1. Introduction
Breast cancer is one of the leading causes of cancer-related mortality among women worldwide and represents a clinically heterogeneous disease. Approximately 10–15% of patients develop highly aggressive tumors with an increased propensity for metastasis [
1]. Triple-negative breast cancer (TNBC) comprises a heterogeneous group of tumors with distinct histological, genomic, and immunological characteristics and is defined by the absence of estrogen and progesterone hormone receptors, as well as human epidermal growth factor receptor 2 (HER2). Several risk factors, including obesity, are associated with poor prognosis, as the interaction between adipocytes and tumor cells creates a permissive tumor microenvironment that promotes cancer cell proliferation, invasion, metastatic dissemination, and resistance to cell death [
2].
In the cancer context, liver metastasis represents one of the most challenging complications in TNBC and is associated with high rates of morbidity and mortality. The liver is among the most frequent sites of breast cancer dissemination and constitutes the initial metastatic organ in approximately 30% of individuals with metastatic breast cancer. The development of liver metastases is associated with an unfavorable prognosis and reduced overall survival, even in the context of advances in diagnostic and therapeutic strategies achieved in recent decades. Notably, TNBC patients with liver involvement exhibit a poorer clinical outcome compared with those affected by other breast cancer subtypes. Consequently, the identification of novel therapeutic strategies capable of improving outcomes in patients with metastatic breast cancer remains a critical unmet clinical need.
The FDA has approved several drugs with therapeutic uses targeting members of the protein kinase superfamily (PKs) associated with various diseases. Because the ATP-binding site is conserved across the human kinome, ATP-mimicking compounds often interact with multiple kinases. Drugs such as Abemaciclib, Palbociclib, Ribociclib, and Trilaciclib target the CDK4/6 proteins at the ATP-binding site; therefore, kinase-like proteins such as CDK4/6, AKT, HER2, ErbB1/2/4, and FKBP12/mTOR are considered therapeutic targets for breast cancer treatment [
3]. In contrast, inhibitors designed for a single target, like the kinase inhibitor Capivasertib, which targets the AKT oncogene—a member of the PI3K/Akt/mTOR signaling pathway—have become key in treating cancer, diabetes, obesity, neurodegenerative diseases, aging, and autoimmunity [
3]. Among the primary treatment options for metastatic breast cancer are taxanes, anthracyclines, methotrexate (MTX), carboplatin, immune checkpoint inhibitors targeting PD-1 or PD-L1, PARP inhibitors, and combinations of these agents with immunotherapy, radiotherapy, or surgery [
4]. However, a significant number of patients respond poorly to these treatments, leading to low survival rates and a reduced quality of life. As a result, the pursuit of new therapeutic strategies that can improve the prognosis for patients with metastatic breast cancer remains a vital priority.
Cyclodipeptides (CDPs) are a class of molecules of the diketopiperazine family, produced by a wide range of organisms that exhibit notable biological activities, including cytotoxic effects against multiple cancer cell lines [
5,
6,
7,
8]. Previously, we reported that CDPs, such as cyclo(L-Pro-L-Tyr), cyclo(L-Pro-L-Val), cyclo(L-Pro-L-Leu), and cyclo(L-Pro-L-Phe), isolated from the
Pseudomonas aeruginosa PAO1 bacterium, induce apoptosis in cervical, colon, leukemia, and melanoma cancer cells. In HeLa cells, CDPs inhibit cell proliferation, including arresting cells at the G0–G1 stage, and induce apoptosis via an intrinsic pathway that relies on caspase-9 and -3 activation. Mechanistically, the antiproliferative effects of CDPs involve modulation of multiple signaling networks associated with tumor growth, survival, and plasticity, including the PI3K/Akt/mTOR, Ras/Raf/MEK/ERK1/2, PI3K/JNK/PKA, p27Kip1/CDK1/survivin, MAPK, HIF-1, Wnt/β-catenin, HSP27, epithelial–mesenchymal transition (EMT), and cancer stem cell (CSCs) pathways [
3,
6,
7]. Transcriptomic profiling of HeLa cells treated with CDPs revealed 151 differentially expressed genes distributed across 15 major biological processes, including regulation, signaling, development, locomotion, adhesion, metabolism, growth, proliferation, and immune response. Pathway enrichment analysis suggested that CDPs modulate multiple signaling networks, encompassing the insulin/IGF/MAP kinase cascade, P38/MAPK signaling, interleukin-mediated pathways, epidermal growth factor receptor (EGFR), PI3K signaling, platelet-derived growth factor (PDGF), Notch, cadherin, p53, Hedgehog, transforming growth factor-β (TGF-β), fibroblast growth factor (FGF), FAS-mediated signaling, angiogenesis, cholesterol biosynthesis, and pathways associated with inflammation, oxidative stress responses, apoptosis, cytoskeletal organization, and T- and B-cell activation [
9]. In a murine melanoma model, CDP treatment reduced tumor growth by decreasing the expression of proteins that mediate pathways involved in energy metabolism, lipid synthesis, EMT, invasion, and metastasis [
10]. In this context, cyclic peptides have emerged as promising therapeutic agents for kinase inhibition, as dysregulated kinases play central roles in cancer cell survival, proliferation, metastasis, and drug resistance. Given this background, we sought to determine whether bacterial CDPs have a promising effect on metastatic breast cancer, including advanced stages of tumorigenesis using a xenograft model of triple-negative breast cancer established with MDA-MB-231 (ER-, PR-, HER2-) cells.
3. Discussion
CDPs have become promising options for cancer treatment due to their stability, high selectivity, and ability to block key cancer signaling pathways [
11]. Their cytotoxic and pro-apoptotic effects have been shown in multiple cancer cell lines [
3,
5,
6,
7,
8]. Beyond their direct anti-proliferative activity, several diketopiperazines have been shown to interfere with mechanisms associated with tumor aggressiveness and therapeutic resistance. For example, fumitremorgin C analogs can overcome multidrug resistance by inhibiting the breast cancer resistance protein (BCRP), a key efflux transporter that limits intracellular drug accumulation. Similarly, the HLY838 diketopiperazine functions as an O-GlcNAc transferase (OGT) inhibitor, enhancing the anti-tumor effects of CDK9 inhibitors by downregulating c-Myc and E2F1 [
12].
Additionally, Verticillin A inhibits c-Met phosphorylation and the downstream Ras/Raf/MEK/ERK signaling pathway, resulting in reduced metastatic potential in colon cancer models [
13]. Together, these studies highlight the potential of CDPs and related diketopiperazines to serve as multitarget anticancer and anti-metastatic agents. Consistent with this, earlier research in mouse melanoma showed that CDPs from Pseudomonas aeruginosa significantly decreased key EMT markers, including MMP-1, E-cadherin, N-cadherin, HIF-1α, Vimentin, and CK-1, supporting their ability to inhibit tumor cell migration and invasion [
6,
10]. These observations support investigating the anti-invasive and metastasis-related effects of bacterial CDPs in aggressive breast cancer subtypes like triple-negative breast cancer.
In this study, functional assays further confirm the ability of bacterial CDPs to suppress invasion and metastasis-associated behaviors in the TNBC model. Wound-healing assays demonstrated that CDPs inhibit the migratory capacity of MDA-MB-231 cells more effectively than methotrexate (MTX) (
Figure 1a). Similarly, transwell invasion assays show that CDPs significantly reduce the invasive ability of MDA-MB-231 cells under both monoculture and macrophage co-culture conditions compared to MTX, with their combination being even more effective. Tumor-associated macrophages (TAMs) play a key role in tumor progression by secreting chemokines and growth factors that enhance cancer cell proliferation, migration, and invasiveness [
14]. Our data indicate that CDPs interfere with TAM–cancer cell interactions in the MDA-MB-231 cell line, highlighting their dual role in promoting apoptosis and reducing metastasis-related processes. This supports their function as inhibitors of tumor cell migration and invasiveness. The data are particularly relevant because the interaction between cancer cells and macrophages, along with the release of chemokines, enhances malignancy and the invasive capacity of cancer cells. The greatest inhibition observed with the combination of CDPs and MTX (up to 90% reduction in invasiveness) suggests a synergistic effect between these agents.
Three-dimensional spheroid models further demonstrated the anti-metastatic and anti-tumor effects of CDPs. Spheroid cultures are enriched with cancer stem cell (CSC)-like populations that exhibit increased self-renewal and resistance to therapy [
14]. Therefore, the spheroid structure in MDA-MB-231 cells promotes resistance to cytotoxic compounds. Our results showed that CDPs significantly reduced spheroid size and number, lowered cell viability, and increased apoptosis in MDA-MB-231 spheroids compared to MTX-treated cultures. The effects were more pronounced when using the CDPs + MTX combination or cyclodextrins (
Figure 1c–e). Additionally, the spheroid integration assay, which mimics three-dimensional tumor growth and plasticity, supports the ability of CDPs to disrupt key stages of tumor progression and stemness-related properties. These findings highlight the potential of CDPs to overcome CSC-associated resistance mechanisms in aggressive breast cancer models.
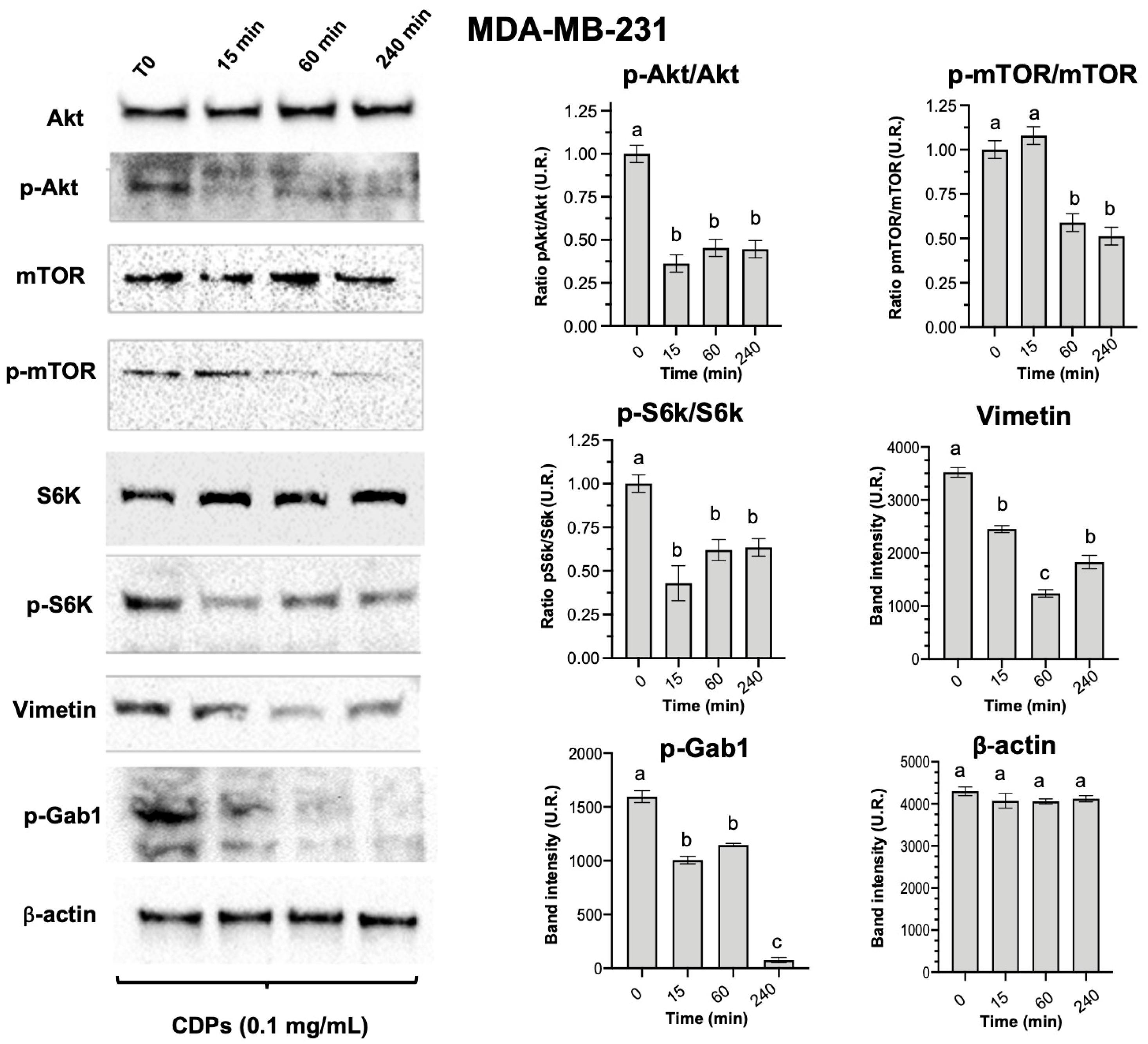
Mechanistically, our results further support previous findings that indicate CDPs act through the PI3K/Akt/mTOR pathway [
3,
5,
6,
10]. In MDA-MB-231 cells, CDPs significantly decreased phosphorylation of Akt, mTOR, and S6K (
Figure 2), as well as reducing levels of Vimentin and Gab1—two proteins closely linked to EMT-related and metastasis-associated signaling [
15]. Thus, CDPs effectively target the PI3K/Akt/mTOR signaling pathway, a key regulator of cell survival, proliferation, and growth. This aligns with the observed suppression of p-S6K phosphorylation and the decrease in Vimentin and p-Gab1 expression. Vimentin is an essential marker of EMT, a process crucial for invasiveness and metastasis. Therefore, the downregulation of Vimentin and inhibition of the PI3K/Akt/mTOR pathway are primary molecular mechanisms responsible for the reduced migration and invasiveness observed in vitro in MDA-MB-231 cells.
Beyond protein-coding genes, growing evidence highlights the role of non-coding RNAs (ncRNAs), including lncRNAs and miRNAs, as key regulators of EMT, metastasis, and therapeutic resistance in breast cancer. Our previous transcriptomic analysis of bacterial cyclodipeptides (CDPs) in the HeLa cell line revealed modulation of transcription factors and regulatory genes involved in epigenetic control and the regulation of miRNAs, circRNAs, and lncRNAs, suggesting that ncRNA-mediated mechanisms may contribute to the anticancer effects of CDPs [
9]. Although ncRNAs were not directly examined in this study, the observed suppression of the PI3K/Akt/mTOR/FOXO1 pathway and EMT-related markers aligns with signaling pathways known to be regulated by metastasis-associated lncRNAs. Notably, MALAT1 has been shown to promote EMT, activate PI3K/Akt/mTOR signaling, and inhibit FOXO1-mediated tumor suppressive functions in breast cancer [
16]. In this context, the coordinated suppression of Akt phosphorylation, EMT markers, and FOXO1 inactivation after CDP treatment may indicate the involvement of molecular pathways controlled by MALAT1 and related ncRNAs.
Meanwhile, emerging therapies focus on targeting metastasis-related non-coding RNAs with advanced delivery systems. Notably, exosome-mediated siRNA delivery has been shown to inhibit postoperative breast cancer metastasis by disrupting pro-metastatic signaling and pre-metastatic niche formation in triple-negative breast cancer models [
17]. Compared to earlier research, the current findings indicate that CDPs might have anti-metastatic effects by indirectly affecting signaling pathways often regulated by ncRNAs, such as the PI3K/Akt/mTOR/FOXO1 pathway, without requiring complex delivery methods.
In the orthotopic TNBC xenograft model, implanting MDA-MB-231 cells directly into the mammary gland of animals creates a tumor microenvironment that exhibits key features of aggressive breast cancer progression. Importantly, before assessing antitumor efficacy, histopathological evaluation of major organs combined with continuous monitoring of body weight showed no evidence of systemic toxicity related to CDP treatment, supporting a favorable in vivo safety profile (
Figure 8 and
Figure S1). Treatment with CDPs significantly reduced tumor volume and weight, especially when administered after the tumor was established, and many treated animals showed no detectable tumors at the end of the experiment (
Figure 4). Besides suppressing primary tumor growth, CDP treatment also alleviated tumor-associated systemic changes. Untreated TNBC-bearing mice had increased weights of the lungs, kidneys, liver, and spleen, along with histological changes such as metastatic foci indicating organ damage caused by the tumor (
Figure 4). Conversely, groups treated with CDPs and the combination of CDPs plus MTX showed normalization of organ weights and no visible metastatic foci. Histopathological analysis of primary tumors revealed that untreated TNBC displayed extensive hyperchromasia. In contrast, CDP-treated tumors showed less hyperchromasia, with areas of cell aggregation observed (
Figure 6). These findings suggest that in tumors from animals treated with CDPs and MTX, cell proliferation is inhibited, as indicated by the size and weight of the tumors dissected from each group (
Figure 4).
Metastatic progression in breast cancer patients is often linked to systemic changes, including increased levels of serum ALT, AST, gamma-glutamyl transferase (GGT), alkaline phosphatase (ALP), and LDH [
18]. In our TNBC model, untreated mice showed significant increases in AST and LDH (
Figure 5). Treatment with CDPs normalized these enzyme levels and reduced organ weight gains. Changes in leukocyte profiles, especially a higher neutrophil-to-lymphocyte ratio (NLR), are recognized markers of poor prognosis in breast cancer [
19]. In our study, TNBC mice exhibited the highest NLR, which was reduced in the CDP-treated groups, approaching control levels (
Figure 5c), indicating decreased tumor aggressiveness. Anemia, another common cancer complication, was also improved with CDP treatment. These findings suggest that CDPs mitigate tumor-related systemic and liver dysfunction, supporting a reduction in tumor aggressiveness and metastatic potential.
It is important to emphasize the safety profile and systemic benefits. CDP treatment did not change the activity of liver enzymes ALT, AST, and LDH in healthy control mice (C + CDPs). Conversely, CDPs reversed the significant increase in AST and LDH activity observed in the TNBC group. These results, along with the restoration of histological structures in the lung and spleen, show that CDPs not only target the tumor locally but also help normalize tumor-related systemic changes. Histological analysis revealed a clear reduction in vascular structures in the liver of the TNBC group. In contrast, structural damage was observed in the lung and spleen tissues, which was significantly reduced after CDP treatment (
Figure 8). These findings further suggest that, in our model, metastasis of the MDA-MB-231 line occurred in the animals following implantation into target organs such as the liver, lungs, and spleen. The ability of CDPs to restore normal tissue architecture supports their role in reducing tumor-related organ dysfunction and decreasing metastatic risk.
At the molecular level, protein and gene expression analyses revealed coordinated modulation of signaling pathways involved in metastasis, immune evasion, and DNA damage response. To further explore tumor-associated signaling in TNBC, we assess metastasis in the tumor of the xenografted TNBC model. Molecular analyses also showed reduced p-Akt levels in tumors from CDP-treated animals, along with decreased total Akt expression (
Figure 7), indicating suppression of the PI3K/Akt/mTOR signaling pathway. FOXO1, a forkhead transcription factor that regulates apoptosis, autophagy, and cell cycle arrest, is negatively regulated by Akt-mediated phosphorylation [
20]. In our study, CDP-treated animals showed decreased FOXO1 phosphorylation. Although total FOXO1 protein levels were reduced, the decrease in FOXO1 phosphorylation corresponds with increased FOXO1 activity, allowing its nuclear functions and the activation of growth-suppressive and stress-response pathways [
21]. This activation aligns with the induction of apoptotic pathways. These findings match our previous transcriptomic data in HeLa cells, showing CDP-mediated upregulation of FOXO1-related genes like GADD45A and SGK1 [
9].
Gab1, an adaptor protein that mediates signals for proliferation, angiogenesis, and invasion [
22], was overexpressed and phosphorylated in TNBC tumors but was downregulated by CDPs, especially when combined with MTX (
Figure 7). This indicates that inhibiting Gab1 is a key mechanism behind the suppression of invasion- and metastasis-related processes caused by CDPs. Protein analysis in the xenografted tumors confirmed the inhibition of the Akt pathway. Consistent with the in vitro results, the expression of phosphorylated Akt protein was reduced in the CDP-treated groups, as Gab1, p-Gab1, FOXO1, and its phosphorylated form p-FOXO1 were markedly decreased in tumors from mice treated with CDPs and MTX.
The study of gene expression in the liver provided crucial data on tumor-related systemic changes influenced by the CDPs. To further explore the metastasis-related signaling pathways targeted by the CDPs, we analyzed the hepatic expression of gene markers associated with multiple pathways involved in tumor progression, immune regulation, and cellular plasticity (
Figure 9). RT-qPCR analysis showed that CDP treatment significantly reversed the overexpression of key genes seen in the TNBC group, including SNAIL, which is linked to promoting EMT, invasiveness, metastasis, and drug resistance. Among immune checkpoint components, such as PD-L1, which contributes to immune evasion, overexpression was observed in the TNBC group and returned to normal levels after CDP treatment, approaching those seen in healthy controls. Regarding genes involved in DNA damage/repair and the cell cycle—such as BRCA1 and GADD45A, which exhibited a marked increase in the TNBC group consistent with tumor-induced systemic stress and genomic instability—these were normalized by CDP treatment. Conversely, genes like PTEN, CXCL12, and CDKN1A (p21) were overexpressed in the livers of animals treated with both CDPs and MTX compared to untreated healthy mice, indicating activation of compensatory tumor-suppressive and cell cycle–regulatory mechanisms related to EMT, invasiveness, metastasis, and drug resistance, reflecting an adaptive hepatic response aimed at reinforcing immune regulation, growth suppression, and homeostasis despite tumor burden. Overall, these findings suggest that bacterial CDPs in the TNBC model modulate tumor-associated systemic gene expression programs related to DNA damage responses, immune regulation, and cellular stress, thereby affecting molecular mechanisms connected to tumor aggressiveness and metastatic progression. This modulation targets both tumor cell–intrinsic signaling pathways and systemic responses linked to the tumor. The inhibition of Akt/mTOR/S6K signaling, suppression of EMT markers, modulation of immune-related genes, and the favorable safety profile collectively position CDPs as promising therapeutic agents. Moreover, the enhanced efficacy observed when combining CDPs with MTX supports the potential of combination strategies to improve treatment outcomes in aggressive breast cancer subtypes.
In summary, in the TNBC mouse model, CDP administration significantly reversed primary tumor growth in the mammary glands, with greater effectiveness when combined with MTX. Beyond local tumor control, CDP treatment reduced systemic changes associated with tumors in distant organs, such as the liver, which are commonly involved in metastatic progression. The treatment with CDPs reduced metastatic foci in organs such as the liver. At the molecular level, CDPs modulated the expression of genes linked to tumor aggressiveness, immune regulation, and stress responses, such as SNAIL, GADD45A, and PD-L1, while increasing the expression of growth-suppressing and homeostatic regulators, including PTEN, CXCL12, and CDKN1A (p21). The data provide new insights into the molecular mechanisms behind the anti-metastatic and anti-invasive effects of CDPs, showing that these effects are connected to genes involved in EMT, invasiveness, and metastasis. This indicates that the signal transduction mechanism involves inhibition of phosphorylation in the Akt/mTOR/S6K pathway and of metastasis markers, such as Vimentin, Gab1, and FOXO1. The findings suggest that combining CDPs with MTX enhances the anti-metastatic effect in a TNBC xenograft model using the MDA-MB-231 line, indicating that bacterial CDPs could be a promising therapeutic agent. In conclusion, bacterial cyclic peptides represent a promising multi-target therapy for TNBC, showing greater efficacy than MTX in vivo. Their mechanism involves blocking the PI3K/Akt/mTOR/FOXO1 pathway and suppressing EMT, resulting in strong anti-tumor and anti-metastatic effects with a good safety profile.
4. Materials and Methods
4.1. Chemicals, Reagents, and Cell Culture
The chemicals and reagents used include Dulbecco’s Modified Eagle Medium (DMEM; Sigma-Aldrich, St. Louis, MO, USA), fetal bovine serum (FBS; Gibco Life Technologies, Grand Island, NY, USA), and trypsin solution (Sigma-Aldrich). A mixture mainly composed of cyclo(L-Pro-L-Tyr), cyclo(L-Pro-L-Val), cyclo(L-Pro-L-Leu), and cyclo(L-Pro-L-Phe) is isolated from the cell-free supernatant of
Pseudomonas aeruginosa PAO1 bacteria, with a purity of over 95% [
23,
24]. This mixture is dissolved in a DMSO-water ratio of 1:3 to prepare stock solutions (100 mg/mL). The MDA-MB-231 breast tumor cell line was obtained from invasive ductal carcinoma (ATCC, Manassas, VA, USA) and cultured in complete media [DMEM supplemented with 10% (
v/
v) FBS, 100 units/mL penicillin, 40 µg/mL streptomycin, and 1 µg/mL amphotericin B (Sigma-Aldrich)], with 1.6 g/L glucose added. Cell culture media were changed twice a week, with cells incubated at 37 °C, 80% humidity, and 5% CO
2. Cells were then trypsinized and counted using a hemocytometer. All cell cultures and procedures were performed in class II biological safety cabinets.
4.2. Cell Viability and Apoptosis Determination
Cell viability was determined with the MTT method. Briefly, cell cultures were grown in 96-well flat-bottomed plates in DMEM medium containing FBS for 24 h, then incubated with CDPs for 24 h at 37 °C with 5% CO2. MTT (50 mg/mL) in PBS was added to each well, and the mixture was incubated for 4 h at 37 °C. Finally, 100 μL of 2-propanol/1M HCl (19:1, v/v) was added to dissolve the formazan crystals, and the absorbance was measured at 595 nm using a microplate reader (BioTek Instruments, Winooski, VT, USA).
Necrosis and apoptosis were evaluated in cell cultures incubated in DMEM medium with FBS for 4 h with CDP treatment. Following incubation, cells were collected by centrifugation at 2000× g for 10 min. The pellet was suspended in 20 μL and incubated with annexin V and propidium iodide (PI) (Molecular Probes, Invitrogen, Life Technologies, Carlsbad, CA, USA). Fluorescence was immediately quantified by FACS using an Accuri-C6 Flow Cytometer (BD Biosciences, San Jose, CA, USA). At least 20,000 cellular events were used for calculations.
4.3. Wound Closure Migration Assay
The MDA-MB-231 cell line was grown to 95% confluence, and three wounds were created per plate using a sterile pipette tip. All assays were performed in triplicate. After wounding, plates were washed twice with phosphate-buffered saline (PBS) and replenished with fresh complete medium. Cells were then treated with CDPs (0.01 mg/mL), methotrexate (MTX; 0.05 mg/mL), or their combination for 48 h. Representative images were taken every 24 h, and wound dimensions were quantified using ImageJ 1.53k software (NIH, Bethesda, MA, USA).
4.4. Invasion Assay
The invasive capacity of the MDA-MB-231 cell line was evaluated using a transwell chamber pre-coated with Matrigel (Corning Life Sciences, Union City, CA, USA). Cells were seeded in the upper chamber in complete DMEM medium. For co-culture experiments, RAW 264.7 macrophages were seeded in the lower chamber containing complete DMEM medium. Cells were treated with CDPs (0.01 mg/mL) or methotrexate (MTX; 0.05 mg/mL) incubated by 24 h at 37 °C with 5% CO
2. Inserts were then briefly washed with PBS, and cells that had invaded through the Matrigel and adhered to the lower surface of the membrane were fixed with 70% ethanol for 15 min at room temperature. Fixed cells were stained with 0.2% (w/v) crystal violet for 5 min, washed thoroughly with PBS to remove excess dye, and air-dried. Invaded cells were visualized by optical microscopy [
25].
4.5. Multicellular Spheroids
MDA-MB-231 cell line (2 x10
5 cells) was cultured under non-adherent conditions using 0.6% agarose in complete DMEM medium to generate multicellular spheroids. Cultures were maintained for 14 days with periodic medium replacement until the spheroids reached a diameter of 40–50 µm [
14]. Spheroids were then treated with CDPs (0.1 mg/mL) or methotrexate (MTX; 0.05 mg/mL) and incubated for 4 h, a time point selected to correspond with the determination of lethal (LD
50) and apoptotic effective (ED
50) doses. Following treatment, spheroids were visualized by optical microscopy, and cell viability was assessed using the MTT assay.
4.6. Western Blot Assays
Confluent MDA-MB-231 cells were treated with CDPs (0.1 mg/mL) and incubated for 15 min, 1 h, and 4 h. Cells were then harvested by trypsinization, washed twice with PBS, and centrifuged at 5000× g for 10 min at 4 °C. Cells pellets or tumor tissue were resuspended in RIPA lysis buffer, and lysed by three cycles of low- intensity sonication (20 kHz, 5 W, 30 s each) at 4 °C using an Hielscher LS24 ultrasonic processor. The protein extract free of cellular debris was obtained by centrifugation at 7500× g for 15 min, and the protein concentration was determined by the Bradford method (BioRad, Hercules, CA, USA). Equal amounts of protein were separated by 10% SDS-PAGE and transferred onto PVDF membranes (Millipore, Billerica, MA, USA) using a Bio-Rad transfer system at 15 volts for 45 min. Membranes were blocked with TBS-T (Tris-HCL 10 mM; NaCl 0.9%; tween-20 0.1%, dry milk 5%, pH 7.8), washed three times for 6 min each, and incubated overnight at 4 °C with primary antibodies diluted in TBS-T (1:5000). The following primary antibodies were used: anti-Akt (cat. 2938), anti-p-Akt (cat. 4060), anti-mTOR (cat. 2983), anti-p-mTOR (cat. 5536), anti-S6K (cat. 2708), anti-p-S6K (cat. 9234), anti-Vimentin (cat. 3932); anti-Gab1 (cat. 3232), anti-p-Gab1 (cat. 3234), anti-FOXO1 (cat. 2880), anti-p-FOXO1 (cat. 9464), and anti-β-actin (cat. 4967) (Cell Signaling Technology, Boston, MA, USA and Santa Cruz Biotechnology, Santa Cruz, CA, USA). After three washes with TBS-T (6 min each), membranes were incubated with HRP-conjugated secondary antibody (BioRad) diluted 1:10,000 in TBS-T for 2 h at room temperature. Immunoreactive bands were detected using SuperSignal™ West Pico chemiluminescent substrate (Pierce; Thermo Fisher Scientific, Waltham, MA, USA) and visualized with a ChemiDoc™ MP imaging system (Bio-Rad). Assays were conducted at least three times, and representative images are shown. ImageJ (NIH) was used to quantify image band intensities.
4.7. Xenografted Metastatic Breast Cancer Model
Immunosuppressed female BALB/c nu/nu mice (8 weeks old) were acclimated for 15 days in a pathogen-free environment with a standard diet and water provided ad libitum. All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC) from the Universidad Michoacana de San Nicolás de Hidalgo (IIIQB-UMSNH-IACUC-2022-35). Experiments complied with standard guidelines for the welfare of animals following the Institutional Committee and recommendations of the Mexican Official Regulations for the Use and Care of Animals (NOM 062-ZOO-1999; Ministry of Agriculture, Mexico).
Once the acclimation period concluded with 100% of animals survival, mice were orthotopically implanted with 1 × 10
5 MDA-MB-231 metastatic breast cancer cells suspended in Matrigel and injected into the mammary fat pad adjacent to the right axillary region. Animals were anesthetized by intraperitoneal administration of ketamine (80 mg/kg) and xylazine (10–15 mg/kg). Body weight was monitored throughout the experiment, and tumor dimensions were measured with a caliper. Tumor volume (TV) was calculated using the formula TV = (0.4) (ab
2), where “a” represents the longest tumor diameter and “
b” the shortest diameter. In addition, a pilot study was carried out to select the doses of CDPs to be tested in the TNBC mice model; thus, we decided to use 0.25 mg/kg of mouse body weight. Treatments were administered intraperitoneally and consisted of CDPs (0.25 mg/kg), methotrexate (MTX; 0.05 mg/kg), or a combination of CDPs and MTX (0.25 mg/kg CDPs + 0.05 mg/kg MTX). The CDP treatments involved three rounds of administration, each consisting of three doses given every 3 days (with a 2-day rest between doses), and a week of rest between rounds, totaling 9 doses per mouse. The animal groups, each consisting of five mice (n = 5) as follows (
Figure 3): healthy mice without treatment (C)—procedure 1; healthy mice treated with CDPs (C + CDPs)—procedure 1; mice with TNBC tumors without treatment (TNBC)—procedure 2; mice with tumors treated with CDPs starting from cell inoculation in early stage (TNBC + CDPs (2))—procedure 2; mice with tumors treated with CDPs at the 35th day post-inoculation in advanced stage (TNBC + CDPs (3))—procedure 3; mice with advanced-stage tumors treated with MTX (TNBC + MTX (3))—procedure 3; and mice with tumors treated with a combination of CDPs and MTX (TNBC + CDPs + MTX (3))—procedure 3.
4.8. Animal Euthanasia and Biological Sample Collection
All mice were euthanized by intraperitoneal administration of a lethal dose of sodium pentobarbital (100–150 mg/Kg body weight), as recommended by IIQB/UMSNH/-IACUC and NOM 062-ZOO-1999. Following confirmation of death, tumors were excised, and whole blood was collected by cardiac puncture into microtubes containing EDTA as an anticoagulant. Major organs were subsequently harvested and weighed.
Blood hemoglobin was determined using heparinized capillary tubes by centrifugation at 3500 rpm for 5 min to separate red blood cells from plasma. Subsequently, serum biochemical parameters, including aspartate aminotransferase (AST), alanine aminotransferase (ALT), and lactate dehydrogenase (LDH), were quantified using a Fuji Dry-Chem NX700 (Fujifilm, Santa Ana, CA, USA).
4.9. Histological Analysis of Tissues
After euthanasia, tumors, liver, lungs, and spleen were excised and processed for histological evaluation. Tissues were fixed in 4% paraformaldehyde and remained until dehydration. The samples were subjected to a 4 h dehydration process at varying ethanol and xylene concentrations, then embedded in paraffin. Paraffin-embedded tissues were sectioned at a thickness of 4 µm, mounted on glass slides, and stained with hematoxylin and eosin (H&E). Photographs were taken under an optical microscope and recorded using an Accu-Scope EXC-120 LED microscope camera at magnifications of 4×, 10×, and 40×.
4.10. RNA Extraction and RT-qPCR
Total RNA from liver samples of each animal group was pooled, homogenized using a tissue homogenizer (Biospec Products, Bartlesville, OK, USA), and extracted using TRIzol reagent (Invitrogen; Thermo Fisher Scientific) according to the manufacturer’s protocol. The RNA concentration and quality were assessed using a spectrophotometer (NanoDrop 2000; Thermo Fisher Scientific). According to the manufacturer’s instructions, complementary DNA (cDNA) was synthesized using a QuantiNova Reverse Transcription Kit (Bio-Rad). Quantitative PCR (qPCR) was performed using the Quantinova SYBR Green PCR Kit (Bio-Rad) in a reaction volume of 10 µL. The thermocycling conditions were set as follows: initial denaturation at 95 °C for 10 min, followed by 40 cycles of denaturation at 95 °C for 5 s and annealing/extension at 60 °C for 30 s and extension to 68 °C for 30 s using a thermocycler (QuantStudio 3; Applied Biosystems by Thermo Fisher Scientific). Relative gene expression levels was calculated using the comparative 2
−ΔΔCt method, normalizing to the internal reference gene
ACTB (β-actin). All experiments were conducted in triplicate. The sequences of the gene-specific primers are provided in
Table S1.
4.11. Statistical Analysis
All quantitative data are presented as mean ± standard error (SE). Statistical analyses were performed using one-way analysis of variance (ANOVA) when comparing more than two experimental groups. When ANOVA indicated significant differences, Tukey’s post hoc multiple comparison test was applied to identify statistically significant pairwise differences between groups. A p-value < 0.05 was considered statistically significant. All analyses were performed using GraphPad Prism version 6.0.