Anti-Inflammatory Effects of Bisacurone Isolated from Curcuma longa (Ryudai Gold): An In Vivo and In Silico Study
Abstract
1. Introduction
2. Results
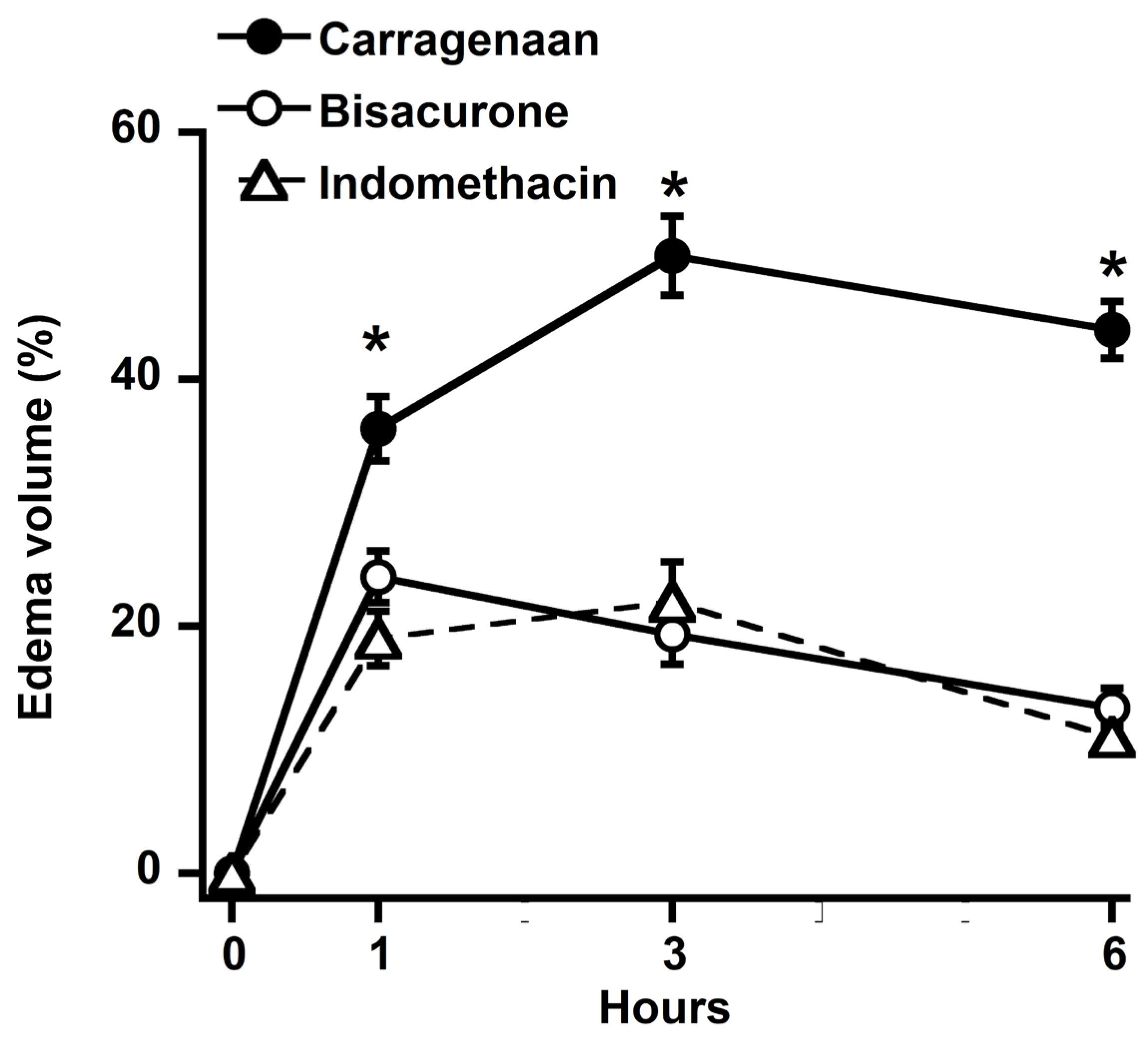
2.1. Effects of Bisacurone on Carrageenan-Induced Paw Edema in Rat
2.2. Histopathological Analysis of Rat Paws Tissue
2.3. Results of In Silico Studies
2.3.1. Conceptual Density Functional Theory (DFT)
2.3.2. Molecular Docking Studies
2.3.3. In Silico Pharmacokinetics and Toxicity Analysis
2.3.4. Molecular Dynamics Simulation
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Isolation and Structural Elucidation of Bisacurone
4.3. Experimental Animal
4.4. Anti-Inflammatory Study
4.5. Histopathological Examination of the Paw Tissues
4.6. Statistical Analysis
4.7. In Silico Analysis
4.7.1. Preparation of Ligand and Conceptual Density Functional Theory (DFT) Calculations
4.7.2. Preparation of Macromolecule
4.7.3. Molecular Docking
4.7.4. In Silico Drug-Likeness and Toxicity Predictions
4.7.5. Molecular Dynamics Simulation Protocol
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef]
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 65, S140–S146. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.S.; Ghosh, S. Shared principles in NF-κB signaling. Cell 2008, 132, 344–362. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef]
- Vane, J.R.; Botting, R.M. Mechanism of action of anti-inflammatory drugs. Scand J. Rheumatol. 1996, 102, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Glucocorticosteroids: Current and future directions. Br. J. Pharmacol. 2011, 163, 29–43. [Google Scholar] [CrossRef]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Harikumar, K.B. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef]
- Hosseini, A.; Hosseinzadeh, H. Antidotal or protective effects of Curcuma longa (turmeric) and its active ingredient, curcumin, against natural and chemical toxicities: A review. Biomed. Pharmacother. 2018, 99, 411–421. [Google Scholar] [CrossRef]
- Li, S.; Yuan, W.; Deng, G.; Wang, P.; Yang, P.; Aggarwal, B.B. Chemical composition and product quality control of turmeric (Curcuma longa L.). Pharm. Crops 2011, 2, 28–54. Available online: https://scholarworks.sfasu.edu/agriculture_facultypubs/1/ (accessed on 29 December 2025). [CrossRef]
- Gupta, S.C.; Sung, B.; Kim, J.H.; Prasad, S.; Li, S.; Aggarwal, B.B. Multitargeting by turmeric, the golden spice: From kitchen to clinic. Mol. Nutr. Food Res. 2013, 57, 1510–1528. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; El Rayess, Y.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocińska, K.; Zielińska, D.; et al. Turmeric and Its Major Compound Curcumin on Health: Bioactive Effects and Safety Profiles for Food, Pharmaceutical, Biotechnological and Medicinal Applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef] [PubMed]
- Adhimoolam, K.; Thi, C.T.D.; Chin, S.; Sureshbabu, A.; Smirnova, E.; Mohammad, M.; Senthil, K.; Min, T.; Kim, I.-J. Turmeric (Curcuma longa L.): The multifaceted golden plant—Insights and uses. Plant Biotechnol. Rep. 2025, 19, 593–625. [Google Scholar] [CrossRef]
- Akter, J.; Hossain, M.A.; Takara, K.; Islam, M.Z.; Hou, D.X. Antioxidant activity of different species and varieties of turmeric (Curcuma spp.): Isolation of active compounds. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 215, 9–17. [Google Scholar] [CrossRef]
- He, C.; Miyazawa, T.; Abe, C.; Ueno, T.; Suzuki, M.; Mizukami, M.; Kurihara, K.; Toda, M. Hypolipidemic and anti-inflammatory effects of Curcuma longa-derived bisacurone in high-fat diet-fed mice. Int. J. Mol. Sci. 2023, 24, 9366. [Google Scholar] [CrossRef]
- Uchio, R.; Okuda-Hanafusa, C.; Sakaguchi, H.; Saji, R.; Muroyama, K.; Murosaki, S.; Yamamoto, Y.; Hirose, Y. Curcuma longa extract reduces serum inflammatory markers and postprandial hyperglycemia in healthy but borderline participants with overweight and glycemia in the normal/prediabetes range: A randomized, double-blind, and placebo-controlled trial. Front. Nutr. 2024, 11, 1324196. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, Z.; Zhang, L. Bisacurone attenuates diabetic nephropathy by ameliorating oxidative stress, inflammation and apoptosis in rats. Hum. Exp. Toxicol. 2022, 41, 09603271221143713. [Google Scholar] [CrossRef]
- Yan, Z.; Li, S.; Gong, Z. Bisacurone gel ameliorates burn wounds in experimental rats via anti-inflammatory, antioxidant, and angiogenic properties. Acta Cir. Bras. 2023, 38, e382423. [Google Scholar] [CrossRef]
- Liao, X.; Wang, Q.; Yang, X.; Yao, Y.; Zhu, D.; Feng, J.; Wang, K. Bisacurone ameliorates myocardial ischemia/reperfusion injury in rats: Regulation of inflammatory and apoptosis pathways via CHOP/GRP78 proteins. BMC Pharmacol. Toxicol. 2025, 26, 115. [Google Scholar] [CrossRef] [PubMed]
- Domingo, L.R.; Ríos-Gutiérrez, M.; Pérez, P. Applications of conceptual density functional theory indices to organic chemistry reactivity. Molecules 2016, 21, 748. [Google Scholar] [CrossRef] [PubMed]
- Geerlings, P.; Chamorro, E.; Chattaraj, P.K.; De Proft, F.; Gázquez, J.L.; Liu, S.; Morell, C.; Toro-Labbé, A.; Vela, A.; Ayers, P. Conceptual density functional theory: Status and prospects. Theor. Chem. Acc. 2020, 139, 36. [Google Scholar] [CrossRef]
- Nagarajan, S.K.; Babu, S.; Sohn, H.; Devaraju, P.; Madhavan, T. Toward a better understanding of the interaction between somatostatin receptor 2 and its ligands: A structural characterization study using molecular dynamics and conceptual density functional theory. J. Biomol. Struct. Dyn. 2019, 37, 3081–3102. [Google Scholar] [CrossRef]
- Feng, L.P.; Lu, L.H.; Yuan, M.R.; Mei, S.X.; Li, X.H. Two pairs of bisabolane sesquiterpenoid stereoisomers, bisacurone D-G, from the rhizome of Curcuma longa L. Fitoterapia 2020, 146, 104701. [Google Scholar] [CrossRef]
- Guo, L.Y.; Cai, X.F.; Lee, J.J.; Kang, S.S.; Shin, E.M.; Zhou, H.Y.; Jung, J.W.; Kim, Y.S. Comparison of suppressive effects of demethoxycurcumin and bisdemethoxycurcumin on expressions of inflammatory mediators In Vitro and In Vivo. Arch. Pharm. Res. 2008, 31, 490–496. [Google Scholar] [CrossRef]
- Jyotirmayee, B.; Mahalik, G. A review on selected pharmacological activities of Curcuma longa L. Int. J. Food Prop. 2022, 25, 1377–1398. [Google Scholar] [CrossRef]
- Ti, H.; Mai, Z.; Wang, Z.; Zhang, W.; Xiao, M.; Yang, Z.; Shaw, P. Bisabolane-type sesquiterpenoids from Curcuma longa L. exert anti-influenza and anti-inflammatory activities through NF-κB/MAPK and RIG-1/STAT1/2 signaling pathways. Food Funct. 2021, 12, 6697–6711. [Google Scholar] [CrossRef]
- Banerjee, P.; Kemmler, E.; Dunkel, M.; Preissner, R. ProTox 3.0: A webserver for the prediction of toxicity of chemicals. Nucleic Acids Res. 2024, 52, W513–W520. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.K. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Veber, D.F.; Johnson, S.R.; Cheng, H.Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular properties that influence the oral bioavailability of drug candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- Kurcinski, M.; Oleniecki, T.; Ciemny, M.P.; Kuriata, A.; Kolinski, A.; Kmiecik, S. CABS-flex standalone: A simulation environment for fast modeling of protein flexibility. Bioinformatics 2019, 35, 694–695. [Google Scholar] [CrossRef] [PubMed]
- López-Blanco, J.R.; Aliaga, J.I.; Quintana-Ortí, E.S.; Chacón, P. iMODS: Internal coordinates normal mode analysis server. Nucleic Acids Res. 2014, 42, W271–W276. [Google Scholar] [CrossRef]
- Akter, J.; Islam, M.Z.; Takara, K.; Hossain, M.A.; Sano, A. Isolation and structural elucidation of antifungal compounds from Ryudai gold (Curcuma longa) against Fusarium solani sensu lato isolated from American manatee. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 219, 87–94. [Google Scholar] [CrossRef]
- Akter, J.; Islam, M.Z.; Hossain, M.A.; Takara, K. Pharmacological activities of 4-methylene-8-hydroxybisabola-2,10-diene-9-one, a new compound isolated from Ryudai gold (Curcuma longa). Naunyn Schmiedebergs Arch. Pharmacol. 2020, 393, 191–201. [Google Scholar] [CrossRef]
- Uehara, S.; Yasuda, I.; Takeya, K.; Itokawa, H.; Iitaka, Y. New bisabolane sesquiterpenoids from the rhizomes of curcuma xanthorhiza (zingiberaceae) ii. Chem. Pharm. Bull. 1990, 38, 261–263. [Google Scholar] [CrossRef][Green Version]
- Islam, M.Z.; Akter, J.; Hossain, M.A.; Islam, M.S.; Islam, P.; Goswami, C.; Nguyen, H.T.T.; Miyamoto, A. Anti-Inflammatory, Wound Healing, and Anti-Diabetic Effects of Pure Active Compounds Present in the Ryudai Gold Variety of Curcuma longa. Molecules 2024, 29, 2795. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B Condens Matter 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Fukui, K. Role of frontier orbitals in chemical reactions. Science 1982, 218, 747–754. [Google Scholar] [CrossRef]
- Hohenberg, P.; Kohn, W. Inhomogeneous electron gas. Phys. Rev. 1964, 136, B864–B871. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B. Gaussian 09, Revision D.01; Gaussian Inc.: Wallingford, CT, USA, 2009.
- Dennington, R.; Keith, T.; Millam, J. GaussView, Version 6.0.16; Semichem Inc.: Shawnee Mission, KS, USA, 2016.
- Liu, S.; Misquitta, Y.R.; Olland, A.; Johnson, M.A.; Kelleher, K.S.; Kriz, R.; Lin, L.L.; Stahl, M.; Mosyak, L. Crystal structure of a human IκB kinase β asymmetric dimer. J. Biol. Chem. 2013, 288, 22758–22767. [Google Scholar] [CrossRef] [PubMed]
- Park, B.S.; Song, D.H.; Kim, H.M.; Choi, B.-S.; Lee, H.; Lee, J.-O. The structural basis of lipopolysaccharide recognition by the TLR4-MD-2 complex. Nature 2009, 458, 1191–1195. [Google Scholar] [CrossRef] [PubMed]
- Stroud, J.C.; Oltman, A.; Han, A.; Bates, D.L.; Chen, L. Structural Basis of HIV-1 Activation by NF-κB—A Higher-Order Complex of p50:RelA Bound to the HIV-1 LTR. J. Mol. Biol. 2009, 393, 98–112. [Google Scholar] [CrossRef]
- Wang, Z.; Canagarajah, B.J.; Boehm, J.C.; Kassisà, S.; Cobb, M.H.; Young, P.R.; Abdel-Meguid, S.; Adams, J.L.; Goldsmith, E.J. Structural basis of inhibitor selectivity in MAP kinases. Structure 1998, 6, 1117–1128. [Google Scholar] [CrossRef]
- Oza, V.; Ashwell, S.; Almeida, L.; Brassil, P.; Breed, J.; Deng, C.; Gero, T.; Grondine, M.; Horn, C.; Ioannidis, S.; et al. Discovery of checkpoint kinase inhibitor (S)-5-(3-fluorophenyl)-N-(piperidin-3-yl)-3-ureidothiophene-2-carboxamide (AZD7762) by structure-based design and optimization of thiophenecarboxamide ureas. J. Med. Chem. 2012, 55, 5130–5142. [Google Scholar] [CrossRef]
- Garcin, E.D.; Arvai, A.S.; Rosenfeld, R.J.; Kroeger, M.D.; Crane, B.R.; Andersson, G.; Andrews, G.; Hamley, P.J.; Mallinder, P.R.; Nicholls, D.J.; et al. Anchored plasticity in nitric oxide synthase. Nat. Chem. Biol. 2008, 4, 700–707. [Google Scholar] [CrossRef]
- Rimon, G.; Sidhu, R.S.; Lauver, D.A.; Lee, J.Y.; Sharma, N.P.; Yuan, C.; Frieler, R.A.; Trievel, R.C.; Lucchesi, B.R.; Smith, W.L. Coxibs interfere with the action of aspirin by binding tightly to one monomer of cyclooxygenase-1. Proc. Natl. Acad. Sci. USA 2010, 107, 28–33. [Google Scholar] [CrossRef]
- Orlando, B.J.; Malkowski, M.G. Substrate-selective Inhibition of Cyclooxygeanse-2 by Fenamic Acid Derivatives Is Dependent on Peroxide Tone. J. Biol. Chem. 2016, 291, 15069–15081. [Google Scholar] [CrossRef]
- Guex, N.; Peitsch, M.C.; Schwede, T. Automated comparative protein structure modeling with SWISS-MODEL and Swiss-PdbViewer: A historical perspective. Electrophoresis 2009, 30, S162–S173. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Yao, X.-Q.; Skjærven, L.; Grant, B.J. Rapid characterization of allosteric networks with ensemble normal mode analysis. J. Phys. Chem. B 2016, 120, 8276–8288. [Google Scholar] [CrossRef]
















| Total Energy (E γ) (eV) | Molecular Dipole Moment (Debye) | EHOMO (eV) | ELUMO (eV) | Egap (eV) | Absolute Hardness (η) | Global Softness (σ) (eV−1) | Electronegativity (χ) | Chemical Potential (μ) | Electrophilicity Index (ω) (eV) |
|---|---|---|---|---|---|---|---|---|---|
| −22,093.6 | 6.518061 | −6.754 | −1.464 | 5.29 | 2.645 eV | 0.378 | 4.109 eV | −4.109 | 3.19 |
| Receptors | Binding Energy (kcal/mol) | Information on Hydrogen Bond | Information on Other Interactions | ||||
|---|---|---|---|---|---|---|---|
| Number of Hydrogen Bonds | Amino Acids Involved in H-Bonding | Hydrogen Bond Distance | Number of Bonds | Amino Acids Involved | Bond Type | ||
| IKKβ (4KIK) | −7.3 | 2 | ASN28, ASN28 | 2.64462 Å 2.39063 Å | 10 | ILE165, VAL29, LYS44, MET96, LEU21, VAL152, CYS99, VAL152, TYR98 | Hydrophobic |
| TLR4–MD-2 (3FXI) | −5.4 | 3 | GLU111, SER184, ARG106 | 2.63259 Å 2.03719 Å 2.510782 Å | 4 | ARG106, ALA107, LEU212, HIS159 | Hydrophobic |
| NF-κB p65 (RelA) (3GUT) | −5.4 | 1 | ARG605 | 2.42814 Å | 11 | VAL248, ARG605, ARG246, LYS572, LYS218, PHE607 | Hydrophobic |
| p38 MAPK (1A9U) | −6.2 | 1 | LYS53 | 1.94967 Å | 10 | ALA51, VAL30, ILE84, ILE85, ILE86, ILE87, ILE88, ILE89, ILE90, ILE91 | Hydrophobic |
| JNK1 (3PZE) | −6.7 | 0 | - | - | 11 | VAL40, ALA53, LEU168, ILE32, ILE86, MET111, VAL158, LEU110 | Hydrophobic |
| iNOS (3E7G) | −5.6 | 3 | ASN482, ASN482, GLY117 | 2.67859 Å 1.94651 Å 3.35344 Å | 6 | CYS110, LEU108, PRO122, LEU125 | Hydrophobic |
| COX-1 (3KK6) | −7.2 | 2 | ARG120, ILE523 | 2.2323 Å 2.76692 Å | 16 | VAL116, VAL349, LEU352, LEU359, ILE523, LEU531, ALA527, TYR355, TRP387, PHE518. | Hydrophobic |
| COX-2 (5F19) | −4.1 | 1 | ARG216 | 2.70304 Å | 7 | ARG216, ALA219, PRO218, PHE220 | Hydrophobic |
| Mol. Wt. (g/mol) | NHD | NHA | NRB | Lipophilicity | Log S (ESOL) Water Solubility | LV | VV | GV | EV | MV | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Log P (iLOGP) | Log P (MLOGP) | ||||||||||
| 252.35 | 2 | 3 | 4 | 2.74 | 1.88 | −2.32 | 0 | 0 | 0 | 0 | 0 |
| (Log Kp) cm/s | GIA | BBB | Inhibitor Interaction | |||||
|---|---|---|---|---|---|---|---|---|
| P-gp | CYP1A2 Inhibitor | CYP2C19 Inhibitor | CYP2C9 Inhibitor | CYP2D6 Inhibitor | CYP3A4 Inhibitor | |||
| −6.5 | High | Yes | No | No | No | No | No | No |
| LD50 (mg/kg) | Toxicity Class | Organ Toxicity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Hepato | Carcino | Immuno | Mutagen | Cytoto | Nephro | Respi | Cardio | ||
| 1170 | 4 | Inactive | Inactive | Inactive | Inactive | Inactive | Inactive | Inactive | Inactive |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Anjum, M.; Hossain, M.A.; Akter, J.; Miyamoto, A.; Islam, M.Z. Anti-Inflammatory Effects of Bisacurone Isolated from Curcuma longa (Ryudai Gold): An In Vivo and In Silico Study. Molecules 2026, 31, 548. https://doi.org/10.3390/molecules31030548
Anjum M, Hossain MA, Akter J, Miyamoto A, Islam MZ. Anti-Inflammatory Effects of Bisacurone Isolated from Curcuma longa (Ryudai Gold): An In Vivo and In Silico Study. Molecules. 2026; 31(3):548. https://doi.org/10.3390/molecules31030548
Chicago/Turabian StyleAnjum, Mahir, Md. Amzad Hossain, Jesmin Akter, Atsushi Miyamoto, and Md. Zahorul Islam. 2026. "Anti-Inflammatory Effects of Bisacurone Isolated from Curcuma longa (Ryudai Gold): An In Vivo and In Silico Study" Molecules 31, no. 3: 548. https://doi.org/10.3390/molecules31030548
APA StyleAnjum, M., Hossain, M. A., Akter, J., Miyamoto, A., & Islam, M. Z. (2026). Anti-Inflammatory Effects of Bisacurone Isolated from Curcuma longa (Ryudai Gold): An In Vivo and In Silico Study. Molecules, 31(3), 548. https://doi.org/10.3390/molecules31030548








