β-Glucuronidase at the Microbiota—Host Interface: Dual Regulatory Roles and Precision Modulation by Natural Products
Abstract
1. Introduction
2. GUS Overview
2.1. GUS Structure and Classification
2.2. Diversity and Functional Specialization of GUS
3. GUS Mediates Host Health and Disease
3.1. A Central Role for GUS in Systemic Homeostasis
3.2. GUS Mediated Drug Metabolism and Toxicity
4. Modulating the GUS–Microbiota Axis with Natural Products
4.1. Inhibitors Derived from Natural Products Targeting GUS
4.1.1. Flavonoid Inhibitors
4.1.2. Natural Amide Inhibitors
4.1.3. Tannin Inhibitors
4.1.4. Catechin and Theaflavin Inhibitors
4.1.5. Other Inhibitors
4.2. Modulation of GUS Activity by Natural Product-Induced Microbial Changes
4.3. GUS-Mediated Biotransformation of Natural Products
5. Natural Product Interactions with GUS in Health and Disease
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Baker, J.M.; Al-Nakkash, L.; Herbst-Kralovetz, M.M. Estrogen-gut microbiome axis: Physiological and clinical implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef]
- Zhou, C.B.; Zhou, Y.L.; Fang, J.Y. Gut Microbiota in Cancer Immune Response and Immunotherapy. Trends Cancer 2021, 7, 647–660. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Cong, Y. Gut microbiota-derived metabolites in the regulation of host immune responses and immune-related inflammatory diseases. Cell Mol. Immunol. 2021, 18, 866–877. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Yu, T.; Huang, X.; Bilotta, A.J.; Xu, L.; Lu, Y.; Sun, J.; Pan, F.; Zhou, J.; Zhang, W.; et al. Intestinal microbiota-derived short-chain fatty acids regulation of immune cell IL-22 production and gut immunity. Nat. Commun. 2020, 11, 4457. [Google Scholar] [CrossRef] [PubMed]
- Rawat, P.S.; Seyed Hameed, A.S.; Meng, X.; Liu, W. Utilization of glycosaminoglycans by the human gut microbiota: Participating bacteria and their enzymatic machineries. Gut Microbes 2022, 14, 2068367. [Google Scholar] [CrossRef]
- Weersma, R.K.; Zhernakova, A.; Fu, J. Interaction between drugs and the gut microbiome. Gut 2020, 69, 1510–1519. [Google Scholar] [CrossRef]
- Odell, L.D.; Fishman, W.H.; Hepner, W.R. Beta-Glucuronidase. Science 1948, 108, 355–356. [Google Scholar] [CrossRef]
- Järvinen, E.; Deng, F.; Kiander, W.; Sinokki, A.; Kidron, H.; Sjöstedt, N. The Role of Uptake and Efflux Transporters in the Disposition of Glucuronide and Sulfate Conjugates. Front. Pharmacol. 2021, 12, 802539. [Google Scholar] [CrossRef]
- Awolade, P.; Cele, N.; Kerru, N.; Gummidi, L.; Oluwakemi, E.; Singh, P. Therapeutic significance of β-glucuronidase activity and its inhibitors: A review. Eur. J. Med. Chem. 2020, 187, 111921. [Google Scholar] [CrossRef]
- Gloux, K.; Berteau, O.; El Oumami, H.; Béguet, F.; Leclerc, M.; Doré, J. A metagenomic β-glucuronidase uncovers a core adaptive function of the human intestinal microbiome. Proc. Natl. Acad. Sci. USA 2011, 108, 4539–4546. [Google Scholar] [CrossRef]
- Gao, S.; Sun, R.; Singh, R.; Yu So, S.; Chan, C.T.Y.; Savidge, T.; Hu, M. The role of gut microbial β-glucuronidase in drug disposition and development. Drug Discov. Today 2022, 27, 103316. [Google Scholar] [CrossRef] [PubMed]
- Fishman, W.H.; Mitchell, G.W., Jr.; Dimitrakis, H.; Hayashi, M. Enzymorphology of adenocarcinoma of the endometrium; beta-glucuronidase, acid phosphatase, reduced diphosphopyridine nucleotide (DPNH) diaphorase, and alpha-naphthyl esterase. Cancer 1963, 16, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Sui, Y.; Wu, J.; Chen, J. The Role of Gut Microbial β-Glucuronidase in Estrogen Reactivation and Breast Cancer. Front. Cell Dev. Biol. 2021, 9, 631552. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, A.P.; Pellock, S.J.; Biernat, K.A.; Walton, W.G.; Wallace, B.D.; Creekmore, B.C.; Letertre, M.M.; Swann, J.R.; Wilson, I.D.; Roques, J.R.; et al. Targeted inhibition of gut bacterial β-glucuronidase activity enhances anticancer drug efficacy. Proc. Natl. Acad. Sci. USA 2020, 117, 7374–7381. [Google Scholar] [CrossRef]
- Zhang, L.; Song, J.; Kong, L.; Yuan, T.; Li, W.; Zhang, W.; Hou, B.; Lu, Y.; Du, G. The strategies and techniques of drug discovery from natural products. Pharmacol. Ther. 2020, 216, 107686. [Google Scholar] [CrossRef]
- Wang, C.; Teng, X.; Wang, C.; Liu, B.; Zhou, R.; Xu, X.; Qiu, H.; Fu, Y.; Sun, R.; Liang, Z.; et al. Insight into the mechanism of Xiao-Chai-Hu-Tang alleviates irinotecan-induced diarrhea based on regulating the gut microbiota and inhibiting Gut β-GUS. Phytomedicine 2023, 120, 155040. [Google Scholar] [CrossRef]
- Fishman, W.H. Studies on β-Glucuronidase: I. A Method of Preparation and Purification. J. Biol. Chem. 1939, 127, 367–373. [Google Scholar] [CrossRef]
- Jain, S.; Drendel, W.B.; Chen, Z.W.; Mathews, F.S.; Sly, W.S.; Grubb, J.H. Structure of human beta-glucuronidase reveals candidate lysosomal targeting and active-site motifs. Nat. Struct. Mol. Biol. 1996, 3, 375–381. [Google Scholar] [CrossRef]
- Wallace, B.D.; Wang, H.; Lane, K.T.; Scott, J.E.; Orans, J.; Koo, J.S.; Venkatesh, M.; Jobin, C.; Yeh, L.A.; Mani, S.; et al. Alleviating cancer drug toxicity by inhibiting a bacterial enzyme. Science 2010, 330, 831–835. [Google Scholar] [CrossRef]
- Sun, C.P.; Yan, J.K.; Yi, J.; Zhang, X.Y.; Yu, Z.L.; Huo, X.K.; Liang, J.H.; Ning, J.; Feng, L.; Wang, C.; et al. The study of inhibitory effect of natural flavonoids toward β-glucuronidase and interaction of flavonoids with β-glucuronidase. Int. J. Biol. Macromol. 2020, 143, 349–358. [Google Scholar] [CrossRef]
- Pollet, R.M.; D’Agostino, E.H.; Walton, W.G.; Xu, Y.; Little, M.S.; Biernat, K.A.; Pellock, S.J.; Patterson, L.M.; Creekmore, B.C.; Isenberg, H.N.; et al. An Atlas of β-Glucuronidases in the Human Intestinal Microbiome. Structure 2017, 25, 967–977.e5. [Google Scholar] [CrossRef] [PubMed]
- Creekmore, B.C.; Gray, J.H.; Walton, W.G.; Biernat, K.A.; Little, M.S.; Xu, Y.; Liu, J.; Gharaibeh, R.Z.; Redinbo, M.R. Mouse Gut Microbiome-Encoded β-Glucuronidases Identified Using Metagenome Analysis Guided by Protein Structure. mSystems 2019, 4, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Pellock, S.J.; Walton, W.G.; Ervin, S.M.; Torres-Rivera, D.; Creekmore, B.C.; Bergan, G.; Dunn, Z.D.; Li, B.; Tripathy, A.; Redinbo, M.R. Discovery and Characterization of FMN-Binding β-Glucuronidases in the Human Gut Microbiome. J. Mol. Biol. 2019, 431, 970–980. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.B.; Walker, M.E.; Sekela, J.J.; Ivey, S.M.; Jariwala, P.B.; Storch, C.M.; Kowalewski, M.E.; Graboski, A.L.; Lietzan, A.D.; Walton, W.G.; et al. Gut microbial β-glucuronidases influence endobiotic homeostasis and are modulated by diverse therapeutics. Cell Host Microbe 2024, 32, 925–944.e910. [Google Scholar] [CrossRef]
- Pellock, S.J.; Walton, W.G.; Biernat, K.A.; Torres-Rivera, D.; Creekmore, B.C.; Xu, Y.; Liu, J.; Tripathy, A.; Stewart, L.J.; Redinbo, M.R. Three structurally and functionally distinct β-glucuronidases from the human gut microbe Bacteroides uniformis. J. Biol. Chem. 2018, 293, 18559–18573. [Google Scholar] [CrossRef]
- Pellock, S.J.; Redinbo, M.R. Glucuronides in the gut: Sugar-driven symbioses between microbe and host. J. Biol. Chem. 2017, 292, 8569–8576. [Google Scholar] [CrossRef]
- Candeliere, F.; Raimondi, S.; Ranieri, R.; Musmeci, E.; Zambon, A.; Amaretti, A.; Rossi, M. β-Glucuronidase Pattern Predicted From Gut Metagenomes Indicates Potentially Diversified Pharmacomicrobiomics. Front. Microbiol. 2022, 13, 826994. [Google Scholar] [CrossRef]
- Wallace, B.D.; Roberts, A.B.; Pollet, R.M.; Ingle, J.D.; Biernat, K.A.; Pellock, S.J.; Venkatesh, M.K.; Guthrie, L.; O’Neal, S.K.; Robinson, S.J.; et al. Structure and Inhibition of Microbiome β-Glucuronidases Essential to the Alleviation of Cancer Drug Toxicity. Chem. Biol. 2015, 22, 1238–1249. [Google Scholar] [CrossRef]
- Miners, J.O.; Polasek, T.M.; Hulin, J.A.; Rowland, A.; Meech, R. Drug-drug interactions that alter the exposure of glucuronidated drugs: Scope, UDP-glucuronosyltransferase (UGT) enzyme selectivity, mechanisms (inhibition and induction), and clinical significance. Pharmacol. Ther. 2023, 248, 108459. [Google Scholar] [CrossRef]
- Fernández-Murga, M.L.; Gil-Ortiz, F.; Serrano-García, L.; Llombart-Cussac, A. A New Paradigm in the Relationship between Gut Microbiota and Breast Cancer: β-glucuronidase Enzyme Identified as Potential Therapeutic Target. Pathogens 2023, 12, 1086. [Google Scholar] [CrossRef]
- Cartmell, A.; Muñoz-Muñoz, J.; Briggs, J.A.; Ndeh, D.A.; Lowe, E.C.; Baslé, A.; Terrapon, N.; Stott, K.; Heunis, T.; Gray, J.; et al. A surface endogalactanase in Bacteroides thetaiotaomicron confers keystone status for arabinogalactan degradation. Nat. Microbiol. 2018, 3, 1314–1326. [Google Scholar] [CrossRef] [PubMed]
- Samar Ballabha, M.; Narayanan, M. Chapter 12—Overview of structure-function relationships of glucuronidases. In Glycoside Hydrolases; Academic Press: Cambridge, MA, USA, 2023; pp. 255–278. [Google Scholar] [CrossRef]
- Sly, W.S.; Quinton, B.A.; McAlister, W.H.; Rimoin, D.L. Beta glucuronidase deficiency: Report of clinical, radiologic, and biochemical features of a new mucopolysaccharidosis. J. Pediatr. 1973, 82, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Sakurama, H.; Kishino, S.; Uchibori, Y.; Yonejima, Y.; Ashida, H.; Kita, K.; Takahashi, S.; Ogawa, J. β-Glucuronidase from Lactobacillus brevis useful for baicalin hydrolysis belongs to glycoside hydrolase family 30. Appl. Microbiol. Biotechnol. 2014, 98, 4021–4032. [Google Scholar] [CrossRef] [PubMed]
- Hameleers, L.; Pijning, T.; Gray, B.B.; Fauré, R.; Jurak, E. Novel β-galactosidase activity and first crystal structure of Glycoside Hydrolase family 154. Nat. Biotechnol. 2024, 80, 1–11. [Google Scholar] [CrossRef]
- Lebreton, A.; Garron, M.L.; Vuillemin, M.; Pilgaard, B.; Hornung, B.V.H.; Drula, E.; Lombard, V.; Helbert, W.; Henrissat, B.; Terrapon, N. Division of the large and multifunctional glycoside hydrolase family 2: High functional specificity and biochemical assays in the uncharacterized subfamilies. Biotechnol. Biofuels Bioprod. 2025, 18, 68. [Google Scholar] [CrossRef]
- Neun, S.; Brear, P.; Campbell, E.; Tryfona, T.; El Omari, K.; Wagner, A.; Dupree, P.; Hyvönen, M.; Hollfelder, F. Functional metagenomic screening identifies an unexpected β-glucuronidase. Nat. Chem. Biol. 2022, 18, 1096–1103. [Google Scholar] [CrossRef]
- Maha, A.A.; Faris, F.A.A.; Hassan, A.R.; Ahmed, A.A.; Al Mokhtar, L.; Emadeldin, M.K. Mechanistic insights into β-glucuronidase inhibition by isoprenylated flavonoids from Centaurea scoparia: Bridging experimental and computational approaches. J. Mol. Struct. 2025, 1322, 140354. [Google Scholar] [CrossRef]
- Grant, D.M. Detoxification pathways in the liver. J. Inherit. Metab. Dis. 1991, 14, 421–430. [Google Scholar] [CrossRef]
- Trottier, J.; Verreault, M.; Grepper, S.; Monté, D.; Bélanger, J.; Kaeding, J.; Caron, P.; Inaba, T.T.; Barbier, O. Human UDP-glucuronosyltransferase (UGT)1A3 enzyme conjugates chenodeoxycholic acid in the liver. Hepatology 2006, 44, 1158–1170. [Google Scholar] [CrossRef]
- Drevland, O.M.; de Muinck, E.J.; Trosvik, P.; Hammerstad, M.; Kvitne, K.E.; Midtvedt, K.; Åsberg, A.; Robertsen, I. Microbiome-derived reactivation of mycophenolate explains variations in enterohepatic recirculation in kidney transplant recipients. Microbiome 2025, 13, 169. [Google Scholar] [CrossRef]
- Zhang, A.; Gao, S.; Shen, C.; Wu, S.; Wang, X.; Shen, Q.; Hua, J.; Wang, J.; Shi, J.; Chen, S.; et al. Bupleuri Radix polysaccharides enhance the efficacy and intestinal absorption of baicalin via regulating intestinal β-glucuronidase activity in MASH mice. Phytomedicine 2025, 145, 157092. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C. Irinotecan: 25 years of cancer treatment. Pharmacol. Res. 2019, 148, 104398. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Ye, A.; Jiang, N. The role of bacteria in gallstone formation. Folia Microbiol. 2024, 69, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, O.; Sie, C.; Ng, K.M.; Cotton, S.; Rosete, C.; Hamden, J.E.; Singh, A.P.; Lee, K.; Choudhary, J.; Kim, J.; et al. Early-life gut inflammation drives sex-dependent shifts in the microbiome-endocrine-brain axis. Brain Behav. Immun. 2025, 125, 117–139. [Google Scholar] [CrossRef]
- Hu, S.; Ding, Q.; Zhang, W.; Kang, M.; Ma, J.; Zhao, L. Gut microbial beta-glucuronidase: A vital regulator in female estrogen metabolism. Gut Microbes 2023, 15, 2236749. [Google Scholar] [CrossRef]
- Cross, T.L.; Simpson, A.M.R.; Lin, C.Y.; Hottmann, N.M.; Bhatt, A.P.; Pellock, S.J.; Nelson, E.R.; Loman, B.R.; Wallig, M.A.; Vivas, E.I.; et al. Gut microbiome responds to alteration in female sex hormone status and exacerbates metabolic dysfunction. Gut Microbes 2024, 16, 2295429. [Google Scholar] [CrossRef]
- Ervin, S.M.; Li, H.; Lim, L.; Roberts, L.R.; Liang, X.; Mani, S.; Redinbo, M.R. Gut microbial β-glucuronidases reactivate estrogens as components of the estrobolome that reactivate estrogens. J. Biol. Chem. 2019, 294, 18586–18599. [Google Scholar] [CrossRef]
- Wei, Y.; Tan, H.; Yang, R.; Yang, F.; Liu, D.; Huang, B.; OuYang, L.; Lei, S.; Wang, Z.; Jiang, S.; et al. Gut dysbiosis-derived β-glucuronidase promotes the development of endometriosis. Fertil. Steril. 2023, 120, 682–694. [Google Scholar] [CrossRef]
- Whiting, J.F.; Narciso, J.P.; Chapman, V.; Ransil, B.J.; Swank, R.T.; Gollan, J.L. Deconjugation of bilirubin-IX alpha glucuronides: A physiologic role of hepatic microsomal beta-glucuronidase. J. Biol. Chem. 1993, 268, 23197–23201. [Google Scholar] [CrossRef]
- Li, S.; Wu, W.; Zhou, Y.; Zhang, S.; Wei, D.; Zhu, M.; Ying, X.; Sun, X.; Liu, H.; Zhu, W.; et al. Gut microbiota-regulated unconjugated bilirubin metabolism drives renal calcium oxalate crystal deposition. Gut Microbes 2025, 17, 2546158. [Google Scholar] [CrossRef]
- Hall, B.; Levy, S.; Dufault-Thompson, K.; Arp, G.; Zhong, A.; Ndjite, G.M.; Weiss, A.; Braccia, D.; Jenkins, C.; Grant, M.R.; et al. BilR is a gut microbial enzyme that reduces bilirubin to urobilinogen. Nat. Microbiol. 2024, 9, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Shao, X.; Zhou, Y.; Yu, Y.; Kuai, X.; Zhou, C. Bidirectional regulation of bile acid on colorectal cancer through bile acid-gut microbiota interaction. Am. J. Transl. Res. 2021, 13, 10994–11003. [Google Scholar] [PubMed]
- Asano, Y.; Hiramoto, T.; Nishino, R.; Aiba, Y.; Kimura, T.; Yoshihara, K.; Koga, Y.; Sudo, N. Critical role of gut microbiota in the production of biologically active, free catecholamines in the gut lumen of mice. J. Physiol.-Gastrointest. Liver Physiol. 2012, 303, G1288–G1295. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lazar, M.A. The mechanism of action of thyroid hormones. Annu. Rev. Physiol. 2000, 62, 439–466. [Google Scholar] [CrossRef]
- Korenman, S.G.; Lipsett, M.B. Is Testosterone Glucuronoside Uniquely Derived from Plasma Testosterone? J. Clin. Investig. 1964, 43, 2125–2131. [Google Scholar] [CrossRef]
- Pavelková, M.; Kubová, K.; Vysloužil, J.; Kejdušová, M.; Vetchý, D.; Celer, V.; Molinková, D.; Lobová, D.; Pechová, A.; Vysloužil, J.; et al. Biological Effects of Drug-Free Alginate Beads Cross-Linked by Copper Ions Prepared Using External Ionotropic Gelation. AAPS PharmSciTech 2017, 18, 1343–1354. [Google Scholar] [CrossRef]
- Sallustio, B.C.; Sabordo, L.; Evans, A.M.; Nation, R.L. Hepatic disposition of electrophilic acyl glucuronide conjugates. Curr. Drug Metab. 2000, 1, 163–180. [Google Scholar] [CrossRef]
- Boelsterli, U.A. Xenobiotic acyl glucuronides and acyl CoA thioesters as protein-reactive metabolites with the potential to cause idiosyncratic drug reactions. Curr. Drug Metab. 2002, 3, 439–450. [Google Scholar] [CrossRef]
- Sabolovic, N.; Heydel, J.M.; Li, X.; Little, J.M.; Humbert, A.C.; Radominska-Pandya, A.; Magdalou, J. Carboxyl nonsteroidal anti-inflammatory drugs are efficiently glucuronidated by microsomes of the human gastrointestinal tract. Biochim. Biophys. Acta 2004, 1675, 120–129. [Google Scholar] [CrossRef]
- Mano, N.; Goto, T.; Nikaido, A.; Narui, T.; Goto, J. Inhibition of the rat hepatic microsomal flurbiprofen acyl glucuronidation by bile acids. J. Pharm. Sci. 2003, 92, 2098–2108. [Google Scholar] [CrossRef]
- Elmassry, M.M.; Kim, S.; Busby, B. Predicting drug-metagenome interactions: Variation in the microbial β-glucuronidase level in the human gut metagenomes. PLoS ONE 2021, 16, e0244876. [Google Scholar] [CrossRef]
- Simpson, J.B.; Sekela, J.J.; Graboski, A.L.; Borlandelli, V.B.; Bivins, M.M.; Barker, N.K.; Sorgen, A.A.; Mordant, A.L.; Johnson, R.L.; Bhatt, A.P.; et al. Metagenomics combined with activity-based proteomics point to gut bacterial enzymes that reactivate mycophenolate. Gut Microbes 2022, 14, 2107289. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.R.; Flannigan, K.L.; Rahim, H.; Mohamud, A.; Lewis, I.A.; Hirota, S.A.; Greenway, S.C. Vancomycin relieves mycophenolate mofetil-induced gastrointestinal toxicity by eliminating gut bacterial β-glucuronidase activity. Sci. Adv. 2019, 5, eaax2358. [Google Scholar] [CrossRef] [PubMed]
- Chamseddine, A.N.; Ducreux, M.; Armand, J.P.; Paoletti, X.; Satar, T.; Paci, A.; Mir, O. Intestinal bacterial β-glucuronidase as a possible predictive biomarker of irinotecan-induced diarrhea severity. Pharmacol. Ther. 2019, 199, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.; Gao, R.; Zhao, L.; Liu, D.; Lv, C.; Wang, Z.; Ai, F.; Zhang, B.; Yu, Z.; Geng, X.; et al. β-Glucuronidase-Expressing Lactobacillus reuteri Triggers Irinotecan Enterotoxicity Through Depleting the Regenerative Epithelial Stem/Progenitor Pool. Adv. Sci. 2025, 12, 2411052. [Google Scholar] [CrossRef]
- Wang, P.; Jia, Y.; Wu, R.; Chen, Z.; Yan, R. Human gut bacterial β-glucuronidase inhibition: An emerging approach to manage medication therapy. Biochem. Pharmacol. 2021, 190, 114566. [Google Scholar] [CrossRef]
- Harvey, A.L.; Edrada-Ebel, R.; Quinn, R.J. The re-emergence of natural products for drug discovery in the genomics era. Nat. Rev. Drug Discov. 2015, 14, 111–129. [Google Scholar] [CrossRef]
- Kamel, E.M.; Maodaa, S.; Al-Shaebi, E.M.; Mokhtar Lamsabhi, A. Molecular Insights Into β-Glucuronidase Inhibition by Alhagi Graecorum Flavonoids: A Computational and Experimental Approach. ChemistryOpen 2025, 14, e202400325. [Google Scholar] [CrossRef]
- Tian, X.G.; Yan, J.K.; Sun, C.P.; Li, J.X.; Ning, J.; Wang, C.; Huo, X.K.; Zhao, W.Y.; Yu, Z.L.; Feng, L.; et al. Amentoflavone from Selaginella tamariscina as a potent inhibitor of gut bacterial β-glucuronidase: Inhibition kinetics and molecular dynamics stimulation. Chem. Biol. Interact. 2021, 340, 109453. [Google Scholar] [CrossRef]
- Weng, Z.M.; Wang, P.; Ge, G.B.; Dai, Z.R.; Wu, D.C.; Zou, L.W.; Dou, T.Y.; Zhang, T.Y.; Yang, L.; Hou, J. Structure-activity relationships of flavonoids as natural inhibitors against E. coli β-glucuronidase. Food Chem. Toxicol. 2017, 109, 975–983. [Google Scholar] [CrossRef]
- Kamel, E.M.; Alkhayl, F.F.A.; Alqhtani, H.A.; Bin-Jumah, M.; Rudayni, H.A.; Lamsabhi, A.M. Dissecting molecular mechanisms underlying the inhibition of β-glucuronidase by alkaloids from Hibiscus trionum: Integrating in vitro and in silico perspectives. Comput. Biol. Med. 2024, 180, 108969. [Google Scholar] [CrossRef]
- Kamel, E.M.; Alqhtani, H.A.; Bin-Jumah, M.; Rudayni, H.A.; El-Bassuony, A.A.; Mokhtar Lamsabhi, A. Deciphering molecular mechanisms underlying the inhibition of β-glucuronidase by xanthones from Centaurium spicatum. Bioorg Chem. 2024, 150, 107609. [Google Scholar] [CrossRef]
- Bai, Y.; Chen, L.; Cao, Y.F.; Hou, X.D.; Jia, S.N.; Zhou, Q.; He, Y.Q.; Hou, J. Beta-Glucuronidase Inhibition by Constituents of Mulberry Bark. Planta Med. 2021, 87, 631–641. [Google Scholar] [CrossRef]
- Bai, Y.; Chen, L.; Wang, P.P.; Tang, Y.Q.; Wu, D.C.; Zhang, C.L.; Zhou, Q.; Yan, R.; Hou, J. Discovery of a naturally occurring broad-spectrum inhibitor against gut bacterial β-glucuronidases from Ginkgo biloba. Food Funct. 2021, 12, 11190–11201. [Google Scholar] [CrossRef]
- Yang, F.; Zhu, W.; Sun, S.; Ai, Q.; Edirisuriya, P.; Zhou, K. Isolation and Structural Characterization of Specific Bacterial β-Glucuronidase Inhibitors from Noni (Morinda citrifolia) Fruits. J. Nat. Prod. 2020, 83, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Nguyen Viet, P.; Seo Young, Y.; Kang-Hyun, H.; Byung Sun, M.; Jeong Ah, K. Inhibition of β-glucuronidase by amide alkaloids isolated from the fruits of Piper longum L.: Enzyme kinetics, molecular docking, and molecular dynamics simulations. J. Mol. Struct. 2025, 1329, 141433. [Google Scholar] [CrossRef]
- Lu, C.; Xu-Dong, H.; Guang-Hao, Z.; Jian, H.; Zhao-Bin, G.; Ya-Ni, Z.; Jian-Ming, S.; Li-Juan, M.; Shou-De, Z.; Jie, H.; et al. Discovery of a botanical compound as a broad-spectrum inhibitor against gut microbial β-glucuronidases from the Tibetan medicine Rhodiola crenulata. Int. J. Biol. Macromol. 2024, 267, 131150. [Google Scholar] [CrossRef]
- Sun, C.P.; Tian, X.G.; Feng, L.; Wang, C.; Li, J. Inhibition of gut bacterial β-glucuronidase by chemical components from black tea: Inhibition interactions and molecular mechanism. Arab. J. Chem. 2021, 14, 103457. [Google Scholar] [CrossRef]
- Emadeldin, M.K.; Faris, F.; Haifa, A.A.; May, B.-J.; Hassan, A.R.; Al Mokhtar, L. Bridging in silico and in vitro perspectives to unravel molecular mechanisms underlying the inhibition of β-glucuronidase by coumarins from Hibiscus trionum. Biophys. Chem. 2024, 313, 107304. [Google Scholar] [CrossRef]
- Nguyen Viet, P.; Myung-Sook, H.; Le Ba, V.; Young Ho, K.; Seo Young, Y. Investigation of the inhibitory activity of triterpenoids isolated from Actinidia polygama stems against β-glucuronidase via enzyme kinetics, molecular docking, and molecular dynamics analyses. J. Mol. Struct. 2024, 1317, 139135. [Google Scholar] [CrossRef]
- Higashi, R.; Ohta, E.; Ômura, H.; Uy, M.M.; Ohta, S. Triterpenoid Saponins from the Fruits of Symplocos lucida as β-Glucuronidase Inhibitors. Chem. Biodivers. 2022, 19, e202200774. [Google Scholar] [CrossRef] [PubMed]
- Reina, H.; Emi, O.; Tatsuo, N.; Hisashi, Ô.; Mylene, M.U.; Shinji, O. Acylated secoiridoid glucosides isolated from the fruit extract of Symplocos lucida inhibit β-glucuronidase. Phytochem. Lett. 2022, 51, 127–131. [Google Scholar] [CrossRef]
- Ying, Y.; Lei, P.; Xu, Y.; Lin, Y.; Yang, N.; Han, Y.; Zhang, Z.; Shan, W.; Rao, G.; Wang, J. Secondary metabolites from Penicillium sp. HS-11, a fungal endophyte of Huperzia serrata. Fitoterapia 2024, 175, 105943. [Google Scholar] [CrossRef] [PubMed]
- Ying, Y.; Qiu, Y.; Tang, J.; Lin, Y.; Xu, S.; Ni, J.; Zhang, Y.; Tong, C.; Mao, W. A new sesterterpenoid with β-glucuronidase inhibitory activity from the Cordyceps-colonising fungus Aspergillus versicolour ZJUTE2. Nat. Prod. Res. 2025, 1–8. [Google Scholar] [CrossRef]
- Bhanukiran, K.; Priya, V. Exploring multitarget potential of Piper nigrum fruit constituents for Alzheimer’s disease: An AI-driven strategy. Phytomedicine 2024, 135, 156108. [Google Scholar] [CrossRef]
- Kim, D.H.; Shim, S.B.; Kim, N.J.; Jang, I.S. Beta-glucuronidase-inhibitory activity and hepatoprotective effect of Ganoderma lucidum. Biol. Pharm. Bull. 1999, 22, 162–164. [Google Scholar] [CrossRef]
- Edwinson, A.L.; Yang, L.; Peters, S.; Hanning, N.; Jeraldo, P.; Jagtap, P.; Simpson, J.B.; Yang, T.Y.; Kumar, P.; Mehta, S.; et al. Gut microbial β-glucuronidases regulate host luminal proteases and are depleted in irritable bowel syndrome. Nat. Microbiol. 2022, 7, 680–694. [Google Scholar] [CrossRef]
- Uoti, A.; Neulasalmi, O.; Hiippala, K.; Oksanen, T.; Arkkila, P.; Puustinen, L.; Satokari, R.; Sjöstedt, N. Characterization of fecal deglucuronidation activity in healthy subjects and in patients treated with fecal microbiota transplantation. Drug Metab. Dispos. 2025, 53, 100205. [Google Scholar] [CrossRef]
- Chen, J.; Li, Y.; Tang, S.; Jin, W.; Yan, R. Gut microbial β-glucuronidases and their role in the microbiome-metabolite axis in colorectal cancer. Nat. Commun. 2025, 16, 10660. [Google Scholar] [CrossRef]
- Cheng, K.W.; Tseng, C.H.; Chen, I.J.; Huang, B.C.; Liu, H.J.; Ho, K.W.; Lin, W.W.; Chuang, C.H.; Huang, M.Y.; Leu, Y.L.; et al. Inhibition of gut microbial β-glucuronidase effectively prevents carcinogen-induced microbial dysbiosis and intestinal tumorigenesis. Pharmacol. Res. 2022, 177, 106115. [Google Scholar] [CrossRef]
- Chrysostomou, D.; Roberts, L.A.; Marchesi, J.R.; Kinross, J.M. Gut Microbiota Modulation of Efficacy and Toxicity of Cancer Chemotherapy and Immunotherapy. Gastroenterology 2023, 164, 198–213. [Google Scholar] [CrossRef]
- Gao, R.; Yue, B.; Lv, C.; Geng, X.; Yu, Z.; Wang, H.; Zhang, B.; Ai, F.; Wang, Z.; Liu, D.; et al. Targeted inhibition of Gus-expressing Enterococcus faecalis to promote intestinal stem cell and epithelial renovation contributes to the relief of irinotecan chemotoxicity by dehydrodiisoeugenol. Acta Pharm. Sin. B 2024, 14, 5286–5304. [Google Scholar] [CrossRef] [PubMed]
- Lam, W.; Bussom, S.; Guan, F.; Jiang, Z.; Zhang, W.; Gullen, E.A.; Liu, S.H.; Cheng, Y.C. The four-herb Chinese medicine PHY906 reduces chemotherapy-induced gastrointestinal toxicity. Sci. Transl. Med. 2010, 2, 45ra59. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Xie, X.; Chen, Y.; Pan, S.; Wu, Z.; Yang, C.; Liang, J.; Zhang, M.; Wang, Q.; Chen, J.; et al. Huang Qin Decoction inhibits the initiation of experimental colitis associated carcinogenesis by controlling the PAD4 dependent NETs. Phytomedicine 2022, 107, 154454. [Google Scholar] [CrossRef] [PubMed]
- Teng, X.; Wu, B.; Liang, Z.; Zhang, L.; Yang, M.; Liu, Z.; Liang, Q.; Wang, C. Three bioactive compounds from Huangqin decoction ameliorate Irinotecan-induced diarrhea via dual-targeting of Escherichia coli and bacterial β-glucuronidase. Cell Biol. Toxicol. 2024, 40, 88. [Google Scholar] [CrossRef]
- Li, P.; Zhang, R.; Zhou, J.; Guo, P.; Liu, Y.; Shi, S. Vancomycin relieves tacrolimus-induced hyperglycemia by eliminating gut bacterial beta-glucuronidase enzyme activity. Gut Microbes 2024, 16, 2310277. [Google Scholar] [CrossRef]
- Zhu, X.; Ding, G.; Ren, S.; Xi, J.; Liu, K. The bioavailability, absorption, metabolism, and regulation of glucolipid metabolism disorders by quercetin and its important glycosides: A review. Food Chem. 2024, 458, 140262. [Google Scholar] [CrossRef]
- Xing, J.; Chen, X.; Zhong, D. Absorption and enterohepatic circulation of baicalin in rats. Life Sci. 2005, 78, 140–146. [Google Scholar] [CrossRef]
- Tuli, H.S.; Aggarwal, V.; Kaur, J.; Aggarwal, D.; Parashar, G.; Parashar, N.C.; Tuorkey, M.; Kaur, G.; Savla, R.; Sak, K.; et al. Baicalein: A metabolite with promising antineoplastic activity. Life Sci. 2020, 259, 118183. [Google Scholar] [CrossRef]
- Zeng, X.; Sheng, Z.; Zhang, Y.; Xiao, J.; Li, Y.; Zhang, J.; Xu, G.; Jia, J.; Wang, M.; Li, L. The therapeutic potential of glycyrrhizic acid and its metabolites in neurodegenerative diseases: Evidence from animal models. Eur. J. Pharmacol. 2024, 985, 177098. [Google Scholar] [CrossRef]
- Wang, X.; Guo, L.; Zheng, L.; Zhao, W.; Li, L. Natural Sweetener Glycyrrhetinic Acid Monoglucuronide Improves Glucose Homeostasis in Healthy Mice. J. Agric. Food Chem. 2024, 72, 3483–3494. [Google Scholar] [CrossRef] [PubMed]
- Afkhami-Poostchi, A.; Mashreghi, M.; Iranshahi, M.; Matin, M.M. Use of a genetically engineered E. coli overexpressing β-glucuronidase accompanied by glycyrrhizic acid, a natural and anti-inflammatory agent, for directed treatment of colon carcinoma in a mouse model. Int. J. Pharm. 2020, 579, 119159. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.Y.; Zhang, L.; Eibl, G.; Go, V.L. Overestimation of flavonoid aglycones as a result of the ex vivo deconjugation of glucuronides by the tissue β-glucuronidase. J. Pharm. Biomed. Anal. 2014, 88, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.L.; Zhao, Q.Y.; Zhang, K.; Zhang, J.M.; Zhang, H.Y. Insights into inhibitory mechanisms: Unraveling the structure-activity relationship of dietary flavonoids on gut bacterial β-glucuronidase. J. Funct. Foods 2024, 122, 106510. [Google Scholar] [CrossRef]
- Xia, L.J.; Wan, L.; Gao, A.; Yu, Y.X.; Zhou, S.Y.; He, Q.; Li, G.; Ren, H.; Lian, X.L.; Zhao, D.H.; et al. Targeted inhibition of gut bacterial β-glucuronidases by octyl gallate alleviates mycophenolate mofetil-induced gastrointestinal toxicity. Int. J. Biol. Macromol. 2024, 264, 130145. [Google Scholar] [CrossRef]
- Kon, R.; Ikarashi, N.; Yamaguchi, A.; Teshima, Y.; Yamaguchi, T.; Miyaoka, K.; Fukuda, M.; Noguchi, H.; Tomimoto, R.; Sakai, H.; et al. Green tea extract prevents CPT-11-induced diarrhea by regulating the gut microbiota. Sci. Rep. 2023, 13, 6537. [Google Scholar] [CrossRef]
- Bahareh, J.; Ahmad Reza, B.; Maryam, M.M. Targeted bacteria-mediated therapy of mouse colorectal cancer using baicalin, a natural glucuronide compound, and E. coli overexpressing β–glucuronidase. Int. J. Pharm. 2023, 642, 123099. [Google Scholar] [CrossRef]
- Yue, B.; Gao, R.; Lv, C.; Yu, Z.; Wang, H.; Geng, X.; Wang, Z.; Dou, W. Berberine Improves Irinotecan-Induced Intestinal Mucositis Without Impairing the Anti-colorectal Cancer Efficacy of Irinotecan by Inhibiting Bacterial β-glucuronidase. Front. Pharmacol. 2021, 12, 774560. [Google Scholar] [CrossRef]


| GUS Categories | Description | Abundance (%) | Representative Sources | Membrane Traversal | References |
|---|---|---|---|---|---|
| Loop 1 | Initially termed the “Bacterial Loop” in EcGUS, >15 residues | 5.5% (human) 8.6% (mouse) | Bacillota | Intracellular | [21,22] |
| Mini-Loop 1 | Located in the same position as L1, 10–15 residues | 15–21% (human) 6.5% (mouse) | Bacillota Bacteroidota | Secreted by Bacteroidota: Yes Secreted by Bacillota: No | [21,22] |
| Loop 2 | Adjacent in structure to L1, a full size of ≥12 residues | 12–14% (human) 6.8% (mouse) | Bacillota Bacteroidota | Able to traverse the inner microbial membrane | [21,22] |
| Mini-Loop 2 | Located in the same position as Mini-Loop 2, 9–11 residues | 4–7% (human) 15.3% (mouse) | Bacteroidota Verrucomicrobiota | Able to traverse the inner microbial membrane | [21,22] |
| Mini-Loop 1,2 | Contain both mL1 and mL2 motifs, exhibit 10–15 residues in the L1 and 9–12 residues in L2. | 1–2% | Verrucomicrobiota Bacteroidota | Able to traverse the inner microbial membrane | [21,22] |
| NL | NL GUS enzymes lack a loop in either position | 49–57% | Bacillota Bacteroidota | Secreted by Bacteroidota: Yes Secreted by Bacillota: No | [21,22] |
| FMN | FMN-binding glycoside hydrolase | - | Bacillota | - | [23] |
| Compound Class | Compound Name | Structure | GUS Source | IC50/Ki (μM) | Inhibition Type | Binding Interactions/Binding Site | Substrate/Positive Control | Reference |
|---|---|---|---|---|---|---|---|---|
| 2′-Hydroxychrysin |  | EcGUS | 3.16 ± 0.34/3.07 | Non-competitive | Hydrophobic and polar interactions/Phe554, Phe448, His162, Asp163, Tyr472, Glu413 | PNPG/ EGCG | [38] | |
| Jaceosidin |  | EcGUS | 5.70 ± 0.08/6.18 | Mixed | Hydrophobic and polar interactions/Met447, Leu361, Phe448, Asn566, Trp549, His330, Asp163, Asn412, Glu413, Tyr468 | PNPG/ EGCG | [38] | |
| 5,7,4′,5′-Tetrahydroxy-6,3′-dimethoxyflavone |  | EcGUS | 3.82 ± 0.10/3.55 | Mixed | Hydrogen bonding/Lys568, Trp549, Asn566, Met447 | PNPG/ EGCG | [38] | |
| 3′-Geranyl genistein |  | EcGUS | 3.65 ± 0.28/– | Mixed | Hydrogen bonding and hydrophobic interactions/Glu413, Glu504, Phe161, His162, Asp163, Tyr472, Met447 | PNPG/ EGCG | [38] | |
| 3′-Geranyl-5,7,2′,4′-tetrahydroxyisoflavone | 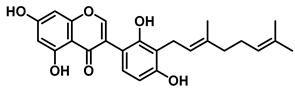 | EcGUS | 3.97 ± 0.11/– | Non-competitive | Hydrogen bonding and hydrophobic interactions/Glu413, Met447, Tyr472, Arg562 | PNPG/ EGCG | [38] | |
| Myricetin |  | EcGUS | 3.95 ± 0.04/3.07 | Non-competitive | Hydrogen bonding and electrostatic interactions/Glu504, Glu413, Trp549 | PNPG/ EGCG | [69] | |
| Chrysoeriol |  | EcGUS | 4.94 ± 0.11/4.58 | Non-competitive | Hydrophobic interactions/Leu361, Val355, Val446, Met447 | PNPG/ EGCG | [69] | |
| Amentoflavone |  | EcGUS | 0.62 ± 0.072 (DDAOG)/– 0.49 ± 0.03 (SN-38G)/– | Mixed (DDAOG) Competitive (SN-38G) | Hydrogen bonding/Leu361, Ile363, Glu413 | DDAOG/SN38G | [70] | |
| Scutellarein |  | EcGUS | 5.76 ± 1.53/– | Competitive | Hydrogen bonding/Glu413, Arg562 | PNPG/ DSL | [71] | |
| Luteolin |  | EcGUS | 8.68 ± 2.02/– | Competitive | Hydrogen bonding/Glu413, Arg562 | PNPG/ DSL | [71] | |
| Steppogenin |  | EcGUS | 6.21 ± 0.20/– | Mixed | Hydrophobic interactions and hydrogen bonding/Phe161, Phe448, Glu413, Tyr472 | PNPG/ EGCG | [72] | |
| Azaleatin |  | EcGUS | 0.57 ± 0.04/– | Non-competitive | Hydrophobic interactions and hydrogen bonding/Phe448, Tyr468, Glu413 | PNPG/ EGCG | [73] | |
| Morin |  | EcGUS | 1.12 ± 0.09/– | Non-competitive | Hydrogen bonding/Tyr160, Gln158, Lys157, His162, Ser159, Ser557, Ile560 | PNPG/ DSL | [74] | |
| Sanggenon C |  | EcGUS | 2.07 ± 0.06/– | Non-competitive | Hydrogen bonding and pi-alkyl interactions/Ser159, Ser557, Tyr160, Lys157, Leu361, Ile149 | PNPG/ DSL | [74] | |
| Kuwanon G |  | EcGUS | 2.37 ± 0.11/– | Non-competitive | Hydrophobic interactions and hydrogen bonding/Ser159, Tyr160, His162, Asp163, Lys157, Gln158, Leu361, Ile149, Ser557, Ile560 | PNPG/ DSL | [74] | |
| Sanggenol A |  | EcGUS | 3.27 ± 0.17/– | Non-competitive | Hydrophobic interactions and hydrogen bonding/Ser159, Tyr160, His162, Asp163, Lys157, Gln158, Leu361, Ile149, Ser557, Ile560 | PNPG/ DSL | [74] | |
| Kuwanon C |  | EcGUS | 4.27 ± 0.32/– | Non-competitive | Hydrophobic interactions and hydrogen bonding/Ser159, Tyr160, His162, Asp163, Lys157, Gln158, Leu361, Ile149, Ser557, Ile560 | PNPG/ DSL | [74] | |
| Amentoflavone |  | C. perfringens (CpGUS) | 2.36 ± 0.12/– | Non-competitive | Hydrogen bonding and pi-alkyl interactions/Arg574, Lys315, Glu55, Ala556, Lys577 | PNPG/ DSL | [75] | |
| S. pasteuri (SpasGUS) | 2.88 ± 0.38/– | Non-competitive | Hydrogen bonding and pi-alkyl interactions/His336, Glu508, Asn414, His364, Ile565, Arg566 | PNPG/ DSL | [75] | |||
| EcGUS | 3.43 ± 0.32/– | Non-competitive | Hydrogen bonding/Asp163, Ser557, Trp160, Lys157 | PNPG/ DSL | [75] | |||
| Moricitrin B |  | EcGUS | 0.95 ± 0.01/– | Not specified | –/– | 4-MUG/ DSL | [76] | |
| Americanol B |  | EcGUS | 4.02 ± 0.06/– | Not specified | –/– | 4-MUG/ DSL | [76] | |
| Isoamericanol B |  | EcGUS | 6.91 ± 0.16/– | Not specified | –/– | 4-MUG/ DSL | [76] | |
| Natural Amides | Grossamide |  | EcGUS | 0.73 ± 0.03/– | Non-competitive | Electrostatic and hydrophobic Interactions/Glu413, Glu504, Arg562 | PNPG/ EGCG | [72] |
| Grossamide K |  | EcGUS | 1.24 ± 0.06/– | Non-competitive | Hydrogen bonding and hydrophobic interactions/Ser159, Asn358, Leu561, Arg562 | PNPG/ EGCG | [72] | |
| N-Feruloyltyramine | 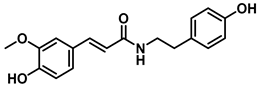 | EcGUS | 12.16 ± 0.7/– | Not specified | Hydrogen bonding/Asp163, His520, Arg562 | PNPG/ EGCG | [72] | |
| Piperlongumamide F |  | EcGUS | 4.9 ± 0.9/3.1 | Non-competitive | Van der waals interactions and hydrogen bonding/Trp340, Ala316 | PNPG/ DSL | [77] | |
| 1-(Eicosa-2E,14Z-dienoyl)piperidine |  | EcGUS | 8.1 ± 0.7/12.8 | Non-competitive | Hydrogen bonding and hydrophobic interactions/Val312, Asp319 | PNPG/ DSL | [77] | |
| N-Isobutyl-(2E,4E)-undeca-2,4-dienamide |  | EcGUS | 5.8 ± 1.2/8.8 | Uncompetitive | Hydrophobic interactions/Tyr160, Leu361, Tyr472 | PNPG/ DSL | [77] | |
| (2E,4E,12Z)-N-Isobutyloctadeca-2,4,12-trienamide) |  | EcGUS | 6.3 ± 0.9/5.3 | Uncompetitive | Alkyl interactions/Ile363, Phe365 | PNPG/ DSL | [77] | |
| (2E,4E,14Z)-N-Isobutyleicosa-2,4,14-trienamide |  | EcGUS | 6.2 ± 2.4/8.8 | Uncompetitive | Van der waals interactions and hydrophobic interactions/Tyr160, His162, Asp163 | PNPG/ DSL | [77] | |
| (2E,4E,16Z)-N-Isobutyldocosa-2,4,16-trienamide |  | EcGUS | 5.1 ± 1.5/9.0 | Uncompetitive | Van der waals interactions and hydrophobic interactions/Tyr160, Gln158, Phe161 | PNPG/ DSL | [77] | |
| Tannin | 1,2,3,4,6-Penta-O-galloyl-β-D-glucopyranose (PGG) | 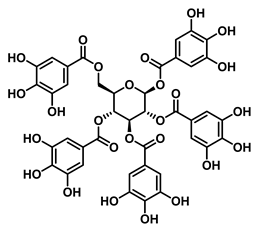 | EcGUS | 0.13 ± 0.004/0.30 | Non-competitive | Hydrogen bonding/Glu106, His107, Glu108, Gly109 | 4-MUG/ DSL | [78] |
| CpGUS | 0.19 ± 0.001/0.17 | Non-competitive | Hydrogen bonding/Lys147, Val161, Asp164, Met364 | 4-MUG/ DSL | [78] | |||
| Streptococcus agalactiae (SaGUS) | 0.14 ± 0.003/0.12 | Non-competitive | Hydrogen bonding/Asp46, Asp550, Glu552, Glu561 | 4-MUG/ DSL | [78] | |||
| Eubacterium eligens (EeGUS) | 0.55 ± 0.003/1.29 | Non-competitive | Hydrogen bonding/Asp58, His62, Tyr63, Asn134 | 4-MUG/ DSL | [78] | |||
| Catechins And Theaflavins | Catechin gallate |  | EcGUS | 1.48/– | Mixed | Hydrogen bonding and hydrophobic interactions/Glu413, Ser360, Ile560 | DDAOG/- | [79] |
| Theaflavin-3′ monogallate |  | EcGUS | 1.03/– | Mixed | Hydrogen bonding/Leu359, Asn369, Arg417 | DDAOG/- | [79] | |
| Theaflavin-3,3′-digallate |  | EcGUS | 1.41/– | Mixed | Hydrogen bonding/Ser360, Glu413, Ile560 | DDAOG/- | [79] | |
| Alkaloids | Angustine |  | EcGUS | 4.55 ± 0.13/– | Non-competitive | Hydrophobic interactions and π-π stacking/Leu361, Met47 | PNPG/ EGCG | [72] |
| Phenylpropanoids | Cleomiscosin A |  | EcGUS | 3.97 ± 0.35/– | Mixed | Hydrophobic interactions and hydrogen bonding/Phe161, Asp163, Tyr472, Arg562 | PNPG/ EGCG | [80] |
| Quinones | Mansonone H |  | EcGUS | 10.32 ± 1.85/– | Non-competitive | Hydrophobic interactions and polar interaction/Glu413, Leu361, Val446, Met447 | PNPG/ EGCG | [80] |
| Terpenoids | 2α,3α,24-trihydroxyurs-12-en-28-oic acid |  | EcGUS | 14.36 ± 0.42/12.8 | Non-competitive | Hydrogen bonding and salt bridge/Asp203, Ser231, Gly232 | PNPG/ DSL | [81] |
| Hemslonin B |  | EcGUS | 1.0 ± 0.1/– | Not specified | –/– | PNPG/ DSL | [82] | |
| Momordin IIa |  | EcGUS | 8.3 ± 0.3/– | Not specified | –/– | PNPG/ DSL | [82] | |
| 3β-hydroxyolean-12-en-28-oic acid-28-O-β-D-glucopyranoside |  | EcGUS | 14.0 ± 1.3/– | Not specified | –/– | PNPG/ DSL | [82] | |
| Symplolucidin B |  | EcGUS | 17.2 ± 0.1/– | Not specified | –/– | PNPG/ DSL | [83] | |
| Sorbicillinoids | Trichodimerol |  | EcGUS | 92.0 ± 9.4/– | Not specified | –/– | PNPG/ DSL | [84] |
| Sesterterpenoids | Versicolorin A |  | EcGUS | 49.82 ± 1.21/– | Not specified | –/– | PNPG/ DSL | [85] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Shen, J.; Xu, S.; Zhao, Q.; Zhang, J.; Zhang, H. β-Glucuronidase at the Microbiota—Host Interface: Dual Regulatory Roles and Precision Modulation by Natural Products. Molecules 2026, 31, 601. https://doi.org/10.3390/molecules31040601
Shen J, Xu S, Zhao Q, Zhang J, Zhang H. β-Glucuronidase at the Microbiota—Host Interface: Dual Regulatory Roles and Precision Modulation by Natural Products. Molecules. 2026; 31(4):601. https://doi.org/10.3390/molecules31040601
Chicago/Turabian StyleShen, Jialu, Shuai Xu, Qingyu Zhao, Junmin Zhang, and Huiyan Zhang. 2026. "β-Glucuronidase at the Microbiota—Host Interface: Dual Regulatory Roles and Precision Modulation by Natural Products" Molecules 31, no. 4: 601. https://doi.org/10.3390/molecules31040601
APA StyleShen, J., Xu, S., Zhao, Q., Zhang, J., & Zhang, H. (2026). β-Glucuronidase at the Microbiota—Host Interface: Dual Regulatory Roles and Precision Modulation by Natural Products. Molecules, 31(4), 601. https://doi.org/10.3390/molecules31040601







