Enhancing the Conjugation of Nickel(II) Schiff Bases for High-Contrast Electrochromism
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization of Nickel(II) Complexes of Schiff Bases
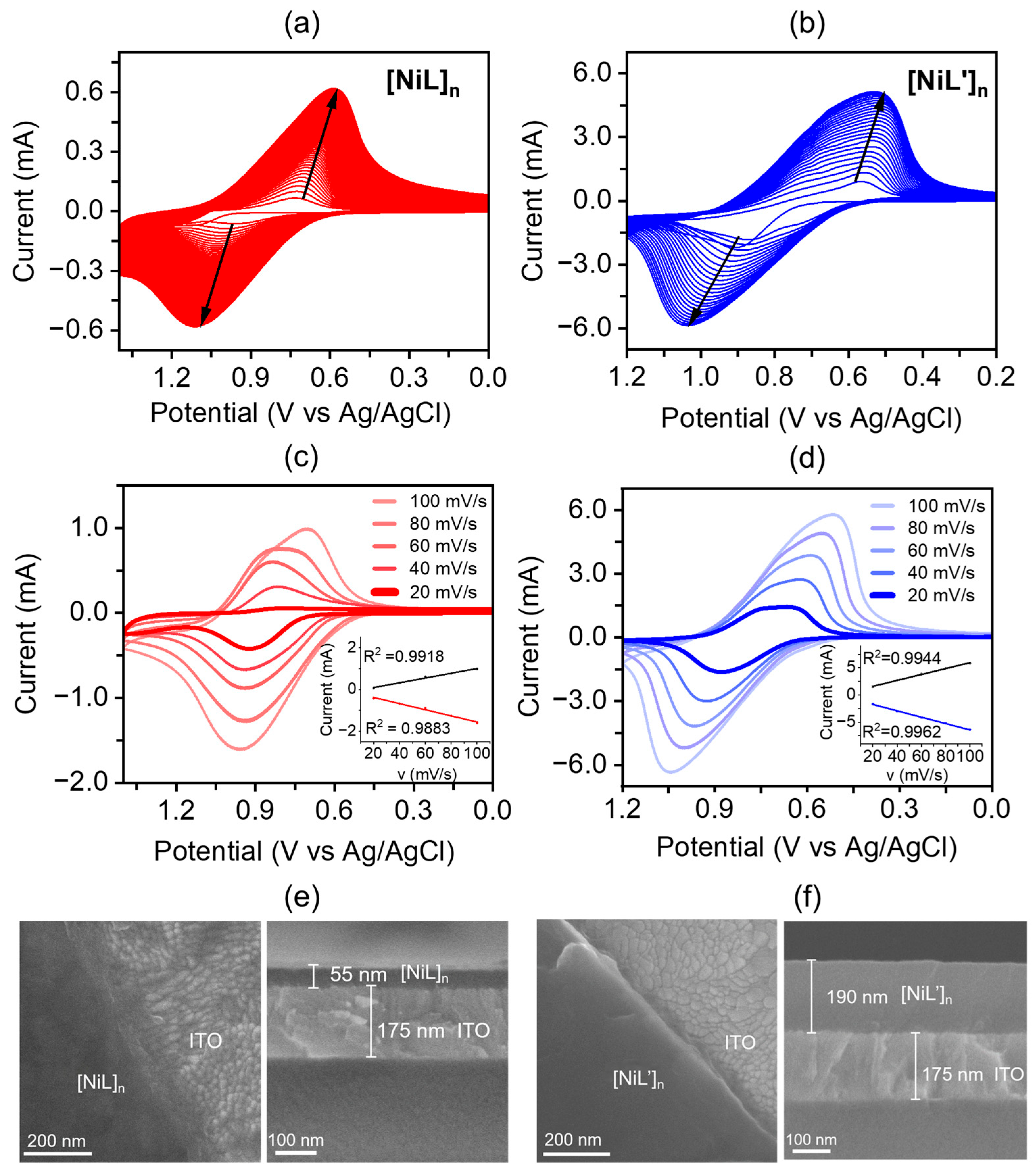
2.2. Preparation and Characterization of Metallopolymer Films
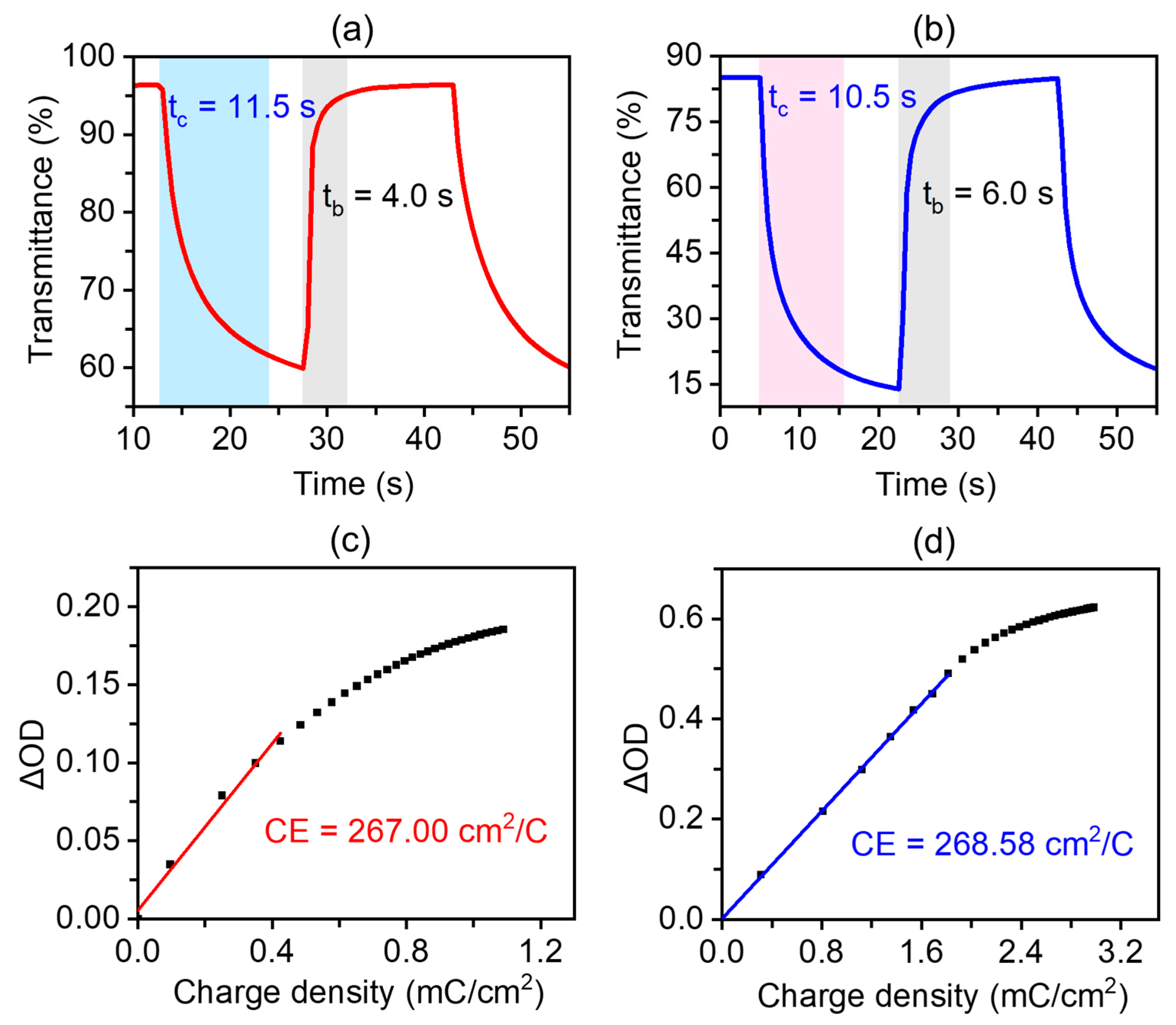
2.3. Electrochromic Properties of Metallopolymer Films
2.4. Solid-State Electrochromic Devices
3. Materials and Methods
3.1. General Information
3.2. Synthesis of Complex NiL
3.3. Synthesis of Complex NiL’
3.4. X-Ray Crystallography
3.5. Preparation of Metallopolymer Films
3.6. Characterization of [NiL]n and [NiL’]n Film
3.7. Fabrication of Solid-State Electrochromic Device
3.8. Characterization of Solid-State Electrochromic Device
3.9. Computational Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EC | electrochromism |
| ECD | electrochromic device |
| HOMO | highest occupied molecular orbital |
| LUMO | lowest unoccupied molecular orbital |
| MLCT | metal-to-ligand charge transfer |
| ITO | indium tin oxide |
| CV | cyclic voltammetry |
| CE | coloring efficiency |
| BE | bleaching efficiency |
References
- Jang, Y.J.; Kim, S.Y.; Kim, Y.M.; Lee, J.K.; Moon, H.C. Unveiling the diffusion-controlled operation mechanism of all-in-one type electrochromic supercapacitors: Overcoming slow dynamic response with ternary gel electrolytes. Energy Storage Mater. 2021, 43, 20–29. [Google Scholar] [CrossRef]
- Ke, Y.; Chen, J.; Lin, G.; Wang, S.; Zhou, Y.; Yin, J.; Lee, P.S.; Long, Y. Smart Windows: Electro-, Thermo-, Mechano-, Photochromics, and Beyond. Adv. Energy Mater. 2019, 9, 1902066. [Google Scholar] [CrossRef]
- Li, J.; Levitt, A.; Kurra, N.; Juan, K.; Noriega, N.; Xiao, X.; Wang, X.; Wang, H.; Alshareef, H.N.; Gogotsi, Y. MXene-conducting polymer electrochromic microsupercapacitors. Energy Storage Mater. 2019, 20, 455–461. [Google Scholar] [CrossRef]
- Poh, W.C.; Gong, X.; Yu, F.; Lee, P.S. Electropolymerized 1D Growth Coordination Polymer for Hybrid Electrochromic Aqueous Zinc Battery. Adv. Sci. 2021, 8, 2101944. [Google Scholar] [CrossRef] [PubMed]
- Rai, V.; Singh, R.S.; Blackwood, D.J.; Zhili, D. A Review on Recent Advances in Electrochromic Devices: A Material Approach. Adv. Eng. Mater. 2020, 22, 2000082. [Google Scholar] [CrossRef]
- Hee, W.J.; Alghoul, M.A.; Bakhtyar, B.; Elayeb, O.; Shameri, M.A.; Alrubaih, M.S.; Sopian, K. The role of window glazing on daylighting and energy saving in buildings. Renew. Sustain. Energy Rev. 2015, 42, 323–343. [Google Scholar] [CrossRef]
- Gorgolis, G.; Karamanis, D. Solar energy materials for glazing technologies. Sol. Energy Mater. Sol. Cells 2016, 144, 559–578. [Google Scholar] [CrossRef]
- Chou, H.-H.; Nguyen, A.; Chortos, A.; To, J.W.F.; Lu, C.; Mei, J.; Kurosawa, T.; Bae, W.-G.; Tok, J.B.H.; Bao, Z. A chameleon-inspired stretchable electronic skin with interactive colour changing controlled by tactile sensing. Nat. Commun. 2015, 6, 8011. [Google Scholar] [CrossRef]
- Argun, A.A.; Cirpan, A.; Reynolds, J.R. The First Truly All-Polymer Electrochromic Devices. Adv. Mater. 2003, 15, 1338–1341. [Google Scholar] [CrossRef]
- Mortimer, R.J. Electrochromic Materials. Annu. Rev. Mater. Res. 2011, 41, 241–268. [Google Scholar] [CrossRef]
- Oi, T. Electrochromic Materials. Annu. Rev. Mater. Res. 1986, 16, 185–201. [Google Scholar] [CrossRef]
- Wang, Y.; Runnerstrom, E.L.; Milliron, D.J. Switchable Materials for Smart Windows. Annu. Rev. Chem. Biomol. Eng. 2016, 7, 283–304. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, F.; Huang, H.; Wang, J.; Yu, H.A.; Jiang, J.; Yan, D.; Wang, Z. Near infrared electrochromism of lutetium phthalocyanine. Synth. Met. 2005, 148, 123–126. [Google Scholar] [CrossRef]
- Wu, Y.; Cong, B.; Yuan, J.; Chao, D.; Lv, Y.; Zhao, X.; Zhou, H.; Chen, C. Multicolored electrochromic metallopolymers based on triphenylamine-substituted terpyridine coordination with transition metal ions. Sol. Energy Mater. Sol. Cells 2025, 290, 113736. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, W.; Li, T.; Xia, Q.; Yang, S.; Weng, J.; Chen, K.; Chen, W.; Liu, M.; Du, S.; et al. Electrochromic Smart Window Based on Transition-Metal Phthalocyanine Derivatives. Inorg. Chem. 2024, 63, 3181–3190. [Google Scholar] [CrossRef] [PubMed]
- Beduoğlu, A.; Budak, Ö.; Sevim, A.M.; Koca, A.; Bayır, Z.A. Double-decker lutetium phthalocyanine functionalized with 4-phenylthiazol-2-thiol moieties: Synthesis, characterization, electrochemistry, spectroelectrochemistry and electrochromism. Polyhedron 2021, 209, 115479. [Google Scholar] [CrossRef]
- Banasz, R.; Kubicki, M.; Wałęsa-Chorab, M. Yellow-to-brown and yellow-to-green electrochromic devices based on complexes of transition metal ions with a triphenylamine-based ligand. Dalton Trans. 2020, 49, 15041–15053. [Google Scholar] [CrossRef]
- Sicard, L.; Navarathne, D.; Skalski, T.; Skene, W.G. On-Substrate Preparation of an Electroactive Conjugated Polyazomethine from Solution-Processable Monomers and its Application in Electrochromic Devices. Adv. Funct. Mater. 2013, 23, 3549–3559. [Google Scholar] [CrossRef]
- Nguyen, V.-Q.; Schaming, D.; Martin, P.; Lacroix, J.-C. Highly Resolved Nanostructured PEDOT on Large Areas by Nanosphere Lithography and Electrodeposition. ACS Appl. Mater. Interfaces 2015, 7, 21673–21681. [Google Scholar] [CrossRef]
- Stockhausen, V.; Ghilane, J.; Martin, P.; Trippé-Allard, G.; Randriamahazaka, H.; Lacroix, J.-C. Grafting Oligothiophenes on Surfaces by Diazonium Electroreduction: A Step toward Ultrathin Junction with Well-Defined Metal/Oligomer Interface. J. Am. Chem. Soc. 2009, 131, 14920–14927. [Google Scholar] [CrossRef]
- Stockhausen, V.; Trippé-Allard, G.; Van Quynh, N.; Ghilane, J.; Lacroix, J.-C. Grafting π-Conjugated Oligomers Incorporating 3,4-Ethylenedioxythiophene (EDOT) and Thiophene Units on Surfaces by Diazonium Electroreduction. J. Phys. Chem. C 2015, 119, 19218–19227. [Google Scholar] [CrossRef]
- Wałęsa-Chorab, M.; Skene, W.G. On-substrate polymerization—A versatile approach for preparing conjugated polymers suitable as electrochromes and for metal ion sensing. RSC Adv. 2014, 4, 19053–19060. [Google Scholar] [CrossRef]
- Ling, H.; Zhang, J.; Wang, Y.; Zeng, X. One-step achieving high performance all-solid-state and all-in-one flexible electrochromic supercapacitor by polymer dispersed electrochromic device strategy. J. Colloid Interface Sci. 2024, 665, 969–976. [Google Scholar] [CrossRef]
- Dai, J.; Zu, Y.; Yang, Y.; Yang, C.; Yu, Y.; Zhang, S.; Hou, J. Thermal Cross-Linking preparation of Aqueous-Based cathodic films for enhanced stability in organic electrochromic devices. Chem. Eng. J. 2024, 487, 150473. [Google Scholar] [CrossRef]
- Chen, F.; Ren, Y.; Guo, J.; Yan, F. Thermo- and electro-dual responsive poly(ionic liquid) electrolyte based smart windows. Chem. Commun. 2017, 53, 1595–1598. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Qiu, L.; Deng, Z.; Yan, F. Plastic reusable pH indicator strips: Preparation via anion-exchange of poly(ionic liquids) with anionic dyes. Polym. Chem. 2013, 4, 1309–1312. [Google Scholar] [CrossRef]
- Nguyen, V.Q.; Sun, X.; Lafolet, F.; Audibert, J.-F.; Miomandre, F.; Lemercier, G.; Loiseau, F.; Lacroix, J.-C. Unprecedented Self-Organized Monolayer of a Ru(II) Complex by Diazonium Electroreduction. J. Am. Chem. Soc. 2016, 138, 9381–9384. [Google Scholar] [CrossRef]
- Maldonado, S.; Smith, T.J.; Williams, R.D.; Morin, S.; Barton, E.; Stevenson, K.J. Surface Modification of Indium Tin Oxide via Electrochemical Reduction of Aryldiazonium Cations. Langmuir 2006, 22, 2884–2891. [Google Scholar] [CrossRef]
- Tao, Y.; Liu, H.; Kong, H.-Y.; Wang, T.-X.; Sun, H.; Li, Y.J.; Ding, X.; Sun, L.; Han, B.-H. Electrochemical Preparation of Porous Organic Polymer Films for High-Performance Memristors. Angew. Chem. Int. Ed. 2022, 61, e202205796. [Google Scholar] [CrossRef]
- Cihaner, A.; Algı, F. A Novel Neutral State Green Polymeric Electrochromic with Superior n- and p-Doping Processes: Closer to Red-Blue-Green (RGB) Display Realization. Adv. Funct. Mater. 2008, 18, 3583–3589. [Google Scholar] [CrossRef]
- Ak, M.; Ak, M.S.; Kurtay, G.; Güllü, M.; Toppare, L. Synthesis and electropolymerization of 1,2-bis(thiophen-3-ylmethoxy)benzene and its electrochromic properties and electrochromic device application. Solid State Sci. 2010, 12, 1199–1204. [Google Scholar] [CrossRef]
- Camurlu, P.; Şahmetlioğlu, E.; Şahin, E.; Akhmedov, İ.M.; Tanyeli, C.; Toppare, L. Fine tuning of color via copolymerization and its electrochromic device application. Thin Solid Film. 2008, 516, 4139–4144. [Google Scholar] [CrossRef]
- Seddiki, I.; Maris, T.; Skene, W.G. Violet Anthraquinone for Expanding the Color Palette of Electrochromes with Three Discrete Colors and Full Color Bleaching. Molecules 2026, 31, 879. [Google Scholar] [CrossRef]
- Nunes, M.; Araújo, M.; Fonseca, J.; Moura, C.; Hillman, R.; Freire, C. High-Performance Electrochromic Devices Based on Poly[Ni(salen)]-Type Polymer Films. ACS Appl. Mater. Interfaces 2016, 8, 14231–14243. [Google Scholar] [CrossRef]
- Chen, P.; Tan, R.; Nazir, M.; Li, J.; Song, W. Research Progress on the Preparation and Performance of Nickel Oxide Electrochromic Films. Nanoenergy Adv. 2026, 6, 10. [Google Scholar] [CrossRef]
- Grundhoefer, J.P.; Hardy, E.E.; West, M.M.; Curtiss, A.B.; Gorden, A.E.V. Mononuclear Cu(II) and Ni(II) complexes of bis(naphthalen-2-ol) Schiff base ligands. Inorg. Chim. Acta 2019, 484, 125–132. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.; Jin, W.; Xu, X.; Liu, H.; Ding, Y.; Wang, G.; Zhang, T.; Peng, Q.; He, J.; et al. π-π stacking controlled photochromic/thermochromic properties of salicylaldehyde Schiff base in solid state. Dye. Pigment. 2022, 202, 110295. [Google Scholar] [CrossRef]
- Liu, J.; Han, L.; Geng, J.; Hua, J.; Wang, Z. Metal-Ligand Coordination Induced Ionochromism for π-Conjugated Materials. Front. Chem. 2020, 8, 589106. [Google Scholar] [CrossRef]
- Seth, P.; Figuerola, A.; Jover, J.; Ruiz, E.; Ghosh, A. Antiferro- to ferromagnetic crossover in diphenoxido bridged NiII2MnII complexes derived from N2O2 donor Schiff base ligands. Polyhedron 2016, 117, 57–63. [Google Scholar] [CrossRef]
- Durmuş, S.; Ergun, Ü.; Jaud, J.C.; Emregül, K.C.; Fuess, H.; Atakol, O. Thermal decomposition of some linear trinuclear schiff base complexes with acetate bridges. J. Therm. Anal. Calorim. 2006, 86, 337–346. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, M.; Sun, F.; Sun, Q.; Cao, G.; Wu, X.; Ling, H.; Su, F.; Tian, Y.; Liu, Y.J.; et al. High-Efficiency and High-Capacity Aqueous Electrochromic Energy Storage Devices Enabled by Decoupled Titanium Oxide/Viologen Derivative Hybrid Materials. Research 2025, 8, 0909. [Google Scholar] [CrossRef] [PubMed]
- Meana-Esteban, B.; Kvarnström, C.; Geschke, B.; Heinze, J.; Ivaska, A. Electrochemical polymerization of 2-methoxynaphthalene. Synth. Met. 2003, 139, 133–143. [Google Scholar] [CrossRef]
- Liu, S.; Lu, Y.; Sun, S.; Wang, H.; Gao, W.; Wang, Y.; Jia, X.; Chen, J. Electrode material promoted dehydrogenative homo-/cross-coupling of weakly activated naphthalenes. Green Synth. Catal. 2023, 4, 71–75. [Google Scholar] [CrossRef]
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of silver and molybdenum microfocus X-ray sources for single-crystal structure determination. J. Appl. Crystallogr. 2015, 48, 3–10. [Google Scholar] [CrossRef]
- Hubschle, C.B.; Sheldrick, G.M.; Dittrich, B. ShelXle: A Qt Graphical User Interface for SHELXL. J. Appl. Crystallogr. 2011, 44, 1281–1284. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16; Revision A.03; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar]
- Casida, M.E.; Jamorski, C.; Casida, K.C.; Salahub, D.R. Molecular excitation energies to high-lying bound states from time-dependent density-functional response theory: Characterization and correction of the time-dependent local density approximation ionization threshold. J. Chem. Phys. 1998, 108, 4439–4449. [Google Scholar] [CrossRef]
- Stratmann, R.E.; Scuseria, G.E.; Frisch, M.J. An efficient implementation of time-dependent density-functional theory for the calculation of excitation energies of large molecules. J. Chem. Phys. 1998, 109, 8218–8224. [Google Scholar] [CrossRef]
- Hay, P.J.; Wadt, W.R. Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J. Chem. Phys. 1985, 82, 270–283. [Google Scholar] [CrossRef]
- Hay, P.J.; Wadt, W.R. Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J. Chem. Phys. 1985, 82, 299–310. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Lu, T.; Chen, Q.X. An sp-hybridized all-carboatomic ring, cyclo[18]carbon: Electronic structure, electronic spectrum, and optical nonlinearity. Carbon 2020, 165, 461–467. [Google Scholar] [CrossRef]
- Liu, L.; Wang, T.; He, Z.; Yi, Y.; Wang, M.; Luo, Z.; Liu, Q.; Huang, J.; Zhong, X.; Du, K.; et al. All-solid-state electrochromic Li-ion hybrid supercapacitors for intelligent and wide-temperature energy storage. Chem. Eng. J. 2021, 414, 128892. [Google Scholar] [CrossRef]
- Li, W.; Zhang, X.; Chen, X.; Zhao, Y.; Wang, L.; Chen, M.; Zhao, J.; Li, Y.; Zhang, Y. Effect of independently controllable electrolyte ion content on the performance of all-solid-state electrochromic devices. Chem. Eng. J. 2020, 398, 125628. [Google Scholar] [CrossRef]
- Song, K.; Cao, Z.; Weng, S.; Chen, W.; Jiang, R.; Rogachev, A.A.; Yarmolenko, M.A.; Zhou, J.; Zhang, H. Extraordinarily fast response all-solid-state electrochromic devices. Sol. Energy Mate. Sol. Cells 2024, 278, 113193. [Google Scholar] [CrossRef]
- Jensen, J.; Krebs, F.C. From the bottom up--flexible solid state electrochromic devices. Adv. mater. 2014, 26, 7231–7234. [Google Scholar] [CrossRef]
- Eren, E. Improved performance and stability of solid state electrochromic devices with eco-friendly chitosan-based electrolytes. Solid State Ionics 2019, 334, 152–159. [Google Scholar] [CrossRef]
- Zhang, S.; Zhao, Y.; Du, Z.; Luo, Y.; Chen, S.; Gu, Y.; Yan, B.; Yang, F.; Cao, Y. Solution-processable three-dimensional honeycomb-like poly(3,4-ethylenedioxythiophene) nanostructure networks with very fast response speed for patterned electrochromic devices. Sol. Energy Mater. Sol. Cells 2020, 207, 110354. [Google Scholar] [CrossRef]
- Pan, M.; Ke, Y.; Ma, L.; Zhao, S.; Wu, N.; Xiao, D. Single-layer electrochromic device based on hydroxyalkyl viologens with large contrast and high coloration efficiency. Electrochim. Acta 2018, 266, 395–403. [Google Scholar] [CrossRef]
- Li, H.; Zhang, W.; Elezzabi, A.Y. Transparent Zinc-Mesh Electrodes for Solar-Charging Electrochromic Windows. Adv. Mater. 2020, 32, 2003574. [Google Scholar] [CrossRef]
- Thai, L.H.; Thanh Nhi, L.T.; Hiep, N.M.; Khan, D.T.; Dat, T.N.; Truong Son, L.V.; Sinh, L.H. All-in-one high performance electrochromic device from new photocurable methacrylated viologen and polyaniline/UiO-66/NiO/ZnO composite. Mater. Today Chem. 2025, 43, 102511. [Google Scholar] [CrossRef]
- Li, M.-D.; Zhang, L.-Y.; Wang, J.-Y.; Han, J.-Y.; Qian, X.-Y.; Ye, X.-L.; Dai, F.-R.; Chen, Z.-N. Metallopolymer Films of a Manganese(II) Chelate with Hexadentate Ligand for High-Performance Electrochromism. Angew. Chem. Int. Ed. 2026, 65, e25500. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Dalchiele, E.A.; Martín, F.; Leinen, D.; Ramos-Barrado, J.R. Electrochromic behaviour of Nb2O5 thin films with different morphologies obtained by spray pyrolysis. Sol. Energy Mater. Sol. Cells 2009, 93, 222–229. [Google Scholar] [CrossRef]
- Zhi, M.; Huang, W.; Shi, Q.; Peng, B.; Ran, K. Enhanced Electrochromic Performance of Vanadium Pentoxide/Reduced Graphene Oxide Nanocomposite Film Prepared by the Sol–Gel Method. J. Electrochem. Soc. 2016, 163, H891. [Google Scholar] [CrossRef]
- Yin, Y.; Li, W.; Zeng, X.; Xu, P.; Murtaza, I.; Guo, Y.; Liu, Y.; Li, T.; Cao, J.; He, Y.; et al. Design Strategy for Efficient Solution-Processable Red Electrochromic Polymers Based on Unconventional 3,6-Bis(dodecyloxy)thieno[3,2-b]thiophene Building Blocks. Macromolecules 2018, 51, 7853–7862. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, X.; Jia, X.; Chao, D. Dual-electrochromic polymer bearing oligoaniline and viologen pendants: Synthesis and properties. Eur. Polym. J. 2019, 111, 43–48. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, J.-X.; Zhang, L.-Y.; Wang, J.-Y.; Dai, F.-R.; Chen, Z.-N. Enhancing the Conjugation of Nickel(II) Schiff Bases for High-Contrast Electrochromism. Molecules 2026, 31, 1433. https://doi.org/10.3390/molecules31091433
Li J-X, Zhang L-Y, Wang J-Y, Dai F-R, Chen Z-N. Enhancing the Conjugation of Nickel(II) Schiff Bases for High-Contrast Electrochromism. Molecules. 2026; 31(9):1433. https://doi.org/10.3390/molecules31091433
Chicago/Turabian StyleLi, Jia-Xin, Li-Yi Zhang, Jin-Yun Wang, Feng-Rong Dai, and Zhong-Ning Chen. 2026. "Enhancing the Conjugation of Nickel(II) Schiff Bases for High-Contrast Electrochromism" Molecules 31, no. 9: 1433. https://doi.org/10.3390/molecules31091433
APA StyleLi, J.-X., Zhang, L.-Y., Wang, J.-Y., Dai, F.-R., & Chen, Z.-N. (2026). Enhancing the Conjugation of Nickel(II) Schiff Bases for High-Contrast Electrochromism. Molecules, 31(9), 1433. https://doi.org/10.3390/molecules31091433







