. [Google Scholar]
- Chapleau, N; Huneault, MA; Li, H. Biaxial orientation of polylactide/thermoplastic starch blends. Int. Polym. Process 2007, 22, 402–409. [Google Scholar]
- Mihai, M; Huneault, MA; Favis, BD. Crystallinity development in cellular poly(lactic acid) in the presence of supercritical carbon dioxide. J. Appl. Polym. Sci 2009, 113, 2920–2932. [Google Scholar]
- Kokturk, G; Serhatkulu, TF; Cakmak, M; Piskin, E. Evolution of phase behavior and orientation in uniaxially deformed polylactic acid films. Polym. Eng. Sci 2002, 42, 1619–1628. [Google Scholar]
- Zhang, Z; Handa, YP. CO2-assisted melting of semicrystalline polymers. Macromolecules 1997, 30, 8505–8507. [Google Scholar]
- Handa, YP; Roovers, J; Wang, F. Effect of thermal annealing and supercritical fluids on the crystallization behavior of methyl-substituted poly(aryl ether ether ketone). Macromolecules 1994, 27, 5511–5516. [Google Scholar]
- Gross, SM; Roberts, GW; Kiserow, DJ; Desimone, JM. Crystallization and solid-state polymerization of poly(bisphenol A carbonate) facilitated by supercritical CO2. Macromolecules 2000, 33, 40–45. [Google Scholar]
- Becjman, E; Porter, RS. Crystallization of bisphenol A polycarbonate induced by supercritical carbon dioxide. J. Polym. Sci. Part B: Polym. Phys 1987, 25, 1511–1517. [Google Scholar]
- Liao, X; Wang, J; Li, G; He, JS. Effect of supercritical carbon dioxide on the crystallization and melting behavior of linear bisphenol A polycarbonate. J. Polym. Sci. Part B: Polym. Phys 2004, 42, 280–285. [Google Scholar]
- Zhai, WT; Yu, J; Ma, WM; He, JS. Cosolvent effect of water in supercritical carbon dioxide facilitating induced crystallization of polycarbonate. Polym. Eng. Sci 2007, 47, 1338–1343. [Google Scholar]
- Zhai, WT; Yu, J; Ma, WM; He, JS. Influence of long-chain branching on the crystallization and melting of polycarbonates in supercritical CO2. Macromolecules 2007, 40, 73–80. [Google Scholar]
- Handa, YP; Zhang, ZY; Roovers, J. Compressed-gas-induced crystallization in tert-butyl poly(ether ether ketone). J. Polym. Sci. Part B: Polym.Phys 2001, 39, 1505–1512. [Google Scholar]
- Yu, L; Liu, H; Dean, K. Thermal behavior of poly(lactic acid) in contact with compressed carbon dioxide. Polym. Int 2009, 58, 368–372. [Google Scholar]
- Yu, L; Liu, H; Dean, K; Chen, L. Cold crystallization and postmelting crystallization of PLA plasticized by compressed carbon dioxide. J. Polym. Sci. Part B: Polym. Phys 2008, 46, 2630–2636. [Google Scholar]
- Wang, X; Kumar, V; Li, W. Low density sub-critical CO2-blown solid-state PLA foams. Cell. Polym 2007, 26, 11–35. [Google Scholar]
- López-Periago, A; García-González, CA; Domingo, C. Solvent- and thermal-induced crystallization of poly-l-lactic acid in supercritical CO2 medium. J. Appl. Polym. Sci 2009, 111, 291–300. [Google Scholar]
- Takada, M; Hasegawa, S; Ohahima, M. Crystallization kinetics of poly(l-lactide) in contact with pressurized CO2. Polym. Eng. Sci 2004, 44, 186–196. [Google Scholar]
- Marubayashi, H; Akaishi, S; Akasaka, S; Asai, S; Sumita, M. Crystalline structure and morphology of poly(l-lactide) formed under high-pressure CO2. Macromolecules 2008, 41, 9192–9203. [Google Scholar]
- Hoogsteen, W; Postema, AR; Pennings, AJ; Ten Brinke, G; Zugenmaier, P. Crystal structure, conformation, and morphology of solution-spun poly(l-lactide) fibers. Macromolecules 1990, 23, 634–642. [Google Scholar]
- Keith, HD. Physics and Chemistry of the Organic Solid State; Fox, D, Labes, MM, Weissberger, A, Eds.; Interscience Publishers: New York, NY, USA, 1963; Volume 1. [Google Scholar]
- Xu, Z; Jiang, X; Liu, T; Hu, G; Zhao, L; Zhu, Z; Yuan, W. Foaming of polypropylene with supercritical carbon dioxide. J. Super. Fluids 2007, 41, 299–310. [Google Scholar]
- Lips, PAM; Velthoen, IW; Dijkstra, PJ; Wessling, M; Feijen, J. Gas foaming of segmented poly(ester amide) films. Polymer 2005, 46, 9396–9403. [Google Scholar]
- Mascia, L; Delre, G; Ponti, PP; Bologna, S; Di Giacomo, G; Haworth, B. Crystallization effects on autoclave foaming of polycarbonate using supercritical carbon dioxide. Adv. Polym. Tech 2006, 25, 255–235. [Google Scholar]
- Fischer, EW; Sterzel, HJ; Wegner, G. Investigation of the structure of solution grown crystals of lactide copolymers by means of chemical reaction. Kolloid-Zu Z-Polymer 1973, 251, 980–990. [Google Scholar]




© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhai, W.; Ko, Y.; Zhu, W.; Wong, A.; Park, C.B. A Study of the Crystallization, Melting, and Foaming Behaviors of Polylactic Acid in Compressed CO2. Int. J. Mol. Sci. 2009, 10, 5381-5397. https://doi.org/10.3390/ijms10125381
Zhai W, Ko Y, Zhu W, Wong A, Park CB. A Study of the Crystallization, Melting, and Foaming Behaviors of Polylactic Acid in Compressed CO2. International Journal of Molecular Sciences. 2009; 10(12):5381-5397. https://doi.org/10.3390/ijms10125381
Chicago/Turabian StyleZhai, Wentao, Yoorim Ko, Wenli Zhu, Anson Wong, and Chul B. Park. 2009. "A Study of the Crystallization, Melting, and Foaming Behaviors of Polylactic Acid in Compressed CO2" International Journal of Molecular Sciences 10, no. 12: 5381-5397. https://doi.org/10.3390/ijms10125381
APA StyleZhai, W., Ko, Y., Zhu, W., Wong, A., & Park, C. B. (2009). A Study of the Crystallization, Melting, and Foaming Behaviors of Polylactic Acid in Compressed CO2. International Journal of Molecular Sciences, 10(12), 5381-5397. https://doi.org/10.3390/ijms10125381




