Geraniol Exerts Cytotoxic Effects in Red Cells Through Ca2+ Elevation and Membrane Hyperpolarization: Attenuating Effects of COX/CK1α/Rac1 GTPase Inhibition
Abstract
1. Introduction
2. Results
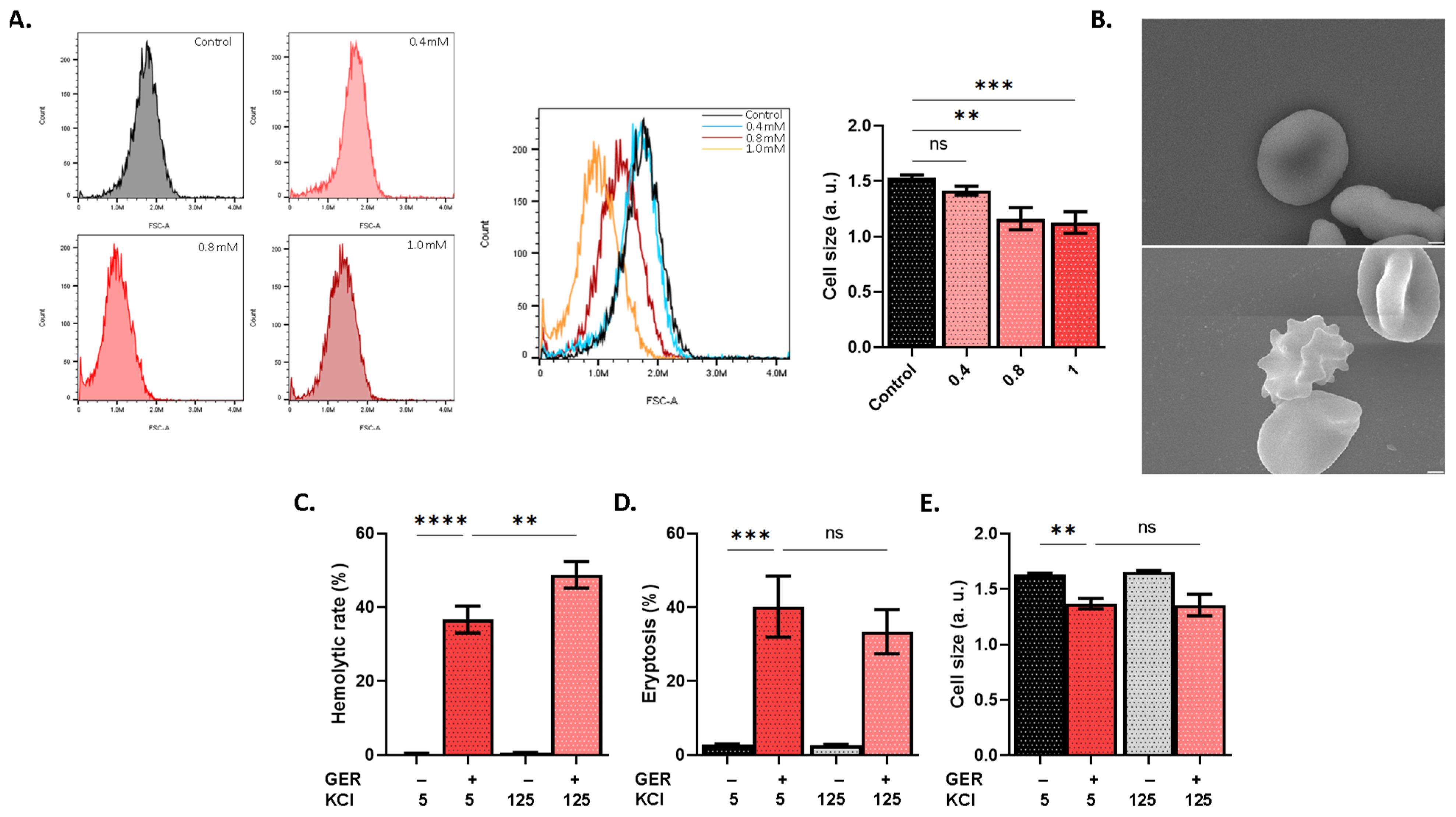
2.1. GER Is Cytotoxic to RBCs Through Hemolysis and Eryptosis
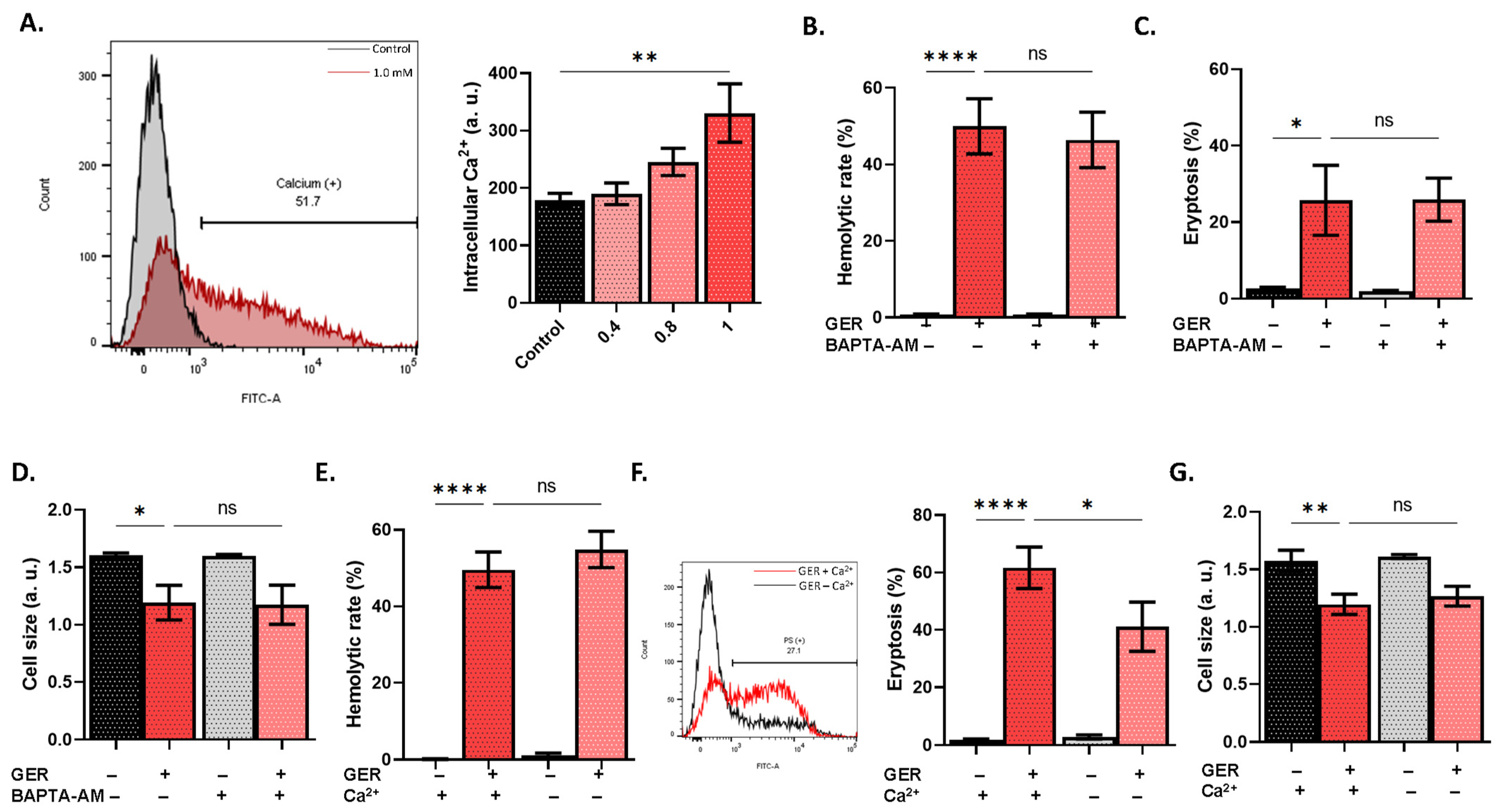
2.2. GER Stimulates Ca2+ Elevation and Membrane Hyperpolarization
2.3. GER Cytotoxicity Is Independent of Oxidative Stress
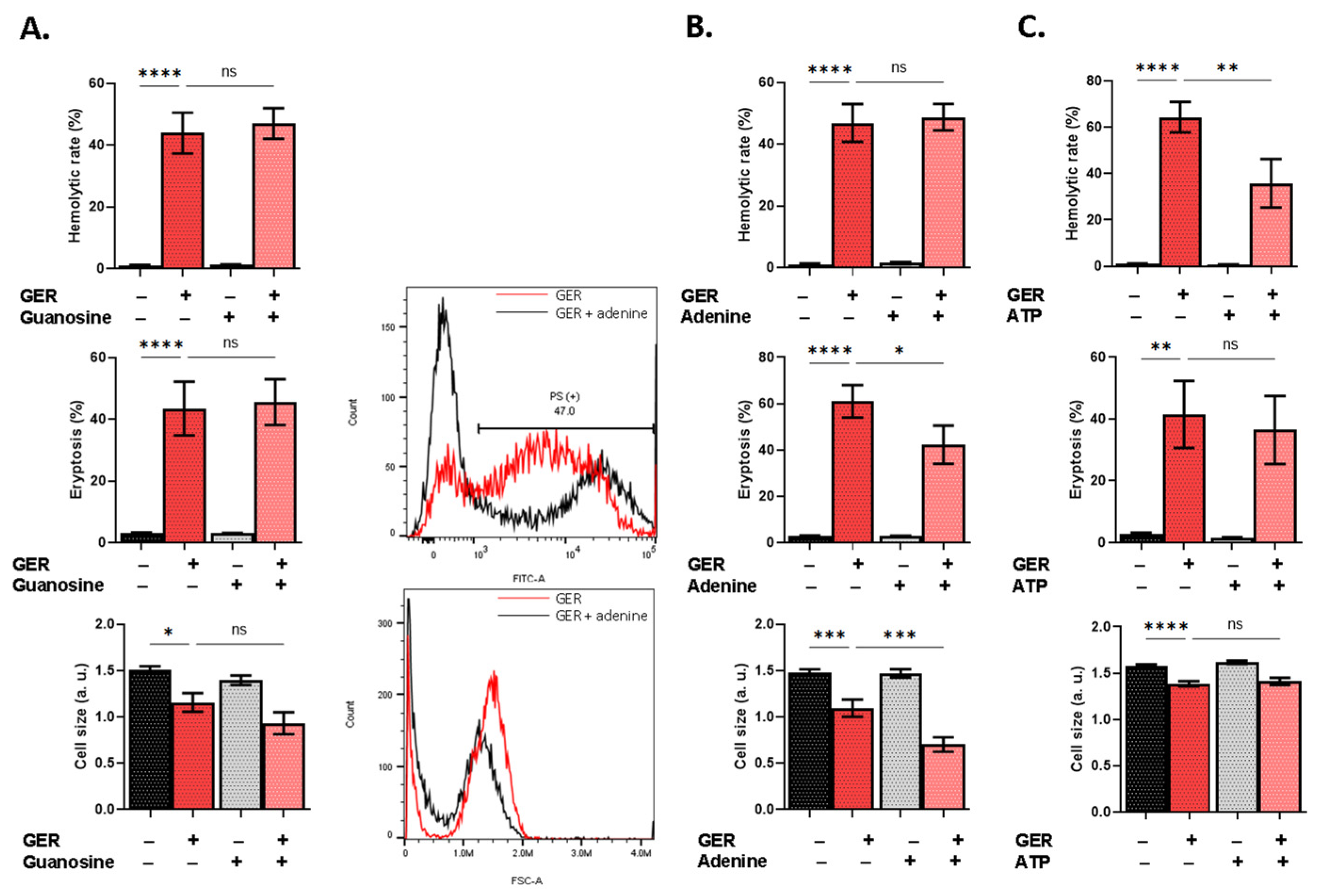
2.4. Inhibition of Rac1 GTPase and CK1α Attenuates GER Cytotoxicity
2.5. Effect of Metabolic Substrates on GER Cytotoxicity
3. Discussion
4. Materials and Methods
4.1. RBC Purification
4.2. Experimental Design
4.3. Hemolysis
4.4. Eryptosis
4.5. Erythrocyte Sedimentation Rate (ESR)
4.6. Electron Microscopy
4.7. Statistics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lei, Y.; Fu, P.; Jun, X.; Cheng, P. Pharmacological Properties of Geraniol—A Review. Planta Med. 2019, 85, 48–55. [Google Scholar] [CrossRef]
- Maczka, W.; Winska, K.; Grabarczyk, M. One Hundred Faces of Geraniol. Molecules 2020, 25, 3303. [Google Scholar] [CrossRef]
- Kim, S.H.; Park, E.J.; Lee, C.R.; Chun, J.N.; Cho, N.H.; Kim, I.G.; Lee, S.; Kim, T.W.; Park, H.H.; So, I.; et al. Geraniol induces cooperative interaction of apoptosis and autophagy to elicit cell death in PC-3 prostate cancer cells. Int. J. Oncol. 2012, 40, 1683–1690. [Google Scholar]
- Qi, F.; Yan, Q.; Zheng, Z.; Liu, J.; Chen, Y.; Zhang, G. Geraniol and geranyl acetate induce potent anticancer effects in colon cancer Colo-205 cells by inducing apoptosis, DNA damage and cell cycle arrest. J. BUON 2018, 23, 346–352. [Google Scholar]
- Shen, X.; Cui, X.; Cui, H.; Jin, Y.; Jin, W.; Sun, H. Geraniol and lupeol inhibit growth and promote apoptosis in human hepatocarcinoma cells through the MAPK signaling pathway. J. Cell. Biochem. 2019, 120, 5033–5041. [Google Scholar] [CrossRef] [PubMed]
- Abo El-Ella, D.M. Autophagy/Apoptosis Induced by Geraniol through HIF-1alpha/BNIP3/Beclin-1 Signaling Pathway in A549 CoCl2 Treated Cells. Adv. Pharm. Bull. 2022, 12, 155–162. [Google Scholar] [PubMed]
- Kuzu, B.; Cuce, G.; Ayan, I.C.; Gultekin, B.; Canbaz, H.T.; Dursun, H.G.; Sahin, Z.; Keskin, I.; Kalkan, S.S. Evaluation of Apoptosis Pathway of Geraniol on Ishikawa Cells. Nutr. Cancer 2021, 73, 2532–2537. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, Y.; Cui, J.; Hao, Z. Apoptosis-Mediated anticancer activity of geraniol inhibits NF-κB, MAPK, and JAK-STAT-3 signaling pathways in human thyroid cancer cells. Pharmacogn. Mag. 2022, 18, 1183–1189. [Google Scholar]
- Ju, R.; Wang, X.; Han, X. Geraniol inhibits cell growth and promotes caspase-dependent apoptosis in nasopharyngeal cancer C666-1 cells via inhibiting PI3K/Akt/mTOR signaling pathway. Arab. J. Chem. 2024, 17, 105373. [Google Scholar] [CrossRef]
- Yuan, T.; Jia, Q.; Zhu, B.; Chen, D.; Long, H. Synergistic immunotherapy targeting cancer-associated anemia: Prospects of a combination strategy. Cell Commun. Signal. 2023, 21, 117. [Google Scholar] [CrossRef]
- Bryer, E.; Henry, D. Chemotherapy-induced anemia: Etiology, pathophysiology, and implications for contemporary practice. Int. J. Clin. Transfus. Med. 2018, 6, 21–31. [Google Scholar] [CrossRef]
- Tkachenko, A.; Alfhili, M.A.; Alsughayyir, J.; Attanzio, A.; Al Mamun Bhuyan, A.; Bukowska, B.; Cilla, A.; Quintanar-Escorza, M.A.; Foller, M.; Havranek, O.; et al. Current understanding of eryptosis: Mechanisms, physiological functions, role in disease, pharmacological applications, and nomenclature recommendations. Cell Death Dis. 2025, 16, 467. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Yan, F.; Shen, J.; Wang, T.; Li, M.; Ni, H. Geraniol attenuates oxygen-glucose deprivation/reoxygenation-induced ROS-dependent apoptosis and permeability of human brain microvascular endothelial cells by activating the Nrf-2/HO-1 pathway. J. Bioenerg. Biomembr. 2024, 56, 193–204. [Google Scholar] [CrossRef]
- Zhang, X.H.; Morad, M. Regulation of SR and mitochondrial Ca2+ signaling by L-type Ca2+ channels and Na/Ca exchanger in hiPSC-CMs. Cell Calcium 2025, 125, 102985. [Google Scholar] [CrossRef]
- Guerrero-Hue, M.; Rubio-Navarro, A.; Sevillano, A.; Yuste, C.; Gutierrez, E.; Palomino-Antolin, A.; Roman, E.; Praga, M.; Egido, J.; Moreno, J.A. Adverse effects of the renal accumulation of haem proteins. Novel therapeutic approaches. Nefrologia 2018, 38, 13–26. [Google Scholar] [CrossRef]
- Attanzio, A.; Frazzitta, A.; Cilla, A.; Livrea, M.A.; Tesoriere, L.; Allegra, M. 7-Keto-Cholesterol and Cholestan-3beta, 5alpha, 6beta-Triol Induce Eryptosis through Distinct Pathways Leading to NADPH Oxidase and Nitric Oxide Synthase Activation. Cell. Physiol. Biochem. 2019, 53, 933–947. [Google Scholar]
- Sangha, G.S.; Smith, L.V.; Kheradmand, M.; Munir, K.M.; Rangachar, N.; Weber, C.M.; Safari, Z.; Rogers, S.C.; Doctor, A.; Clyne, A.M. Piezo1 activates nitric oxide synthase in red blood cells via protein kinase C with increased activity in diabetes. Mechanobiol. Med. 2025, 3, 100145. [Google Scholar] [CrossRef]
- Gao, M.; Wong, S.Y.; Lau, P.M.; Kong, S.K. Ferutinin induces in vitro eryptosis/erythroptosis in human erythrocytes through membrane permeabilization and calcium influx. Chem. Res. Toxicol. 2013, 26, 1218–1228. [Google Scholar] [CrossRef]
- Restivo, I.; Attanzio, A.; Giardina, I.C.; Di Gaudio, F.; Tesoriere, L.; Allegra, M. Cigarette Smoke Extract Induces p38 MAPK-Initiated, Fas-Mediated Eryptosis. Int. J. Mol. Sci. 2022, 23, 14730. [Google Scholar] [CrossRef] [PubMed]
- Borst, O.; Abed, M.; Alesutan, I.; Towhid, S.T.; Qadri, S.M.; Foller, M.; Gawaz, M.; Lang, F. Dynamic adhesion of eryptotic erythrocytes to endothelial cells via CXCL16/SR-PSOX. Am. J. Physiol. Cell Physiol. 2012, 302, C644–C651. [Google Scholar] [CrossRef]
- Bogdanova, A.; Makhro, A.; Wang, J.; Lipp, P.; Kaestner, L. Calcium in red blood cells-a perilous balance. Int. J. Mol. Sci. 2013, 14, 9848–9872. [Google Scholar] [CrossRef]
- Ataga, K.I.; Staffa, S.J.; Brugnara, C.; Stocker, J.W. Haemoglobin response to senicapoc in patients with sickle cell disease: A re-analysis of the Phase III trial. Br. J. Haematol. 2021, 192, e129–e132. [Google Scholar] [CrossRef]
- Alasmari, B.G.; Wafa, S.; Alsari Alqahtani, A.; Sameh, B.; Elzubair, L. Dehydrated Hereditary Stomatocytosis (DHS): A Rare Inherited Hemolytic Disorder with Unusual Hypochromic Microcytic Anemia. Cureus 2025, 17, e81335. [Google Scholar] [CrossRef]
- Lang, P.A.; Kempe, D.S.; Myssina, S.; Tanneur, V.; Birka, C.; Laufer, S.; Lang, F.; Wieder, T.; Huber, S.M. PGE(2) in the regulation of programmed erythrocyte death. Cell Death Differ. 2005, 12, 415–428. [Google Scholar] [CrossRef]
- George, A.; Pushkaran, S.; Li, L.; An, X.; Zheng, Y.; Mohandas, N.; Joiner, C.H.; Kalfa, T.A. Altered phosphorylation of cytoskeleton proteins in sickle red blood cells: The role of protein kinase C, Rac GTPases, and reactive oxygen species. Blood Cells Mol. Dis. 2010, 45, 41–45. [Google Scholar] [CrossRef]
- Alfhili, M.A.; Alghareeb, S.A.; Alotaibi, G.A.; Alsughayyir, J. Galangin Triggers Eryptosis and Hemolysis Through Ca2+ Nucleation and Metabolic Collapse Mediated by PKC/CK1alpha/COX/p38/Rac1 Signaling Axis. Int. J. Mol. Sci. 2024, 25, 12267. [Google Scholar] [CrossRef] [PubMed]
- Paone, S.; D’Alessandro, S.; Parapini, S.; Celani, F.; Tirelli, V.; Pourshaban, M.; Olivieri, A. Characterization of the erythrocyte GTPase Rac1 in relation to Plasmodium falciparum invasion. Sci. Rep. 2020, 10, 22054. [Google Scholar] [CrossRef] [PubMed]
- Zelenak, C.; Eberhard, M.; Jilani, K.; Qadri, S.M.; Macek, B.; Lang, F. Protein kinase CK1alpha regulates erythrocyte survival. Cell. Physiol. Biochem. 2012, 29, 171–180. [Google Scholar] [CrossRef] [PubMed]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Alfhili, M.A.; Alruwaili, S.H.; Alsughayyir, J. Geraniol Exerts Cytotoxic Effects in Red Cells Through Ca2+ Elevation and Membrane Hyperpolarization: Attenuating Effects of COX/CK1α/Rac1 GTPase Inhibition. Molecules 2026, 31, 1621. https://doi.org/10.3390/molecules31101621
Alfhili MA, Alruwaili SH, Alsughayyir J. Geraniol Exerts Cytotoxic Effects in Red Cells Through Ca2+ Elevation and Membrane Hyperpolarization: Attenuating Effects of COX/CK1α/Rac1 GTPase Inhibition. Molecules. 2026; 31(10):1621. https://doi.org/10.3390/molecules31101621
Chicago/Turabian StyleAlfhili, Mohammad A., Shaymah H. Alruwaili, and Jawaher Alsughayyir. 2026. "Geraniol Exerts Cytotoxic Effects in Red Cells Through Ca2+ Elevation and Membrane Hyperpolarization: Attenuating Effects of COX/CK1α/Rac1 GTPase Inhibition" Molecules 31, no. 10: 1621. https://doi.org/10.3390/molecules31101621
APA StyleAlfhili, M. A., Alruwaili, S. H., & Alsughayyir, J. (2026). Geraniol Exerts Cytotoxic Effects in Red Cells Through Ca2+ Elevation and Membrane Hyperpolarization: Attenuating Effects of COX/CK1α/Rac1 GTPase Inhibition. Molecules, 31(10), 1621. https://doi.org/10.3390/molecules31101621






