Predicting Vapour Pressures of Organic Compounds from Their Chemical Structure for Classification According to the VOCDirective and Risk Assessment in General
Abstract
:Introduction
| Compound | DIPPR [3] [Pa] | TRC [4] [Pa] | IUCLID [5] [Pa] | N’Guimbi et al. [6] [Pa] |
|---|---|---|---|---|
| 1-Hexanol | 72.38 | 47.32 | 100 | 71.73 |
| 1-Decanol | 0.63 | 5.34 | NA | 0.67 |
| Methoxyacetic acid | 7.65 | 18.6 | NA | NA |
| Triethanolamine | 0.00024 | 0.00791) | 0.00004772) | NA |
| Vapour pressure[Pa] | Method used for the determination 1) |
|---|---|
| 85.3 | Trouton’s rule + Clausius-Clapeyron [7] |
| 76.0 | Vetere + Clausius-Clapeyron [8] |
| 61.3 | Riedel + Clausius-Clapeyron [8] |
| 53.3 | Ciacolone + Clausius-Clapeyron [8] |
| 34.72) | Chastrette: experimental data, correlated by Antoine’s model [9] |
| 25.3 | Riedels model in absolute temperature [8] |
| 20.0 | P_PREDICT, general model |
| 16.0 | Vapour pressure measured at our laboratory using a gas saturation method3). |
| 13.3 | Limit between VOC and non VOC in the Danish MAL-code system (=0.1 mmHg) |
| 13.3 | P_PREDICT, hydrocarbon model |
| 13.3 | P_PREDICT, n-alkane model |
| 12.0 | Allemand et al.: experimental data measured between 25 and 45°C, extrapolated to 20°C [10] |
| 11.8 | TRC (1998) [4] |
| 11.7 | DIPPR experimental data, correlated by Riedel’s model (DIPPR, 1999) [3] |
| 10 | Limit between VOC and non-VOC in the EU VOC-directive (same limit as in the USA) |
| 9.3 | Riedel-Plank-Miller’s model [11] |
Existing Models
- The molar gas volume is assumed to be much larger than the molar liquid volume.
- The gas phase is assumed to be ideal.
- ∆hVap is assumed to be constant.
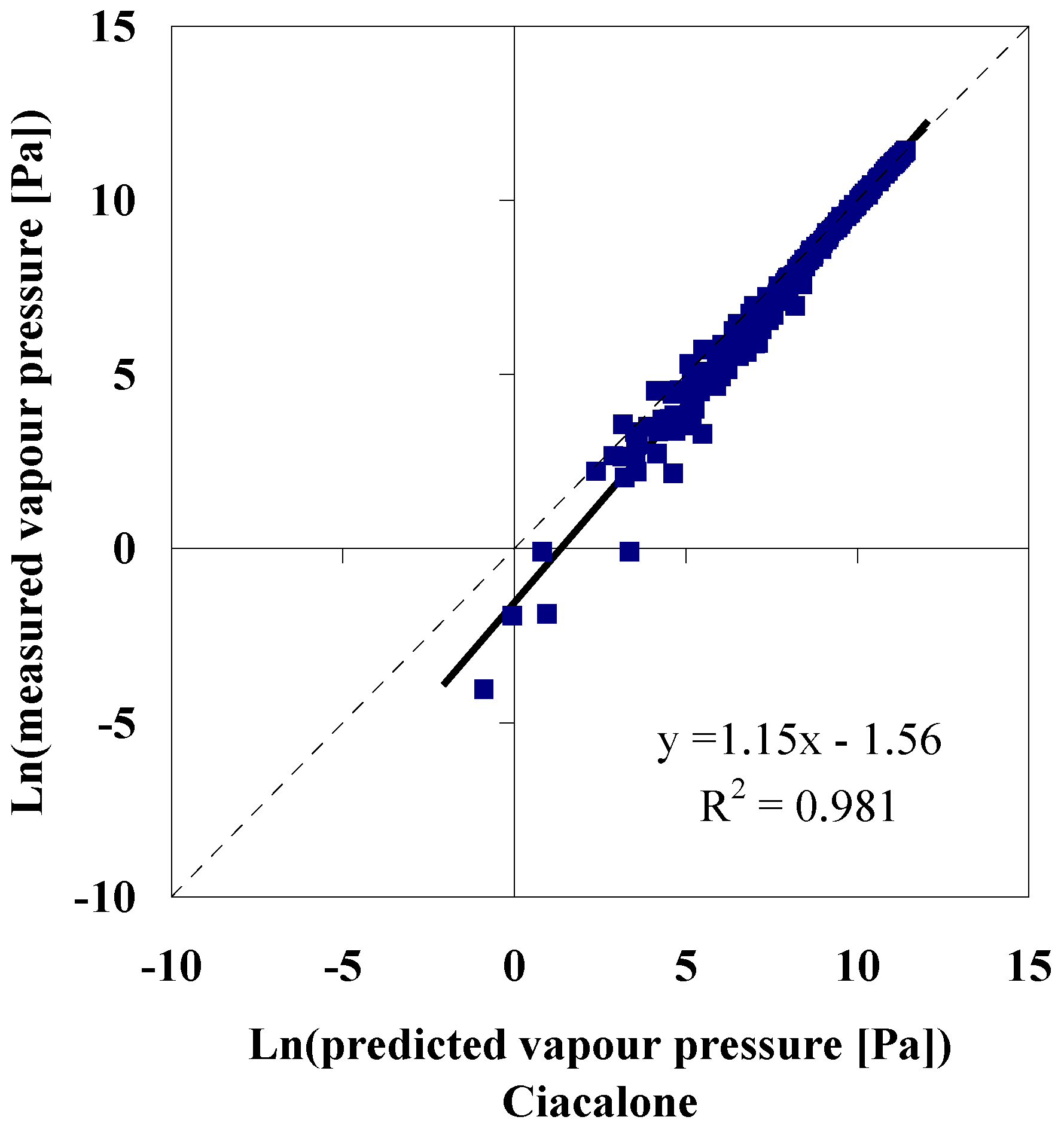
Knowing the Normal Boiling Temperature
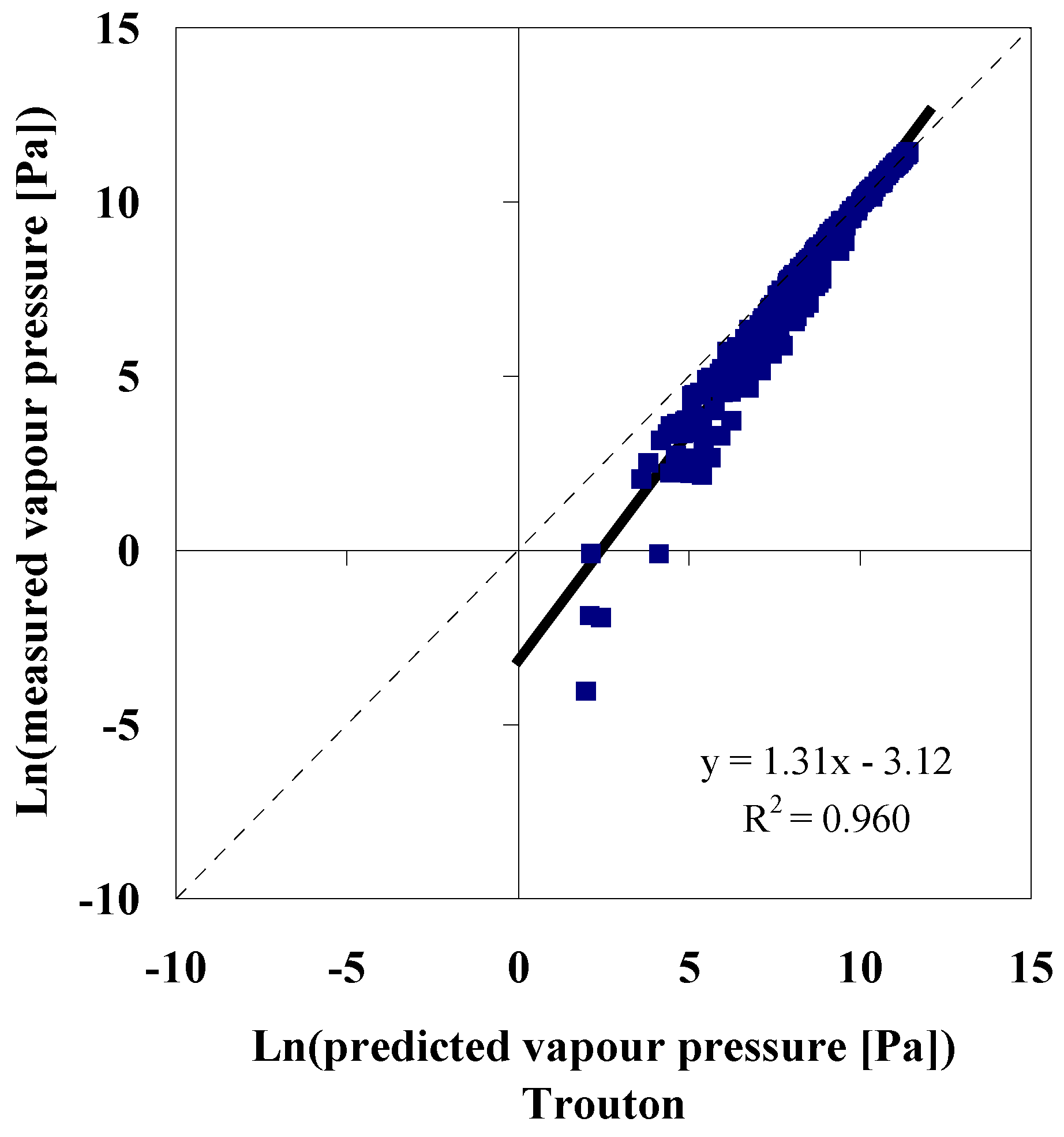
Knowing the Normal Boiling Temperature, Critical Pressure and Critical Temperature
Polynomial in Absolute Temperature
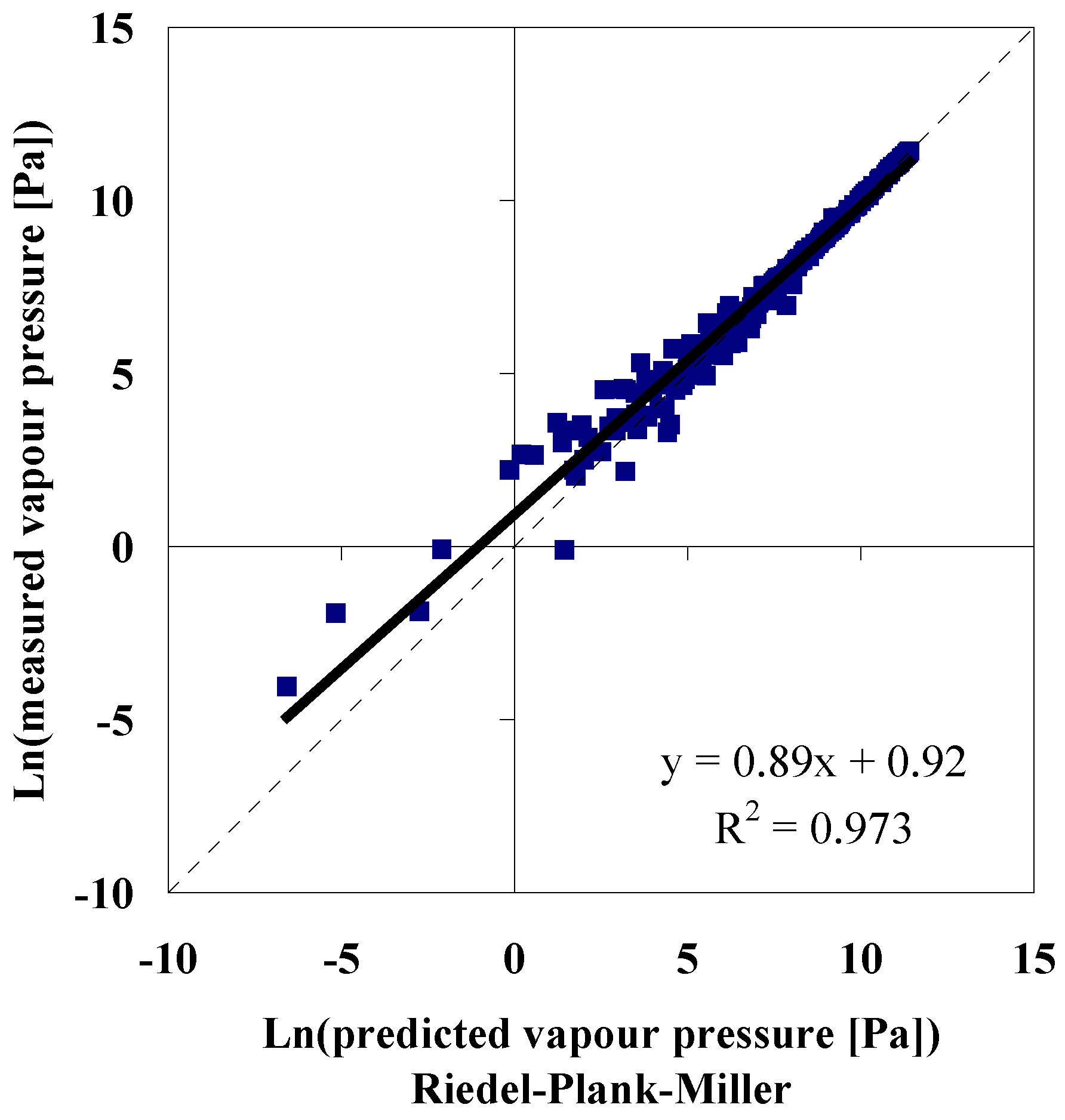
Accuracy of Model Predictions
Needs
Objectives
Methods
- Vapour pressure data should be available at ambient temperature, i.e. between -40 and 40°C.
- Data should be available from independent sources, which do not deviate more than 50%.
The Chemical (UNIFAC) Structure of Molecules
| Compound | 1st order groups | 2nd order groups | |||||||
|---|---|---|---|---|---|---|---|---|---|
| -CH3 | -CH2- | >CH- | -OH | > ACH 1) | -CH=CH | >CHOH | -CH2OH | CH2OHCHOH | |
| Hexane | 2 | 4 | - | - | - | - | - | - | - |
| 2-Pentene | 2 | 1 | - | - | - | 1 | - | - | - |
| Ethanol | 1 | 1 | - | 1 | - | - | - | 1 | - |
| Butanol | 1 | 3 | - | 1 | - | - | - | 1 | - |
| Benzene | - | - | - | - | 6 | - | - | - | - |
| Glycerol | - | 2 | 1 | 3 | - | - | 1 | 2 | 2 |
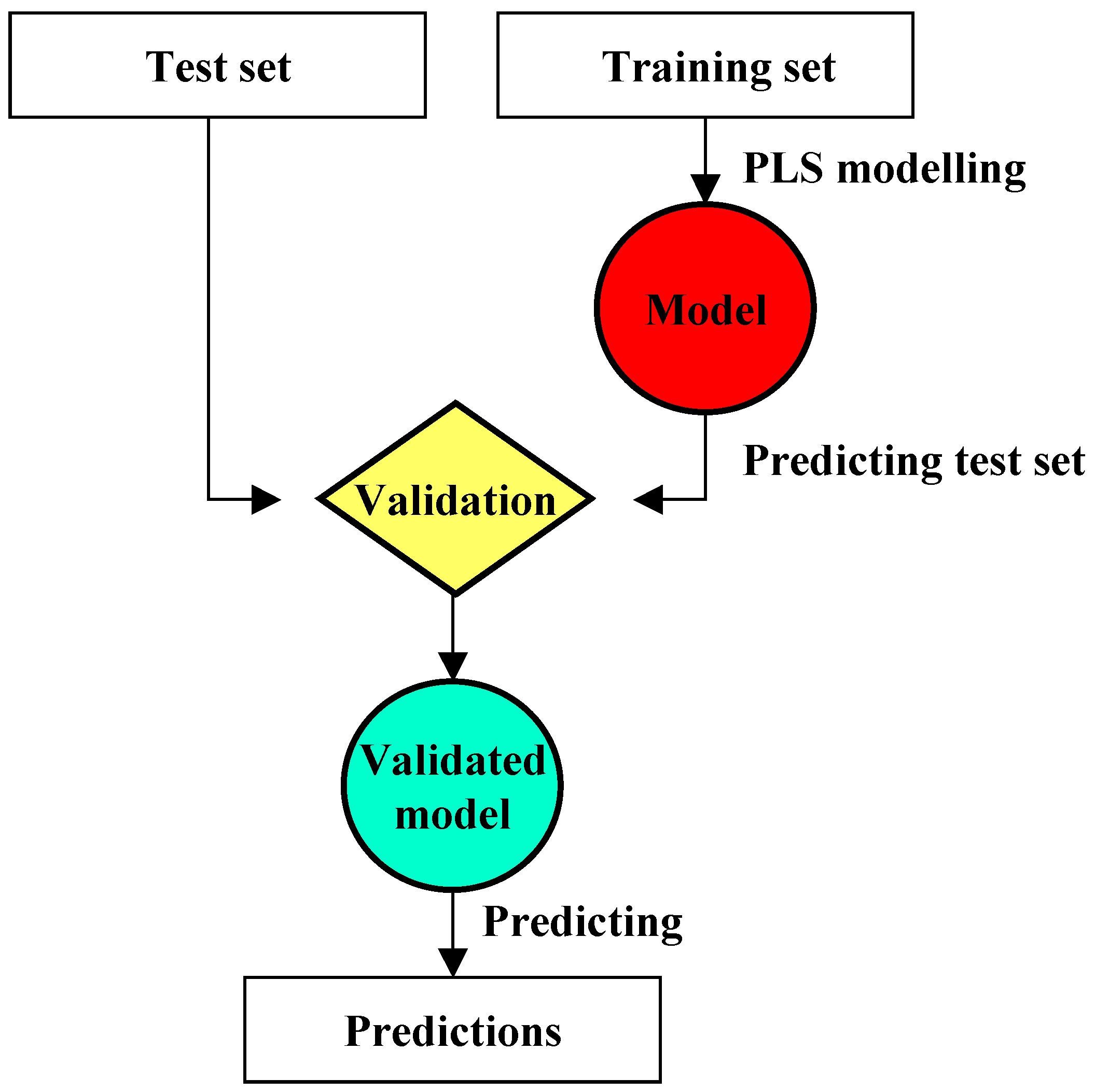
PLS
| Average bias | Standard Error of Performance | Root Mean Squared Error of Prediction |
|---|---|---|
Model validation
Results
P_PREDICT
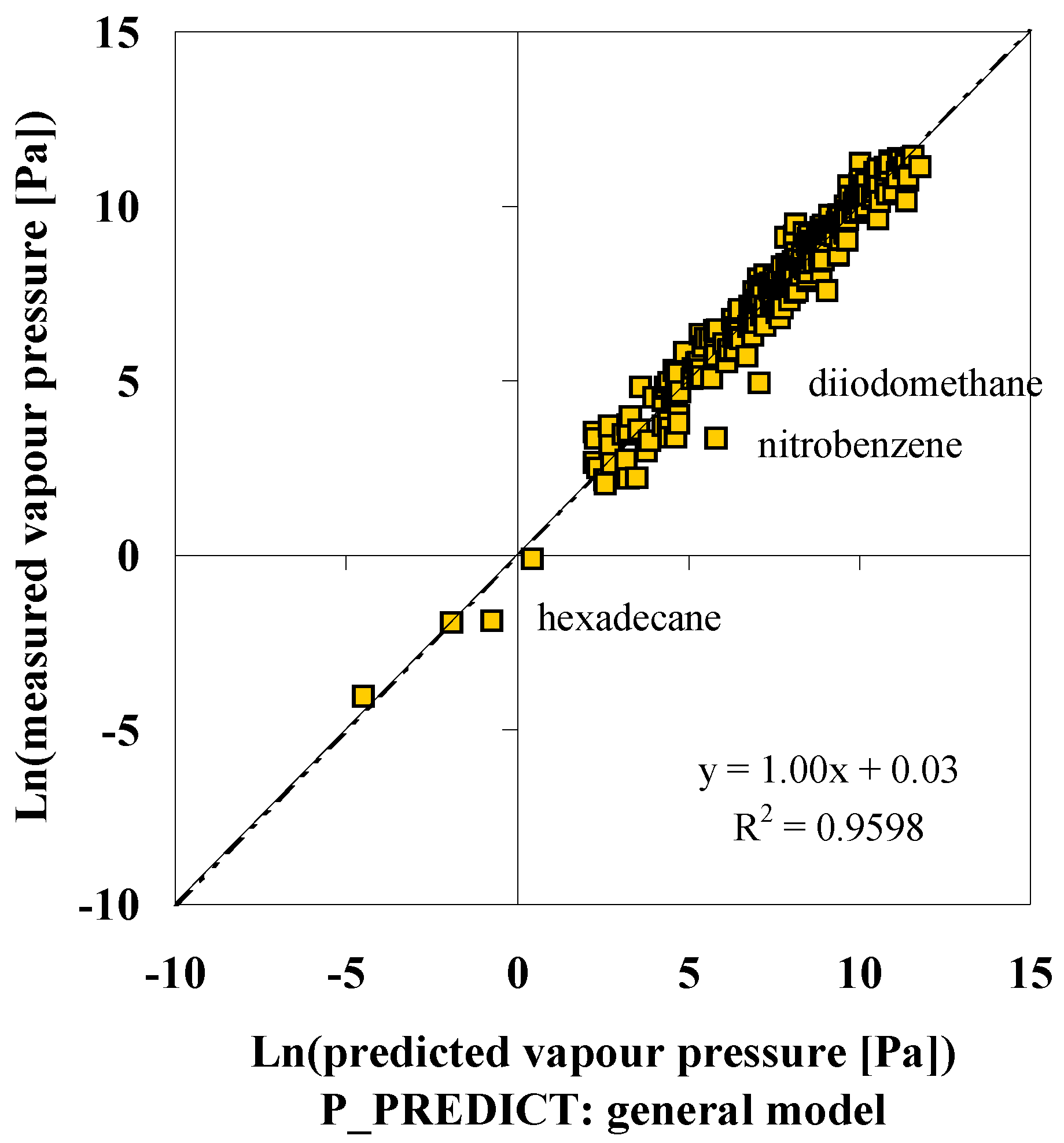
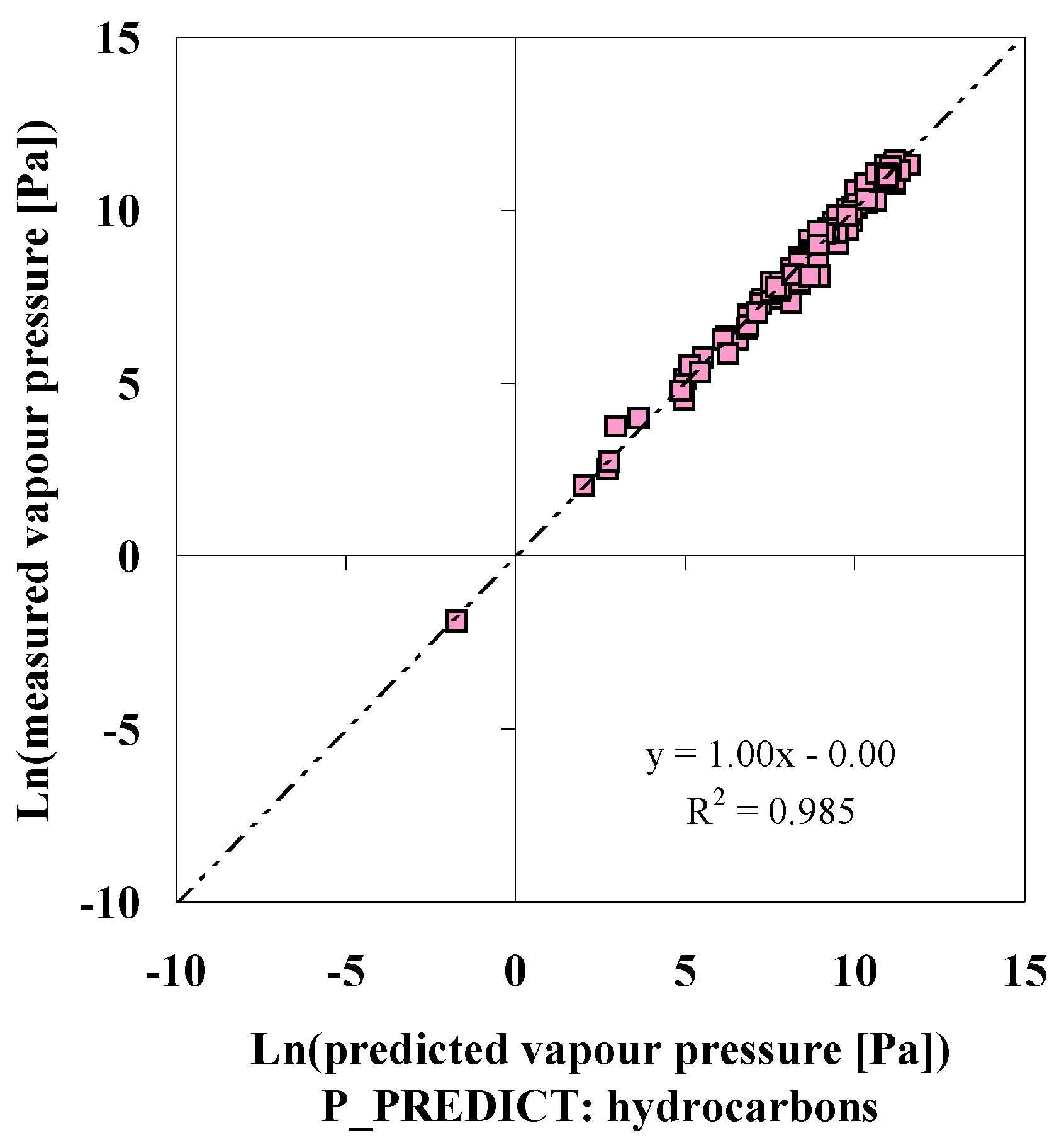
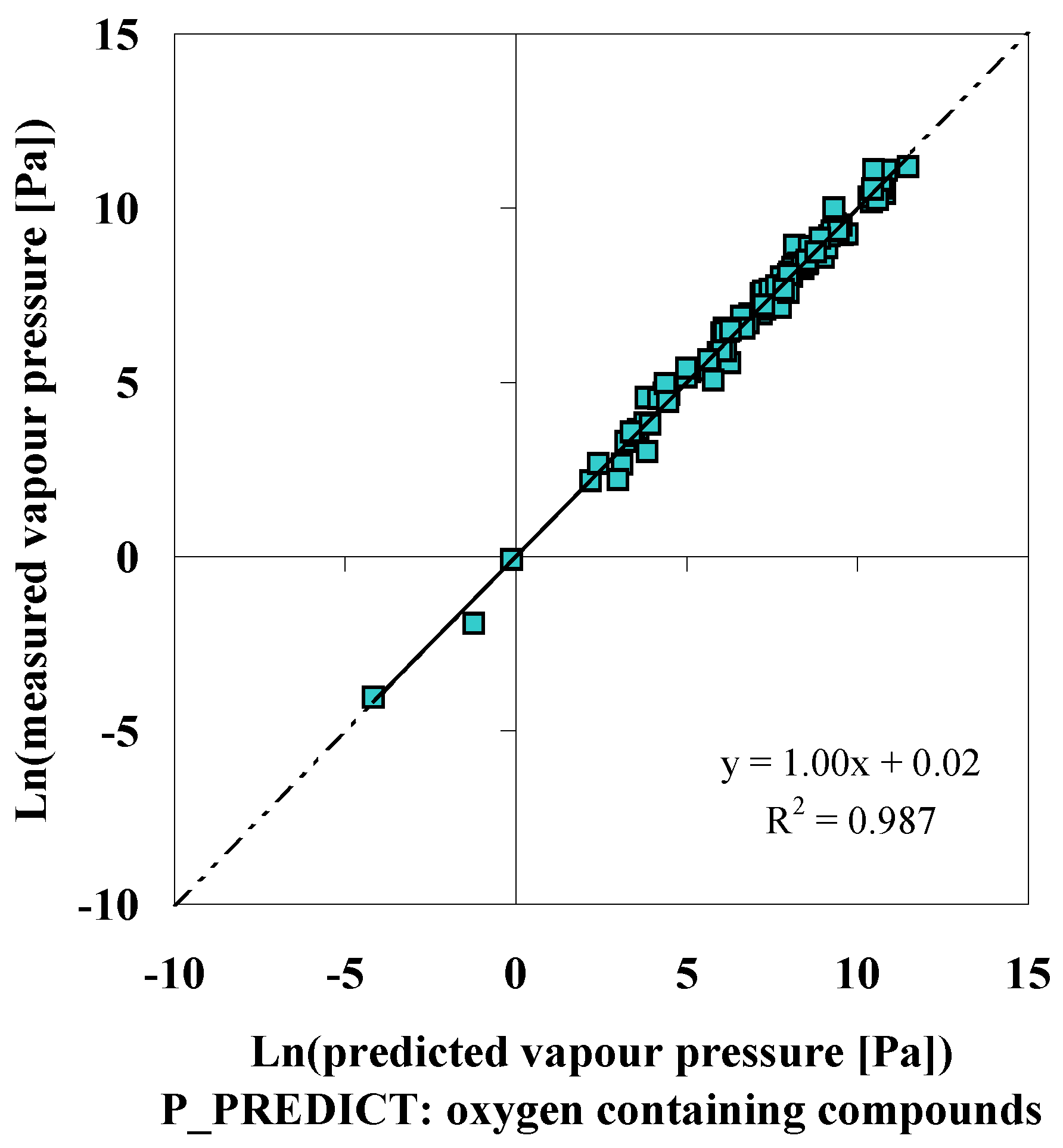
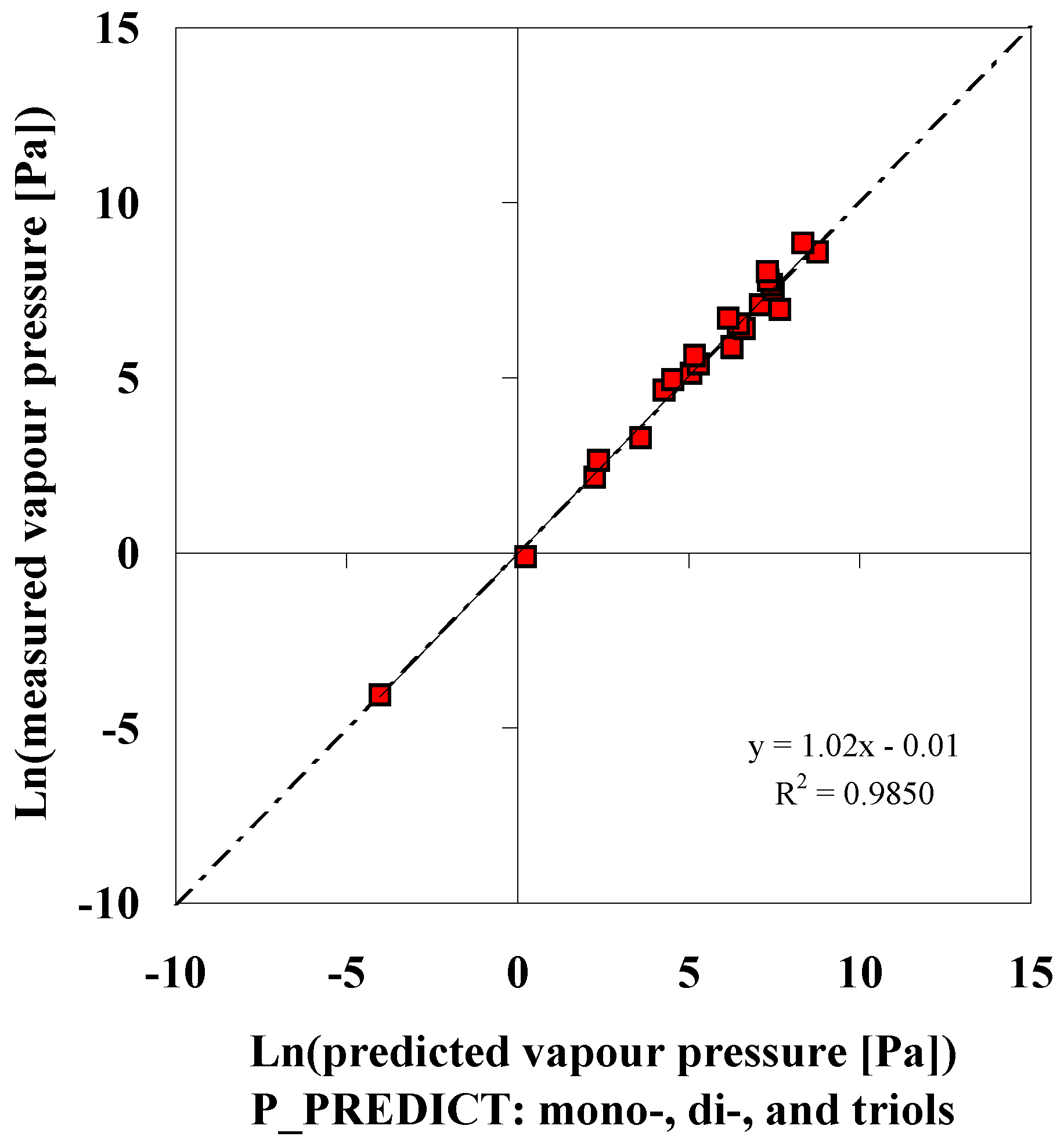
| Compound | p (DIPPR [3]) [Pa] | p (P_PREDICTgeneral model) [Pa] | ∆ [Pa] | ∆% [%] | p (IUCLID [5]) [Pa] |
|---|---|---|---|---|---|
| Acetone | 24711.5 | 24370.5 | -341.0 | -1.4 | 23300 |
| Hexane | 16231.7 | 11798.8 | -4432.9 | -27.3 | 16000 |
| Butanone | 10039.0 | 8385.8 | -1653.2 | -16.5 | 10100. 10600 |
| Benzene | 9985.0 | 8652.5 | -1332.5 | -13.3 | 9970 |
| Ethanol | 5901.3 | 6692.7 | 791.4 | 13.4 | 5730 , 5900 |
| Heptane | 4703.3 | 4052.9 | -650.4 | -13.8 | 4800 |
| Toluene | 2926.2 | 2613.1 | -313.1 | -10.7 | 2890 , 2930, 3000 |
| 1-Propanol | 1973.1 | 2306.4 | 333.3 | 16.9 | 1900 , 1940, 2000, 2030 |
| 2-Propanol | 4444.1 | 5772.8 | 1328.7 | 29.9 | 4200 , 4300 |
| Octane | 1404.1 | 1399.9 | -4.2 | -0.3 | 1500 |
| 1-Butanol | 648.5 | 792.1 | 143.6 | 22.1 | , 560, 670, 700, |
| 2-Butanol | 1735.8 | 1983.8 | 248.0 | 14.3 | 1596 |
| Ethylbenzene | 954.0 | 634.6 | -319.4 | -33.5 | 950 |
| p-Xylene | 872.1 | 790.7 | -81.4 | -9.3 | NA |
| m-Xylene | 834.1 | 790.7 | -43.4 | -5.2 | 660 |
| o-Xylene | 655.1 | 790.7 | 135.6 | 20.7 | 671) |
| Nonane | 420.6 | 480.0 | 59.4 | 14.1 | NA |
| Decane | 126.9 | 165.2 | 38.3 | 30.2 | 170, 173 |
| 1-Pentanol | 221.4 | 272.0 | 50.6 | 22.9 | 600 |
| 2-Pentanol | 568.1 | 682.6 | 114.5 | 20.2 | NA |
| 1-Hexanol | 72.4 | 93.7 | 21.3 | 29.4 | 71.7 |
| 1-Octanol | 6.4 | 11.1 | 4.7 | 73.4 | 3.1 |
| 2-Octanol | 20.1 | 27.7 | 7.6 | 37.8 | NA |
| 1-Decanol | 0.6 | 1.3 | 0.7 | 106.3 | NA |
| Methoxyacetic acid | 7.7 | 16.3 | 8.7 | 113.1 | NA |
| Glycerol | 0.0120 | 0.0093 | -0.0027 | -22.5 | NA |
| Triethanolamine | 0.0002 | 0.0007 | 0.0005 | 191.7 | 0.000004772) |
| Compound | p (DIPPR [3]) [Pa] | p (P_PREDICTgeneral model) [Pa] | ∆ [Pa] | ∆% [%] | p (IUCLID [5]) [Pa] |
|---|---|---|---|---|---|
| Hexadecane | 0.11 | 0.31) | 0.2 | 145.5 | NA |
| Dodecane | 11.66 | 19.6 | 7.9 | 68.1 | NA |
| Dicyclohexane | 9.60 | 8.5 | -1.1 | -11.8 | NA |
| 1-Methylnaphthalene | 6.75 | 10.8 | 4.0 | 59.9 | NA |
| 1,2,3,4-Tetrahydronaphthalene | 33.97 | 24.7 | -9.3 | -27.3 | 20, 24, 34 |
| cis-Decahydronaphthalene | 73.24 | 72.0 | -1.2 | -1.7 | NA |
| trans-Decahydronaphthalene | 115.37 | 71.3 | -44.1 | -38.2 | NA |
| n-Butylbenzene | 100.87 | 77.5 | -23.4 | -23.2 | NA |
| sec-Butylbenzene | 157.64 | 173.2 | 15.5 | 9.9 | 150 |
| tert-Butylbenzene | 208.10 | 142.7 | -65.5 | -31.5 | NA |
“…, or having a corresponding volatility under the particular condition of use” [1]
| Compound | p(20°C) [Pa] | p(30°C) [Pa] | p(30°C)c [Pa] | ∆ [Pa] | ∆% [%] | p(40 °C) [Pa] | p(40 °C)c [Pa] | ∆ [Pa] | ∆% [%] |
| Trichloromethane | 20997.10 | 32315.22 | 31373.4 | -941.8 | -2.9 | 48092.88 | 45690.7 | -2402.2 | -5.0 |
| Methanol | 12890.90 | 21781.29 | 19573.9 | -2207.4 | -10.1 | 35413.16 | 28939.2 | -6474.0 | -18.3 |
| Heptane | 4703.29 | 7745.54 | 7383.1 | -362.4 | -4.7 | 12278.99 | 11260.8 | -1018.2 | -8.3 |
| Dodecane | 11.66 | 26.90 | 22.3 | -4.6 | -17.0 | 58.07 | 41.0 | -17.1 | -29.5 |
| 2-Nonanol | 5.55 | 14.35 | 10.9 | -3.5 | -24.2 | 34.45 | 20.4 | -14.0 | -40.7 |
| Tridecane | 3.59 | 8.84 | 7.2 | -1.7 | -19.1 | 20.25 | 13.6 | -6.6 | -32.8 |
| 1-Nonanol | 1.65 | 5.01 | 3.4 | -1.6 | -32.8 | 13.45 | 6.6 | -6.9 | -51.2 |
| Heptacosane | 6.571E-7 | 3.369E-6 | 2.2E-6 | 1.5E-6 | -41.2 | 18.4E-6 | 6.7E-6 | -12E-6 | -63.6 |
Discussion
- has a vapour pressure above 10 Pa at 20°C, or
- having a corresponding volatility under the particular condition of use.
Organic compounds used at room temperature
Organic Compounds Used at Elevated Temperatures
Conclusions
Future work
Acknowledgements
References
- EU VOC-directive: Council Directive 1999/13/EC of 11 March 1999 on the limitation of emissions of volatile organic compounds due to the use of organic solvents in certain activities and installations. Official Journal of the European Communities: Brussels, 1999.
- Verbelen, R. Exxon Chemical, Europe. Private communication, 1998. [Google Scholar]
- DIPPR. Data Compilation of Pure Compound Properties. U.S. Department of Commerce. National Institute of Standard and Technology: Gaithersburg, Now available from Technical Database Services Inc. 135 West 50th Street. New York, 1999. www.tds-tds.com.
- TRC. Vapor Pressure Data File. The Texas Engineering Experiment. Texas A&M University System: Station. College Station, Available from Technical Database Services Inc. 135 West 50th Street. New York, 1998. www.tds-tds.com.
- IUCLID. International Uniform Chemical Information Database on CD-ROM. European Commission EUR 17283. European Chemicals Bureau Existing Chemicals. Joint Research Centre: Ispra (VA) Italy, 2000; http://ecb.ei.jrc.it.
- N’Guimbi, J.; Kasehgari, H.; Mokbel, I.; Jose, J. Tensions de vapeur d’alcohools primaires dans le domaine 0,3 Pa à 1,5 kPa (in French: Vapour Pressure of Primary Alcohols in the Range of 0.3 to 1.5 kPa). Thermochimica Acta. 1992, 196, 367–377. [Google Scholar]
- Horvarth, A.L. Molecular Design. Elsevier: London, 1992. [Google Scholar]
- Reid, R.C.; Prausnitz, J.M.; Poling, B.E. The Properties of Gases and Liquids. McGraw-Hill: New York, 1997. [Google Scholar]
- Chastrette, M.; Crétin, D. Structure-Property Relationship of the Vapour Pressure of Hydrocarbons and Oxygenated Compounds Using Multifunctional Autocorrelation Method (MAM). In SAR and QSAR in Environment Research Vol. 3: 131-149; Overseas Publishers Association: Amsterdam, 1995. [Google Scholar]
- Allemand, N.; Rose, J.; Merlin, J.C. Mesure des pressions de vapeur d’hydrocarbures C10 a C18 nalcanes et n-alkylbenzenes dans le domaine 3-1000 Pascal (in French: Vapour Pressure Measurements of C10 to C18 n-alkanes and n-alkylbenzenes in the Range of 3 – 1000 Pascal). Thermochimica Acta. 1986, 105, 79–90. [Google Scholar] [CrossRef]
- Reid, R.C.; Prausnitz, J.M.; Poling, B.E. The Properties of Gases and Liquids. McGraw-Hill: New York, 1981. [Google Scholar]
- Gmehling, J.; Kolbe. Thermodynamik. (In German: Thermodynamics); Georg Thieme Verlag: Stuttgart, 1988. [Google Scholar]
- Olsen, E.; Olsen, I.; Wallström, E.; Rasmussen, D. The SUBTEC Software Package. A Tool for Risk Assessment and Substitution. Occupational Hygiene 1997, 5, 1–21. [Google Scholar]
- Constantinou, L.; Gani, R. New Group Contribution Method for Estimating Properties of Pure Components. A.I.Ch. J. 1994, 40, 1697–1710. [Google Scholar]
- Jensen, T.; Fredenslund, Aa.; Rasmussen, P. Pure Component Vapor Pressures Using UNIFAC Group Contribution. I&EC Fundamentals 1981, 20, 239–246. [Google Scholar]
- Fredenslund, Aa..; Gmehling, J.; Rasmussen, P. Vapor-liquid equilibria using UNIFAC. Elsevier: Amsterdam, 1977. [Google Scholar]
- Martens, H.; Næs, T. Multivariate Calibration. John Wiley & Sons: Chichester, 1989. [Google Scholar]
- Unscrambler Version 7.5. Camo A/S: Trondheim, 2000.
- Esbensen, K; Schönkopf, S; Midtgaard, T. Multivariate analysis-in practice. Camo: Trondheim, 1996. [Google Scholar]
- Draft EU VOC-directive: Proposal for a Council Directive 96/0276 (SYN). Commission of the European Communities: Brussels, 1997.
- Pryde, E. Letter of 19 January 2000 from Health and Safety Executives, United Kingdom.
- Technical Guidance Document in support of commission directive 93/67/EEC on risk assessment for new notified substances and commission regulation (EC) No 1488/94 on risk assessment for existing substances. European Commission: Luxembourg, 1996.
| # | Group | # | Group | # | Group | # | Group |
| 1 | CH3 | 15 | OH | 29 | CH2N | 43 | ACF |
| 2 | CH2 | 16 | ACOH | 30 | CN | 44 | CF3 |
| 3 | CH | 17 | CH3CO | 31 | COOH | 45 | CH3S |
| 4 | C | 18 | CH2CO | 32 | CH2Cl | 46 | CH2S |
| 5 | CH2=CH | 19 | CHO | 33 | CHCl | 201 | CH2OH |
| 6 | CH2=C | 20 | CH3COO | 34 | CCl3 | 202 | CHOH |
| 7 | CH=CH | 21 | CH2COO | 35 | ACCl | 203 | CHm(OH)CHn(OH) |
| 8 | CH=C | 22 | COO | 36 | ACNO2 | 204 | CHm(OH)CHn(NH2) |
| 9 | C=C | 23 | HCOO | 37 | CH2SH | 205 | ACCHO |
| 10 | ACH | 24 | CH3O | 38 | I | 206 | ACCOO |
| 11 | AC | 25 | CH2O | 39 | Br | 207 | AC(OH)ACX |
| 12 | ACCH3 | 26 | CH2NH2 | 40 | C≡CH | 208 | AC(NH2) ACX |
| 13 | ACCH2 | 27 | ACNH2 | 41 | C≡C | 209 | AC(NO2) ACX |
| 14 | ACCH | 28 | CH2NH | 42 | Cl(C=C) |
- Sample Availability: Not applicable
© 2001 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes
Share and Cite
Olsen, E.; Nielsen, F. Predicting Vapour Pressures of Organic Compounds from Their Chemical Structure for Classification According to the VOCDirective and Risk Assessment in General. Molecules 2001, 6, 370-389. https://doi.org/10.3390/60400370
Olsen E, Nielsen F. Predicting Vapour Pressures of Organic Compounds from Their Chemical Structure for Classification According to the VOCDirective and Risk Assessment in General. Molecules. 2001; 6(4):370-389. https://doi.org/10.3390/60400370
Chicago/Turabian StyleOlsen, Erik, and Frands Nielsen. 2001. "Predicting Vapour Pressures of Organic Compounds from Their Chemical Structure for Classification According to the VOCDirective and Risk Assessment in General" Molecules 6, no. 4: 370-389. https://doi.org/10.3390/60400370
APA StyleOlsen, E., & Nielsen, F. (2001). Predicting Vapour Pressures of Organic Compounds from Their Chemical Structure for Classification According to the VOCDirective and Risk Assessment in General. Molecules, 6(4), 370-389. https://doi.org/10.3390/60400370




