Formulation and Characterization of Hydrogel Chitosan–Pectin Active Films Containing Silymarin
Abstract
1. Introduction
2. Results and Discussion
2.1. Physicochemical Analysis of Hydrogel Chitosan and Chitosan–Pectin Films
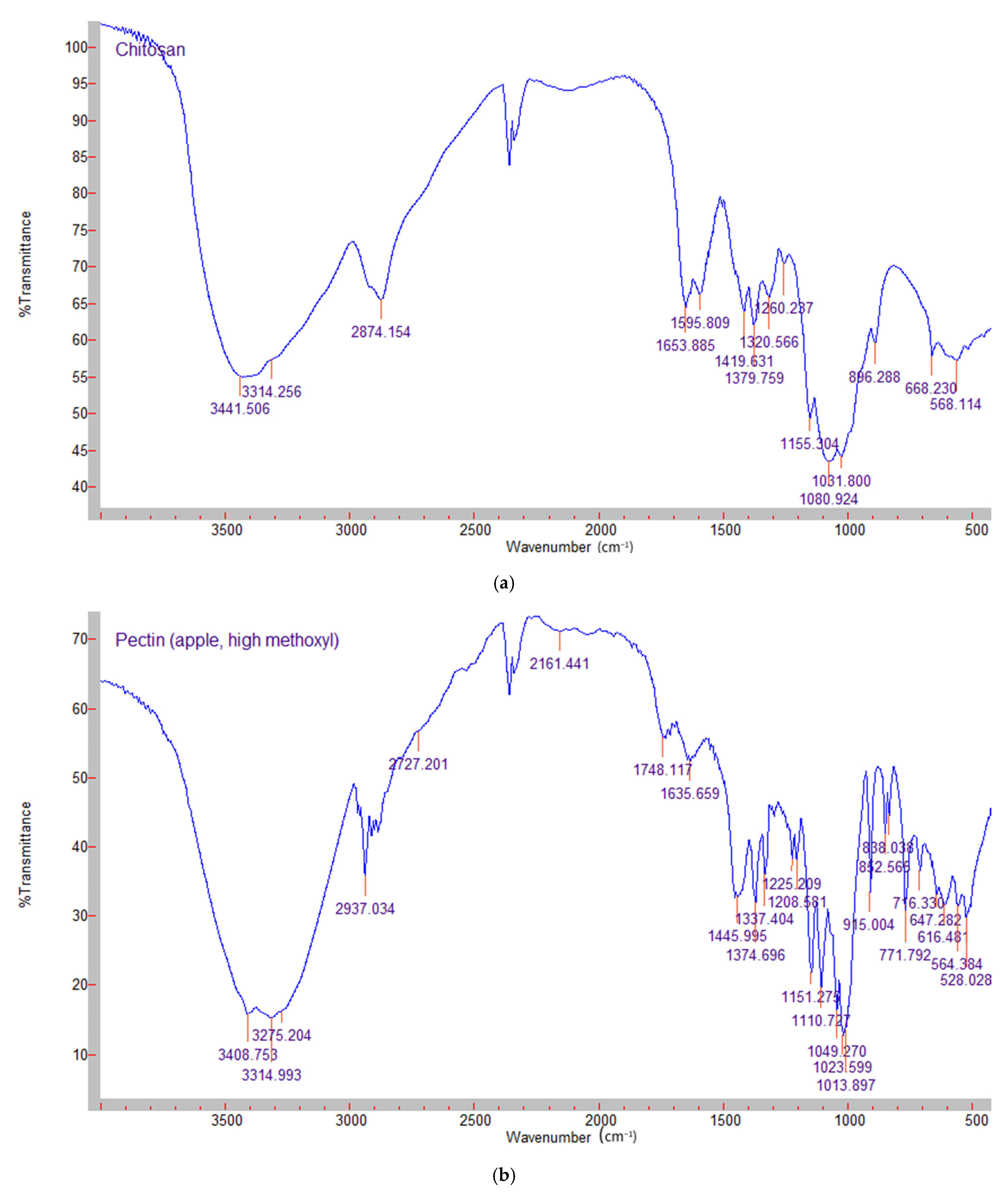
2.2. Fourier Transform Infrared Spectroscopy (FTIR)
2.3. Mechanical Properties
2.4. Optical Properties
2.4.1. Color
2.4.2. Light Transmission
2.5. Antioxidant Properties
2.6. Dissolution Study of Silymarin
3. Materials and Methods
3.1. Materials
3.2. Preparation of Hydrogel Films (With and Without Silymarin)
3.3. Infrared Spectroscopy Analysis (FTIR)
3.4. Physicochemical Analysis of Films
3.4.1. Moisture Content
3.4.2. Water Activity
3.4.3. Thickness
3.4.4. Water Absorption
3.4.5. pH Measurement
3.5. Optical Properties
3.5.1. Color Measurement
3.5.2. Light Transmission (LT)
3.6. Mechanical Tests
3.7. Antioxidant Activity Measurement
3.8. In Vitro Dissolution of Silymarin
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marmouzi, I.; Bouyahya, A.; Ezzat, S.M.; El Jemli, M.; Kharbach, M. The food plant Silybum marianum (L.) Gaertn.: Phytochemistry, Ethnopharmacology and clinical evidence. J. Ethnopharmacol. 2021, 265, 113303. [Google Scholar] [CrossRef]
- Kvasnička, F.; Bıba, B.; Ševčík, R.; Voldřich, M.; Kratka, J. Analysis of the active components of silymarin. J. Chromatogr. A 2003, 990, 239–245. [Google Scholar] [CrossRef]
- Saller, R.; Meier, R.; Brignoli, R. The use of silymarin in the treatment of liver diseases. Drugs 2001, 61, 2035–2063. [Google Scholar] [CrossRef]
- Aslan, E.; Aladağ, T.; Demirel, H.H.; Pektaş, M.B. Silymarin promotes wound healing through regulating epithelial-mesenchymal transition in rat model: Histopathological and immunohistochemical evidences. Health Sci. Q. 2024, 4, 195–205. [Google Scholar]
- Fehér, P.; Vecsernyés, M.; Fenyvesi, F.; Váradi, J.; Kiss, T.; Újhelyi, Z.; Nagy, K.; Bácskay, I. Topical application of Silybum marianum extract. J. Med. Ar. 2011, 14, 5–8. [Google Scholar]
- Vostálová, J.; Tinková, E.; Biedermann, D.; Kosina, P.; Ulrichová, J.; Rajnochová Svobodová, A. Skin Protective Activity of Silymarin and its Flavonolignans. Molecules 2019, 24, 1022. [Google Scholar] [CrossRef]
- Mady, F.M.; Essa, H.; El-Ammawi, T.; Abdelkader, H.; Hussein, A.K. Formulation and clinical evaluation of silymarin pluronic-lecithin organogels for treatment of atopic dermatitis. Drug Des. Devel. Ther. 2016, 10, 1101–1110. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aliabadi, A.; Yousefi, A.; Mahjoor, A.; Farahmed, M. Evaluation of wound healing activity of silymarin (Silybum marianum). J. Anim. Vet. Adv. 2011, 10, 3287–3292. [Google Scholar]
- Sharifi, R.; Rastegar, H.; Kamalinejad, M.; Dehpour, A.R.; Tavangar, S.M.; Paknejad, M.; Mehrabani Natanzi, M.; Ghannadian, N.; Akbari, M.; Pasalar, P. Effect of topical application of silymarin (Silybum marianum) on excision wound healing in albino rats. Acta Med. Iran. 2012, 50, 583–588. [Google Scholar] [PubMed]
- Oryan, A.; Tabatabaei-Naeini, A.; Moshiri, A.; Mohammadalipour, A.; Tabandeh, M.R. Modulation of cutaneous wound healing by silymarin in rats. J. Wound Care 2012, 21, 457–464. [Google Scholar] [CrossRef]
- Tabandeh, M.R.; Oryan, A.; Mohhammadalipour, A.; Tabatabaei-Naieni, A. Silibinin regulates matrix metalloproteinase 3 (stromelysine 1) gene expression, hexoseamines and collagen production during rat skin wound healing. Phytother. Res. 2013, 27, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodi-Nesheli, M.; Alizadeh, S.; Solhi, H.; Mohseni, J.; Mahmoodi-Nesheli, M. Adjuvant effect of oral Silymarin on patients’ wound healing process caused by thermal injuries. Caspian J. Intern. Med. 2018, 9, 341–346. [Google Scholar] [CrossRef]
- Woo, J.S.; Kim, T.S.; Park, J.H.; Chi, S.C. Formulation and biopharmaceutical evaluation of silymarin using SMEDDS. Arch. Pharm. Res. 2007, 30, 82–89. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Martineau, L.; Shek, P.N. Evaluation of a bi-layer wound dressing for burn care. II. In vitro and in vivo bactericidal properties. Burns 2006, 32, 172–179. [Google Scholar] [CrossRef]
- Jose, J.; Athira, V.P.; Michel, H.; Hafeela, A.R.; Bhat, S.G.; Thomas, S.; Pereira Maria, L. Chapter 1–Hydrogels: An overview of the history, classification, principles, applications, and kinetics. In Sustainable Hydrogels; Thomas, S., Sharma, B., Jain, P., Shekhar, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 1–22. [Google Scholar]
- Kertmen, A.; Dziedzic, I.; Ehrlich, H. Patentology of chitinous biomaterials. Part II: Chitosan. Carbohydr. Polym. 2023, 301, 120224. [Google Scholar] [CrossRef]
- Feng, Y.; Jie, D.; Yong, C.; Zheng, Z.; Zhi, D.; Chaoqun, N. Chitosan Gel Film with Patterned Aptamer as well as Preparation Method and Application Thereof. CN 107375253 A, 2017. [Google Scholar]
- Athanasiadis, T.; Hanton, L.R.; Moratti, S.C.; Robinson, B.H.; Robinson, S.R.; Wormald, P.J. Polymer Comprising a Dicarboxy-derivatised Chitosan Polymer Cross-Linked to an Aldehyde-Derivatised Dextran Polymer Useful for Preventing Tissue Adhesion and for Promoting Wound Healing. NZ 583819 A, 2008. [Google Scholar]
- Neufeld, L.; Bianco-Peled, H. Pectin-chitosan physical hydrogels as potential drug delivery vehicles. Int. J. Biol. Macromol. 2017, 101, 852–861. [Google Scholar] [CrossRef]
- Vildanova, R.R.; Petrova, S.F.; Kolesov, S.V.; Khutoryanskiy, V.V. Biodegradable Hydrogels Based on Chitosan and Pectin for Cisplatin Delivery. Gels 2023, 9, 342. [Google Scholar] [CrossRef] [PubMed]
- Ignaczak, A.; Woźniak, Ł.; Salamon, A.; Szczepańska-Stolarczyk, J.; Trych, U.; Chobot, M.; Kowalska, J.; Kowalska, H. Shaping the Physicochemical and Health-Promoting Properties of Carrot Snacks Produced by Microwave-Vacuum Drying with Preliminary Thermal and Enriching Treatment. Molecules 2024, 29, 5100. [Google Scholar] [CrossRef]
- Sabet-Bokati, K.; Plucknett, K. Water-induced failure in polymer coatings: Mechanisms, impacts and mitigation strategies—A comprehensive review. Polym. Degrad. Stab. 2024, 230, 111058. [Google Scholar] [CrossRef]
- Fernandes Queiroz, M.; Melo, K.R.T.; Sabry, D.A.; Sassaki, G.L.; Rocha, H.A.O. Does the Use of Chitosan Contribute to Oxalate Kidney Stone Formation? Mar. Drugs 2015, 13, 141–158. [Google Scholar] [CrossRef]
- Muslu Can, A.; Metin Yildirim, R.; Karadag, A. The Properties of Pectin Extracted from the Residues of Vinegar-Fermented Apple and Apple Pomace. Fermentation 2024, 10, 556. [Google Scholar] [CrossRef]
- Tsai, Y.H.; Yang, Y.N.; Ho, Y.C.; Tsai, M.L.; Mi, F.L. Drug release and antioxidant/antibacterial activities of silymarin-zein nanoparticle/bacterial cellulose nanofiber composite films. Carbohydr. Polym. 2018, 180, 286–296. [Google Scholar] [CrossRef]
- Younis, H.G.R.; Zhao, G. Physicochemical properties of the edible films from the blends of high methoxyl apple pectin and chitosan. Int. J. Biol. Macromol. 2019, 131, 1057–1066. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi, Y. The properties of chitosan and gelatin films incorporated with ethanolic red grape seed extract and Ziziphora clinopodioides essential oil as biodegradable materials for active food packaging. Int. J. Biol. Macromol. 2017, 99, 746–753. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.Y.; Zhang, Z.H.; Li, L.; Yuan, M.L.; Fan, J.; Zhao, T.R. Physio-mechanical properties of an active chitosan film incorporated with montmorillonite and natural antioxidants extracted from pomegranate rind. J. Food Sci. Technol. 2013, 52, 1471–1479. [Google Scholar] [CrossRef]
- Pereda, M.; Ponce, A.G.; Marcovich, N.E.; Ruseckaite, R.A.; Martucci, J.F. Chitosan-gelatin composites and bi-layer films with potential antimicrobial activity. Food Hydrocoll. 2011, 25, 1372–1381. [Google Scholar] [CrossRef]
- Ojagh, S.M.; Rezaei, M.; Razavi, S.H.; Hosseini, S.M.H. Development and evaluation of a novel biodegradable film made from chitosan and cinnamon essential oil with low affinity toward water. Food Chem. 2010, 122, 161–166. [Google Scholar] [CrossRef]
- Bajić, M.; Ročnik, T.; Oberlintner, A.; Scognamiglio, F.; Novak, U.; Likozar, B. Natural plant extracts as active components in chitosan-based films: A comparative study. Food Packag. Shelf Life 2019, 21, 100365. [Google Scholar] [CrossRef]
- Wang, Z.; Yan, Y.; Zhang, Z.; Li, C.; Mei, L.; Hou, R.; Liu, X.; Jiang, H. Effect of Chitosan and Its Water-Soluble Derivatives on Antioxidant Activity. Polymers 2024, 16, 867. [Google Scholar] [CrossRef]
- Ro, J.; Kim, Y.; Kim, H.; Jang, S.B.; Lee, H.J.; Chakma, S.; Jeong, J.H.; Lee, J. Anti-oxidative activity of pectin and its stabilizing effect on retinyl palmitate. Korean J. Physiol. Pharmacol. 2013, 17, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F. Silymarin as a Natural Antioxidant: An Overview of the Current Evidence and Perspectives. Antioxidants 2015, 4, 204–247. [Google Scholar] [CrossRef]
- Pientaweeratch, S.; Panapisal, V.; Tansirikongkol, A. Antioxidant, anti-collagenase and anti-elastase activities of Phyllanthus emblica, Manilkara zapota and silymarin: An in vitro comparative study for anti-aging applications. Pharm. Biol. 2016, 54, 1865–1872. [Google Scholar] [CrossRef]
- Asghar, Z.; Masood, Z. Evaluation of antioxidant properties of silymarin and its potential to inhibit peroxyl radicals in vitro. Pak. J. Pharm. Sci. 2008, 21, 249–254. [Google Scholar] [PubMed]
- BS EN 13726-1:2002; Test Methods for Primary Wound Dressings—ASPECTS of Absorbency. Science and Education: St. Petersburg, FL, USA, 2013.
- ISO 527-3:2003; Plastics—Determination of Tensile Properties—Part 3: Test Conditions for Films and Sheets. ISO: Geneva, Switzerland, 2003.
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]





| Formulation Code | Thickness (mm) | Moisture Content (%) | Water Activity (aw) | Water Absorption (g/g) | pH (10% Water Extract) |
|---|---|---|---|---|---|
| Ch (0) | 0.094 ± 0.011 | 12.02 ± 0.77 | 0.321 ± 0.002 | 1.75 ± 0.11 | 5.12 ± 0.15 |
| Ch (SM) | 0.085 ± 0.010 | 10.67 ± 0.35 | 0.312 ± 0.004 | 2.13 ± 0.13 | 5.08 ± 0.13 |
| ChP1 (0) | 0.078 ± 0.007 | 13.71 ± 0.73 | 0.354 ± 0.005 | 1.66 ± 0.14 | 4.86 ± 0.20 |
| ChP1 (SM) | 0.118 ± 0.012 | 13.05 ± 0.11 | 0.341 ± 0.004 | 2.35 ± 0.12 | 4.85 ± 0.16 |
| ChP2 (0) | 0.088 ± 0.014 | 14.57 ± 0.81 | 0.362 ± 0.009 | 1.56 ± 0.11 | 4.76 ± 0.18 |
| ChP2 (SM) | 0.121 ± 0.008 | 13.83 ± 0.88 | 0.352 ± 0.011 | 2.51 ± 0.10 | 4.75 ± 0.14 |
| Formulation Code | Yield Strength (MPa) | Tensile Strength (MPa) | Young’s Modulus (MPa) | Elongation at Break (%) | Toughness (J/mm3) |
|---|---|---|---|---|---|
| Ch (0) | 12.69 ± 1.21 | 23.35 ± 1.74 | 60.14 ± 3.02 | 25.03 ± 2.68 | 2.97 ± 0.26 |
| Ch (SM) | 8.35 ± 0.46 | 22.01 ± 2.67 | 39.17 ± 1.78 | 90.15 ± 11.13 | 6.15 ± 1.03 |
| ChP1 (0) | 13.30 ± 1.08 | 19.98 ± 1.20 | 182.09 ± 17.23 | 32.81 ± 6.62 | 3.52 ± 0.60 |
| ChP1 (SM) | 1.28 ± 0.32 | 12.72 ± 2.11 | 17.05 ± 1.25 | 127.35 ± 12.47 | 6.98 ± 1.09 |
| ChP2 (0) | 4.48 ± 0.33 | 14.57 ± 0.81 | 197.70 ± 18.40 | 22.34 ± 2.14 | 0.88 ± 0.19 |
| ChP2 (SM) | 3.55 ± 0.39 | 11.51 ± 2.27 | 84.40 ± 14.10 | 28.12 ± 4.17 | 1.30 ± 0.24 |
| Formulation Code | L | a | b | ΔE |
|---|---|---|---|---|
| Ch (0) | 87.31 ± 1.88 | 1.01 ± 0.61 | 13.60 ± 4.02 | - |
| Ch (SM) | 61.73 ± 1.31 | 10.71 ± 0.53 | 37.84 ± 0.36 | 34.71 ± 7.36 |
| ChP1(0) | 81.29 ± 1.34 | 6.74 ± 1.64 | 31.79 ± 4.02 | - |
| ChP1(SM) | 54.72 ± 0.83 | 21.63 ± 0.65 | 44.32 ± 1.02 | 31.74 ± 1.56 |
| ChP2(0) | 66.64 ± 0.83 | 13.48 ± 0.91 | 43.55 ± 2.94 | - |
| ChP2(SM) | 53.26 ± 2.69 | 23.49 ± 1.34 | 63.30 ± 1.40 | 22.87± 4.85 |
| Control Films | Antioxidant Activity (mg TE/g) | Silymarin Loaded Films | Antioxidant Activity (mg TE/g) |
|---|---|---|---|
| Ch (0) | 0.081 ± 0.012 | Ch (SM) | 2.060 ± 0.091 |
| ChP1 (0) | 0.114 ± 0.018 | ChP1(SM) | 2.106 ± 0.190 |
| ChP2 (0) | 0.093 ± 0.005 | ChP2(SM) | 2.020 ± 0.048 |
| Formulation Code | Chitosan (g) | Pectin (g) | Silymarin (g) | Ethanol 95% (mL) | 1% Acetic Acid (mL) | Water (mL) | Glycerol (g) |
|---|---|---|---|---|---|---|---|
| Ch (0) | 1.40 | - | - | 5.0 | 85.0 | 10.0 | 1.0 |
| Ch (SM) | 1.40 | - | 0.10 | 5.0 | 85.0 | 10.0 | 1.0 |
| ChP1(0) | 1.40 | 0.14 | - | 5.0 | 85.0 | 10.0 | 1.0 |
| ChP1(SM) | 1.40 | 0.14 | 0.10 | 5.0 | 85.0 | 10.0 | 1.0 |
| ChP2(0) | 1.40 | 0.28 | - | 5.0 | 85.0 | 10.0 | 1.0 |
| ChP2(SM) | 1.40 | 0.28 | 0.10 | 5.0 | 85.0 | 10.0 | 1.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Dyankova, S.; Bozakova, N.; Boneva, V.; Solak, A.; Ivanov, V. Formulation and Characterization of Hydrogel Chitosan–Pectin Active Films Containing Silymarin. Molecules 2026, 31, 322. https://doi.org/10.3390/molecules31020322
Dyankova S, Bozakova N, Boneva V, Solak A, Ivanov V. Formulation and Characterization of Hydrogel Chitosan–Pectin Active Films Containing Silymarin. Molecules. 2026; 31(2):322. https://doi.org/10.3390/molecules31020322
Chicago/Turabian StyleDyankova, Svetla, Nadya Bozakova, Vanya Boneva, Ayten Solak, and Veselin Ivanov. 2026. "Formulation and Characterization of Hydrogel Chitosan–Pectin Active Films Containing Silymarin" Molecules 31, no. 2: 322. https://doi.org/10.3390/molecules31020322
APA StyleDyankova, S., Bozakova, N., Boneva, V., Solak, A., & Ivanov, V. (2026). Formulation and Characterization of Hydrogel Chitosan–Pectin Active Films Containing Silymarin. Molecules, 31(2), 322. https://doi.org/10.3390/molecules31020322






