Controllable Room-Temperature Synthesis of Highly Stable CsPbBr3 Perovskite Quantum Dots via Synergistic Optimization of Br/Pb and OA/OAm Ratios
Abstract
1. Introduction
2. Results and Discussion
2.1. The Effect of the Cs-Pb Ratio on Quantum Dot Properties
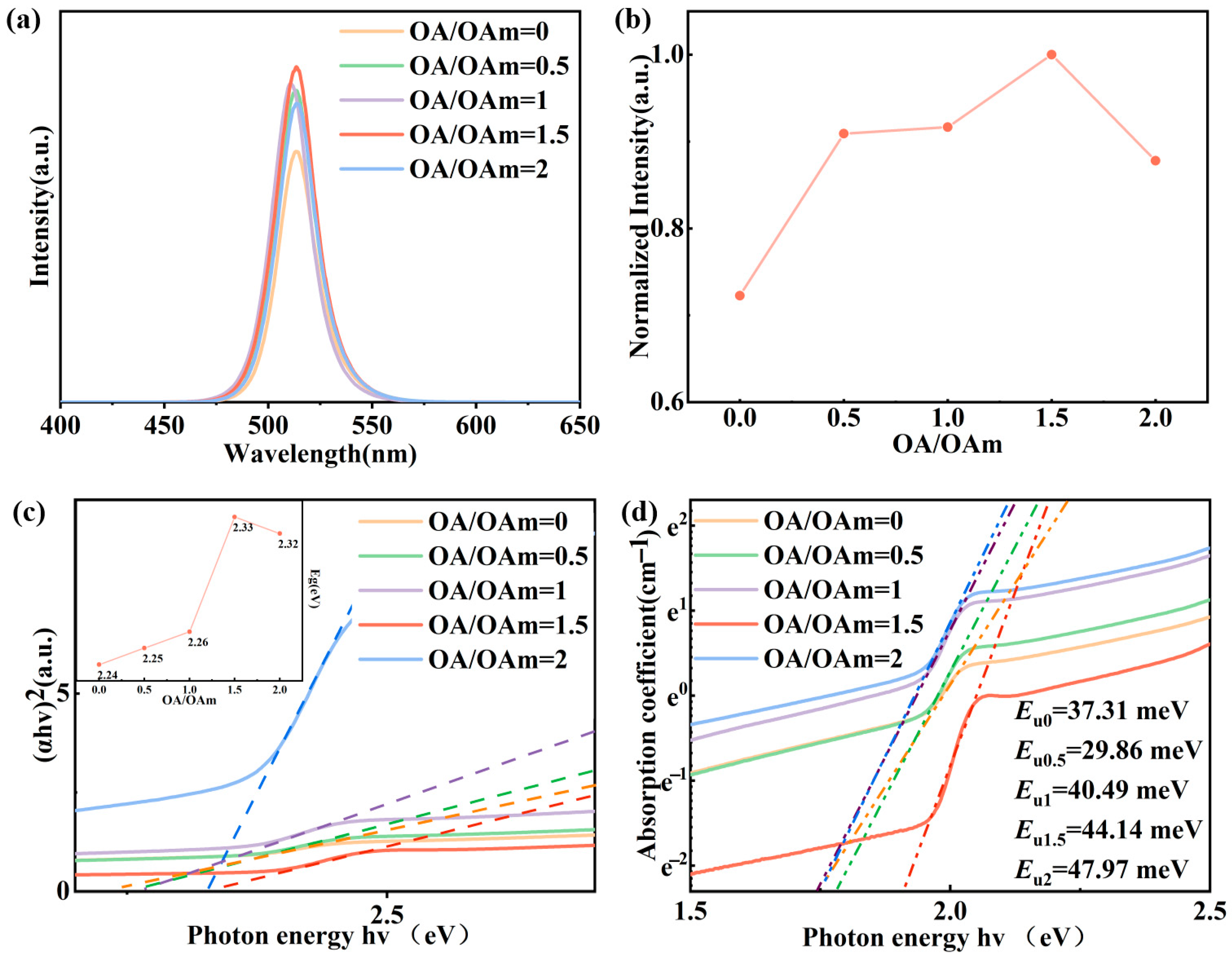
2.2. Effect of OA to OAm Ratio on Quantum Dot Properties
2.3. Applications of the QDs in LED
3. Experimental Section
3.1. Raw Materials
3.2. Synthesis of CsPbBr3 QDs
3.3. QDs Characterization
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ye, J.; Gaur, D.; Mi, C.; Chen, Z.; Fernández, I.L.; Zhao, H.; Dong, Y.; Polavarapu, L.; Hoye, R.L.Z. Strongly-Confined Colloidal Lead-Halide Perovskite Quantum Dots: From Synthesis to Applications. Chem. Soc. Rev. 2024, 53, 8095–8122. [Google Scholar] [CrossRef]
- Suresh, S.; Subramaniam, M.R.; Batabyal, S.K. Improved Photoluminescence Stability and Defect Passivation in SbBr3 Post-treated CsPbBr3 Quantum Dots under Ambient Conditions. Luminescence 2024, 39, e4706. [Google Scholar] [CrossRef]
- Wei, S.; Hu, J.; Bi, C.; Ren, K.; Wang, X.; De De Leeuw, N.H.; Lu, Y.; Sui, M.; Wang, W. Strongly-Confined CsPbBr3 Perovskite Quantum Dots with Ultralow Trap Density and Narrow Size Distribution for Efficient Pure-Blue Light-Emitting Diodes. Small 2024, 20, 2400885. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-Y.; Li, H.; Wu, Y.; Lin, C.-H.; Guan, X.; Hu, L.; Kim, J.; Zhu, X.; Zeng, H.; Wu, T. Inorganic Halide Perovskite Quantum Dots: A Versatile Nanomaterial Platform for Electronic Applications. Nano-Micro Lett. 2023, 15, 16. [Google Scholar] [CrossRef]
- Wang, C.; Cai, J.; Ye, Y.; Hu, X.; Zhong, L.; Xie, H.; Chen, E.; Ye, Y.; Xu, S.; Sun, J.; et al. Full-Visible-Spectrum Perovskite Quantum Dots by Anion Exchange Resin Assisted Synthesis. Nanophotonics 2022, 11, 1355–1366. [Google Scholar] [CrossRef] [PubMed]
- Salih Ajaj, C.; Sadiq, D. Enhancement of Optical Properties and Stability in CsPbBr3 Using CQD and TOP Doping for Solar Cell Applications. J. Nanotechnol. 2024, 2024, 5555895. [Google Scholar] [CrossRef]
- Asghar, K.; Ngulimi, M.F.; Kim, S.; Seo, B.K.; Bertrand, G.H.V.; Roh, C. State of the Art Challenges and Prospects of Advanced Materials in Radiation Detection for Nuclear Energy: A Review. Progress. Mater. Sci. 2026, 158, 101616. [Google Scholar] [CrossRef]
- Li, K.; Li, W.; Nie, Q.; Luo, X. Innovations in Scintillator Materials for X-Ray Detection. Inorg. Chem. Front. 2025, 12, 4741–4768. [Google Scholar] [CrossRef]
- Ogunleye, A.M.; Adeshina, M.A.; Kim, G.; Kim, H.; Park, J. Bright Prospects, Lingering Challenges: CsPbBr3 Quantum Dots for Environmental Sensing. Cryst. Growth Des. 2025, 25, 3238–3252. [Google Scholar] [CrossRef]
- Gao, G.; Xi, Q.; Zhou, H.; Zhao, Y.; Wu, C.; Wang, L.; Guo, P.; Xu, J. Novel Inorganic Perovskite Quantum Dots for Photocatalysis. Nanoscale 2017, 9, 12032–12038. [Google Scholar] [CrossRef]
- Hirotsu, S.; Harada, J.; Iizumi, M.; Gesi, K. Structural Phase Transitions in CsPbBr3. J. Phys. Soc. Jpn. 1974, 37, 1393–1398. [Google Scholar] [CrossRef]
- Li, C.; Lu, X.; Ding, W.; Feng, L.; Gao, Y.; Guo, Z. Formability of ABX3 (X = F, Cl, Br, I) Halide Perovskites. Acta Crystallogr. B Struct. Sci. 2008, 64, 702–707. [Google Scholar] [CrossRef]
- Lee, J.-W.; Tan, S.; Seok, S.I.; Yang, Y.; Park, N.-G. Rethinking the A Cation in Halide Perovskites. Science 2022, 375, eabj1186. [Google Scholar] [CrossRef]
- Ko, Y.-H.; Prabhakaran, P.; Jalalah, M.; Lee, S.-J.; Lee, K.-S.; Park, J.-G. Correlating Nano Black Spots and Optical Stability in Mixed Halide Perovskite Quantum Dots. J. Mater. Chem. C 2018, 6, 7803–7813. [Google Scholar] [CrossRef]
- Kim, D.; Yun, T.; An, S.; Lee, C.-L. How to Improve the Structural Stabilities of Halide Perovskite Quantum Dots: Review of Various Strategies to Enhance the Structural Stabilities of Halide Perovskite Quantum Dots. Nano Converg. 2024, 11, 4. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Debroye, E.; Caliandro, G.; Janssen, K.P.F.; Van Loon, J.; Kirschhock, C.E.A.; Martens, J.A.; Hofkens, J.; Roeffaers, M.B.J. Photoluminescence Blinking of Single-Crystal Methylammonium Lead Iodide Perovskite Nanorods Induced by Surface Traps. ACS Omega 2016, 1, 148–159. [Google Scholar] [CrossRef]
- Chouhan, L.; Ito, S.; Thomas, E.M.; Takano, Y.; Ghimire, S.; Miyasaka, H.; Biju, V. Real-Time Blinking Suppression of Perovskite Quantum Dots by Halide Vacancy Filling. ACS Nano 2021, 15, 2831–2838. [Google Scholar] [CrossRef]
- Shen, J.; Zhu, Q. Stability Strategies of Perovskite Quantum Dots and Their Extended Applications in Extreme Environment: A Review. Mater. Res. Bull. 2022, 156, 111987. [Google Scholar] [CrossRef]
- Wei, Y.; Cheng, Z.; Lin, J. An Overview on Enhancing the Stability of Lead Halide Perovskite Quantum Dots and Their Applications in Phosphor-Converted LEDs. Chem. Soc. Rev. 2019, 48, 310–350. [Google Scholar] [CrossRef]
- Park, Y.; Nishikubo, R.; Pylnev, M.; Shimomura, R.; Saeki, A. Trivalent Metal Chloride Doping for Interfacial Passivation and Enhanced Charge Transfer in Wide Bandgap Perovskite Solar Cells. ACS Appl. Energy Mater. 2024, 7, 11818–11826. [Google Scholar] [CrossRef]
- Jiang, Q.; Zhao, Y.; Zhang, X.; Yang, X.; Chen, Y.; Chu, Z.; Ye, Q.; Li, X.; Yin, Z.; You, J. Surface Passivation of Perovskite Film for Efficient Solar Cells. Nat. Photonics 2019, 13, 460–466. [Google Scholar] [CrossRef]
- Khvichia, M.; Zeitz, D.C.; Chou, K.-C.; Ping, Y.; Zhang, J.Z. Temperature Dependence of Photoinduced Carrier Spin Relaxation Dynamics in CsPbBr3 and MAPbBr3 Perovskite Quantum Dots. J. Phys. Chem. Lett. 2025, 16, 5681–5686. [Google Scholar] [CrossRef]
- Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Krieg, F.; Caputo, R.; Hendon, C.H.; Yang, R.X.; Walsh, A.; Kovalenko, M.V. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Lett. 2015, 15, 3692–3696. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, L.C.; Pertegás, A.; González-Carrero, S.; Malinkiewicz, O.; Agouram, S.; Mínguez Espallargas, G.; Bolink, H.J.; Galian, R.E.; Pérez-Prieto, J. Nontemplate Synthesis of CH3NH3PbBr3 Perovskite Nanoparticles. J. Am. Chem. Soc. 2014, 136, 850–853. [Google Scholar] [CrossRef]
- Li, X.; Wu, Y.; Zhang, S.; Cai, B.; Gu, Y.; Song, J.; Zeng, H. CsPbX3 Quantum Dots for Lighting and Displays: Room-Temperature Synthesis, Photoluminescence Superiorities, Underlying Origins and White Light-Emitting Diodes. Adv. Funct. Mater. 2016, 26, 2435–2445. [Google Scholar] [CrossRef]
- Jana, A.; Mittal, M.; Singla, A.; Sapra, S. Solvent-Free, Mechanochemical Syntheses of Bulk Trihalide Perovskites and Their Nanoparticles. Chem. Commun. 2017, 53, 3046–3049. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Hu, Z.; Cong, P.; Zhou, F.; Yue, Q.; Xue, Z.; Lin, C.; Jiang, Y.; Du, J.; Pan, A.; et al. Spatial and Chemical Dual Nano-Confined Ultrastable Perovskite Quantum Dots Glass Manifesting Exciton Modulation. Adv. Opt. Mater. 2024, 12, 2400630. [Google Scholar] [CrossRef]
- Hassanabadi, E.; Latifi, M.; Gualdrón-Reyes, A.F.; Masi, S.; Yoon, S.J.; Poyatos, M.; Julián-López, B.; Mora-Seró, I. Ligand & Band Gap Engineering: Tailoring the Protocol Synthesis for Achieving High-Quality CsPbI3 Quantum Dots. Nanoscale 2020, 12, 14194–14203. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, X.; Zhu, C.; Yu, Q. Advancing the Synthesis Strategy and Interface Modification for Efficient Perovskite Quantum Dot Solar Cells. J. Mater. Chem. A 2025, 13, 29688–29705. [Google Scholar] [CrossRef]
- Wang, L.; Ma, D.; Guo, C.; Jiang, X.; Li, M.; Xu, T.; Zhu, J.; Fan, B.; Liu, W.; Shao, G.; et al. CsPbBr3 Nanocrystals Prepared by High Energy Ball Milling in One-Step and Structural Transformation from CsPbBr3 to CsPb2Br5. Appl. Surf. Sci. 2021, 543, 148782. [Google Scholar] [CrossRef]
- Zhang, F.; Chen, J.; Zhou, Y.; He, R.; Zheng, K. Effect of Synthesis Methods on Photoluminescent Properties for CsPbBr3 Nanocrystals: Hot Injection Method and Conversion Method. J. Lumin. 2020, 220, 117023. [Google Scholar] [CrossRef]
- Souza, G.F.D.; Menon, L.; Izumi, C.M.S.; Ludwig, Z.M.C. Challenges and Perspectives in the Use of Perovskite Nanocrystals in Solar Cell. IOP Conf. Ser. Earth Environ. Sci. 2025, 1536, 012009. [Google Scholar] [CrossRef]
- Deng, J.; Wang, H.; Xun, J.; Wang, J.; Yang, X.; Shen, W.; Li, M.; He, R. Room-Temperature Synthesis of Excellent-Performance CsPb1−xSnxBr3 Perovskite Quantum Dots and Application in Light Emitting Diodes. Mater. Des. 2020, 185, 108246. [Google Scholar] [CrossRef]
- Mojed, A.; Ashiri, R.; Shafyei, A. A Mechanistic Study on Efficient Room Temperature Sonocolloidal Obtainment of SrTiO3 Nanocrystals. Ultrason. Sonochem. 2023, 99, 106568. [Google Scholar] [CrossRef] [PubMed]
- Soosaimanickam, A.; Saura, A.; Farinós, N.; Abargues, R. Influence of Binary Ligands in Designing Cesium Lead Halide (CsPbX3, X = Cl, Br, I) Perovskite Nanocrystals-Oleic Acid and Oleylamine. Nanoenergy Adv. 2023, 3, 376–400. [Google Scholar] [CrossRef]
- Nair, G.B.; Tamboli, S.; Dhoble, S.J.; Swart, H.C. Striking the Balance between the Capping Ligands for CsPbBr3 Nanocrystal Synthesis via an Emulsion LARP Approach. Phys. Chem. Chem. Phys. 2025, 27, 12886–12898. [Google Scholar] [CrossRef]
- Atteberry, M.L.; Mi, C.; Chandra, S.; Fiaz, S.; Dong, Y. Synthesis, Properties, and Applications of Morphology-Controlled Perovskite Quantum Dots. ACS Appl. Opt. Mater. 2026, 4, 278–292. [Google Scholar] [CrossRef]
- Chen, W.; Hu, L.; Wang, Y.; Huang, L.; Wang, Z.; Tang, X. Surface Passivation Strategies for CsPbBr3 Quantum Dots Aiming at Nonradiative Suppression and Enhancement of Electroluminescent Light-Emitting Diodes. Dalton Trans. 2025, 54, 2156–2165. [Google Scholar] [CrossRef]
- Kim, J.; Kim, W.; Jang, J.I.; Kim, W.; Yoo, D.; Kim, J.W.; Lee, Y.; Choi, M.-J.; Choi, J.; Kim, H.M.; et al. SN2-Mediated Decoupled Precursor Provision Enables Large-Scale Production of Monodisperse Lead Halide Perovskite Quantum Dots in a Single Reactor. Adv. Compos. Hybrid. Mater. 2025, 8, 125. [Google Scholar] [CrossRef]
- Boehme, S.C.; Bodnarchuk, M.I.; Burian, M.; Bertolotti, F.; Cherniukh, I.; Bernasconi, C.; Zhu, C.; Erni, R.; Amenitsch, H.; Naumenko, D.; et al. Strongly Confined CsPbBr3 Quantum Dots as Quantum Emitters and Building Blocks for Rhombic Superlattices. ACS Nano 2023, 17, 2089–2100. [Google Scholar] [CrossRef] [PubMed]
- Cheng, O.H.-C.; Qiao, T.; Sheldon, M.; Son, D.H. Size- and Temperature-Dependent Photoluminescence Spectra of Strongly Confined CsPbBr3 Quantum Dots. Nanoscale 2020, 12, 13113–13118. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Di Stasio, F.; Dang, Z.; Canale, C.; Khan, A.H.; Shamsi, J.; Brescia, R.; Prato, M.; Manna, L. Colloidal Synthesis of Strongly Fluorescent CsPbBr3 Nanowires with Width Tunable down to the Quantum Confinement Regime. Chem. Mater. 2016, 28, 6450–6454. [Google Scholar] [CrossRef]
- Cho, S.; Noh, S.K.; Kim, H.; Im, H. Distinctive Spectroscopic Characteristics Observed in Colloidal Perovskite CsPbBr3 Quantum Dot. Appl. Sci. Converg. Technol. 2021, 30, 107–110. [Google Scholar] [CrossRef]
- Nuber, M.; Tan, Q.Y.; Sandner, D.; Yin, J.; Kienberger, R.; Soci, C.; Iglev, H. Accelerated Polaron Formation in Perovskite Quantum Dots Monitored via Picosecond Infrared Spectroscopy. J. Mater. Chem. C 2023, 11, 3581–3587. [Google Scholar] [CrossRef]
- Ravi, V.K.; Santra, P.K.; Joshi, N.; Chugh, J.; Singh, S.K.; Rensmo, H.; Ghosh, P.; Nag, A. Origin of the Substitution Mechanism for the Binding of Organic Ligands on the Surface of CsPbBr3 Perovskite Nanocubes. J. Phys. Chem. Lett. 2017, 8, 4988–4994. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Jia, X.; Nie, Y.; Zhu, Y.; Ye, H. The Thermodynamical and Optical Properties of Surface Bromine Vacancy in Two-Dimensional CsPbBr3: A First Principles Study. Appl. Surf. Sci. 2022, 584, 152626. [Google Scholar] [CrossRef]
- Lin, Z.; Song, J.; Wu, H.; Li, H.; Huang, R. Mechanistic Identification of Oxygen Species in the Degradation of CsPbBr3 Quantum Dot Films Through Real-Time In Situ Monitoring. Materials 2025, 18, 5467. [Google Scholar] [CrossRef]
- Zheng, C.; Bi, C.; Huang, F.; Binks, D.; Tian, J. Stable and Strong Emission CsPbBr3 Quantum Dots by Surface Engineering for High-Performance Optoelectronic Films. ACS Appl. Mater. Interfaces 2019, 11, 25410–25416. [Google Scholar] [CrossRef]
- Zu, Y.; Zhang, T.; Jing, L.; Gao, H.; Wang, W.; Yang, K.; Zeng, B.; Gan, R.; Gong, D.; Liu, P.; et al. Bifunctional Ligand Passivation Enables Robust-Stability Perovskite Nanocrystals for Backlit Display. Dye. Pigment. 2025, 242, 112983. [Google Scholar] [CrossRef]
- Wu, R.; Yue, Y.; Zhang, A.; Wei, J.; Liu, F.; Li, H. Unveiling the Doping Effect of Mixed-Halide CsPb(Br1−nXn)3 (X = I, Cl) Single Crystals toward High-Sensitivity Radiation Detection. Nanoscale 2025, 17, 18843–18851. [Google Scholar] [CrossRef]
- Zhao, Y.; Yang, R.; Wan, W.; Jing, X.; Wen, T.; Ye, S. Stabilizing CsPbBr3 Quantum Dots with Conjugated Aromatic Ligands and Their Regulated Optical Behaviors. Chem. Eng. J. 2020, 389, 124453. [Google Scholar] [CrossRef]
- Cai, H.; Sun, Y.; Zhang, X.; Zhang, L.; Liu, H.; Li, Q.; Bo, T.; Zhou, D.; Wang, C.; Lian, J. Reduction Temperature-Dependent Nanoscale Morphological Transformation and Electrical Conductivity of Silicate Glass Microchannel Plate. Materials 2019, 12, 1183. [Google Scholar] [CrossRef] [PubMed]
- Hills-Kimball, K.; Yang, H.; Cai, T.; Wang, J.; Chen, O. Recent Advances in Ligand Design and Engineering in Lead Halide Perovskite Nanocrystals. Adv. Sci. 2021, 8, 2100214. [Google Scholar] [CrossRef] [PubMed]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
He, Y.; Zhu, X.; Li, A.; Lin, S.; Li, B.; Liu, S.; Ye, X. Controllable Room-Temperature Synthesis of Highly Stable CsPbBr3 Perovskite Quantum Dots via Synergistic Optimization of Br/Pb and OA/OAm Ratios. Molecules 2026, 31, 1006. https://doi.org/10.3390/molecules31061006
He Y, Zhu X, Li A, Lin S, Li B, Liu S, Ye X. Controllable Room-Temperature Synthesis of Highly Stable CsPbBr3 Perovskite Quantum Dots via Synergistic Optimization of Br/Pb and OA/OAm Ratios. Molecules. 2026; 31(6):1006. https://doi.org/10.3390/molecules31061006
Chicago/Turabian StyleHe, Yiting, Xiayu Zhu, Ajun Li, Shuyuan Lin, Bo Li, Songbin Liu, and Xinyu Ye. 2026. "Controllable Room-Temperature Synthesis of Highly Stable CsPbBr3 Perovskite Quantum Dots via Synergistic Optimization of Br/Pb and OA/OAm Ratios" Molecules 31, no. 6: 1006. https://doi.org/10.3390/molecules31061006
APA StyleHe, Y., Zhu, X., Li, A., Lin, S., Li, B., Liu, S., & Ye, X. (2026). Controllable Room-Temperature Synthesis of Highly Stable CsPbBr3 Perovskite Quantum Dots via Synergistic Optimization of Br/Pb and OA/OAm Ratios. Molecules, 31(6), 1006. https://doi.org/10.3390/molecules31061006




