Nanoparticle-Catalysed Microwave-Driven MCRs for Sustainable Heterocycle Synthesis
Abstract
1. Introduction
1.1. Role of Nanoparticle Catalysts in Sustainable MCRs
1.2. Microwave Irradiation as a Facilitating Eco-Friendly Method
1.3. Nanoparticle-Catalysed Microwave-Assisted MCRs for Heterocycle Synthesis
1.4. Purpose and Scope of the Current Review
2. Synthesis of Nanoparticles: Approaches and Sustainable Methods
3. Understanding the Catalytic Function of Nanoparticles
4. Microwave–Nanoparticle Interaction at the Molecular Level
5. Recent Advances in Microwave-Assisted MCRs Using Nanoparticle Catalysts
5.1. Synthesis of Pyridine and Pyrimidine Derivatives
5.2. Microwave-Assisted Synthesis of Imidazole and Indazole Derivatives
5.3. Microwave-Assisted Synthesis of Pyran and Chromene Derivatives
5.4. Microwave-Assisted Synthesis of Propargylamine Derivatives
5.5. Microwave-Assisted Synthesis of Phenazine Derivatives
5.6. Microwave-Assisted Synthesis of Quinazolinone and Quinoxaline Derivatives
5.7. Microwave-Assisted Synthesis of Xanthene Derivatives
5.8. Microwave-Assisted Synthesis of Acridine Derivatives
5.9. Microwave-Assisted Synthesis of Azlactone Derivatives
5.10. Microwave-Assisted Synthesis of Thiophene Derivatives
5.11. Microwave-Assisted Synthesis of Benzoxazine Derivatives
5.12. Microwave-Assisted Synthesis of Benzodiazepine Derivatives
5.13. Microwave-Assisted Synthesis of Tetrazole Derivatives
5.14. Microwave-Assisted Synthesis of Pyranopyrazole Derivatives
5.15. Microwave-Assisted Synthesis of Indole Derivatives
5.16. Microwave-Assisted Synthesis of Benzoxazole Derivatives
5.17. Microwave-Assisted Synthesis of Triazole Derivatives
6. Mechanistic Actions in MW–Nanoparticle-Catalysed MCRs
7. Green Metrics and Sustainability Analysis of MCRs Catalysed by NPs Under MW Irradiation
8. Challenges and Future Perspectives
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eckert, H. Diversity oriented syntheses of conventional heterocycles by smart multi component reactions (MCRs) of the last decade. Molecules 2012, 17, 1074–1102. [Google Scholar] [CrossRef] [PubMed]
- Hoult, J.R.S.; Payá, M. Pharmacological and biochemical actions of simple coumarins: Natural products with therapeutic potential. Gen. Pharmacol. Vasc. Syst. 1996, 27, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Ugi, I.; Dömling, A.; Hörl, W. Multicomponent reactions in organic chemistry. Endeavour 1994, 18, 115–122. [Google Scholar] [CrossRef]
- Lambat, T.L.; Chaudhary, R.G.; Abdala, A.A.; Mishra, R.K.; Mahmood, S.H.; Banerjee, S. Mesoporous PbO nanoparticle-catalyzed synthesis of arylbenzodioxy xanthenedione scaffolds under solvent-free conditions in a ball mill. RSC Adv. 2019, 9, 31683–31690. [Google Scholar] [CrossRef]
- Fang, G.; Bi, X. Silver-catalysed reactions of alkynes: Recent advances. Chem. Soc. Rev. 2015, 44, 8124–8173. [Google Scholar] [CrossRef]
- Saha, A.; Payra, S.; Banerjee, S. One-pot multicomponent synthesis of highly functionalized bio-active pyrano[2,3-c]pyrazole and benzylpyrazolyl coumarin derivatives using ZrO2 nanoparticles as a reusable catalyst. Green Chem. 2015, 17, 2859–2866. [Google Scholar] [CrossRef]
- Koley, S.; Chowdhury, S.; Chanda, T.; Ramulu, B.J.; Singh, M.S. Diversity oriented catalyst-free and solvent-free one-pot MCR at room temperature: Rapid and regioselective convergent approach to highly functionalized dihydro-4H-thiopyrans. Tetrahedron 2013, 69, 8013–8018. [Google Scholar] [CrossRef]
- Nandi, S.; Jamatia, R.; Sarkar, R.; Sarkar, F.K.; Alam, S.; Pal, A.K. One-Pot Multicomponent Reaction: A Highly Versatile Strategy for the Construction of Valuable Nitrogen-Containing Heterocycles. ChemistrySelect 2022, 7, e202201901. [Google Scholar] [CrossRef]
- Zhao, W.; Chen, F.E. One-pot synthesis and its practical application in pharmaceutical industry. Curr. Org. Synth. 2012, 9, 873–897. [Google Scholar] [CrossRef]
- Astruc, D.; Lu, F.; Aranzaes, J.R. Nanoparticles as recyclable catalysts: The frontier between homogeneous and heterogeneous catalysis. Angew. Chem. Int. Ed. 2005, 44, 7852–7872. [Google Scholar] [CrossRef]
- Clark, J.H.; Macquarrie, D.J. Handbook of Green Chemistry and Technology; Blackwell Science: Hoboken, NJ, USA, 2002. [Google Scholar]
- Polshettiwar, V.; Varma, R.S. Green chemistry by nano-catalysis. Green Chem. 2010, 12, 743–754. [Google Scholar] [CrossRef]
- Ramón, D.J.; Yus, M. Asymmetric multicomponent reactions (AMCRs): The new frontier. Angew. Chem. Int. Ed. 2005, 44, 1602–1634. [Google Scholar] [CrossRef] [PubMed]
- Rotstein, B.H.; Zaretsky, S.; Rai, V.; Yudin, A.K. Small heterocycles in multicomponent reactions. Chem. Rev. 2014, 114, 8323–8359. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A. Metrics of green chemistry and sustainability: Past, present, and future. ACS Sustain. Chem. Eng. 2018, 6, 32–48. [Google Scholar] [CrossRef]
- Polshettiwar, V.; Luque, R.; Fihri, A.; Zhu, H.; Bouhrara, M.; Basset, J.M. Magnetically recoverable nanocatalysts. Chem. Rev. 2011, 111, 3036–3075. [Google Scholar] [CrossRef]
- Hootifard, G.; Sheikhhosseini, E.; Ahmadi, S.A.; Yahyazadehfar, M. Fe3O4@iron-based metal–organic framework nanocomposite [Fe3O4@MOF (Fe) NC] as a recyclable magnetic nano-organocatalyst for the environment-friendly synthesis of pyrano[2,3-d]pyrimidine derivatives. Front. Chem. 2023, 11, 1193080. [Google Scholar] [CrossRef]
- Kasi, V.; EISayed Abdelsalam Zaki, M.; Nabisahebgari, H.B.; Shaik, H.; Chang, S.K.; Wong, L.S.; Parasuraman, K.; Gomha, S.M. Magnetic nanoparticle-catalysed one-pot multicomponent reactions (MCRs): A green chemistry approach. Catalysts 2025, 15, 800. [Google Scholar] [CrossRef]
- Varma, R.S. Journey on greener pathways: From the use of alternate energy inputs and benign reaction media to sustainable applications of nano-catalysts in synthesis and environmental remediation. Green Chem. 2014, 16, 2027–2041. [Google Scholar] [CrossRef]
- Varma, R.S. Greener and sustainable trends in synthesis of organics and nanomaterials. ACS Sustain. Chem. Eng. 2016, 4, 5866–5878. [Google Scholar] [CrossRef]
- Gao, H.Y. Recent advances in organic molecule reactions on metal surfaces. Phys. Chem. Chem. Phys. 2024, 26, 19052–19068. [Google Scholar] [CrossRef]
- Rai, P.; Gupta, D. Magnetic nanoparticles as green catalysts in organic synthesis-a review. Synth. Commun. 2021, 51, 3059–3083. [Google Scholar] [CrossRef]
- Wang, D.; Wen, L.; Liu, P.; Li, P.; Xi, J. Boosting catalytic performance of ternary FeCoNi yolk-shelled nanoreactors for organic reactions via low-content active metal decoration. Sci. China Technol. Sci. 2025, 68, 1720203. [Google Scholar] [CrossRef]
- Li, J.; Feng, Y.; Li, X.; Zhang, T.; Liu, X.; Wang, N.; Sun, Q. Sub-2 nm ternary metallic alloy encapsulated within montmorillonite interlayers for efficient hydrogen generation from ammonia borane hydrolysis. ACS Catal. 2024, 14, 14665–14677. [Google Scholar] [CrossRef]
- Gawande, M.B.; Shelke, S.N.; Zboril, R.; Varma, R.S. Microwave-assisted chemistry: Synthetic applications for rapid assembly of nanomaterials and organics. Acc. Chem. Res. 2014, 47, 1338–1348. [Google Scholar] [CrossRef]
- Zhu, J.; Bienaymé, H. (Eds.) Multicomponent Reactions; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Zhu, J.; Wang, Q.; Wang, M. (Eds.) Multicomponent Reactions in Organic Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Damera, T.; Pagadala, R.; Rana, S.; Jonnalagadda, S.B. A concise review of multicomponent reactions using novel heterogeneous catalysts under microwave irradiation. Catalysts 2023, 13, 1034. [Google Scholar] [CrossRef]
- Adhikari, A.; Bhakta, S.; Ghosh, T. Microwave-assisted synthesis of bioactive heterocycles: An overview. Tetrahedron 2022, 126, 133085. [Google Scholar] [CrossRef]
- Dubasi, N.; Varala, R.; Kamsali, M.M.A.; Alam, M.M. Visible Light-Driven Multicomponent Reactions for the Synthesis of Diverse Heterocyclic Frameworks. Eur. J. Org. Chem. 2025, 28, e2500216. [Google Scholar] [CrossRef]
- Gulati, S.; John, S.E.; Shankaraiah, N. Microwave-assisted multicomponent reactions in heterocyclic chemistry and mechanistic aspects. Beilstein J. Org. Chem. 2021, 17, 819–865. [Google Scholar] [CrossRef]
- Leadbeater, N.E. Microwave Heating as a Tool for Sustainable Chemistry; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar] [CrossRef]
- Tandi, M.; Sharma, V.; Gopal, B.; Sundriyal, S. Multicomponent reactions (MCRs) yielding medicinally relevant rings: A recent update and chemical space analysis of the scaffolds. RSC Adv. 2025, 15, 1447–1489. [Google Scholar] [CrossRef]
- Mukherjee, R.; Saha, I.; Mitra, P.; Majhi, S. Nanoparticle-Assisted Green Synthesis of N, N-Heterocycles with Anti-Breast Cancer Activity: A Decade of Advances. ChemistrySelect 2025, 10, e00767. [Google Scholar] [CrossRef]
- Yadav, M.; Dutta, M.; Tanwar, P.; Jain, R.; Srivastava, A.; Sharma, R.K. Microwave-assisted CC, CO, CN, CS bond formation and multicomponent reactions using magnetic retrievable nanocatalysts. Curr. Microw. Chem. 2021, 8, 96–116. [Google Scholar] [CrossRef]
- Rahman, M.; Ghosh, S.; Bhattacherjee, D.; Zyryanov, G.V.; Kumar Bagdi, A.; Hajra, A. Recent Advances in Microwave-assisted Cross-Coupling Reactions. Asian J. Org. Chem. 2022, 11, e202200179. [Google Scholar] [CrossRef]
- Sah, M.K.; Ettarhouni, Z.O.; Pathak, R.; Gawad, J.; Bonde, C.; Arya, S.P.; Bhattarai, A. Green Chemistry: Strategies and Sustainable Approaches for Bridging UN SDGS. ChemistrySelect 2025, 10, e00847. [Google Scholar] [CrossRef]
- Kümmerer, K. Sustainable chemistry: A future guiding principle. Angew. Chem. Int. Ed. 2017, 56, 16420–16421. [Google Scholar] [CrossRef] [PubMed]
- Matlin, S.A.; Mehta, G.; Cornell, S.E.; Krief, A.; Hopf, H. Chemistry and pathways to net zero for sustainability. RSC Sustain. 2023, 1, 1704–1721. [Google Scholar] [CrossRef]
- Nathanael, A.J.; Kannaiyan, K.; Kunhiraman, A.K.; Ramakrishna, S.; Kumaravel, V. Global opportunities and challenges on net-zero CO2 emissions towards a sustainable future. React. Chem. Eng. 2021, 6, 2226–2247. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Arends, I.; Hanefeld, U. Green Chemistry and Catalysis; John Wiley & Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- Jadoun, S.; Arif, R.; Jangid, N.K.; Meena, R.K. Green synthesis of nanoparticles using plant extracts: A review. Environ. Chem. Lett. 2021, 19, 355–374. [Google Scholar] [CrossRef]
- Iravani, S. Green synthesis of metal nanoparticles using plants. Green Chem. 2011, 13, 2638–2650. [Google Scholar] [CrossRef]
- Liang, Y.P.; Chan, Y.B.; Aminuzzaman, M.; Shahinuzzaman, M.; Djearamane, S.; Thiagarajah, K.; Leong, S.Y.; Wong, L.S.; Tey, L.H. Green Synthesis and Characterization of Copper Oxide Nanoparticles from Durian (Durio zibethinus) Husk for Environmental Applications. Catalysts 2025, 15, 275. [Google Scholar] [CrossRef]
- Hano, C.; Abbasi, B.H. Plant-based green synthesis of nanoparticles: Production, characterization and applications. Biomolecules 2021, 12, 31. [Google Scholar] [CrossRef]
- Bahrulolum, H.; Nooraei, S.; Javanshir, N.; Tarrahimofrad, H.; Mirbagheri, V.S.; Easton, A.J.; Ahmadian, G. Green synthesis of metal nanoparticles using microorganisms and their application in the agrifood sector. J. Nanobiotechnology 2021, 19, 86. [Google Scholar] [CrossRef]
- Tahir, K.; Nazir, S.; Ahmad, A.; Li, B.; Khan, A.U.; Khan, Z.U.H.; Khan, F.U.; Khan, Q.U.; Khan, A.; Rahman, A.U. Facile and green synthesis of phytochemicals capped platinum nanoparticles and in vitro their superior antibacterial activity. J. Photochem. Photobiol. B Biol. 2017, 166, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Zuhrotun, A.; Oktaviani, D.J.; Hasanah, A.N. Biosynthesis of gold and silver nanoparticles using phytochemical compounds. Molecules 2023, 28, 3240. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Kumar, R.; Alhan, S.; Sharma, H.; Singh, N.; Yogi, R.; Chhokar, V.; Beniwal, V.; Ghosh, M.K.; Chandraker, S.K.; et al. Lycium shawii mediated green synthesis of silver nanoparticles, characterization and assessments of their phytochemical, antioxidant, antimicrobial properties. Inorg. Chem. Commun. 2024, 159, 111735. [Google Scholar] [CrossRef]
- Phang, Y.K.; Aminuzzaman, M.; Akhtaruzzaman; Muhammad, G.; Ogawa, S.; Watanabe, A.; Tey, L.H. Green synthesis and characterization of CuO nanoparticles derived from papaya peel extract for the photocatalytic degradation of palm oil mill effluent (POME). Sustainability 2021, 13, 796. [Google Scholar] [CrossRef]
- Cheah, S.Y.; Phang, Y.K.; Koh, M.X.; Chia-Yen Lim, S.; Djearamane, S.; Subramaniam, H.; Lim, B.H.; Li, F.; Aminuzzaman, M.; Wong, L.S.; et al. Eco-sustainable synthesis of chromium oxide (Cr2O3) nanoparticles via pomegranate husk extract: Calcination-driven control of structure and properties. Green Process. Synth. 2025, 15, 20250162. [Google Scholar] [CrossRef]
- Cheah, S.Y.; Aminuzzaman, M.; Phang, Y.K.; Lim, S.C.Y.; Koh, M.X.; Djearamane, S.; Subramaniam, H.; Lim, B.H.; Li, F.; Wong, L.S.; et al. Green-synthesized chromium oxide nanoparticles using pomegranate husk extract: Multifunctional bioactivity in antioxidant potential, lipase and amylase inhibition, and cytotoxicity. Green Process. Synth. 2025, 14, 20240246. [Google Scholar] [CrossRef]
- Chan, Y.B.; Aminuzzaman, M.; Chuah, X.T.; Li, K.; Balu, P.; Wong, L.S.; Guha, S.K.; Tey, L.H. Review in green synthesis mechanisms, application, and future prospects for Garcinia mangostana L. (mangosteen)-derived nanoparticles. Nanotechnol. Rev. 2025, 14, 20250157. [Google Scholar] [CrossRef]
- Mohammad Abu-Taweel, G.; Ibrahim, M.M.; Khan, S.; Al-Saidi, H.M.; Alshamrani, M.; Alhumaydhi, F.A.; Alharthi, S.S. Medicinal importance and chemosensing applications of pyridine derivatives: A review. Crit. Rev. Anal. Chem. 2024, 54, 599–616. [Google Scholar] [CrossRef]
- Lokesh Kumar, S.; Tabassum, S.; Sagar, K.S.; Govindaraju, S. A mini review on the multicomponent synthesis of pyridine derivatives. ChemistrySelect 2022, 7, e202203668. [Google Scholar] [CrossRef]
- De, S.; Ashok Kumar, S.K.; Shah, S.K.; Kazi, S.; Sarkar, N.; Banerjee, S.; Dey, S. Pyridine: The scaffolds with significant clinical diversity. RSC Adv. 2022, 12, 15385–15406. [Google Scholar] [CrossRef]
- Patil, S.B. Recent medicinal approaches of novel pyrimidine analogs: A review. Heliyon 2023, 9, e16773. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.W.; Islam, M.M.; Akter, R.; Limon, T.R.; Vasquez, E.S.; Shaikh, M.A.A.; Habib, A. A review on pyrimidine-based derivatives: Synthesis and their biological application. J. Heterocycl. Chem. 2024, 61, 1159–1179. [Google Scholar] [CrossRef]
- Venkatesan, K.; Satyanarayana, V.S.V.; Sivakumar, A. Synthesis of pyrimidine carboxamide derivatives catalyzed by uranyl nitrate hexa Hydrate with their antibacterial and antioxidant studies. Bull. Chem. Soc. Ethiop. 2016, 30, 119–128. [Google Scholar] [CrossRef]
- Hosseinzadeh, Z.; Ramazani, A.; Ahankar, H.; Ślepokura, K.; Lis, T. Synthesis of 2-amino-4,6-diarylnicotinonitrile in the presence of CoFe2O4@SiO2-SO3H as a reusable solid acid nanocatalyst under microwave irradiation in solvent-freeconditions. Silicon 2019, 11, 2169–2176. [Google Scholar] [CrossRef]
- Afradi, M.; Pour, S.A.; Dolat, M.; Yazdani-Elah-Abadi, A. Nanomagnetically modified vitamin B3 (Fe3O4@Niacin): An efficient and reusable green biocatalyst for microwave-assisted rapid synthesis of 2-amino-3-cyanopyridines in aqueous medium. Appl. Organomet. Chem. 2018, 32, e4103. [Google Scholar] [CrossRef]
- Dehghan, P.; Mohebat, R. A highly efficient and green synthesis of pyrimido-fused benzophenazines via microwave-assisted and H3PW12O40@Nano-ZnO catalyzed a sequential one-pot cyclization in aqueous medium. Polycycl. Aromat. Compd. 2018, 40, 1164–1174. [Google Scholar] [CrossRef]
- Pan, Y. Magnetic nanocatalyst for microwave-assisted synthesis of Benzo[4,5]imidazo[1,2-a]pyrimidines via A3 coupling. Front. Chem. 2025, 13, 1631183. [Google Scholar] [CrossRef]
- Paul, D.; Reddy, R.G.; Rajendran, S.P. Facile ecofriendly one pot synthesis of heterocyclic priviledged medicinal scaffolds via biginelli reaction using retrievable nickel nanoparticles as catalyst. J. Chil. Chem. Soc. 2018, 63, 3974–3982. [Google Scholar] [CrossRef]
- Achary, L.S.K.; Kumar, A.; Rout, L.; Kunapuli, S.V.; Dhaka, R.S.; Dash, P. Phosphate functionalized graphene oxide with enhanced catalytic activity for Biginelli type reaction under microwave condition. Chem. Eng. J. 2018, 331, 300–310. [Google Scholar] [CrossRef]
- Jain, S.; Jain, S.; Verma, D. Novel ZnO-Co3O4-CuO Nanocomposite Catalyzed Microwave-Assisted Facile Synthesis of Polyhydroquinoline Derivatives. Appl. Organomet. Chem. 2025, 39, e7835. [Google Scholar] [CrossRef]
- Moradnia, F.; Taghavi Fardood, S.; Ramazani, A. Green synthesis and characterization of NiFe2O4@ ZnMn2O4 magnetic nanocomposites: An efficient and reusable spinel nanocatalyst for the synthesis of tetrahydropyrimidine and polyhydroquinoline derivatives under microwave irradiation. Appl. Organomet. Chem. 2024, 38, e7315. [Google Scholar] [CrossRef]
- Moradi, L.; Tadayon, M. Green synthesis of 3,4-dihydropyrimidinones using nano Fe3O4@ meglumine sulfonic acid as a new efficient solid acid catalyst under microwave irradiation. J. Saudi Chem. Soc. 2018, 22, 66–75. [Google Scholar] [CrossRef]
- Kumar, P.V.; Madhumitha, G. Microwave-assisted synthesis of 4-amino pyrimidine analogues using eco-friendly NiTiO3 nanoparticle supported montmorillonite K30 as a heterogeneous catalyst. New J. Chem. 2024, 48, 14410–14419. [Google Scholar] [CrossRef]
- Rout, L.; Kumar, A.; Chand, P.K.; Achary, L.S.K.; Dash, P. Microwave-Assisted Efficient One-Pot Multi-Component Synthesis of Octahydroquinazolinone Derivatives Catalyzed by Cu@Ag Core-Shell Nanoparticle. ChemistrySelect 2019, 4, 5696–5706. [Google Scholar] [CrossRef]
- Thongni, A.; Nongkhlaw, R.; Pandya, C.; Sivaramakrishna, A.; Gannon, P.M.; Kaminsky, W. Microwave-assisted synthesis of benzo[4,5]imidazo[1,2-a]pyrimidines and pyrano[4,3-b]pyrans catalyzed by L-glutamine functionalized magnetic nanoparticles in water: Ethanol mixture. J. Heterocycl. Chem. 2024, 61, 581–599. [Google Scholar] [CrossRef]
- Kumari, M.; Jain, Y.; Yadav, P.; Laddha, H.; Gupta, R. Synthesis of Fe3O4-DOPA-Cu magnetically separable nanocatalyst: A versatile and robust catalyst for an array of sustainable multicomponent reactions under microwave irradiation. Catal. Lett. 2019, 149, 2180–2194. [Google Scholar] [CrossRef]
- Alshahrani, M.Y.; Al-dulaimi, A.A.; Sead, F.; Ganesan, S.; Al-Hetty, H.R.A.K.; Garg, G.; Pramanik, A.; Ray, S.; Hammady, F.J.; Salih, S.A. Synergistic effect of silica nanoparticles and microwave irradiation in catalysis of pyrimidine derivatives synthesis. Res. Chem. Intermed. 2025. [Google Scholar] [CrossRef]
- Ouakki, M.; Galai, M.; Cherkaoui, M. Imidazole derivatives as efficient and potential class of corrosion inhibitors for metals and alloys in aqueous electrolytes: A review. J. Mol. Liq. 2022, 345, 117815. [Google Scholar] [CrossRef]
- Rani, N.; Singh, R.; Kumar, P. Imidazole and derivatives drugs synthesis: A review. Curr. Org. Synth. 2023, 20, 630–662. [Google Scholar] [CrossRef]
- Devi, M.M.; Devi, K.S.; Singh, O.M.; Singh, T.P. Synthesis of imidazole derivatives in the last 5 years: An update. Heterocycl. Commun. 2024, 30, 20220173. [Google Scholar] [CrossRef]
- Claramunt, R.M.; López, C.; López, A.; Pérez-Medina, C.; Pérez-Torralba, M.; Alkorta, I.; Elguero, J.; Escames, G.; Acuña-Castroviejo, D. Synthesis and biological evaluation of indazole derivatives. Eur. J. Med. Chem. 2011, 46, 1439–1447. [Google Scholar] [CrossRef] [PubMed]
- Mal, S.; Malik, U.; Mahapatra, M.; Mishra, A.; Pal, D.; Paidesetty, S.K. A review on synthetic strategy, molecular pharmacology of indazole derivatives, and their future perspective. Drug Dev. Res. 2022, 83, 1469–1504. [Google Scholar] [CrossRef] [PubMed]
- Sedaghat, M.; Moeinpour, F.; Mohseni-Shahri, F.S. Copper(II)/polyimide linked covalent organic framework as a powerful catalyst for the solvent-free microwave irradiation-based synthesis of 2,4,5-trisubstituted imidazoles. Anal. Sci. Adv. 2023, 4, 302–311. [Google Scholar] [CrossRef]
- Kafi-Ahmadi, L.; Khademinia, S.; Poursattar Marjani, A.; Nozad, E. Microwave-assisted preparation of polysubstituted imidazoles using Zingiber extract synthesized green Cr2O3 nanoparticles. Sci. Rep. 2022, 12, 19942. [Google Scholar] [CrossRef]
- Bheemayya, L.; Kamble, R.R.; Shettar, A.K.; Nadoni, V.B.; Nayak, M.R.; Joshi, S.D.; Bayannavar, P.K.; Metre, T.V.; Keri, R.S.; Hoskeri, J.H. Microwave-Assisted Fe3O4 Nanoparticles Catalyzed Cascade Synthesis of 3-(1,4,5-Triaryl-1H-imidazol-2yl)quinolin-2-amines as COX-1, COX-2 Inhibitors and Antioxidant Agents. Appl. Organomet. Chem. 2025, 39, e7812. [Google Scholar] [CrossRef]
- Taheri, M.; Jawhar, Z.H. Microwave-assisted Multi-component Reaction for the Green Synthesis of Novel 4-(5-hydroxybenzo[a]phenazin-6-yl)-5-phenyl-1,3-dihydro-2H-imidazol-2-one Using H3PW12O40@nano-TiO2 as Recyclable Catalyst. Green Chem. Lett. Rev. 2022, 15, 813–824. [Google Scholar] [CrossRef]
- Radhi, S.M.; Taresh, B.H.; Shareef, N.K.; Ahmed, L.M. One-pot, Three Component, and Green Synthesis of 2-(Aryl)-3-((2-oxo-2-(2-oxo-2H-chromen-3-yl) ethyl) amino) imidazolidin-4-one Derivatives using Graphene Oxide Nanosheets under Microwave Irradiation: A Comparison Study Between One-pot and Step by Step Synthesis. Iran. J. Catal. 2022, 12, 373–387. [Google Scholar] [CrossRef]
- Zahedi, N.; Javid, A.; Mohammadi, M.K.; Tavakkoli, H. Microwave-promoted solvent free one-pot synthesis of triazolo[1,2-a]indazole-triones catalyzed by silica-supported La0.5Ca0.5CrO3 nanoparticles as a new and reusable perovskite-type oxide. Bull. Chem. Soc. Ethiop. 2018, 32, 239–248. [Google Scholar] [CrossRef]
- Maddila, S.; Kerru, N.; Jonnalagadda, S.B. Recent progress in the multicomponent synthesis of pyran derivatives by sustainable catalysts under green conditions. Molecules 2022, 27, 6347. [Google Scholar] [CrossRef]
- El-Bana, G.G.; Salem, M.A.; Helal, M.H.; Alharbi, O.; Gouda, M.A. A review on the recent multicomponent synthesis of 4H-pyran derivatives. Mini-Rev. Org. Chem. 2024, 21, 73–91. [Google Scholar] [CrossRef]
- Venkatesan, K.; Rao, T.S.; Sridhar, V.; Yacobe, J.; Satyanarayana, T.V.V.; Pasupathi, M. Ultrasound Assisted Synthesis of Pyran Derivatives Catalysed by Uranyl Nitrate and Their Molecular Docking Against Glycogen Synthase Kinase-3 Beta Receptor. Russ. J. Bioorganic Chem. 2024, 50, 2580–2588. [Google Scholar] [CrossRef]
- Kumar Maurya, R.; Dey, A.; Kumara, V.; Khatravath, M. Recent Advances on Synthesis of 2H-Chromenes, and Chromenes Fused Hetrocyclic Compounds. Asian J. Org. Chem. 2024, 13, e202400259. [Google Scholar] [CrossRef]
- Chadha, M.; Garg, A.; Bhalla, A.; Berry, S. Green methods mediated synthesis of chromene derivatives using magnetic nanoparticles as heterogeneous and reusable nanocatalyst: A review. Tetrahedron 2024, 150, 133741. [Google Scholar] [CrossRef]
- Sharon, K.N.; Padmaja, P.; Reddy, P.N. A Brief Review on the Synthesis of 4H-Chromene-Embedded Heterocycles. ChemistrySelect 2024, 9, e202400565. [Google Scholar] [CrossRef]
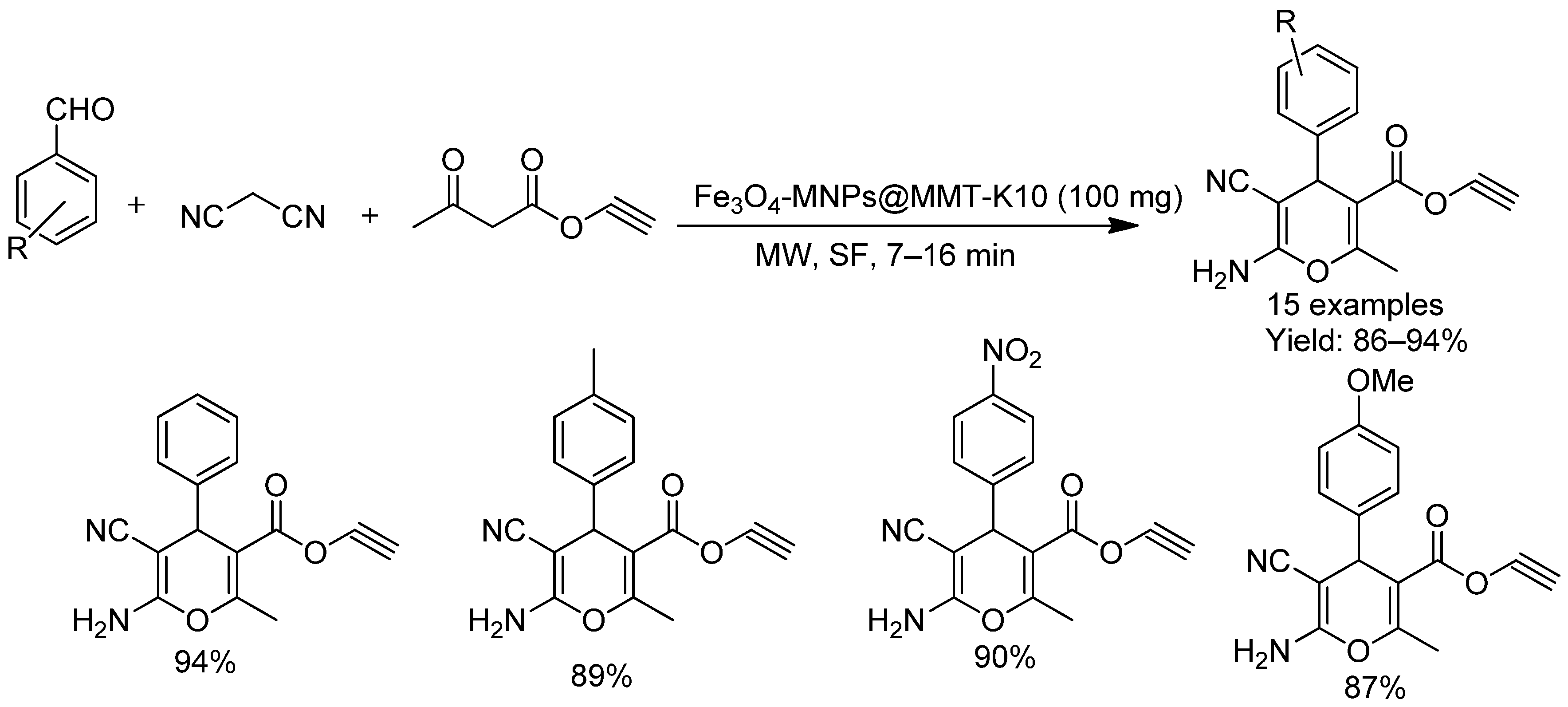
- Thanh, N.D.; Hai, D.S.; Huyen, L.T.; Thuy, V.T.T.; Tung, D.T.; Van, H.T.K.; Toan, V.N.; Giang, N.T.K.; Tri, N.M. Fe3O4-MNPs@MMT-K10: A reusable catalyst for synthesis of propargyl 4-aryl-4 H-pyran-3-carboxyles via one pot three-component reaction under microwave-assisted solvent-free conditions. Res. Chem. Intermed. 2023, 49, 525–555. [Google Scholar] [CrossRef]
- Okram, M.; Bharti, R.; Sharma, R.; Verma, M.; Bansal, R. Microwave-assisted Cr2O3 nanoparticle-catalyzed synthesis of 2-amino-4H-chromenes: Evaluation of antioxidant, corrosion inhibition, and molecular docking studies. Res. Chem. Intermed. 2025, 51, 6049–6086. [Google Scholar] [CrossRef]
- Jopale, M.K.; Shelke, B.N.; Mharsale, N.; Patil, S.; Chine, S.S.; Lokhande, D.D.; Rote, R.; Gade, V.; Kategaonkar, A.H. Synthesis and characterisation of mixed oxides of Co–Ni catalyst and its application in microwave mediated synthesis of Benzo[b]Pyrans. J. Indian Chem. Soc. 2024, 101, 101265. [Google Scholar] [CrossRef]
- Kafi-Ahmadi, L.; Poursattar Marjani, A.; Nozad, E. Ultrasonic-assisted preparation of Co3O4 and Eu-doped Co3O4 nanocatalysts and their application for solvent-free synthesis of 2-amino-4H-benzochromenes under microwave irradiation. Appl. Organomet. Chem. 2021, 35, e6271. [Google Scholar] [CrossRef]
- Ahankar, H.; Fardood, S.T.; Ramazani, A. One-pot three-component synthesis of tetrahydrobenzo[b]pyrans in the presence of Ni0.5Cu0.5Fe2O4 magnetic nanoparticles under microwave irradiation in solvent-free conditions. Iran. J. Catal. 2020, 10, 195–201. Available online: https://oiccpress.com/ijc/article/view/3561.
- Taheri, M.; Mohebat, R. Synthesis of one-pot pyrazolo [4′, 3′: 5, 6] pyrano [2, 3-c] phenazin-15-yl) methanone derivatives via a multi-component using Fe3O4@ TiO2-SO3H as a recoverable magnetic catalyst under microwave irradiation. Green Chem. Lett. Rev. 2020, 13, 165–178. [Google Scholar] [CrossRef]
- Behera, B.K.; Ghosh, P.; Saikia, A.K. Recent advances in the synthesis of N-heterocycles via Lewis acid-catalyzed/mediated cyclization of propargyl-and homopropargyl-amines and their derivatives. Tetrahedron 2024, 162, 134123. [Google Scholar] [CrossRef]
- Carneiro, A.; Uriarte, E.; Borges, F.; Matos, M.J. Propargylamine: An important moiety in drug discovery. Future Med. Chem. 2023, 15, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Hasan, K.; Joseph, R.G.; Patole, S.P.; Al-Qawasmeh, R.A. Development of magnetic Fe3O4-chitosan immobilized Cu (II) Schiff base catalyst: An efficient and reusable catalyst for microwave assisted one-pot synthesis of propargylamines via A3 coupling. Catal. Commun. 2023, 174, 106588. [Google Scholar] [CrossRef]
- Shah, A.P.; Sharma, A.S.; Jain, S.; Shimpi, N.G. Microwave assisted one pot three component synthesis of propargylamine, tetra substituted propargylamine and pyrrolo[1,2-a]quinolines using CuNPs@ ZnO–PTh as a heterogeneous catalyst. New J. Chem. 2018, 42, 8724–8737. [Google Scholar] [CrossRef]
- Patel, S.B.; Vasava, D.V. Carbon Nitride-Supported Silver Nanoparticles: Microwave-Assisted Synthesis of Propargylamine and Oxidative C-C Coupling Reaction. ChemistrySelect 2018, 3, 471–480. [Google Scholar] [CrossRef]
- Che, Y.X.; Qi, X.N.; Lin, Q.; Yao, H.; Qu, W.J.; Shi, B.; Zhang, Y.M.; Wei, T.B. Design strategies and applications of novel functionalized phenazine derivatives: A review. J. Mater. Chem. C 2022, 10, 11119–11174. [Google Scholar] [CrossRef]
- Huang, W.; Wan, Y.; Zhang, S.; Wang, C.; Zhang, Z.; Su, H.; Xiong, P.; Hou, F. Recent advances in phenazine natural products: Chemical structures and biological activities. Molecules 2024, 29, 4771. [Google Scholar] [CrossRef]
- Nadtochiy, V.V.; Nikonov, I.L.; Zyryanov, G.V. Modern approaches to the synthesis of phenazine derivatives (microreview). Chem. Heterocycl. Compd. 2024, 60, 233–235. [Google Scholar] [CrossRef]
- Taheri, M.; Yousefi, M.; Mehdipourrad, M.; Jawhar, Z.H.; Alnoaemi, B.M.; Shahcheragh, S.K. Synthesis of benzo[a]furo[2,3-c]phenazine derivatives via microwave irradiation by catalyzed Fe3O4@ MCM-48@ IL/Pd for high-performance removal of rhodamine B and determination of losartan using modified glassy carbon electrode. Res. Chem. Intermed. 2024, 50, 2221–2245. [Google Scholar] [CrossRef]
- Taheri, M.; Mohebat, R.; Moslemin, M.H. Microwave-Assisted Multi-Component Green Synthesis of Benzo[α]furo[2,3-c]phenazine Derivatives via a Magnetically-Separable Fe3O4@rGO@ZnO-HPA Nanocatalyst under Solvent-Free Conditions. Polycycl. Aromat. Compd. 2023, 43, 586–596. [Google Scholar] [CrossRef]
- Taheri, M.; Mohebat, R.; Moslemin, M.H. Synthesis of benzo[a]furo[2,3-c] phenazine derivatives through an efficient, rapid and via microwave irradiation under solvent-free conditions catalyzed by H3PW12O40@Fe3O4 ZnO for high-performance removal of methylene blue. Artif. Cells Nanomed. Biotechnol. 2021, 49, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Tomar, V.; Joshi, R.K.; Nemiwal, M. Nanocatalyzed synthetic approach for quinazoline and quinazolinone derivatives: A review (2015–present). Synth. Commun. 2022, 52, 795–826. [Google Scholar] [CrossRef]
- Varala, R.; Kamsali, M.M.A.; Hussein, M.; Alam, M.M. Visible Light-Mediated Synthesis of Quinazoline and Quinazolinone Derivatives: A Quadrennial Update. J. Heterocycl. Chem. 2025, 62, 848–879. [Google Scholar] [CrossRef]
- Avula, B.; Reddivari, C.K.R.; Muchumarri, R.M.R.; Eraganaboyina, S.; Zyryanov, G.V.; Nemallapudi, B.R. Recent advances in the synthesis of quinoxalines. a mini review. Polycycl. Aromat. Compd. 2024, 44, 634–670. [Google Scholar] [CrossRef]
- Sharma, A.; Narasimhan, B.; Marwaha, R.K. Advances in Quinoxaline Derivatives: Multi-Target Anticancer Potential and Nanocatalyst-Driven Synthesis. ChemistrySelect 2025, 10, e02264. [Google Scholar] [CrossRef]
- Sharma, A.; Deep, A.; Marwaha, M.G.; Marwaha, R.K. Quinoxaline: A chemical moiety with spectrum of interesting biological activities. Mini Rev. Med. Chem. 2022, 22, 927–948. [Google Scholar] [CrossRef]
- Aswar, A.; Salunkhe, N.G.; Arun Ladole, C.; Thakare, N.V.; Barabde, J.M. A simple and convenient synthesis of 2,3-dihydroquinazolin-4 (1H)-one derivatives using MgFe2O4@ SiO2-SO3H catalyst. Adv. Mater. Lett. 2021, 12, 1–5. [Google Scholar] [CrossRef]
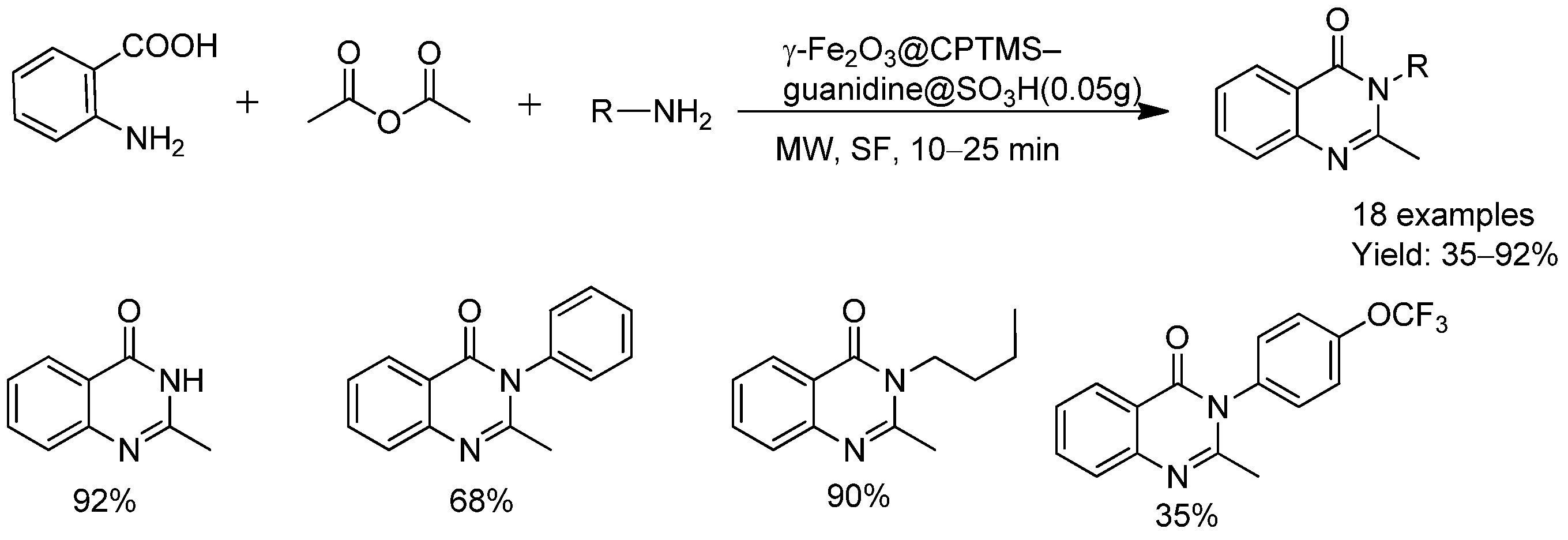
- Norouzi, F.H.; Foroughifar, N.; Khajeh-Amiri, A.; Pasdar, H. A novel superparamagnetic powerful guanidine-functionalized γ-Fe2O3 based sulfonic acid recyclable and efficient heterogeneous catalyst for microwave-assisted rapid synthesis of quinazolin-4(3H)-one derivatives in Green media. RSC Adv. 2021, 11, 29948–29959. [Google Scholar] [CrossRef]
- Taheri, M.; Mohebat, R.; Moslemin, M.H.; Jawhar, Z.H. Nano-Fe3O4@rGO@ZnO-H3PW12O40 as a recoverable magnetic catalyst for one-pot synthesis of benzo[f]furo[2,3-h]quinoxaline-5,6-dicarbonitrile under solvent-free conditions. Mol. Cryst. Liq. Cryst. 2023, 755, 62–79. [Google Scholar] [CrossRef]
- Abualhasan, M.; Hawash, M.; Aqel, S.; Al-Masri, M.; Mousa, A.; Issa, L. Biological evaluation of xanthene and thioxanthene derivatives as antioxidant, anticancer, and COX inhibitors. ACS Omega 2023, 8, 38597–38606. [Google Scholar] [CrossRef]
- Ahmed, L.A.; Sharmah, H. A Concise Review of Synthetic Xanthone Derivatives for Alzheimer’s Disease. Brain Disord. 2025, 100256. [Google Scholar] [CrossRef]
- Ran, X.Y.; Wei, Y.F.; Wu, Y.L.; Dai, L.R.; Xia, W.L.; Zhou, P.Z.; Li, K. Xanthene-based NIR organic phototheranostics agents: Design strategies and biomedical applications. J. Mater. Chem. B 2025, 13, 2952–2977. [Google Scholar] [CrossRef]
- Alsalhi, M.S.; Tarek, M.; Said, G.E.; Almehizia, A.A.; Naglah, A.M.; Khatab, T.K. Microwave-assisted synthesis of a zirconium-based MOF as an efficient catalyst for one-pot synthesis of xanthene derivatives: In silico study as a potential anti-HIV RNA. RSC Adv. 2025, 15, 16654–16666. [Google Scholar] [CrossRef]
- Lambat, T.L.; Mahmood, S.H.; Ledade, P.V.; Banerjee, S. Microwave Assisted One-Pot Multicomponent Synthesis Using ZnO-β Zeolite Nanoparticle: An Easy Access to 7-Benzodioxolo[4,5-b]xanthene-dione and 4-Oxo-tetrahydroindole Scaffolds. ChemistrySelect 2020, 5, 8864–8874. [Google Scholar] [CrossRef]
- Goni, L.K.; Jafar Mazumder, M.A.; Tripathy, D.B.; Quraishi, M.A. Acridine and its derivatives: Synthesis, biological, and anticorrosion properties. Materials 2022, 15, 7560. [Google Scholar] [CrossRef]
- Gupta, S.; Tiwari, A.; Sahoo, N.K.; Tiwari, V. Emerging Insights and Innovations in Acridine Derivatives: A Review. Curr. Bioact. Compd. 2025, 22, e15734072346755. [Google Scholar] [CrossRef]
- Majhi, S. Recent developments in the synthesis and anti-cancer activity of acridine and xanthine-based molecules. Phys. Sci. Rev. 2023, 8, 2405–2439. [Google Scholar] [CrossRef]
- Nguyen, P.N.; Nguyen, G.L.N.; Duong, T.-A.T.; Le, M.P.T.; Nguyen, L.P.; Kim, J.; Tran, P.H.; Truong, H.H.T.; Nguyen, H.T. High-yield, fast, and green synthesis of acridine derivatives using a Co/C catalyst from rice husks with a microwave-assisted method. React. Chem. Eng. 2024, 9, 2034–2049. [Google Scholar] [CrossRef]
- de Castro, P.P.; Carpanez, A.G.; Amarante, G.W. Azlactone reaction developments. Chem. Eur. J. 2016, 22, 10294–10318. [Google Scholar] [CrossRef]
- Mistry, R.N.; Desai, K.R. Studies on synthesis of some novel heterocyclic azlactone derivatives and imidazolinone derivatives and their antimicrobial activity. J. Chem. 2005, 2, 42–51. [Google Scholar] [CrossRef]
- Kushwaha, N.; Kushwaha, S. Synthetic approaches and biological significance of oxazolone moieties: A review. Biointerface Res. Appl. Chem. 2022, 12, 6460–6486. [Google Scholar] [CrossRef]
- Buddiga, L.R.; Gajula, G.R.; Brahmayya, M.; Bhanuchander, P. Zr/P Co-Doped TiO2 Nanonanocatalyst for Microwave-Assisted Green Synthesis of Bioactive Azlactones: Enhanced Catalytic Performance and Mechanistic Insights. Catal. Lett. 2026, 156, 23. [Google Scholar] [CrossRef]
- Mishra, I.; Sharma, V.; Kumar, N.; Krishna, G.; Sethi, V.A.; Mittal, R.; Dhakad, P.K.; Mishra, R. Exploring Thiophene Derivatives: Synthesis Strategies and Biological Significance. Med. Chem. 2025, 21, 11–31. [Google Scholar] [CrossRef]
- Shah, R.; Verma, P.K. Therapeutic importance of synthetic thiophene. Chem. Cent. J. 2018, 12, 137. [Google Scholar] [CrossRef]
- Archna; Pathania, S.; Chawla, P.A. Thiophene-based derivatives as anticancer agents: An overview on decade’s work. Bioorganic Chem. 2020, 101, 104026. [Google Scholar] [CrossRef]
- Zargari, M.; Ardeshiri, H.H.; Ghafuri, H.; Hassanzadeh, M.M. Fe3O4 nanoparticles impregnated eggshell as an efficient biocatalyst for eco-friendly synthesis of 2-amino thiophene derivatives. Heliyon 2024, 10, e29674. [Google Scholar] [CrossRef]
- Zampieri, D.; Mamolo, M.G.; Filingeri, J.; Fortuna, S.; De Logu, A.; Sanna, A.; Zanon, D. Design, synthesis and antimycobacterial activity of benzoxazinone derivatives and open-ring analogues: Preliminary data and computational analysis. Bioorganic Med. Chem. Lett. 2019, 29, 2468–2474. [Google Scholar] [CrossRef]
- Sebbar, N.K.; El Ghayati, L.; Hlimi, F.; El Haddad, S.; Mokhtar Essassi, E. Synthetic Strategies and Therapeutic Profile of Some 1,4-benzoxazine Derivatives: A Review. Curr. Org. Chem. 2025, 30, 40–58. [Google Scholar] [CrossRef]
- Salunkhe, N.G.; Ladole, C.A.; Thakare, N.V.; Aswar, A.S. MgFe2O4@ SiO2–SO3H: An efficient, reusable catalyst for the microwave-assisted synthesis of benzoxazinone and benzthioxazinone via multicomponent reaction under solvent free condition. Res. Chem. Intermed. 2018, 44, 355–372. [Google Scholar] [CrossRef]
- Haji Norouzi, F.; Foroughifar, N.; Khajeh-Amiri, A.; Pasdar, H. A Novel Powerful γ-Fe2O3@ CPTMS-Diethylene Triamine (DETA)@SO3H as a Heterogeneous Nanocatalyst, Recyclable and Efficient via Microwave-assisted for the Synthesis of Benzoxazinone-4 (3H)-one Derivatives in Green Media. Lett. Org. Chem. 2023, 20, 519–532. [Google Scholar] [CrossRef]
- An, Y.S.; Hao, Z.F.; Zhang, X.J.; Wang, L.Z. Efficient Synthesis and Biological Evaluation of a Novel Series of 1,5-Benzodiazepine Derivatives as Potential Antimicrobial Agents. Chem. Biol. Drug Des. 2016, 88, 110–121. [Google Scholar] [CrossRef]
- Tolu-Bolaji, O.O.; Sojinu, S.O.; Okedere, A.P.; Ajani, O.O. A review on the chemistry and pharmacological properties of benzodiazepine motifs in drug design. Arab. J. Basic Appl. Sci. 2022, 29, 287–306. [Google Scholar] [CrossRef]
- Pourghasem, S.; Moeinpour, F.; Mohseni-Shahri, F.S. Cu (II)/polyimide linked COF: An effective mesoporous catalyst for solvent-free 1,5-benzodiazepine synthesis. Arab. J. Chem. 2023, 16, 104694. [Google Scholar] [CrossRef]
- Kaushik, N.; Kumar, N.; Kumar, A.; Singh, U.K. Tetrazoles: Synthesis and biological activity. Immunol. Endocr. Metab. Agents Med. Chem. 2018, 18, 3–21. [Google Scholar] [CrossRef]
- Vishwakarma, R.; Gadipelly, C.; Mannepalli, L.K. Advances in tetrazole synthesis–an overview. ChemistrySelect 2022, 7, e202200706. [Google Scholar] [CrossRef]
- Hussein, M.; Varala, R.; Kamsali, M.M.A.; Seema, V.; Beda, D.P.; Syed, M.A.; Alam, M.M. Recent Advances in the Chemistry of Tetrazole Derivatives-A Quinquennial Update (Mid-2019 to date). Mini-Rev. Org. Chem. 2025, 22, 1–18. [Google Scholar] [CrossRef]
- Ramírez-Coronel, A.A.; Sivaraman, R.; Ahmed, Y.M.; Abass, R.R.; Kareem, H.A. A Green and Ecofriendly Catalytic System for One-Pot Three-Component Synthesis of 5-Substituted 1H Tetrazoles Under Microwave Irradiation. Polycycl. Aromat. Compd. 2024, 44, 577–590. [Google Scholar] [CrossRef]
- Sikandar, S.; Zahoor, A.F. Synthesis of pyrano[2,3-c]pyrazoles: A review. J. Heterocycl. Chem. 2021, 58, 685–705. [Google Scholar] [CrossRef]
- Myrboh, B.; Mecadon, H.; Rohman, M.R.; Rajbangshi, M.; Kharkongor, I.; Laloo, B.M.; Kharbangar, I.; Kshiar, B. Synthetic developments in functionalized pyrano[2,3-c]pyrazoles. A review. Org. Prep. Proced. Int. 2013, 45, 253–303. [Google Scholar] [CrossRef]
- Farooq, S.; Ngaini, Z. Recent Synthesis of Mono-& Bis-Pyranopyrazole Derivatives. ChemistrySelect 2024, 9, e202400028. [Google Scholar] [CrossRef]
- Thakare, N.V.; Aswar, A.S.; Salunkhe, N.G. Magnetically recoverable nanocatalyst for the synthesis of pyranopyrazoles: CoFe2O4@SiO2-HClO4. Eur. J. Chem. 2023, 14, 385–392. [Google Scholar] [CrossRef]
- Sun, P.; Huang, Y.; Chen, S.; Ma, X.; Yang, Z.; Wu, J. Indole derivatives as agrochemicals: An overview. Chin. Chem. Lett. 2024, 35, 109005. [Google Scholar] [CrossRef]
- Chen, Q.; Wu, C.; Zhu, J.; Li, E.; Xu, Z. Therapeutic potential of indole derivatives as anti-HIV agents: A mini-review. Curr. Top. Med. Chem. 2022, 22, 993–1008. [Google Scholar] [CrossRef]
- Gohain, S.B.; Thakur, A.J. Au Nanostructures with Controlled Morphology, Biosynthesized from Garcinia cowa Fruit Extract, and Their Use in Microwave-Mediated Bisindole Synthesis. ChemistrySelect 2021, 6, 6773–6780. [Google Scholar] [CrossRef]
- Shanbhag, G.S.; Bhargava, A.; Singh, G.P.; Joshi, S.D.; Chundawat, N. Synthesis, molecular simulation studies, in vitro biological assessment of 2-substituted benzoxazole derivatives as promising antimicrobial agents. Turk. J. Chem. 2023, 47, 263–279. [Google Scholar] [CrossRef]
- Abdullahi, A.; Yeong, K.Y. Targeting disease with benzoxazoles: A comprehensive review of recent developments. Med. Chem. Res. 2024, 33, 406–438. [Google Scholar] [CrossRef]
- Naeimi, H.; Rouzegar, Z.; Rahmatinejad, S. Catalyst-free microwave-promoted one pot synthesis of 2-aryl benzoxazoles using MnO2 nanoparticles as a convenient oxidant under mild condition. Res. Chem. Intermed. 2017, 43, 4745–4758. [Google Scholar] [CrossRef]
- Vaishnani, M.J.; Bijani, S.; Rahamathulla, M.; Baldaniya, L.; Jain, V.; Thajudeen, K.Y.; Ahmed, M.M.; Farhana, S.A.; Pasha, I. Biological importance and synthesis of 1,2,3-triazole derivatives: A review. Green Chem. Lett. Rev. 2024, 17, 2307989. [Google Scholar] [CrossRef]
- Salma, U.; Ahmad, S.; Alam, M.Z.; Khan, S.A. A review: Synthetic approaches and biological applications of triazole derivatives. J. Mol. Struct. 2024, 1301, 137240. [Google Scholar] [CrossRef]
- Attia, Y.A.; Mohamed, Y.M. Microwave-assisted synthesis of 1,4-disubstituted 1,2,3-triazole derivatives utilizing NiO/Cu2O nano-photocatalyst. J. Chem. Technol. Biotechnol. 2024, 99, 2311–2319. [Google Scholar] [CrossRef]















































| Parameter | Conventional Catalysts (Bulk/Homogeneous) | Nanoparticle Catalysts |
|---|---|---|
| Surface Area | Relatively low | Very high surface-to-volume ratio |
| Active Sites | Limited catalytic active sites | Large number of accessible active sites |
| Reaction Rate | Moderate to slow | Significantly faster |
| Reaction Conditions | Often requires higher temperature and longer reaction time | Mild conditions with shorter reaction time |
| Product Yield | Moderate yield | Higher yield |
| Catalyst Recovery | Difficult for homogeneous catalysts | Easy recovery and recyclability |
| Energy Efficiency | Lower | Higher due to efficient microwave absorption |
| Selectivity | Moderate | Often improved selectivity |
| Reaction Type | Nanocatalyst | MW Time | Conventional Time | Yield (%) | Solvent | Catalyst Reuse (Cycles) | Atom Economy |
|---|---|---|---|---|---|---|---|
| Biginelli reaction | PGO | 5–10 min | 20 min | 95–98 | Solvent-free | 4 | High |
| Pyran | Co–Ni Oxide | 10 min | 1 h | 84–96 | Solvent-free | 12 | Moderate–High |
| Benzodiazepine | Cu@COF | 10 min | 5 h | 93–98 | Solvent-free | 5 | High |
| Propargylamine | CuNPs@ZnO–PTh | 6 min | 2 h | 78–97 | Ethylene glycol | 4 | Moderate |
| Pyrido-pyrimidine | SiNPs (rice husk) | 7 min | 3 h | 90–98 | Water | 8–9 | High |
| Tetrazole | MNPs-Picolylamine-Cu(OAc)2 | 15 min | 2 h | 81–99% | Water | 8 | High |
| dioxolo-xanthenone | ZnO-β Zeolite. | 4 min | 3 h | 84–95% | EtOH | 4 | Moderate–High |
| Indazole | S-LCCO NPs | 4 min | 20 min | 84–94% | Solvent-free | 4 | Moderate–High |
| Thiophene | eggshell/Fe3O4 | 4–15 min | 180 min | 82–97% | EtOH | 5 | Moderate–High |
| Effect | Illustration | Caption |
|---|---|---|
| Localized superheating | Hot spots around NP surface | NP absorbs MW energy more efficiently than bulk medium |
| Enhanced dielectric loss | Polar reactants clustering near NP | Increased local temperature at catalytic sites |
| Surface charge polarization | +/− charges on NP surface | Facilitates adsorption of polar intermediates |
| Metrices | Measured Parameter | Green Significance |
|---|---|---|
| Atom Economy (AE) | Efficiency of atom incorporation into product | Higher AE: less theoretical waste; MCRs typically show high AE |
| E-Factor | Amount of waste generated per mass of product | Lower value indicates greener process; solvent-free MW reactions reduce E-factor |
| Reaction Mass Efficiency (RME) | Practical mass efficiency considering yield | Combines AE and yield; reflects real process efficiency |
| Process Mass Intensity (PMI) | Total material used per mass of product | Lower PMI means fewer resources consumed |
| Carbon Efficiency (CE) | Fraction of carbon ending in product | Useful for carbon-rich heterocycle synthesis |
| Energy Efficiency | Energy required per unit product | MW reduces time but must be measured, not assumed |
| Catalyst Turnover Number (TON) | Catalyst productivity | Higher TON: less catalyst waste |
| Catalyst Recyclability | Reuse capability | Indicates long-term sustainability of nanocatalyst |
| Nanocatalyst | Heterocycle | Reaction Conditions | Reaction Time | Yield (%) | Recyclability (Cycles) | Distinct Green Chemistry Attributes |
|---|---|---|---|---|---|---|
| CoFe2O4@SiO2–SO3H | Pyridine derivatives | Solvent-free, MW irradiation | Short (minutes) | 86–92 | 5 | Solvent-free, magnetic recovery |
| Fe3O4@niacin | Cyanopyridines | Water, MW irradiation | Rapid | 73–95 | 6 | Aqueous medium, bio-functionalized catalyst |
| H3PW12O40@nano-ZnO | Pyrimido-phenazine derivatives | Water, MW irradiation | Short | 84–92 | 5 | Non-toxic solid acid, aqueous medium |
| NiFe2O4@MCM-41@IL/Pt | Imidazo-pyrimidines | MW irradiation, short time | 15–25 min | 89–96 | 5 | Magnetic separation, rapid synthesis |
| Ni(II)-Zeolite Y | Dihydropyrimidinones (DHPMs) | MW irradiation | Rapid | 62–81 | 4 | Heterogeneous catalyst, reduced waste |
| Phosphate-Graphene Oxide (PGO) | Pyrimidinones | MW irradiation | Short | 91–98 | 5 | High atom economy, reusable nanocomposite |
| ZnO–Co3O4–CuO nanocomposite | Polyhydroquinolines | PEG, 400 W MW | Short | 88–94 | 6 | Green solvent (PEG), multimetal synergy |
| NiFe2O4@ZnMn2O4 | Tetrahydropyrimidines & polyhydroquinolines | Solvent-free, MW | Rapid | 86–97 | 4 | Solvent-free, magnetic recoverability |
| Fe3O4@meglumine-SO3H | Dihydropyrimidinones | H2O/EtOH (1:1), MW | Short | 90–98 | 4 | Green solvent mixture, magnetic reuse |
| Cu@Ag core–shell NPs | Octahydroquinazolinones | MeOH, MW | Short | 58–96 | 5 | Core–shell recyclable catalyst |
| Fe3O4@SiO2@L-glutamine | Benzo-imidazo-pyrimidines | Water, MW | Rapid | 87–96 | 5 | Bio-functionalized, aqueous medium |
| Cu(II)/PL-COF | Triaryl imidazoles | Solvent-free, MW | Short | 93–98 | 5 | Solvent-free, recyclable COF |
| Cr2O3 NPs | Imidazoles | Water, MW | Rapid | 89–98 | 6 | Plant-mediated synthesis, aqueous system |
| Graphene Oxide | Imidazolidinones | MW irradiation | Short | 65–89 | 7 | Metal-free catalyst, high stability |
| Fe3O4-MMT-K10 | Pyran/Chromene derivatives | Solvent-free, MW | Short | 86–94 | 6 | Solvent-free, magnetic nanocomposite |
| Co–Ni Mixed Oxide | Benzo-pyrans | MW irradiation | Rapid | 84–96 | 12 | Bio-derived preparation, high durability |
| Fe3O4@CS@Schiff base@Cu | Propargylamines | MW irradiation | Short | 65–97 | 6 | Magnetic support, high retention of activity |
| AgNPs@g-C3N4 | Propargylamines | Ethanol, MW | Rapid | 89–97 | 6 | Recyclable, eco-friendly solvent |
| Fe3O4@MCM-48@IL/Pd | Benzo-furo-phenazines | Solvent-free, MW | Short | 85–93 | 6 | Magnetic, solvent-free, dual functionality |
| MgFe2O4@SiO2–SO3H | Quinazolinones | Solvent-free, MW | Rapid | 78–95 | 5 | Magnetic separation, reduced solvent use |
| Zr-MOF (Zr/VitB3) | Tetrahydroxanthenes | MW irradiation | Short | 58–95 | 3 | MOF-based, reusable framework |
| Zr/P co-doped TiO2 | Azlactones | MW irradiation | Short | 89–96 | Not reported | Reduced reaction time, improved efficiency |
| Eggshell/Fe3O4 | 2-Aminothiophenes | Ethanol, MW | 10 min | 82–97 | 5 | Waste-derived biocatalyst |
| Cu@PI-COF | Benzodiazepines | Solvent-free, MW | Rapid | 93–98 | 5 | High atom economy, solvent-free |
| MNPs–picolylamine–Cu(OAc)2 | Tetrazoles | Water, MW | Short | 81–99 | 8 | Aqueous medium, magnetic reuse |
| CoFe2O4@SiO2–HClO4 | Pyranopyrazoles | MW irradiation | Rapid | 78–96 | 4 | Magnetically recoverable |
| Entry | Reaction Type | Reaction Conditions | Catalyst System | Reaction Time | Yield (%) | Solvent System | Estimated E-Factor | PMI | Sustainability Observation |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Multicomponent heterocycle synthesis | Conventional heating | No catalyst | 8–12 h | 35–50 | Organic solvent | 40–60 | 65–85 | Low conversion and high waste generation due to prolonged heating |
| 2 | Multicomponent heterocycle synthesis | Microwave irradiation | No catalyst | 2–3 h | 45–60 | Organic solvent | 30–50 | 50–70 | Moderate yield but still significant solvent contribution |
| 3 | Multicomponent heterocycle synthesis | Microwave irradiation | Cu nanoparticle catalyst | 10–20 min | 85–95 | Ethanol/solvent-free | 6–14 | 12–22 | Dramatic improvement in efficiency, reduced solvent use and waste |
| 4 | A3 coupling reaction | Conventional heating | No catalyst | 10–15 h | 30–45 | Organic solvent | 45–70 | 70–95 | Poor atom utilization and high environmental footprint |
| 5 | A3 coupling reaction | Microwave irradiation | Metal nanoparticle catalyst | 15–30 min | 80–92 | Water/ethanol | 10–18 | 18–28 | Improved atom economy and reduced mass intensity |
| 6 | One-pot multicomponent synthesis | Conventional heating | No catalyst | 6–10 h | 40–55 | Organic solvent | 35–60 | 60–80 | Higher waste formation and energy consumption |
| 7 | One-pot multicomponent synthesis | Microwave irradiation | Recyclable nanoparticle catalyst | 10–25 min | 88–96 | Solvent-free/green solvent | 6–12 | 10–20 | Excellent sustainability performance with minimal waste |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kasi, V.; Jeleń, M.; Chu, X.-H.; Karthikeyan, P.; Młodawska, B.M.; Tey, L.-H. Nanoparticle-Catalysed Microwave-Driven MCRs for Sustainable Heterocycle Synthesis. Molecules 2026, 31, 1031. https://doi.org/10.3390/molecules31061031
Kasi V, Jeleń M, Chu X-H, Karthikeyan P, Młodawska BM, Tey L-H. Nanoparticle-Catalysed Microwave-Driven MCRs for Sustainable Heterocycle Synthesis. Molecules. 2026; 31(6):1031. https://doi.org/10.3390/molecules31061031
Chicago/Turabian StyleKasi, Venkatesan, Malgorzata Jeleń, Xiao-Hui Chu, Parasuraman Karthikeyan, Beata Morak Młodawska, and Lai-Hock Tey. 2026. "Nanoparticle-Catalysed Microwave-Driven MCRs for Sustainable Heterocycle Synthesis" Molecules 31, no. 6: 1031. https://doi.org/10.3390/molecules31061031
APA StyleKasi, V., Jeleń, M., Chu, X.-H., Karthikeyan, P., Młodawska, B. M., & Tey, L.-H. (2026). Nanoparticle-Catalysed Microwave-Driven MCRs for Sustainable Heterocycle Synthesis. Molecules, 31(6), 1031. https://doi.org/10.3390/molecules31061031








